Equine Genital Squamous Cell Carcinoma Associated with EcPV2 Infection: RANKL Pathway Correlated to Inflammation and Wnt Signaling Activation
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Histopathological Diagnosis and Immunohistochemistry
2.3. DNA Extraction and EcPV2 Detection
2.4. RNA Extraction and EcPV2 Gene Expression Evaluation
2.5. RT-qPCR for Host Gene Study
2.6. Statistical Analysis
3. Results
3.1. Case Selection, Histological Diagnosis of Tumors, Mitotic Count, and Ki67 Index
3.2. EcPV2 DNA Detection and EcPV2 Gene Expression
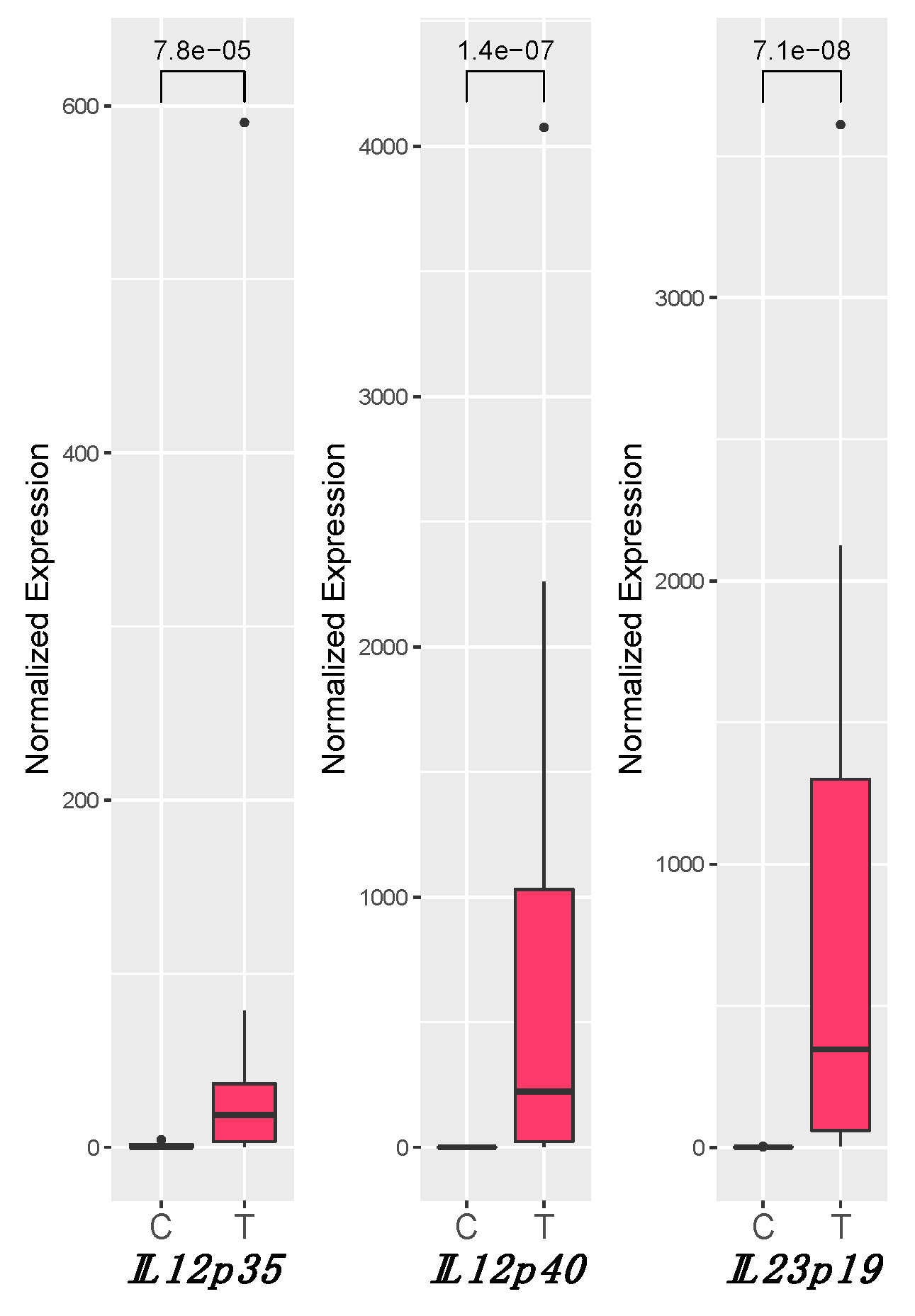
3.3. IL12 and IL23 Gene Expression
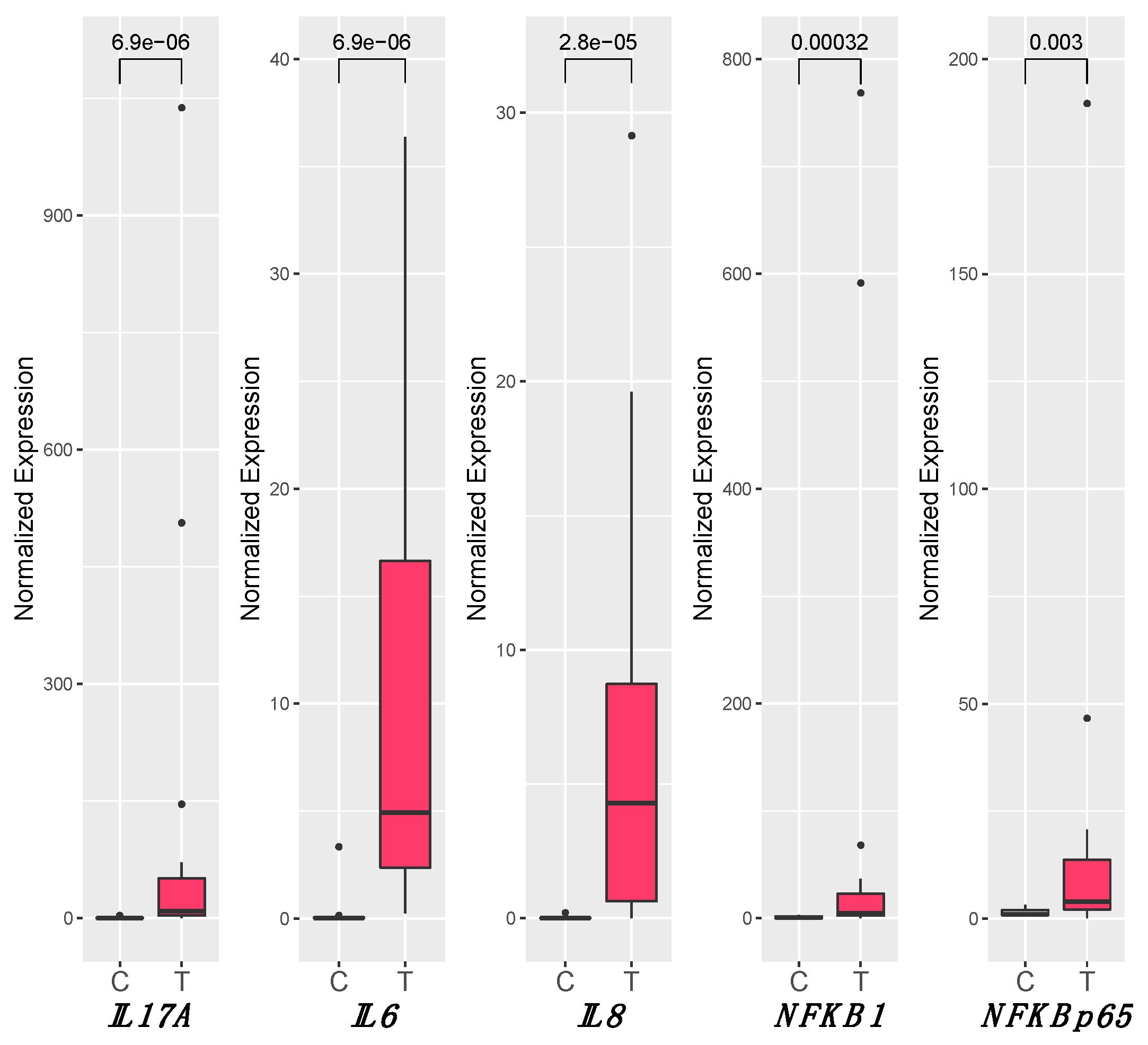
3.4. Inflammation
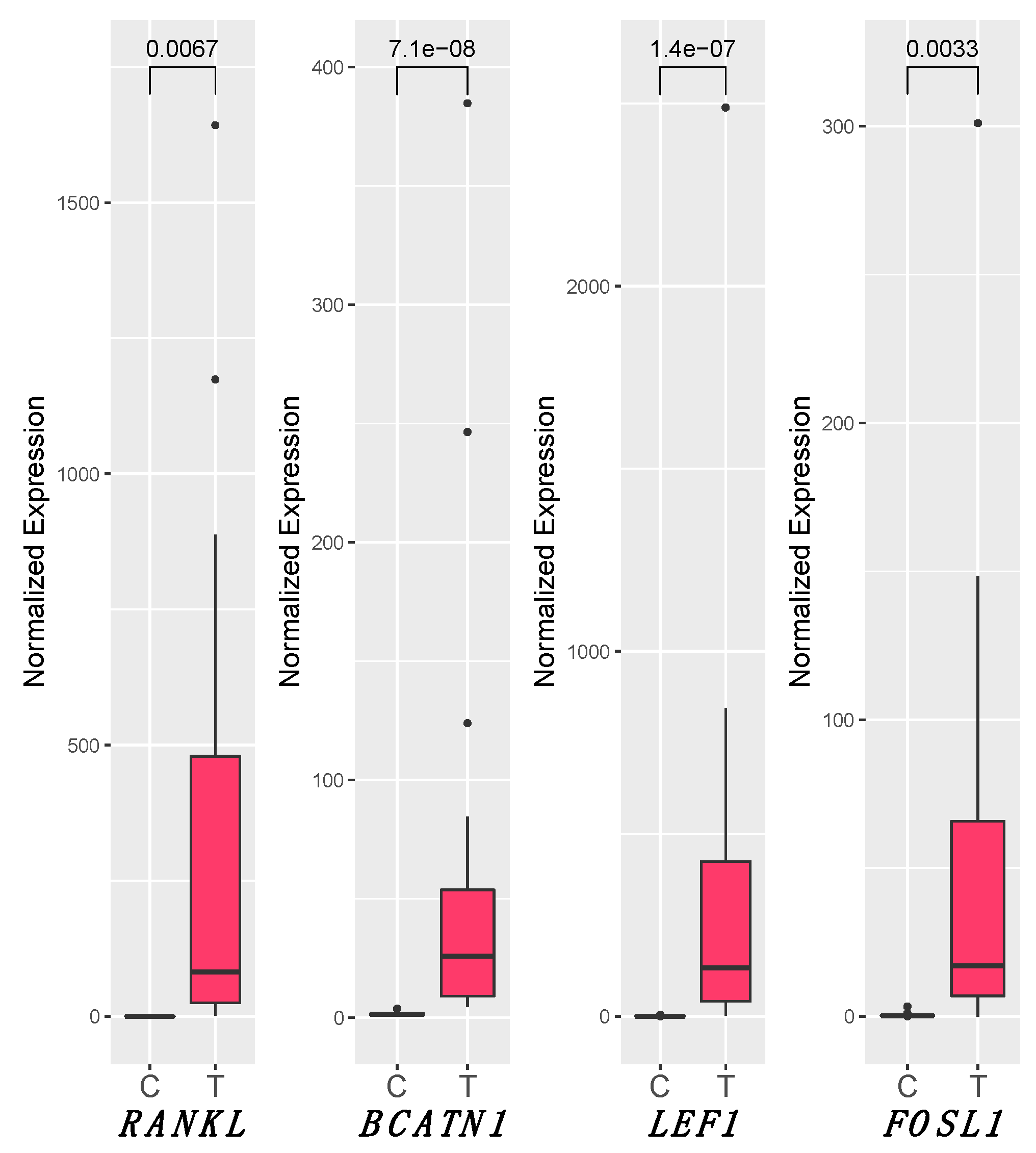
3.5. RANKL and Wnt Signaling Pathways
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arthurs, C.; Suarez-Bonnet, A.; Willis, C.; Xie, B.; Machulla, N.; Mair, T.S.; Cao, K.; Millar, M.; Thrasivoulou, C.; Priestnall, S.L.; et al. Equine Penile Squamous Cell Carcinoma: Expression of Biomarker Proteins and EcPV2. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Munday, J.S. Bovine and Human Papillomaviruses: A Comparative Review. Vet. Pathol. 2014, 51, 1063–1075. [Google Scholar] [CrossRef]
- Christensen, N.D.; Budgeon, L.R.; Cladel, N.M.; Hu, J. Recent Advances in Preclinical Model Systems for Papillomaviruses. Virus Res. 2017, 231, 108–118. [Google Scholar] [CrossRef]
- Porcellato, I.; Mecocci, S.; Mechelli, L.; Cappelli, K.; Brachelente, C.; Pepe, M.; Orlandi, M.; Gialletti, R.; Passeri, B.; Ferrari, A.; et al. Equine Penile Squamous Cell Carcinomas as a Model for Human Disease: A Preliminary Investigation on Tumor Immune Microenvironment. Cells 2020, 9, 2364. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Karin, M. Inflammation and Oncogenesis: A Vicious Connection. Curr. Opin. Genet. Dev. 2010, 20, 65–71. [Google Scholar] [CrossRef] [PubMed]
- de Visser, K.E.; Korets, L.V.; Coussens, L.M. De Novo Carcinogenesis Promoted by Chronic Inflammation Is B Lymphocyte Dependent. Cancer Cell 2005, 7, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Niu, Z.; Zhang, H.; Kong, Y.; Wang, Z.; Yang, X.; Yuan, F. Imbalance of Th1/Th2 and Th17/Treg during the Development of Uterine Cervical Cancer. Int. J. Clin. Exp. Pathol. 2019, 9, 3604. [Google Scholar]
- Clevers, H.; Nusse, R. Wnt/β-Catenin Signaling and Disease. Cell 2012, 149, 1192–1205. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; He, H.; Lei, G. Wnt/β-Catenin Pathway in Bone Cancers. Tumor Biol. 2014, 35, 9439–9445. [Google Scholar] [CrossRef]
- Rampias, T.; Boutati, E.; Pectasides, E.; Sasaki, C.; Kountourakis, P.; Weinberger, P.; Psyrri, A. Activation of Wnt Signaling Pathway by Human Papillomavirus E6 and E7 Oncogenes in HPV16-Positive Oropharyngeal Squamous Carcinoma Cells. Mol. Cancer Res. 2010, 8, 433–443. [Google Scholar] [CrossRef]
