The Adaptor Protein Lurap1 Is Required for Cell Cohesion during Epiboly Movement in Zebrafish
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Zebrafish
2.2. Capped mRNA Synthesis and Microinjections
2.3. UV-Mediated Photo-Conversion of Kaede GFP
2.4. Cell Transplantation
2.5. Immunofluorescence
2.6. Analysis of EVL and DEL Epiboly
2.7. Live Time-Lapse Imaging
2.8. Cell Sorting and Adhesion Assays
2.9. In Situ Hybridization
2.10. Statistical Analyses
3. Results
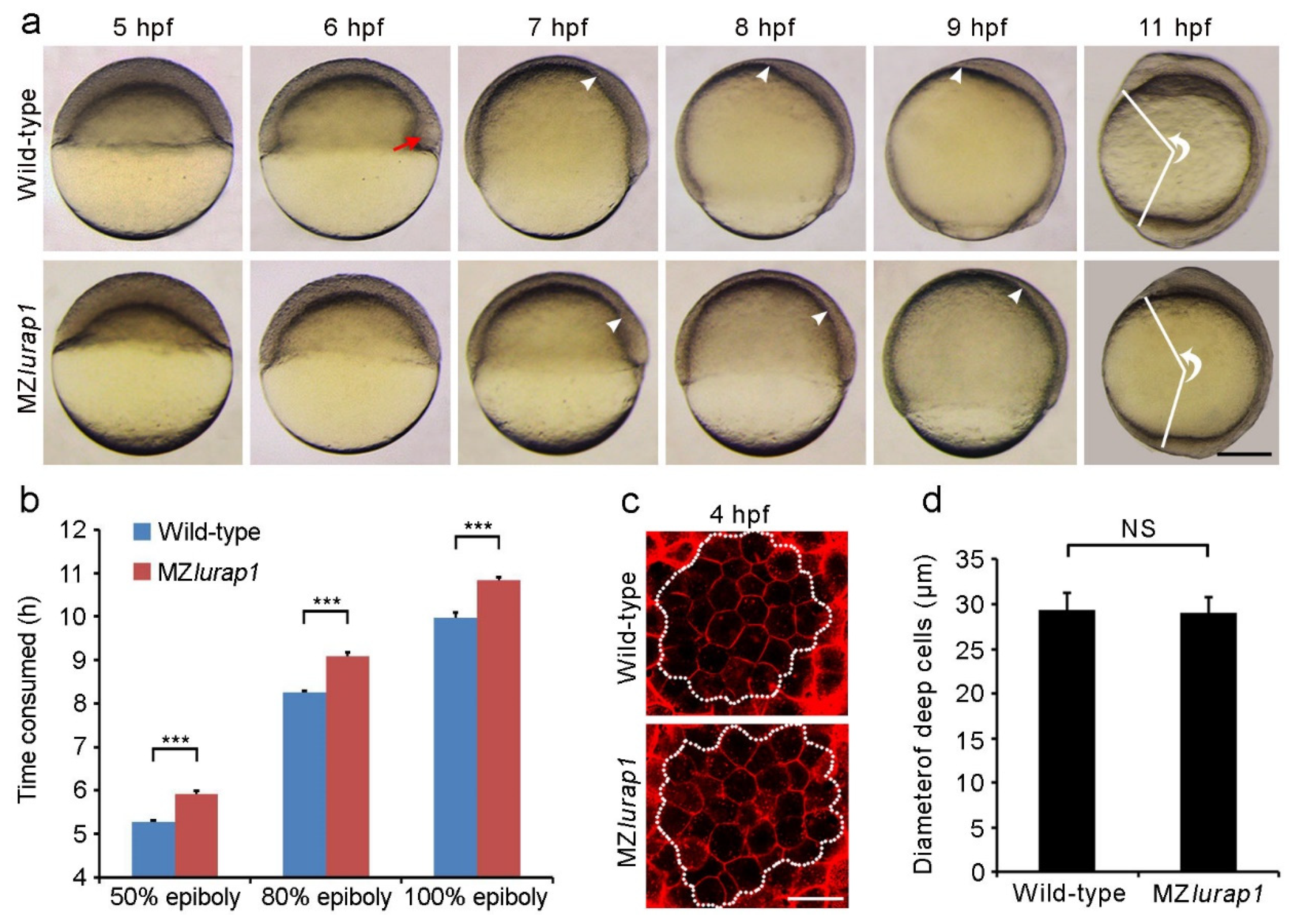
3.1. Loss of Maternal Lurap1 Delays Epiboly Movement
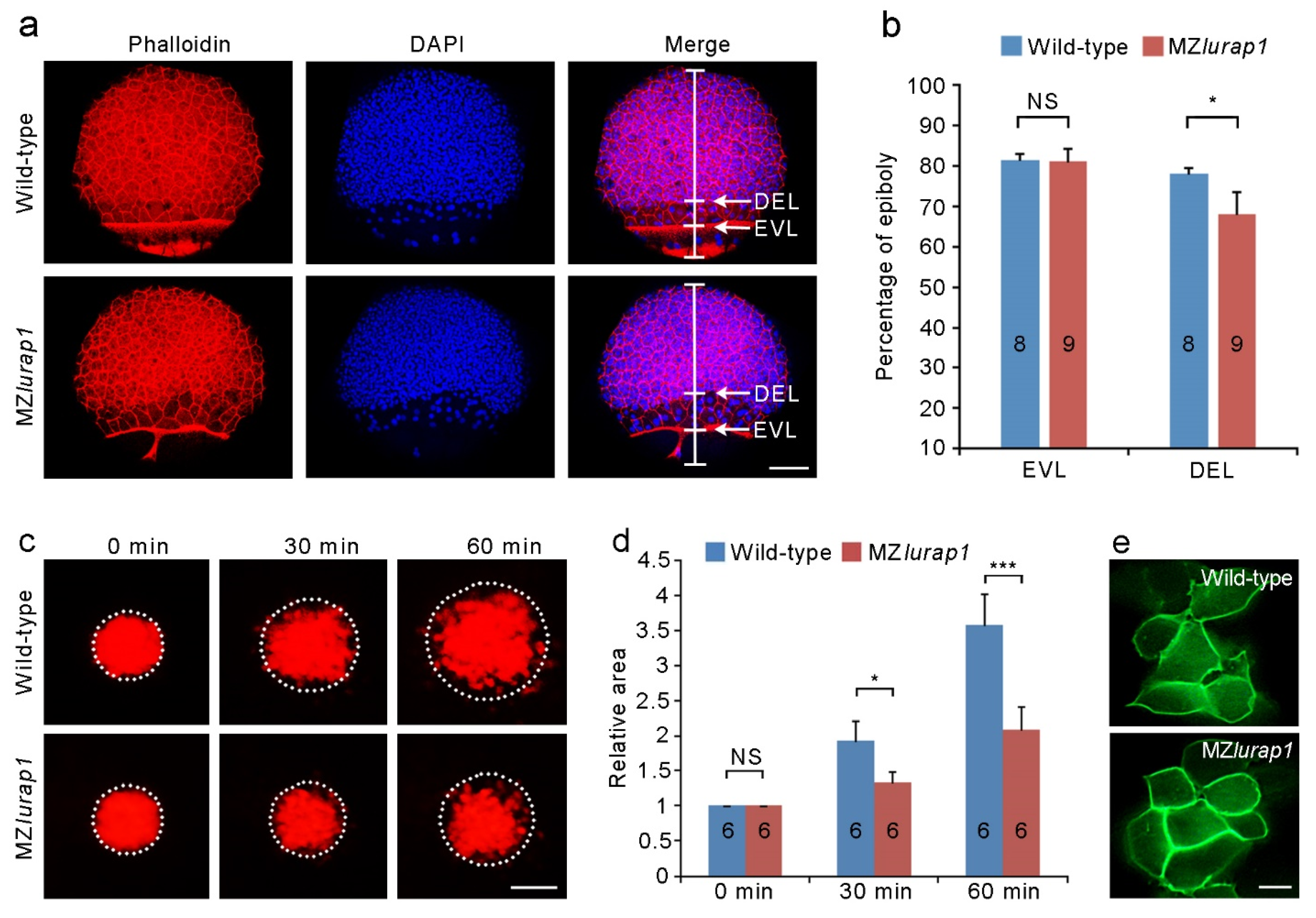
3.2. Delayed DEL Epiboly in MZlurap1 Mutants
3.3. Reduced Blastoderm Cell Cohesion in MZlurap1 Mutants
3.4. Disrupted Cortical F-actin Organization in the Blastoderm of MZlurap1 Mutants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
References
- Solnica-Krezel, L. Conserved patterns of cell movements during vertebrate gastrulation. Curr. Biol. 2005, 15, R213–R228. [Google Scholar] [CrossRef]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Warga, R.M.; Kimmel, C.B. Cell movements during epiboly and gastrulation in zebrafish. Development 1990, 108, 569–580. [Google Scholar] [CrossRef]
- Bruce, A.E.E. Zebrafish epiboly: Spreading thin over the yolk. Dev. Dyn. 2016, 245, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Bensch, R.; Song, S.; Ronneberger, O.; Driever, W. Non-directional radial intercalation dominates deep cell behavior during zebrafish epiboly. Biol. Open 2013, 2, 845–854. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bruce, A.E.E.; Heisenberg, C.P. Mechanisms of zebrafish epiboly: A current view. Curr. Top. Dev. Biol. 2020, 136, 319–341. [Google Scholar] [CrossRef]
- Solnica-Krezel, L. Gastrulation in zebrafish—All just about adhesion? Curr. Opin. Genet. Dev. 2006, 16, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Kane, D.A.; McFarland, K.N.; Warga, R.M. Mutations in half baked/E-cadherin block cell behaviors that are necessary for teleost epiboly. Development 2005, 132, 1105–1116. [Google Scholar] [CrossRef]
