Extraction of Aloesin from Aloe vera Rind Using Alternative Green Solvents: Process Optimization and Biological Activity Assessment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Experimental Design for Extraction Process Optimization
2.3. Extraction Process
2.4. Chromatographic Analysis of Aloesin
2.5. Extraction Process Modelling and Statistical Analysis
2.6. Dose–Response Analysis of the Solid/Liquid Ratio and Models Validation
2.7. Evaluation of Bioactive Properties
2.7.1. Lipid Peroxidation Inhibition Capacity
2.7.2. Antibacterial Activity
2.7.3. Antifungal Activity
2.7.4. Cytotoxic Activity
3. Results and Discussion
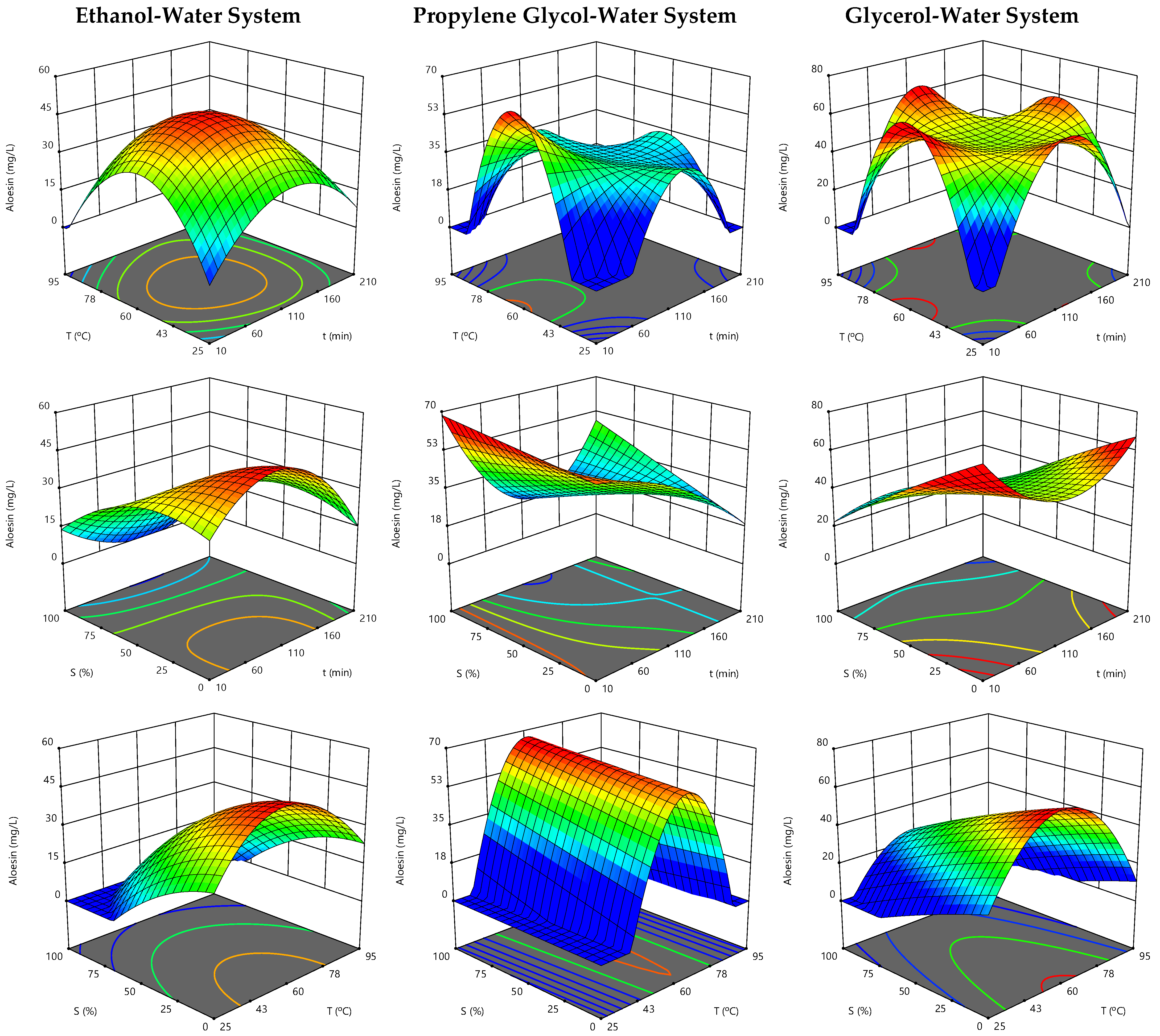
3.1. Experimental Data for Extraction Process Optimization
3.2. Models Fitting and Statistical Verification
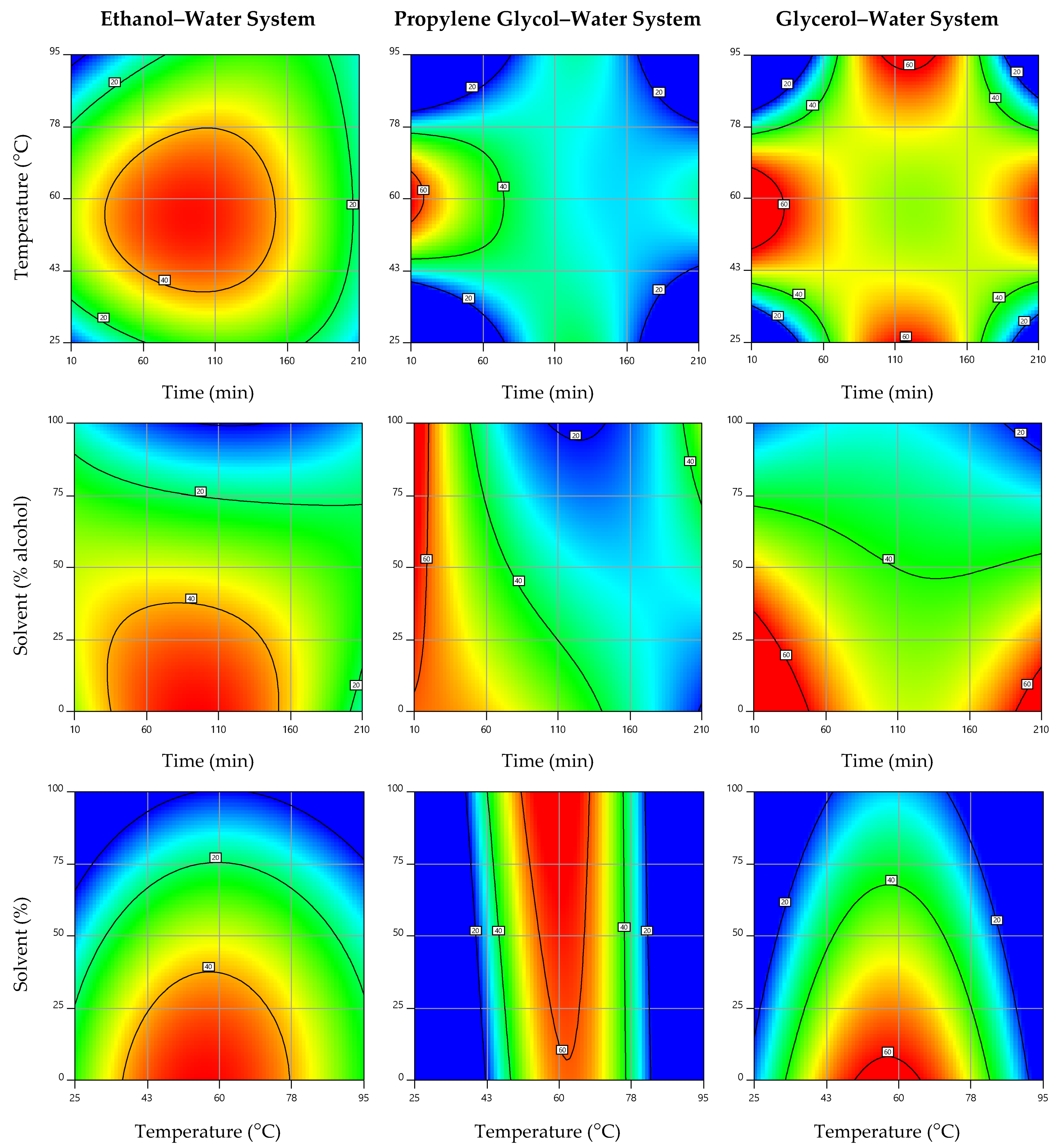
3.3. Effect of the Extraction Parameters on Aloesin Content and Optimal Extraction Conditions
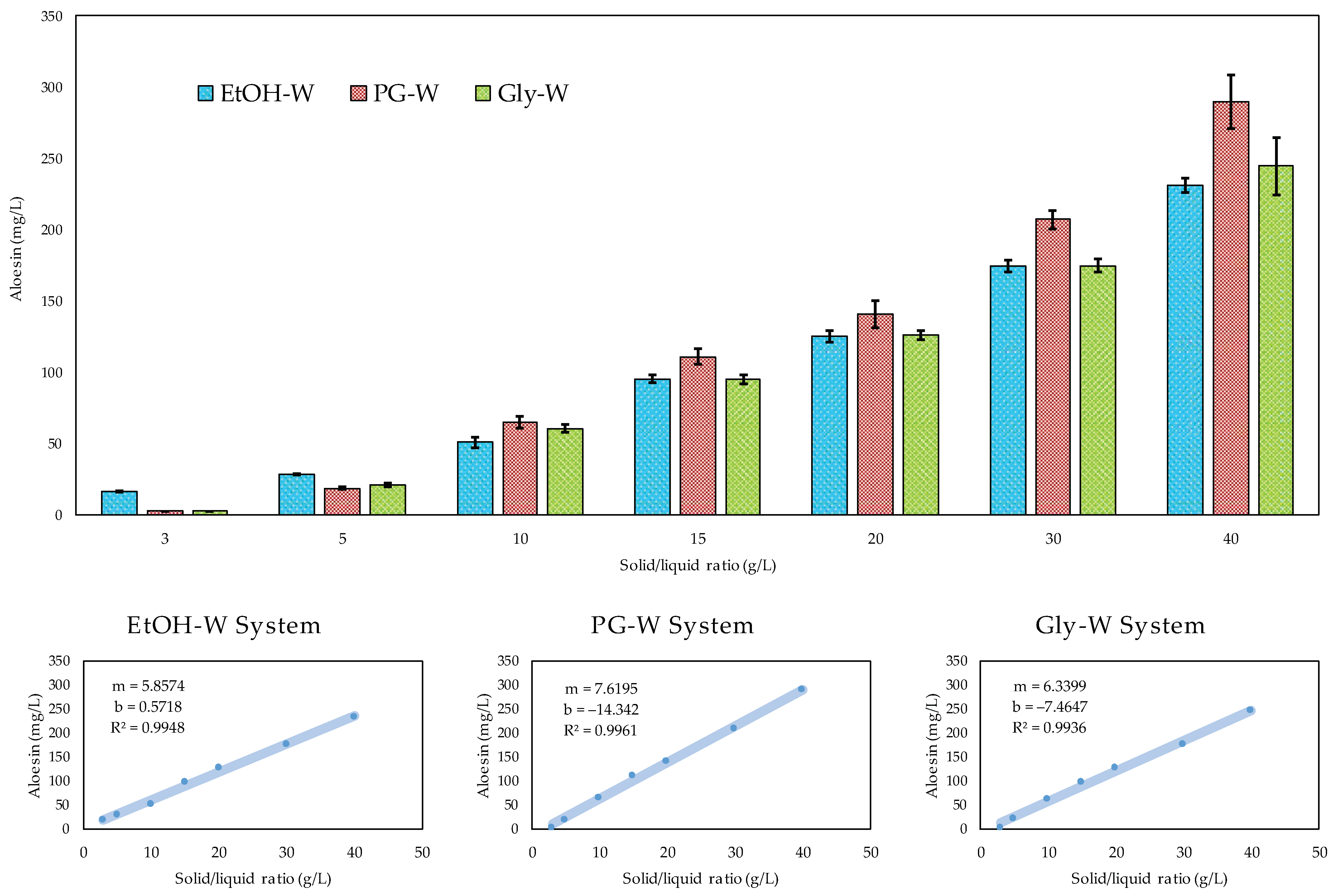
3.4. Models Validation and Effect of the Solid–Liquid Ratio
3.5. Bioactive Properties of the Aloesin Extracts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sánchez-Machado, D.I.; López-Cervantes, J.; Sendón, R.; Sanches-Silva, A. Aloe vera: Ancient knowledge with new frontiers. Trends Food Sci. Technol. 2017, 61, 94–102. [Google Scholar] [CrossRef]
- Akaberi, M.; Sobhani, Z.; Javadi, B.; Sahebkar, A.; Emami, S.A. Therapeutic effects of Aloe spp. in traditional and modern medicine: A review. Biomed. Pharmacother. 2016, 84, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Radha, M.H.; Laxmipriya, N.P. Evaluation of biological properties and clinical effectiveness of Aloe vera: A systematic review. J. Tradit. Complement. Med. 2015, 5, 21–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Añibarro-Ortega, M.; Pinela, J.; Barros, L.; Ćirić, A.; Silva, S.P.; Coelho, E.; Mocan, A.; Calhelha, R.C.; Soković, M.; Coimbra, M.A.; et al. Compositional features and bioactive properties of Aloe vera leaf (fillet, mucilage, and rind) and flower. Antioxidants 2019, 8, 444. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Yin, S.; Zhong, J.; Ding, W.; Wan, J.; Xie, Z. Mushroom tyrosinase inhibitors from Aloe barbadensis Miller. Fitoterapia 2012, 83, 1706–1711. [Google Scholar] [CrossRef]
