Asparagopsis armata Exudate Cocktail: The Quest for the Mechanisms of Toxic Action of an Invasive Seaweed on Marine Invertebrates
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Organisms
2.2. Experimental Setup
2.3. Biochemical Analysis
2.3.1. Tissue Preparation
2.3.2. Protein Quantification
2.3.3. Antioxidant and Detoxification Defenses
2.3.4. Oxidative Damage
2.3.5. Neuromuscular Biomarker
2.3.6. Fatty Acid Profile
2.4. Statistical Analysis
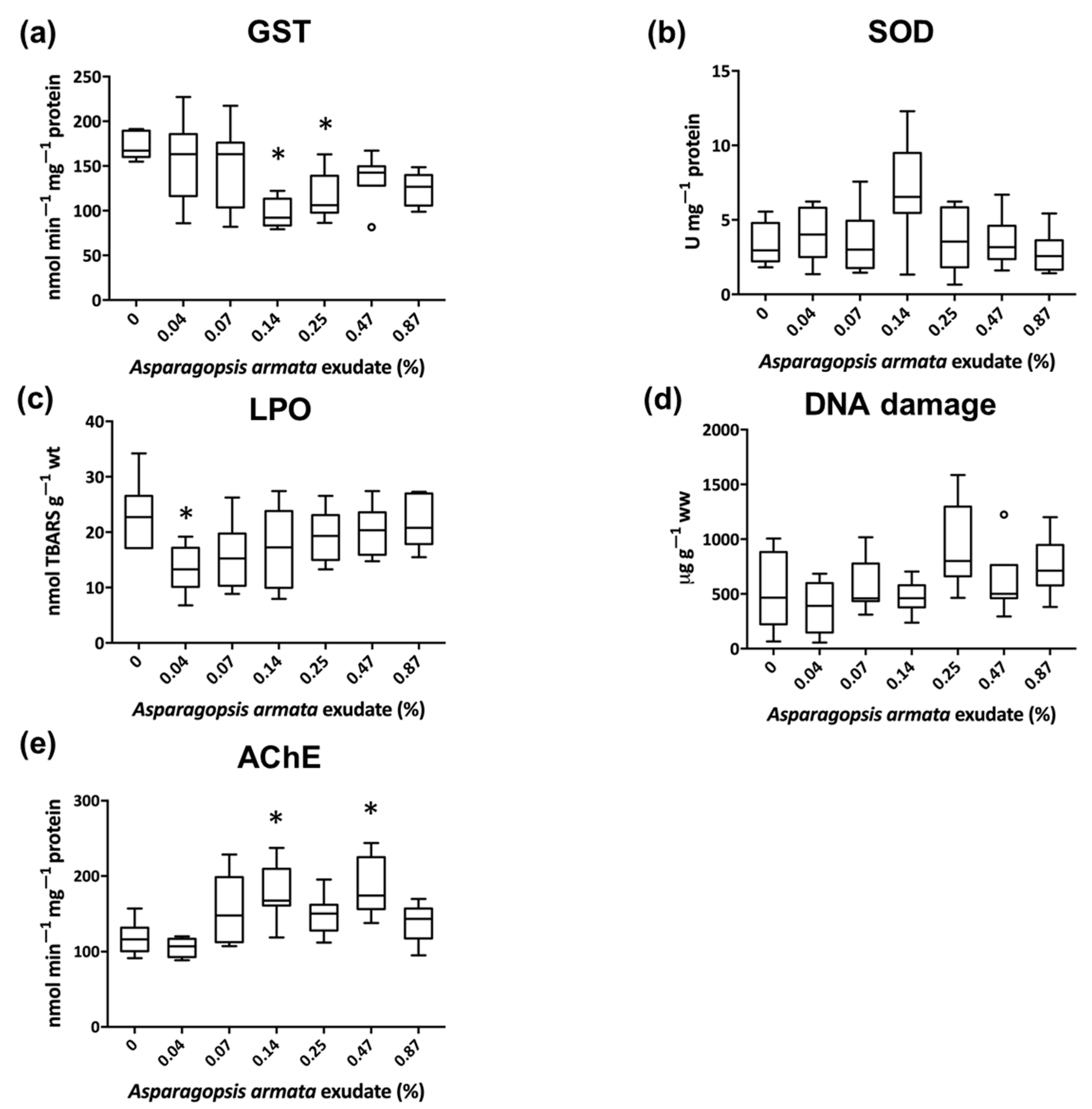
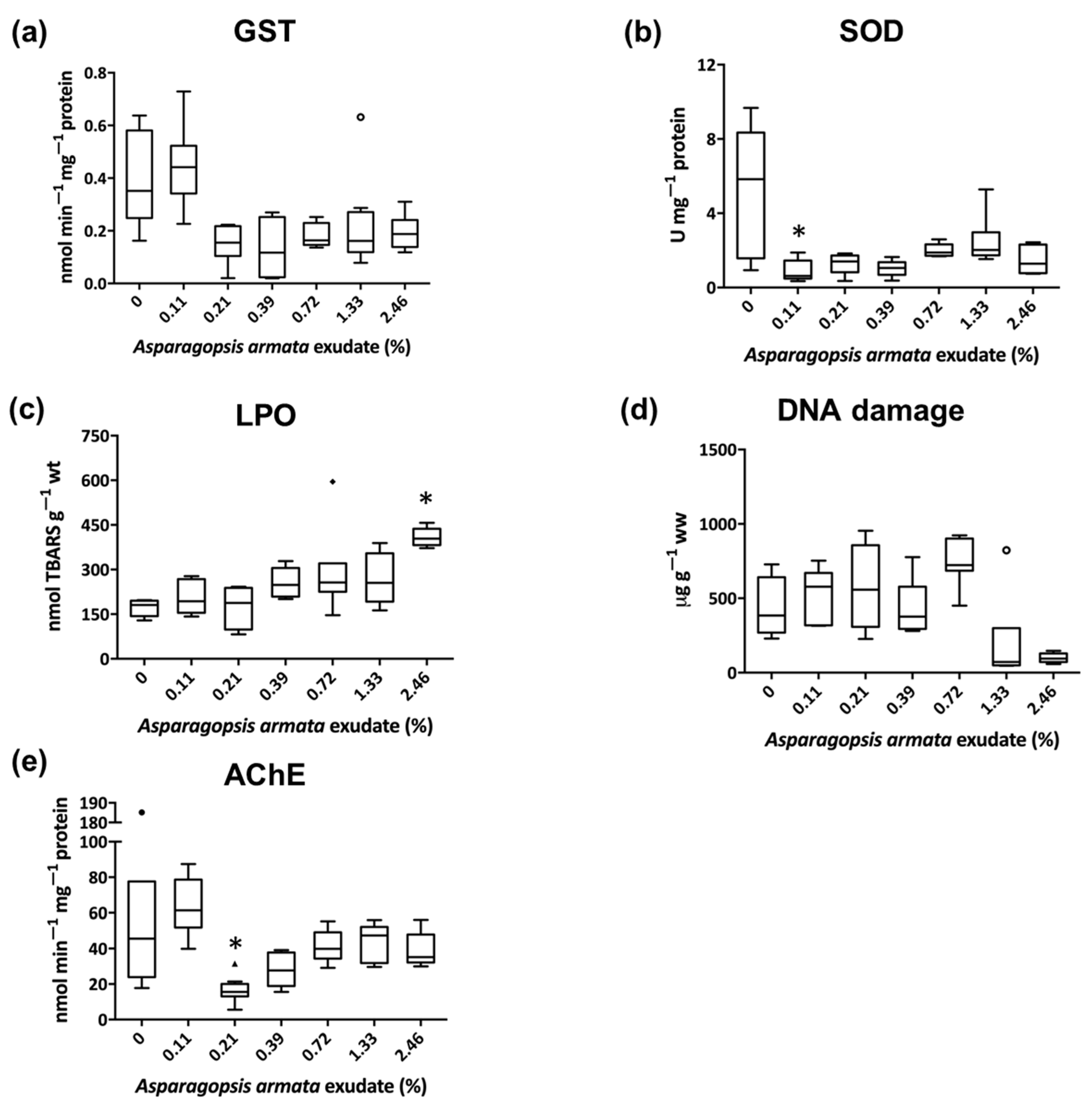
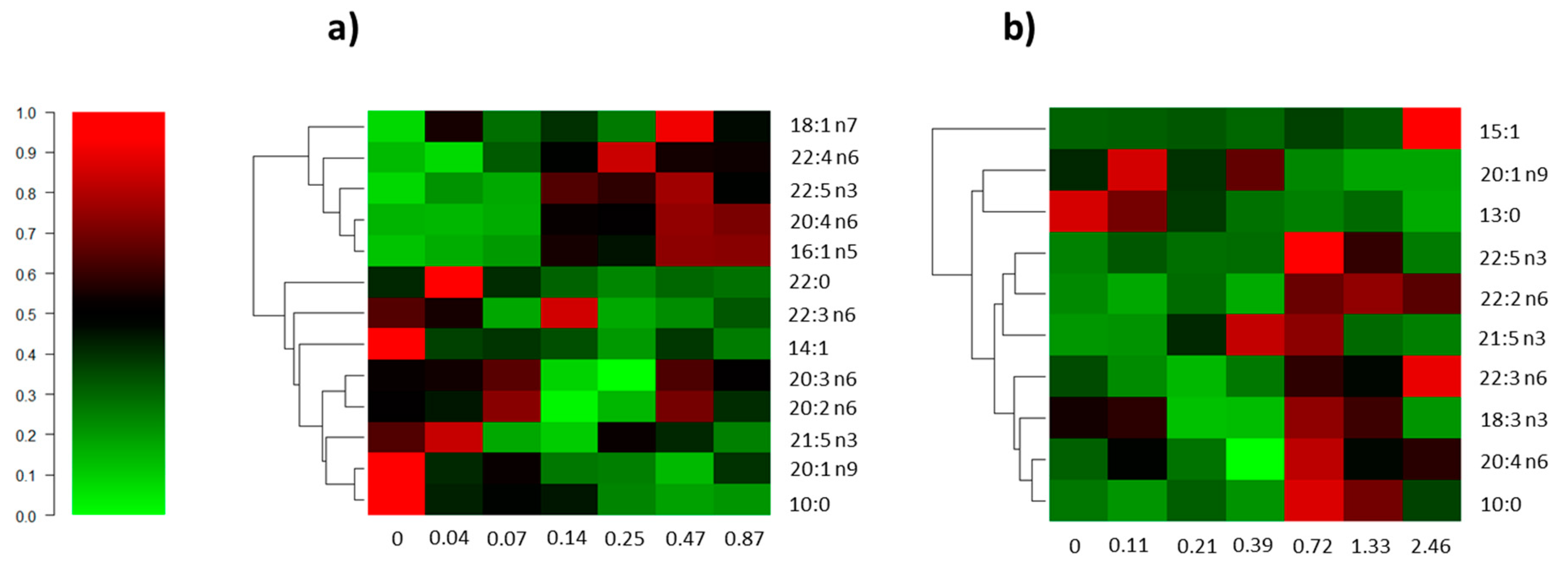
3. Results
3.1. Biochemical Biomarkers
3.2. Fatty Acid Profile
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gribble, G.W. The diversity of naturally produced organohalogens. Chemosphere 2003, 52, 289–297. [Google Scholar] [CrossRef]
- Amsler, C.D. Algal Chemical Ecology; Springer: Berlin/Heidelberg, Germany, 2008; Volume 468. [Google Scholar]
- McConnell, O.; Fenical, W. Halogen chemistry of the red alga Asparagopsis. Phytochemistry 1977, 16, 367–374. [Google Scholar] [CrossRef]
- Paul, N.A.; Cole, L.; De Nys, R.; Steinberg, P.D. Ultrastructure of the gland cells of the red alga Asparagopsis armata (Bonnemaisoniaceae. J. Phycol. 2006, 42, 637–645. [Google Scholar] [CrossRef]
- Paul, N.A.; de Nys, R.; Steinberg, P.D. Chemical defence against bacteria in the red alga Asparagopsis armata: Linking structure with function. Mar. Ecol. Prog. Ser. 2006, 306, 87–101. [Google Scholar] [CrossRef]
- Paul, N.A.; De Nys, R.; Steinberg, P.D. Seaweed–herbivore interactions at a small scale: Direct tests of feeding deterrence by filamentous algae. Mar. Ecol. Prog. Ser. 2006, 323, 1–9. [Google Scholar] [CrossRef]
