Antarctic Epilithic Lichens as Niches for Black Meristematic Fungi
Abstract
:1. Introduction
2. Experimental Section
| Lichen species | Location | Coordinates | Sampling date | Fungal strains (CCFEE) |
|---|---|---|---|---|
| Acarospora sp. | Ford Peak, NVL | 75°41'26.3''S 160°26'25.3''E | 28/01/2004 | - |
| Acarospora flavocordia Castello & Nimis | Kay Island, NVL | 75°04'13.7''S 165°19'02.0''E | 30/01/2004 | 5324 |
| Buellia frigida Darb. | Inexpressible Island, NVL | 75°52'23.2''S 163°42'16.5''E | 17/01/2004 | - |
| Lecanora fuscobrunnea Dodge & Baker | Edmonson Point, NVL | 74°19'43.7''S 165°08'00.7''E | 29/01/2004 | 5320 * |
| Lecanora fuscobrunnea Dodge & Baker | Convoy Range, Terra SVL | 76°54'33.0''S 160°50'00.0''E | 25/01/2004 | 5303 |
| Lecanora sp. | Inexpressible Island, NVL | 75°52'23.2''S 163°42'16.5''E | 17/01/2004 | 5319 *, 5323 |
| Lecidea sp. | Starr Nunatak, NVL | 75°53'55.7''S 162°35'31.3''E | 15/02/2004 | 5318 |
| Lecidea sp. | Starr Nunatak, Terra Vittoria del Nord | 75°53'55.7''S 162°35'31.3''E | 15/02/2004 | 5326 |
| Lecidea cancriformis Dodge & Baker | Widowmaker Pass, NVL | 74°55'23.5''S 162°24'17.0''E | 12/02/2004 | 5321 ** |
| Rhizocarpon sp. | Vegetation Island, NVL | 74°47'05.2''S 163°38'40.3''E | 16/01/2004 | 5312 |
| Umbilicaria aprina Nyl. | Kay Island, NVL | 75°04'13.7''S 165°19'02.0''E | 30/01/2004 | - |
| Umbilicaria decussata (Vill.) Zahlbr. | Kay Island, NVL | 75°04'13.7''S 165°19'02.0''E | 02/02/2004 | - |
| Umbilicaria decussata (Vill.) Zahlbr. | Vegetation Island, NVL | 74°47'05.2''S 163°38'40.3''E | 16/01/2004 | 5317 |
| Usnea antarctica Du Rietz | Kay Island, NVL | 75°04'13.7''S 165°19'02.0''E | 30/01/2004 | - |
| Usnea antarctica Du Rietz | Vegetation Island, NVL | 74°47'05.2''S 163°38'40.3''E | 16/01/2004 | 5313 * |
| Xanthoria elegans (Link) th. Fr. | Kay Island, NVL | 75°04'13.7''S 165°19'02.0''E | 30/01/2004 | 5314, 5322 |
| Species | Strains no. | Source | Location | SSU |
|---|---|---|---|---|
| Acidomyces acidophilum | C2 | acid mine drainage | CA, USA | AY374300 |
| Acidomyces acidophilum | A3-7 | acid mine drainage | CA, USA | AY374299 |
| Acidomyces acidophilum | B1 | acid mine drainage | CA, USA | AY374298 |
| Aureobasidium pullulans | 28v1 | - | - | AY137505 |
| Aureobasidium pullulans | 30v4 | - | - | AY137507 |
| Botryosphaeria ribis | CBS 121.26 | Ribes rubrum | - | U42477 |
| Botryosphaeria ribis | CBS 115475 | Ribes | - | DQ678000 |
| Capnobotryella renispora | CBS 214.90 | Abies | Japan | EF137360 |
