Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrument
2.2. Preparation of CMC/SA/CS Composite Films
2.3. Thickness
2.4. Light Transmittance
2.5. Water Absorption
2.6. Moisture Permeability Coefficient
2.7. Tensile Strength
2.8. Elongation after Fracture
2.9. Oil Permeability Coefficient
2.10. Surface Morphology
2.11. Infrared Spectroscopy Analysis
2.12. Antibacterial Testing
3. Results and Discussion
3.1. Results of the Completely Randomized Design
3.2. Results of Orthogonal Experiment
3.3. The Morphology of Films
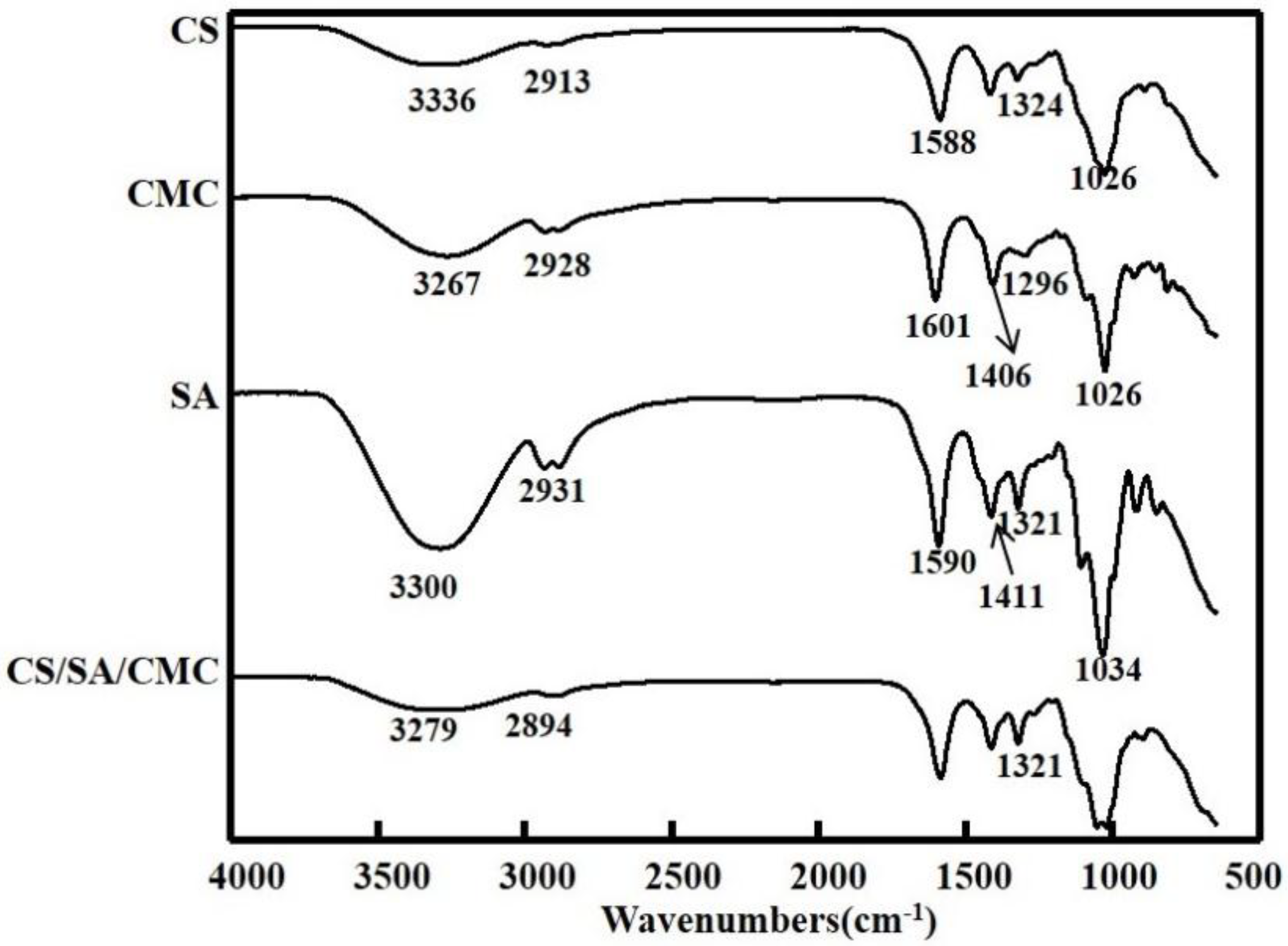
3.4. FTIR Analysis
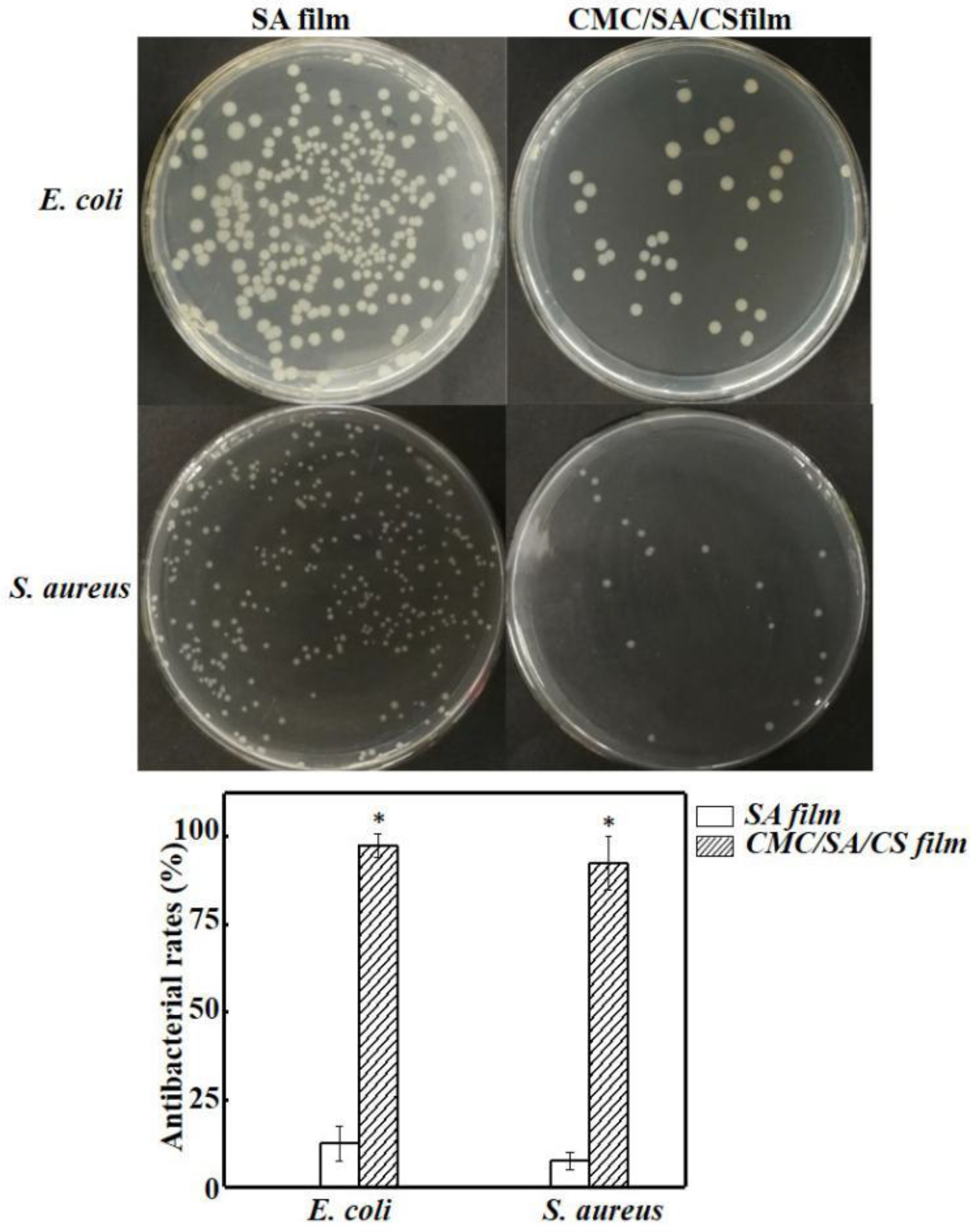
3.5. Analysis of the Antibacterial Activity
3.6. Film Performance Comparison
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Steinmetz, Z.; Wollmann, C.; Schaefer, M.; Buchmann, C.; David, J.; Troger, J.; Munoz, K.; Fror, O.; Schaumann, G.E. Plastic mulching in agriculture. Trading short-term agronomic benefits for long-term soil degradation. Sci. Total. Environ. 2016, 550, 690–705. [Google Scholar] [CrossRef] [PubMed]
- Onwudili, J.A.; Williams, P.T. Catalytic supercritical water gasification of plastics with supported RuO2: A potential solution to hydrocarbons-water pollution problem. Process Saf. Environ. Prot. 2016, 102, 140–149. [Google Scholar] [CrossRef]
- Yang, S.; Lei, P.; Shan, Y.; Zhang, D. Preparation and characterization of antibacterial electrospun chitosan/poly (vinyl alcohol)/graphene oxide composite nanofibrous membrane. Appl. Surf. Sci. 2018, 435, 832–840. [Google Scholar] [CrossRef]
- Xu, C.; Chen, C.; Wu, D. The starch nanocrystal filled biodegradable poly(ε-caprolactone) composite membrane with highly improved properties. Carbohydr. Polym. 2018, 182, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Chen, C.; Rosswurm, K.; Yao, T.; Janaswamy, S. A facile route to prepare cellulose-based films. Carbohydr. Polym. 2016, 149, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Song, X.; Ma, Z.; Zhang, X.; Shu, D.; Nan, J. Al2O3/PVdF-HFP-CMC/PE separator prepared using aqueous slurry and post-hot-pressing method for polymer lithium-ion batteries with enhanced safety. Electrochim. Acta 2016, 212, 416–425. [Google Scholar] [CrossRef]
- Yang, W.; Wang, J.; Yang, Q.; Pei, H.; Hu, N.; Suo, Y.; Li, Z.; Zhang, D.; Wang, J. Facile fabrication of robust MOF membranes on cloth via a CMC macromolecule bridge for highly efficient Pb(II) removal. Chem. Eng. J. 2018, 339, 230–239. [Google Scholar] [CrossRef]
