Characterization of Ultrasonic-Assisted Electrochemical Deposition of Ni-Co-ZrO2
Abstract
1. Introduction
2. Materials and Methods
3. Results
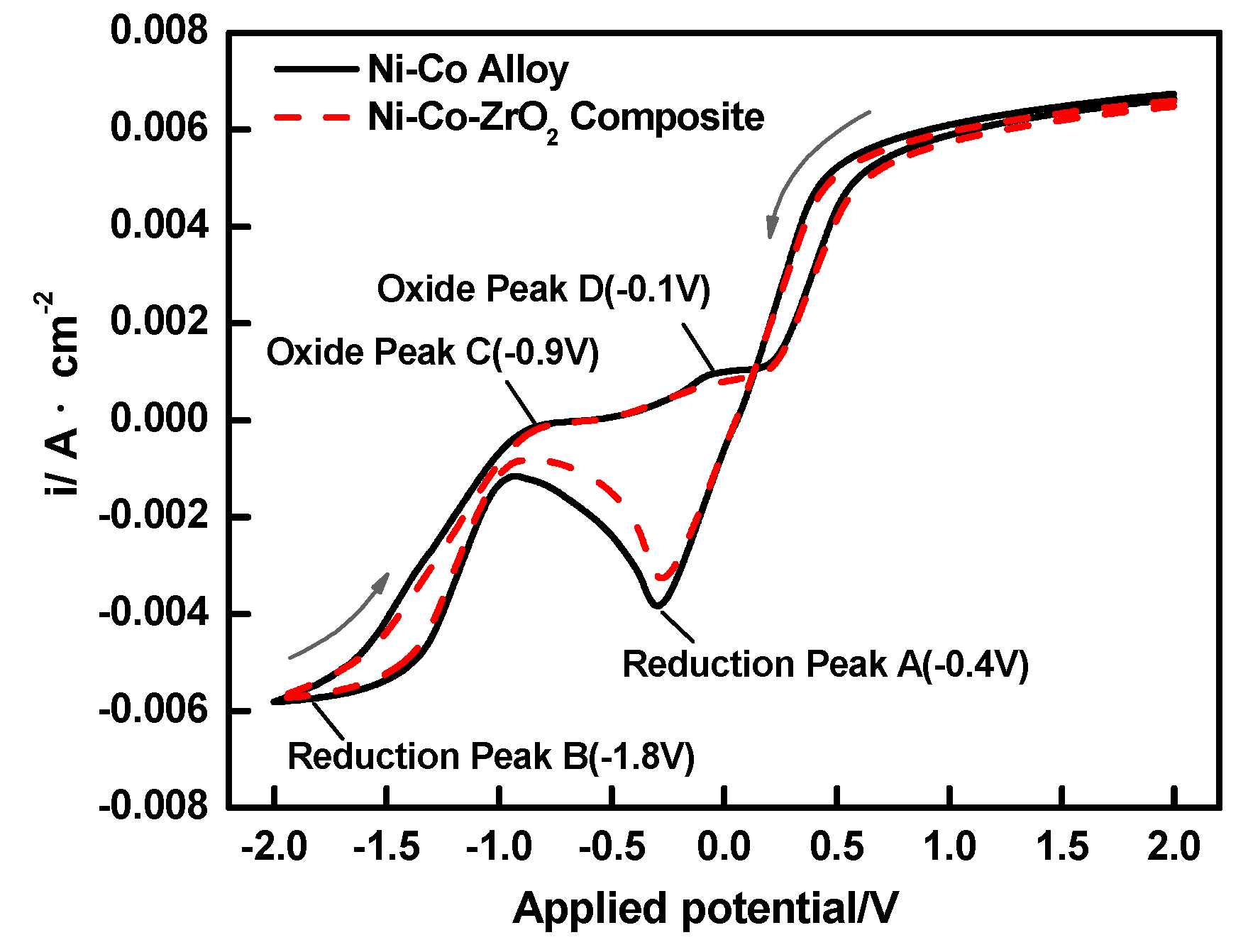
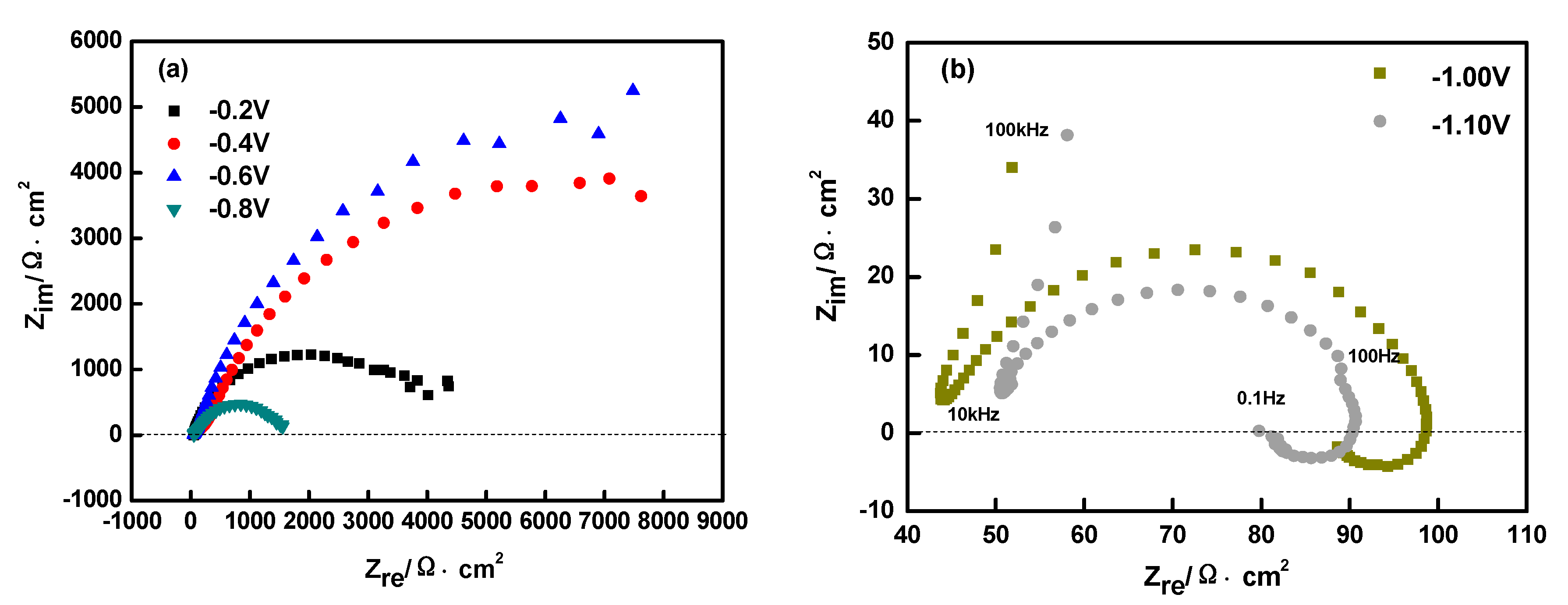
3.1. Cylic Voltammetry (CV) Studies
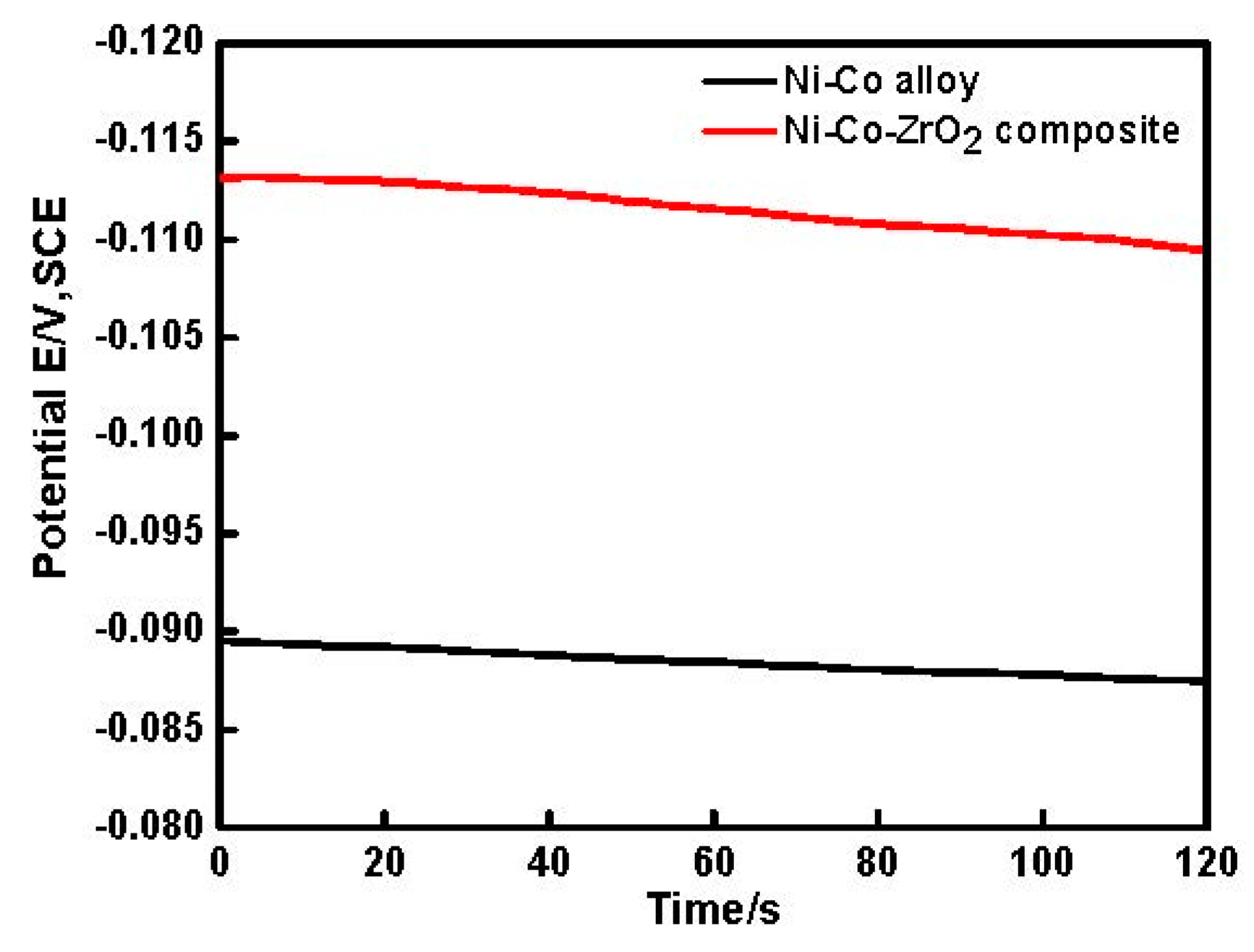
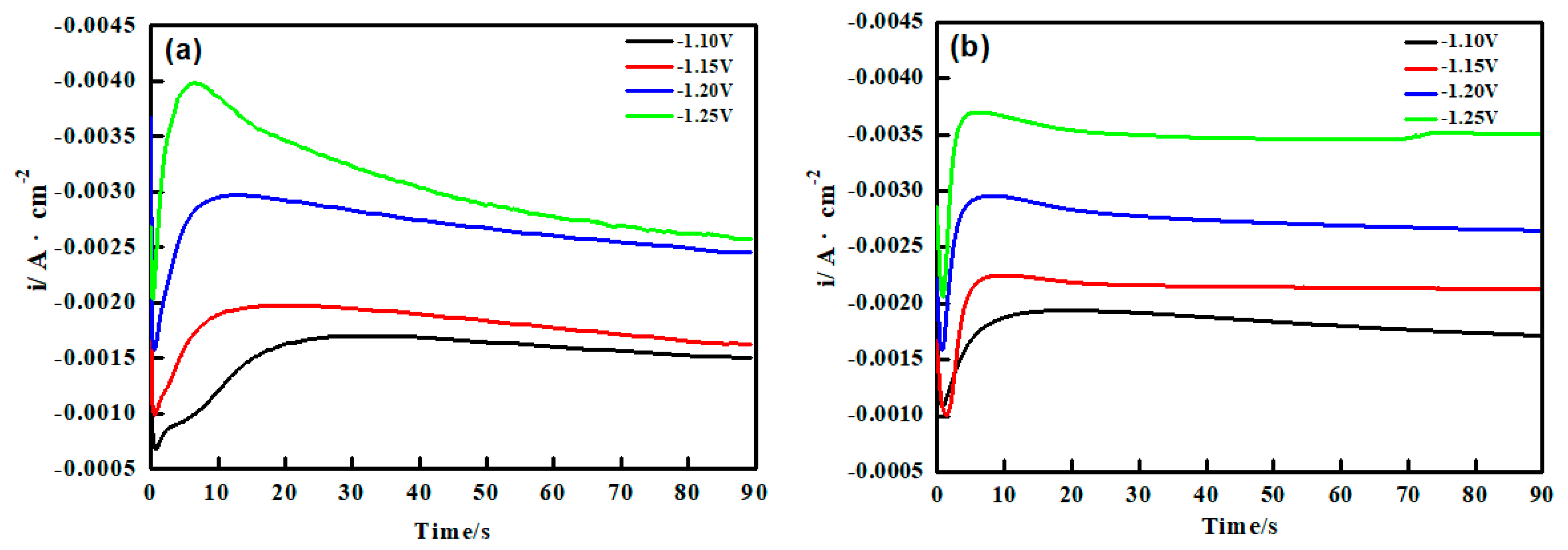
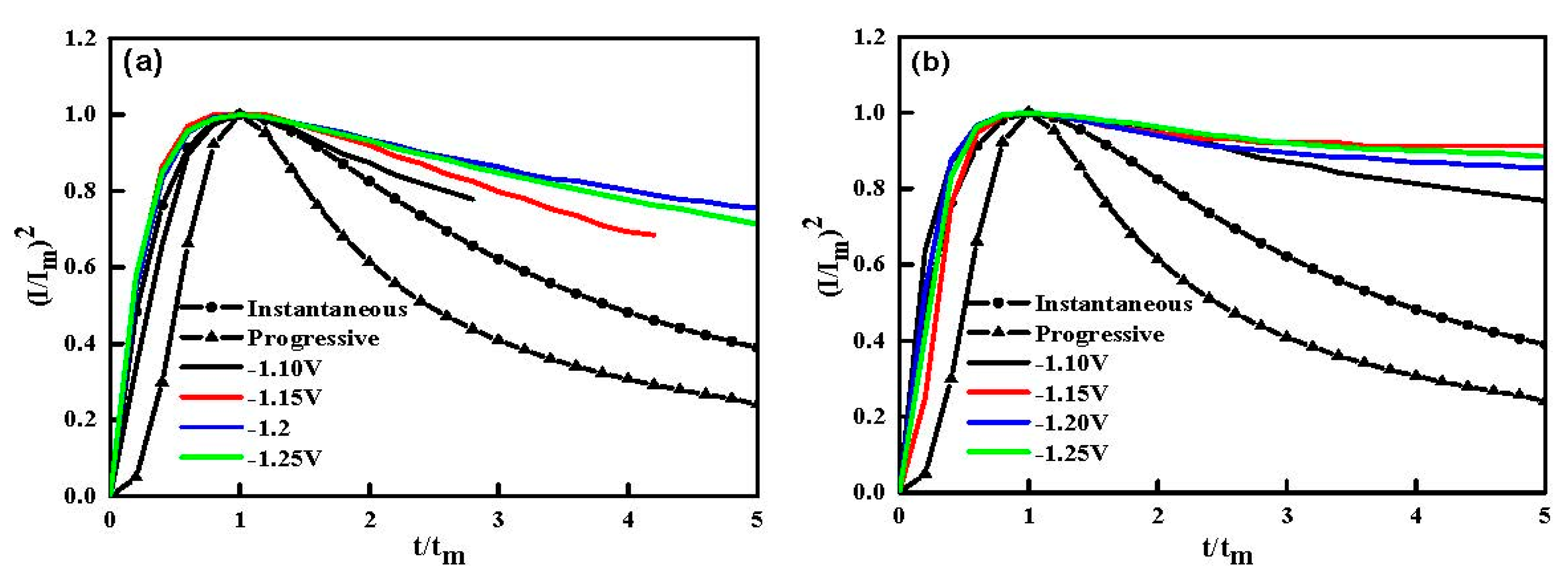
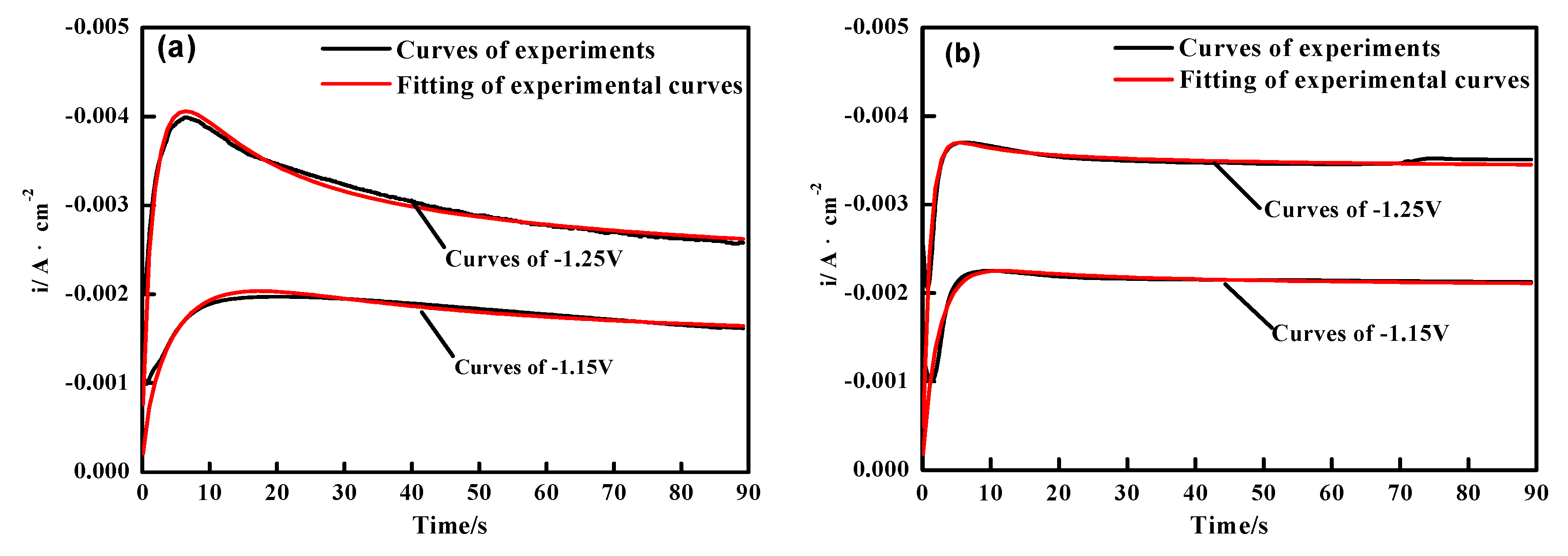
3.2. Chronoamperometry (CA) Studies
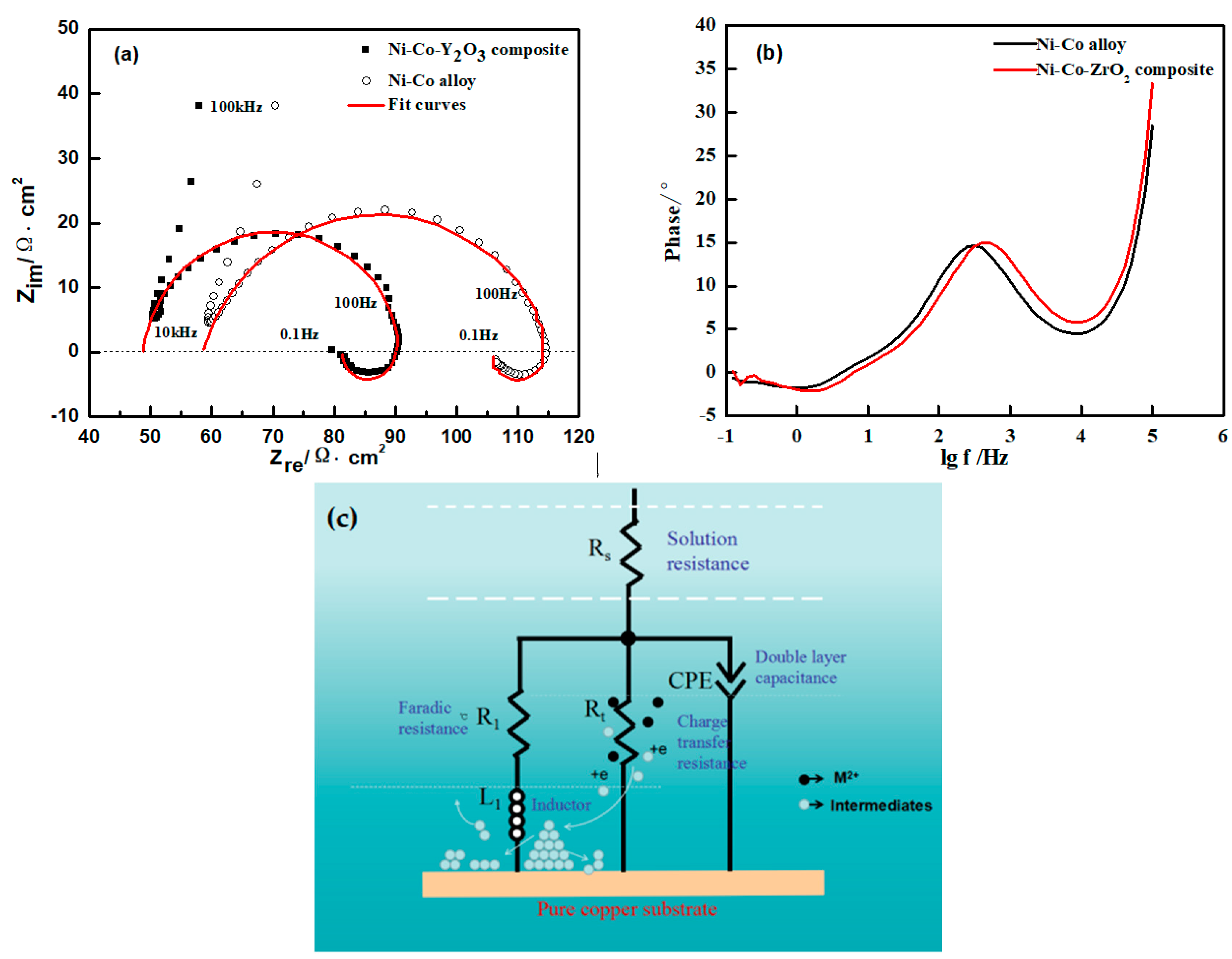
3.3. Electrochemical Impedance Spectroscopy (EIS) Studies
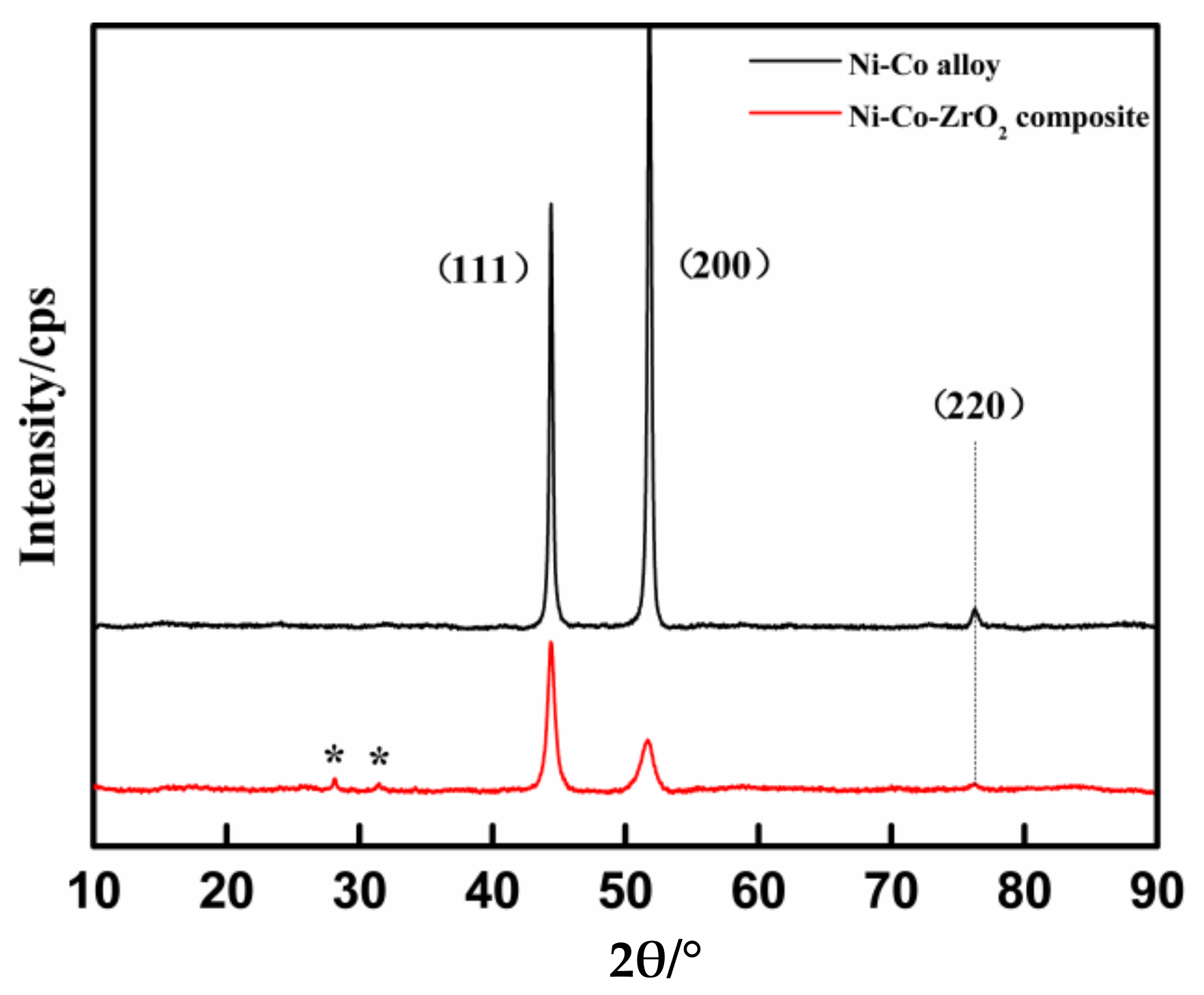
3.4. X-ray Diffraction Texture and Surface Morphology of Coatings
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kainer, K.U. Metal Matrix Composites: Custom-Made Materials for Automotive and Aerospace Engineering; Wiley VCH: Weinheim, Gemany, 2006; Volume 300. [Google Scholar] [CrossRef]
- Benea, L. Electrodeposition and tribocorrosion behaviour of ZrO2–Ni composite coatings. J. Appl. Electrochem. 2009, 39, 1671–1681. [Google Scholar] [CrossRef]
- Da Costa Pereira, R.F.; de Oliveira, E.S.D.; Silva, D.L.A.E.; Souza Ribeiro, A.; de Araújo Filho, O.O.; de Andrade e Lima, M.A.G.; Urtiga Filho, S.L. Influence of composite nano-coating of Ni-Co-SiC obtained by electrodeposition on the corrosion resistance of API 5L X80 Steel. Mater. Sci. Forum 2017, 899, 317–322. [Google Scholar] [CrossRef]