- Ahlawat, K.S.; Khatkar, B.S. Processing, food applications and safety of Aloe vera products: A review. J. Food Sci. Technol. 2011, 48, 525–533. [Google Scholar] [CrossRef] [Green Version]
- Chandegara, V.K.; Varshney, A. Aloe vera L. processing and products: A review. Int. J. Med. Aromat. Plants 2013, 3, 492–506. [Google Scholar]
- Wahedi, H.M.; Jeong, M.; Chae, J.K.; Do, S.G.; Yoon, H.; Kim, S.Y. Aloesin from Aloe vera accelerates skin wound healing by modulating MAPK/Rho and Smad signaling pathways in vitro and in vivo. Phytomedicine 2017, 28, 19–26. [Google Scholar] [CrossRef]
- Mikayoulou, M.; Mayr, F.; Temml, V.; Pandian, A.; Vermaak, I.; Chen, W.; Komane, B.; Stuppner, H.; Viljoen, A. Anti-tyrosinase activity of South African Aloe species and isolated compounds plicataloside and aloesin. Fitoterapia 2021, 150, 104828. [Google Scholar] [CrossRef]
- Asthana, N.; Pal, K.; Aljabali, A.A.A.; Tambuwala, M.M.; de Souza, F.G.; Pandey, K. Polyvinyl alcohol (PVA) mixed green–clay and Aloe vera based polymeric membrane optimization: Peel-off mask formulation for skin care cosmeceuticals in green nanotechnology. J. Mol. Struct. 2020, 1229, 129592. [Google Scholar] [CrossRef]
- Grollier, J.F.; Lang, G.; Forestier, S.; Rosenbaum, G. Cosmetic Composition Containing Aloesin as an Agent for Protection against Sunlight and Its Use for Skin and Hair Protection. U.S. Patent US4656029A, 7 April 1987. [Google Scholar]
- Do, S.-G.; Kim, J.-W.; Jin, Y.-J.; Kim, S.-Y.; Moon, J.-H.; Lee, C.-H.; Lee, S.-R. Composition for Preventing Skin Aging Containing Aloesin or Derivative Thereof. U.S. Patent US20160175221A1, 11 July 2014. [Google Scholar]
- Aldayel, T.S.; Grace, M.H.; Lila, M.A.; Yahya, M.A.; Omar, U.M.; Alshammary, G. LC-MS characterization of bioactive metabolites from two Yemeni Aloe spp. with antioxidant and antidiabetic properties. Arab. J. Chem. 2020, 13, 5040–5049. [Google Scholar] [CrossRef]
- Majumder, R.; Das, C.K.; Mandal, M. Lead bioactive compounds of Aloe vera as potential anticancer agent. Pharmacol. Res. 2019, 148, 104416. [Google Scholar] [CrossRef]
- Lynch, B.; Simon, R.; Roberts, A. In vitro and in vivo assessment of the genotoxic activity of aloesin. Regul. Toxicol. Pharmacol. 2011, 61, 215–221. [Google Scholar] [CrossRef]
- CMI Cosmetic ingredients market to reach $23.8 bn globally by 2027. Focus Surfactants 2020, 2020, 6. [CrossRef]
