Age-Related Palatal Wound Healing: An Experimental In Vivo Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Preparation of Experimental Model
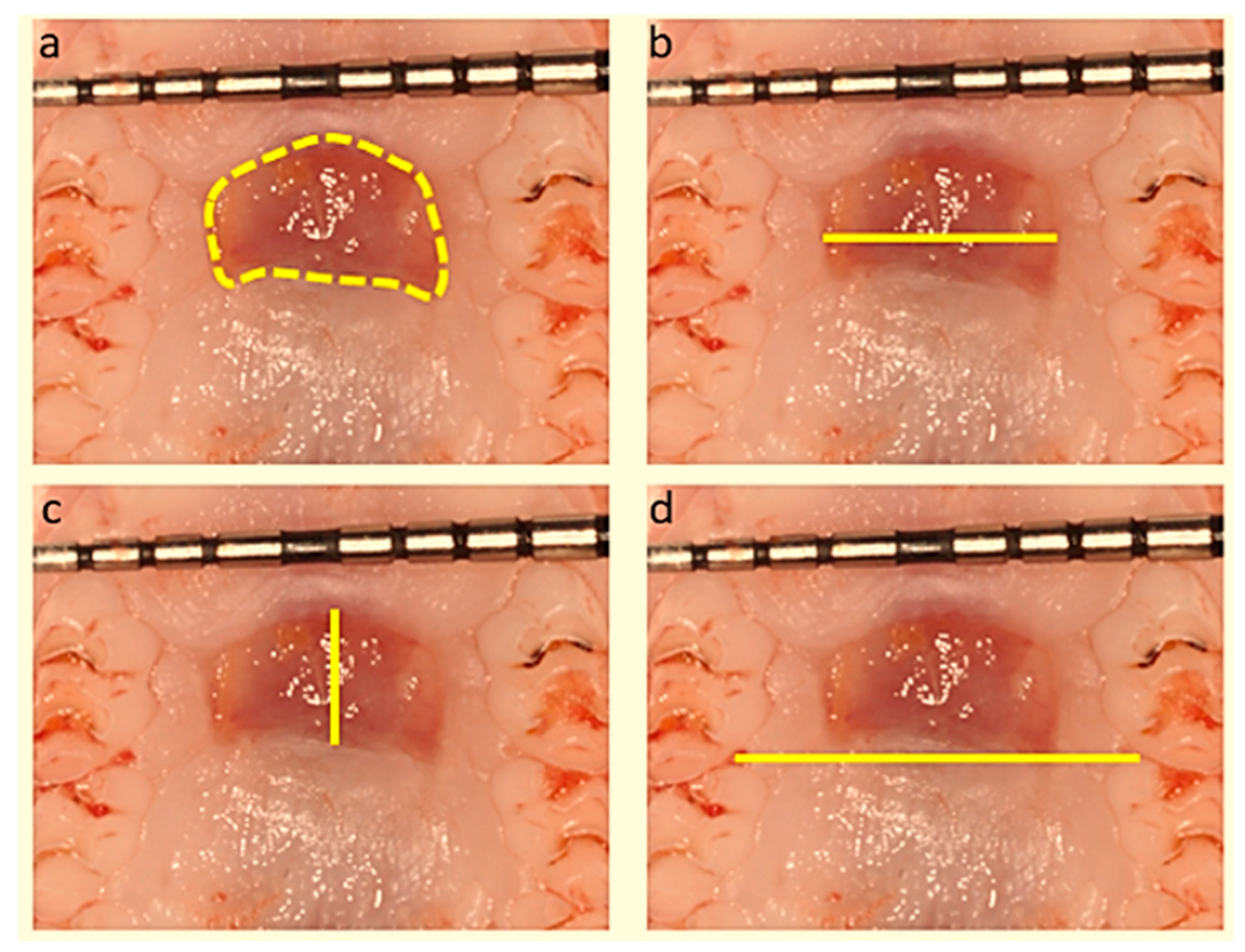
2.2. Macroscopic Evaluation
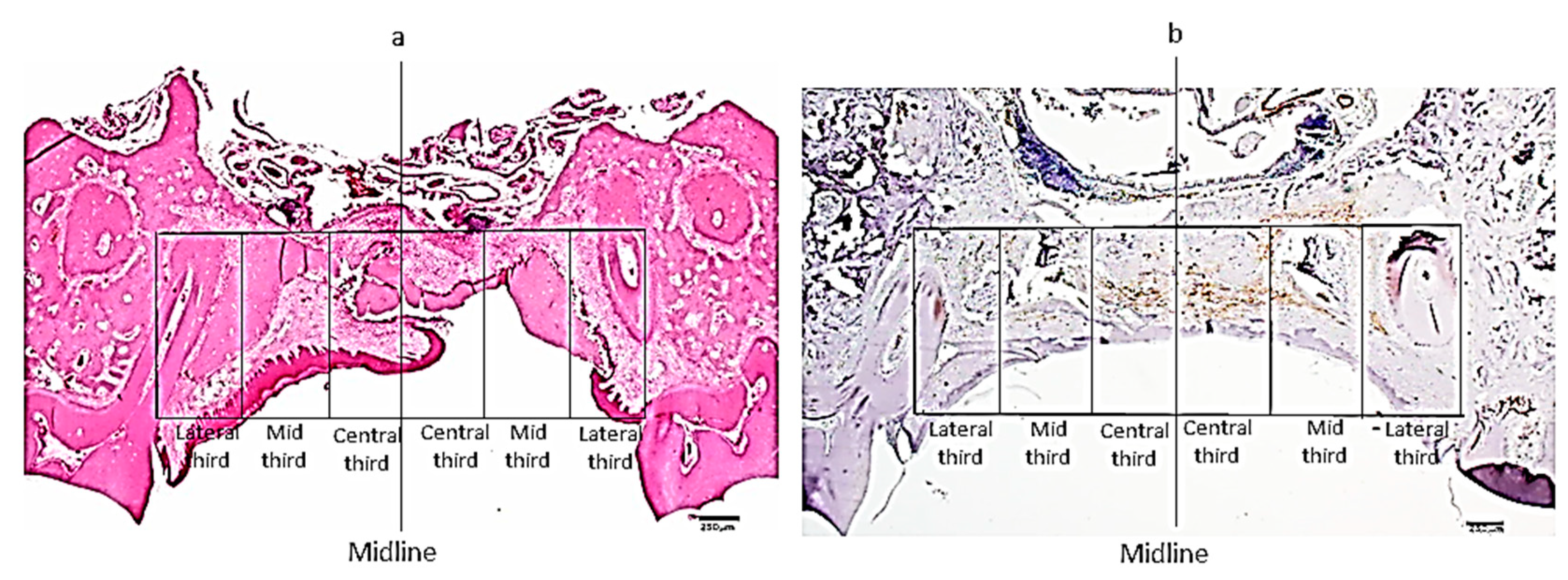
2.3. Microscopic Examination
2.4. Immunohistochemistry for Identification of Myofibroblasts
2.5. Histomorphometry for Assessment of the Intensity of Inflammation and Density of Myofibroblasts
2.6. Statistical Analysis
3. Results
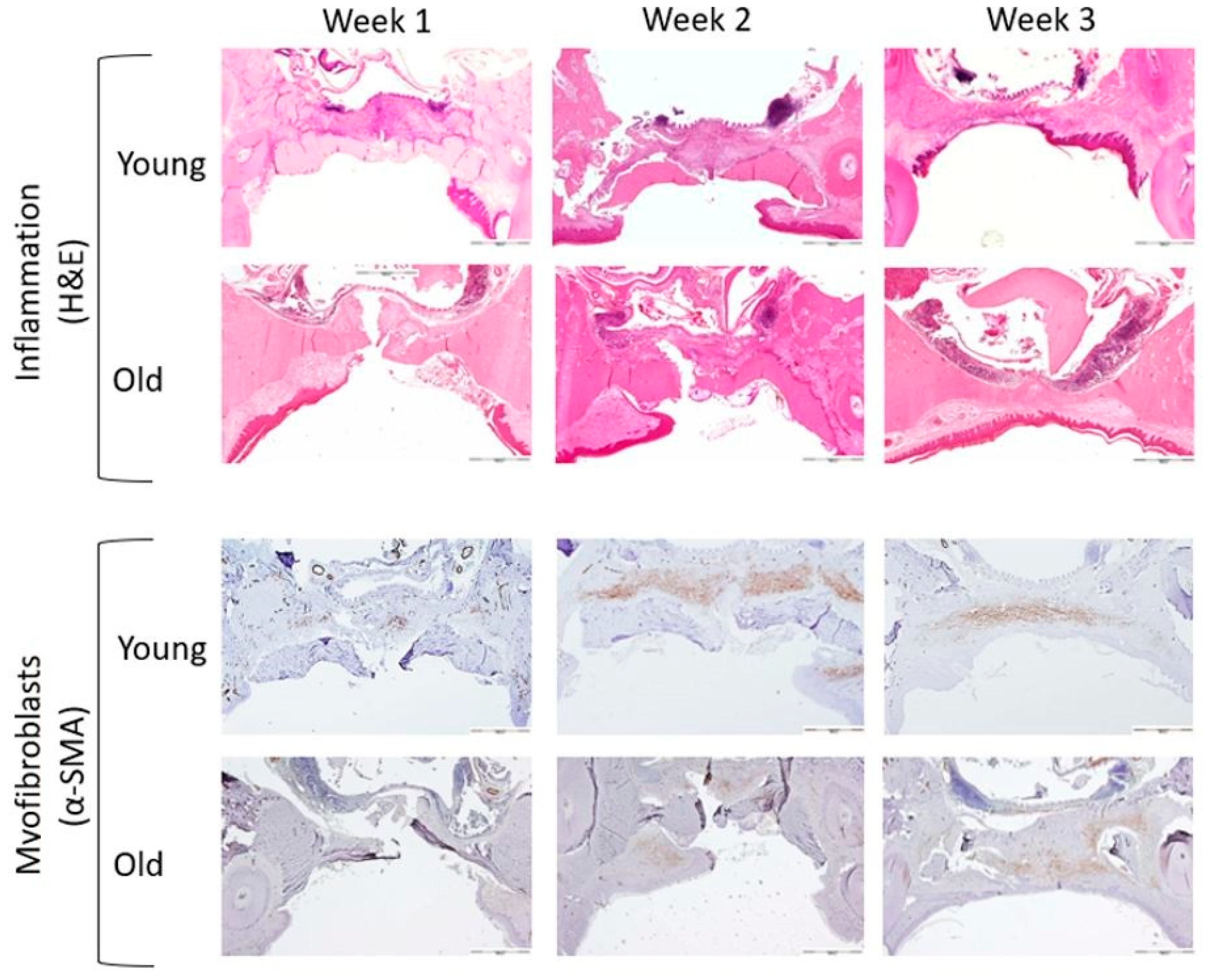
3.1. Microscopic Evaluation
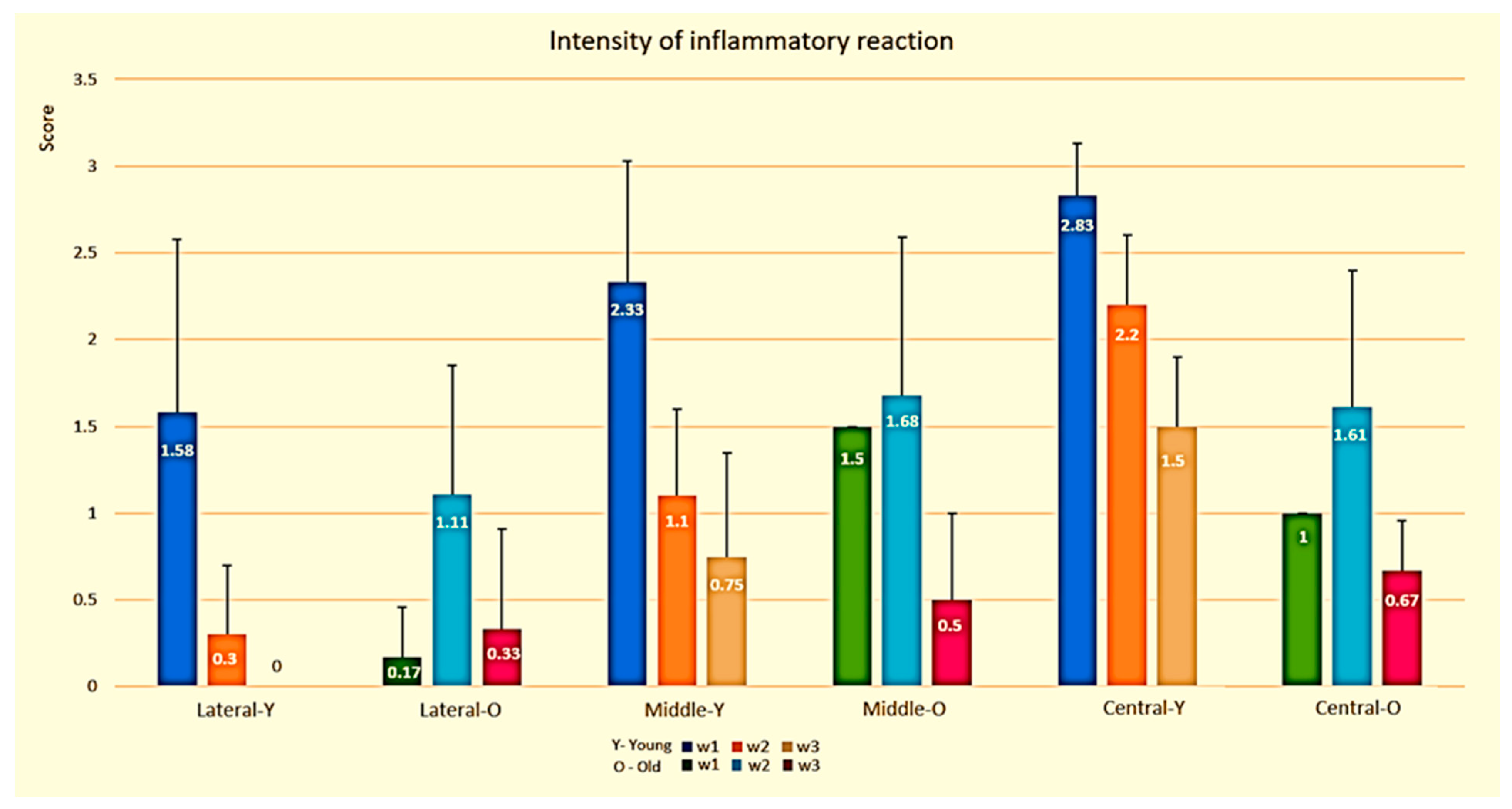
3.1.1. Semi-Quantitative Evaluation of Inflammatory Phase
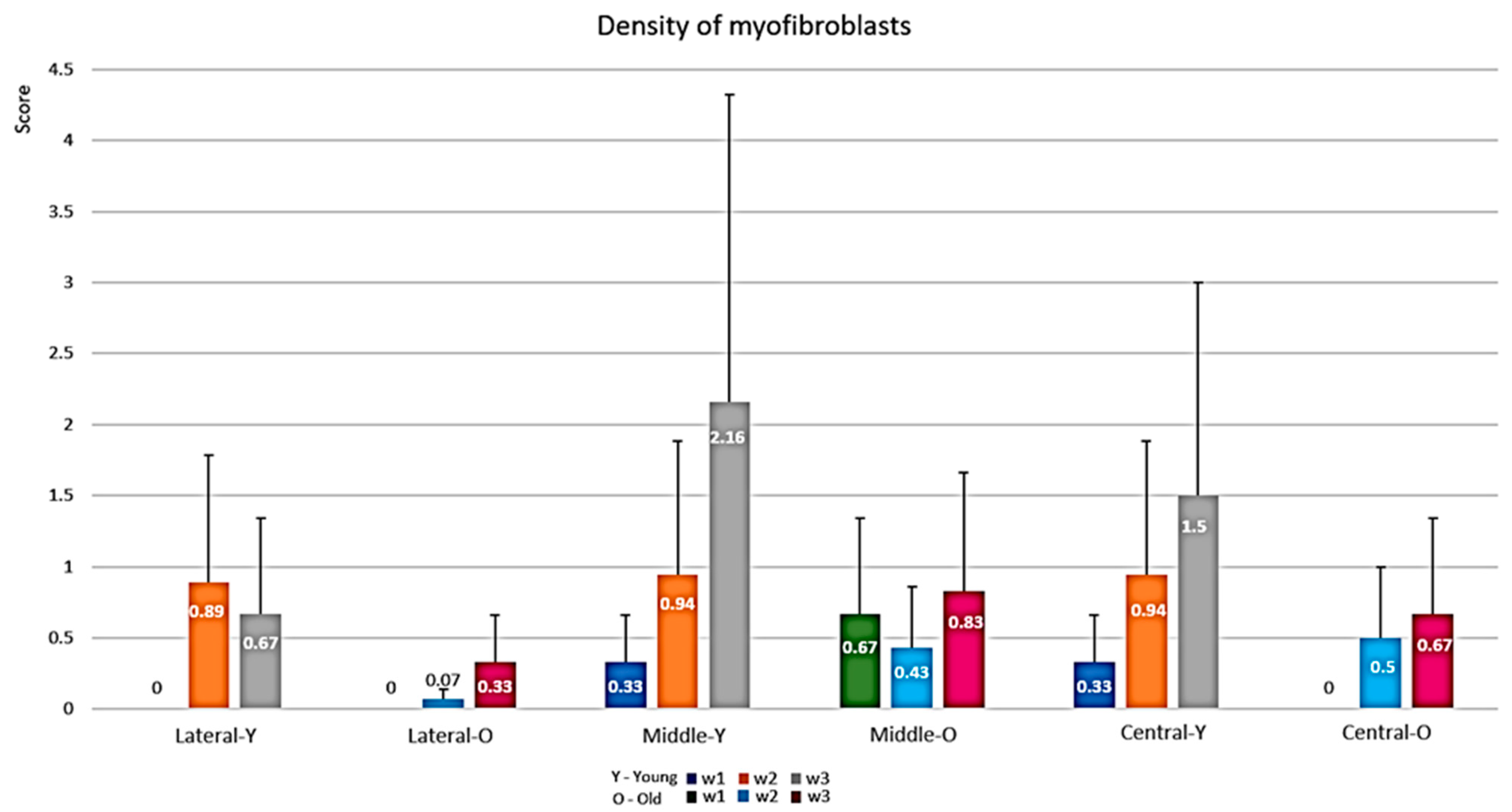
3.1.2. Semi-Quantitative Evaluation of the Proliferative Phase (Myofibroblasts)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deo, S.D.; Shetty, S.K.; Kulloli, A.; Chavan, R.; Dholakia, P.; Ligade, S.; Dharmarajan, G. Efficacy of free gingival graft in the treatment of Miller Class I and Class II localized gingival recessions: A systematic review. J. Indian Soc. Periodontol. 2019, 23, 93–99. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Nakajima, T. Tracheoplasty with palatal mucoperiosteal graft. Plast. Reconstr. Surg. 1990, 86, 558–562. [Google Scholar] [CrossRef]