- Silva, C.O.; Lemos, M.F.L.; Gaspar, R.; Gonçalves, C.; Neto, J.M. The effects of the invasive seaweed Asparagopsis armata on native rock pool communities: Evidences from experimental exclusion. Ecol. Indic. 2021, 125, 107463. [Google Scholar] [CrossRef]
- Silva, C.O.; Novais, S.C.; Soares, A.M.V.M.; Barata, C.; Lemos, M.F.L. Impacts of the Invasive Seaweed Asparagopsis armata Exudate on Energetic Metabolism of Rock Pool Invertebrates. Toxins 2021, 13, 15. [Google Scholar] [CrossRef] [PubMed]
- Harlin, M. Allelochemistry in marine macroalgae. Crit. Rev. Toxicol. 1996, 5, 237–249. [Google Scholar] [CrossRef]
- Nelson, T.A.; Lee, D.J.; Smith, B.C. Are “green tides” harmful algal blooms? Toxic properties of water-soluble extracts from two bloom-forming macroalgae, Ulva fenestrata and Ulvaria obscura (Ulvophyceae). J. Phycol. 2003, 39, 874–879. [Google Scholar] [CrossRef]
- Gaudêncio, M.J.; Guerra, M.T. Preliminary observations on Gibbula umbilicalis (da Costa, 1778) on the Portuguese coast. In Long-Term Changes in Coastal Benthic Communities; Springer: Dordrecht, The Netherlands, 1987; pp. 23–30. [Google Scholar]
- Livingstone, D.R. Oxidative stress in aquatic organisms in relation to pollution and aquaculture. Rev. De Med. Vet. 2003, 154, 427–430. [Google Scholar]
- Stegeman, J.J.; Brouwer, M.; Richard, T.D.G.; Forlin, L.; Fowler, B.A.; Sanders, B.M.; van Veld, P.A. Molecular responses to environmental contamination: Enzyme and protein systems as indicators of chemical exposure and effect. In Biomarkers: Biochemical, Physiological and Histological Markers of Anthropogenic Stress; Huggett, R.J., Kimerly, R.A., Mehrle, P.M., Jr., Bergman, H.L., Eds.; Lewis Publishers: Chelsea, MI, USA, 1992; pp. 235–335. [Google Scholar]
- Boutet, I.; Tanguy, A.; Moraga, D. Response of the Pacific oyster Crassostrea gigas to hydrocarbon contamination under experimental conditions. Gene 2004, 329, 147–157. [Google Scholar] [CrossRef]