- Muñoz Bello, J.O.; Olmedo Nieva, L.; Contreras Paredes, A.; Fuentes Gonzalez, A.M.; Rocha Zavaleta, L.; Lizano, M. Regulation of the Wnt/β-Catenin Signaling Pathway by Human Papillomavirus E6 and E7 Oncoproteins. Viruses 2015, 7, 4734–4755. [Google Scholar] [CrossRef]
- Corda, G.; Sala, A. Non-Canonical WNT/PCP Signalling in Cancer: Fzd6 Takes Centre Stage. Oncogenesis 2017, 6, e364. [Google Scholar] [CrossRef]
- Moparthi, L.; Koch, S. Wnt Signaling in Intestinal Inflammation. Differentiation 2019, 108, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Arya, M.; Thrasivoulou, C.; Henrique, R.; Millar, M.; Hamblin, R.; Davda, R.; Aare, K.; Masters, J.R.; Thomson, C.; Muneer, A.; et al. Targets of Wnt/ß-Catenin Transcription in Penile Carcinoma. PLoS ONE 2015, 10, e0124395. [Google Scholar] [CrossRef]
- Yang, M.; Wang, M.; Li, X.; Xie, Y.; Xia, X.; Tian, J.; Zhang, K.; Tang, A. Wnt Signaling in Cervical Cancer? J. Cancer 2018, 9, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sastre, M.A.; González-Maya, L.; Delgado, R.; Lizano, M.; Tsubaki, G.; Mohar, A.; García-Carrancá, A. Abnormal Distribution of E-Cadherin and β-Catenin in Different Histologic Types of Cancer of the Uterine Cervix. Gynecol. Oncol. 2005, 97, 330–336. [Google Scholar] [CrossRef]
- Kovács, B.; Vajda, E.; Nagy, E.E. Regulatory Effects and Interactions of the Wnt and OPG-RANKL-RANK Signaling at the Bone-Cartilage Interface in Osteoarthritis. Int. J. Mol. Sci. 2019, 20, 4653. [Google Scholar] [CrossRef]
- Okamoto, K.; Nakashima, T.; Shinohara, M.; Negishi-Koga, T.; Komatsu, N.; Terashima, A.; Sawa, S.; Nitta, T.; Takayanagi, H. Osteoimmunology: The Conceptual Framework Unifying the Immune and Skeletal Systems. Physiol. Rev. 2017, 97, 1295–1349. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Shi, Y.; Xu, L.; Peng, Q.; Wang, F.; Wang, X.; Sun, W.; Lu, Y.; Tsao, B.P.; Zhang, M.; et al. Modulation of IL-6 Induced RANKL Expression in Arthritic Synovium by a Transcription Factor SOX5. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Anderson, D.M.; Maraskovsky, E.; Billingsley, W.L.; Dougall, W.C.; Tometsko, M.E.; Roux, E.R.; Teepe, M.C.; DuBose, R.F.; Cosman, D.; Galibert, L. A Homologue of the TNF Receptor and Its Ligand Enhance T-Cell Growth and Dendritic-Cell Function. Nature 1997, 390, 175–179. [Google Scholar] [CrossRef]
- Leibbrandt, A.; Penninger, J.M. Novel Functions of RANK(L) Signaling in the Immune System. In Osteoimmunology; Choi, Y., Ed.; Advances in Experimental Medicine and Biology; Springer US: Boston, MA, USA, 2009; Volume 658, pp. 77–94. ISBN 978-1-4419-1049-3. [Google Scholar]
- Ono, T.; Hayashi, M.; Sasaki, F.; Nakashima, T. RANKL Biology: Bone Metabolism, the Immune System, and Beyond. Inflamm. Regen. 2020, 40. [Google Scholar] [CrossRef] [PubMed]