- Aydogmus, U.; Topkara, A.; Akbulut, M.; Ozkan, A.; Turk, F.; Sahin, B.; Yuncu, G. Effectiveness of palatal mucosa graft in surgical treatment of sub-glottic stenosis. Clin. Exp. Otorhinolaryngol. 2016, 9, 358–365. [Google Scholar] [CrossRef][Green Version]
- Yoshimura, Y.; Nakajima, T.; Yoneda, K. Use of the palatal mucosal graft for reconstruction of the eye socket. J. Cranio-Maxillofac. Surg. 1995, 23, 27–30. [Google Scholar] [CrossRef]
- Hatoko, M.; Kuwahara, M.; Tanaka, A.; Tada, H.; Muramatsu, T. Correction of a posttraumatic nasal deformity using a hard palate mucosa graft. Aesthetic. Plast. Surg. 2000, 24, 34–38. [Google Scholar] [CrossRef]
- Fernández-Mejía, S.; Domínguez-Cherit, J.; Pichardo-Velázquez, P.; González-Olvera, S. Treatment of nail bed defects with hard palate mucosal grafts. J. Cutan. Med. Surg. 2006, 10, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Reiser, G.M.; Bruno, J.F.; Mahan, P.E.; Larkin, L.H. The subepithelial connective tissue graft palatal donor site: Anatomic considerations for surgeons. Int. J. Periodontics Restor. Dent. 1996, 16, 130–137. [Google Scholar]