- Barata, C.; Solayan, A.; Porte, C. Role of B-esterases in assessing toxicity of organophosphorus (chlorpyrifos, malathion) and carbamate (carbofuran) pesticides to Daphnia magna. Aquat. Toxicol. 2004, 66, 125–139. [Google Scholar] [CrossRef]
- Monteiro, H.R.; Lemos, M.F.L.; Novais, S.C.; Soares, A.M.V.M.; Pestana, J.L.T. Amitraz toxicity to the midge Chironomus riparius: Life-history and biochemical responses. Chemosphere 2019, 221, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.O.; Novais, S.C.; Alves, L.M.F.; Soares, A.M.V.M.; Barata, C.; Lemos, M.F.L. Linking cholinesterase inhibition with behavioural changes in the sea snail Gibbula umbilicalis: Effects of the organophosphate pesticide chlorpyrifos. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 225, 108570. [Google Scholar] [CrossRef] [PubMed]
- Filimonova, V.; Goncalves, F.; Marques, J.C.; De Troch, M.; Goncalves, A.M. Fatty acid profiling as bioindicator of chemical stress in marine organisms: A review. Ecol. Indic. 2016, 67, 657–672. [Google Scholar] [CrossRef]
- Silva, C.O.; Simões, T.; Novais, S.C.; Pimparel, I.; Granada, L.; Soares, A.M.V.M.; Barata, C.; Lemos, M.F.L. Fatty acid profile of the sea snail Gibbula umbilicalis as a biomarker for coastal metal pollution. Sci. Total Environ. 2017, 586, 542–550. [Google Scholar] [CrossRef]
- Silva, C.S.E.; Novais, S.C.; Simões, T.; Caramalho, M.; Gravato, C.; Rodrigues, M.J.; Maranhão, P.; Lemos, M.F.L. Using biomarkers to address the impacts of pollution on limpets (Patella depressa) and their mechanisms to cope with stress. Ecol. Indic. 2018, 95, 1077–1086. [Google Scholar] [CrossRef]
