Simple Summary
Transgenic applications have largely focused on constitutive promoters in plants. However, strong and continuous over-expression of certain genes may be redundant and even harmful to plant growth. Thus, tissue-specific promoters are the most suitable for regulating target gene expression. Although several tissue-specific promoters have been identified, the regulatory mechanism of tissue-specific gene expression remains unclear. By a series of GUS staining of 5′ and 3′ deletions, we uncover tissue-specific cis-acting elements in GSX7R, including ten light-responsive elements. The results reveal that GSX7R is a reverse green tissue-specific promoter, except in endosperm. In contrast, strong tissue-specific promoters that can be used for rice improvements are limited. In this study, we successfully showed that the GSX7R promoter can drive the Cry1Ab gene to resistant rice yellow stem borer. In addition, our study demonstrates an effective promoter to drive foreign genes for crop improvement.
Abstract
Plant promoters play a vital role in the initiation and regulation of gene transcription. In this study, a rice protein/gene of unknown expression, named Os8GSX7, was gained from a rice T-DNA capture line. The semi-quantitative RT-PCR analysis showed that the gene was only expressed in root, glume, and flower, but not in stem, leaf, embryo, and endosperm of japonica rice. The GUS activity analysis of the GSX7R promoter showed that it was a reverse green tissue expression promoter, except in endosperm. The forward promoter of GSX7 cannot normally drive the expression of the foreign GUS gene, while the reverse promoter of GSX7 is a green tissue-specific expression promoter, which can drive the expression of the foreign GUS gene. The region from −2097 to −1543 bp was the key region for controlling the green tissue-specific expression. The regulatory sequences with different lengths from the 2097 bp reverse sequence from the upstream region of the Os8GSX7 were fused with the GUS reporter gene and stably expressed in rice. Furthermore, transgenic rice plants carrying Cry1Ab encoding Bacillus thuringiensis endotoxin, regulated by GSX7R, were resistant to yellow stem borer. The analysis suggested that 10 light responsive elements of tissue-specific expression were found, including ACE, Box4, CAT-box, G-Box, G-box, GATA motif, GC motif, I-box, Sp1, and chs-unit1 M1. In addition, the results of 5′ and 3′ deletions further speculated that ACE and I-box may be the key elements for determining the green tissue-specific expression of GSX7R promoter.
1. Introduction
Promoters are the key regulators of plant genetic engineering, during the process of gene transcription [1]. They are mainly divided into three types, i.e., constitutive, inducible, and tissue-specific. Constitutive promoters are usually used to analyze gene function and character modification, which can cause foreign genes to obtain a stable expression level in plants [2]. There are three most commonly used constitutive promoters in genetic engineering, such as 35S, actin, and ubiquitin [3,4,5]. However, a large number of efficient promoters which are continuously driving the expression of foreign genes are not only unnecessary for plant growth, but also consume a lot of nutrients in plants, so far as to have a negative impact on plant growth and development [6]. The transcription of constitutive promoters often inhibits plant growth [7]. Tissue-specific promoters are generally a 5′-UTR of genes expressed in specific tissues, organs, or specific development stages, which can effectively avoid negative effects on plant growth and development [1]. Although some tissue-specific expression promoters have been cloned, mostly including root [8], leaf sheath [9], phloem [10], pollen grain [11], embryo [12], endosperm [13] and green tissue [14,15,16,17], but only a small part of cis-acting regulatory elements are recognized [18]. In addition, the cloned green tissue-specific expression promoters are mainly some genes closely related to light induction and photosynthetic system, which participate in plant photosynthesis and realize the function of converting light energy into sugar [19], such as RBCs [20], DX1 [14], D540 [21]. Isolation and identification of rice green tissue specific expression promoters related to the non-photosynthetic system are helpful to avoiding the influence of light effect, and have a wider range of applications.
The broader expression pattern mainly focuses on the forward promoter [22]. The reverse promoter mainly means that its forward promoter cannot drive the expression of foreign genes. However, a few studies are reported on plant reverse promoters, most of the studies are focused on bi-directional promoters, such as BEAP [23], Cab1/2 [24] and BDDP [25]. Rice is one of the most important food crops in the world [26,27]. The application of strong specific promoters is very important for rice genetic improvement [28]. In this study, a green tissue-specific promoter was obtained through the screening of the T-DNA capture line. The gene information and regulation of the promoter have not been unknown. The reverse promoter fragment 2097 bp upstream of the Os8GSX7 gene was obtained by the PCR cloning method. Furthermore, the 5′ and 3′ end deletion of the reverse promoter GSX7R was analyzed, and the corresponding plant expression vectors were constructed. Through agrobacterium-mediated genetic transformation of rice and GUS tissue staining, we characterized a core region of green-specific control of gene expression. It also provides a reference theoretical basis for the expression and regulation function of the unknown protein. More importantly, due to its strong tissue-specific expression, it can provide an application basis for transgenic safety in the future.
