From Oxytocin to Compassion: The Saliency of Distress
Abstract
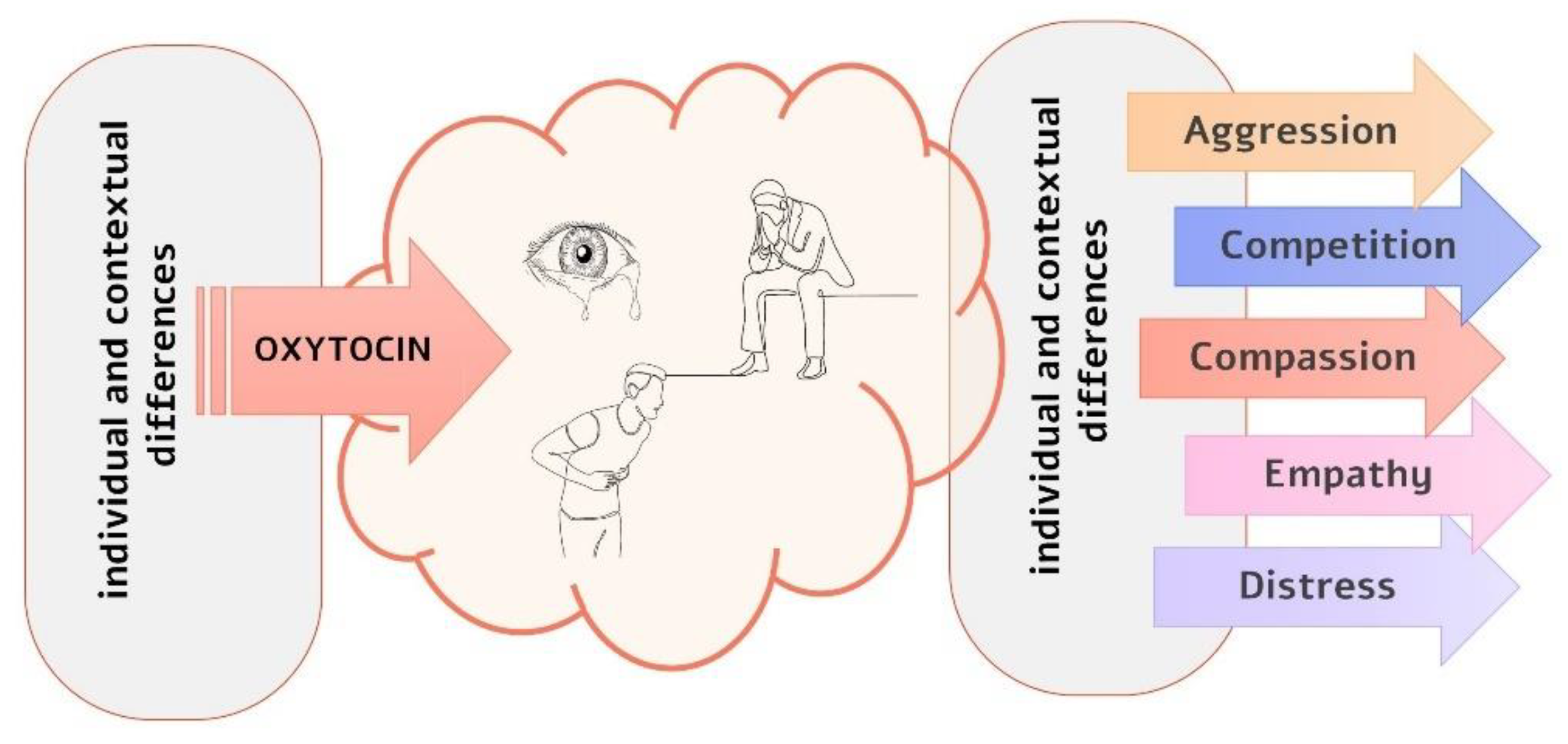
Simple Summary
Abstract
1. Introduction
2. The Social Saliency Hypothesis
3. From Social Saliency to Compassion
4. A Putative Neurobiological Mechanism of Social Saliency Evaluation
5. Individual Differences in Compassion
6. Through the Lens of Attachment
7. Final Notes and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Goetz, J.L.; Keltner, D.; Simon-Thomas, E. Compassion: An Evolutionary Analysis and Empirical Review. Psychol. Bull. 2010, 136, 351–374. [Google Scholar] [CrossRef] [PubMed]
- Spikins, P. Prehistoric origins: The compassion of far distant strangers. In Compassion; Routledge: London, UK, 2017; pp. 16–30. [Google Scholar]
- Gilbert, P. Explorations into the nature and function of compassion. Curr. Opin. Psychol. 2019, 28, 108–114. [Google Scholar] [CrossRef]
- Weisz, E.; Zaki, J. Motivated empathy: A social neuroscience perspective. Curr. Opin. Psychol. 2018, 24, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Leiberg, S.; Klimecki, O.; Singer, T. Short-Term Compassion Training Increases Prosocial Behavior in a Newly Developed Prosocial Game. PLoS ONE 2011, 6, e17798. [Google Scholar] [CrossRef] [PubMed]
- Donald, J.N.; Sahdra, B.K.; Van Zanden, B.; Duineveld, J.J.; Atkins, P.W.; Marshall, S.L.; Ciarrochi, J. Does your mindfulness benefit others? A systematic review and meta-analysis of the link between mindfulness and prosocial behaviour. Br. J. Psychol. 2019, 110, 101–125. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P. Compassion: From Its Evolution to a Psychotherapy. Front. Psychol. 2020, 11, 3123. [Google Scholar] [CrossRef] [PubMed]
- Ricard, M. Altruism: The Power of Compassion to Change Yourself and the World; Little, Brown: Boston, MA, USA, 2015; Available online: https://scholar.google.com/scholar_lookup?author=M.+Ricard&publication_year=2015&journal=Altruism:+The+power+of+compassion+to+change+itself+and+the+world (accessed on 14 October 2022).