- Alvarez-Rivera, G.; Llompart, M.; Garcia-Jares, C.; Lores, M. Identification of unwanted photoproducts of cosmetic preservatives in personal care products under ultraviolet-light using solid-phase microextraction and micro-matrix solid-phase dispersion. J. Chromatogr. A 2015, 1390, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lores, M.; Llompart, M.; Alvarez-Rivera, G.; Guerra, E.; Vila, M.; Celeiro, M.; Lamas, J.P.; Garcia-Jares, C. Positive lists of cosmetic ingredients: Analytical methodology for regulatory and safety controls—A review. Anal. Chim. Acta 2016, 915, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Bom, S.; Jorge, J.; Ribeiro, H.M.; Marto, J. A step forward on sustainability in the cosmetics industry: A review. J. Clean. Prod. 2019, 225, 270–290. [Google Scholar] [CrossRef]
- Steenkamp, L.H.; Mitra, R.K.; Heggie, S.J.; Phehane, V.N. Method for Converting Aloeresin A to Aloesin. U.S. Patent US9434710B2, 6 September 2006. [Google Scholar]
- Steenkamp, L.; Mathiba, K.; Steenkamp, P.; Phehane, V.; Mitra, R.; Heggie, S.; Brady, D. Biocatalytic conversion of aloeresin A to aloesin. J. Ind. Microbiol. Biotechnol. 2012, 39, 1091–1097. [Google Scholar] [CrossRef]
- Ummat, V.; Sivagnanam, S.P.; Rajauria, G.; O’Donnell, C.; Tiwari, B.K. Advances in pre-treatment techniques and green extraction technologies for bioactives from seaweeds. Trends Food Sci. Technol. 2021, 110, 90–106. [Google Scholar] [CrossRef]
- Lesellier, E.; Lefebvre, T.; Destandau, E. Recent developments for the analysis and the extraction of bioactive compounds from Rosmarinus officinalis and medicinal plants of the Lamiaceae family. TrAC—Trends Anal. Chem. 2021, 135, 116158. [Google Scholar] [CrossRef]
- Rocha, R.; Pinela, J.; Abreu, R.M.V.; Añibarro-Ortega, M.; Pires, T.C.S.P.; Saldanha, A.L.; Alves, M.J.; Nogueira, A.; Ferreira, I.C.F.R.; Barros, L. Extraction of anthocyanins from red raspberry for natural food colorants development: Processes optimization and in vitro bioactivity. Processes 2020, 8, 1447. [Google Scholar] [CrossRef]