- Renema, N.; Navet, B.; Heymann, M.-F.; Lezot, F.; Heymann, D. RANK–RANKL Signalling in Cancer. Biosci. Rep. 2016, 36. [Google Scholar] [CrossRef]
- Ahern, E.; Smyth, M.J.; Dougall, W.C.; Teng, M.W.L. Roles of the RANKL–RANK Axis in Antitumour Immunity—Implications for Therapy. Nat. Rev. Clin. Oncol. 2018, 15, 676–693. [Google Scholar] [CrossRef] [PubMed]
- de Groot, A.F.; Appelman-Dijkstra, N.M.; van der Burg, S.H.; Kroep, J.R. The Anti-Tumor Effect of RANKL Inhibition in Malignant Solid Tumors–A Systematic Review. Cancer Treat. Rev. 2018, 62, 18–28. [Google Scholar] [CrossRef]
- van Dam, P.A.; Verhoeven, Y.; Jacobs, J.; Wouters, A.; Tjalma, W.; Lardon, F.; Van den Wyngaert, T.; Dewulf, J.; Smits, E.; Colpaert, C.; et al. RANK-RANKL Signaling in Cancer of the Uterine Cervix: A Review. Int. J. Mol. Sci. 2019, 20, 2183. [Google Scholar] [CrossRef]
- Meuten, D.J.; Moore, F.M.; George, J.W. Mitotic Count and the Field of View Area: Time to Standardize. Vet. Pathol. 2016, 53, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Bacci, B.; Stent, A.W.; Walmsley, E.A. Equine Intestinal Lymphoma: Clinical-Pathological Features, Immunophenotype, and Survival. Vet. Pathol 2020, 57, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Porcellato, I.; Modesto, P.; Cappelli, K.; Varello, K.; Peletto, S.; Brachelente, C.; Martini, I.; Mechelli, L.; Ferrari, A.; Ghelardi, A.; et al. Equus Caballus Papillomavirus Type 2 (EcPV2) in Co-Occurring Vulvar and Gastric Lesions of a Pony. Res. Vet. Sci. 2020, 132, 167–171. [Google Scholar] [CrossRef]
- Cappelli, K.; Felicetti, M.; Capomaccio, S.; Nocelli, C.; Silvestrelli, M.; Verini-Supplizi, A. Effect of Training Status on Immune Defence Related Gene Expression in Thoroughbred: Are Genes Ready for the Sprint? Vet. J. 2013, 195, 373–376. [Google Scholar] [CrossRef]
- Brachelente, C.; Cappelli, K.; Capomaccio, S.; Porcellato, I.; Silvestri, S.; Bongiovanni, L.; Maria, R.D.; Supplizi, A.V.; Mechelli, L.; Sforna, M. Transcriptome Analysis of Canine Cutaneous Melanoma and Melanocytoma Reveals a Modulation of Genes Regulating Extracellular Matrix Metabolism and Cell Cycle. Sci. Rep. 2017, 7, 6386. [Google Scholar] [CrossRef]
- Porcellato, I.; Brachelente, C.; Cappelli, K.; Menchetti, L.; Silvestri, S.; Sforna, M.; Mecocci, S.; Iussich, S.; Leonardi, L.; Mechelli, L. FoxP3, CTLA-4, and IDO in Canine Melanocytic Tumors. Vet. Pathol. 2021, 58, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Bonnet, A.; Willis, C.; Pittaway, R.; Smith, K.; Mair, T.; Priestnall, S.L. Molecular Carcinogenesis in Equine Penile Cancer: A Potential Animal Model for Human Penile Cancer. Urol. Oncol. Semin. Orig. Investig. 2018, 36, 532.e9–532.e18. [Google Scholar] [CrossRef]
- Alloway, E.; Linder, K.; May, S.; Rose, T.; DeLay, J.; Bender, S.; Tucker, A.; Luff, J. A Subset of Equine Gastric Squamous Cell Carcinomas Is Associated with Equus Caballus Papillomavirus-2. Vet. Pathol. 2020, 57, 427–431. [Google Scholar] [CrossRef]
