Microplastics in Different Tissues of a Commonly Consumed Fish, Scomberomorus guttatus, from a Large Subtropical Estuary: Accumulation, Characterization, and Contamination Assessment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fish Sample Collection and Preparation
2.3. Digestion of Fish Tissue and Separation of MPs
2.4. Microscopic Analysis and Polymer Identification of MPs
2.5. Contamination Assessment of MPs in Fish
2.6. Control of Contamination
2.7. Statistical Analysis
3. Results and Discussion
3.1. MPs Occurrence and Abundance in Fish
| Country | Study Region | Fish Species | No. of Fish | MPs/g DT | MPs/g Gill | MPs/g Muscle | MPs/ind. | References |
|---|---|---|---|---|---|---|---|---|
| Bangladesh | Meghna River estuary | S. guttatus | 10 | 0.84 ± 0.45 | 2.56 ± 0.73 | 2.3 ± 1.72 | 48.7 ± 20.27 | Present study |
| Karnafully River | Setipinna phasa | 30 | 8.29 ± 1.75 | - | - | 13.17 ± 0.76 | Hossain et al. [46] | |
| Polynemus Paradiseus | 30 | 5.44 ± 0.51 | - | - | 10.83 ± 0.81 | |||
| Otolithoides pama | 15 | 1.65 ± 0.19 | - | - | 5.93 ± 0.62 | |||
| Bay of Bengal | Priacanthus hamrur | 10 | 2.53 | - | - | 3.8 | Ghosh et al. [15] | |
| Setipinna tenuifilis | 10 | 6.45 | - | - | 3.2 | |||
| Sciades sona | 10 | 1.67 | - | - | 3 | |||
| Carangoides Chrysophrys | 10 | 2.5 | - | - | 2 | |||
| Sardinella brachysoma | 10 | 1.82 | - | - | 2 | |||
| Harpadon nehereus | 10 | 1 | - | - | 1.8 | |||
| Otolithoides pama | 10 | 1.2 | - | - | 1.8 | |||
| Coilia neglecta | 10 | 0.94 | - | - | 1.5 | |||
| Anodontostoma chacunda | 10 | 0.45 | - | - | 1.4 | |||
| Megalaspis cordyla | 10 | 0.63 | - | - | 1 | |||
| Bay of Bengal | Sardinella gibbose | 25 | 1.55 ± 0.48 | - | - | 3.20 ± 1.16 | Hossain et al. [18] | |
| Harpadon translucens | 25 | 1.10 ± 0.30 | - | - | 5.80 ± 1.41 | |||
| Harpadon nehereus | 25 | 0.37 ± 0.10 | - | - | 8.72 ± 1.54 | |||
| India | Gulf of Mannar coast | Sufflamen fraenatus | 20 | 0.22 | - | 0.18 | - | Selvam et al. [47] |
| Heniochus acuminatus | 25 | 0.10 | - | 0.06 | - | |||
| Pseudotriacanthus | 20 | 0.35 | - | 0.36 | - | |||
| Leiognathus brevirostris | 15 | 0.12 | - | 0.10 | - | |||
| Japan | West coast of Kyushu | Scomber japonicus | 40 | 38 | - | - | 0.95 | Yagi et al. [43] |
| Trichiurus japonicus | 38 | 17 | - | - | 0.45 | |||
| D. tumifrons | 15 | 1 | - | - | 0.07 | |||
| Z. faber | 9 | 3 | - | - | 0.33 | |||
| M. scolopax | 39 | 3 | - | - | 0.08 | |||
| C. equula | 73 | 11 | - | - | 0.15 |
3.2. Morphological Characteristics of Fish MPs
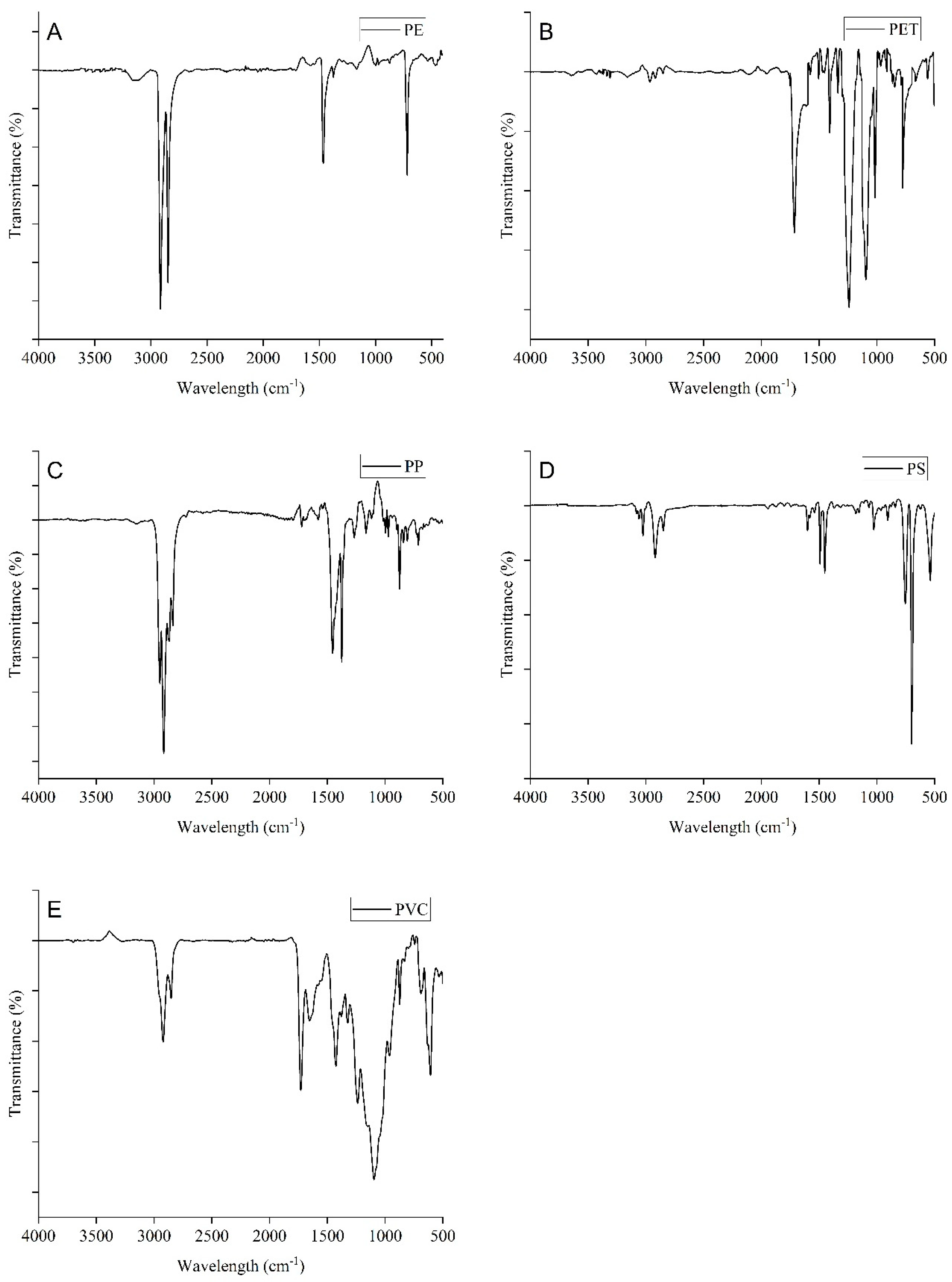
3.3. Polymer Characteristics of MPs