2. Materials and Methods
2.1. Experimental Materials
Japonica rice (Oryza sativa subsp. Japonica), a fine variety was used as a receptor. An independently constructed rice T-DNA insertion mutant library promoter (Figure 1), Escherichia coli strain DH5α, and Agrobacterium tumefaciens strain LBA4404 were preserved in the laboratory. The promoter expression verification vector pCXGUS-P was constructed and provided by Dr. Chen Songbiao [29].
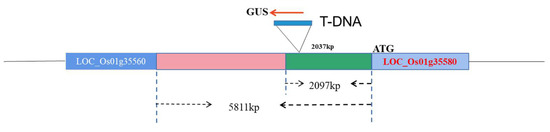
Figure 1.
A schematic diagram showing the insertion position of T-DNA.
2.2. Expression Analysis of Candidate Genes in Rice Organs
An unknown expression gene (LOC_Os01g35580) was obtained by T-DNA capture line, named Os8GSX7, and its promoter was reverse promoter GSX7R. Using RNA extraction kit (Tiangen Biotech Co., Ltd., Beijing, China), total RNA was extracted from various tissues and organs of japonica rice, including root, stem, leaf, leaf sheath, glume, flower, embryo, and endosperm. A total of 2 µg sample of RNA was reverse transcribed into cDNA (Tiangen Biotech Co., Ltd., Beijing, China).
Using the cDNA obtained from the above reverse transcription as the template, RT-PCR semi-quantitative analysis was carried out, and primers were designed using primer 6.0 software to detect the expression of genes (Table 1). The PCR reaction conditions were: 94 °C denaturation for 3 min, 94 °C 15 s, 58.5 °C 15 s, 72 °C 30 s, 28 cycles, extension at 72 °C for 5 min, and storage at 4 °C. Os8GSX7 and GUS genes were amplified with rice actin gene as internal reference. The amplified fragments were 114 bp and 203 bp respectively. PCR products were analyzed by 1.5% agarose gel electrophoresis.

Table 1.
PCR primers used in the study.
2.3. Promoter Cloning and Cis-Acting Element Analysis
Using the 2097 bp sequence truncated from the upstream region of the coding gene sequence ATG of japonica rice, primer 6.0 software was used for primer design (Table 1). Primer design PCR method was used to construct the 5′ and 3′ deletion promoters of forward promoter GSX7F and reverse promoter GSX7R series.
The 5′ and 3′ deletion fragments were cloned into the promoter cloning vector pCXGUS-P by TA cloning through PCR (Figure 2). Agrobacterium tumefaciens LBA4404 was transformed by electric shock, and the mature embryo callus of rice was transformed by Agrobacterium tumefaciens and introduced into Nipponbare.
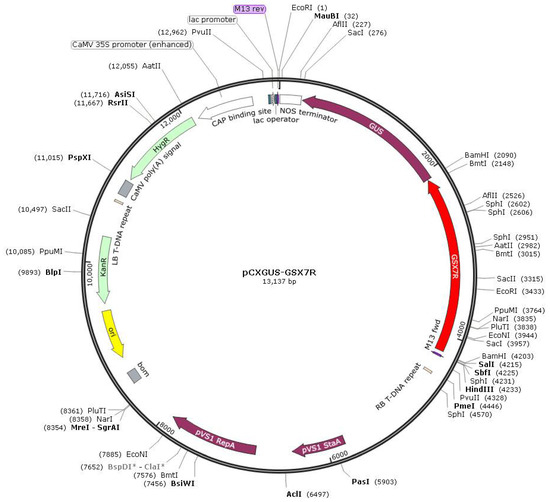
Figure 2.
Schematic diagram of the T-DNA region of binary vector pCXGUS-P with GSX7R or GSX7R deletions.
2.4. Construction of Plant Expression Vector
PCR system (50 µL): 2× Phanta Max Buffer 25 µL, dNTP Mix (10 mM each) 1 µL, template DNA (100 ng/µL), upstream primer (10 µM) 2 µL, downstream primer (10 µM) 2 µL, Phanta Max Super-Fidelity DNA Polymerase 1 µL, insufficient supplement ddH2O to 50 µL. PCR double stress condition: 95 °C 3 min; 95 °C 15 s, 60 °C 15 s, 72 °C for 2 min 30 s, 35 cycles; 72 °C for 5 min. After adding a tail to the PCR product, it was constructed on the skeleton vector pCXGUS-P digested by XcmI and sent to Xiamen Boson Biotech Co., Ltd., Xiamen, China for sequencing verification.