| Capnobotryella renispora | CBS 215.90 | Sphagnum | Japan | AY220613 |
| Capnobotryella renispora | CBS 572.89 | Roof tile | Sweeden | AY220614 |
| Capnobotryella renispora | UAMH 9870 | Sphagnum | - | AY220611 |
| Capronia coronata | CBS 617.96 | Decorticated wood | New Zealand | AJ232939 |
| Capronia semiimmersa | CBS 840.69 | Decaying timber | Finland | AY554291 |
| Catenulostroma abietis | CBS 459.93 | Abies | Germany | DQ678040 |
| Cladophialophora carrionii | CBS 260.83 | Skin lesion | - | AY554285 |
| Cladophialophora sp. | CBS 985.96 | Brain | USA | AJ232953 |
| Coccodinium bartschii | UME30232 | - | - | U77668 |
| Coniosporium sp. | MA 4597 | Marble | Turkey | AJ972863 |
| Cyphellophora laciniata | MUCL 9569 | - | - | AY342010 |
| Cryomyces antarcticus | CCFEE 514 | Rock | Antarctica | GU250319 |
| Cryomyces antarcticus | CCFEE 515 | Rock | Antarctica | GU250320 |
| Cryomyces antarcticus | CBS 116301T; CCFEE 534 | Sandstone | Antarctica | DQ028269 |
| Cryomyces minteri | CBS 116302; CCFEE 5187 | Sandstone | Antarctica | DQ028270 |
| Discosphaerina fagi | CBS 171.93 | Populus leaf | UK | AY016342 |
| Elasticomyces elasticus | CBS 122538; CCFEE 5313 | Lichen | Antarctica | FJ415474 |
| Elasticomyces elasticus | CBS 122539; CCFEE 5319 | Lichen | Antarctica | GU250332 |
| Elasticomyces elasticus | CBS 122540; CCFEE 5320 | Lichen | Antarctica | GU250333 |
| Elsinoe centrolobii | CBS 222.50 | Centrolobium robustum | Brazil | DQ678041 |
| Exophiala salmonis | CBS 157.67 | Salmo clarkii | Canada | JN856020 |
| Exophiala salmonis | AFTOL-ID 671 | - | - | EF413608 |
| Friedmanniomyces endolithicus | CCFEE 670 | Rock | Antarctica | GU250322 |
| Friedmanniomyces endolithicus | CCFEE 5208 | Rock | Antarctica | Unpublished |
| Friedmanniomyces endolithicus | CCFEE 5321 | Lichen | Antarctic | Unpublished |
| Fonsecaea pedrosoi | CBS 272.37 | - | - | AY554290 |
| Guignardia mangiferae | IFO 33119 | Rhododendron pulchrum | - | AB041247 |
| Guignardia mangiferae | CBS 226.77 | Paphiopedilum callosum | - | AB041248 |
| Guignardia mangiferae | CBS 398.80 | Orchid | - | AB041249 |
| Hobsonia santessonii | - | - | - | AF289658 |
| Hortaea werneckii | dH10921 | Marble | - | Y18700 |
| Hortaea werneckii | CBS 107.67 | human Tinea nigra | - | Y18693 |
| Knufia chersonesos | CBS 600.93; dH16058 | Marble | Greece | Y18702 |
| Knufia chersonesos | CBS 726.95 | Marble | Italy | Unpublished |
| Knufia perforans | CBS 885.95 | Marble | Delos, Greece | Y11714 |