3.4. Contamination Level Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frias, J.P.G.L.; Nash, R. Microplastics: Finding a consensus on the definition. Mar. Pollut. Bull. 2019, 138, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Lusher, A.L.; Hernandez-Milian, G.; O’Brien, J.; Berrow, S.; O’Connor, I.; Officer, R. Microplastic and macroplastic ingestion by a deep diving, oceanic cetacean: The True’s beaked whale Mesoplodon mirus. Environ. Pollut. 2015, 199, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Zhao, H.; Sun, H.; Sun, Y.; Zhao, J.; Xia, T. Investigation of microplastics in sludge from five wastewater treatment plants in Nanjing, China. J. Environ. Manag. 2022, 301, 113793. [Google Scholar] [CrossRef] [PubMed]
- Banik, P.; Hossain, M.B.; Nur, A.A.U.; Choudhury, T.R.; Liba, S.I.; Yu, J.; Noman, M.A.; Sun, J. Microplastics in Sediment of Kuakata Beach, Bangladesh: Occurrence, Spatial Distribution, and Risk Assessment. Front. Mar. Sci. 2022, 9, 348. [Google Scholar] [CrossRef]
- Rochman, C.M.; Brookson, C.; Bikker, J.; Djuric, N.; Earn, A.; Bucci, K.; Athey, S.; Huntington, A.; McIlwraith, H.; Munno, K.; et al. Rethinking microplastics as a diverse contaminant suite. Environ. Toxicol. Chem. 2019, 38, 703–711. [Google Scholar] [CrossRef]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.Q. Heavy metals and pesticides toxicity in agricultural soil and plants: Ecological risks and human health implication. Toxics 2021, 9, 42. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Bakir, A.; Burton, G.A.; Janssen, C.R. Microplastic as a Vector for Chemicals in the Aquatic Environment: Critical Review and Model-Supported Reinterpretation of Empirical Studies. Environ. Sci. Technol. 2016, 50, 3315–3326. [Google Scholar] [CrossRef]
- Lusher, A.L.; Welden, N.A.; Sobral, P.; Cole, M. Sampling, Isolating and Identifying Microplastics Ingested by Fish and Invertebrates. In Analysis of Nanoplastics and Microplastics in Food; CRC Press: Boca Raton, FL, USA, 2020; pp. 119–148. [Google Scholar] [CrossRef]
- Wang, F.; Yu, Y.; Wu, H.; Wu, W.; Wang, L.; An, L.; Cai, W. Microplastics in spotted seal cubs (Phoca largha): Digestion after ingestion? Sci. Total Environ. 2021, 785, 147426. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, X.; Xu, J.; Zhu, L.; Peng, G.; Xu, P.; Li, D. Food-web transfer of microplastics between wild caught fish and crustaceans in East China Sea. Mar. Pollut. Bull. 2019, 146, 173–182. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.K.; Fileman, E.; Clark, J.; Lewis, C.; Halsband, C.; Galloway, T.S. Microplastics Alter the Properties and Sinking Rates of Zooplankton Faecal Pellets. Environ. Sci. Technol. 2016, 50, 3239–3246. [Google Scholar] [CrossRef] [PubMed]
