1. Introduction
Colorfastness to light is an essential quality for any dye/fabric combination intended for sunlight exposure to sunlight for a significant length of time. One need only look at garments or fabric that has been in a shop display window to see that fading due to sunlight is a problem for most fabric dyes.
Textiles can be quickly and efficiently dyed with wood-staining (spalting) fungal pigments, without the use of water [
1]. Spalting pigments also have unusually long persistence of color, both under laboratory conditions and in applications such as ancient marquetry and intarsia [
2,
3,
4]. These characteristics indicate that spalting pigments could be more eco-friendly than commercial dyes, many of which are carcinogenic [
5] and will exhibit superior colorfastness to light without the use of water or heat.
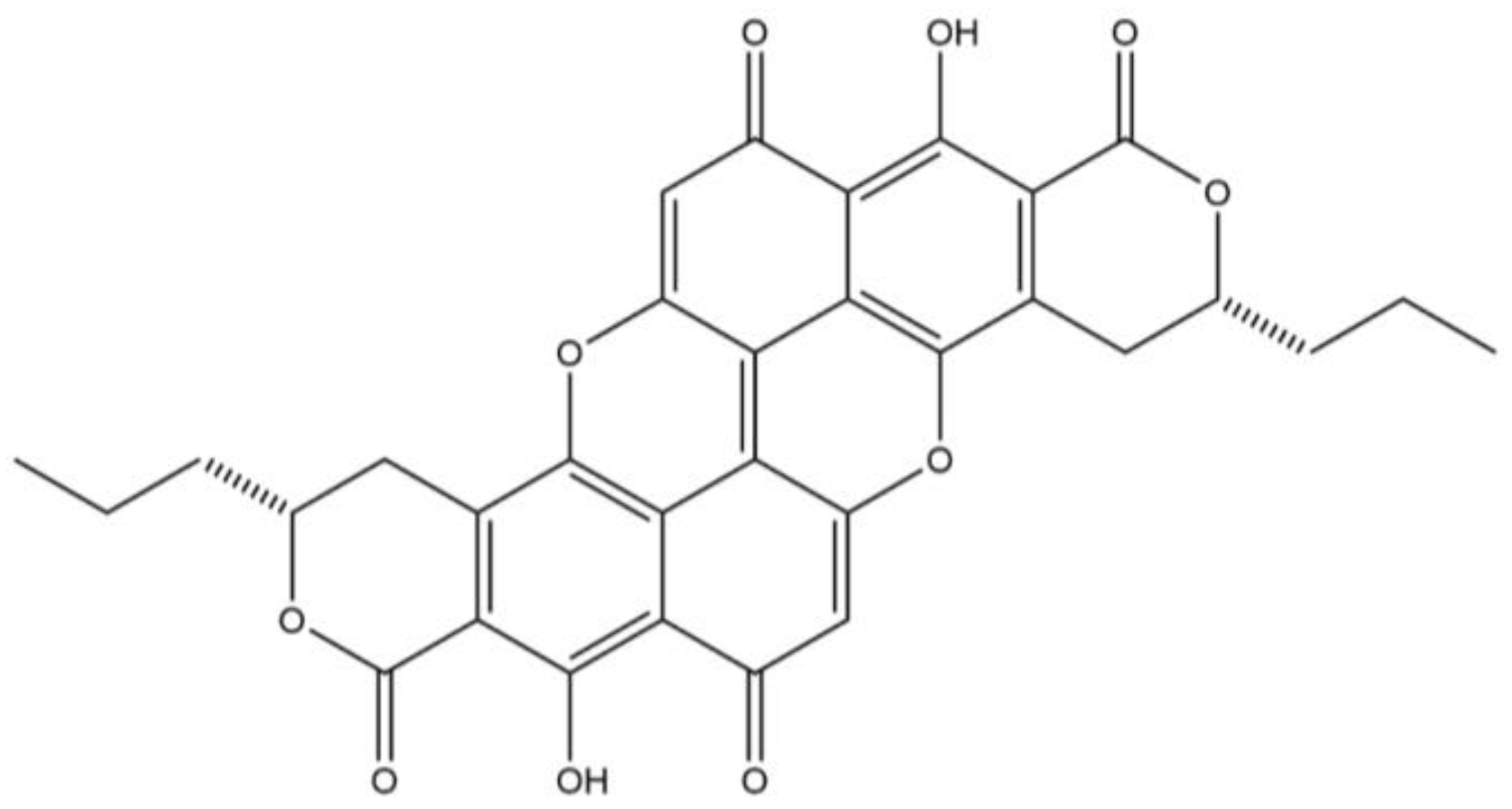
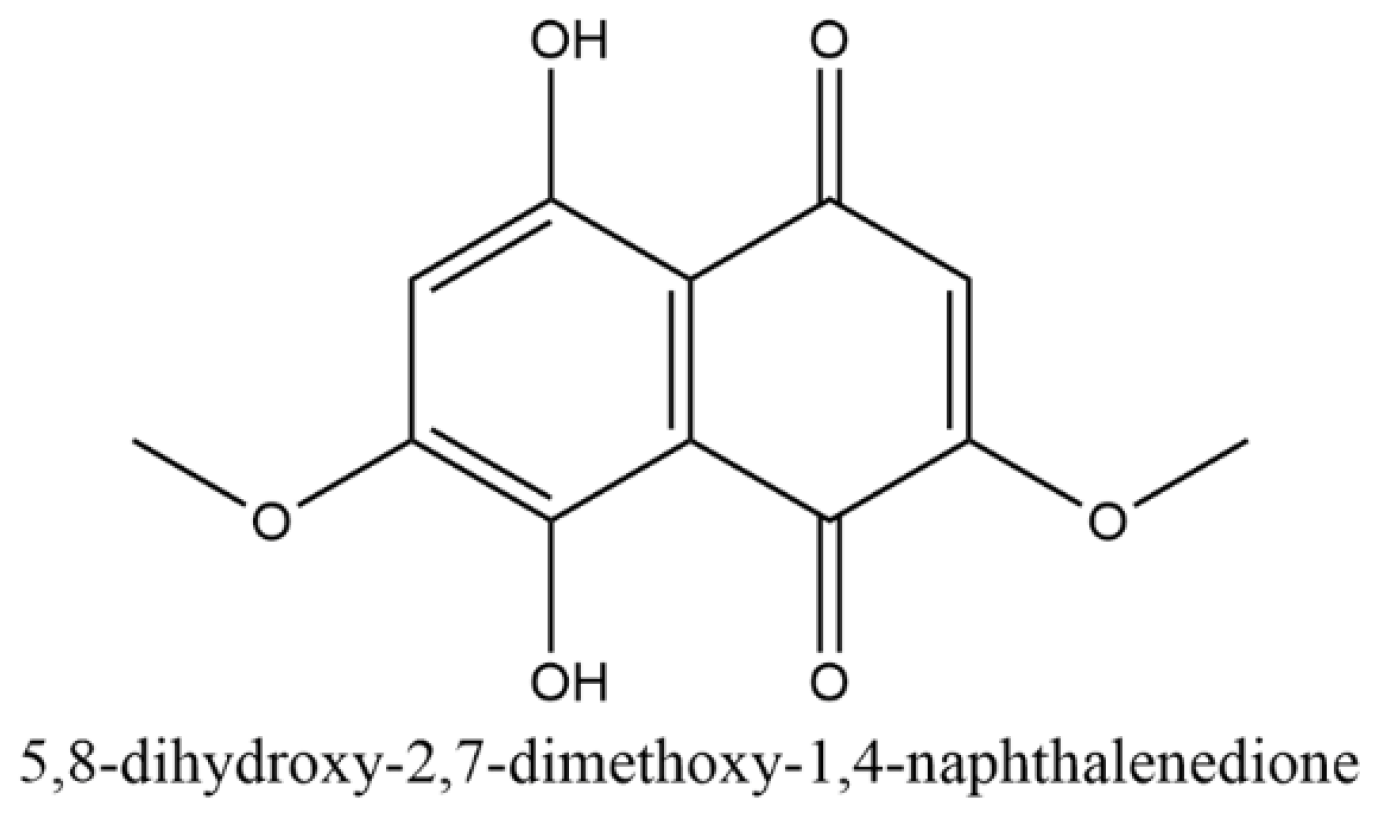
Wood-staining fungal pigments, such as xylindein and draconin red, are anthraquinones (
Figure 1 and
Figure 2) [
6,
7], which are produced by the fungi in response to stress. Many commercial dyes, such as disperse dyes, are also anthraquinone dyes, which is one of the reason these fungal pigments were chosen for the current study. Furthermore, the ease of laboratory culturing of the fungi and extraction of the pigments make them an excellent choice for experimentation [
8]. Extracted wood-staining fungal pigments have only recently been used to dye textiles, and previous tests on colorfastness to washing, perspiration, and crocking have shown promising results [
1,
9,
10].