- Liu, S.; Li, Y.; Li, L. Enhanced stability and mechanical strength of sodium alginate composite films. Carbohydr. Polym. 2017, 160, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Varaprasad, K.; Raghavendra, G.M.; Jayaramudu, T.; Seo, J. Nano zinc oxide–sodium alginate antibacterial cellulose fibres. Carbohydr. Polym. 2016, 135, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Bano, S.; Mahmood, A.; Kim, S.J.; Lee, K.H. Chlorine resistant binary complexed NaAlg/PVA composite membrane for nanofiltration. Sep. Purif. Technol. 2014, 137, 21–27. [Google Scholar] [CrossRef]
- Habiba, U.; Afifi, A.M.; Salleh, A.; Ang, B.C. Chitosan/(polyvinyl alcohol)/zeolite electrospun composite nanofibrous membrane for adsorption of Cr6+, Fe3+ and Ni2+. Sep. Purif. Technol. 2017, 322, 182–194. [Google Scholar] [CrossRef]
- Ibarra, V.G.; Sendón, R.; de Quiros, A.R.-B. Antimicrobial food packaging based on biodegradable materials. Antimicrob. Food Packag. 2016, 363–384. [Google Scholar] [CrossRef]
- Wang, B.; Yang, X.; Qiao, C.; Li, Y.; Li, T.; Xu, C. Effects of chitosan quaternary ammonium salt on the physicochemical properties of sodium carboxymethyl cellulose-based films. Carbohydr. Polym. 2018, 184, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Yu, M.; Wang, L. Physical and antimicrobial properties of sodium alginate/carboxymethyl cellulose films incorporated with cinnamon essential oil. Food Pack. Shelf Life 2017, 15, 35–42. [Google Scholar] [CrossRef]
- Kawasaki, T.; Nakaji-Hirabayashi, T.; Masuyama, K.; Fujita, S.; Kitano, H. Complex film of chitosan and carboxymethyl cellulose nanofibers. Colloids Surf. B 2016, 139, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Noshirvani, N.; Ghanbarzadeh, B.; Gardrat, C.; Rezaei, M.R.; Hashemi, M.; Le Coz, C.; Coma, V. Cinnamon and ginger essential oils to improve antifungal, physical and mechanical properties of chitosan-carboxymethyl cellulose films. Food Hydrocolloid 2017, 70, 36–45. [Google Scholar] [CrossRef]
- Zhuang, C.; Jiang, Y.; Zhong, Y.; Zhao, Y.; Deng, Y.; Yue, J.; Wang, D.; Jiao, S.; Gao, H.; Chen, H.; et al. Development and characterization of nano-bilayer films composed of polyvinyl alcohol, chitosan and alginate. Food Control 2018, 86, 191–199. [Google Scholar] [CrossRef]