- Albuquerque, B.R.; Pinela, J.; Barros, L.; Oliveira, M.B.P.P.; Ferreira, I.C.F.R. Anthocyanin-rich extract of jabuticaba epicarp as a natural colorant: Optimization of heat- and ultrasound-assisted extractions and application in a bakery product. Food Chem. 2020, 316, 126364. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Prieto, M.A.; Barreiro, F.M.F.; Oliveira, M.B.P.; Ferreira, I.C.F.R. Extraction of rosmarinic acid from Melissa officinalis L. by heat-, microwave- and ultrasound-assisted extraction techniques: A comparative study through response surface analysis. Sep. Purif. Technol. 2017, 186, 297–308. [Google Scholar] [CrossRef] [Green Version]
- Clarke, C.J.; Tu, W.-C.; Levers, O.; Bröhl, A.; Hallett, J.P. Green and Sustainable Solvents in Chemical Processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef]
- European Commission. Commission Decision of 9 February 2006 amending Decision 96/335/EC establishing an inventory and a common nomenclature of ingredients employed in cosmetic products. Off. J. Eur. Union 2006, 49, 1–528. [Google Scholar]
- Bessada, S.M.F.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb.f.: An underexploited and highly disseminated species. Ind. Crops Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef] [Green Version]
- Pinela, J.; Barros, L.; Carvalho, A.M.; Ferreira, I.C.F.R. Nutritional composition and antioxidant activity of four tomato (Lycopersicon esculentum L.) farmer’ varieties in Northeastern Portugal homegardens. Food Chem. Toxicol. 2012, 50, 829–834. [Google Scholar] [CrossRef]
- Soković, M.; Glamočlija, J.; Marin, P.D.; Brkić, D.; van Griensven, L.J.L.D. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef] [Green Version]
- Soković, M.; van Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol. 2006, 116, 211–224. [Google Scholar] [CrossRef]
- Pereira, C.; Calhelha, R.C.; Barros, L.; Ferreira, I.C.F.R. Antioxidant properties, anti-hepatocellular carcinoma activity and hepatotoxicity of artichoke, milk thistle and borututu. Ind. Crops Prod. 2013, 49, 61–65. [Google Scholar] [CrossRef]
- Karakashov, B.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Optimisation of polyphenol extraction from Hypericum perforatum (St. John’s Wort) using aqueous glycerol and response surface methodology. J. Appl. Res. Med. Aromat. Plants 2015, 2, 1–8. [Google Scholar] [CrossRef]
- Huang, H.; Belwal, T.; Jiang, L.; Hu, J.; Limwachiranon, J.; Li, L.; Ren, G.; Zhang, X.; Luo, Z. Valorization of lotus byproduct (Receptaculum Nelumbinis) under green extraction condition. Food Bioprod. Process. 2019, 115, 110–117. [Google Scholar] [CrossRef]