- Hibi, H.; Hatama, S.; Obata, A.; Shibahara, T.; Kadota, K. Laryngeal Squamous Cell Carcinoma and Papilloma Associated with Equus Caballus Papillomavirus 2 in a Horse. J. Vet. Med. Sci. 2019, 81, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Massa, C.; Seliger, B. The Tumor Microenvironment: Thousand Obstacles for Effector T Cells. Cell. Immunol. 2019, 343, 103730. [Google Scholar] [CrossRef]
- Smola, S. Immunopathogenesis of HPV-Associated Cancers and Prospects for Immunotherapy. Viruses 2017, 9, 254. [Google Scholar] [CrossRef]
- Lange, C.E.; Tobler, K.; Lehner, A.; Grest, P.; Welle, M.M.; Schwarzwald, C.C.; Favrot, C. EcPV2 DNA in Equine Papillomas and In Situ and Invasive Squamous Cell Carcinomas Supports Papillomavirus Etiology. Vet. Pathol. 2013, 50, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J. Host Control of Human Papillomavirus Infection and Disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 47, 27–41. [Google Scholar] [CrossRef]
- van den Top, J.G.B.; Harkema, L.; Lange, C.; Ensink, J.M.; van de Lest, C.H.A.; Barneveld, A.; van Weeren, P.R.; Gröne, A.; Martens, A. Expression of P53, Ki67, EcPV2- and EcPV3 DNA, and Viral Genes in Relation to Metastasis and Outcome in Equine Penile and Preputial Squamous Cell Carcinoma. Equine Vet. J. 2015, 47, 188–195. [Google Scholar] [CrossRef] [PubMed]
- de Sanjosé, S.; Alemany, L.; Ordi, J.; Tous, S.; Alejo, M.; Bigby, S.M.; Joura, E.A.; Maldonado, P.; Laco, J.; Bravo, I.G.; et al. Worldwide Human Papillomavirus Genotype Attribution in over 2000 Cases of Intraepithelial and Invasive Lesions of the Vulva. Eur. J. Cancer 2013, 49, 3450–3461. [Google Scholar] [CrossRef]
- Boda, D.; Docea, A.O.; Calina, D.; Ilie, M.A.; Caruntu, C.; Zurac, S.; Neagu, M.; Constantin, C.; Branisteanu, D.E.; Voiculescu, V.; et al. Human Papilloma Virus: Apprehending the Link with Carcinogenesis and Unveiling New Research Avenues (Review). Int. J. Oncol. 2018, 52, 637–655. [Google Scholar] [CrossRef]
- Burt, L.M.; Shrieve, D.C.; Tward, J.D. Stage Presentation, Care Patterns, and Treatment Outcomes for Squamous Cell Carcinoma of the Penis. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Ueda, Y.; Enomoto, T.; Kimura, T.; Yoshino, K.; Fujita, M.; Kimura, T. Two Distinct Pathways to Development of Squamous Cell Carcinoma of the Vulva. J. Skin Cancer 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Teh, J.; O’Connor, E.; O’Brien, J.; Lim, W.M.; Taylor, M.; Heriot, A.; Ramsay, R.; Lawrentschuk, N. Future Directions in Advanced Penile Cancer–Mechanisms of Carcinogenesis and a Search for Targeted Therapy. Future Oncol. 2020, 16, 2357–2369. [Google Scholar] [CrossRef] [PubMed]