| Knufia perforans | CBS 665.80 | Marble | Delos, Greece | Y11712 |
| Mycocalicium victoriae | CBS 109863 | Soil | Italy | Unpublished |
| Myriangium duriaei | CBS 260.36 | Chrysomphalus | Argentina | NG_013129 |
| Pseudotaeniolina globosa | CBS 109889 | Rock | Italy | GU214576 |
| Saxomyces alpinus | CCFEE 5466 | Rock | Alps, Italy | GU250350 |
| Saxomyces alpinus | CCFEE 5469 | Rock | Alps, Italy | KC315860 |
| Saxomyces alpinus | CCFEE 5470 | Rock | Alps, Italy | KC315861 |
| Saxomyces penninicus | CCFEE 5495 | Rock | Alps, Italy | KC315864 |
| Recurvomyces mirabilis | CBS 119434; CCFEE 5264 | Rock | Antarctica | GU250329 |
| Rhinocladiella atrovirens | CBS 688.76 | Pinus | Australia | AJ232937 |
| Rock black fungus | CCFEE 451 | Rock | Antarctic | GU250314 |
| Rock black fungus | CCFEE 457 | Rock | Antarctic | GU250317 |
| Rock black fungus | CCFEE 507 | Rock | Antarctic | Unpublished |
| Rock black fungus | CCFEE 5176 | Rock | Antarctic | GU250325 |
| Rock black fungus | CCFEE 5177 | Rock | Antarctic | Unpublished |
| Rock black fungus | CCFEE 5205 | Rock | Antarctic | GU250327 |
| Rock black fungus | CCFEE 5207 | Rock | Antarctic | Unpublished |
| Rock black fungus | CCFEE 5267 | Rock | Antarctic | Unpublished |
| Rock black fungus | CCFEE 5284 | Rock | Antarctic | GU250330 |
| Rock black fungus | CCFEE 5303 | Rock | Antarctic | GU250331 |
| Rock black fungus | CCFEE 5329 | Rock | Antarctic | Unpublished |
| Teratosphaeria microspora | CBS 101951; STE-U 1960 | Leaf | South Africa | EU167572 |
| Teratosphaeria molleriana | CPC 1214 | Eucalyptus globulus | Portugal | GU214606 |
| Teratosphaeria molleriana | CPC 4577 | Eucalyptus | Australia | GU214582 |
| Teratosphaeria molleriana | CPC 10397 | Eucalyptus globulus | Spain | GU214607 |
| Teratosphaeria nubilosa | CPC 933 | Eucalyptus nitens | South Africa | GU214608 |
| Teratosphaeria nubilosa | CPC 937 | Eucalyptus globulus | Australia | GU214609 |
| Unknown black fungus | CCFEE 5304 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5312 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5314 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5317 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5318 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5322 | Lichen | Antarctic | GU250334 |
| Unknown black fungus | CCFEE 5323 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5324 | Lichen | Antarctic | Unpublished |
| Unknown black fungus | CCFEE 5326 | Lichen | Antarctic | Unpublished |
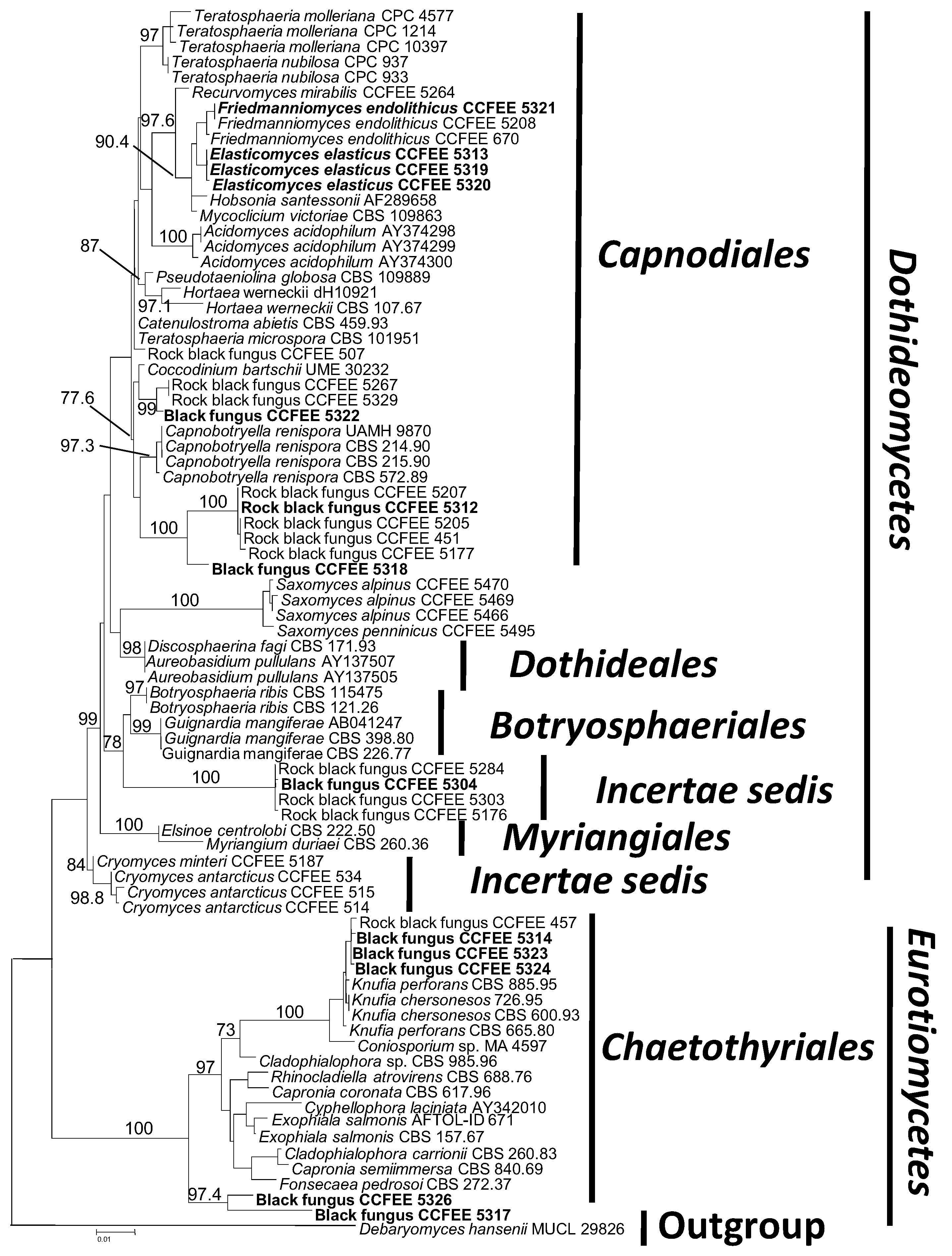