The p1300GSAb vector was constructed by one step cloning kit (Vazyme Biotech, code: C112). The pCAMBIA1300 vector was designed by enzyme digestion by HindIII and EcoRI. GSX7R promoter, Cry1Ab and Tnos were amplified by PCR, and then connected to pCAMBIA1300.
2.5. Analysis of Genetic Transmission and Transgenic Detection
After mechanical shelling of Nipponbare seeds, they were pretreated with 75% ethanol for 2 min, soaked in 3% sodium hypochlorite for 20 min, rinsed with sterile water for 4–5 times, and placed on an N6 medium containing 3 mg/L 2,4-D, dark cultured at 25 °C to induce callus, transformed by Agrobacterium-mediated rice genetic transformation method, and regenerated rice plants [30]. The PCR amplification product of HPT gene was 832 bp [31].
2.6. GUS Histochemical Staining
GUS chemical staining was performed on different tissues and organs of transgenic positive plants according to the previously described method [14]. The roots, stems, leaves, leaf sheaths, glumes, and seeds of positive transgenic plants were cut into appropriate sizes with blades, and put into EP tubes. An appropriate amount of GUS dye was added to make them fully colored [GUS dye composition: 50 mM sodium phosphate (pH 7.0), 10 mmol Na2EDTA, 0.1% Triton X-100, 1 mg/mL x-gluc, 100 μg/mL chloramphenicol, 1 mM potassium ferricyanide, 1 mm/L potassium ferrocyanide and 20% methanol]. The treating samples were placed at 37 °C overnight. After dyeing, it was decolorized with absolute ethanol and 70% (v/v) ethanol for about 1 h. At least 10 repetitions were set for each transformation event, and the photos were taken with a stereomicroscope (OLYMPUS-SZ61, Olympus Corporation, Tokyo, Japan).
3. Results
3.1. The Expression Pattern Analysis of Candidate Gene Os8GSX7 in Rice
The cDNA was obtained by double transcription of total RNA from different tissues and organs of japonica rice. It was used as a template for PCR double correspondence. The expression pattern of candidate gene Os8GSX7 was analyzed by semi-quantitative RT-PCR. The results showed that the Os8GSX7 gene was only expressed in roots, glumes, and flowers (Figure 3A). The reverse promoter GSX7R-2097 derived the expression of exogenous GUS gene in the green tissue of rice, stem, leaf, glume, flower, and embryo, as well as in root but not in the endosperm (Figure 3B).
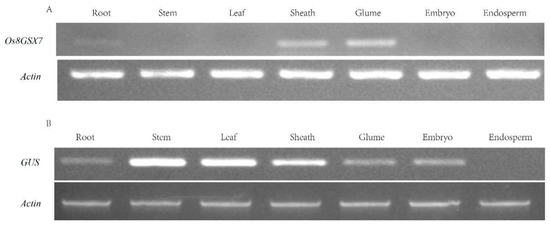
Figure 3.
Expression profiles of Os8GSX7 and GUS by RT-PCR analysis. (A) Expression profiles of Os8GSX7 gene by RT-PCR analysis in the wild type; (B) Expression profiles of GUS gene in transgenic positive lines of GSX7R-2097 by RT-PCR analysis. The rice Actin gene was used as an internal control.
3.2. Isolation and Cloning of the Full-Length Promoter GSX7R and Its Deletion Fragment
Using the genomic DNA of Nipponbare rice as the template, a reverse 2097 bp promoter fragment from the upstream region of the gene was cloned by PCR and named GSX7R. Using pCXGUS-GSX7R as a template, other 5′ and 3′ deletion fragments were cloned respectively (Figure 4). The fragment was connected to the plant expression vector by the TA cloning method, and Agrobacterium tumefaciens was transformed into rice callus. All transgenic plants with positive fragments were identified by hygromycin marker gene detection PCR and planted in a greenhouse.
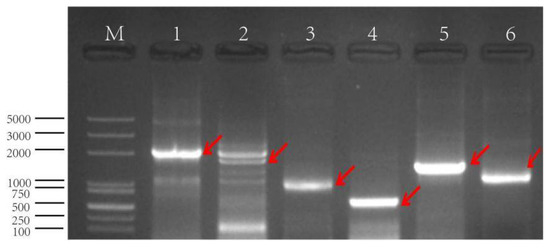
Figure 4.
The PCR amplification of promoter GSX7R and its deletions. M: M5 DL2000 plus DNA marker, 1: GSX7R-2097, 2: GSX7R-1765, 3: GSX7R-1198, 4: GSX7R-554, 5: GSX7R-1543, 6: GSX7R-1212. The amplified bands are indicated by red arrows.