- Studer, S.P.; Allen, E.P.; Rees, T.C.; Kouba, A. The thickness of masticatory mucosa in the human hard palate and tuberosity as potential donor sites for ridge augmentation procedures. J. Periodontol. 1997, 68, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Maino, G.N.E.; Valles, C.; Santos, A.; Pascual, A.; Esquinas, C.; Nart, J. Influence of suturing technique on wound healing and patient morbidity after connective tissue harvesting. A randomized clinical trial. J. Clin. Periodontol. 2018, 45, 977–985. [Google Scholar] [CrossRef]
- Farnoush, A. Techniques for the protection and coverage of the donor sites in free soft tissue grafts. J. Periodontol. 1978, 49, 403–405. [Google Scholar] [CrossRef]
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Hämmerle, C.H.F.; Giannobile, W.V. Biology of soft tissue wound healing and regeneration—Consensus report of group 1 of the 10th European workshop on periodontology. J. Clin. Periodontol. 2014, 41, S1–S5. [Google Scholar] [CrossRef] [PubMed]
- Kahnberg, K.E.; Thilander, H. Healing of experimental excisional wounds in the rat palate. (I) Histological study of the interphase in wound healing after sharp dissection. Int. J. Oral Surg. 1982, 11, 44–51. [Google Scholar] [CrossRef]
