Synthesis of CoFe2O4 through Wet Ferritization Method Using an Aqueous Extract of Eucalyptus Leaves
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Plant Extract
2.3. Synthesis of Cobalt Ferrites
2.4. Characterization Techniques
2.5. Antimicrobial Activity Assays
3. Results and Discussion
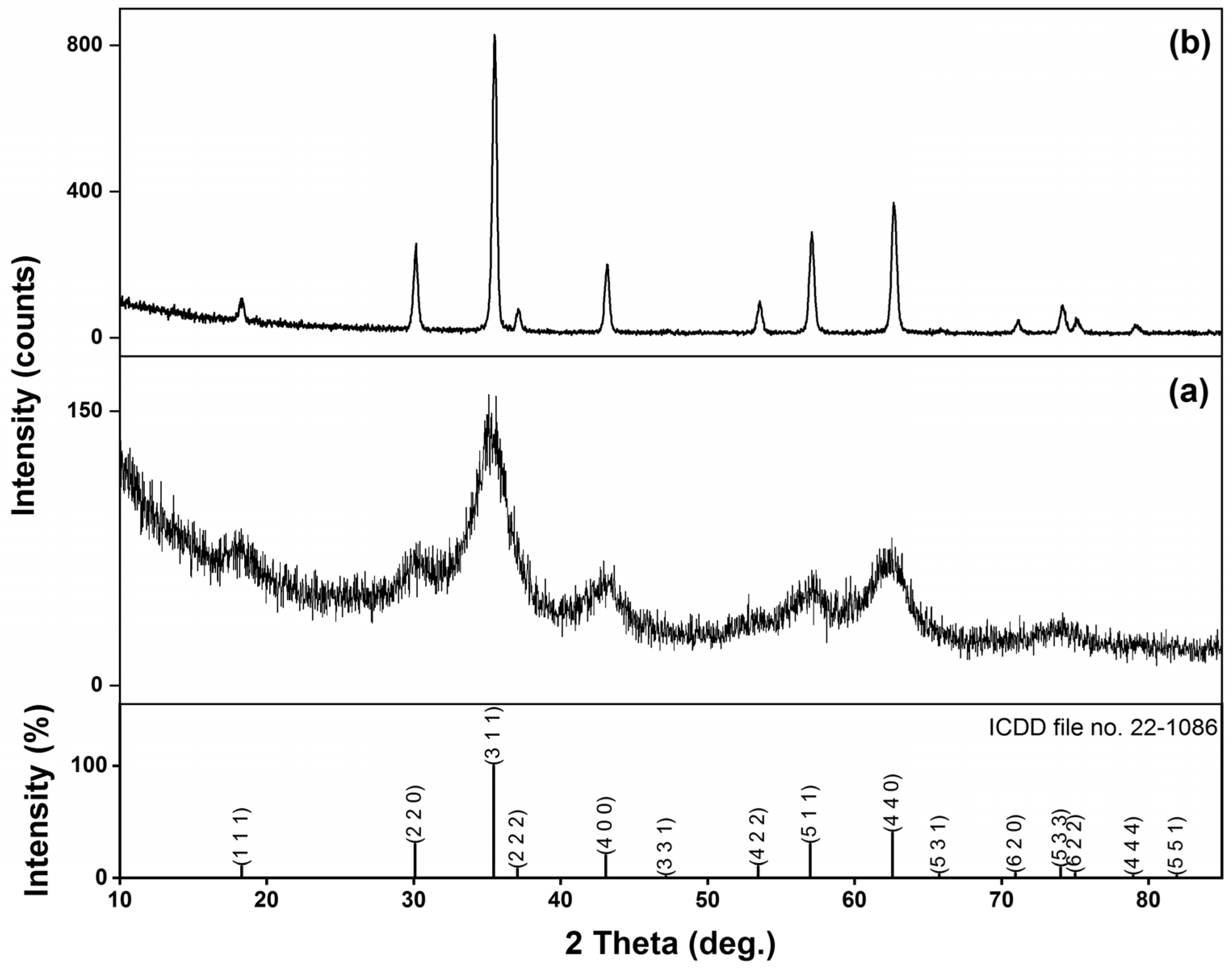
3.1. X-ray Fluorescence and X-ray Diffraction
3.2. Scanning Electron Microscopy
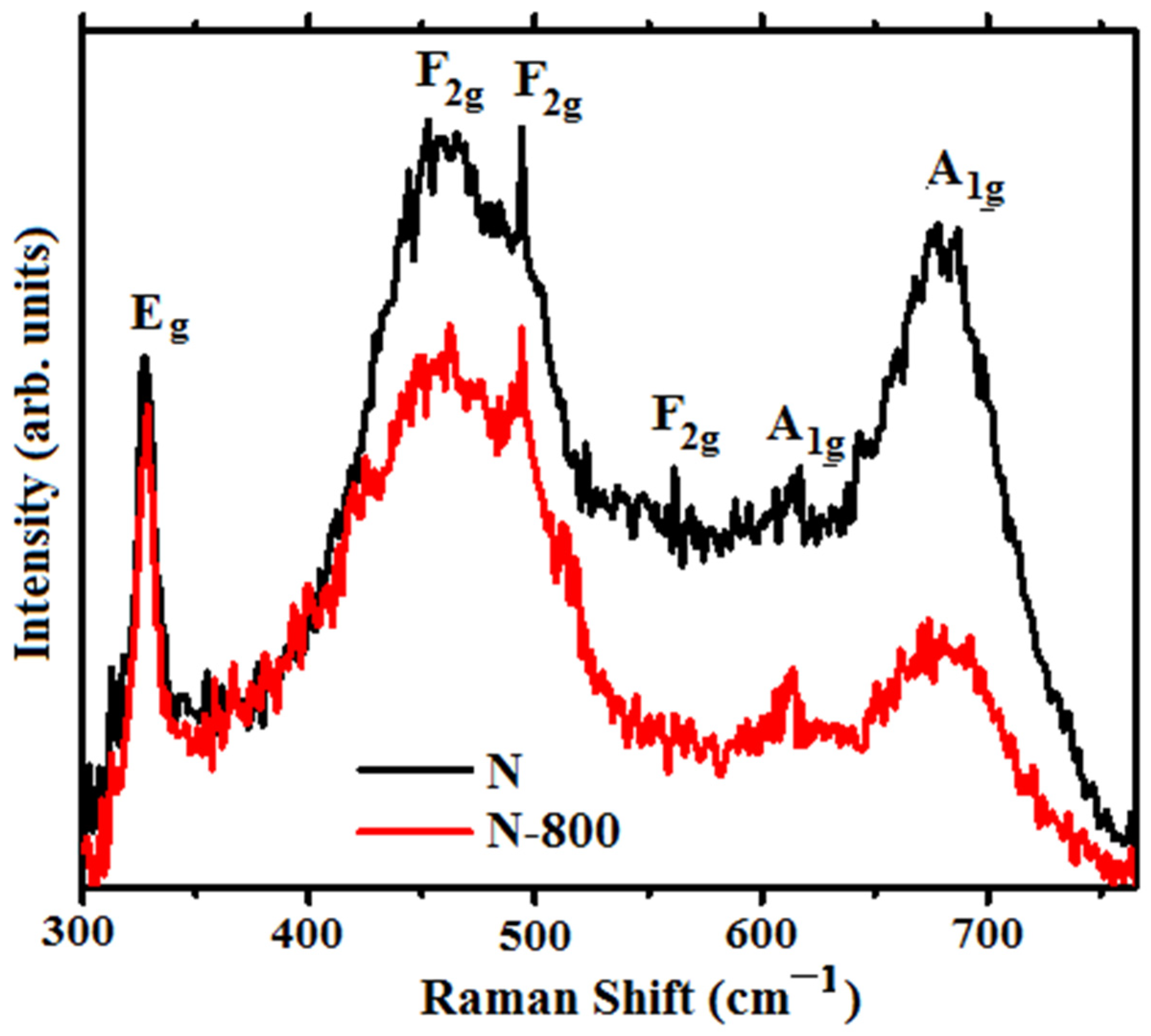
3.3. FTIR and Raman Spectroscopy
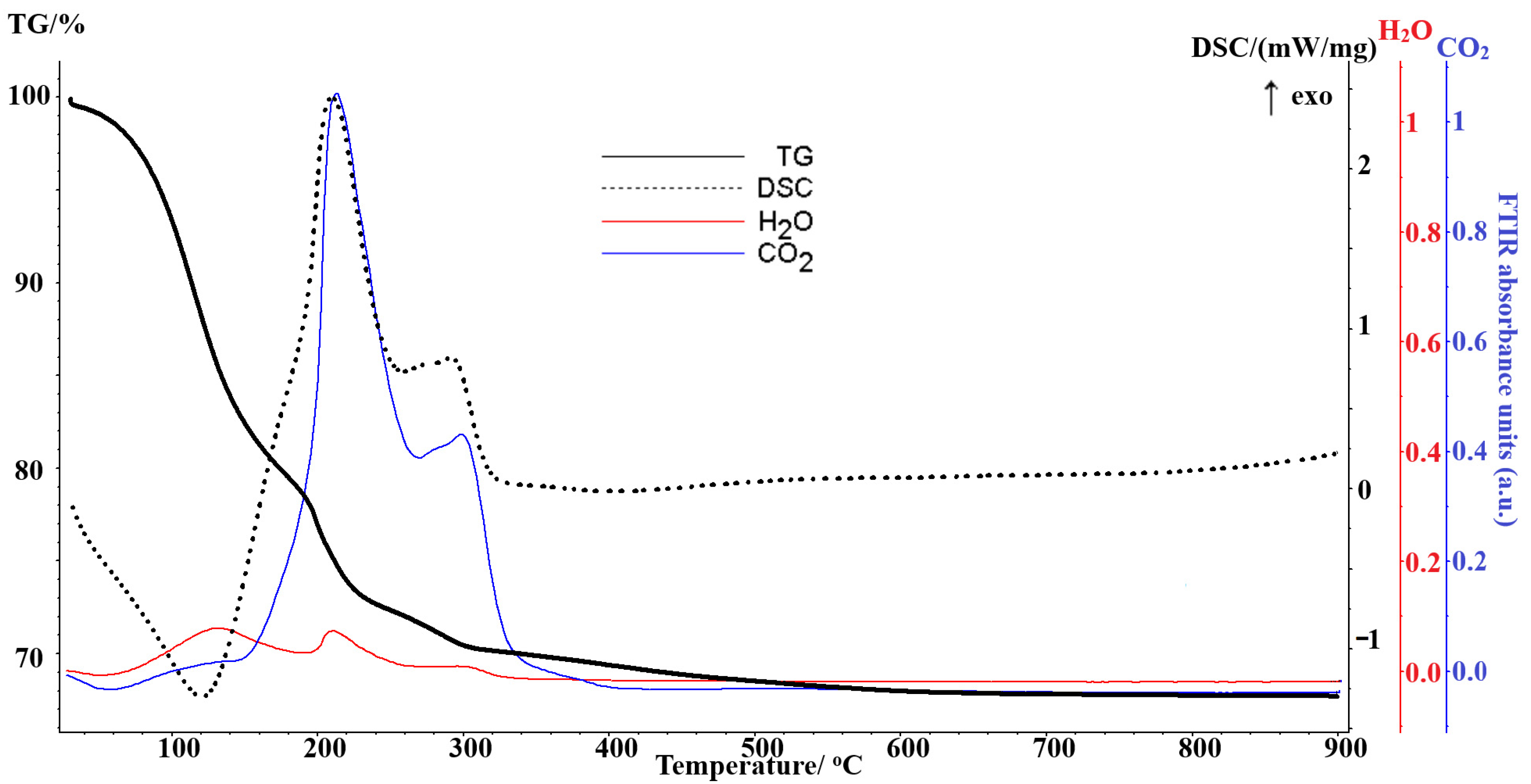
