Stress Reduction Potential in Mice Ingesting DNA from Salmon Milt
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Reagents
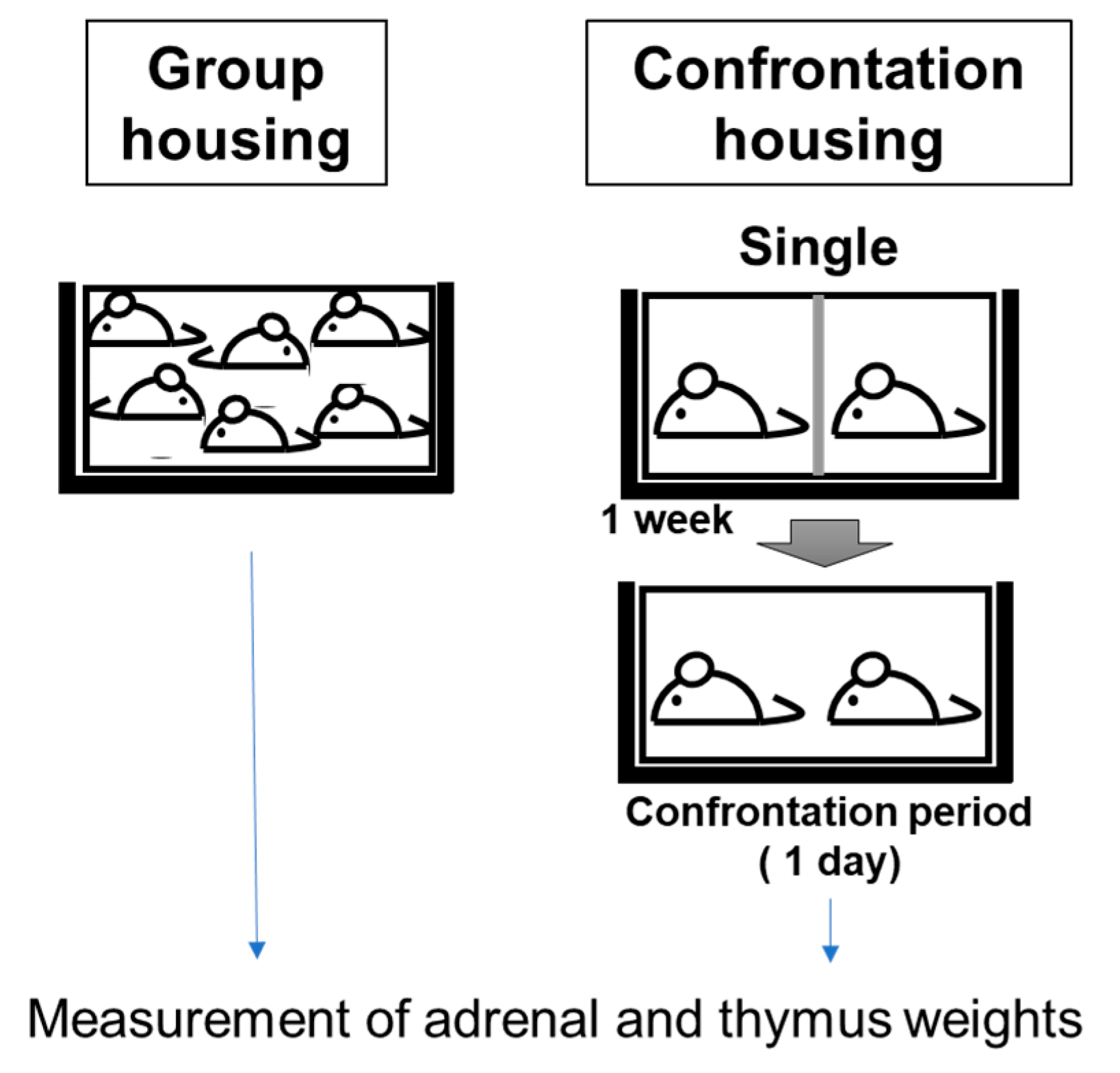
2.3. Screening Method for Anti-Stress Effects
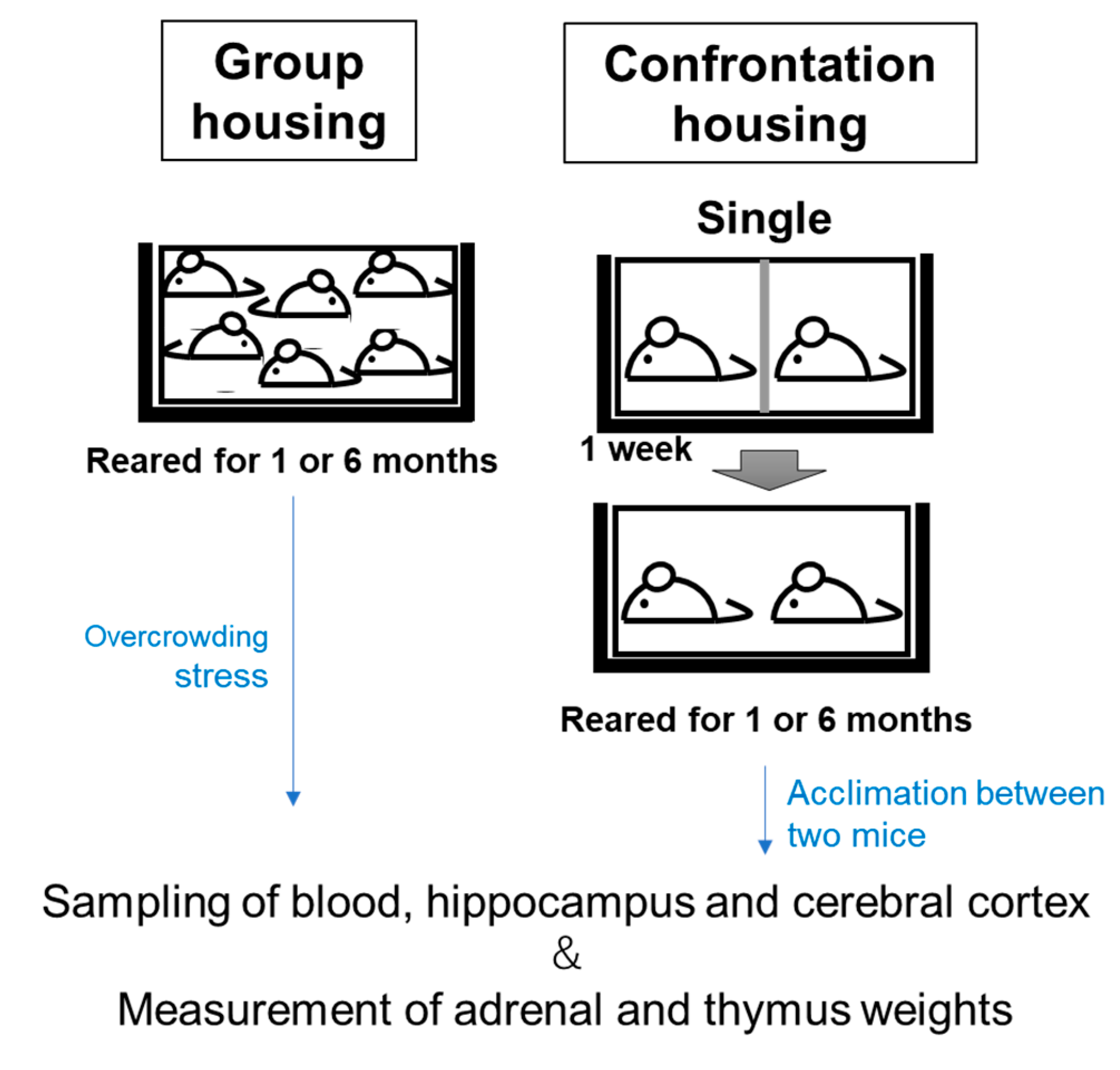
2.4. Anti-Stress Effects of DNA-Na on Chronic Crowding Stress in Older Mice
2.5. Quantitative Real-Time Reverse-Transcription PCR (qRT-PCR)
2.6. Measurement of Oxidative Damage in the Brain and Serum
2.7. Statistical Analyses
3. Results
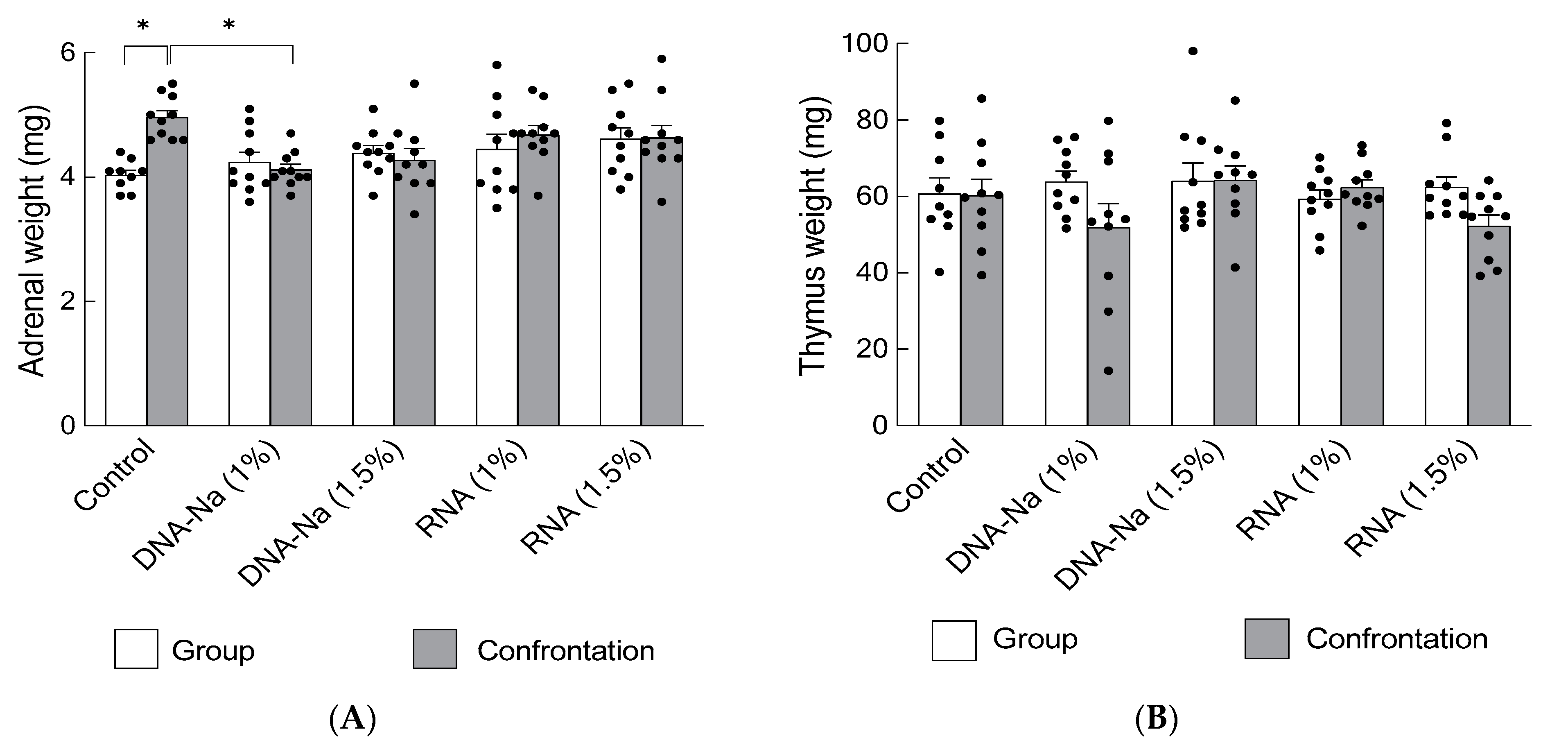
3.1. Suppression of Adrenal Hypertrophy by Ingestion of DNA-Na from Salmon Milt
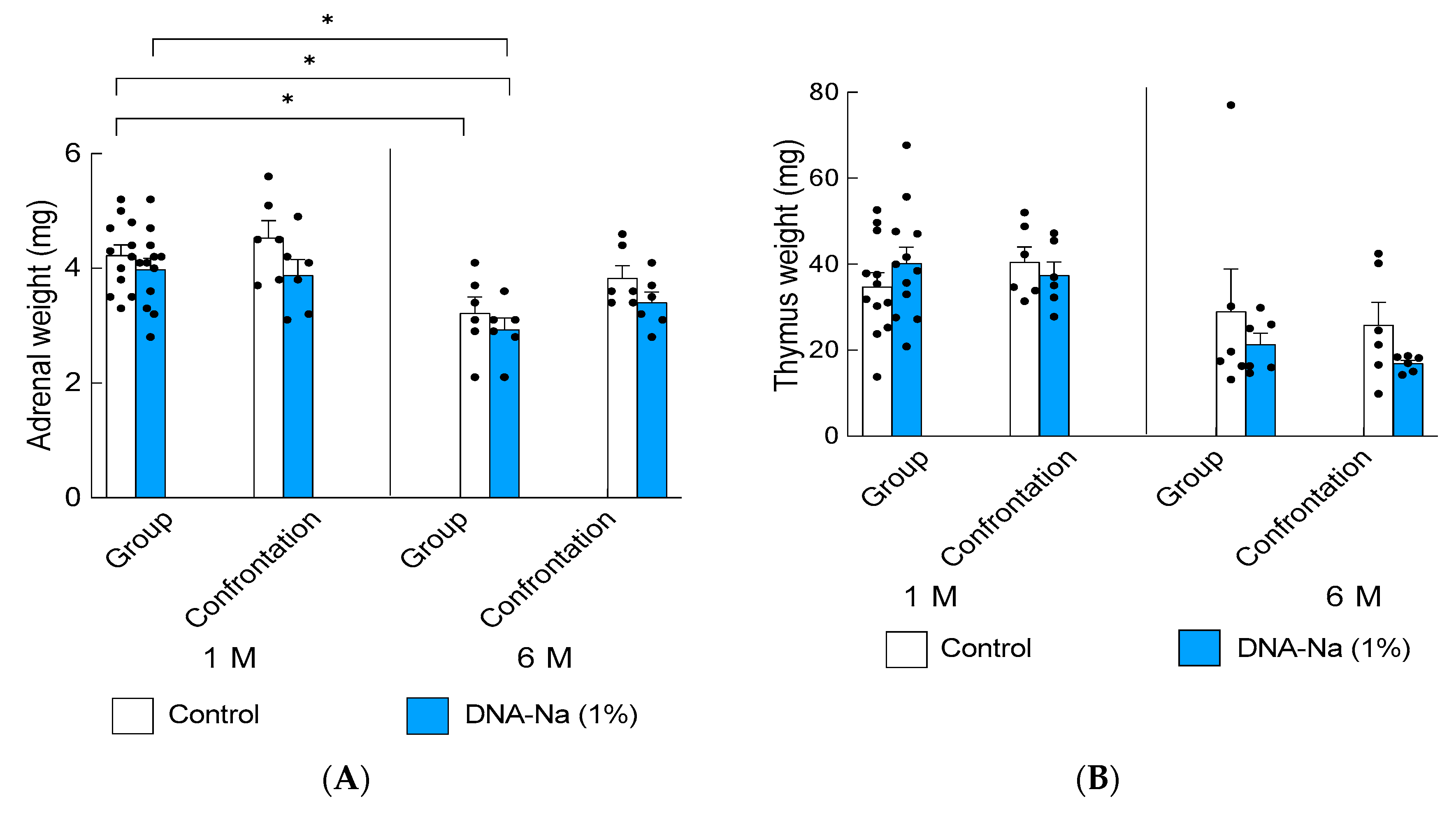
3.2. Effects of DNA-Na Ingestion on Chronic Crowding Stress in Older Mice
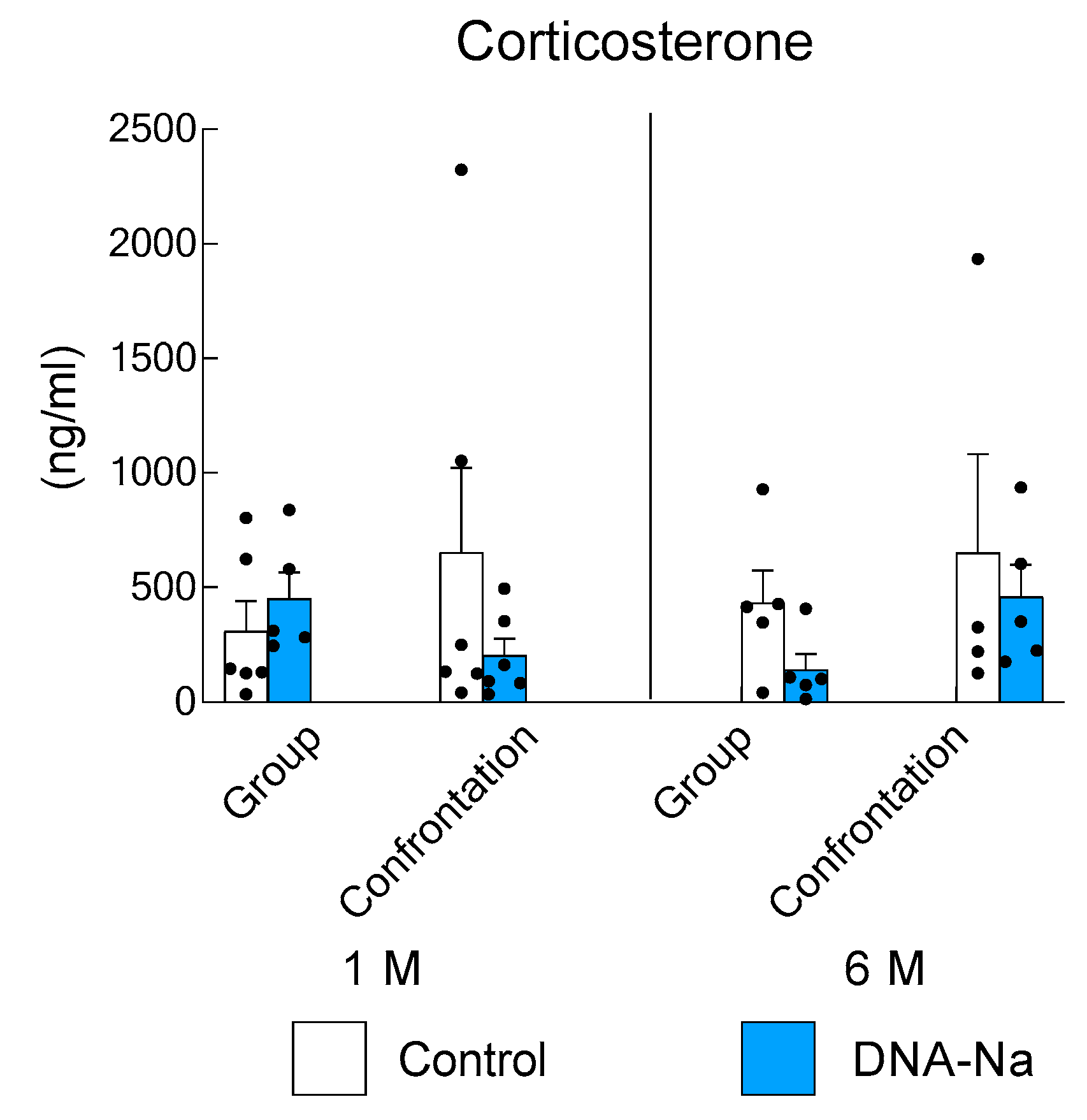
3.3. Effects of DNA-Na Intake on Serum Corticosterone Levels
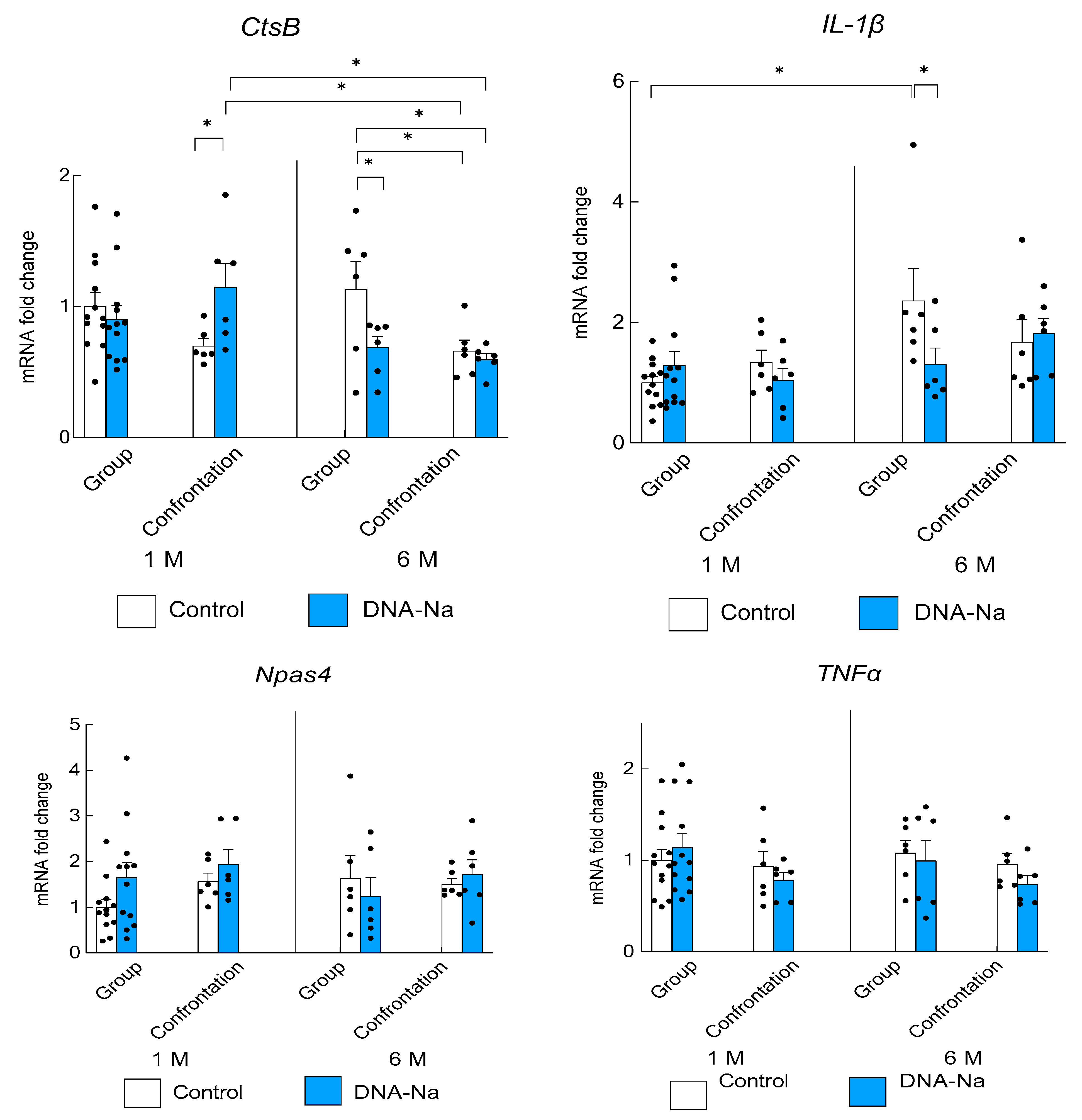
3.4. Effects of DNA-Na Intake on Gene Expression in the Brain
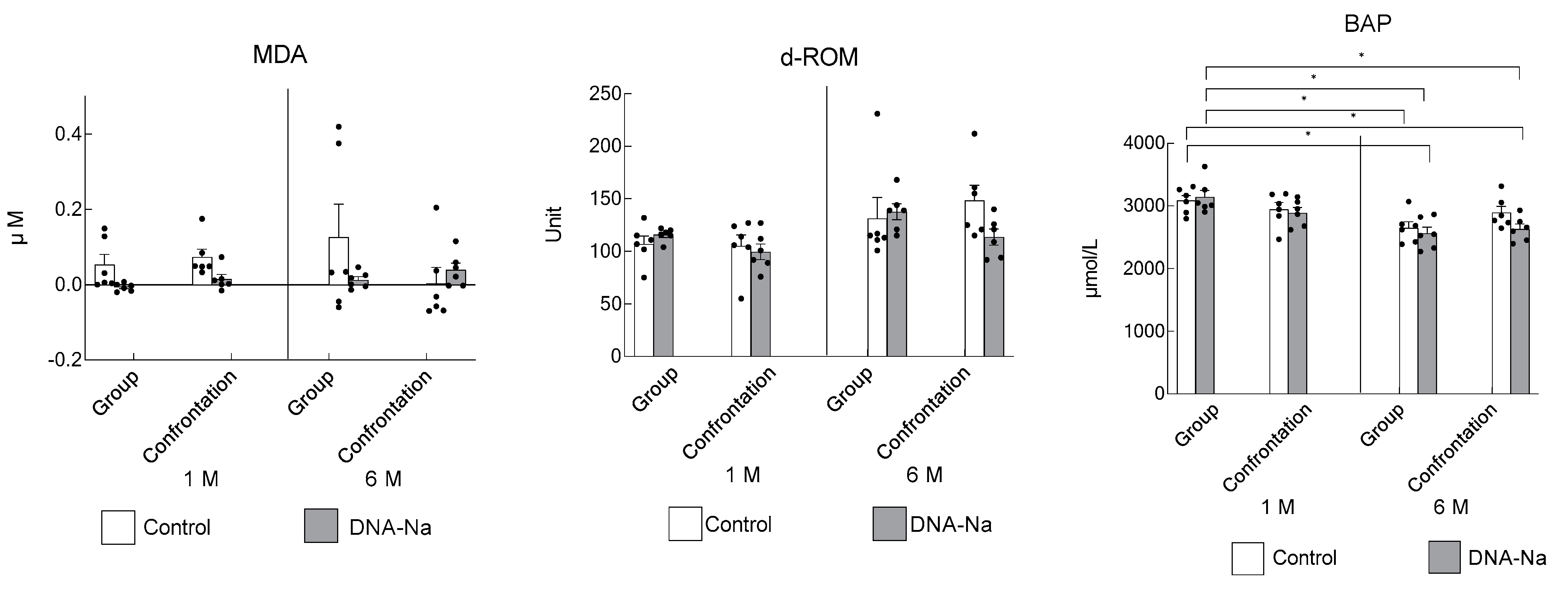
3.5. Effects of DNA-Na Intake on Oxidative Damage
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sauer, N.; Mosenthin, R.; Bauer, E. The role of dietary nucleotides in single-stomached animals. Nutr. Res. Rev. 2011, 24, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, T.; Tatibana, M. Metabolic fate of pyrimidines and purines in dietary nucleic acids ingested by mice. Biochim. Biophys. Acta 1978, 521, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Hess, J.R.; Greenberg, N.A. The role of nucleotides in the immune and gastrointestinal systems: Potential clinical applications. Nutr. Clin. Pract. 2012, 27, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Aggett, P.; Leach, J.L.; Rueda, R.; MacLean, W.C., Jr. Innovation in infant formula development: A reassessment of ribonucleotides in 2002. Nutrition 2003, 19, 375–384. [Google Scholar] [CrossRef]