- Bajpai, S.K.; Chand, N.; Ahuja, S. Investigation of curcumin release from chitosan/cellulose micro crystals (CMC) antimicrobial films. Int. J. Boil. Macromol. 2015, 79, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, Y.; He, W.; Wang, C.; Sun, Y.; Qiao, K.; Wang, X.; Gao, L. Reprint of: Preparation of a novel sodium alginate/polyvinyl formal composite with a double crosslinking interpenetrating network for multifunctional biomedical application. Compos. Part B Eng. 2017, 121, 9–22. [Google Scholar] [CrossRef]
- Ahmed, S.; Ikram, S. Chitosan and gelatin based biodegradable packaging films with UV-light protection. J. Photochem. Photobiol. B 2016, 163, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Alsammarraie, F.K.; Nayigiziki, F.X.; Wang, W.; Vardhanabhuti, B.; Mustapha, A.; Lin, M. Effect and mechanism of cellulose nanofibrils on the active functions of biopolymer-based nanocomposite films. Food Res. Int. 2017, 99, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Ercelik, M.; Ozden, A.; Devrim, Y.; Colpan, C.O. Investigation of Nafion based composite membranes on the performance of DMFCs. Int. J. Hydrogen Energy 2017, 42, 2658–2668. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, K.; Wu, J.; Ren, G.; Chen, H.; Xu, J. Bio-nanocomposite films reinforced with organo-modified layered double hydroxides: Preparation, morphology and properties. Appl. Clay Sci. 2016, 126, 72–80. [Google Scholar] [CrossRef]
- Wang, P.; Wang, H.; Liu, J.; Wang, P.; Jiang, S.; Li, X.; Jiang, S. Montmorillonite@chitosan-poly(ethylene oxide) nanofibrous membrane enhancing poly(vinyl alcohol-co-ethylene) composite film. Carbohydr. Polym. 2018, 181, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Kida, T.; Ohta, T.; Kondo, K.; Akashi, M. Fabrication of poly(vinyl alcohol)s (PVAs) nanotubes through the fusion of nanocapsules composed of PVAs multilayer films. Polymer 2014, 55, 2841–2847. [Google Scholar] [CrossRef]
- Liu, Y.; Ji, P.; Lv, H.; Qin, Y.; Deng, L. Gentamicin modified chitosan film with improved antibacterial property and cell biocompatibility. Int. J. Biol. Macromol. 2017, 98, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Nouri, A.; Yaraki, M.T.; Ghorbanpour, M.; Agarwal, S.; Gupta, V.K. Enhanced antibacterial effect of chitosan film using Montmorillonite/CuO nanocomposite. Int. J. Biol. Macromol. 2017, 109, 1219–1231. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Wang, H.; Wang, L. Physical properties and antibacterial activity of quaternized chitosan/carboxymethyl cellulose blend films. LWT-Food Sci. Technol. 2016, 65, 398–405. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, G.; Wang, X.; Wang, L.; Hu, M.; Wang, Z.; Tao, Y. Efforts on membrane properties and enzymes by adding divalent cations and sodium carboxymethyl cellulose. Carbohydr. Polym. 2014, 106, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Zhivkov, A.M.; Hristov, R.P. Adsorption of carboxymethyl cellulose on alumina particles. J. Colloid Interface Sci. 2015, 447, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Vild, A.; Teixeira, S.; Kühn, K.; Cuniberti, G.; Sencadas, V. Orthogonal experimental design of titanium dioxide-poly(methyl methacrylate) electrospun nanocomposite membranes for photocatalytic applications. J. Environ. Chem. Eng. 2016, 4, 3151–3158. [Google Scholar] [CrossRef]
- Ebrahimi, B.; Mohammadi, R.; Rouhi, M.; Mortazavian, A.M.; Shojaee-Aliabadi, S.; Koushki, M.R. Survival of probiotic bacteria in carboxymethyl cellulose-based edible film and assessment of quality parameters. LWT-Food Sci. Technol. 2018, 87, 54–60. [Google Scholar] [CrossRef]
- Atef, M.; Rezaei, M.; Behrooz, R. Preparation and characterization agar-based nanocomposite film reinforced by nanocrystalline cellulose. Int. J. Biol. Macromol. 2014, 70, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Li, D.; Huo, Q.; Zhao, Q.; Lan, Q.; Cui, M.; Pan, W.; Yang, X. Two kinds of ketoprofen enteric gel beads (CA and CS-SA) using biopolymer alginate. Asian J. Pharm. Sci. 2017, 13, 120–130. [Google Scholar] [CrossRef]
- Kingkaew, J.; Kirdponpattara, S.; Sanchavanakit, N.; Pavasant, P.; Phisalaphong, M. Effect of molecular weight of chitosan on antimicrobial properties and tissue compatibility of chitosan-impregnated bacterial cellulose films. Biotechnol. Bioprocess E 2014, 19, 534–544. [Google Scholar] [CrossRef]
- Kanmani, P.; Lim, S.T. Development and characterization of novel probiotic-residing pullulan/starch edible films. Food Chem. 2013, 141, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimzadeh, S.; Ghanbarzadeh, B.; Hamishehkar, H. Physical properties of carboxymethyl cellulose based nano-biocomposites with graphene nano-platelets. Int. J. Biol. Macromol. 2016, 84, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Paunonen, S. Nanocellulose-based food packaging materials—A review. Nord. Pulp Pap. Res. J. 2013, 28, 165–181. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Fernando, A.L.; Pires, J.R.A.; Rodrigues, P.F.; Lopes, A.A.S.; Fernandes, F.M.B. Physical properties of chitosan films incorporated with natural antioxidants. Ind. Crop. Prod. 2017, 107, 565–572. [Google Scholar] [CrossRef]