- Vieira, V.; Calhelha, R.C.; Barros, L.; Coutinho, J.A.P.; Ferreira, I.C.F.R.; Ferreira, O. Insights on the extraction performance of alkanediols and glycerol: Using Juglans regia L. leaves as a source of bioactive compounds. Molecules 2020, 25, 2497. [Google Scholar] [CrossRef] [PubMed]
- Tubtimdee, C.; Shotipruk, A. Extraction of phenolics from Terminalia chebula Retz with water-ethanol and water-propylene glycol and sugaring-out concentration of extracts. Sep. Purif. Technol. 2011, 77, 339–346. [Google Scholar] [CrossRef]
- Shehata, E.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Extraction optimisation using water/glycerol for the efficient recovery of polyphenolic antioxidants from two Artemisia species. Sep. Purif. Technol. 2015, 149, 462–469. [Google Scholar] [CrossRef]
- Mourtzinos, I.; Anastasopoulou, E.; Petrou, A.; Grigorakis, S.; Makris, D.; Biliaderis, C.G. Optimization of a green extraction method for the recovery of polyphenols from olive leaf using cyclodextrins and glycerin as co-solvents. J. Food Sci. Technol. 2016, 53, 3939–3947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Commission. Directive 2009/32/EC of the European Parliament and of the Council of 23 April 2009 on the approximation of the laws of the Member States on extraction solvents used in the production of foodstuffs and food ingredients. Off. J. Eur. Union 2009, 52, 3–11. [Google Scholar]
- Leichtweis, M.G.; Pereira, C.; Prieto, M.A.; Barreiro, M.F.; Beraldi, I.J.; Barros, L.; Ferreira, I.C.F.R. Ultrasound as a rapid and low-cost extraction procedure to obtain anthocyanin-based colorants from Prunus spinosa L. fruit epicarp: Comparative study with conventional heat-based extraction. Molecules 2019, 24, 573. [Google Scholar] [CrossRef] [Green Version]
- Pinela, J.; Prieto, M.A.; Pereira, E.; Jabeur, I.; Barreiro, M.F.; Barros, L.; Ferreira, I.C.F.R. Optimization of heat- and ultrasound-assisted extraction of anthocyanins from Hibiscus sabdariffa calyces for natural food colorants. Food Chem. 2019, 275, 309–321. [Google Scholar] [CrossRef] [Green Version]