- Yeo-Teh, N.S.L.; Ito, Y.; Jha, S. High-Risk Human Papillomaviral Oncogenes E6 and E7 Target Key Cellular Pathways to Achieve Oncogenesis. Int. J. Mol. Sci. 2018, 19, 1706. [Google Scholar] [CrossRef]
- Stankiewicz, E.; Ng, M.; Cuzick, J.; Mesher, D.; Watkin, N.; Lam, W.; Corbishley, C.; Berney, D.M. The Prognostic Value of Ki-67 Expression in Penile Squamous Cell Carcinoma. J. Clin. Pathol. 2012, 65, 534–537. [Google Scholar] [CrossRef]
- Silva, D.C.; Gonçalves, A.K.; Cobucci, R.N.; Mendonça, R.C.; Lima, P.H.; Cavalcanti, G. Immunohistochemical Expression of P16, Ki-67 and P53 in Cervical Lesions–A Systematic Review. Pathol. Res. Pract. 2017, 213, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Ramsauer, A.S.; Wachoski-Dark, G.L.; Fraefel, C.; Tobler, K.; Brandt, S.; Knight, C.G.; Favrot, C.; Grest, P. Paving the Way for More Precise Diagnosis of EcPV2-Associated Equine Penile Lesions. BMC Vet. Res. 2019, 15. [Google Scholar] [CrossRef]
- Palafox, M.; Ferrer, I.; Pellegrini, P.; Vila, S.; Hernandez-Ortega, S.; Urruticoechea, A.; Climent, F.; Soler, M.T.; Muñoz, P.; Viñals, F.; et al. RANK Induces Epithelial–Mesenchymal Transition and Stemness in Human Mammary Epithelial Cells and Promotes Tumorigenesis and Metastasis. Cancer Res. 2012, 72, 2879–2888. [Google Scholar] [CrossRef]
- González-Suárez, E.; Sanz-Moreno, A. RANK as a Therapeutic Target in Cancer. FEBS J. 2016, 283, 2018–2033. [Google Scholar] [CrossRef] [PubMed]
- Barger, A.M.; Fan, T.M.; de Lorimier, L.-P.; Sprandel, I.T.; O’Dell-Anderson, K. Expression of Receptor Activator of Nuclear Factor K-B Ligand (RANKL) in Neoplasms of Dogs and Cats. J. Vet. Intern. Med. 2007, 21, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The Hallmarks of Cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Veeck, J.; Niederacher, D.; An, H.; Klopocki, E.; Wiesmann, F.; Betz, B.; Galm, O.; Camara, O.; Dürst, M.; Kristiansen, G.; et al. Aberrant Methylation of the Wnt Antagonist SFRP1 in Breast Cancer Is Associated with Unfavourable Prognosis. Oncogene 2006, 25, 3479–3488. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Zhou, X.; Liu, Y.; Tang, Z. Mutation and Overexpression of the β-Catenin Gene May Play an Important Role in Primary Hepatocellular Carcinoma among Chinese People. J. Cancer Res. Clin. Oncol. 2001, 127, 577–581. [Google Scholar] [CrossRef]
- Hofmann, B.M.; Buchfelder, M.; Buslei, R. Nuclear B-Catenin Accumulation as Reliable Marker for the Differentiation Between Cystic Craniopharyngiomas and Rathke Cleft Cysts: A Clinico-Pathologic Approach. Am. J. Surg. Pathol. 2006, 30, 9. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.; Germinaro, M.; Micsenyi, A.; Monga, N.K.; Bell, A.; Sood, A.; Malhotra, V.; Sood, N.; Midda, V.; Monga, D.K.; et al. Aberrant Wnt/β-Catenin Signaling in Pancreatic Adenocarcinoma. Neoplasia 2006, 8, 279–289. [Google Scholar] [CrossRef]