- Kutralam-Muniasamy, G.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Shruti, V.C. An overview of recent advances in micro/nano beads and microfibers research: Critical assessment and promoting the less known. Sci. Total Environ. 2020, 740, 139991. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Zheng, R.; Zhang, Y.; Hong, F.; Mu, J.; Chen, M.; Song, P.; Lin, L.; Lin, H.; Le, F.; et al. Microplastic contamination in benthic organisms from the Arctic and sub-Arctic regions. Chemosphere 2018, 209, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, G.C.; Akter, S.M.; Islam, R.M.; Habib, A.; Chakraborty, T.K.; Zaman, S.; Kabir, A.H.M.E.; Shipin, O.V.; Wahid, M.A. Microplastics contamination in commercial marine fish from the Bay of Bengal. Reg. Stud. Mar. Sci. 2021, 44, 101728. [Google Scholar] [CrossRef]
- Pan, Z.; Zhang, C.; Wang, S.; Sun, D.; Zhou, A.; Xie, S.; Xu, G.; Zou, J. Occurrence of microplastics in the gastrointestinal tract and gills of fish from Guangdong, South China. J. Mar. Sci. Eng. 2021, 9, 981. [Google Scholar] [CrossRef]
- Siddique, M.A.M.; Uddin, A.; Rahman, S.M.A.; Rahman, M.; Islam, M.S.; Kibria, G. Microplastics in an anadromous national fish, Hilsa shad Tenualosa ilisha from the Bay of Bengal, Bangladesh. Mar. Pollut. Bull. 2022, 174, 113236. [Google Scholar] [CrossRef]
- Hossain, M.S.; Sobhan, F.; Uddin, M.N.; Sharifuzzaman, S.M.; Chowdhury, S.R.; Sarker, S.; Chowdhury, M.S.N. Microplastics in fishes from the Northern Bay of Bengal. Sci. Total Environ. 2019, 690, 821–830. [Google Scholar] [CrossRef]
- Hossain, M.S.; Rahman, M.S.; Uddin, M.N.; Sharifuzzaman, S.M.; Chowdhury, S.R.; Sarker, S.; Chowdhury, M.S.N. Microplastic contamination in Penaeid shrimp from the Northern Bay of Bengal. Chemosphere 2020, 238, 124688. [Google Scholar] [CrossRef]
- Neves, D.; Sobral, P.; Ferreira, J.L.; Pereira, T. Ingestion of microplastics by commercial fish off the Portuguese coast. Mar. Pollut. Bull. 2015, 101, 119–126. [Google Scholar] [CrossRef]
- Rahman, M.S.; Hossain, M.S.; Ahmed, M.K.; Akther, S.; Jolly, Y.N.; Akhter, S.; Kabir, M.J.; Choudhury, T.R. Assessment of heavy metals contamination in selected tropical marine fish species in Bangladesh and their impact on human health. Environ. Nanotechnol. Monit. Manag. 2019, 11, 100210. [Google Scholar]