- Uauy, R.; Quan, R.; Gil, A. Role of nucleotides in intestinal development and repair: Implications for infant nutrition. J. Nutr. 1994, 124, 1436S–1441S. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, F.B.; Kulkarni, A.D.; Fanslow, W.C.; Pizzini, R.P.; Kumar, S.; Van Buren, C.T. Role of RNA as a dietary source of pyrimidines and purines in immune function. Nutrition 1990, 6, 45–52, discussion 59–62. [Google Scholar]
- Sánchez-Pozo, A.; Gil, A. Nucleotides as semiessential nutritional components. Br. J. Nutr. 2002, 87 (Suppl. S1), S135–S137. [Google Scholar] [CrossRef]
- Young, J.D.; Yao, S.Y.; Baldwin, J.M.; Cass, C.E.; Baldwin, S.A. The human concentrative and equilibrative nucleoside transporter families, SLC28 and SLC29. Mol. Aspects Med. 2013, 34, 529–547. [Google Scholar] [CrossRef]
- Ermakov, A.V.; Konkova, M.S.; Kostyuk, S.V.; Izevskaya, V.L.; Baranova, A.; Veiko, N.N. Oxidized extracellular DNA as a stress signal in human cells. Oxid. Med. Cell Longev. 2013, 2013, 649747. [Google Scholar] [CrossRef]
- Kostyuk, S.V.; Tabakov, V.J.; Chestkov, V.V.; Konkova, M.S.; Glebova, K.V.; Baydakova, G.V.; Ershova, E.S.; Izhevskaya, V.L.; Baranova, A.; Veiko, N.N. Oxidized DNA induces an adaptive response in human fibroblasts. Mutat. Res. 2013, 747–748, 6–18. [Google Scholar] [CrossRef]
- Filev, A.D.; Shmarina, G.V.; Ershova, E.S.; Veiko, N.N.; Martynov, A.V.; Borzikova, M.A.; Poletkina, A.A.; Dolgikh, O.A.; Veiko, V.P.; Bekker, A.A.; et al. Oxidized Cell-Free DNA Role in the Antioxidant Defense Mechanisms under Stress. Oxid. Med. Cell Longev. 2019, 2019, 1245749. [Google Scholar] [CrossRef]
- Kostyuk, S.V.; Porokhovnik, L.N.; Ershova, E.S.; Malinovskaya, E.M.; Konkova, M.S.; Kameneva, L.V.; Dolgikh, O.A.; Veiko, V.P.; Pisarev, V.M.; Martynov, A.V.; et al. Changes of KEAP1/NRF2 and IKB/NF-κB Expression Levels Induced by Cell-Free DNA in Different Cell Types. Oxid. Med. Cell Longev. 2018, 2018, 1052413. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Protective and damaging effects of stress mediators: Central role of the brain. Dialogues Clin. Neurosci. 2006, 8, 367–381. [Google Scholar] [CrossRef] [PubMed]