- Seppala, E.M.; Hutcherson, C.A.; Nguyen, D.T.; Doty, J.R.; Gross, J.J. Loving-kindness meditation: A tool to improve healthcare provider compassion, resilience, and patient care. J. Compassionate Health Care 2014, 1, 5. [Google Scholar] [CrossRef]
- Batson, C.D. Empathy-induced altruistic motivation. In Prosocial Motives, Emotions, and Behavior: The Better Angels of Our Nature; Mikulincer, M., Shaver, P.R., Eds.; American Psychological Association: Washington, DC, USA, 2010; pp. 15–34. [Google Scholar] [CrossRef]
- Brown, S.L.; Brown, R.M.; Penner, L.A. Moving Beyond Self-Interest: Perspectives from Evolutionary Biology, Neuroscience, and the Social Sciences; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Carter, C.S.; Bartal, I.B.-A.; Porges, E.C. The Roots of Compassion. In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017; Available online: https://www.oxfordhandbooks.com/view/10.1093/oxfordhb/9780190464684.001.0001/oxfordhb-9780190464684-e-14 (accessed on 9 February 2021).
- de Waal, F.B.M. Putting the Altruism Back into Altruism: The Evolution of Empathy. Annu. Rev. Psychol. 2008, 59, 279–300. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.L. Is altruism part of human nature? J. Pers. Soc. Psychol. 1981, 40, 121. [Google Scholar] [CrossRef]
- Gordon, I.; Zagoory-Sharon, O.; Leckman, J.F.; Feldman, R. Oxytocin and the Development of Parenting in Humans. Biol. Psychiatry 2010, 68, 377–382. [Google Scholar] [CrossRef]
- Brown, S.L.; Brown, R.M. Compassionate neurobiology and health. In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017; Volume 10, pp. 159–172. [Google Scholar]
- Marsh, A.A. The Caring Continuum: Evolved Hormonal and Proximal Mechanisms Explain Prosocial and Antisocial Extremes. Annu. Rev. Psychol. 2019, 70, 347–371. [Google Scholar] [CrossRef]
- Domes, G.; Ower, N.; von Dawans, B.; Spengler, F.B.; Dziobek, I.; Bohus, M.; Matthies, S.; Philipsen, A.; Heinrichs, M. Effects of intranasal oxytocin administration on empathy and approach motivation in women with borderline personality disorder: A randomized controlled trial. Transl. Psychiatry 2019, 9, 1. [Google Scholar] [CrossRef]
- Geng, Y.; Zhao, W.; Zhou, F.; Ma, X.; Yao, S.; Hurlemann, R.; Becker, B.; Kendrick, K.M. Oxytocin Enhancement of Emotional Empathy: Generalization Across Cultures and Effects on Amygdala Activity. Front. Neurosci. 2018, 12, 512. [Google Scholar] [CrossRef]
- Hurlemann, R.; Patin, A.; Onur, O.A.; Cohen, M.X.; Baumgartner, T.; Metzler, S.; Dziobek, I.; Gallinat, J.; Wagner, M.; Kendrick, K.M.; et al. Oxytocin Enhances Amygdala-Dependent, Socially Reinforced Learning and Emotional Empathy in Humans. J. Neurosci. 2010, 30, 4999–5007. [Google Scholar] [CrossRef] [PubMed]
- Le, J.; Kou, J.; Zhao, W.; Fu, M.; Zhang, Y.; Becker, B.; Kendrick, K.M. Oxytocin Facilitation of Emotional Empathy Is Associated With Increased Eye Gaze Toward the Faces of Individuals in Emotional Contexts. Front. Neurosci. 2020, 14, 803. [Google Scholar] [CrossRef]
- Palgi, S.; Klein, E.; Shamay-Tsoory, S.G. Intranasal administration of oxytocin increases compassion toward women. Soc. Cogn. Affect. Neurosci. 2015, 10, 311–317. [Google Scholar] [CrossRef]
- Palgi, S.; Klein, E.; Shamay-Tsoory, S.G. Oxytocin improves compassion toward women among patients with PTSD. Psychoneuroendocrinology 2016, 64, 143–149. [Google Scholar] [CrossRef]
- Krueger, F.; Parasuraman, R.; Moody, L.; Twieg, P.; de Visser, E.; McCabe, K.; O’Hara, M.; Lee, M.R. Oxytocin selectively increases perceptions of harm for victims but not the desire to punish offenders of criminal offenses. Soc. Cogn. Affect. Neurosci. 2013, 8, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Barraza, J.A.; Zak, P.J. Empathy toward Strangers Triggers Oxytocin Release and Subsequent Generosity. Ann. N. Y. Acad. Sci. 2009, 1167, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Procyshyn, T.L.; Watson, N.V.; Crespi, B.J. Experimental empathy induction promotes oxytocin increases and testosterone decreases. Horm. Behav. 2020, 117, 104607. [Google Scholar] [CrossRef]
- Speck, L.G.; Schöner, J.; Bermpohl, F.; Heinz, A.; Gallinat, J.; Majić, T.; Montag, C. Endogenous oxytocin response to film scenes of attachment and loss is pronounced in schizophrenia. Soc. Cogn. Affect. Neurosci. 2019, 14, 109–117. [Google Scholar] [CrossRef]