- Neves, M.; Castro, B.B.; Vidal, T.; Vieira, R.; Marques, J.C.; Coutinho, J.A.P.; Gonçalves, F.; Gonçalves, A.M.M. Biochemical and populational responses of an aquatic bioindicator species, Daphnia longispina, to a commercial formulation of a herbicide (Primextra® Gold TZ) and its active ingredient (S-metolachlor). Ecol. Indic. 2015, 53, 220–230. [Google Scholar] [CrossRef]
- Lemos, M.F.L.; Soares, A.M.V.M.; Correia, A.C.; Esteves, A.C. Proteins in ecotoxicology–how, why and why not? Proteomics 2010, 10, 873–887. [Google Scholar] [CrossRef]
- Cabecinhas, A.S.; Novais, S.C.; Santos, S.C.; Rodrigues, A.C.M.; Pestana, J.L.T.; Soares, A.M.V.M.; Lemos, M.F.L. Sensitivity of the sea snail Gibbula umbilicalis to mercury exposure–linking endpoints from different biological organization levels. Chemosphere 2015, 119, 490–497. [Google Scholar] [CrossRef]
- Oliveira, C.; Almeida, J.R.; Guilhermino, L.; Soares, A.M.V.M.; Gravato, C. Swimming velocity, avoidance behavior and biomarkers in Palaemon serratus exposed to fenitrothion. Chemosphere 2013, 90, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Frasco, M.F.; Fournier, D.; Carvalho, F.; Guilhermino, L. Cholinesterase from the common prawn (Palaemon serratus) eyes: Catalytic properties and sensitivity to organophosphate and carbamate compounds. Aquat. Toxicol. 2006, 77, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Frasco, M.F.; Guilhermino, L. Effects of dimethoate and beta-naphtho flavone on selected biomarkers of Poecilia reticulata. Fish Physiol. Biochem. 2002, 26, 149–156. [Google Scholar] [CrossRef]
- McCord, J.M.; Fridovich, I. Superoxide dismutase an enzymic function for erythrocuprein (hemocuprein). J. Biol. Chem. 1969, 244, 6049–6055. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Bird, R.P.; Draper, H.H. Comparative studies on different methods of malonaldehyde determination. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1984; Volume 105, pp. 299–305. [Google Scholar]