- McFarland, K.N.; Warga, R.M.; Kane, D.A. Genetic locus half baked is necessary for morphogenesis of the ectoderm. Dev. Dyn. 2005, 233, 390–406. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Yabe, T.; Muraoka, O.; Yonemura, S.; Aramaki, S.; Hatta, K.; Bae, Y.K.; Nojima, H.; Hibi, M. E-cadherin is required for gastrulation cell movements in zebrafish. Mech. Dev. 2005, 122, 747–763. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, N.E.; Wong, A.H.; Bruce, A.E.E. Spatiotemporal characterization of dynamic epithelial filopodia during zebrafish epiboly. Dev. Dyn. 2019, 248, 997–1008. [Google Scholar] [CrossRef]
- Lee, S.J. Dynamic regulation of the microtubule and actin cytoskeleton in zebrafish epiboly. Biochem. Biophys. Res. Commun. 2014, 452, 1–7. [Google Scholar] [CrossRef]
- Cheng, J.C.; Miller, A.L.; Webb, S.E. Organization and function of microfilaments during late epiboly in zebrafish embryos. Dev. Dyn. 2004, 231, 313–323. [Google Scholar] [CrossRef]
- Köppen, M.; Fernandez, B.G.; Carvalho, L.; Jacinto, A.; Heisenberg, C.P. Coordinated cell-shape changes control epithelial movement in zebrafish and Drosophila. Development 2006, 133, 2671–2681. [Google Scholar] [CrossRef] [PubMed]
- Lachnit, M.; Kur, E.; Driever, W. Alterations of the cytoskeleton in all three embryonic lineages contribute to the epiboly defect of Pou5f1/Oct4 deficient MZspg zebrafish embryos. Dev. Biol. 2008, 315, 1–17. [Google Scholar] [CrossRef]
- Lin, F.; Chen, S.; Sepich, D.S.; Panizzi, J.R.; Clendenon, S.G.; Marrs, J.A.; Hamm, H.E.; Solnica-Krezel, L. Galpha12/13 regulate epiboly by inhibiting E-cadherin activity and modulating the actin cytoskeleton. J. Cell Biol. 2009, 184, 909–921. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, X.; Gong, B.; Wu, D.; Meng, A.; Jia, S. Alkbh4 and atrn act maternally to regulate zebrafish epiboly. Int. J. Biol. Sci. 2017, 13, 1051–1066. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.L.; Shao, M.; Shi, D.L. Rac1 signalling coordinates epiboly movement by differential regulation of actin cytoskeleton in zebrafish. Biochem. Biophys. Res. Commun. 2017, 490, 1059–1065. [Google Scholar] [CrossRef]
