Anti-SARS-Cov-2 IgA Response in Tears of COVID-19 Patients
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Samples
2.2. IgA Detection
2.3. Statistics
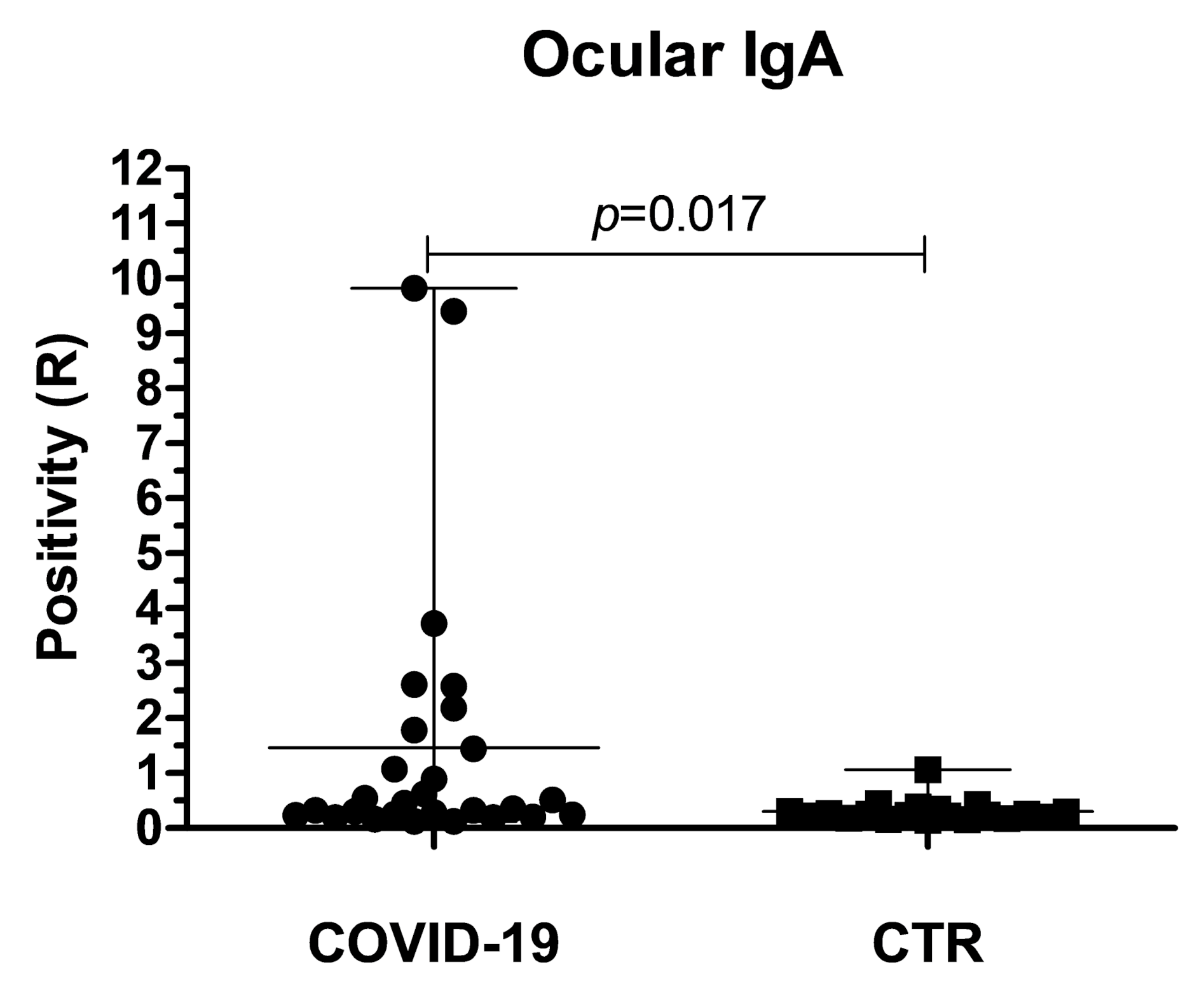
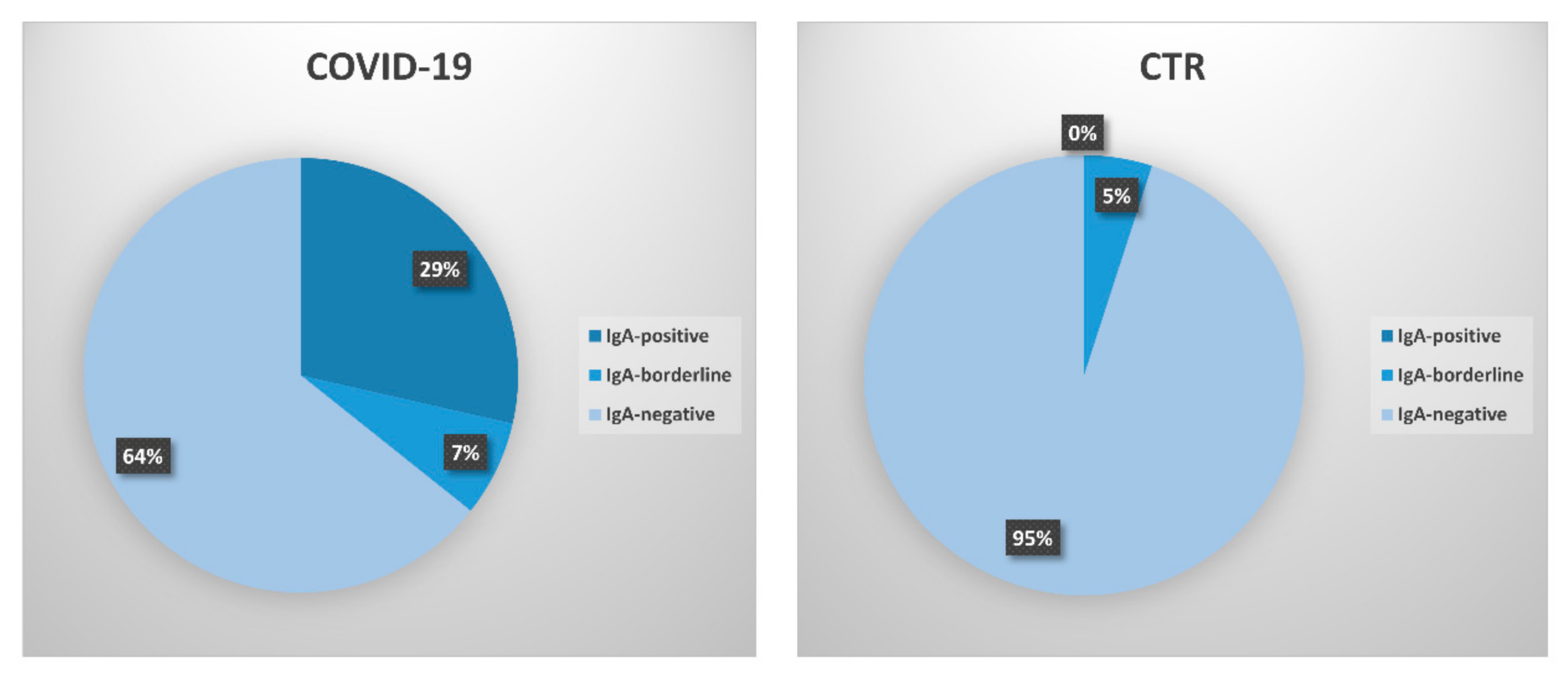
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Contini, C.; Caselli, E.; Martini, F.; Maritati, M.; Torreggiani, E.; Seraceni, S.; Vesce, F.; Perri, P.; Rizzo, L.; Tognon, M. Covid-19 is a multifaceted challenging pandemic which needs urgent public health interventions. Microorganisms 2020, 8, 1228. [Google Scholar] [CrossRef]
- WHO. Diagnostic Detection of 2019-nCoV by Real-time rt-pcr. Available online: https://www.who.int/docs/default-source/coronaviruse/protocol-v2-1.pdf (accessed on 3 April 2020).
- WHO. Cleaning and Disinfection of Environmental Surfaces in the Context of COVID-19. Available online: https://www.who.int/publications/i/item/cleaning-and-disinfection-of-environmental-surfaces-inthe-context-of-COVID-19 (accessed on 10 July 2020).
- Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-Cov-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, A.A.T.; Fatima, K.; Mohammad, T.; Fatima, U.; Singh, I.K.; Singh, A.; Atif, S.M.; Hariprasad, G.; Hasan, G.M.; Hassan, M.I. Insights into sars--CoV-2 genome, structure, evolution, pathogenesis and therapies: Structural genomics approach. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165878. [Google Scholar] [CrossRef] [PubMed]
- Benvenuto, D.; Giovanetti, M.; Ciccozzi, A.; Spoto, S.; Angeletti, S.; Ciccozzi, M. The 2019-new coronavirus epidemic: Evidence for virus evolution. J. Med. Virol. 2020, 92, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Giovanetti, M.; Benvenuto, D.; Angeletti, S.; Ciccozzi, M. The first two cases of 2019-nCoV in italy: Where they come from? J. Med. Virol. 2020, 92, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Wang, W.; Zhao, X.; Zai, J.; Li, X. Cross-species transmission of the newly identified coronavirus 2019-nCoV. J. Med. Virol. 2020, 92, 433–440. [Google Scholar] [CrossRef]
- Wan, Y.; Shang, J.; Graham, R.; Baric, R.S.; Li, F. Receptor recognition by the novel coronavirus from wuhan: An analysis based on decade-long structural studies of sars coronavirus. J. Virol. 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Riordan, J.F. Angiotensin-i-converting enzyme and its relatives. Genome Biol. 2003, 4, 225. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘cytokine storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with sars--CoV-2 pneumonia in wuhan, china: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Bitko, V.; Musiyenko, A.; Barik, S. Viral infection of the lungs through the eye. J. Virol. 2007, 81, 783–790. [Google Scholar] [CrossRef]
- Belser, J.A.; Rota, P.A.; Tumpey, T.M. Ocular tropism of respiratory viruses. Microbiol. Mol. Biol. Rev. 2013, 77, 144–156. [Google Scholar] [CrossRef]
- Almeida, F.P.; Paula, J.S.; Martins, M.C.; Sena, D.F.; Cervi, M.C.; Rodrigues, M.L. Ocular manifestations in pediatric patients with hiv infection in the post-haart era in southern brazil. Eye 2007, 21, 1017–1018. [Google Scholar] [CrossRef] [PubMed]
- Dockery, D.M.; Rowe, S.G.; Murphy, M.A.; Krzystolik, M.G. The ocular manifestations and transmission of COVID-19: Recommendations for prevention. J. Emerg. Med. 2020, 59, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.; Wolf, J.; Auw-Haedrich, C.; Schlecht, A.; Boneva, S.; Lapp, T.; Horres, R.; Agostini, H.; Martin, G.; Reinhard, T.; et al. Expression of the COVID-19 receptor ace2 in the human conjunctiva. J. Med. Virol 2020. [Google Scholar] [CrossRef]
- Senanayake, P.; Drazba, J.; Shadrach, K.; Milsted, A.; Rungger-Brandle, E.; Nishiyama, K.; Miura, S.; Karnik, S.; Sears, J.E.; Hollyfield, J.G. Angiotensin ii and its receptor subtypes in the human retina. Investig. Ophthalmol. Vis. Sci. 2007, 48, 3301–3311. [Google Scholar] [CrossRef]
- Ma, D.; Chen, C.B.; Jhanji, V.; Xu, C.; Yuan, X.L.; Liang, J.J.; Huang, Y.; Cen, L.P.; Ng, T.K. Expression of sars-CoV-2 receptor ace2 and tmprss2 in human primary conjunctival and pterygium cell lines and in mouse cornea. Eye 2020, 34, 1212–1219. [Google Scholar] [CrossRef]