- Bellosta-Batalla, M.; Blanco-Gandía, M.C.; Rodríguez-Arias, M.; Cebolla, A.; Pérez-Blasco, J.; Moya-Albiol, L. Increased Salivary Oxytocin and Empathy in Students of Clinical and Health Psychology After a Mindfulness and Compassion-Based Intervention. Mindfulness 2020, 11, 1006–1017. [Google Scholar] [CrossRef]
- Christ, C.C.; Carlo, G.; Stoltenberg, S.F. Oxytocin Receptor (OXTR) Single Nucleotide Polymorphisms Indirectly Predict Prosocial Behavior Through Perspective Taking and Empathic Concern. J. Pers. 2016, 84, 204–213. [Google Scholar] [CrossRef]
- Gong, P.; Fan, H.; Liu, J.; Yang, X.; Zhang, K.; Zhou, X. Revisiting the impact of OXTR rs53576 on empathy: A population-based study and a meta-analysis. Psychoneuroendocrinology 2017, 80, 131–136. [Google Scholar] [CrossRef]
- Kogan, A.; Saslow, L.R.; Impett, E.A.; Oveis, C.; Keltner, D.; Saturn, S.R. Thin-slicing study of the oxytocin receptor (OXTR) gene and the evaluation and expression of the prosocial disposition. Proc. Natl. Acad. Sci. USA 2011, 108, 19189–19192. [Google Scholar] [CrossRef]
- Rodrigues, S.M.; Saslow, L.R.; Garcia, N.; John, O.P.; Keltner, D. Oxytocin receptor genetic variation relates to empathy and stress reactivity in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 21437–21441. [Google Scholar] [CrossRef]
- Wu, N.; Li, Z.; Su, Y. The association between oxytocin receptor gene polymorphism (OXTR) and trait empathy. J. Affect. Disord. 2012, 138, 468–472. [Google Scholar] [CrossRef]
- Swain, J.E.; Kim, P.; Spicer, J.; Ho, S.S.; Dayton, C.J.; Elmadih, A.; Abel, K.M. Approaching the biology of human parental attachment: Brain imaging, oxytocin and coordinated assessments of mothers and fathers. Brain Res. 2014, 1580, 78–101. [Google Scholar] [CrossRef]
- Atzil, S.; Hendler, T.; Feldman, R. Specifying the Neurobiological Basis of Human Attachment: Brain, Hormones, and Behavior in Synchronous and Intrusive Mothers. Neuropsychopharmacology 2011, 1, 13. [Google Scholar] [CrossRef]
- Carter, C.S. Oxytocin Pathways and the Evolution of Human Behavior. Annu. Rev. Psychol. 2014, 65, 10–11. [Google Scholar] [CrossRef]
- Feldman, R. The Neurobiology of Human Attachments. Trends Cogn. Sci. 2017, 21, 80–99. [Google Scholar] [CrossRef]
- Shamay-Tsoory, S.G.; Abu-Akel, A. The Social Salience Hypothesis of Oxytocin. Biol. Psychiatry 2016, 79, 194–202. [Google Scholar] [CrossRef]
- Stallen, M.; De Dreu, C.K.W.; Shalvi, S.; Smidts, A.; Sanfey, A.G. The Herding Hormone: Oxytocin Stimulates In-Group Conformity. Psychol. Sci. 2012, 23, 1288–1292. [Google Scholar] [CrossRef]
- Kosfeld, M.; Heinrichs, M.; Zak, P.J.; Fischbacher, U.; Fehr, E. Oxytocin Increases Trust in Humans. Nature 2005, 435, 673–676. [Google Scholar] [CrossRef]
- Ne’eman, R.; Perach-Barzilay, N.; Fischer-Shofty, M.; Atias, A.; Shamay-Tsoory, S.G. Intranasal administration of oxytocin increases human aggressive behavior. Horm. Behav. 2016, 80, 125–131. [Google Scholar] [CrossRef]
- De Dreu, C.K.W. Oxytocin modulates cooperation within and competition between groups: An integrative review and research agenda. Horm. Behav. 2012, 61, 419–428. [Google Scholar] [CrossRef]
- De Dreu, C.K.W.; Greer, L.L.; Kleef, G.A.V.; Shalvi, S.; Handgraaf, M.J.J. Oxytocin Promotes Human Ethnocentrism. Proc. Natl. Acad. Sci. USA 2011, 108, 1262–1266. [Google Scholar] [CrossRef]
- De Dreu, C.K.W.; Greer, L.L.; Handgraaf, M.J.J.; Shalvi, S.; Van Kleef, G.A.; Baas, M.; Ten Velden, F.S.; Van Dijk, E.; Feith, S.W.W. The Neuropeptide Oxytocin Regulates Parochial Altruism in Intergroup Conflict Among Humans. Science 2010, 328, 1408–1411. [Google Scholar] [CrossRef]
- Shamay-Tsoory, S.G.; Fischer, M.; Dvash, J.; Harari, H.; Perach-Bloom, N.; Levkovitz, Y. Intranasal Administration of Oxytocin Increases Envy and Schadenfreude (Gloating). Biol. Psychiatry 2009, 66, 864–870. [Google Scholar] [CrossRef]
- Declerck, C.H.; Boone, C.; Pauwels, L.; Vogt, B.; Fehr, E. A Registered Replication Study on Oxytocin and Trust. Nat. Hum. Behav. 2020, 4, 646–655. [Google Scholar] [CrossRef]
- Lane, A.; Mikolajczak, M.; Treinen, E.; Samson, D.; Corneille, O.; de Timary, P.; Luminet, O. Failed Replication of Oxytocin Effects on Trust: The Envelope Task Case. PLoS ONE 2015, 10, e0137000. [Google Scholar] [CrossRef]
- Bartz, J.A.; Zaki, J.; Bolger, N.; Ochsner, K.N. Social effects of oxytocin in humans: Context and person matter. Trends Cogn. Sci. 2011, 15, 301–309. [Google Scholar] [CrossRef]