- Li, Y.L.; Cheng, X.N.; Lu, T.; Shao, M.; Shi, D.L. Syne2b/nesprin-2 is required for actin organization and epithelial integrity during epiboly movement in zebrafish. Front. Cell Dev. Biol. 2021, 9, 671887. [Google Scholar] [CrossRef] [PubMed]
- Tan, I.; Yong, J.; Dong, J.M.; Lim, L.; Leung, T. A tripartite complex containing MRCK modulates lamellar actomyosin retrograde flow. Cell 2008, 135, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.N.; Shao, M.; Li, J.T.; Wang, Y.F.; Qi, J.; Xu, Z.G.; Shi, D.L. Leucine repeat adaptor protein 1 interacts with Dishevelled to regulate gastrulation cell movements in zebrafish. Nat. Commun. 2017, 8, 1353. [Google Scholar] [CrossRef]
- Carreira-Barbosa, F.; Kajita, M.; Morel, V.; Wada, H.; Okamoto, H.; Martinez Arias, A.; Fujita, Y.; Wilson, S.W.; Tada, M. Flamingo regulates epiboly and convergence/extension movements through cell cohesive and signalling functions during zebrafish gastrulation. Development 2009, 136, 383–392. [Google Scholar] [CrossRef]
- Schepis, A.; Sepich, D.; Nelson, W.J. αE-catenin regulates cell-cell adhesion and membrane blebbing during zebrafish epiboly. Development 2012, 139, 537–546. [Google Scholar] [CrossRef][Green Version]
- Yap, A.S.; Crampton, M.S.; Hardin, J. Making and breaking contacts: The cellular biology of cadherin regulation. Curr. Opin. Cell Biol. 2007, 19, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Troyanovsky, R.B.; Troyanovsky, S.M. Binding to F-actin guides cadherin cluster assembly, stability, and movement. J. Cell Biol. 2013, 201, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.K.; Gomez, G.A.; Michael, M.; Verma, S.; Cox, H.L.; Lefevre, J.G.; Parton, R.G.; Hamilton, N.A.; Neufeld, Z.; Yap, A.S. Cortical F-actin stabilization generates apical-lateral patterns of junctional contractility that integrate cells into epithelia. Nat. Cell Biol. 2014, 16, 167–178. [Google Scholar] [CrossRef]