- Chen, X.; Yu, H.; Mei, T.; Chen, B.; Chen, L.; Li, S.; Zhang, X.; Sun, X. Sars-CoV-2 on the ocular surface: Is it truly a novel transmission route? Br. J. Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Jorstad, O.K.; Moe, M.C.; Eriksen, K.; Petrovski, G.; Bragadottir, R. Coronavirus disease 2019 (COVID-19) outbreak at the department of ophthalmology, oslo university hospital, norway. Acta Ophthalmol. 2020, 98, e388–e389. [Google Scholar] [CrossRef]
- Lu, C.W.; Liu, X.F.; Jia, Z.F. 2019-nCoV transmission through the ocular surface must not be ignored. Lancet 2020, 395, e39. [Google Scholar] [CrossRef]
- Li, J.O.; Lam, D.S.C.; Chen, Y.; Ting, D.S.W. Novel coronavirus disease 2019 (COVID-19): The importance of recognising possible early ocular manifestation and using protective eyewear. Br. J. Ophthalmol. 2020, 104, 297–298. [Google Scholar] [CrossRef]
- Xia, J.; Tong, J.; Liu, M.; Shen, Y.; Guo, D. Evaluation of coronavirus in tears and conjunctival secretions of patients with sars-CoV-2 infection. J. Med. Virol. 2020, 92, 589–594. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report of the Who-China Joint Mission on Coronavirus Disease 2019 (COVID-19); World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Colavita, F.; Lapa, D.; Carletti, F.; Lalle, E.; Bordi, L.; Marsella, P.; Nicastri, E.; Bevilacqua, N.; Giancola, M.L.; Corpolongo, A.; et al. Sars-CoV-2 isolation from ocular secretions of a patient with COVID-19 in Italy with prolonged viral rna detection. Ann. Intern. Med. 2020. [Google Scholar] [CrossRef]
- Taylor, A.W. Ocular immune privilege and transplantation. Front. Immunol. 2016, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Secchiero, P.; Lamberti, G.; Corallini, F.; Melloni, E.; Guarnotta, C.; Sebastiani, A.; Zauli, G. Conjunctival sac fluid contains elevated levels of soluble trail: Implications for the anti-tumoral surveillance of the anterior surface of the eye. J. Cell Physiol. 2009, 218, 199–204. [Google Scholar] [CrossRef]
- Meyer, B.; Torriani, G.; Yerly, S.; Mazza, L.; Calame, A.; Arm-Vernez, I.; Zimmer, G.; Agoritsas, T.; Stirnemann, J.; Spechbach, H.; et al. Validation of a commercially available SARS-CoV-2 serological immunoassay. Clin. Microbiol. Infect. 2020, 26, 1386–1394. [Google Scholar] [CrossRef]
- Xie, H.T.; Jiang, S.Y.; Xu, K.K.; Liu, X.; Xu, B.; Wang, L.; Zhang, M.C. SARS-CoV-2 in the ocular surface of COVID-19 patients. Eye Vis. 2020, 7, 23. [Google Scholar] [CrossRef]
- Guemes-Villahoz, N.; Burgos-Blasco, B.; Arribi-Vilela, A.; Arriola-Villalobos, P.; Rico-Luna, C.M.; Cuina-Sardina, R.; Delgado-Iribarren, A.; Garcia-Feijoo, J. Detecting sars-CoV-2 rna in conjunctival secretions: Is it a valuable diagnostic method of COVID-19? J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Atum, M.; Boz, A.A.E.; Cakir, B.; Karabay, O.; Koroglu, M.; Ogutlu, A.; Alagoz, G. Evaluation of conjunctival swab pcr results in patients with sars--CoV-2 infection. Ocul. Immunol. Inflamm. 2020, 28, 745–748. [Google Scholar] [CrossRef] [PubMed]