- Torres, M.A.; Testa, C.P.; Gáspari, C.; Masutti, M.B.; Panitz, C.M.N.; Curi-Pedrosa, R.; de Almeida, E.A.; Di Mascio, P.; Filho, D.W. Oxidative stress in the mussel Mytella guyanensis from polluted mangroves on Santa Catarina Island, Brazil. Mar. Pollut. Bull. 2002, 44, 923–932. [Google Scholar] [CrossRef]
- Olive, P.L. DNA Precipitation Assay: A Rapid and Simple Method for Detecting DNA Damage in Mammalian Cells. Environ. Mol. Mutagenesis 1988, 11, 487–495. [Google Scholar] [CrossRef]
- de Lafontaine, Y.; Gagné, F.; Blaise, C.; Costan, G.; Gagnon, P.; Chan, H.M. Biomarkers in zebra mussels (Dreissena polymorpha) for the assessment and monitoring of water quality of the St Lawrence River (Canada). Aquat. Toxicol. 2000, 50, 51–71. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Guo, X. (Ed.) Advances in Gas Chromatography; BoD–Books on Demand: Norderstedt, Germany, 2014. [Google Scholar]
- R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria. Available online: https://www.R-project.org/ (accessed on 1 November 2020).
- RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA. Available online: http://www.rstudio.com/ (accessed on 1 November 2020).
- Pohlert, T. PMCMR: Calculate Pairwise Multiple Comparisons of Mean Rank Sums. Ver. 4.3. R Package. 2018. Available online: http://CRAN.R-project.org/package=PMCMR (accessed on 1 November 2020).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Stevens, M.H.H. Package ‘Vegan’. R Package Version 2.5-6. 2019. Available online: https://CRAN.R-project.org/package=vegan (accessed on 1 November 2020).
- Maechler, M.; Rousseeuw, P.; Struyf, A.; Hubert, M.; Hornik, K. Cluster: Cluster Analysis Basics and Extensions. 2019. Available online: https://CRAN.R-project.org/package=cluster (accessed on 1 November 2020).