- Ory, N.; Chagnon, C.; Felix, F.; Fernández, C.; Ferreira, J.L.; Gallardo, C.; Garcés Ordóñez, O.; Henostroza, A.; Laaz, E.; Mizraji, R.; et al. Low prevalence of microplastic contamination in planktivorous fish species from the southeast Pacific Ocean. Mar. Pollut. Bull. 2017, 127, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Karami, A.; Golieskardi, A.; Keong Choo, C.; Larat, V.; Galloway, T.S.; Salamatinia, B. The presence of microplastics in commercial salts from different countries. Sci. Rep. 2017, 71, 46173. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.; Webb, H.; Lindeque, P.K.; Fileman, E.S.; Halsband, C.; Galloway, T.S. Isolation of microplastics in biota-rich seawater samples and marine organisms. Sci. Rep. 2014, 41, 4528. [Google Scholar] [CrossRef] [PubMed]
- Avio, C.G.; Gorbi, S.; Regoli, F. Experimental development of a new protocol for extraction and characterization of microplastics in fish tissues: First observations in commercial species from Adriatic Sea. Mar. Environ. Res. 2015, 111, 18–26. [Google Scholar] [CrossRef]
- Mathalon, A.; Hill, P. Microplastic fibers in the intertidal ecosystem surrounding Halifax Harbor, Nova Scotia. Mar. Pollut. Bull. 2014, 81, 69–79. [Google Scholar] [CrossRef]
- Masura, J.; Baker, J.; Foster, G.; Arthur, C. Laboratory Methods for the Analysis of Microplastics in the Marine Environment: Recommendations for Quantifying Synthetic Particles in Waters and Sediments; Silver Spring: Montgomery County, MA, USA, 2015. [Google Scholar] [CrossRef]
- Coppock, R.L.; Cole, M.; Lindeque, P.K.; Queirós, A.M.; Galloway, T.S. A small-scale, portable method for extracting microplastics from marine sediments. Environ. Pollut. 2017, 230, 829–837. [Google Scholar] [CrossRef]
- De Witte, B.; Devriese, L.; Bekaert, K.; Hoffman, S.; Vandermeersch, G.; Cooreman, K.; Robbens, J. Quality assessment of the blue mussel (Mytilus edulis): Comparison between commercial and wild types. Mar. Pollut. Bull. 2014, 85, 146–155. [Google Scholar] [CrossRef]
- Devriese, L.I.; van der Meulen, M.D.; Maes, T.; Bekaert, K.; Paul-Pont, I.; Frère, L.; Robbens, J.; Vethaak, A.D. Microplastic contamination in brown shrimp (Crangon crangon, Linnaeus 1758) from coastal waters of the Southern North Sea and Channel area. Mar. Pollut. Bull. 2015, 98, 179–187. [Google Scholar] [CrossRef]
- Shruti, V.C.; Kutralam-Muniasamy, G. Blanks and bias in microplastic research: Implications for future quality assurance. Trends Environ. Anal. Chem. 2023, 2023, e00203. [Google Scholar] [CrossRef]
- Hossain, M.B.; Banik, P.; Nur, A.A.U.; Rahman, T. Abundance and characteristics of microplastics in sediments from the world’s longest natural beach, Cox’s Bazar, Bangladesh. Mar. Pollut. Bull. 2021, 163, 111956. [Google Scholar] [CrossRef]