- Emparan, J.P.O.; Sardi-Correa, C.; Lopez-Ulloa, J.A.; Viteri-Soria, J.; Penniecook, J.A.; Jimenez-Roman, J.; Lansingh, V.C. COVID-19 and the eye: How much do we really know? A best evidence review. Arq. Bras. Oftalmol. 2020, 83, 250–261. [Google Scholar]
- Hu, Y.; Chen, T.; Liu, M.; Zhang, L.; Wang, F.; Zhao, S.; Liu, H.; Xia, H.; Wang, Y.; Li, L. Positive detection of sars--CoV-2 combined hsv1 and hhv6b virus nucleic acid in tear and conjunctival secretions of a non-conjunctivitis COVID-19 patient with obstruction of common lacrimal duct. Acta Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Seah, I.; Agrawal, R. Can the coronavirus disease 2019 (COVID-19) affect the eyes? A review of coronaviruses and ocular implications in humans and animals. Ocul. Immunol. Inflamm. 2020, 28, 391–395. [Google Scholar] [CrossRef]
- Al-Sharif, E.; Strianese, D.; AlMadhi, N.H.; D’Aponte, A.; dell’Omo, R.; Di Benedetto, R.; Costagliola, C. Ocular tropism of coronavirus (covs): A comparison of the interaction between the animal-to-human transmitted coronaviruses (SARS-CoV-1, SARS-CoV-2, MERS-CoV, CoV-229E, NL63, OC43, HKU1) and the eye. Int. Ophthalmol. 2020, 1–14. [Google Scholar] [CrossRef]
- Peng, M.; Dai, J.; Sugali, C.K.; Rayana, N.P.; Mao, W. The role of the ocular tissue in SARS-CoV-2 transmission. Clin. Ophthalmol. 2020, 14, 3017–3024. [Google Scholar] [CrossRef]
- Marinho, P.M.; Marcos, A.A.A.; Romano, A.C.; Nascimento, H.; Belfort, R., Jr. Retinal findings in patients with COVID-19. Lancet 2020, 395, 1610. [Google Scholar] [CrossRef]
- Deng, W.; Bao, L.; Gao, H.; Xiang, Z.; Qu, Y.; Song, Z.; Gong, S.; Liu, J.; Liu, J.; Yu, P.; et al. Ocular conjunctival inoculation of sars-cov-2 can cause mild COVID-19 in rhesus macaques. Nat. Commun. 2020, 11, 4400. [Google Scholar] [CrossRef]
- Wu, P.; Duan, F.; Luo, C.; Liu, Q.; Qu, X.; Liang, L.; Wu, K. Characteristics of ocular findings of patients with coronavirus disease 2019 (COVID-19) in hubei province, china. JAMA Ophthalmol. 2020, 138, 575–578. [Google Scholar] [CrossRef]
- Saitoh-Inagawa, W.; Hiroi, T.; Yanagita, M.; Iijima, H.; Uchio, E.; Ohno, S.; Aoki, K.; Kiyono, H. Unique characteristics of lacrimal glands as a part of mucosal immune network: High frequency of iga-committed b-1 cells and nk1.1+ alphabeta t cells. Invest. Ophthalmol. Vis. Sci. 2000, 41, 138–144. [Google Scholar]
- Lang, J.; Yang, N.; Deng, J.; Liu, K.; Yang, P.; Zhang, G.; Jiang, C. Inhibition of sars pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans. PLoS ONE 2011, 6, e23710. [Google Scholar] [CrossRef]
| No | Age | Gender | COVID-19 Symptoms | Ocular Symptoms | Blood Lymphocytes/µL | PCR (mL/L) | Time * | IgA § |
|---|---|---|---|---|---|---|---|---|
| 1 | 66 | F | Fever, epigastralgia | None | 370 | 0.19 | 6 | 1.076 ± 0.02 |
| 2 | 89 | F | Fever, dyspnea | None | 1010 | 1.04 | 47 | 2.618 ± 0.05 |
| 3 | 73 | F | Pneumonia | None | 470 | 3.17 | 47 | 9.822 ± 0.13 |
| 4 | 74 | F | General malaise, hyporexia, nausea | None | 31,430 | 7.45 | 43 | |
| 5 | 44 | F | Pharyngodynia, anosmia | Mild redness | 2290 | 0.09 | 54 | |
| 6 | 77 | F | Bowel obstruction | None | 1690 | 11.25 | 11 | |