- Ranganatha, S.; Venkatesha, T.V.; Vathsala, K. Electrochemical studies on Zn/nano-CeO2 electrodeposited composite coatings. Surf. Coat. Technol. 2012, 208, 64–72. [Google Scholar] [CrossRef]
- Ghaziof, S.; Kilmartin, P.A.; Gao, W. Electrochemical studies of sol-enhanced Zn–Ni–Al2O3 composite and Zn–Ni alloy coatings. J. Electroanal. Chem. 2015, 755, 63–70. [Google Scholar] [CrossRef]
- Popoola, A.P.I.; Aigbodion, V.S.; Fayomi, O.S.I. Surface characterization, mechanical properties and corrosionbehaviour of ternary based Zn–ZnO–SiO2 composite coating of mildsteel. J. Alloys Compd. 2016, 654, 561–566. [Google Scholar] [CrossRef]
- Daniyan, A.A.; Umoru, L.E.; Popoola, A.P.I.; Fayomi, O.S.I. Comparative studies of microstructural, tribological and corrosion properties of Zn-TiO2 and Zn-TiO2-WO3 nano-composite coatings. Result. Phys. 2017, 7, 3222–3229. [Google Scholar] [CrossRef]
- Chen, Y.; Hao, Y.; Huang, W.; Ji, Y.; Yang, W.Z.; Yin, X.S.; Liu, Y.; Ling, X. Corrosion behavior of Ni-P-nano-Al2O3 composite coating in the presence of anionic and cationic surfactants. Surf. Coat. Technol. 2017, 310, 122–128. [Google Scholar] [CrossRef]