3.3. Bioinformatics Analysis of Reverse Promoter GSX7R Sequence
Through the promoter prediction software plantCARE cis-acting element database, the promoter GSX7R sequence of 2097 bp reverse fragment upstream of the Os8GSX7 gene was analyzed. There were mainly 46 elements. In addition to the core elements of the CAAT box and TATA box of the promoter, it was found that there were 10 potential key cis-regulatory elements specifically expressed by green tissue (Table S1, Figure 5), including ACE, Box4, CAT box, G-Box, G-box, GATA motif, GC motif, I-box, Sp1, and chs-unit1 M1 components.
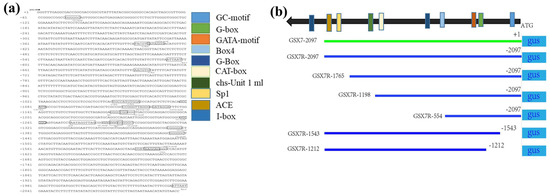
Figure 5.
The location of putative cis-acting elements in GSX7R predicted by the PlantCARE database and schematic diagrams of promoter deletion constructs. (a) Putative cis-acting elements in GSX7R, The reverse 5′-region of the Os8GSX7 gene containing the 2097 bp promoter sequence from the translational start site. The transcription initiation site is defined as +1. The GC-motif, G-box, and other key cis-acting elements are underlined with and indicated by black frame as shown in the legend and different colors outside. The position of each element is also indicated by schematic diagrams; (b) The schematic diagrams of the truncated GSX7R constructs. The numbers to the left of these diagrams indicate the position of the 5′-deletion or 3′-deletion.
3.4. Molecular Detection of Transgenic Rice
The screening marker hygromycin gene (hpt) was used to detect the genes of rice-positive transgenic plants with different vector skeletons. The results showed that the positive progeny plants were isolated from different transformed plants (Figure 6). The size of the hygromycin gene was in line with the expected 832 bp.
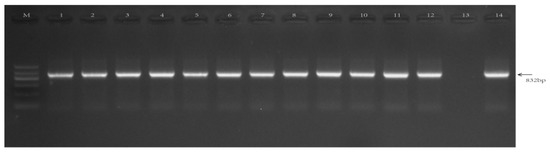
Figure 6.
PCR analysis of hpt in different transgenic rice. M: M5 Marker II DNA Marker, 1–2: GSX7R-2097; 3–4: GSX7R-1765, 5–6: GSX7R-1198, 7–8: GSX7R-554, 9–10: GSX7R-1543, 11–12: GSX7R-1212, 13: non-transgenic rice, 14: Positive plasmid control.
3.5. GUS Activity Analysis of GSX7R and Truncated Promoter
The roots, stems, leaves, leaf sheaths, glumes, and seeds of transgenic positive plants were selected for GUS staining analysis. The GUS gene driven by constitutive act promoter was positive. The results showed that the forward GSX7 full-length promoter could not start GUS protein expression, while the reverse GSX7R full-length promoter could start GUS protein expression, so GSX7R was a reverse expression promoter. The GSX7R promoter was positive in other green tissues except for the expression of GUS protein in the endosperm (Figure 7a), which further confirmed that the reverse promoter GSX7R was a green tissue-specific expression promoter.
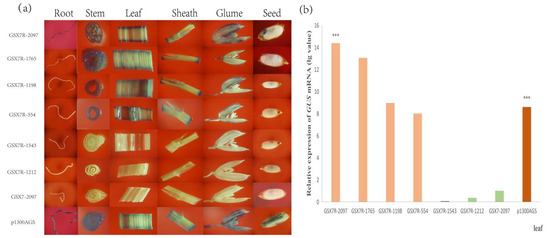
Figure 7.
GUS staining and quantification of GUS mRNA transcript of transgenic rice containing different 5′ and 3′-deletion. (a) Histochemical analysis of transgenic rice plants containing various GSX7R promoter fragment/GUS fusions in different tissues; (b) GUS mRNA expression in the leaf of various rice transformants. Quantitative qRT-PCR analysis of GUS transcripts from the leaf in transgenic rice plants. Data are shown as mean ± SD (n = 3), Student’s t-test. *** indicates highly significant differences from all tested tissues for each transgenic plant. Relative expression was calculated as lg2−ΔΔCt.
The results showed that GSX7R-1765, GSX7R-1198, and GSX7R-554 deleted at the 5′ ends could drive the expression of the GUS gene in the stem, leaf, leaf sheath, and glume, but not in root and seed. However, GSX7R-1543 and GSX7R-1212 with 3′ deletions could not drive the expression of the GUS reporter gene. Therefore, the promoter element at the 3′ ends is very critical to the function of the promoter. To determine the strength of the seven GSX7R promoter variants, the quantitative expression of GUS mRNA in leaves was determined. Compared to the histochemical staining of GUS, the levels of GUS transcript were designated as “no expression” in GSX7 transformed rice. Compared with the GUS expression driven by only the Actin promoter of p1300AGS, GSX7R-2097 and GSX7R-1765 had significantly strong expression (Figure 7b). These results demonstrated that the GSX7R promoter might play an important role in the regulation of gene expression in the green tissue of plants.