- Harari-Dahan, O.; Bernstein, A. A general approach-avoidance hypothesis of Oxytocin: Accounting for social and non-social effects of oxytocin. Neurosci. Biobehav. Rev. 2014, 47, 506–519. [Google Scholar] [CrossRef]
- Quintana, D.S.; Guastella, A.J. An Allostatic Theory of Oxytocin. Trends Cogn. Sci. 2020, 24, 515–528. [Google Scholar] [CrossRef]
- Mierop, A.; Mikolajczak, M.; Stahl, C.; Béna, J.; Luminet, O.; Lane, A.; Corneille, O. How can intranasal oxytocin research be trusted? A systematic review of the interactive effects of intranasal oxytocin on psychosocial outcomes. Perspect. Psychol. Sci. 2020, 15, 1228–1242. [Google Scholar] [CrossRef]
- Grillon, C.; Krimsky, M.; Charney, D.R.; Vytal, K.; Ernst, M.; Cornwell, B. Oxytocin increases anxiety to unpredictable threat. Mol. Psychiatry 2013, 18, 9. [Google Scholar] [CrossRef]
- Striepens, N.; Scheele, D.; Kendrick, K.M.; Becker, B.; Schäfer, L.; Schwalba, K.; Reul, J.; Maier, W.; Hurlemann, R. Oxytocin facilitates protective responses to aversive social stimuli in males. Proc. Natl. Acad. Sci. USA 2012, 109, 18144–18149. [Google Scholar] [CrossRef]
- Tracy, L.M.; Georgiou-Karistianis, N.; Gibson, S.J.; Giummarra, M.J. Oxytocin and the modulation of pain experience: Implications for chronic pain management. Neurosci. Biobehav. Rev. 2015, 55, 53–67. [Google Scholar] [CrossRef]
- Eckstein, M.; Bamert, V.; Stephens, S.; Wallen, K.; Young, L.J.; Ehlert, U.; Ditzen, B. Oxytocin increases eye-gaze towards novel social and non-social stimuli. Soc. Neurosci. 2019, 14, 594–607. [Google Scholar] [CrossRef]
- Marlin, B.J.; Mitre, M.; D’amour, J.A.; Chao, M.V.; Froemke, R.C. Oxytocin Enables Maternal Behavior by Balancing Cortical Inhibition. Nature 2015, 520, 499–504. [Google Scholar] [CrossRef]
- Pfundmair, M.; Zwarg, C.; Paulus, M.; Rimpel, A. Oxytocin promotes attention to social cues regardless of group membership. Horm. Behav. 2017, 90, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Putnam, P.T.; Roman, J.M.; Zimmerman, P.E.; Gothard, K.M. Oxytocin enhances gaze-following responses to videos of natural social behavior in adult male rhesus monkeys. Psychoneuroendocrinology 2016, 72, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ma, X.; Zhao, W.; Luo, L.; Yao, S.; Kendrick, K.M. Oxytocin enhances attentional bias for neutral and positive expression faces in individuals with higher autistic traits. Psychoneuroendocrinology 2015, 62, 352–358. [Google Scholar] [CrossRef]
- Xu, X.; Li, J.; Chen, Z.; Kendrick, K.M.; Becker, B. Oxytocin reduces top-down control of attention by increasing bottom-up attention allocation to social but not non-social stimuli—A randomized controlled trial. Psychoneuroendocrinology 2019, 108, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Auyeung, B.; Lombardo, M.V.; Heinrichs, M.; Chakrabarti, B.; Sule, A.; Deakin, J.B.; Bethlehem, R.; Dickens, L.; Mooney, N.; Baron-Cohen, S.; et al. Oxytocin increases eye contact during a real-time, naturalistic social interaction in males with and without autism. Transl. Psychiatry 2015, 5, e507. [Google Scholar] [CrossRef]
- Bradley, E.R.; Seitz, A.; Niles, A.N.; Rankin, K.P.; Mathalon, D.H.; O’Donovan, A.; Woolley, J.D. Oxytocin increases eye gaze in schizophrenia. Schizophr. Res. 2019, 212, 177–185. [Google Scholar] [CrossRef]
- Domes, G.; Steiner, A.; Porges, S.W.; Heinrichs, M. Oxytocin differentially modulates eye gaze to naturalistic social signals of happiness and anger. Psychoneuroendocrinology 2013, 38, 1198–1202. [Google Scholar] [CrossRef] [PubMed]
- Guastella, A.J.; Mitchell, P.B.; Dadds, M.R. Oxytocin Increases Gaze to the Eye Region of Human Faces. Biol. Psychiatry 2008, 63, 3–5. [Google Scholar] [CrossRef]
- Nishizato, M.; Fujisawa, T.X.; Kosaka, H.; Tomoda, A. Developmental changes in social attention and oxytocin levels in infants and children. Sci. Rep. 2017, 7, 1. [Google Scholar] [CrossRef]
- Gamer, M.; Zurowski, B.; Büchel, C. Different amygdala subregions mediate valence-related and attentional effects of oxytocin in humans. Proc. Natl. Acad. Sci. USA 2010, 107, 9400–9405. [Google Scholar] [CrossRef] [PubMed]
- Le, J.; Kou, J.; Zhao, W.; Fu, M.; Zhang, Y.; Becker, B.; Kendrick, K.M. Oxytocin biases eye-gaze to dynamic and static social images and the eyes of fearful faces: Associations with trait autism. Transl. Psychiatry 2020, 10, 1. [Google Scholar] [CrossRef]