3. Results and Discussion


| Temperature (°C) | |||||||
|---|---|---|---|---|---|---|---|
| Isolates | 0 | 5 | 10 | 15 | 20 | 25 | 30 |
| CCFEE 5303 | 6.8 ± 1.8 | 4.3 ± 2.5 | 11.3 ± 0.4 | 13.8 ± 1.8 | - | - | - |
| CCFEE 5312 | 6.9 ± 1.3 | 7.8 ± 1.1 | 11.8 ± 0.4 | 18.2 ± 1.9 | 5.2 ± 1.6 | - | - |
| CCFEE 5313 | 16.4 ± 0.8 | 12.9 ± 0.6 | 25.8 ± 0.4 | 31.9 ± 3.7 | 30.8 ± 1.1 | 19.5 ± 3.5 | - |
| CCFEE 5314 | 4.4 ± 0.6 | 5.2 ± 1.6 | 7 ± 0 | 31.9 ± 3.7 | - | - | - |
| CCFEE 5317 | 4.8 ± 0.4 | 3.3 ± 0.4 | 8 ± 1.4 | 12 ± 0 | - | - | - |
| CCFEE 5318 | 7.3 ± 0.4 | 4.1 ± 1.8 | 13.3 ± 0.4 | 13 ± 0.4 | 13.5 ± 0.7 | 11.3 ± 3.2 | - |
| CCFEE 5319 | 15.5 ± 0.7 | 11.9 ± 0.1 | 22 ± 0 | 30 ± 0.7 | 24 ± 0.7 | 11.8 ± 1.1 | - |
| CCFEE 5320 | 14 ± 0.7 | 10.8 ± 1.1 | 23 ± 1.4 | 27.7 ± 0.9 | 22.5 ± 0.7 | 16.8 ± 1.8 | - |
| CCFEE 5321 | 3.5 ± 0.7 | 4.7 ± 0.9 | 8.8 ± 0 | 10.3 ± 1.8 | - | - | - |
| CCFEE 5322 | 10.3 ± 1.8 | 9.8 ± 1.1 | 14 ± 1.4 | 18.4 ± 2.3 | 4 ± 0 | - | - |
| CCFEE 5323 | 7 ± 0.7 | 4.7 ± 0.9 | 7 ± 2.1 | 18 ± 0.7 | 20.5 ± 0.7 | - | - |
| CCFEE 5324 | 3.5 ± 0.7 | 4.1 ± 1.8 | 5.3 ± 0.4 | 8.8 ± 0 | - | - | - |
| CCFEE 5326 | 2.5 ± 0 | 3.5 ± 0.7 | 4.1 ± 1.8 | 7 ± 0.7 | - | - | - |
4. Conclusions
Acknowledgements
References
- Plemenitaš, A.; Gunde-Cimerman, N. Cellular responses in the halophilic black yeast Hortaea werneckii to high environmental salinity. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 455–470. [Google Scholar]
- Baker, B.J.; Lutz, M.A.; Dawson, S.C.; Bond, P.L.; Banfield, J.F. Metabolically active eukaryotic communities in extremely acidic mine drainage. Appl. Environ. Microbiol. 2004, 70, 6264–6271. [Google Scholar] [CrossRef]
- Selbmann, L.; Egidi, E.; Isola, D.; Onofri, S.; Zucconi, Z.; de Hoog, G.S.; Chinaglia, S.; Testa, L.; Tosi, S.; Balestrazzi, A.; et al. Biodiversity, evolution and adaptation of fungi in extreme environments. Plant Biosyst. 2012, 147. [Google Scholar] [CrossRef]
- Isola, D.; Selbmann, L.; de Hoog, G.S.; Fenice, M.; Onofri, S.; Prenafeta-Boldú, F.X.; Zucconi, L. Isolation and screening of black fungi as degraders of volatile aromatic hydrocarbons. Mycopathologia 2013. [Google Scholar] [CrossRef]
- Staley, J.T.; Palmer, F.; Adams, J.B. Microcolonial fungi: Common inhabitants on desert rocks? Science 1982, 215, 1093–1095. [Google Scholar]
- Ruibal, C.; Gonzalo, P.; Bills, G.F. Isolation and characterization of melanized fungi from limestone formations in Mallorca. Mycol. Progress 2005, 4, 23–38. [Google Scholar] [CrossRef]