- Lin, C.W.; Yen, S.T.; Chang, H.T.; Chen, S.J.; Lai, S.L.; Liu, Y.C.; Chan, T.H.; Liao, W.L.; Lee, S.J. Loss of cofilin 1 disturbs actin dynamics, adhesion between enveloping and deep cell layers and cell movements during gastrulation in zebrafish. PLoS ONE 2010, 5, e15331. [Google Scholar] [CrossRef]
- Huang, H.; Lu, F.I.; Jia, S.; Meng, S.; Cao, Y.; Wang, Y.; Ma, W.; Yin, K.; Wen, Z.; Peng, J.; et al. Amotl2 is essential for cell movements in zebrafish embryo and regulates c-Src translocation. Development 2007, 134, 979–988. [Google Scholar] [CrossRef]
- Tada, M.; Kai, M. Planar cell polarity in coordinated and directed movements. Curr. Top. Dev. Biol. 2012, 101, 77–110. [Google Scholar] [CrossRef]
- Montero, J.A.; Carvalho, L.; Wilsch-Bräuninger, M.; Kilian, B.; Mustafa, C.; Heisenberg, C.P. Shield formation at the onset of zebrafish gastrulation. Development 2005, 132, 1187–1198. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Schilling, T.F. Chemokine signaling controls endodermal migration during zebrafish gastrulation. Science 2008, 322, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Love, A.M.; Prince, D.J.; Jessen, J.R. Vangl2-dependent regulation of membrane protrusions and directed migration requires a fibronectin extracellular matrix. Development 2018, 145, dev165472. [Google Scholar] [CrossRef]
- Prince, D.J.; Jessen, J.R. Dorsal convergence of gastrula cells requires Vangl2 and an adhesion protein-dependent change in protrusive activity. Development 2019, 146, dev182188. [Google Scholar] [CrossRef] [PubMed]
- Miller, N.L.; Lawson, C.; Chen, X.L.; Lim, S.T.; Schlaepfer, D.D. Rgnef (p190RhoGEF) knockout inhibits RhoA activity, focal adhesion establishment, and cell motility downstream of integrins. PLoS ONE 2012, 7, e37830. [Google Scholar] [CrossRef]