- Thorfve, A.; Dehne, T.; Lindahl, A.; Brittberg, M.; Pruss, A.; Ringe, J.; Sittinger, M.; Karlsson, C. Characteristic Markers of the WNT Signaling Pathways Are Differentially Expressed in Osteoarthritic Cartilage. Cartilage 2012, 3, 43–57. [Google Scholar] [CrossRef]
- Liu, X.; Mazanek, P.; Dam, V.; Wang, Q.; Zhao, H.; Guo, R.; Jagannathan, J.; Cnaan, A.; Maris, J. Deregulated Wnt/b-Catenin Program in High-Risk Neuroblastomas without MYCN Amplification. Oncogene 2008, 27, 1478–1488. [Google Scholar] [CrossRef]
- Mann, B.; Gelos, M.; Siedow, A.; Hanski, M.L.; Gratchev, A.; Ilyas, M.; Bodmer, W.F.; Moyer, M.P.; Riecken, E.O.; Buhr, H.J.; et al. Target Genes of -Catenin-T Cell-Factor/Lymphoid-Enhancer-Factor Signaling in Human Colorectal Carcinomas. Proc. Natl. Acad. Sci. USA 1999, 96, 1603–1608. [Google Scholar] [CrossRef]
- Katoh, M. WNT/PCP Signaling Pathway and Human Cancer (Review). Oncol. Rep. 2005. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, Z.; Tang, Y.; Xiao, Q. The Involvement of Noncanonical Wnt Signaling in Cancers. Biomed. Pharmacother. 2021, 133, 110946. [Google Scholar] [CrossRef]
- Liu, J.; Huang, B.; Xiu, Z.; Zhou, Z.; Liu, J.; Li, X.; Tang, X. PI3K/Akt/HIF-1α Signaling Pathway Mediates HPV-16 Oncoprotein-Induced Expression of EMT-Related Transcription Factors in Non-Small Cell Lung Cancer Cells. J. Cancer 2018, 9, 3456–3466. [Google Scholar] [CrossRef]
- Gaffen, S.L. Structure and Signalling in the IL-17 Receptor Family. Nat. Rev. Immunol. 2009, 9, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Kireva, T.; Erhardt, A.; Tiegs, G.; Tilg, H.; Denk, H.; Haybaeck, J.; Aigner, E.; Moschen, A.; Distler, J.H.; Schett, G.; et al. Transcription Factor Fra-1 Induces Cholangitis and Liver Fibrosis. Hepatology 2011, 53, 1287–1297. [Google Scholar] [CrossRef]
- Xue, J.; Wang, Y.; Chen, C.; Zhu, X.; Zhu, H.; Hu, Y. Effects of Th17 Cells and IL-17 in the Progression of Cervical Carcinogenesis with High-risk Human Papillomavirus Infection. Cancer Med. 2017, 7, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, Y. The IL-23/IL-17 Axis in Inflammation. J. Clin. Investig. 2006, 116, 1218–1222. [Google Scholar] [CrossRef]
- Ma, W.-T.; Yao, X.-T.; Peng, Q.; Chen, D.-K. The Protective and Pathogenic Roles of IL-17 in Viral Infections: Friend or Foe? Open Biol. 2019, 9, 190109. [Google Scholar] [CrossRef]
| Gene | Sequences | Amplicon Length | Accession Number |
|---|---|---|---|
| EcPV2-E2 | F-5′-AAAAGGGAGGGTACGTTGTC-3′ R-5′-CCTGGTAGTAGACATGCTGC-3′ | 90 | NC_012123.1:2767–4017 |
| p-EcPV2-E2 | FAM- GCCAAGACAGCCACGACGCCAT-TAMRA | 22 | |
| EcPV2-E6 | F-5′-CGTTGGCCTTCTTTGCATCT-3′ R-5′-AGGTTCAGGTCTGCTGTGTT-3′ | 81 | NC_012123.1:5–622 |
| p-EcPV2-E6 | FAM- CCGTGTGGCTATGCTGATGACATTTGG -TAMRA | 27 | |
| B2M DNA detection | F-5′-CTGATGTTCTCCAGGTGTTCC-3′ R-5′-TCAATCTCAGGCGGATGGAA-3′ | 114 | NM_001082502.3 chr1:145961271-145961384 |