- Olvera Alvarez, H.A.; Kubzansky, L.D.; Campen, M.J.; Slavich, G.M. Early life stress, air pollution, inflammation, and disease: An integrative review and immunologic model of social-environmental adversity and lifespan health. Neurosci. Biobehav. Rev. 2018, 92, 226–242. [Google Scholar] [CrossRef] [PubMed]
- Juruena, M.F.; Eror, F.; Cleare, A.J.; Young, A.H. The Role of Early Life Stress in HPA Axis and Anxiety. Adv. Exp. Med. Biol. 2020, 1191, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Dar, T.; Radfar, A.; Abohashem, S.; Pitman, R.K.; Tawakol, A.; Osborne, M.T. Psychosocial Stress and Cardiovascular Disease. Curr. Treat. Options Cardiovasc. Med. 2019, 21, 23. [Google Scholar] [CrossRef]
- Unno, K.; Nakamura, Y. Green Tea Suppresses Brain Aging. Molecules 2021, 26, 4897. [Google Scholar] [CrossRef]
- Smith, S.M.; Vale, W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006, 8, 383–395. [Google Scholar] [CrossRef]
- Spencer, R.L.; Deak, T. A users guide to HPA axis research. Physiol. Behav. 2017, 178, 43–65. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Figueiredo, H.F.; Ostrander, M.M.; Choi, D.C.; Engeland, W.C.; Herman, J.P. Chronic stress induces adrenal hyperplasia and hypertrophy in a subregion-specific manner. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E965–E973. [Google Scholar] [CrossRef]
- Unno, K.; Iguchi, K.; Tanida, N.; Fujitani, K.; Takamori, N.; Yamamoto, H.; Ishii, N.; Nagano, H.; Nagashima, T.; Hara, A.; et al. Ingestion of theanine, an amino acid in tea, suppresses psychosocial stress in mice. Exp. Physiol. 2013, 98, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Taguchi, K.; Konishi, T.; Ozeki, M.; Nakamura, Y. Theanine, a Tea-Leaf-Specific Amino Acid, Alleviates Stress through Modulation of Npas4 Expression in Group-Housed Older Mice. Int. J. Mol. Sci. 2023, 24, 3983. [Google Scholar] [CrossRef]