3.6. Evaluation of the Ability of GSX7R Promoter to Drive Foreign Genes
To evaluate the ability of GSX7R promoter to drive foreign genes, a transgenic vector was constructed (Figure 8a) and insect resistance was carried between transgenic and wild-type (WT) plants in the greenhouse paddy field. The results showed that WT plants were severely damaged by yellow stem borer (Figure 8b(B,D)), whereas the transgenic plants grew well (Figure 8b(A,C)). It demonstrated that the GSX7R promoter had the strong ability to drive the expression of foreign genes.

Figure 8.
Schematic diagram of vector construction and bioassays in the field. (a) GSX7R:Cry1Ab:p1300GSAb plant transformation vector containing the Cry1Ab gene driven by the GSX7R promoter; (b) (A,C) Transgenic plants without any symptom of infestation in the whole-plant bioassay; (B,D) Wild-type plants showed white heads in the whole-plant bioassay. Red arrow indicates damage to YSB.
4. Discussion
The plant promoters have significant importance with respect to plant biotechnology [32]. The precise regulation of genes mainly depends on the promoters. The characteristics of promoter elements basically determine the spatio-temporal expression and transcription level of the gene [33]. Generally, there are significant differences in promoters within and between genomes, which shows that some promoters contain specific types of bases, while others have diverse and complex sequence characteristics [34]. In this study, the surface characteristics of a 2097 bp reverse promoter upstream of the Os8GSX7 (LOC_Os01g35580) gene in rice were studied. The forward promoter could drive the expression of rice endogenous gene Os8GSX7 in roots, glumes, and flowers, but it could not drive the expression of the GUS reporter gene. The reverse promoter GSX7R had high intensity and could drive the specific expression of the GUS reporter gene in rice green tissue (Figure 7a). Compared with the constitutive promoter p1300AGS, except that the expression was not detected in endosperm and was detected in the root, other green tissue parts were expressed, including stem, leaf, leaf sheath, glume, and embryo.
In order to further study the function of green tissue-specific expression cis-acting elements, we analyzed the 5′ and 3′ deletion truncation of GSX7R promoter, constructed seven different full-length and deletion promoter expression vectors, transformed them into rice, and obtained their transgenic positive plants by hygromycin resistance screening and PCR detection of resistance genes. In addition to the promoter core element TATA box, CAAT box, and GC motif that enhance transcription efficiency, 10 elements closely related to the specific expression of light-response were found e.g., ACE, Box4, CAT box, G-Box, G-box, GATA motif, GC motif, I-box, Sp1, and chs-unit1 M1. From the distribution position, they were mainly distributed in the −1021 to −1621 region of the promoter. When GSX7R promoter 5′ was deleted to −1765, −1198, and −554, GUS expression was not detected in roots and embryos, but was still strongly expressed in stems, leaves, leaf sheaths, and glumes. When the 3′ ends were deleted to −1212 and −1543, the promoter lost the activity of driving foreign genes and GUS expression was not detected (Figure 7a). Combined with the GUS mRNA expression of the 3′ end fragment (Figure 7b), it can be shown that ACE, SP1, GC motif, GATA motif, I-box, and Box4 elements are very important for the tissue-specific expression of promoter GSX7R. Analysis together with the results of 5′ end fragment deletion showed that ACE and I-box may play a decisive role in the expression of green tissue promoter GSX7R. Previous studies have shown that the ACE element in the promoter is regulated by HY5, and the lack of HY5 will widely reduce the accumulation of other photosystem proteins except for PSII protein [35]. I-box elements mainly regulate the activity of promoters in leaves, not fruits, and can control light regulatory genes [36,37]. In addition, I-box and G-box elements act as light response enhancers of CMA5 activity in plants [38,39].
In addition to light response-related elements, the promoter GSX7R also had meristem element (CAT box), MYBHv1 binding site (CCAAT box), MYB transcription element (MBS, MYB, MYB recognition site, MYB-like, and MYB binding site), seed-specific expression element (RY-element), auxin response element (TGA element), jasmonic acid response element (CGTCA motif), etc. Studies have shown that genes with MYB cis-acting elements can combine with jasmonic acid response to improve cold tolerance in rice [40]. In addition, the rice OsMYB4 transcription factor can directly or indirectly regulate the tolerance of target genes by interacting with transcription factors such as the CCAAT box and MYB [41]. The combination of MYB binding site element and TIMYB2r-1 protein can improve the disease resistance of wheat [42]. Therefore, the functional study of these elements may provide a research direction for revealing the unknown functional genes of Os8GSX7 (LOC_Os01g35580).