- Bodner, L.; Dayan, D.; Oberman, M.; Hirshberg, A.; Tal, H. Healing of experimental wounds in sialadenectomized rat. J. Clin. Periodontol. 1992, 19, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Bodner, L.; Dayan, D.; Pinto, Y.; Hammel, I. Characteristics of palatal wound healing in desalivated rats. Arch. Oral Biol. 1993, 38, 17–21. [Google Scholar] [CrossRef]
- Bodner, L.; Dayan, D. Epithelium and connective tissue regeneration during palatal wound healing in desalivated rats--a comparative study. Comp. Biochem. Physiol. A Physiol. 1995, 111, 415–419. [Google Scholar] [CrossRef]
- Smith, P.C.; Cáceres, M.; Martínez, C.; Oyarzún, A.; Martínez, J. Gingival wound healing: An essential response disturbed by aging? J. Dent. Res. 2015, 94, 395–402. [Google Scholar] [CrossRef]
- Engeland, C.G.; Sabzehei, B.; Marucha, P.T. Sex hormones and mucosal wound healing. Brain. Behav. Immun. 2009, 23, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Lamster, I.B.; Asadourian, L.; Del Carmen, T.; Friedman, P.K. The aging mouth: Differentiating normal aging from disease. Periodontol. 2000 2016, 72, 96–107. [Google Scholar] [CrossRef]
- Cáceres, M.; Oyarzun, A.; Smith, P.C. Defective wound-healing in aging gingival tissue. J. Dent. Res. 2014, 93, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. Osteoarthr. Cart. 2012, 20, 256–260. [Google Scholar] [CrossRef]