- Domes, G.; Lischke, A.; Berger, C.; Grossmann, A.; Hauenstein, K.; Heinrichs, M.; Herpertz, S.C. Effects of intranasal oxytocin on emotional face processing in women. Psychoneuroendocrinology 2010, 35, 83–93. [Google Scholar] [CrossRef]
- Hubble, K.; Daughters, K.; Manstead, A.S.R.; Rees, A.; Thapar, A.; van Goozen, S.H.M. Oxytocin Reduces Face Processing Time but Leaves Recognition Accuracy and Eye-Gaze Unaffected. J. Int. Neuropsychol. Soc. JINS 2017, 23, 23–33. [Google Scholar] [CrossRef]
- Lischke, A.; Berger, C.; Prehn, K.; Heinrichs, M.; Herpertz, S.C.; Domes, G. Intranasal oxytocin enhances emotion recognition from dynamic facial expressions and leaves eye-gaze unaffected. Psychoneuroendocrinology 2012, 37, 475. [Google Scholar] [CrossRef]
- Chen, J.; Luo, Y.L.L.; Xie, Y.; Yang, Z.; Cai, H. Oxytocin intensifies the mortality salience effect: Novel evidence for the social salience model of oxytocin. Horm. Behav. 2021, 129, 104920. [Google Scholar] [CrossRef]
- Goetz, J.L.; Simon-Thomas, E. The landscape of compassion: Definitions and scientific approaches. In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017. [Google Scholar]
- Collins, N.L.; Feeney, B.C. A safe haven: An attachment theory perspective on support seeking and caregiving in intimate relationships. J. Pers. Soc. Psychol. 2000, 78, 1053–1073. [Google Scholar] [CrossRef]
- Hammer, J.L.; Marsh, A.A. Why do fearful facial expressions elicit behavioral approach? Evidence from a combined approach-avoidance implicit association test. Emotion 2015, 15, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Marlin, B.J.; Froemke, R.C. Oxytocin modulation of neural circuits for social behavior: Oxytocin Modulation of Neural Circuits for Social Behavior. Dev. Neurobiol. 2017, 77, 169–189. [Google Scholar] [CrossRef]
- Batson, C.D. The empathy altruism hypothesis what and so what? In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017; pp. 27–40. [Google Scholar]
- Gilbert, P. The Evolution and Social Dynamics of Compassion. Soc. Personal. Psychol. Compass 2015, 9, 239–254. [Google Scholar] [CrossRef]
- Halifax, J. A heuristic model of enactive compassion. Curr. Opin. Support. Palliat. Care 2012, 6, 228–235. [Google Scholar] [CrossRef]
- Weng, H.Y.; Lapate, R.C.; Stodola, D.E.; Rogers, G.M.; Davidson, R.J. Visual Attention to Suffering After Compassion Training Is Associated With Decreased Amygdala Responses. Front. Psychol. 2018, 9, 771. [Google Scholar] [CrossRef]
- Shahrestani, S.; Kemp, A.H.; Guastella, A.J. The Impact of a Single Administration of Intranasal Oxytocin on the Recognition of Basic Emotions in Humans: A Meta-Analysis. Neuropsychopharmacology 2013, 38, 1929–1936. [Google Scholar] [CrossRef]
- Van IJzendoorn, M.H.; Bakermans-Kranenburg, M.J. A sniff of trust: Meta-analysis of the effects of intranasal oxytocin administration on face recognition, trust to in-group, and trust to out-group. Psychoneuroendocrinology 2012, 37, 438–443. [Google Scholar] [CrossRef]
- Voorthuis, A.; Riem, M.M.E.; Van IJzendoorn, M.H.; Bakermans-Kranenburg, M.J. Reading the mind in the infant eyes: Paradoxical effects of oxytocin on neural activity and emotion recognition in watching pictures of infant faces. Brain Res. 2014, 1580, 151–159. [Google Scholar] [CrossRef]
- Leppanen, J.; Ng, K.W.; Kim, Y.-R.; Tchanturia, K.; Treasure, J. Meta-analytic review of the effects of a single dose of intranasal oxytocin on threat processing in humans. J. Affect. Disord. 2018, 225, 167–179. [Google Scholar] [CrossRef]
- Bartz, J.A.; Zaki, J.; Bolger, N.; Hollander, E.; Ludwig, N.N.; Kolevzon, A.; Ochsner, K.N. Oxytocin Selectively Improves Empathic Accuracy. Psychol. Sci. 2010, 21, 1426–1428. [Google Scholar] [CrossRef]
- Berridge, K.C. From prediction error to incentive salience: Mesolimbic computation of reward motivation: From prediction error to incentive salience. Eur. J. Neurosci. 2012, 35, 1124–1143. [Google Scholar] [CrossRef]
- Salamone, J.D.; Correa, M. The Mysterious Motivational Functions of Mesolimbic Dopamine. Neuron 2012, 76, 470–485. [Google Scholar] [CrossRef]
- Walum, H.; Young, L.J. The neural mechanisms and circuitry of the pair bond. Nat. Rev. Neurosci. 2018, 19, 643–654. [Google Scholar] [CrossRef]
- Love, T.M. Oxytocin, motivation and the role of dopamine. Pharmacol. Biochem. Behav. 2014, 119, 49–60. [Google Scholar] [CrossRef]