- Ingebrigtson, D.N.; Smith, A.L. Infrared analysis of solids by potassium bromide pellet technique. Anal. Chem. 1954, 26, 1765–1768. [Google Scholar] [CrossRef]
- Noda, I.; Dowrey, A.E.; Haynes, J.L.; Marcott, C. Group Frequency Assignments for Major Infrared Bands Observed in Common Synthetic Polymers. In Physical Properties of Polymers Handbook; Springer: New York, NY, USA, 2007; pp. 395–406. [Google Scholar] [CrossRef]
- Jung, M.R.; Horgen, F.D.; Orski, S.V.; Rodriguez, C.V.; Beers, K.L.; Balazs, G.H.; Jones, T.T.; Work, T.M.; Brignac, K.C.; Royer, S.J.; et al. Validation of ATR FT-IR to identify polymers of plastic marine debris, including those ingested by marine organisms. Mar. Pollut. Bull. 2018, 127, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, D.L.; Wilson, J.G.; Harris, C.R.; Jeffrey, D.W. Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgoländer Meeresun. 1980, 33, 566–575. [Google Scholar] [CrossRef]
- Haque, M.R.; Ali, M.M.; Ahmed, W.; Siddique, M.A.B.; Akbor, M.A.; Islam, M.S.; Rahman, M.M. Assessment of microplastics pollution in aquatic species (fish, crab, and snail), water, and sediment from the Buriganga River, Bangladesh: An ecological risk appraisals. Sci. Total Environ. 2023, 857, 159344. [Google Scholar] [CrossRef] [PubMed]
- Peters, C.A.; Thomas, P.A.; Rieper, K.B.; Bratton, S.P. Foraging preferences influence microplastic ingestion by six marine fish species from the Texas Gulf Coast. Mar. Pollut. Bull. 2017, 124, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Güven, O.; Gökdağ, K.; Jovanović, B.; Kıdeyş, A.E. Microplastic litter composition of the Turkish territorial waters of the Mediterranean Sea, and its occurrence in the gastrointestinal tract of fish. Environ. Pollut. 2017, 223, 286–294. [Google Scholar] [CrossRef]
- Bessa, F.; Barría, P.; Neto, J.M.; Frias, J.P.G.L.; Otero, V.; Sobral, P.; Marques, J.C. Occurrence of microplastics in commercial fish from a natural estuarine environment. Mar. Pollut. Bull. 2018, 128, 575–584. [Google Scholar] [CrossRef]
- Jabeen, K.; Su, L.; Li, J.; Yang, D.; Tong, C.; Mu, J.; Shi, H. Microplastics and mesoplastics in fish from coastal and fresh waters of China. Environ. Pollut. 2017, 221, 141–149. [Google Scholar] [CrossRef]
- Silva-Cavalcanti, J.S.; Silva, J.D.B.; França, E.J.; Araújo, M.C.B.; Gusmão, F. Microplastics ingestion by a common tropical freshwater fishing resource. Environ. Pollut. 2017, 221, 218–226. [Google Scholar] [CrossRef]