- Cao, J.M.; Cheng, X.N.; Li, S.Q.; Heller, S.; Xu, Z.G.; Shi, D.L. Identification of novel MYO18A interaction partners required for myoblast adhesion and muscle integrity. Sci. Rep. 2016, 6, 36768. [Google Scholar] [CrossRef]
- Cao, J.M.; Li, S.Q.; Shao, M.; Cheng, X.N.; Xu, Z.G.; Shi, D.L. The PDZ-containing unconventional myosin XVIIIA regulates embryonic muscle integrity in zebrafish. J. Genet. Genom. 2014, 41, 417–428. [Google Scholar] [CrossRef]
- Taft, M.H.; Latham, S.L. Myosin XVIII. Adv. Exp. Med. Biol. 2020, 1239, 421–438. [Google Scholar] [CrossRef]
- Ouyang, Z.; Zhao, S.; Yao, S.; Wang, J.; Cui, Y.; Wei, K.; Jiu, Y. Multifaceted function of myosin-18, an unconventional class of the myosin superfamily. Front. Cell Dev. Biol. 2021, 9, 632445. [Google Scholar] [CrossRef]
- Miller, N.L.; Kleinschmidt, E.G.; Schlaepfer, D.D. RhoGEFs in cell motility: Novel links between Rgnef and focal adhesion kinase. Curr. Mol. Med. 2014, 14, 221–234. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.-T.; Cheng, X.-N.; Zhang, C.; Shi, D.-L.; Shao, M. The Adaptor Protein Lurap1 Is Required for Cell Cohesion during Epiboly Movement in Zebrafish. Biology 2021, 10, 1337. https://doi.org/10.3390/biology10121337
Li J-T, Cheng X-N, Zhang C, Shi D-L, Shao M. The Adaptor Protein Lurap1 Is Required for Cell Cohesion during Epiboly Movement in Zebrafish. Biology. 2021; 10(12):1337. https://doi.org/10.3390/biology10121337
Chicago/Turabian StyleLi, Ji-Tong, Xiao-Ning Cheng, Chong Zhang, De-Li Shi, and Ming Shao. 2021. "The Adaptor Protein Lurap1 Is Required for Cell Cohesion during Epiboly Movement in Zebrafish" Biology 10, no. 12: 1337. https://doi.org/10.3390/biology10121337
APA StyleLi, J.-T., Cheng, X.-N., Zhang, C., Shi, D.-L., & Shao, M. (2021). The Adaptor Protein Lurap1 Is Required for Cell Cohesion during Epiboly Movement in Zebrafish. Biology, 10(12), 1337. https://doi.org/10.3390/biology10121337