5. Conclusions
In this study, a rice reverse promoter (OsCGX7R) was obtained through T-DNA capture line screening. The expression intensity of the reverse promoter was higher than that of the forward promoter, and the reverse promoter was a green tissue efficient expression promoter. Compared with the green tissue-specific expression promoter related to the traditional photosynthetic system, it can avoid the influence of light regulation and has wider application. The results of this study can provide a new basis for the application and development of green tissue-specific expression promoters in transgenic safety.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biology11081092/s1, Table S1: Cis-elements related to green tissue specific expression in promoter sequence by PlantCARE.
Author Contributions
Conceptualization, F.C. and Z.L.; methodology, M.L., J.Y., S.W. and S.T.; software, J.Y.; validation, J.Y., M.M.A. and Z.L.; formal analysis, M.L.; investigation, Z.L.; resources, Z.L.; data curation, J.Y. and M.M.A.; writing—original draft preparation, M.L. and M.M.A.; writing—review and editing, J.Y., F.C. and Z.L.; supervision, Z.L.; project administration, F.C. and Z.L.; funding acquisition, F.C. and Z.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by Special Support for Scientific Research of Fujian Provincial Public Welfare Scientific Research Institutes (2019R1027-6).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Xue, M.; Long, Y.; Zhao, Z.; Huang, G.; Huang, K.; Zhang, T.; Jiang, Y.; Yuan, Q.; Pei, X. Isolation and Characterization of a Green-Tissue Promoter from Common Wild Rice (Oryza rufipogon Griff.). Int. J. Mol. Sci. 2018, 19, 2009. [Google Scholar] [CrossRef] [PubMed]
- Kishi-Kaboshi, M.; Aida, R.; Sasaki, K. Parsley ubiquitin promoter displays higher activity than the CaMV 35S promoter and the chrysanthemum actin 2 promoter for productive, constitutive, and durable expression of a transgene in Chrysanthemum morifolium. Breed. Sci. 2019, 69, 19036. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Lee, Y.-H.; Jiang, Y.; Shi, X.; Kertbundit, S.; Hall, T.C. A Two-Edged Role for the Transposable Element Kiddo in the rice ubiquitin2 Promoter. Plant Cell 2005, 17, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Lin, Z.; McElroy, D.; Wu, R. Identification of a rice Actin 2 gene regulatory region for high-level expression of transgenes in monocots. Plant Biotechnol. J. 2009, 7, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Kishi-Kaboshi, M.; Aida, R.; Sasaki, K. Generation of Gene-Edited Chrysanthemum morifolium Using Multi-Copy Transgenes as Targets and Markers. Plant. Cell Physiol. 2017, 58, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, H.-B.; Jin, H.-L. Light Signaling-Dependent Regulation of PSII Biogenesis and Functional Maintenance. Plant Physiol. 2020, 183, 1855–1868. [Google Scholar] [CrossRef]
- Jeong, H.-J.; Jung, K.-H. Rice tissue-specific promoters and condition-dependent promoters for effective translational application. J. Integr. Plant Biol. 2015, 57, 913–924. [Google Scholar] [CrossRef]
- Li, Y.; Li, C.; Cheng, L.; Yu, S.; Shen, C.; Pan, Y. Over-expression of OsPT2 under a rice root specific promoter Os03g01700. Plant Physiol. Biochem. 2019, 136, 52–57. [Google Scholar] [CrossRef]
- Oshima, M.; Taniguchi, Y.; Akasaka, M.; Abe, K.; Ichikawa, H.; Tabei, Y.; Tanaka, J. Development of a visible marker trait based on leaf sheath—Specific anthocyanin pigmentation applicable to various genotypes in rice. Breed. Sci. 2019, 69, 244–254. [Google Scholar] [CrossRef]
- Dutt, M.; Ananthakrishnan, G.; Jaromin, M.K.; Brlansky, R.H.; Grosser, J.W. Evaluation of four phloem-specific promoters in vegetative tissues of transgenic citrus plants. Tree Physiol. 2012, 32, 83–93. [Google Scholar] [CrossRef]