- Groppe, S.E.; Gossen, A.; Rademacher, L.; Hahn, A.; Westphal, L.; Gründer, G.; Spreckelmeyer, K.N. Oxytocin influences processing of socially relevant cues in the ventral tegmental area of the human brain. Biol. Psychiatry 2013, 74, 172–179. [Google Scholar] [CrossRef]
- Borland, J.M.; Grantham, K.N.; Aiani, L.M.; Frantz, K.J.; Albers, H.E. Role of oxytocin in the ventral tegmental area in social reinforcement. Psychoneuroendocrinology 2018, 95, 128–137. [Google Scholar] [CrossRef]
- Gunaydin, L.A.; Grosenick, L.; Finkelstein, J.C.; Kauvar, I.V.; Fenno, L.E.; Adhikari, A.; Lammel, S.; Mirzabekov, J.J.; Airan, R.D.; Deisseroth, K.; et al. Natural neural projection dynamics underlying social behavior. Cell 2014, 157, 1535–1551. [Google Scholar] [CrossRef]
- Hung, L.W.; Neuner, S.; Polepalli, J.S.; Beier, K.T.; Wright, M.; Walsh, J.J.; Lewis, E.M.; Luo, L.; Deisseroth, K.; Malenka, R.C. Gating of social reward by oxytocin in the ventral tegmental area. Science 2017, 357, 1406–1411. [Google Scholar] [CrossRef]
- Williams, A.V.; Duque-Wilckens, N.; Ramos-Maciel, S.; Campi, K.L.; Bhela, S.K.; Xu, C.K.; Jackson, K.; Chini, B.; Pesavento, P.A.; Trainor, B.C. Social approach and social vigilance are differentially regulated by oxytocin receptors in the nucleus accumbens. Neuropsychopharmacology 2020, 45, 1423–1430. [Google Scholar] [CrossRef]
- Preston, S.D. The rewarding nature of social contact. Science 2017, 357, 1353–1354. [Google Scholar] [CrossRef]
- Steinman, M.Q.; Duque-Wilckens, N.; Trainor, B.C. Complementary Neural Circuits for Divergent Effects of Oxytocin: Social Approach Versus Social Anxiety. Biol. Psychiatry 2019, 85, 792–801. [Google Scholar] [CrossRef]
- Ashar, Y.K.; Andrews-Hanna, J.R.; Dimidjian, S.; Wager, T.D. Empathic Care and Distress: Predictive Brain Markers and Dissociable Brain Systems. Neuron 2017, 94, 1263–1273.e4. [Google Scholar] [CrossRef]
- Engen, H.G.; Singer, T. Compassion-based emotion regulation up-regulates experienced positive affect and associated neural networks. Soc. Cogn. Affect. Neurosci. 2015, 10, 1291–1301. [Google Scholar] [CrossRef]
- Hare, T.A.; Camerer, C.F.; Knoepfle, D.T.; O’Doherty, J.P.; Rangel, A. Value Computations in Ventral Medial Prefrontal Cortex during Charitable Decision Making Incorporate Input from Regions Involved in Social Cognition. J. Neurosci. 2010, 30, 583–590. [Google Scholar] [CrossRef]
- Zaki, J.; Schirmer, J.; Mitchell, J.P. Social Influence Modulates the Neural Computation of Value. Psychol. Sci. 2011, 22, 894–900. [Google Scholar] [CrossRef]
- Singer, T.; Klimecki, O.M. Empathy and compassion. Curr. Biol. 2014, 24, R875–R878. [Google Scholar] [CrossRef] [PubMed]
- Marsh, N.; Marsh, A.A.; Lee, M.R.; Hurlemann, R. Oxytocin and the Neurobiology of Prosocial Behavior. Neuroscientist 2020, 27, 604–619. [Google Scholar] [CrossRef] [PubMed]
- Ross, H.E.; Young, L.J. Oxytocin and the neural mechanisms regulating social cognition and affiliative behavior. Front. Neuroendocrinol. 2009, 30, 534–547. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Mascaro, J. Compassion: Fears, blocks, and resistances: An evolutionary investigation. In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017; pp. 399–420. [Google Scholar]
- Olff, M.; Frijling, J.L.; Kubzansky, L.D.; Bradley, B.; Ellenbogen, M.A.; Cardoso, C.; Bartz, J.F.; Yee, J.R.; Van Zuiden, M. The role of oxytocin in social bonding, stress regulation and mental health: An update on the moderating effects of context and interindividual differences. Psychoneuroendocrinology 2013, 38, 1883–1894. [Google Scholar] [CrossRef]
- Peled-Avron, L.; Abu-Akel, A.; Shamay-Tsoory, S. Exogenous effects of oxytocin in five psychiatric disorders: A systematic review, meta-analyses and a personalized approach through the lens of the social salience hypothesis. Neurosci. Biobehav. Rev. 2020, 114, 70–95. [Google Scholar] [CrossRef] [PubMed]
- Zaki, J.; Cikara, M. Addressing Empathic Failures. Science 2015, 1, 6. [Google Scholar] [CrossRef]
- Tops, M.; Van Peer, J.M.; Korf, J.; Wijers, A.A.; Tucker, D.M. Anxiety, cortisol, and attachment predict plasma oxytocin. Psychophysiology 2007, 44, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Strathearn, L.; Fonagy, P.; Amico, J.; Montague, P.R. Adult Attachment Predicts Maternal Brain and Oxytocin Response to Infant Cues. Neuropsychopharmacology 2009, 34, 13. [Google Scholar] [CrossRef] [PubMed]