- Yu, H.L.; Zhang, W.; Wang, H.M.; Ji, X.C.; Song, Z.Y.; Li, X.Y.; Xu, B.S. In-situ synthesis of TiC/Ti composite coating by high frequency induction cladding. J. Alloys Compd. 2017, 701, 244–255. [Google Scholar] [CrossRef]
- Beltowska-Lehman, E.; Goral, A.; Indyka, P. Electrodeposition and characterization of Ni/Al2O3 nanocomposite coatings. Arch. Metall. Mater. 2011, 56, 919–931. [Google Scholar] [CrossRef]
- Singh, S.; Sribalaji, M.; Wasekar, N.P.; Joshi, S.; Sundararajan, G.; Singh, R.; Keshri, A.K. Microstructural, phase evolution and corrosion properties of silicon carbide reinforced pulse electrodeposited nickel-tungsten composite coatings. Appl. Surf. Sci. 2016, 364, 264–272. [Google Scholar] [CrossRef]
- Tripathi, M.K.; Singh, V.B.; Singh, H.K. Structure and properties of electrodeposited functional Ni–Fe/TiN nanocomposite coatings. Surf. Coat. Technol. 2015, 278, 146–156. [Google Scholar] [CrossRef]
- Beltowska-Lehman, E.; Indyka, O.; Bigos, A.; Kot, M.; Tarkowski, L. Electrodeposition of nanocrystalline Ni–W coatings strengthened by ultrafine alumina particles. Surf. Coat. Technol. 2012, 211, 62–66. [Google Scholar] [CrossRef]
- Chen, W.W.; Gao, W. Sol-enhanced electroplating of nanostructured Ni–TiO2 composite coatings—the effects of sol concentration on the mechanical and corrosion properties. Electrochim. Acta 2010, 55, 6865–6871. [Google Scholar] [CrossRef]