- Vieira, V.; Prieto, M.A.; Barros, L.; Coutinho, J.A.P.; Ferreira, O.; Ferreira, I.C.F.R. Optimization and comparison of maceration and microwave extraction systems for the production of phenolic compounds from Juglans regia L. for the valorization of walnut leaves. Ind. Crops Prod. 2017, 107, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Asnin, L.; Assefa, A.D.; Ko, E.Y.; Sharma, K.; Park, S.W. Extraction of antioxidants from Aloe vera leaf gel: A response surface methodology study. Food Anal. Methods 2014, 7, 1804–1815. [Google Scholar] [CrossRef]
- Bertolín, J.R.; Joy, M.; Blanco, M. Malondialdehyde determination in raw and processed meat products by UPLC-DAD and UPLC-FLD. Food Chem. 2019, 298, 125009. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.N.; Li, W.; Lee, S.H.; Jang, H.D.; Ma, J.Y.; Kim, Y.H. Antioxidant and anti-osteoporotic effects of anthraquinones and related constituents from the aqueous dissolved Aloe exudates. Nat. Prod. Res. 2017, 31, 2810–2813. [Google Scholar] [CrossRef] [PubMed]
- Lucini, L.; Pellizzoni, M.; Pellegrino, R.; Molinari, G.P.; Colla, G. Phytochemical constituents and in vitro radical scavenging activity of different Aloe species. Food Chem. 2015, 170, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Hiruy, M.; Bisrat, D.; Mazumder, A.; Asres, K. Two chromones with antimicrobial activity from the leaf latex of Aloe monticola Reynolds. Nat. Prod. Res. 2021, 35, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Abert Vian, M.; Fabiano-Tixier, A.S.; Nutrizio, M.; Režek Jambrak, A.; Munekata, P.E.S.; Lorenzo, J.M.; Barba, F.J.; Binello, A.; Cravotto, G. A review of sustainable and intensified techniques for extraction of food and natural products. Green Chem. 2020, 22, 2325–2353. [Google Scholar] [CrossRef] [Green Version]
| Run | Experimental CCRD Design Matrix | Aloesin Content (mg/L) | ||||
|---|---|---|---|---|---|---|
| X1: t (min) | X2: T (°C) | X3: S (%) | EtOH-W System | PG-W System | Gly-W System | |
| 1 | 51 (−1) | 39 (−1) | 20 (−1) | 35 ± 1 | 30.0 ± 0.6 | 46 ± 4 |
| 2 | 170 (+1) | 39 (−1) | 20 (−1) | 30.2 ± 0.9 | 24 ± 2 | 45 ± 2 |
| 3 | 51 (−1) | 81 (+1) | 20 (−1) | 30 ± 1 | 38.7 ± 0.9 | 39.7 ± 0.8 |
| 4 | 170 (+1) | 81 (+1) | 20 (−1) | 29.8 ± 0.8 | 31 ± 2 | 44.4 ± 0.6 |
| 5 | 51 (−1) | 39 (−1) | 80 (+1) | 9.5 ± 0.1 | 28± 4 | 27 ± 2 |
| 6 | 170 (+1) | 39 (−1) | 80 (+1) | 9.3 ± 0.1 | 28.1 ± 0.6 | 31.3 ± 0.7 |
| 7 | 51 (−1) | 81 (+1) | 80 (+1) | 9.5 ± 0.1 | 20.6 ± 0.3 | 21 ± 3 |
| 8 | 170 (+1) | 81 (+1) | 80 (+1) | 13.85 ± 0.07 | 20.8 ± 0.1 | 21 ± 1 |