- Dring, M.J. Stress resistance and disease resistance in seaweeds: The role of reactive oxygen metabolism. Adv. Bot. Res. 2005, 43, 175–207. [Google Scholar]
- Cunha, I.; Mangas-Ramirez, E.; Guilhermino, L. Effects of copper and cadmium on cholinesterase and glutathione S-transferase activities of two marine gastropods (Monodonta lineata and Nucella lapillus). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 145, 648–657. [Google Scholar] [CrossRef]
- Lee, K. Glutathione S-transferase activities in phytophagous insects: Induction and inhibition by plant phototoxins and phenols. Insect Biochem. 1991, 21, 353–361. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P. How does hormesis impact biology, toxicology, and medicine? NPJ Aging Mech. Dis. 2017, 3, 13. [Google Scholar] [CrossRef]
- Conolly, R.B.; Lutz, W.K. Nonmonotonic Dose-Response Relationships: Mechanistic Basis, Kinetic Modeling, and Implications for Risk Assessment. Toxicol. Sci. 2004, 77, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Aderemi, A.O.; Novais, S.C.; Lemos, M.F.L.; Alves, L.M.F.; Hunter, C.; Pahl, O. Oxidative stress responses and cellular energy allocation changes in microalgae following exposure to widely used human antibiotics. Aquat. Toxicol. 2018, 203, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.S.E.; Novais, S.C.; Lemos, M.F.L.; Mendes, S.; Oliveira, A.P.; Gonçalves, E.J.; Faria, A.M. Effects of ocean acidification on the swimming ability, development and biochemical responses of sand smelt larvae. Sci. Total Environ. 2016, 563, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.L.N.; Venugopal, N.B.R.K. Effect of fluoride on acetylcholinesterase activity and oxygen consumption in a freshwater field crab, Barytelphusa guerini. Bull. Environ. Contam. Toxicol. 1990, 45, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Badiou, A.; Meled, M.; Belzunces, L.P. Honeybee Apis mellifera acetylcholinesterase—a biomarker to detect deltamethrin exposure. Ecotoxicol. Environ. Saf. 2008, 69, 246–253. [Google Scholar] [CrossRef]
- Hossain, M.M.; Suzuki, T.; Sato, I.; Takewaki, T.; Suzuki, K.; Kobayashi, H. The modulatory effect of pyrethroids on acetylcholine release in the hippocampus of freely moving rats. Neurotoxicology 2004, 25, 825–833. [Google Scholar] [CrossRef]
- Espinoza, B.; Silman, I.; Arnon, R.; Tarrab-Hazdai, R. Phosphatidylinositol-specific Phospholipase C induces biosynthesis of acetylcholinesterase via diacylglycerol in Schistosoma mansoni. Eur. J. Biochem. 1991, 195, 863–870. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, W.; Zhang, J.; Zhao, H.; Zhang, Y.; Quan, X.; Jin, Y. Characterisation of acute toxicity, genotoxicity and oxidative stress posed by textile effluent on zebrafish. J. Environ. Sci. 2012, 24, 2019–2027. [Google Scholar] [CrossRef]
- Hampel, M.; Blasco, J.; Martín Díaz, M.L. Biomarkers and Effects. In Marine Ecotoxicology: Current Knowledge and Future Issues; Elsevier: Amsterdam, The Netherlands, 2016; pp. 121–165. [Google Scholar]
- Box, A.; Sureda, A.; Deudero, S. Antioxidant response of the bivalve Pinna nobilis colonised by invasive red macroalgae Lophocladia lallemandii. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2009, 149, 456–460. [Google Scholar] [CrossRef]
- Natarajan, S.; Shanmugiahthevar, K.P.; Kasi, P.D. Cholinesterase inhibitors from Sargassum and Gracilaria gracilis: Seaweeds inhabiting South Indian coastal areas (Hare Island, Gulf of Mannar). Nat. Prod. Res. 2009, 23, 355–369. [Google Scholar] [CrossRef]