| B2M cDNA expression | F-5′-GGCTACTCTCCCTGACTGG-3′ R-5′- TCAATCTCAGGCGGATGGAA-3′ | 136 | NM_001082502.3 chr1:145961271-145964672 |
| p-B2M | FAM-ACTCACGTCACCCAGCAGAGA-TAMRA | 21 | NM_001082502.3 |
| Gene | Primer Pairs Sequences | Amplicon Length | Accession Number |
|---|---|---|---|
| B2M | F-5′-GGCTACTCTCCCTGACTGG-3′ R-5′-TCAATCTCAGGCGGATGGAA-3′ | 136 | NM_001082502.3 |
| RANKL | F-5′-AGCCTGACACTCAACCTTTTG-3′ R-5′-CCAGGAAGACAGACTCACTTTG-3′ | 86 | XM_014732051.2 |
| NFKBp50 | F-5′-CCAGCTTTTGGTAGATGTGCTG-3′ R-5′-TCGTCTTCTGCCATTCTGGA-3′ | 102 | XM_023637631.1 |
| NFKBp65 | F-5′-GAGCCCATGGAGTTCCAGTA-3′ R-5′-AGGTCTCATATGTCCTTTTGCGT-3′ | 82 | XM_023654462.1 |
| IL8 | F-5′-CTGGCTGTGGCTCTCTTG-3′ R-5′-CAGTTTGGGATTGAAAGGTTTG-3′ | 133 | NM_001083951.2 |
| IL17 | F-5′-ACAACCGCTCCACCTCCC-3′ R-5′-CCTTCGGCATTGACACAGC-3′ | 112 | NM_001143792.1 |
| IL23p19 | F-5′-CTGTACGCTGGCCTGGAG-3′ R-5′-GTGGATCCTTTGCAAGCAGG-3′ | 167 | NM_001082522.2 |
| IL6 | F-5′-TCAAGGGTGAAAAGGAAAACATC-3′ R-5′-GGTGGTTACTTCTGGATTCTTC-3′ | 98 | NM_001082496.2 |
| IL12p35 | F-5′-CTGAGGACCGTCAGCAACAC-3′ R-5′-GTTCGGGGCGAGTTCCAG-3′ | 147 | NM_001082511.2 |
| IL12p40 | F-5′-GATCGTGGTGGATGCTGTTC-3′ R-5′-TCCACCTGCCGAGAATTCTT-3′ | 132 | NM_001082516.1 |
| BCATN1 | F-5′-CCTCTTCAGAACGGAGCCAA-3′ R-5′-CTGGCGATATCCAAGGGGTT-3′ | 91 | XM_023619816.1 |
| FOSL1 | F-5′-TACCGAGACTTCGGGGAAC-3′ R-5′-GCGTTGATACTTGGCACGAG-3′ | 116 | XM_001494776.4 |
| LEF1 | F-5′-GCCAGACAAGCACAAACCTC-3′ R-5′-GGGTCCCTTGCTGTAGAGG-3′ | 102 | XM_023636760.1 |
| Mitoses | Ki67 | BCATN1 | FOSL1 | IL12p35 | IL12p40 | IL17A | IL23p19 | IL6 | IL8 | LEF1 | NFKB1 | NKFBp65 | RANKL | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mitoses | 1.000 | −0.074 | 0.117 | −0.071 | −0.145 | 0.160 | −0.162 | 0.154 | 0.026 | −0.161 | −0.010 | 0.133 | −0.004 | 0.153 |
| Ki67 | 1.000 | −0.370 | −0.518 | −0.389 | −0.141 | −0.071 | −0.101 | −0.155 | −0.156 | −0.171 | −0.405 | −0.402 | −0.272 | |
| BCATN1 | 1.000 | 0.791 ** | 0.864 ** | 0.657 ** | 0.161 | 0.800 ** | 0.888 ** | 0.371 | 0.890 ** | 0.425 * | 0.486 * | 0.773 ** | ||
| FOSL1 | 1.000 | 0.758 ** | 0.282 | −0.011 | 0.412 | 0.698 ** | 0.469 | 0.610 * | 0.352 | 0.533 | 0.632 * | |||
| IL12p35 | 1.000 | 0.742 ** | 0.235 | 0.748 ** | 0.893 ** | 0.451 * | 0.868 ** | 0.520 * | 0.381 | 0.707 ** | ||||
| IL12p40 | 1.000 | 0.432 | 0.775 ** | 0.809 ** | 0.162 | 0.739 ** | 0.481 * | 0.184 | 0.634 ** | |||||
| IL17A | 1.000 | 0.227 | 0.339 | 0.033 | 0.210 | 0.143 | 0.157 | 0.156 | ||||||
| IL23p19 | 1.000 | 0.881 ** | 0.191 | 0.910 ** | 0.458 * | 0.296 | 0.850 ** | |||||||
| IL6 | 1.000 | 0.429 | 0.922 ** | 0.398 | 0.381 | 0.792 ** | ||||||||
| IL8 | 1.000 | 0.247 | 0.162 | 0.356 | 0.248 | |||||||||
| LEF1 | 1.000 | 0.454 * | 0.267 | 0.856 ** | ||||||||||
| NFKB1 | 1.000 | 0.227 | 0.515 * | |||||||||||
| NFKBp65 | 1.000 | 0.205 |