- Bonilla, J.; Talon, E.; Atares, L.; Vargas, M.; Chiralt, A. Effect of the incorporation of antioxidants on physicochemical and antioxidant properties of wheat starchechitosan films. J. Food Eng. 2013, 118, 271–278. [Google Scholar] [CrossRef]
- Sheela, T.; Bhajantri, R.F.; Nambissan, P.M.G.; Ravindrachary, V.; Lobo, B.; Naik, J.; Rathod, S.G. Ionic conductivity and free volume related microstructural properties of LiClO4/PVA/NaAlg polymer composites: Positron annihilation spectroscopic studies. J. Non-Cryst. Solids 2016, 454, 19–30. [Google Scholar] [CrossRef]
- Sheela, T.; Bhajantri, R.F.; Ravindrachary, V.; Rathod, S.G.; Pujari, P.K.; Poojary, B.; Somashekar, R. Effect of UV irradiation on optical, mechanical and microstructural properties of PVA/NaAlg blends. Radiat. Phys. Chem. 2014, 103, 45–52. [Google Scholar] [CrossRef]
- Li, J.; Ma, J.; Chen, S.; He, J.; Huang, Y. Characterization of calcium alginate/deacetylated konjac glucomannan blend films prepared by Ca2+ crosslinking and deacetylation. Food Hydrocolloids 2018, 82, 363–369. [Google Scholar] [CrossRef]
- Gholizadeh, B.S.; Buazar, F.; Hosseini, S.M.; Mousavi, S.M. Enhanced antibacterial activity, mechanical and physical properties of alginate/hydroxyapatite bionanocomposite film. Int. J. Biol. Macromol. 2018, 116, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, H.M.S.; Riaz, A.; Hamed, Y.S.; Abdin, M.; Chen, G.; Wan, P.; Zeng, X. Production and characterization of CMC-based antioxidant and antimicrobial films enriched with chickpea hull polysaccharides. Int. J. Biol. Macromol. 2018, 118, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Esteghlal, S.; Niakousari, M.; Hosseini, S.M.H. Physical and mechanical properties of gelatin-CMC composite films under the influence of electrostatic interactions. Int. J. Biol. Macromol. 2018, 114, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Rhim, J.W. Preparation and characterization of agar/lignin/silver nanoparticles composite films with ultraviolet light barrier and antibacterial properties. Food Hydrocolloids 2017, 71, 76–84. [Google Scholar] [CrossRef]