- Weinberg, E.; Vered, M.; Atzil, S.; Chaushu, G.; Chaushu, L. The dynamics of closure following excisional mid-palatal mucoperiosteal wound in a rat model. Clin. Oral Investig. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kozlovsky, A.; Artzi, Z.; Hirshberg, A.; Israeli-Tobias, C.; Reich, L. Effect of local antimicrobial agents on excisional palatal wound healing: A clinical and histomorphometric study in rats. J. Clin. Periodontol. 2007, 34, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Oda, Y.; Kagami, H.; Ueda, M. Accelerating effects of basic fibroblast growth factor on wound healing of rat palatal mucosa. J. Oral Maxillofac. Surg. 2004, 62, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Dayan, D.; Salo, T.; Salo, S.; Nyberg, P.; Nurmenniemi, S.; Costea, D.E.; Vered, M. Molecular crosstalk between cancer cells and tumor microenvironment components suggests potential targets for new therapeutic approaches in mobile tongue cancer. Cancer Med. 2012, 1, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Lorenzana, E.R.; Allen, E.P. The single-incision palatal harvest technique: A strategy for esthetics and patient comfort. Int. J. Periodontics Restor. Dent. 2000, 20, 297–305. [Google Scholar]
- Sulijaya, B.; Takahashi, N.; Yamazaki, K. Host modulation therapy using anti-inflammatory and antioxidant agents in periodontitis: A review to a clinical translation. Arch. Oral Biol. 2019, 105, 72–80. [Google Scholar] [CrossRef]
- Socransky, S.S.; Haffajee, A.D. The bacterial etiology of destructive periodontal disease: Current concepts. J. Periodontol. 1992, 63, 322–331. [Google Scholar] [CrossRef]
- Bartold, P.M.; Van Dyke, T.E. Host modulation: Controlling the inflammation to control the infection. Periodontol. 2000 2017, 75, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Sima, C.; Glogauer, M. Neutrophil dysfunction and host susceptibility to periodontal inflammation: Current state of knowledge. Curr. Oral Health Rep. 2014, 2014, 95–103. [Google Scholar] [CrossRef]
- Preshaw, P.M. Host modulation therapy with anti-inflammatory agents. Periodontol. 2000 2018, 76, 131–149. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, A.M.; Maltha, J.C.; Von den Hoff, H.W.; Kuijpers-Jagtman, A.M. Palatal mucoperiosteal wound healing in the rat. Eur. J. Oral Sci. 1999, 107, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Keskiner, I.; Aydogdu, A.; Balli, U.; Kaleli, A.E. Quantitative changes in palatal donor site thickness after free gingival graft harvesting: A pilot study. J. Clin. Periodontol. 2016, 43, 976–984. [Google Scholar] [CrossRef] [PubMed]