- Unno, K.; Sumiyoshi, A.; Konishi, T.; Hayashi, M.; Taguchi, K.; Muguruma, Y.; Inoue, K.; Iguchi, K.; Nonaka, H.; Kawashima, R.; et al. Theanine, the Main Amino Acid in Tea, Prevents Stress-Induced Brain Atrophy by Modifying Early Stress Responses. Nutrients 2020, 12, 174. [Google Scholar] [CrossRef] [PubMed]
- Ibi, D.; Takuma, K.; Koike, H.; Mizoguchi, H.; Tsuritani, K.; Kuwahara, Y.; Kamei, H.; Nagai, T.; Yoneda, Y.; Nabeshima, T. Social isolation rearing-induced impairment of the hippocampal neurogenesis is associated with deficits in spatial memory and emotion-related behaviors in juvenile mice. J. Neurochem. 2008, 105, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Takano, S.; Uchida, K.; Miyagi, M.; Inoue, G.; Aikawa, J.; Iwabuchi, K.; Takaso, M. Adrenomedullin Regulates IL-1β Gene Expression in F4/80+ Macrophages during Synovial Inflammation. J. Immunol. Res. 2017, 2017, 9832430. [Google Scholar] [CrossRef]
- Dong-Newsom, P.; Powell, N.D.; Bailey, M.T.; Padgett, D.A.; Sheridan, J.F. Repeated social stress enhances the innate immune response to a primary HSV-1 infection in the cornea and trigeminal ganglia of Balb/c mice. Brain Behav. Immun. 2010, 24, 273–280. [Google Scholar] [CrossRef]
- Rocchi, A.; Carminati, E.; De Fusco, A.; Kowalska, J.A.; Floss, T.; Benfenati, F. REST/NRSF deficiency impairs autophagy and leads to cellular senescence in neurons. Aging Cell 2021, 20, e13471. [Google Scholar] [CrossRef]
- Badcock, N.R.; Zoanetti, G.D.; Martin, E.S. Nonchromatographic assay for malondialdehyde-thiobarbituric acid adduct with HPLC equivalence. Clin. Chem. 1997, 43, 1655–1657. [Google Scholar] [CrossRef]
- Choi, S.; Zhang, B.; Ma, S.; Gonzalez-Celeiro, M.; Stein, D.; Jin, X.; Kim, S.T.; Kang, Y.L.; Besnard, A.; Rezza, A.; et al. Corticosterone inhibits GAS6 to govern hair follicle stem-cell quiescence. Nature 2021, 592, 428–432. [Google Scholar] [CrossRef]