- Custódio, L.; Silvestre, L.; Rocha, M.I.; Rodrigues, M.J.; Vizetto-Duarte, C.; Pereira, H.; Barreira, L.; Varela, J. Methanol extracts from Cystoseira tamariscifolia and Cystoseira nodicaulis are able to inhibit cholinesterases and protect a human dopaminergic cell line from hydrogen peroxide-induced cytotoxicity. Pharm. Biol. 2016, 54, 1687–1696. [Google Scholar] [CrossRef]
- Fokina, N.N.; Ruokolainen, T.R.; Nemova, N.N.; Bakhmet, I.N. Changes of blue mussels Mytilus edulis L. lipid composition under cadmium and copper toxic effect. Biol. Trace Elem. Res. 2013, 154, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Trabelsi, W.; Chetoui, I.; Fouzai, C.; Bejaoui, S.; Rabeh, I.; Telahigue, K.; Soudani, N. Redox status and fatty acid composition of Mactra corallina digestive gland following exposure to acrylamide. Environ. Sci. Pollut. Res. 2019, 26, 22197–22208. [Google Scholar] [CrossRef] [PubMed]
- Maazouzi, C.; Masson, G.; Izquierdo, M.S.; Pihan, J.C. Chronic copper exposure and fatty acid composition of the amphipod Dikerogammarus villosus: Results from a field study. Environ. Pollut. 2008, 156, 221–226. [Google Scholar] [CrossRef]
- Vance, D.E.; Vance, J.E. (Eds.) Biochemistry of Lipids, Lipoproteins and Membranes, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2002; p. 624. [Google Scholar]
- Kaur, G.; Cameron-Smith, D.; Garg, M.; Sinclair, A.J. Docosapentaenoic acid (22:5n-3): A review of its biological effects. Prog. Lipid Res. 2011, 50, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Calder, P. Marine omega-3 fatty acids and inflammation. J. Lipid Nutr. 2010, 19, 233–244. [Google Scholar] [CrossRef][Green Version]
- Delaporte, M.; Soudant, P.; Moal, J.; Giudicelli, E.; Lambert, C.; Séguineau, C.; Samain, J.F. Impact of 20: 4n-6 supplementation on the fatty acid composition and hemocyte parameters of the pacific oyster Crassostrea gigas. Lipids 2006, 41, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Omega-3 fatty acids in inflammation and autoimmune diseases. J. Am. Coll. Nutr. 2002, 21, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Larsen, L.N.; Høvik, K.; Bremer, J.; Holm, K.H.; Myhren, F.; Børretzen, B. Heneicosapentaenoate (21: 5n-3): Its incorporation into lipids and its effects on arachidonic acid and eicosanoid synthesis. Lipids 1997, 32, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Mirmonsef, P.; Zariffard, M.R.; Gilbert, D.; Makinde, H.; Landay, A.L.; Spear, G.T. Short-chain fatty acids induce pro-inflammatory cytokine production alone and in combination with toll-like receptor ligands. Am. J. Reprod. Immunol. 2012, 67, 391–400. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, C.O.; Simões, T.; Félix, R.; Soares, A.M.V.M.; Barata, C.; Novais, S.C.; Lemos, M.F.L. Asparagopsis armata Exudate Cocktail: The Quest for the Mechanisms of Toxic Action of an Invasive Seaweed on Marine Invertebrates. Biology 2021, 10, 223. https://doi.org/10.3390/biology10030223
Silva CO, Simões T, Félix R, Soares AMVM, Barata C, Novais SC, Lemos MFL. Asparagopsis armata Exudate Cocktail: The Quest for the Mechanisms of Toxic Action of an Invasive Seaweed on Marine Invertebrates. Biology. 2021; 10(3):223. https://doi.org/10.3390/biology10030223
Chicago/Turabian StyleSilva, Carla O., Tiago Simões, Rafael Félix, Amadeu M.V.M. Soares, Carlos Barata, Sara C. Novais, and Marco F.L. Lemos. 2021. "Asparagopsis armata Exudate Cocktail: The Quest for the Mechanisms of Toxic Action of an Invasive Seaweed on Marine Invertebrates" Biology 10, no. 3: 223. https://doi.org/10.3390/biology10030223
APA StyleSilva, C. O., Simões, T., Félix, R., Soares, A. M. V. M., Barata, C., Novais, S. C., & Lemos, M. F. L. (2021). Asparagopsis armata Exudate Cocktail: The Quest for the Mechanisms of Toxic Action of an Invasive Seaweed on Marine Invertebrates. Biology, 10(3), 223. https://doi.org/10.3390/biology10030223