- Ataie, S.A.; Zakeri, A. Improving tribological properties of (Zn–Ni)/nano Al2O3 composite coatings produced by ultrasonic assisted pulse plating. J. Alloys Compd. 2016, 674, 315–322. [Google Scholar] [CrossRef]
- Beltowska-Lehman, E.; Indyka, P.; Bigos, A.; Szczerba, M.J.; Kot, M. Ni–W/ZrO2 nanocomposites obtained by ultrasonic DC electrodeposition. Mater. Des. 2015, 80, 1–11. [Google Scholar] [CrossRef]
- Srivastava, M.; Selvi, V.E.; Grips, V.K.W.; Rajam, K.S. Corrosion resistance and microstructure of electrodeposited nickel–cobalt alloy coatings. Surf. Coat. Technol. 2006, 201, 3051–3060. [Google Scholar] [CrossRef]
- Dolati, A.; Sababi, M.; Nouri, E.; Ghorbani, M. A study on the kinetic of the electrodeposited Co–Ni alloy thin films in sulfate solution. Mater. Chem. Phys. 2007, 102, 118–124. [Google Scholar] [CrossRef]
- Marikkannu, K.R.; Kalaignan, G.P.; Vasudevan, T. The role of additives in the electrodeposition of nickel–cobalt alloy from acetate electrolyte. J. Alloys Compd. 2007, 438, 332–336. [Google Scholar] [CrossRef]
- Hart, T.; Watson, A. Electroforming. Met. Finish. 2000, 100, 372–383. [Google Scholar] [CrossRef]