- Selbmann, L.; de Hoog, G.S.; Mazzaglia, A.; Friedmann, E.I.; Onofri, S. Fungi at the edge of life: Cryptoendolithic black fungi from Antarctic desert. Stud. Mycol. 2005, 51, 1–32. [Google Scholar]
- Sert, H.B.; Sümbül, H.; Sterflinger, K. Microcolonial fungi from antique marbles in Perge/Side/Termessos (Antalya/Turkey). Antonie Leeuwenhoek 2007, 91, 217–227. [Google Scholar] [CrossRef]
- Sert, H.B.; Sümbül, H.; Sterflinger, K. Sarcinomyces sideticae, a new black yeast from historical marble monuments in Side (Antalya, Turkey). Bot. J. Linnean Soc. 2007, 154, 373–380. [Google Scholar] [CrossRef]
- Sert, H.B.; Sümbül, H.; Sterflinger, K. A new species of Capnobotryella from monument surfaces. Mycol. Res. 2007, 111, 1235–1241. [Google Scholar] [CrossRef]
- Marvasi, M.; Donnarumma, F.; Frandi, A.; Mastromei, G.; Sterflinger, K.; Tiano, P.; Perito, B. Black microcolonial fungi as deteriogens of two famous marble statues in Florence, Italy. Int. Biodeterior. Biodegrad. 2012, 68, 36–44. [Google Scholar] [CrossRef]
- Zucconi, L.; Gagliardi, M.; Isola, D.; Onofri, S.; Andaloro, M.C.; Pelosi, C.; Pogliani, P.; Selbmann, L. Biodeteriorigenous agents dwelling the wall paintings of the Holy Saviour’s Cave (Vallerano, Italy). Int. Biodeterior. Biodegrad. 2012, 70, 40–46. [Google Scholar] [CrossRef]
- Onofri, S.; Selbmann, L.; Zucconi, L.; de Hoog, G.S.; de los Rios, A.; Ruisi, S.; Grube, M. Fungal associations at the cold edge of life. In Algae and Cyanobacteria in Extreme Environments; Seckbach, J., Ed.; Springer: Dordrecht, The Netherlands, 2007; pp. 735–757. [Google Scholar]
- Harutyunyan, S.; Muggia, L.; Grube, M. Black fungi in lichens from seasonally arid habitats. Stud. Mycol. 2008, 61, 83–90. [Google Scholar] [CrossRef]
- Sterflinger, K. Black Yeast and Meristematic Fungi: Ecology, Diversity and Identification. In The Yeast Handbook. Biodiversity and Ecophysiology of Yeasts; Péter, G., Rosa, C., Eds.; Springer: Berlin, Germany, 2006; pp. 501–514. [Google Scholar]
- Nienow, J.A.; Friedmann, E.I. Terrestrial Litophytic (Rock) Communities. In Antarctic Microbiology; Friedmann, E.I., Ed.; Wiley-Liss: New York, NY, USA, 1993; pp. 343–412. [Google Scholar]
- Selbmann, L.; de Hoog, G.S.; Gerrits van den Ende, A.H.G.; Ruibal, C.; de Leo, F.; Zucconi, L.; Isola, D.; Ruisi, S.; Onofri, S. Drought meets acid: Three new genera in a Dothidealean clade of extremotolerant fungi. Stud. Mycol. 2008, 61, 1–20. [Google Scholar] [CrossRef]