| 9 | 10 (−1.68) | 60 (0) | 50 (0) | 33.4 ± 0.9 | 64 ± 1 | 52 ± 3 |
| 10 | 210 (+1.68) | 60 (0) | 50 (0) | 23 ± 1 | 33.7 ± 0.4 | 42 ± 1 |
| 11 | 110 (0) | 25 (−1.68) | 50 (0) | 11.1 ± 0.2 | 37 ± 1 | 57 ± 3 |
| 12 | 110 (0) | 95 (+1.68) | 50 (0) | 16.5 ± 0.2 | 33 ± 1 | 55.2 ± 0.7 |
| 13 | 110 (0) | 60 (0) | 0 (−1.68) | 48 ± 1 | 47.7 ± 0.3 | 47.9 ± 0.7 |
| 14 | 110 (0) | 60 (0) | 100 (+1.68) | tr | 20.2 ± 0.8 | 28.29 ± 0.01 |
| 15 | 110 (0) | 60 (0) | 50 (0) | 35 ± 1 | 35 ± 3 | 40 ± 2 |
| 16 | 110 (0) | 60 (0) | 50 (0) | 33 ± 1 | 29.0 ± 0.1 | 39 ± 4 |
| 17 | 110 (0) | 60 (0) | 50 (0) | 33 ± 2 | 37 ± 1 | 43 ± 4 |
| 18 | 110 (0) | 60 (0) | 50 (0) | 34 ± 1 | 37 ± 2 | 40 ± 6 |
| 19 | 110 (0) | 60 (0) | 50 (0) | 34 ± 2 | 31 ± 3 | 41 ± 2 |
| 20 | 110 (0) | 60 (0) | 50 (0) | 34 ± 2 | 38 ± 1 | 36.5 ± 0.5 |
| Coefficients # | EtOH-W System | PG-W System | Gly-W System | |
|---|---|---|---|---|
| Intercept | b0 | 33.9 ± 0.4 | 33.0 ± 0.8 | 40 ± 1 |
| Linear terms | b1 | −2.9 ± 0.4 | −9.0 ± 0.9 | −3 ± 1 |
| b2 | −1.3 ± 0.6 * | −1.1 ± 0.9 * | −1 ± 1 * | |
| b3 | −14.1 ± 0.4 | −8.2 ± 0.9 | −6 ± 1 | |
| Quadratic terms | b11 | −2.0 ± 0.3 | 5.6 ± 0.6 | 2.5 ± 0.8 |
| b22 | −7.2 ± 0.3 | ns | 5.7 ± 0.8 | |
| b33 | −3.7 ± 0.3 | ns | ns | |
| Cubic terms | b222 | 0.9 ± 0.3 | ns | ns |
| Interaction terms | b12 | 1.1 ± 0.4 | −0.8 ± 0.3 * | 0.9 ± 0.4 * |
| b13 | 1.1 ± 0.4 | 1.8 ± 0.8 | 0.9 ± 0.4 * | |
| b23 | 1.2 ± 0.4 | −3.9 ± 0.8 | ns | |
| b113 | 3.9 ± 0.6 | 5 ± 1 | −4 ± 1 | |
| b122 | 2.9 ± 0.6 | 7 ± 1 | 4 ± 1 | |
| b1122 | ns | −12 ± 1 | −11 ± 1 | |
| Statistical Data | Model F-value | 232.64 | 29.53 | 23.54 |
| Lack of Fit | ns | ns | ns | |
| R2 | 0.9904 | 0.9291 | 0.9099 | |
| R2adj | 0.9862 | 0.8976 | 0.8699 | |
| Ad. Precision | 57.21 | 24.32 | 17.3697 | |
| C.V. (%) | 5.80 | 9.85 | 9.32 | |
| Optimal Conditions | X1 (min) | 92.9 | 12.0 | 42.2 |
| X2 (°C) | 55.9 | 60.6 | 56.8 | |
| X3 (%, w/w) | 0.0 | 51.1 | 17.5 | |
| Response Optimum | Model-predicted | 48 ± 1 mg/L | 63 ± 2 mg/L | 57 ± 2 mg/L |
| Experimental | 51 ± 4 mg/L | 65 ± 4 mg/L | 61 ± 3 mg/L |
| EtOH-W Extract | PG-W Extract | Gly-W Extract | Positive Control * | |||||
|---|---|---|---|---|---|---|---|---|
| TBARS (EC50, µg/mL) # | 310 ± 21 a | 432 ± 18 b | 610 ± 13 c | 5.4 ± 0.3 | ||||