- Sr, M.S.; Grips, V.K.W.; Jain, A.; Rajam, K.S. Influence of SiC particle size on the structure and tribological properties of Ni–Co composites. Surf. Coat. Technol. 2007, 202, 310–318. [Google Scholar]
- Srivastava, M.; WilliamGrips, V.K.; Rajam, K.S. Electrochemical deposition and tribological behaviour of Ni and Ni–Co metal matrix composites with SiC nano-particles. Appl. Surf. Sci. 2007, 253, 3814–3824. [Google Scholar] [CrossRef]
- Wu, G.; Li, N.; Zhou, D.R. Electrochemical anomalous codeposition of Co-Ni alloys from sulfamate electrolytes and its mechanism. J. Chem. Eng. Chin. Univ. 2005, 19, 48–53. [Google Scholar]
- Kumar, K.; Chandramohan, R.; Kalyanaraman, D. Effect of heat treatment on cobalt and nickel electroplated surfaces with Cr2O3 dispersion. Appl. Surf. Sci. 2004, 227, 383–386. [Google Scholar] [CrossRef]
- Shi, L.; Sun, C.F.; Gao, P.; Zhou, F.; Liu, W.M. Electrodeposition and characterization of Ni–Co–carbon nanotubes composite coatings. Surf. Coat. Technol. 2006, 200, 4870–4875. [Google Scholar] [CrossRef]
- Wang, W.; Hou, F.Y.; Wang, H.; Guo, H.T. Fabrication and characterization of Ni–ZrO2 composite nano-coatings by pulse electrodeposition. Scr. Mater. 2005, 53, 613–618. [Google Scholar] [CrossRef]
- Zhang, K.F.; Ding, S.; Wang, G.F. Different superplastic deformation behavior of nanocrystalline Ni and ZrO2/Ni nanocomposite. Mater. Lett. 2008, 62, 719–722. [Google Scholar] [CrossRef]