The two most common test methods for colorfastness to light in the textiles industry are AATCC Test Method 16.3 [
11] and ISO 105-B02 [
12], both of which involve the use of a xenon arc lamp. If colorfastness to light testing is not a regularly performed task, the cost of a xenon arc lamp testing unit can be cost prohibitive. The same issue arises when using AATCC Test Method 16.2 [
13], Colorfastness to light: Carbon arc. ISO no longer has a standard for colorfastness to light testing using a carbon arc lamp.
Two other alternatives for testing colorfastness to light are AATCC Test Method 16.1 [
14], Colorfastness to light: Outdoor, and ISO 105-B01 [
15], Color fastness to light: Daylight. These two methods are similar and require outdoor exposure in specially constructed frames. The ISO standard is very specific about exposure direction, shadows, grade, etc. The AATCC Test Method is less specific, but requires constant monitoring. Having the space, time, and weather conditions available to perform outdoor daylight tests on many samples may be prohibitive in regions where sunlight is limited.
In the current study, a new test method was developed as an alternative to the above methods using a QUV Accelerated Weathering Machine. The developed method requires a fraction of the time as that of outdoor daylight testing, and less exposure time than the xenon arc lamp method. Additionally, after initial calibration, monitoring is not required during testing. While more costly than simple outdoor exposure, the QUV has a wider range of functions than an arc lamp and can be used for many types of weathering tests on a variety of materials. It can be used to test reactions of fabrics, wood, and other materials to UV exposure, temperature variation, and moisture. The machine can be configured to test a variety of conditions, such as normal outdoor use or thermal shock. Its use in other testing, including another AATCC test standard, AATCC TM186 [
16] therefore, may make it a more suitable machine to purchase than other UV-testing equipment.
The purpose of this study was to compare the colorfastness to light between spalting pigments and commercial dyes, without heat, using a novel, short-duration immersion dyeing method. Both unmordanted and mordanted fabrics were tested and compared for colorfastness. Noting the historic color stability of spalting pigments, it is likely that these pigments will demonstrate superior colorfastness (specifically in regards to light) when compared with commercial fabric dyes, using the proposed methodology.
3. Results and Discussion
For unmordanted fabrics tested with light, only 5 of the 30 tested fabric/dye/color combinations were colorfast: cotton/draconin red, cotton/xylindein, linen/disperse/red, wool/draconin red, and wool/disperse/red (see
Table 4 and
Figure 3). For mordanted fabrics tested with light, 11 of the 38 tested fabric/dye/color combinations were colorfast. Of those 11, 8 were dyed with fungal pigments, 2 with fiber reactive dyes, and one with a natural dye (spirulina) (see
Table 5 and
Figure 4). For mordanted fabrics,
p-values for all highest possible levels of interaction were <0.006 at α = 0.05. For unmordanted fabrics,
p-values for all possible highest levels of interaction were <0.0001 at α = 0.05, except for cotton where the
p-value for color*treatment was 0.1396 at α = 0.05. Average Δ
E* values can be found in
Appendix Table A1 and
Table A2.