- Friedmann, E.I. Endolithic microorganisms in the Antarctic Cold Desert. Science 1982, 215, 1045–1053. [Google Scholar]
- De la Torre, J.R.; Goebel, B.M.; Friedmann, E.I.; Pace, N.R. Microbial diversity of cryptoendolithic communities from the McMurdo Dry Valleys, Antarctica. Appl. Environ. Microbiol. 2003, 215, 3858–3867. [Google Scholar]
- Selbmann, L.; Zucconi, L.; Ruisi, S.; Grube, M.; Cardinale, M.; Onofri, S. Culturable bacteria associated with Antarctic lichens: Affiliation and psychrotolerance. Polar Biol. 2010, 33, 71–83. [Google Scholar] [CrossRef]
- Cardinale, M.; Vieira de Castro, J., Jr.; Müller, H.; Berg, G.; Grube, M. In situ analysis of the bacterial community associated with the reindeer lichen Cladonia arbuscula reveals predominance of Alphaproteobacteria. FEMS Microbiol. Ecol. 2008, 66, 63–71. [Google Scholar] [CrossRef]
- Grube, M.; Cardinale, M.; Vieira de Castro, J.; Müller, H.; Berg, G. Species-specificstructural and functional diversity of bacterial communitiesin lichen symbioses. ISME J. 2009, 3, 1105–1115. [Google Scholar] [CrossRef]
- Hodkinson, B.; Lutzoni, F. A microbiotic survey of lichen-associated bacteria reveals a new lineage from the Rhizobiales. Symbiosis 2009, 49, 163–180. [Google Scholar] [CrossRef]
- Bates, S.T.; Cropsey, G.W.G.; Caporaso, J.G.; Knight, R.; Fierer, N. Bacterial communities associated with the lichen symbiosis. Appl. Environ. Microbiol. 2011, 77, 1309–1314. [Google Scholar] [CrossRef]
- Grube, M.; Berg, G. Microbial consortia of bacteria and fungi with focus on the lichen symbiosis. Fungal Biol. Rev. 2009, 23, 72–85. [Google Scholar] [CrossRef]
- Lawrey, J.D.; Diederich, P. Lichenicolous fungi: Interactions, evolution, and biodiversity. Bryologist 2003, 106, 80–120. [Google Scholar] [CrossRef]
- Lawrey, J.D.; Binder, M.; Diederich, P.; Molina, M.C.; Sikaroodi, M.; Ertz, D. Phylogenetic diversity of lichen-associated homobasidiomycetes. Mol. Phylogenetics Evol. 2007, 44, 778–789. [Google Scholar] [CrossRef]
- Girlanda, M.; Isocrono, D.; Bianco, C.; Luppi-Mosca, A.M. Two foliose lichens as microfungal ecological niches. Mycologia 1997, 89, 531–536. [Google Scholar] [CrossRef]
- Suryanarayanan, T.S.; Thirunavukkarasu, N.; Hariharan, G.N.; Balaji, P. Occurrence of non-obligate microfungi inside lichen thalli. Sydowia 2005, 57, 120–130. [Google Scholar]
- U’Ren, J.; Lutzoni, F.; Miadlikowska, J.; Arnold, A.E. Community analysis reveals close affinities between endophytic and endolichenic fungi in mosses and lichens. Microb. Ecol. 2010, 60, 340–353. [Google Scholar] [CrossRef]