- Cao, J.Q. Effect of Cr (VI) on the deposition of Nickel. J. Nanchang Hangkong Univ. Nat. Sci. 1994, 14–19. (In Chinese) [Google Scholar]
- Tan, C.; Cui, H.; Hu, W.; Liu, Y.; Zheng, Z. Influence of nano-Al2O3 particles on nickel electrocrystallization at initial stage. Rare Met. Mater. Eng. 2010, 39, 10–16. [Google Scholar]
- Vathsala, K. Zn–ZrO2 nanocomposite coatings: Elecrodeposition and evaluation of corrosion resistance. Appl. Surf. Sci. 2011, 257, 8929–8936. [Google Scholar] [CrossRef]
- Scharifker, B.; Hills, G. Theoretical and experimental studies of multiple nucleation. Electrochim. Acta 1983, 28, 879–889. [Google Scholar] [CrossRef]
- Raeissi, K.; Saatchi, A.; Golozar, M.A. Effect of nucleation mode on the morphology and texture of electrodeposited zinc. J. Appl. Electrochem. 2003, 33, 635–642. [Google Scholar] [CrossRef]
- Wu, G.; Li, N.; Wang, D.L.; Zhou, D.R.; Xu, B.Q.; Mitsuo, K. Effect of α-Al2O3 particles on the electrochemical codeposition of Co–Ni alloys from sulfamate electrolytes. Mater. Chem. Phys. 2004, 87, 411–419. [Google Scholar] [CrossRef]
- Palomar-Pardavé, M.; Scharifker, B.R.; Arce, E.M.; Romero-Romo, M. Nucleation and diffusion-controlled growth of electroactive centers: Reduction of protons during cobalt electrodeposition. Electrochim. Acta 2005, 50, 4736–4745. [Google Scholar] [CrossRef]