| Case ID | Histological Diagnosis | Sex | DNA | cDNA | |||
|---|---|---|---|---|---|---|---|
| B2M | E6 | E6 | E2 | E6 Expressing samples | |||
| 1 | SCC | M | + | ++ | 30.4 ± 0.2 | >48 | E6+ |
| 2 | SCC | M | + | + | >48 | >48 | E6− |
| 3 | SCC | M | + | +++ | 32.8 ± 0.3 | >48 | E6+ |
| 4 | CIS | M | + | ++++ | 36.2 ± 0.6 | 44.2 ± 2.4 | E6+ |
| 5 | SCC | F | + | ++++ | 31.5 ± 0.7 | 34.1 ± 0.4 | E6+ |
| 6 | P | M | + | + | >48 | >48 | E6− |
| 7 | SCC | M | + | + | >48 | >48 | E6− |
| 8 | SCC | M | + | +++ | 33.5 ± 0.4 | >48 | E6+ |
| 9 | SCC | M | + | ++++ | 32.7 ± 0.9 | 36.1 ± 0.6 | E6+ |
| 10 | SCC | M | + | +++ | 33.5 ± 0.3 | 39.3 ± 1.1 | E6+ |
| 11 | SCC | M | + | + | >48 | >48 | E6− |
| 12 | SCC | M | + | ++++ | 33.7 ± 0.4 | >48 | E6+ |
| 13 | SCC | M | + | + | >48 | >48 | E6− |
| 14 | SCC | M | + | + | >48 | >48 | E6− |
| 15 | SCC | M | + | ++++ | 33.9 ± 1.9 | 38.5 ± 0.6 | E6+ |
| 16 | SCC | F | + | ++++ | 31.9 ± 0.4 | >48 | E6+ |
| 17 | SCC | M | + | +++ | 35.3 ± 1.5 | 39.2 ± 0.9 | E6+ |
| 18 | SCC | M | + | ++ | >48 | >48 | E6− |
| 19 | SCC | F | + | ++++ | 32.1 ± 1.1 | 35.6 ± 0.7 | E6+ |
| 20 | SCC | M | + | − | ND | ND | E6− |
| 21 | CIS | M | + | ++++ | 30.4 ± 0.6 | >48 | E6+ |
| 22 | SCC | M | + | ++++ | 32.3 ± 0.1 | 41.9 ± 2.6 | E6+ |
| 23 | SCC | M | + | − | ND | ND | E6− |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mecocci, S.; Porcellato, I.; Armando, F.; Mechelli, L.; Brachelente, C.; Pepe, M.; Gialletti, R.; Passeri, B.; Modesto, P.; Ghelardi, A.; et al. Equine Genital Squamous Cell Carcinoma Associated with EcPV2 Infection: RANKL Pathway Correlated to Inflammation and Wnt Signaling Activation. Biology 2021, 10, 244. https://doi.org/10.3390/biology10030244
Mecocci S, Porcellato I, Armando F, Mechelli L, Brachelente C, Pepe M, Gialletti R, Passeri B, Modesto P, Ghelardi A, et al. Equine Genital Squamous Cell Carcinoma Associated with EcPV2 Infection: RANKL Pathway Correlated to Inflammation and Wnt Signaling Activation. Biology. 2021; 10(3):244. https://doi.org/10.3390/biology10030244
Chicago/Turabian StyleMecocci, Samanta, Ilaria Porcellato, Federico Armando, Luca Mechelli, Chiara Brachelente, Marco Pepe, Rodolfo Gialletti, Benedetta Passeri, Paola Modesto, Alessandro Ghelardi, and et al. 2021. "Equine Genital Squamous Cell Carcinoma Associated with EcPV2 Infection: RANKL Pathway Correlated to Inflammation and Wnt Signaling Activation" Biology 10, no. 3: 244. https://doi.org/10.3390/biology10030244
APA StyleMecocci, S., Porcellato, I., Armando, F., Mechelli, L., Brachelente, C., Pepe, M., Gialletti, R., Passeri, B., Modesto, P., Ghelardi, A., Cappelli, K., & Razzuoli, E. (2021). Equine Genital Squamous Cell Carcinoma Associated with EcPV2 Infection: RANKL Pathway Correlated to Inflammation and Wnt Signaling Activation. Biology, 10(3), 244. https://doi.org/10.3390/biology10030244