- Mikulincer, M.; Shaver, P.R. Attachment Security, Compassion, and Altruism. Curr. Dir. Psychol. Sci. 2005, 14, 34–38. [Google Scholar] [CrossRef]
- van Kleef, G.A.; Oveis, C.; van der Löwe, I.; LuoKogan, A.; Goetz, J.; Keltner, D. Power, Distress, and Compassion: Turning a Blind Eye to the Suffering of Others. Psychol. Sci. 2008, 19, 1315–1322. [Google Scholar] [CrossRef] [PubMed]
- Dimitroff, S.J.; Kardan, O.; Necka, E.A.; Decety, J.; Berman, M.G.; Norman, G.J. Physiological dynamics of stress contagion. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R. Oxytocin and social affiliation in humans. Horm. Behav. 2012, 61, 380–391. [Google Scholar] [CrossRef]
- Mikulincer, M.; Shaver, P.R. Adult attachment and compassion: Normative and individual difference components. In The Oxford Handbook of Compassion Science; Oxford University Press: New York, NY, USA, 2017; pp. 79–90. [Google Scholar]
- Bowlby, J. Attachment and loss: Retrospect and prospect. Am. J. Orthopsychiatry 1982, 52, 664–678. [Google Scholar] [CrossRef] [PubMed]
- Sbarra, D.A.; Hazan, C. Coregulation, dysregulation, self-regulation: An integrative analysis and empirical agenda for understanding adult attachment, separation, loss, and recovery. Personal. Soc. Psychol. Rev. 2008, 12, 141–167. [Google Scholar] [CrossRef]
- Joireman, J.; Needham, T.; Cummings, A.-L. Relationships between dimensions of attachment and empathy. North Am. J. Psychol. 2002, 4, 63–80. [Google Scholar]
- Lopez, F.G.; Mauricio, A.M.; Gormley, B.; Simko, T.; Berger, E. Adult Attachment Orientations and College Student Distress: The Mediating Role of Problem Coping Styles. J. Couns. Dev. 2001, 79, 459–464. [Google Scholar] [CrossRef]
- Shaver, P.R.; Mikulincer, M.; Shemesh-Iron, M. A behavioral-systems perspective on prosocial behavior. In Prosocial Motives, Emotions, and Behavior: The Better Angels of Our Nature; American Psychological Association: Washington, DC, USA, 2010; pp. 73–91. [Google Scholar] [CrossRef]
- Wayment, H.A. Attachment style, empathy, and helping following a collective loss: Evidence from the September 11 terrorist attacks. Attach. Hum. Dev. 2006, 8, 1–9. [Google Scholar] [CrossRef]
- Feldman, R.; Bakermans-Kranenburg, M.J. Oxytocin: A parenting hormone. Curr. Opin. Psychol. 2017, 15, 13–18. [Google Scholar] [CrossRef]
- Apter-Levi, Y.; Zagoory-Sharon, O.; Feldman, R. Oxytocin and vasopressin support distinct configurations of social synchrony. Brain Res. 2014, 1580, 124–132. [Google Scholar] [CrossRef]
- Feldman, R.; Zagoory-Sharon, O.; Weisman, O.; Schneiderman, I.; Gordon, I.; Maoz, R.; Shalev, I.; Ebstein, R.P. Sensitive Parenting Is Associated with Plasma Oxytocin and Polymorphisms in the OXTR and CD38 Genes. Biol. Psychiatry 2012, 72, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Collins, N.L.; Ford, M.; Guichard, A.; Kane, H.; Feeney, B. Responding to need in intimate relationships: Social support and caregiving processes in couples. In Prosocial Motives, Emotions, and Behavior: The Better Angels of Our Nature; American Psychological Association: Washington, DC, USA, 2010. [Google Scholar] [CrossRef]
- Collins, N.L.; Ford, M.B. Responding to the needs of others: The caregiving behavioral system in intimate relationships. J. Soc. Pers. Relatsh. 2010, 27, 235–244. [Google Scholar] [CrossRef]
- Feeney, B.C.; Cassidy, J.; Ramos-Marcuse, F. The generalization of attachment representations to new social situations: Predicting behavior during initial interactions with strangers. J. Pers. Soc. Psychol. 2008, 95, 1481. [Google Scholar] [CrossRef] [PubMed]
- Mikulincer, M.; Gillath, O.; Halevy, V.; Avihou, N.; Avidan, S.; Eshkoli, N. Attachment theory and rections to others’ needs: Evidence that activiation of the sense of attachment security promotes empathic responses. J. Pers. Soc. Psychol. 2001, 81, 1205. [Google Scholar] [CrossRef]
- Mikulincer, M.; Gillath, O.; Sapir-Lavid, Y.; Yaakobi, E.; Arias, K.; Tal-Aloni, L.; Bor, G. Attachment theory and concern for others’ welfare: Evidence that activation of the sense of secure base promotes endorsement of self-transcendence values. Basic Appl. Soc. Psychol. 2003, 25, 299–312. [Google Scholar] [CrossRef]
- Westmaas, J.L.; Silver, R.C. The role of attachment in responses to victims of life crises. J. Pers. Soc. Psychol. 2001, 80, 425–438. [Google Scholar] [CrossRef]