- Schirrmacher, V. Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications. Biomedicines 2021, 9, 293. [Google Scholar] [CrossRef]
- Agathokleous, E.; Calabrese, E.J. Hormesis: A General Biological Principle. Chem. Res. Toxicol. 2022, 35, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Kubota, Y.; Toyoda, A. Effects of diet quality on vulnerability to mild subchronic social defeat stress in mice. Nutr. Neurosci. 2016, 19, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Hatmal, M.M.; Al-Hatamleh, M.A.I.; Olaimat, A.N.; Alshaer, W.; Hasan, H.; Albakri, K.A.; Alkhafaji, E.; Issa, N.N.; Al-Holy, M.A.; Abderrahman, S.M.; et al. Immunomodulatory Properties of Human Breast Milk: MicroRNA Contents and Potential Epigenetic Effects. Biomedicines 2022, 10, 1219. [Google Scholar] [CrossRef]
- Goukassian, D.A.; Bagheri, S.; el-Keeb, L.; Eller, M.S.; Gilchrest, B.A. DNA oligonucleotide treatment corrects the age-associated decline in DNA repair capacity. FASEB J. 2002, 16, 754–756. [Google Scholar] [CrossRef]
- Hadshiew, I.M.; Eller, M.S.; Gasparro, F.P.; Gilchrest, B.A. Stimulation of melanogenesis by DNA oligonucleotides: Effect of size, sequence and 5′ phosphorylation. J. Dermatol. Sci. 2001, 25, 127–138. [Google Scholar] [CrossRef]
- Ni, J.; Wu, Z.; Stoka, V.; Meng, J.; Hayashi, Y.; Peters, C.; Qing, H.; Turk, V.; Nakanishi, H. Increased expression and altered subcellular distribution of cathepsin B in microglia induce cognitive impairment through oxidative stress and inflammatory response in mice. Aging Cell. 2019, 18, e12856. [Google Scholar] [CrossRef] [PubMed]
- Yambire, K.F.; Rostosky, C.; Watanabe, T.; Pacheu-Grau, D.; Torres-Odio, S.; Sanchez-Guerrero, A.; Senderovich, O.; Meyron-Holtz, E.G.; Milosevic, I.; Frahm, J.; et al. Impaired lysosomal acidification triggers iron deficiency and inflammation in vivo. Elife 2019, 8, e51031. [Google Scholar] [CrossRef]