| 7 | 87 | M | Pneumonia | None | 1080 | 2.98 | 78 | |
| 8 | 80 | M | Dyspnea, edema | None | 710 | 4.19 | 48 | 1.784 ± 0.03 |
| 9 | 69 | F | Cardiac failure | None | 1280 | 3.87 | 46 | |
| 10 | 59 | F | Fever, dyspnea, cough | None | 990 | 3.58 | 5 | 0.896 ± 0.01 |
| 11 | 72 | F | Gastric hemorrhage | None | 1680 | 0.20 | 10 | |
| 12 | 58 | M | Dyspnea, cough, chest tightness | None | 1620 | 0.83 | 8 | |
| 13 | 63 | M | Fever, pneumonia | None | 750 | 1.31 | 4 | |
| 14 | 72 | M | Heart failure, pleural effusion | None | 850 | 0.60 | 6 | |
| 15 | 59 | F | Fever, asthenia | None | 1520 | 3.76 | 6 | 9.402 ± 0.26 |
| 16 | 89 | M | Respiratory failure, pneumonia | None | 1870 | 2.44 | 6 | |
| 17 | 84 | M | Acute respiratory failure, pneumonia | None | 1540 | 5.80 | 3 | |
| 18 | 72 | M | Fever, cough | None | 1130 | 14.26 | 7 | |
| 19 | 62 | M | Dyspnea, pneumonia | None | 690 | 1.40 | 4 | |
| 20 | 75 | M | Fever | None | 900 | 3.27 | 3 | 2.588 ± 0.51 |
| 21 | 21 | F | Pyelonephritis | None | 1080 | 20.21 | 7 | |
| 22 | 19 | F | Fever | None | 1670 | 8.04 | 2 | 1.444 ± 0.11 |
| 23 | 72 | M | Fever, dyspnea, dysgeusia | None | 690 | 8.31 | 1 | |
| 24 | 47 | M | Recurrent COVID19 recovery | None | 1780 | 0.30 | 2 | |
| 25 | 68 | M | ARDS (acute respiratory distress syndrome) | None | 630 | 28.35 | 15 | 2.186 ± 0.09 |
| 26 | 81 | F | Fever, dyspnea | None | 310 | 0.75 | 7 | |
| 27 | 87 | F | Fever, bronchopneumonia | None | 1710 | 16.68 | 7 | |
| 28 | 53 | M | Pneumonia, dyspnea | None | 1220 | 0.87 | 5 | 3.728 ± 0.23 |
| Parameter | Correlation (r) * | 95% C.I. | p Value |
|---|---|---|---|
| Age | −0.097 | −0.46–0.29 | 0.31 (n.s.) |
| Gender | 0.017 | −0.37–0.39 | 0.46 (n.s.) |
| Lymphocyte number | −0.199 | −054–0.19 | 0.15 (n.s.) |
| Ocular symptoms | −0.107 | −0.47–0.28 | 0.29 (n.s.) |
| Disease severity | −0.133 | −0.48–0.27 | 0.26 (n.s.) |
| PCR | −0.111 | −0.47–0.28 | 0.29 (n.s.) |
| Time | 0.083 | −0.31–0.45 | 0.34 (n.s.) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caselli, E.; Soffritti, I.; Lamberti, G.; D’Accolti, M.; Franco, F.; Demaria, D.; Contoli, M.; Passaro, A.; Contini, C.; Perri, P. Anti-SARS-Cov-2 IgA Response in Tears of COVID-19 Patients. Biology 2020, 9, 374. https://doi.org/10.3390/biology9110374
Caselli E, Soffritti I, Lamberti G, D’Accolti M, Franco F, Demaria D, Contoli M, Passaro A, Contini C, Perri P. Anti-SARS-Cov-2 IgA Response in Tears of COVID-19 Patients. Biology. 2020; 9(11):374. https://doi.org/10.3390/biology9110374
Chicago/Turabian StyleCaselli, Elisabetta, Irene Soffritti, Giuseppe Lamberti, Maria D’Accolti, Filippo Franco, Davide Demaria, Marco Contoli, Angela Passaro, Carlo Contini, and Paolo Perri. 2020. "Anti-SARS-Cov-2 IgA Response in Tears of COVID-19 Patients" Biology 9, no. 11: 374. https://doi.org/10.3390/biology9110374
APA StyleCaselli, E., Soffritti, I., Lamberti, G., D’Accolti, M., Franco, F., Demaria, D., Contoli, M., Passaro, A., Contini, C., & Perri, P. (2020). Anti-SARS-Cov-2 IgA Response in Tears of COVID-19 Patients. Biology, 9(11), 374. https://doi.org/10.3390/biology9110374