- Gordon, I.; Zagoory-Sharon, O.; Schneiderman, I.; Leckman, J.F.; Weller, A.; Feldman, R. Oxytocin and cortisol in romantically unattached young adults: Associations with bonding and psychological distress. Psychophysiology 2008, 45, 349–352. [Google Scholar] [CrossRef]
- Gordon, I.; Martin, C.; Feldman, R.; Leckman, J.F. Oxytocin and social motivation. Dev. Cogn. Neurosci. 2011, 1, 471–493. [Google Scholar] [CrossRef]
- Young, L.J.; Lim, M.M.; Gingrich, B.; Insel, T.R. Cellular mechanisms of social attachment. Horm. Behav. 2001, 40, 133–138. [Google Scholar] [CrossRef]
- Macdonald, K.S. Sex, Receptors, and Attachment: A Review of Individual Factors Influencing Response to Oxytocin. Front. Neurosci. 2013, 6, 194. [Google Scholar] [CrossRef] [PubMed]
- Bakermans-Kranenburg, M.J.; van IJzendoorn, M.H. Oxytocin receptor (OXTR) and serotonin transporter (5-HTT) genes associated with observed parenting. Soc. Cogn. Affect. Neurosci. 2008, 3, 128–134. [Google Scholar] [CrossRef]
- Buchheim, A.; Heinrichs, M.; George, C.; Pokorny, D.; Koops, E.; Henningsen, P.; O’Connor, M.-F.; Gündel, H. Oxytocin enhances the experience of attachment security. Psychoneuroendocrinology 2009, 34, 1417–1422. [Google Scholar] [CrossRef]
- Floyd, K.; Denes, A. Attachment Security and Oxytocin Receptor Gene Polymorphism Interact to Influence Affectionate Communication. Commun. Q. 2015, 63, 272–285. [Google Scholar] [CrossRef]
- Bartz, J.A.; Lydon, J.E.; Kolevzon, A.; Zaki, J.; Hollander, E.; Ludwig, N.; Bolger, N. Differential effects of oxytocin on agency and communion for anxiously and avoidantly attached individuals. Psychol. Sci. 2015, 26, 1177–1186. [Google Scholar] [CrossRef]
- Bernaerts, S.; Prinsen, J.; Berra, E.; Bosmans, G.; Steyaert, J.; Alaerts, K. Long-term oxytocin administration enhances the experience of attachment. Psychoneuroendocrinology 2017, 78, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Landau, D.H.; Friedman, D.; Hasler, B.S.; Levit-Binnun, N.; Golland, Y. Exposure to social suffering in virtual reality boosts compassion and facial synchrony. Comput. Hum. Behav. 2021, 122, 106781. [Google Scholar] [CrossRef]
- Dor-Ziderman, Y.; Cohen, D.; Levit-Binnun, N.; Golland, Y. Synchrony with distress in affective empathy and compassion. Psychophysiology 2021, 58, e13889. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.; Feldman, R. Synchronous Interactions Foster Empathy. J. Exp. Neurosci. 2019, 13, 1179069519865799. [Google Scholar] [CrossRef]
- Condon, P.; Desbordes, G.; Miller, W.B.; DeSteno, D. Meditation Increases Compassionate Responses to Suffering. Psychol. Sci. 2013, 24, 2125–2127. [Google Scholar] [CrossRef] [PubMed]
- Loewenstein, G.; Small, D.A. The Scarecrow and the Tin Man: The Vicissitudes of Human Sympathy and Caring. Rev. Gen. Psychol. 2007, 11, 112–126. [Google Scholar] [CrossRef]
- Luminet, O.; Grynberg, D.; Ruzette, N.; Mikolajczak, M. Personality-dependent effects of oxytocin: Greater social benefits for high alexithymia scorers. Biol. Psychol. 2011, 87, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Sheng, F.; Liu, Y.; Zhou, B.; Zhou, W.; Han, S. Oxytocin modulates the racial bias in neural responses to others’ suffering. Biol. Psychol. 2013, 92, 380–386. [Google Scholar] [CrossRef]
- Dahl, C.J.; Lutz, A.; Davidson, R.J. Cognitive Processes Are Central in Compassion Meditation. Trends Cogn. Sci. 2016, 20, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Bartz, J.A.; Zaki, J.; Ochsner, K.N.; Bolger, N.; Kolevzon, A.; Ludwig, N.; Lydon, J.E. Effects of oxytocin on recollections of maternal care and closeness. Proc. Natl. Acad. Sci. USA 2010, 107, 21371–21375. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kucerova, B.; Levit-Binnun, N.; Gordon, I.; Golland, Y. From Oxytocin to Compassion: The Saliency of Distress. Biology 2023, 12, 183. https://doi.org/10.3390/biology12020183
Kucerova B, Levit-Binnun N, Gordon I, Golland Y. From Oxytocin to Compassion: The Saliency of Distress. Biology. 2023; 12(2):183. https://doi.org/10.3390/biology12020183
Chicago/Turabian StyleKucerova, Barbora, Nava Levit-Binnun, Ilanit Gordon, and Yulia Golland. 2023. "From Oxytocin to Compassion: The Saliency of Distress" Biology 12, no. 2: 183. https://doi.org/10.3390/biology12020183
APA StyleKucerova, B., Levit-Binnun, N., Gordon, I., & Golland, Y. (2023). From Oxytocin to Compassion: The Saliency of Distress. Biology, 12(2), 183. https://doi.org/10.3390/biology12020183