- Bates, S.T.; Berg-Lyons, D.; Lauber, C.L.; Walters, W.A.; Knight, R.; Fierer, N. A preliminary survey of lichen associated eukaryotes using Pyrosequencing. Lichenologist 2012, 44, 137–146. [Google Scholar] [CrossRef]
- Farrar, J.F. The Lichen as an Ecosystem: Observation and Experiment. In Lichenology: Progress and Problems; Brown, D.H., Hawksworth, D.L., Bailey, R.H., Eds.; Academic Press: London, UK, 1976; pp. 385–406. [Google Scholar]
- Castello, M. Lichens of the Terra Nova Bay area, Northern Victoria Land (Continental Antactica). Stud. Geobot. 2003, 22, 3–54. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.B.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols, a Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The ClustalX windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 24, 4876–4882. [Google Scholar]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 40, 783–791. [Google Scholar] [CrossRef]
- Van Uden, N. Temperature profiles of yeasts. Adv. Microbiol. Physiol. 1984, 25, 195–251. [Google Scholar] [CrossRef]
- Schoch, C.L.; Shoemaker, R.A.; Seifert, K.A.; Hambleton, S.; Spatafora, J.W.; Crous, P.W. A multigene phylogeny of the Dothideomycetes using four nuclear loci. Mycologia 2006, 98, 1041–1052. [Google Scholar] [CrossRef]
- Ruibal, C.; Gueidan, C.; Selbmann, L.; Gorbushina, A.A.; Crous, P.W.; Groenewald, J.Z.; Muggia, L.; Grube, M.; Isola, D.; Schoch, C.L.; et al. Phylogeny of rock-inhabiting fungi related to Dothideomycetes. Stud. Mycol. 2009, 64, 123–133. [Google Scholar] [CrossRef]
- Tsuneda, A.; Hambleton, S.; Currah, R.S. The anamorph genus Knufia and its phylogenetically allied species in Coniosporium, Sarcinomyces, and Phaeococcomyces. Botany 2011, 89, 523–536. [Google Scholar] [CrossRef]
- Muggia, L.; Hafellner, J.; Wirtz, N.; Hawksworth, D.L.; Grube, M. The sterile microfilamentous lichenized fungi Cystocoleus ebeneus and Racodium rupestre are relatives of plant pathogens and clinically important dothidealean fungi. Mycol. Res. 2008, 112, 50–56. [Google Scholar] [CrossRef]
- Onofri, S.; Pagano, S.; Zucconi, L.; Tosi, S. Friedmanniomyces endolithicus (Fungi, Hyphomycetes), anam.-gen. and sp.nov., from continental Antarctica. Nova Hedwig. 1999, 68, 175–181. [Google Scholar]
- Onofri, S.; Anastasi, A.; Del Frate, G.; di Piazza, S.; Garnero, N.; Guglielminetti, M.; Isola, D.; Panno, L.; Ripa, C.; Selbmann, L.; et al. Biodiversity of rock, beach and water fungi in Italy. Plant Biosyst. 2011, 45, 978–987. [Google Scholar]