- Terada, K.; Yamada, J.; Hayashi, Y.; Wu, Z.; Uchiyama, Y.; Peters, C.; Nakanishi, H. Involvement of cathepsin B in the processing and secretion of interleukin-1beta in chromogranin A-stimulated microglia. Glia 2010, 58, 114–124. [Google Scholar] [CrossRef]
- Yu, J.; Zhu, H.; Taheri, S.; Monday, W.L.; Perry, S.; Kindy, M.S. Reduced Neuroinflammation and Improved Functional Recovery after Traumatic Brain Injury by Prophylactic Diet Supplementation in Mice. Nutrients 2019, 11, 299. [Google Scholar] [CrossRef]
- Wu, Z.; Ni, J.; Liu, Y.; Teeling, J.L.; Takayama, F.; Collcutt, A.; Ibbett, P.; Nakanishi, H. Cathepsin B plays a critical role in inducing Alzheimer’s disease-like phenotypes following chronic systemic exposure to lipopolysaccharide from Porphyromonas gingivalis in mice. Brain Behav. Immun. 2017, 65, 350–361. [Google Scholar] [CrossRef]
- Meng, J.; Liu, Y.; Xie, Z.; Qing, H.; Lei, P.; Ni, J. Nucleus distribution of cathepsin B in senescent microglia promotes brain aging through degradation of sirtuins. Neurobiol. Aging 2020, 96, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Waititu, S.M.; Heo, J.M.; Patterson, R.; Nyachoti, C.M. Dose-response effects of in-feed antibiotics on growth performance and nutrient utilization in weaned pigs fed diets supplemented with yeast-based nucleotides. Anim. Nutr. 2015, 1, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Valini, G.A.C.; Duarte, M.S.; Calderano, A.A.; Teixeira, L.M.; Rodrigues, G.A.; Fernandes, K.M.; Veroneze, R.; Serão, N.V.L.; Mantovani, H.C.; Rocha, G.C. Dietary nucleotide supplementation as an alternative to in-feed antibiotics in weaned piglets. Animal 2021, 15, 100021. [Google Scholar] [CrossRef]
- Koert, A.; Ploeger, A.; Bockting, C.L.H.; Schmidt, M.V.; Lucassen, P.J.; Schrante, A.; Mul, J.D. The social instability stress paradigm in rat and mouse: A systematic review of protocols, limitations, and recommendations. Neurobiol. Stress 2021, 15, 100410. [Google Scholar] [CrossRef] [PubMed]