- Song, L.X.; Zhang, Z.; Zhang, J.Q.; Chunan, C. Electroplating mechanism of nanostructured black Ni films. Acta Metall. Sin. 2011, 47, 123–128. [Google Scholar]
- Nowak, P.; Socha, R.P.; Kaisheva, M.; Fransaer, J.; Celis, J.P.; Stoinov, Z. Electrochemical investigation of the codeposition of SiC and SiO2 particles with nickel. J. Appl. Electrochem. 2004, 21, 429–437. [Google Scholar]
- Monshi, A.; Foroughi, M.R.; Monshi, M.R. Modified scherrer equation to estimate more accurately nano-crystallite size using XRD. World J. Nano Sci. Eng. 2012, 2, 154–160. [Google Scholar] [CrossRef]

| Bath Constituents | Bath Parameters |
|---|---|
| Ni(NH2SO3)2·4H2O—80 g/L(source of Ni2+) | Current density—5 × 10−2 A/m2 |
| Co(NH2SO3)2·4H2O—20 g/L(source of Co2+) | Temperature—40 ± 2 °C |
| H3BO3—40 g/L | Ultrasonic power—100 W |
| ZrO2—10 g/L | pH = 4 |
| Materials | Potential (V) | P1* (μA cm−2) | P2 (s−1) | P3 (s−1) | P4 (μA cm−2) | A (s−1) |
|---|---|---|---|---|---|---|
| Ni-Co | −1.15 | −1.80 | 0.13974 | 1.72 | −4.38 | 1.72 |
| Ni-Co | −1.25 | −1.95 | 0.33107 | 2.91 | −6.52 | 2.91 |
| Ni-Co-ZrO2 | −1.15 | −2.02 | 0.37832 | 1.95 | −0.89313 | 1.95 |