- Yagi, M.; Kobayashi, T.; Maruyama, Y.; Hoshina, S.; Masumi, S.; Aizawa, I.; Uchida, J.; Kinoshita, T.; Yamawaki, N.; Aoshima, T.; et al. Microplastic pollution of commercial fishes from coastal and offshore waters, Japan. bioRxiv 2021, 10, 465208. [Google Scholar] [CrossRef]
- Jovanović, B.; Gökdağ, K.; Güven, O.; Emre, Y.; Whitley, E.M.; Kideys, A.E. Virgin microplastics are not causing imminent harm to fish after dietary exposure. Mar. Pollut. Bull. 2018, 130, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.; Thiel, M.; Lebreton, L. Nature of Plastic Marine Pollution in the Subtropical Gyres. Handb. Environ. Chem. 2019, 78, 135–162. [Google Scholar]
- Hossain, M.J.; AftabUddin, S.; Akhter, F.; Nusrat, N.; Rahaman, A.; Sikder, M.N.A.; Zhang, J. Surface water, sediment, and biota: The first multi-compartment analysis of microplastics in the Karnafully river, Bangladesh. Mar. Pollut. Bull. 2022, 180, 113820. [Google Scholar] [CrossRef] [PubMed]
- Selvam, S.; Manisha, A.; Venkatramanan, S.; Chung, S.Y.; Paramasivam, C.R.; Singaraja, C. Microplastic presence in commercial marine sea salts: A baseline study along Tuticorin Coastal salt pan stations, Gulf of Mannar, South India. Mar. Pollut. Bull. 2020, 150, 110675. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Deng, H.; Li, B.; Chen, Q.; Pettigrove, V.; Wu, C.; Shi, H. The occurrence of microplastic in specific organs in commercially caught fishes from coast and estuary area of east China. J. Hazard. Mater. 2019, 365, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Batel, A.; Linti, F.; Scherer, M.; Erdinger, L.; Braunbeck, T. Transfer of benzopyrene from microplastics to Artemia nauplii and further to zebrafish via a trophic food web experiment: CYP1A induction and visual tracking of persistent organic pollutants. Environ. Toxicol. Chem. 2016, 35, 1656–1666. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, X.; Huang, W.; Li, J.; Wang, C.; Zhang, D.; Zhang, C. Microplastic pollution in deep-sea sediments and organisms of the Western Pacific Ocean. Environ. Pollut. 2020, 259, 113948. [Google Scholar] [CrossRef]
- Rummel, C.D.; Löder, M.G.J.; Fricke, N.F.; Lang, T.; Griebeler, E.M.; Janke, M.; Gerdts, G. Plastic ingestion by pelagic and demersal fish from the North Sea and Baltic Sea. Mar. Pollut. Bull. 2016, 102, 134–141. [Google Scholar] [CrossRef]
- Zitouni, N.; Bousserrhine, N.; Belbekhouche, S.; Missawi, O.; Alphonse, V.; Boughatass, I.; Banni, M. First report on the presence of small microplastics (≤3 μm) in tissue of the commercial fish Serranus scriba (Linnaeus. 1758) from Tunisian coasts and associated cellular alterations. Environ. Pollut. 2020, 263, 114576. [Google Scholar] [CrossRef]