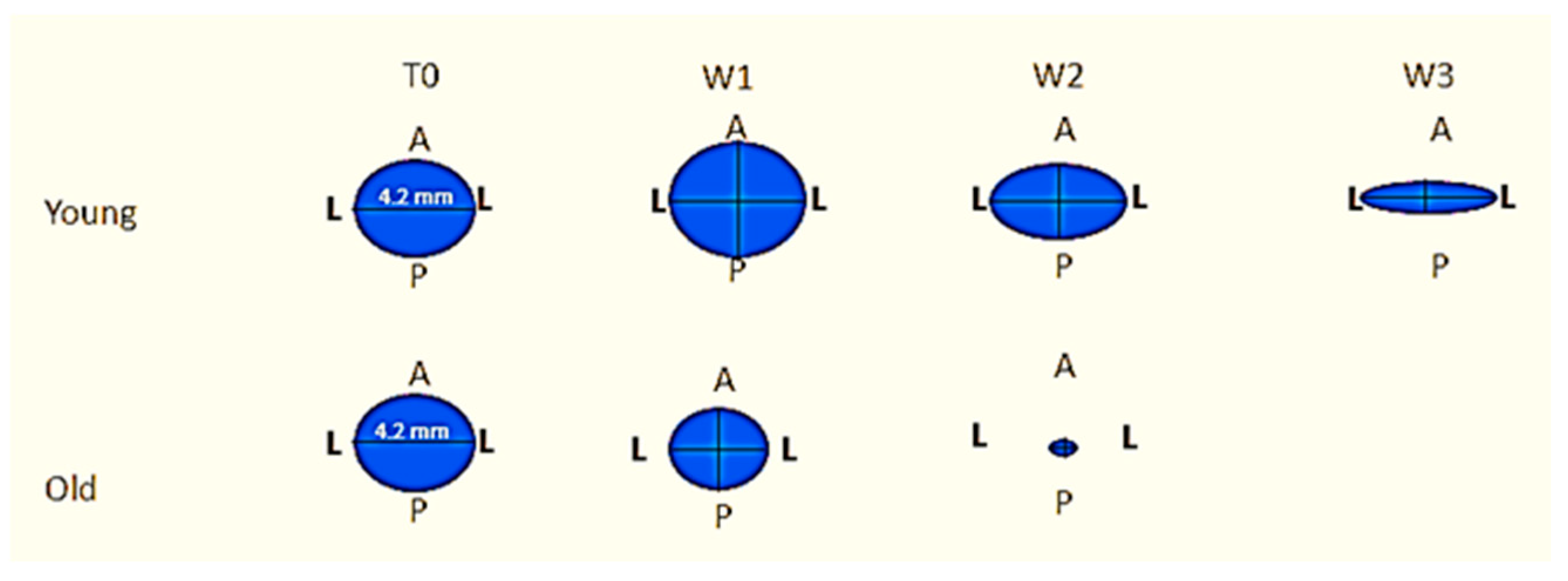
| Measured Parameter | Young (n = 21) | Old (n = 21) | p | Finteraction | pinteraction |
|---|---|---|---|---|---|
| L-L distance (mm) | 5.31 | <0.01 | |||
| Week 1 | 4.03 ± 0.87 | 2.73 ± 0.08 | 0.03 | ||
| Week 2 | 3.16 ± 0.82 | 1.96 ± 0.59 | <0.001 | ||
| Week 3 | 3.18 ± 0.92 | 0.00 ± 0.00 | <0.001 | ||
| A-P distance (mm) | 1.39 | 0.26 | |||
| Week 1 | 4.66 ± 2.65 | 2.68 ± 0.20 | <0.001 | ||
| Week 2 | 3.12 ± 0.75 | 1.90 ± 0.68 | <0.001 | ||
| Week 3 | 1.83 ± 0.45 | 0.00 ± 0.00 | <0.001 | ||
| Total area (mm2) | 1.84 | 0.17 | |||
| Week 1 | 14.71 ± 4.01 | 6.13 ± 1.06 | <0.001 | ||
| Week 2 | 7.96 ± 3.96 | 3.80 ± 2.13 | <0.001 | ||
| Week 3 | 4.89 ± 1.60 | 0.00 ± 0.00 | <0.001 |
| Measured Parameter | Young (n = 21) | Old (n = 21) | p | Finteraction | pinteraction |
|---|---|---|---|---|---|
| L-L fraction relative to W0 (%) | 5.99 | <0.01 | |||
| Week 1 | 91.00 ± 16.70 | 65.33 ± 2.08 | 0.04 | ||
| Week 2 | 66.00 ± 27.55 | 46.73 ± 14.26 | 0.03 | ||
| Week 3 | 75.50 ± 19.53 | 0.00 ± 0.01 | <0.001 | ||
| A-P fraction relative to W0 (%) | 4.83 | 0.04 | |||
| Week 1 | 106.25 ± 16.50 | 64.67 ± 5.16 | 0.002 | ||