| Ni-Co-ZrO2 | −1.25 | −3.36 | 0.83355 | 2.49 | −0.89541 | 2.49 |
| Materials | Potential (V) | Rs/Ω cm2 | CPE1-P/F cm2 | Rt/Ω cm2 | R1/Ω cm2 | L1/H cm2 |
|---|---|---|---|---|---|---|
| Ni-Co | −1.10 | 55.56 | 5.2 × 10−5 | 54.89 | 249.8 | 34.52 |
| Ni-Co-ZrO2 | −1.10 | 45.48 | 4.1 × 10−5 | 43.36 | 133.7 | 7.989 |
| Materials | Ni | Co | O | Zr | Total |
|---|---|---|---|---|---|
| Ni-Co | 63.55 | 36.45 | – | – | 100 |
| Ni-Co-ZrO2 | 59.66 | 21.39 | 12.57 | 6.38 | 100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhou, X.; Liang, Z.; Jin, H. Characterization of Ultrasonic-Assisted Electrochemical Deposition of Ni-Co-ZrO2. Coatings 2018, 8, 211. https://doi.org/10.3390/coatings8060211
Wang Y, Zhou X, Liang Z, Jin H. Characterization of Ultrasonic-Assisted Electrochemical Deposition of Ni-Co-ZrO2. Coatings. 2018; 8(6):211. https://doi.org/10.3390/coatings8060211
Chicago/Turabian StyleWang, Yiyong, Xinyu Zhou, Zhipeng Liang, and Hui Jin. 2018. "Characterization of Ultrasonic-Assisted Electrochemical Deposition of Ni-Co-ZrO2" Coatings 8, no. 6: 211. https://doi.org/10.3390/coatings8060211
APA StyleWang, Y., Zhou, X., Liang, Z., & Jin, H. (2018). Characterization of Ultrasonic-Assisted Electrochemical Deposition of Ni-Co-ZrO2. Coatings, 8(6), 211. https://doi.org/10.3390/coatings8060211