- Basiouni, S.; Tellez-Isaias, G.; Latorre, J.D.; Graham, B.D.; Petrone-Garcia, V.M.; El-Seedi, H.R.; Yalçın, S.; El-Wahab, A.A.; Visscher, C.; May-Simera, H.L.; et al. Anti-Inflammatory and Antioxidative Phytogenic Substances against Secret Killers in Poultry: Current Status and Prospects. Vet. Sci. 2023, 10, 55. [Google Scholar] [CrossRef]
- Mitarai, M.; Hsu, T.-F.; Wang, M.-F.; Hiroshi Hirahara, H.; Sekido, H.; Hoshino, Y.; Enari, H.; Yamamoto, S. Effects of dietary DNA from chum salmon milt on memory and age-related changes in senescence-accelerated mice. Nippon Shokuhin Kagaku Kogaku Kaishi 2008, 55, 461–467. [Google Scholar] [CrossRef]
- Nakamichi, N.; Nakao, S.; Masuo, Y.; Koike, A.; Matsumura, N.; Nishiyama, M.; Al-Shammari, A.H.; Sekiguchi, H.; Sutoh, K.; Usumi, K.; et al. Hydrolyzed Salmon Milt Extract Enhances Object Recognition and Location Memory Through an Increase in Hippocampal Cytidine Nucleoside Levels in Normal Mice. J. Med. Food 2019, 22, 408–415. [Google Scholar] [CrossRef]
- Kawarasaki, M.; Akita, R.; Sugimoto, M.; Enari, H.; Yamamoto, S. Effect of taking DNA-Na from chum salmon milt DNA on ethanol metabolism. Nippon Shokuhin Kagaku Kogaku Kaishi 2008, 55, 632–636. [Google Scholar] [CrossRef]
- Kojima-Yuasa, A.; Goto, M.; Yoshikawa, E.; Morita, Y.; Sekiguchi, H.; Sutoh, K.; Usumi, K.; Matsui-Yuasa, I. Protective Effects of Hydrolyzed Nucleoproteins from Salmon Milt against Ethanol-Induced Liver Injury in Rats. Mar. Drugs 2016, 14, 232. [Google Scholar] [CrossRef]
- Xue, Y.; Li, X.; Tian, Y.; Huang, X.; Zhang, L.; Li, J.; Hou, H.; Dong, P.; Wang, J. Salmon sperm DNA prevents acute liver injury by regulating alcohol-induced steatosis and restores chronic hepatosis via alleviating inflammation and apoptosis. J. Food Biochem. 2022, 46, e14346. [Google Scholar] [CrossRef]