| Week 2 | 65.20 ± 27.51 | 45.27 ± 16.36 | 0.032 | ||
| Week 3 | 43.75 ± 8.73 | 0.00 ± 0.01 | <0.001 | ||
| Total area fraction relative to W0 (%) | 3.47 | 0.02 | |||
| Week 1 | 97.65 ± 29.64 | 51.67 ± 8.60 | 0.030 | ||
| Week 2 | 53.62 ± 21.91 | 35.53 ± 20.87 | 0.025 | ||
| Week 3 | 36.00 ± 13.14 | 0.00 ± 0.01 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaushu, L.; Atzil, S.; Vered, M.; Chaushu, G.; Matalon, S.; Weinberg, E. Age-Related Palatal Wound Healing: An Experimental In Vivo Study. Biology 2021, 10, 240. https://doi.org/10.3390/biology10030240
Chaushu L, Atzil S, Vered M, Chaushu G, Matalon S, Weinberg E. Age-Related Palatal Wound Healing: An Experimental In Vivo Study. Biology. 2021; 10(3):240. https://doi.org/10.3390/biology10030240
Chicago/Turabian StyleChaushu, Liat, Svetlana Atzil, Marilena Vered, Gavriel Chaushu, Shlomo Matalon, and Evgeny Weinberg. 2021. "Age-Related Palatal Wound Healing: An Experimental In Vivo Study" Biology 10, no. 3: 240. https://doi.org/10.3390/biology10030240
APA StyleChaushu, L., Atzil, S., Vered, M., Chaushu, G., Matalon, S., & Weinberg, E. (2021). Age-Related Palatal Wound Healing: An Experimental In Vivo Study. Biology, 10(3), 240. https://doi.org/10.3390/biology10030240