- Prenafeta-Boldú, F.X.; Summerbell, R.C.; de Hoog, G.S. Fungi growing on aromatic hydrocarbons: Biotechnology’s unexpected encounter with biohazard. FEMS Microbiol. Rev. 2006, 30, 109–130. [Google Scholar] [CrossRef]
- Diederich, P.; Lawrey, J.D. New lichenicolous, muscicolous, corticolous and lignicolous taxa of Burgoa s. l. and Marchandiomyces s. l. (anamorphic Basidiomycota), a new genus for Omphalina foliacea, and a catalogue and a key to the non-lichenized, bulbilliferous basidiomycetes. Mycol. Progress 2007, 6, 61–80. [Google Scholar] [CrossRef]
- Muggia, L.; Gueidan, C.; Knudsen, K.; Perlmutter, G.; Grube, M. The lichen connections of black fungi. Mycopathologia 2012. [Google Scholar] [CrossRef]
- Selbmann, L.; Isola, D.; Egidi, E.; Zucconi, L.; Gueidan, C.; de Hoog, G.S.; Onofri, S. Rock inhabiting fungi: Saxomyces gen. nov. and four new species from the Alps. Fungal Diver. 2013, in press. [Google Scholar]
- Gorbushina, A.A.; Beck, A.; Shulte, A. Microcolonial rock inhabiting fungi and lichen photobionts: Evidence for mutualistic interactions. Mycol. Res. 2005, 109, 1288–1296. [Google Scholar] [CrossRef]
- Brunauer, G.; Blaha, J.; Hager, A.; Turk, R.; Stocker-Worgotter, E.; Grube, M. An isolated lichenicolous fungus forms lichenoid structures when co-cultured with various coccoid algae. Symbiosis 2007, 44, 127–136. [Google Scholar]
- Gueidan, C.; Ruibal, C.; de Hoog, G.S.; Gorbushina, A.; Untereiner, W.A.; Lutzoni, F. A rock-inhabiting ancestor for mutualistic and pathogen-rich fungal lineages. Stud. Mycol. 2008, 61, 111–119. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Selbmann, L.; Grube, M.; Onofri, S.; Isola, D.; Zucconi, L. Antarctic Epilithic Lichens as Niches for Black Meristematic Fungi. Biology 2013, 2, 784-797. https://doi.org/10.3390/biology2020784
Selbmann L, Grube M, Onofri S, Isola D, Zucconi L. Antarctic Epilithic Lichens as Niches for Black Meristematic Fungi. Biology. 2013; 2(2):784-797. https://doi.org/10.3390/biology2020784
Chicago/Turabian StyleSelbmann, Laura, Martin Grube, Silvano Onofri, Daniela Isola, and Laura Zucconi. 2013. "Antarctic Epilithic Lichens as Niches for Black Meristematic Fungi" Biology 2, no. 2: 784-797. https://doi.org/10.3390/biology2020784
APA StyleSelbmann, L., Grube, M., Onofri, S., Isola, D., & Zucconi, L. (2013). Antarctic Epilithic Lichens as Niches for Black Meristematic Fungi. Biology, 2(2), 784-797. https://doi.org/10.3390/biology2020784