| Antibacterial Activity | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Staphylococcus aureus | 1.5 | 3 | 1.5 | 2 | 2 | 4 | 0.006 | 0.012 |
| Staphylococcus epidermidis | 1.5 | 2 | 1.5 | 2 | na | na | 0.003 | 0.006 |
| Staphylococcus lugdunensis | 2 | 3 | 1 | 2 | 2 | 4 | 0.025 | 0.05 |
| Micrococcus flavus | 2 | 4 | 1.5 | 2 | na | na | 0.2 | 0.3 |
| Listeria monocytogenes | 2 | 4 | 0.25 | 0.5 | na | na | 0.2 | 0.3 |
| Escherichia coli | 3 | 4 | 1 | 2 | 1.5 | 2 | 0.006 | 0.012 |
| Pseudomonas aeruginosa | na | na | 0.5 | 1 | 1.5 | 2 | 0.025 | 0.05 |
| Salmonella enterica serovar Typhimurium | 2 | 3 | 0.25 | 0.5 | na | na | 0.25 | 0.5 |
| Antifungal Activity | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC |
| Aspergillus flavus | 1.5 | 2 | 1 | 2 | na | na | 0.25 | 0.5 |
| Aspergillus niger | 2 | 4 | 1.5 | 2 | na | na | 0.2 | 0.5 |
| Penicillium funiculosum | 1 | 2 | 1 | 2 | na | na | 0.2 | 0.5 |
| Candida albicans | 2 | 4 | 0.7 | 1 | 2 | 4 | 0.4 | 0.8 |
| Trichophyton mentagrophytes | 1 | 2 | 0.12 | 1 | 2 | 4 | 0.012 | 0.025 |
| Trichophyton tonsurans | 0.5 | 1 | 0.25 | 0.5 | 2 | 4 | 0.0015 | 0.003 |
| Microsporum gypseum | 1 | 2 | 0.5 | 1 | 1 | 2 | 0.006 | 0.012 |
| Microsporum canis | 1 | 2 | 1 | 2 | 0.5 | 1 | 0.003 | 0.006 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Añibarro-Ortega, M.; Pinela, J.; Ćirić, A.; Lopes, E.; Molina, A.K.; Calhelha, R.C.; Soković, M.; Ferreira, O.; Ferreira, I.C.F.R.; Barros, L. Extraction of Aloesin from Aloe vera Rind Using Alternative Green Solvents: Process Optimization and Biological Activity Assessment. Biology 2021, 10, 951. https://doi.org/10.3390/biology10100951
Añibarro-Ortega M, Pinela J, Ćirić A, Lopes E, Molina AK, Calhelha RC, Soković M, Ferreira O, Ferreira ICFR, Barros L. Extraction of Aloesin from Aloe vera Rind Using Alternative Green Solvents: Process Optimization and Biological Activity Assessment. Biology. 2021; 10(10):951. https://doi.org/10.3390/biology10100951
Chicago/Turabian StyleAñibarro-Ortega, Mikel, José Pinela, Ana Ćirić, Elsa Lopes, Adriana K. Molina, Ricardo C. Calhelha, Marina Soković, Olga Ferreira, Isabel C. F. R. Ferreira, and Lillian Barros. 2021. "Extraction of Aloesin from Aloe vera Rind Using Alternative Green Solvents: Process Optimization and Biological Activity Assessment" Biology 10, no. 10: 951. https://doi.org/10.3390/biology10100951