The extracted fungal pigments performed better than other dyes with regards to colorfastness to light, when the short-duration immersion dyeing method was used without heat. Although there is inherently some difference between carrying a dye in water, and carrying the fungal pigments in DCM as the fungal pigments cannot be carried in water, a direct comparison was not possible. It was the authors’ intent to compare a “natural” dye system to that of the fungal pigments, as these colorants will likely share a market. Hence, although different results likely would have been achieved if delivering the dyes in an organic solvent, the comparison would not have been as relevant to the end goal of this investigation.
Draconin red performed better than xylindein, at least for unmordanted fabrics, although no fabric or dye/color was outstanding. Due to xylindein’s characteristically persistent nature on wood [
2,
3], even after centuries, it would have been expected that the pigment would have demonstrated better colorfastness to light. It is UV radiation that causes color fading by inducing a chemical reaction that breaks the carbon-carbon double bonds of quinone-based pigments [
17]. It may be that xylindein binds to certain fibers in such a way as to prevent this chemical reaction yet binds differently in other fibers allowing the chemical reaction to occur. This could explain why some of the fabrics dyed with xylindein were colorfast to light while others were not.
Examination of clothing, fabric, or yarn that has been in a shop display window will show that fading due to exposure to sunlight is a problem with most dyes. With mordanted fabrics dyed with fungal pigments, colorfastness to light was about 1.5 times greater than that of unmordanted fabrics dyed with fungal pigments (21% of samples compared to 13% of samples). This indicates that mordanting may improve colorfastness to light, especially with the fungal pigments.
Every fungal pigment sample on unmordanted fabrics that was colorfast to light was also colorfast when mordanted, with the exception of cotton/draconin red, and five additional fabrics dyed with fungal pigments were colorfast to light when mordanted. Why colorfastness did not function as well with cotton and draconin red is unknown, however it may have something to do with the unique crystalline nature of the draconin red pigment [
18]. Mordanting improved colorfastness to light on cotton, hemp, silk, and wool. In three recent studies, that included testing for colorfastness to light for silk, cotton, and nylon colored with natural dyes, mordanting had variable effects on lightfastness. In the Haar et al. study [
19], aluminum-based mordants did not affect colorfastness to light of cotton dyed with madder or weld, and slightly decreased the colorfastness to light of cotton dyed with coreopsis (AATCC Test Method 16.3). In the Shams-Nateri et al. study [
20] of aluminum potassium sulfate (AlK(SO
4)
2) mordanted nylon dyed with weld and pomegranate peel, colorfastness to light was decreased for weld except when the samples were post-mordanted (mordanted after dyeing). Colorfastness to light for nylon dyed with pomegranate peel increased except when the samples were meta-mordanted (mordanted at the same time they were dyed) (ISO 105-B02).
In the Punrattanasin et al. study [
21] on silk dyed with mangrove bark extract, aluminum potassium sulfate had no effect on colorfastness to light, ferrous sulfate and copper (II) sulfate (CuSO
4) increased colorfastness to light, and stannous chloride (SnCl
2) decreased colorfastness to light (ISO 105-B02). The inconsistency of the results from these studies compared with the results of the current study indicates that, at least for the alum and cream of tartar mordant, mordanting yields more consistent colorfastness to light results for fungal pigments, especially on natural fibers. None of the fungal pigments/fabric combinations exhibited a decreased colorfastness to light when mordanted (excluding cotton/draconin red), and a few combinations exhibited increased colorfastness.
A general statement cannot be made as to whether mordanting improves, does not change, or decreases colorfastness to light for natural dyes. Results are heavily dependent on dye type, mordant type, and fiber type. Despite the apparent improvement of colorfastness to light of fungal pigments when used on mordanted fabrics, fungal pigments may require a UV-protective finish (such as those used on woodworks to prevent UV degradation, or those used on the red-stained boxelder wood to prevent degradation of the tree-red pigment) if the end use of the textile requires prolonged exposure to sunlight.