- Ren, H.; Gao, Z.; Wu, D.; Jiang, J.; Sun, Y.; Luo, C. Efficient Pb(II) removal using sodium alginate–carboxymethyl cellulose gel beads: Preparation, characterization, and adsorption mechanism. Carbohydr. Polym. 2016, 137, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Luo, Y.; Lin, Y.; Li, Q.; Yu, P.; Zeng, M. Study of pervaporation for dehydration of caprolactam through blend NaAlg-poly(vinyl pyrrolidone) membranes on PAN supports. Sep. Purif. Technol. 2010, 74, 242–252. [Google Scholar] [CrossRef]
- Branca, C.; D’Angelo, G.; Crupi, C.; Khouzami, K.; Rifici, S.; Ruello, G.; Wanderlingh, U. Role of the OH and NH vibrational groups in polysaccharide-nanocomposite interactions: A FTIR-ATR study on chitosan and chitosan/clay films. Polymer 2016, 99, 614–622. [Google Scholar] [CrossRef]
- Ashrafi, A.; Jokar, M.; Nafchi, A.M. Preparation and characterization of biocomposite film based on chitosan and kombucha tea as active food packaging. Int. J. Biol. Macromol. 2018, 108, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Campana, R.; Casettari, L.; Ciandrini, E.; Illum, L.; Baffone, W. Chitosans inhibit the growth and the adhesion of klebsiella pneumonia and escherichia coli clinical isolates on urinary catheters. Int. J. Antimicrob. Agents 2017, 50, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Doulabi, A.H.; Mirzadeh, H.; Imani, M.; Samadi, N. Chitosan/polyethylene glycol fumarate blend film: Physical and antibacterial properties. Carbohydr. Polym. 2013, 92, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, S.; Lan, W. Fabrication of antibacterial chitosan-PVA blended film using electrospray technique for food packaging applications. Int. J. Biol. Macromol. 2017, 107, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Rafique, A.; Zia, K.M.; Zuber, M.; Tabasum, S.; Rehman, S. Chitosan functionalized poly(vinyl alcohol) for prospects biomedical and industrial applications: A review. Int. J. Biol. Macromol. 2016, 87, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Khazaei, N.; Esmaiili, M.; Djomeh, Z.E.; Ghasemlou, M.; Jouki, M. Characterization of new biodegradable edible film made from basil seed (Ocimum basilicum L.) gum. Carbohydr. Polym. 2014, 102, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.B.; Bozell, J.J.; Hayes, D.G.; Zivanovic, S. Introduction of primary antioxidant activity to chitosan for application as a multifunctional food packaging material. Food Hydrocolloids 2013, 33, 207–214. [Google Scholar] [CrossRef]