- Wang, M.; Yan, W.; Peng, X.; Chen, Z.; Xu, C.; Wu, J.; Deng, X.W.; Tang, X. Identification of late-stage pollen-specific promoters for construction of pollen-inactivation system in rice. J. Integr. Plant. Biol. 2020, 62, 1246–1263. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jung, I.J.; Kim, D.Y.; Fanata, W.I.; Son, B.H.; Yoo, J.Y.; Harmoko, R.; Ko, K.S.; Moon, J.C.; Jang, H.H.; et al. Proteomic identification of an embryo-specific 1Cys-Prx promoter and analysis of its activity in transgenic rice. Biochem. Biophys. Res. Commun. 2011, 408, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Patti, T.; Bembi, B.; Cristin, P.; Mazzarol, F.; Secco, E.; Pappalardo, C.; Musetti, R.; Martinuzzi, M.; Versolatto, S.; Cariati, R.; et al. Endosperm-specific expression of human acid beta-glucosidase in a waxy rice. Rice 2012, 5, 34. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ye, R.; Zhou, F.; Lin, Y. Two novel positive cis-regulatory elements involved in green tissue-specific promoter activity in rice (Oryza sativa L. ssp.). Plant. Cell Rep. 2012, 31, 1159–1172. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Mei, F.; Zhang, W.; Shen, Z.; Fang, J. Creation of Bt Rice Expressing a Fusion Protein of Cry1Ac and Cry1I-Like Using a Green Tissue-Specific Promoter. J. Econ. Entomol. 2014, 107, 1674–1679. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, R.; Balakrishnan, N.; Sudhakar, D.; Udayasuriyan, V. Development of leaffolder resistant transgenic rice expressing cry2AX1 gene driven by green tissue-specific rbcS promoter. World J. Microbiol. Biotechnol. 2016, 32, 37. [Google Scholar] [CrossRef]
- Bai, J.; Wang, X.; Wu, H.; Ling, F.; Zhao, Y.; Lin, Y.; Wang, R. Comprehensive construction strategy of bidirectional green tissue-specific synthetic promoters. Plant. Biotechnol. J. 2020, 18, 668–678. [Google Scholar] [CrossRef]
- Cai, M.; Yuan, T.; Duan, L.; Li, X.; Wang, S. Identification of potential protein regulators bound to the tissue-specific positive and negative cis-acting elements of a green tissue-specific promoter in rice. Plant. Biol. 2008, 10, 771–777. [Google Scholar] [CrossRef]
- Liu, W.; Mazarei, M.; Ye, R.; Peng, Y.; Shao, Y.; Baxter, H.L.; Sykes, R.W.; Turner, G.B.; Davis, M.F.; Wang, Z.-Y.; et al. Switchgrass (Panicum virgatum L.) promoters for green tissue-specific expression of the MYB4 transcription factor for reduced-recalcitrance transgenic switchgrass. Biotechnol. Biofuels 2018, 11, 122. [Google Scholar] [CrossRef]
- Matsuoka, M.; Kyozuka, J.; Shimamoto, K.; Kano-Murakami, Y. The promoters of two carboxylases in a C4 plant (maize) direct cell-specific, light-regulated expression in a C3 plant (rice). Plant J. 1994, 6, 311–319. [Google Scholar] [CrossRef]
- Cai, M.; Wei, J.; Li, X.; Xu, C.; Wang, S. A rice promoter containing both novel positive and negative cis-elements for regulation of green tissue-specific gene expression in transgenic plants. Plant Biotechnol. J. 2007, 5, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-J.; Yue, Q.-J.; Zhang, W. Structural and functional analysis of an asymmetric bidirectional promoter in Arabidopsis thaliana. J. Integr. Plant. Biol. 2015, 57, 162–170. [Google Scholar] [CrossRef]
- Chennareddy, S.; Cicak, T.; Clark, L.; Russell, S.; Skokut, M.; Beringer, J.; Yang, X.; Jia, Y.; Gupta, M. Expression of a novel bi-directional Brassica napus promoter in soybean. Transgenic Res. 2017, 26, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.; Han, J.; Zhang, Z.J.; Mitra, A. The intergenic region of Arabidopsis thaliana cab1 and cab2 divergent genes functions as a bidirectional promoter. Planta 2009, 229, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.T.; Jayasankar, S.; Gray, D.J. Bi-directional Duplex Promoters with Duplicated Enhancers Significantly Increase Transgene Expression in Grape and Tobacco. Transgenic Res. 2004, 13, 143–154. [Google Scholar] [CrossRef]
- Ladha, J.K.; Tirol-Padre, A.; Reddy, K.; Ventura, W. Prospects and Problems of Biological Nitrogen Fixation in Rice Production: A Critical Assessment; Springer: Cham, Switzerland, 1993; pp. 677–682. [Google Scholar]