- Huang, X.; Chu, X.; Tian, Y.; Xue, Y.; Zhang, L.; Li, J.; Hou, H.; Dong, P.; Wang, J. Preventive effect of salmon sperm DNA on acute carbon tetrachloride-induced liver injury in mice through Nrf2/ARE and mitochondrial apoptosis pathway. Food Sci. Nutr. 2022, 11, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.W.; Stratton, M.S.; Ferguson, B.S. Dietary natural products as epigenetic modifiers in aging-associated inflammation and disease. Nat. Prod. Rep. 2020, 37, 653–676. [Google Scholar] [CrossRef] [PubMed]
- Marino, M.; Mele, E.; Pastorino, G.M.G.; Meccariello, R.; Operto, F.F.; Santoro, A.; Viggiano, A. Neuroinflammation: Molecular Mechanisms and Therapeutic Perspectives. Cent. Nerv. Syst. Agents Med. Chem. 2022, 22, 160–174. [Google Scholar] [CrossRef] [PubMed]
| Gene | Forward Sequence (5′ to 3′) | Reverse Sequence (5′ to 3′) | Ref. |
|---|---|---|---|
| β-actin | TGACAGGATGCAGAAGGAGA | GCTGGAAGGTGGACAGTGAG | |
| Npas4 | AGCATTCCAGGCTCATCTGAA | GGCGAAGTAAGTCTTGGTAGGATT | [24] |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT | [25] |
| TNFα | CTGTCTACTGAACTTCGGGGTGAT | GGTCTGGGCCATAGAACTGATG | [26] |
| CtsB | CTGCTGAAGACCTGCTTA | AATTGTAGACTCCACCTGAA | [27] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unno, K.; Taguchi, K.; Fujita, M.; Sutoh, K.; Nakamura, Y. Stress Reduction Potential in Mice Ingesting DNA from Salmon Milt. Biology 2023, 12, 978. https://doi.org/10.3390/biology12070978
Unno K, Taguchi K, Fujita M, Sutoh K, Nakamura Y. Stress Reduction Potential in Mice Ingesting DNA from Salmon Milt. Biology. 2023; 12(7):978. https://doi.org/10.3390/biology12070978
Chicago/Turabian StyleUnno, Keiko, Kyoko Taguchi, Mica Fujita, Keita Sutoh, and Yoriyuki Nakamura. 2023. "Stress Reduction Potential in Mice Ingesting DNA from Salmon Milt" Biology 12, no. 7: 978. https://doi.org/10.3390/biology12070978
APA StyleUnno, K., Taguchi, K., Fujita, M., Sutoh, K., & Nakamura, Y. (2023). Stress Reduction Potential in Mice Ingesting DNA from Salmon Milt. Biology, 12(7), 978. https://doi.org/10.3390/biology12070978