- Treenate, P.; Monvisade, P.; Yamaguchi, M. Development of hydroxyethylacryl chitosan/alginate hydrogel films for biomedical application. J. Polym. Res. 2014, 21, 601. [Google Scholar] [CrossRef]

| Samples | Thickness (μm) | Tensile Strength (MPa) | Elongation After Fracture (%) |
|---|---|---|---|
| SA-0.5% | 53.7 ± 1.10 | 46.48 ± 6.57 | 6.15 ± 1.07 |
| SA-1.0% | 67.3 ± 1.60 | 59.39 ± 9.89 | 5.03 ± 0.75 |
| SA-1.5% | 74.1 ± 2.10 | 84.19 ± 12.86 | 6.44 ± 1.16 |
| SA-2.0% | 97.8 ± 2.80 | 67.21 ± 10.33 | 7.67 ± 1.31 |
| CS-0.5% | 59.4 ± 1.40 | 54.59 ± 7.55 | 4.47 ± 0.52 |
| CS-1.0% | 67.3 ± 1.50 | 59.39 ± 9.86 | 5.03 ± 0.91 |
| CS-1.5% | 85.5 ± 2.50 | 61.39 ± 10.07 | 5.43 ± 0.86 |
| CS-2.0% | 112.7 ± 3.10 | 54.18 ± 7.59 | 5.68 ± 0.91 |
| CMC-0.5% | 70.2 ± 1.90 | 56.12 ± 8.89 | 9.56 ± 2.17 |
| CMC-1.0% | 65.4 ± 1.50 | 78.23 ± 11.74 | 5.20 ± 0.81 |
| CMC-1.5% | 67.6 ± 1.60 | 59.39 ± 9.86 | 5.03 ± 0.75 |
| CMC-2.0% | 74.9 ± 2.20 | 59.51 ± 9.59 | 8.98 ± 1.51 |
| Level | Factors | ||
|---|---|---|---|
| SA (%) | CS (%) | CMC (%) | |
| 1 | 1.0 | 1.0 | 0.5 |
| 2 | 1.5 | 1.5 | 1.0 |
| 3 | 2.0 | 2.0 | 1.5 |
| Level | Factors | ||
|---|---|---|---|
| A | B | C | |
| 1 | A1 | B1 | C1 |
| 2 | A1 | B2 | C3 |
| 3 | A1 | B3 | C2 |
| 4 | A2 | B1 | C2 |
| 5 | A2 | B2 | C2 |
| 6 | A2 | B3 | C3 |
| 7 | A3 | B1 | C3 |
| 8 | A3 | B2 | C1 |
| 9 | A3 | B3 | C1 |
| K1 | 35.82 | 34.87 | 36.86 |
| K2 | 36.34 | 38.64 | 32.87 |
| K3 | 29.92 | 28.57 | 32.35 |
| R | 6.42 | 10.07 | 4.51 |
| Optimal level | A2 | B2 | C1 |
| Samples | Thickness (mm) | Tensile Strength (MPa) | Elongation after Fracture (%) | Water Uptake (%) |
| 1 | 70 ± 3.10 | 56.12 ± 2.11 | 9.56 ± 0.98 | 2.83 ± 0.12 |
| 2 | 75 ± 2.90 | 54.18 ± 3.12 | 5.68 ± 0.36 | 23.00 ± 1.02 |
| 3 | 96 ± 3.60 | 69.95 ± 2.07 | 15.51 ± 0.32 | 12.50 ± 0.89 |
| 4 | 97 ± 4.00 | 61.12 ± 1.81 | 30.46 ± 0.13 | 6.33 ± 0.09 |
| 5 | 125 ± 5.10 | 49.55 ± 1.98 | 3.47 ± 0.39 | 6.00 ± 0.13 |
| 6 | 134 ± 4.20 | 54.96 ± 2.22 | 13.05 ± 0.87 | 5.50 ± 0.32 |
| 7 | 108 ± 4.90 | 62.34 ± 2.43 | 19.51 ± 1.20 | 2.75 ± 0.17 |
| 8 | 129 ± 4.90 | 52.24 ± 1.19 | 7.35 ± 0.69 | 8.00 ± 0.28 |
| 9 | 153 ± 2.30 | 84.19 ± 0.53 | 6.44 ± 0.31 | 4.00 ± 0.03 |