- Javed, T.; Ali, M.M.; Shabbir, R.; Gull, S.; Ali, A.; Khalid, E.; Abbas, A.N.; Tariq, M. Muqmirah Rice seedling establishment as influenced by cultivars and seed priming with potassium nitrate. J. Appl. Res. Plant Sci. 2020, 1, 65–75. [Google Scholar] [CrossRef]
- Wang, R.; Yan, Y.; Zhu, M.; Yang, M.; Zhou, F.; Chen, H.; Lin, Y. Isolation and Functional Characterization of Bidirectional Promoters in Rice. Front. Plant Sci. 2016, 7, 766. [Google Scholar] [CrossRef]
- Chen, S.; Songkumarn, P.; Liu, J.; Wang, G.-L. A Versatile Zero Background T-Vector System for Gene Cloning and Functional Genomics. Plant Physiol. 2009, 150, 1111–1121. [Google Scholar] [CrossRef]
- Chen, H.; Zhao, Y.; Chong, K. Improved High-efficiency System for Rice Transformation Using Mature Embryo-derived Calli. Chinese Bull. Bot. 2008, 25, 322–331. [Google Scholar]
- Su, J.; Zhang, G.; Yu, W.; Song, H. Exogenous Cry1Ac/CpTI of Common Wild Rice by Introgressing from Transgenic Cultivated Rice can be Stable Inheritance and Expression. GMO Biosaf. Res. 2012, 2, 12–17. [Google Scholar] [CrossRef]
- Nakamura, S.; Nakao, A.; Kawamukai, M.; Kimura, T.; Ishiguro, S.; Nakagawa, T. Development of Gateway Binary Vectors, R4L1pGWBs, for Promoter Analysis in Higher Plants. Biosci. Biotechnol. Biochem. 2009, 73, 2556–2559. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dutt, M.; Dhekney, S.A.; Soriano, L.; Kandel, R.; Grosser, J.W. Temporal and spatial control of gene expression in horticultural crops. Hortic. Res. 2014, 1, 14047. [Google Scholar] [CrossRef] [PubMed]
- Brázda, V.; Bartas, M.; Bowater, R.P. Evolution of Diverse Strategies for Promoter Regulation. Trends Genet. 2021, 37, 730–744. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, R.; Lv, D.; Zhang, C.; Yang, H.; Zhang, J.; Wen, J.; Li, C.; Tan, X. Identification of the Core Pollen-Specific Regulation in the Rice OsSUT3 Promoter. Int. J. Mol. Sci. 2020, 21, 1909. [Google Scholar] [CrossRef]
- Ali, M.M.; Alam, S.M.; Anwar, R.; Ali, S.; Shi, M.; Liang, D.; Lin, Z.; Chen, F. Genome-Wide Identification, Characterization and Expression Profiling of Aluminum-Activated Malate Transporters in Eriobotrya japonica Lindl. Horticulturae 2021, 7, 441. [Google Scholar] [CrossRef]
- Baum, K.; Groning, B.; Meier, I. Improved ballistic transient transformation conditions for tomato fruit allow identification of organ-specific contributions of I-box and G-box to the RBCS2 promoter activity. Plant J. 1997, 12, 463–469. [Google Scholar] [CrossRef]
- Zhi, C.; Ali, M.M.; Alam, S.M.; Gull, S.; Ali, S.; Yousef, A.F.; Ahmed, M.A.A.; Ma, S.; Chen, F. Genome-Wide in Silico Analysis and Expression Profiling of Phosphoenolpyruvate Carboxylase Genes in Loquat, Apple, Peach, Strawberry and Pear. Agronomy 2021, 12, 25. [Google Scholar] [CrossRef]
- Lopez-Ochoa, L.; Acevedo-Hernandez, G.; Martinez-Hernandez, A.; Arguello-Astorga, G.; Herrera-Estrella, L. Structural relationships between diverse cis-acting elements are critical for the functional properties of a rbcS minimal light regulatory unit. J. Exp. Bot. 2007, 58, 4397–4406. [Google Scholar] [CrossRef]
- Mao, D.; Xin, Y.; Tan, Y.; Hu, X.; Bai, J.; Liu, Z.; Yu, Y.; Li, L.; Peng, C.; Fan, T.; et al. Natural variation in the HAN1 gene confers chilling tolerance in rice and allowed adaptation to a temperate climate. Proc. Natl. Acad. Sci. USA 2019, 116, 3494–3501. [Google Scholar] [CrossRef]
- Park, M.-R.; Yun, K.-Y.; Mohanty, B.; Herath, V.; Xu, F.; Wijaya, E.; Bajic, V.B.; YUN, S.-J.; De Los Reyes, B.G. Supra-optimal expression of the cold-regulated OsMyb4 transcription factor in transgenic rice changes the complexity of transcriptional network with major effects on stress tolerance and panicle development. Plant Cell Environ. 2010, 33, 2209–2230. [Google Scholar] [CrossRef]
- Liu, X.; Yang, L.; Zhou, X.; Zhou, M.; Lu, Y.; Ma, L.; Ma, H.; Zhang, Z. Transgenic wheat expressing Thinopyrum intermedium MYB transcription factor TiMYB2R-1 shows enhanced resistance to the take-all disease. J. Exp. Bot. 2013, 64, 2243–2253. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).