- Jeong, C.B.; Won, E.J.; Kang, H.M.; Lee, M.C.; Hwang, D.S.; Hwang, U.K.; Zhou, B.; Souissi, S.; Lee, S.J.; Lee, J.S. Microplastic Size-Dependent Toxicity, Oxidative Stress Induction, and p-JNK and p-p38 Activation in the Monogonont Rotifer (Brachionus koreanus). Environ. Sci. Technol. 2016, 50, 8849–8857. [Google Scholar] [CrossRef]
- Lin, L.; Ma, L.S.; Li, H.X.; Pan, Y.F.; Liu, S.; Zhang, L.; Peng, J.P.; Fok, L.; Xu, X.R.; He, W.H. Low level of microplastic contamination in wild fish from an urban estuary. Mar. Pollut. Bull. 2020, 160, 111650. [Google Scholar] [CrossRef] [PubMed]
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R. Accumulation of microplastic on shorelines woldwide: Sources and sinks. Environ. Sci. Technol. 2011, 45, 9175–9179. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Wu, S.; Lu, S.; Liu, M.; Song, Y.; Fu, Z.; Shi, H.; Raley-Susman, K.M.; He, D. Microplastic particles cause intestinal damage and other adverse effects in zebrafish Danio rerio and nematode. Caenorhabditis elegans. Sci. Total Environ. 2018, 619–620, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Impact of Microplastic Beads and Fibers on Waterflea (Ceriodaphnia dubia) Survival, Growth, and Reproduction: Implications of Single and Mixture Exposures. Environ. Sci. Technol. 2017, 51, 13397–13406. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, H.; Chen, B.; Sun, X.; Qu, K.; Xia, B. Microplastic ingestion in deep-sea fish from the South China Sea. Sci. Total Environ. 2019, 677, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Roch, S.; Friedrich, C.; Brinker, A. Uptake routes of microplastics in fishes: Practical and theoretical approaches to test existing theories. Sci. Rep. 2020, 101, 3896. [Google Scholar] [CrossRef]
- Yan, M.; Nie, H.; Xu, K.; He, Y.; Hu, Y.; Huang, Y.; Wang, J. Microplastic abundance, distribution and composition in the Pearl River along Guangzhou city and Pearl River estuary, China. Chemosphere 2019, 217, 879–886. [Google Scholar] [CrossRef]
- Wu, F.; Misra, M.; Mohanty, A.K. Challenges and new opportunities on barrier performance of biodegradable polymers for sustainable packaging. Prog. Polym. Sci. 2021, 117, 101395. [Google Scholar] [CrossRef]
- Ranjani, M.; Veerasingam, S.; Venkatachalapathy, R.; Mugilarasan, M.; Bagaev, A.; Mukhanov, V.; Vethamony, P. Assessment of potential ecological risk of microplastics in the coastal sediments of India: A meta-analysis. Mar. Pollut. Bull. 2021, 163, 111969. [Google Scholar] [CrossRef]