| Samples | Oil Permeability Coefficient | Water Vapor Permeation | Light Transmittance (%) | |
| 1 | 95 ± 2.09 | 1271.73 ± 5.03 | 81.85 ± 1.07 | |
| 2 | 156 ± 3.18 | 1292.93 ± 6.45 | 77.63 ± 1.78 | |
| 3 | 25 ± 0.75 | 1272.93 ± 2.19 | 68.39 ± 3.56 | |
| 4 | 289 ± 3.19 | 1382.13 ± 4.19 | 23.50 ± 2.13 | |
| 5 | 171 ± 3.98 | 1290.00 ± 3.32 | 33.34 ± 0.19 | |
| 6 | 163 ± 0.47 | 1289.20 ± 2.18 | 54.20 ± 2.00 | |
| 7 | 178 ± 5.00 | 1528.27 ± 3.76 | 82.22 ± 3.13 | |
| 8 | 241 ± 7.20 | 1220.93 ± 4.10 | 97.05 ± 2.50 | |
| 9 | 45 ± 1.45 | 1356.13 ± 1.18 | 55.72 ± 1.72 |
| Samples | Tensile Strength | Elongation after Fracture | Water Uptake (%) | Oil Permeability Coefficient | Water Permeability Coefficient | Light Transmittance (%) | Total Score |
|---|---|---|---|---|---|---|---|
| 1 | 2.71 | 3.01 | 9.87 | 7.60 | 8.44 | 8.14 | 39.77 |
| 2 | 2.20 | 1.73 | 1.00 | 5.52 | 7.82 | 7.63 | 25.90 |
| 3 | 6.30 | 4.97 | 5.62 | 10.00 | 8.40 | 6.49 | 41.78 |
| 4 | 4.01 | 10.00 | 8.33 | 1.00 | 5.24 | 1.00 | 29.58 |
| 5 | 1.00 | 1.00 | 8.48 | 5.01 | 7.91 | 2.20 | 25.60 |
| 6 | 2.41 | 4.16 | 8.70 | 5.28 | 7.93 | 4.76 | 33.24 |
| 7 | 4.33 | 6.29 | 10.00 | 4.77 | 1.00 | 8.19 | 34.58 |
| 8 | 1.70 | 2.28 | 7.60 | 2.63 | 10.00 | 10.00 | 34.21 |
| 9 | 10.00 | 1.98 | 9.36 | 9.30 | 5.99 | 4.94 | 41.57 |
| Index | The Relations of Affect | Range |
|---|---|---|
| Tensile strength | B > C > A | 15.56 > 12.96 > 6.13 |
| Elongation after fracture | B > A > C | 14.17 > 8.86 > 5.5 |
| Water uptake | B > A > C | 9.14 > 7.75 > 6.33 |
| Oil permeability coefficient | A > B > C | 120.67 > 83.33 > 20.33 |
| Moisture Permeability Coefficient | A > B > C | 120.93 > 117.42 > 57.07 |
| Light transmittance | C > A > B | 30.27 > 29.61 > 24.56 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lan, W.; He, L.; Liu, Y. Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film. Coatings 2018, 8, 291. https://doi.org/10.3390/coatings8080291
Lan W, He L, Liu Y. Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film. Coatings. 2018; 8(8):291. https://doi.org/10.3390/coatings8080291
Chicago/Turabian StyleLan, Wenting, Li He, and Yaowen Liu. 2018. "Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film" Coatings 8, no. 8: 291. https://doi.org/10.3390/coatings8080291
APA StyleLan, W., He, L., & Liu, Y. (2018). Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film. Coatings, 8(8), 291. https://doi.org/10.3390/coatings8080291