- Nur, A.A.U.; Hossain, M.B.; Banik, P.; Choudhury, T.R.; Liba, S.I.; Umamaheswari, S.; Albeshr, M.F.; Senapathi, V.; Arai, T.; Yu, J. Microplastic contamination in processed and unprocessed sea salts from a developing country and potential risk assessment. Chemosphere 2022, 308, 136395. [Google Scholar]
- Jang, M.; Shim, W.J.; Cho, Y.; Han, G.M.; Song, Y.K.; Hong, S.H. A close relationship between microplastic contamination and coastal area use pattern. Water Res. 2020, 171, 115400. [Google Scholar] [CrossRef] [PubMed]

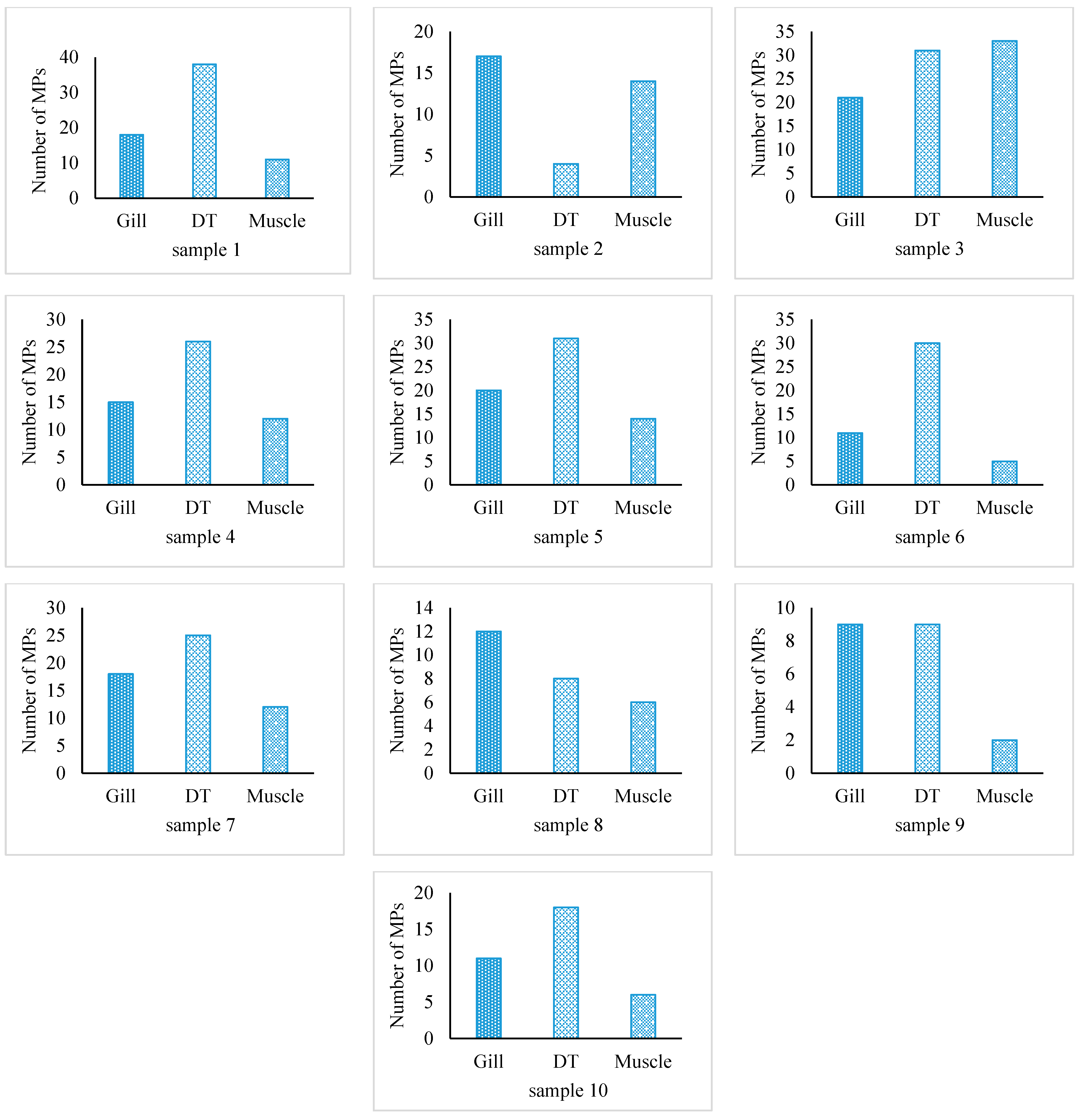
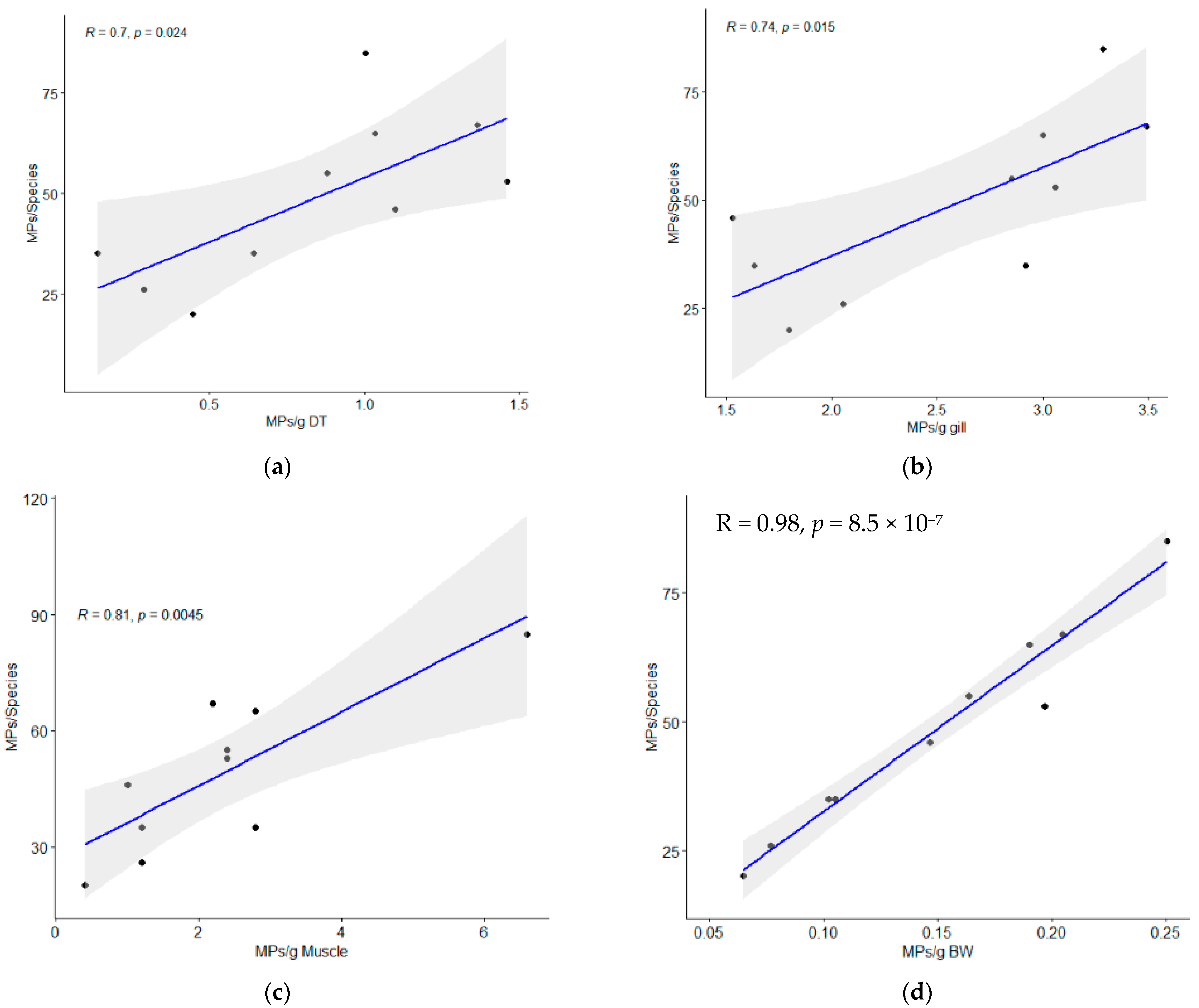

| Scheme. | TL (Range) cm | BW (Range) g | DT Weight (Range) g | Gill Weight (Range) g | MPs/g DT | MPs/g Gill | MPs/g Muscle | MPs/g BW | MPs/ind. |
|---|---|---|---|---|---|---|---|---|---|
| S. guttatus | 36.68 ± 1.53 (33.9–38.2) | 325.22 ± 22.8 (269.3–342.8) | 26.74 ± 4.26 (17.8–30.93) | 6.0 ± 0.79 (4.9–7.2) | 0.84 ± 0.45 | 2.56 ± 0.73 | 2.3 ± 1.72 | 0.15 ± 0.06 | 48.7 ± 20.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossain, M.B.; Pingki, F.H.; Azad, M.A.S.; Nur, A.-A.U.; Banik, P.; Paray, B.A.; Arai, T.; Yu, J. Microplastics in Different Tissues of a Commonly Consumed Fish, Scomberomorus guttatus, from a Large Subtropical Estuary: Accumulation, Characterization, and Contamination Assessment. Biology 2023, 12, 1422. https://doi.org/10.3390/biology12111422
Hossain MB, Pingki FH, Azad MAS, Nur A-AU, Banik P, Paray BA, Arai T, Yu J. Microplastics in Different Tissues of a Commonly Consumed Fish, Scomberomorus guttatus, from a Large Subtropical Estuary: Accumulation, Characterization, and Contamination Assessment. Biology. 2023; 12(11):1422. https://doi.org/10.3390/biology12111422
Chicago/Turabian StyleHossain, Mohammad Belal, Farjana Haque Pingki, Md. Abdus Samad Azad, As-Ad Ujjaman Nur, Partho Banik, Bilal Ahamad Paray, Takaomi Arai, and Jimmy Yu. 2023. "Microplastics in Different Tissues of a Commonly Consumed Fish, Scomberomorus guttatus, from a Large Subtropical Estuary: Accumulation, Characterization, and Contamination Assessment" Biology 12, no. 11: 1422. https://doi.org/10.3390/biology12111422
APA StyleHossain, M. B., Pingki, F. H., Azad, M. A. S., Nur, A.-A. U., Banik, P., Paray, B. A., Arai, T., & Yu, J. (2023). Microplastics in Different Tissues of a Commonly Consumed Fish, Scomberomorus guttatus, from a Large Subtropical Estuary: Accumulation, Characterization, and Contamination Assessment. Biology, 12(11), 1422. https://doi.org/10.3390/biology12111422