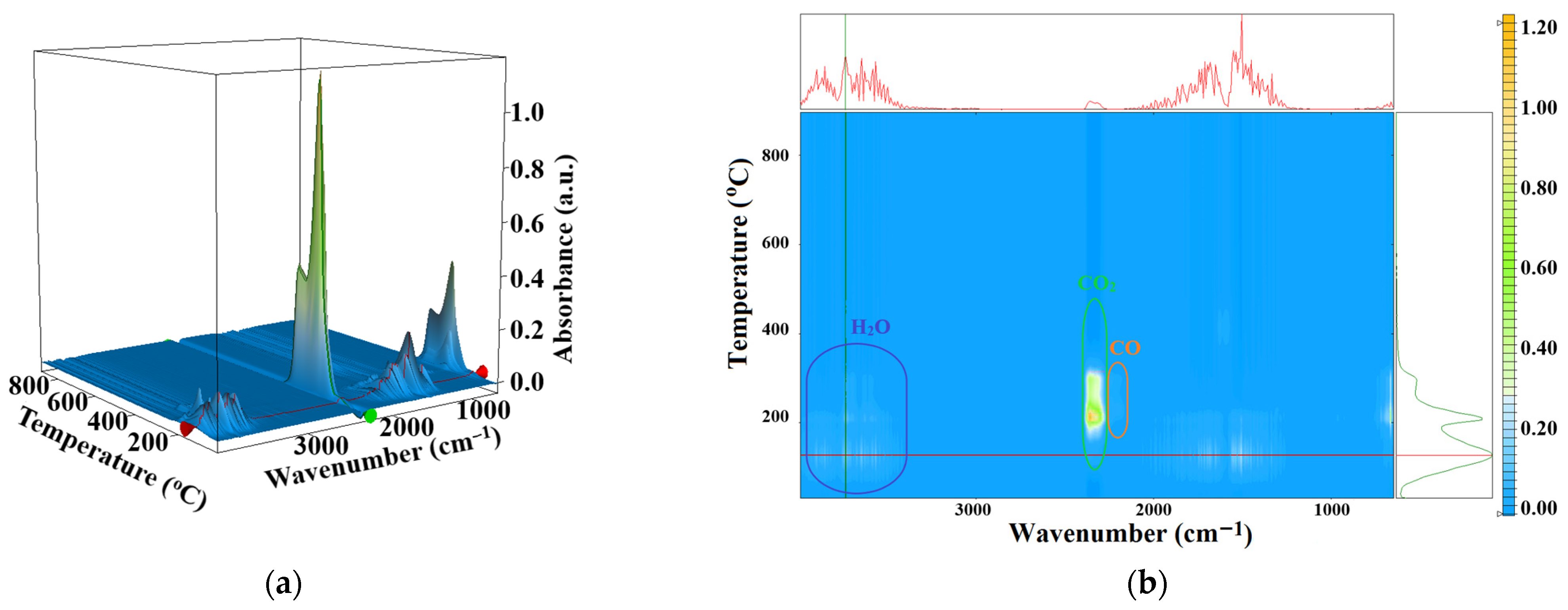
3.4. Thermal Analysis
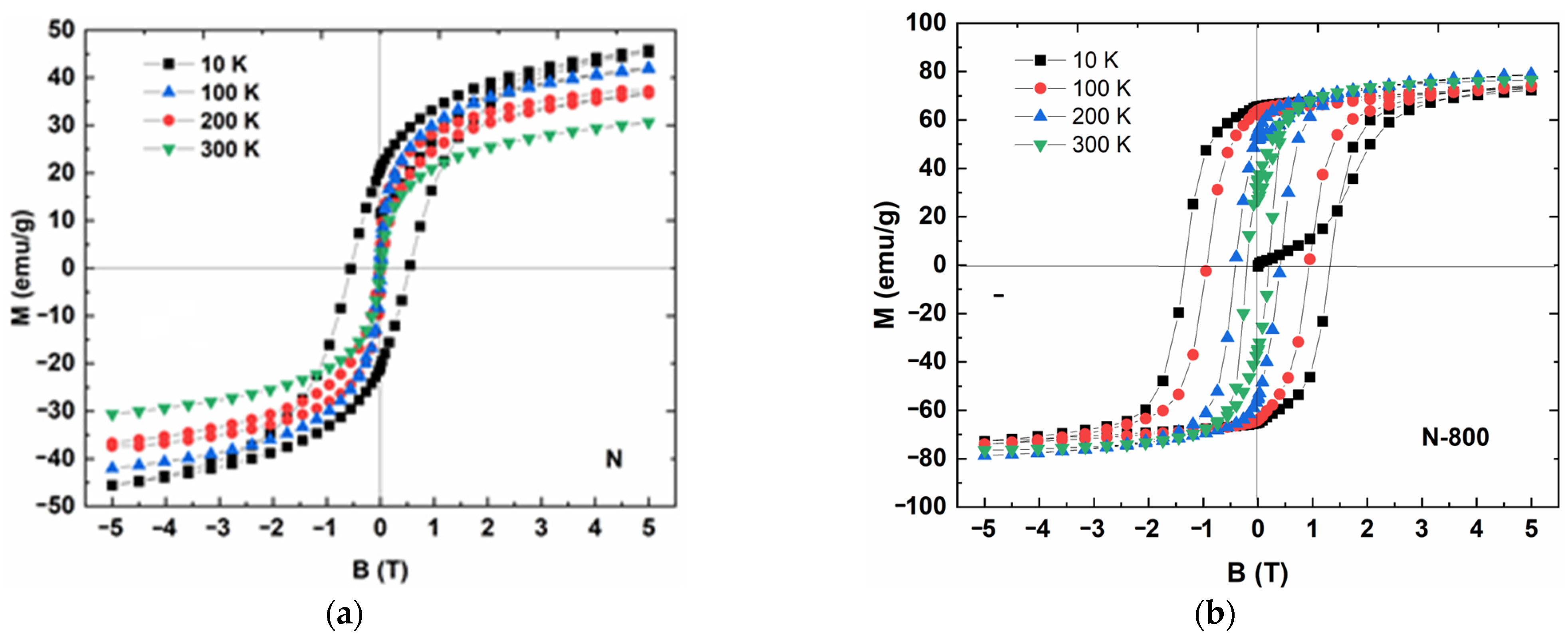
3.5. Magnetic Measurements
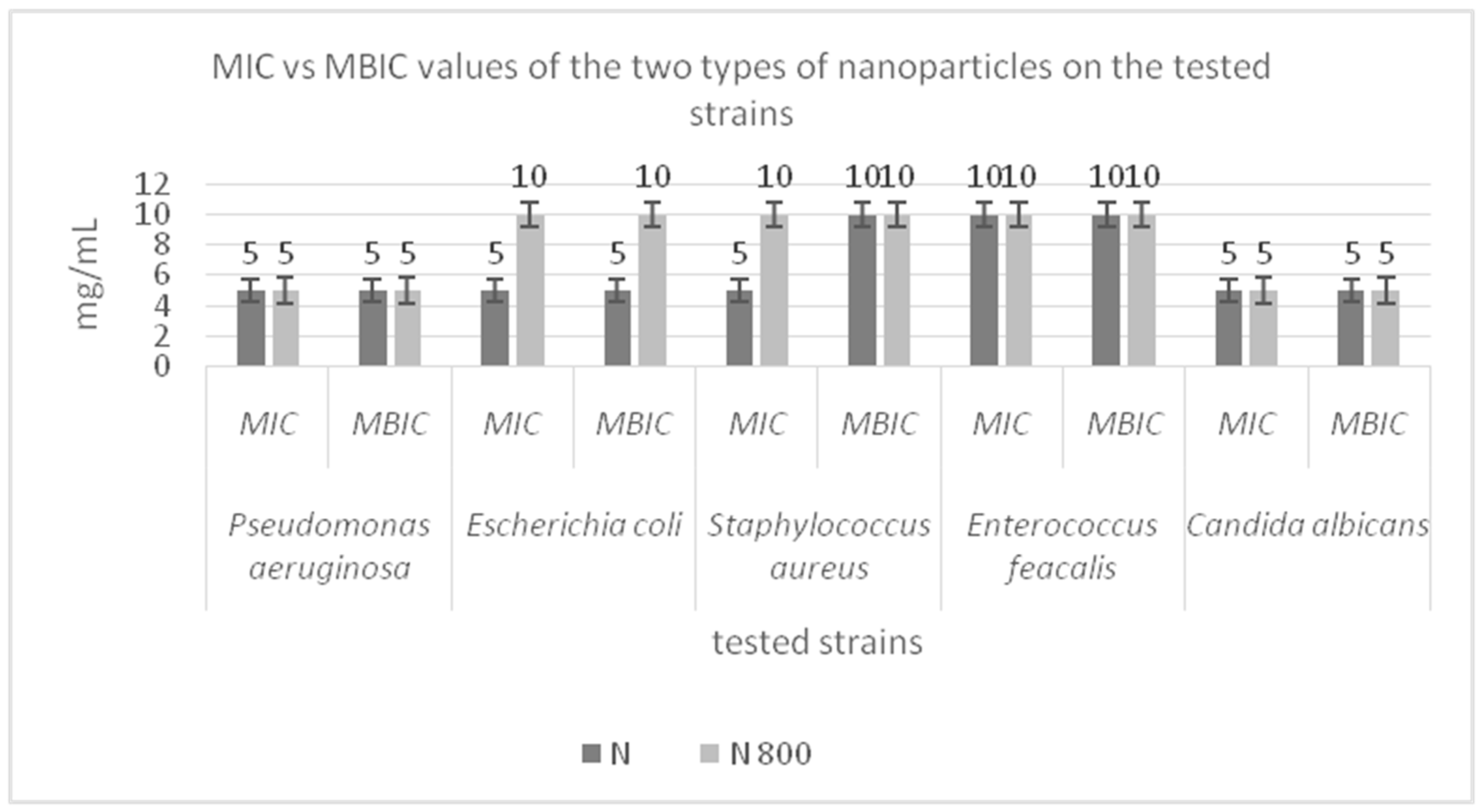
3.6. Antimicrobial Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kefeni, K.K.; Msagati, T.A.M.; Mamba, B.B. Ferrite nanoparticles: Synthesis, characterisation and applications in electronic device. Mater. Sci. Eng. B 2017, 215, 37–55. [Google Scholar] [CrossRef]
- Ahmad, S.I. Nano cobalt ferrites: Doping, structural, low-temperature, and room temperature magnetic and dielectric properties—A comprehensive review. J. Magn. Magn. Mater. 2022, 562, 169840. [Google Scholar] [CrossRef]
- Asadi, R.; Abdollahi, H.; Gharabaghi, M.; Boroumand, Z. Effective removal of Zn (II) ions from aqueous solution by the magnetic MnFe2O4 and CoFe2O4 spinel ferrite nanoparticles with focuses on synthesis, characterization, adsorption, and desorption. Adv. Powder Technol. 2020, 31, 1480–1489. [Google Scholar] [CrossRef]
- Aghrich, K.; Abdellaoui, M.; Mamouni, N.; Bellaouchou, A.; Fekhaoui, M.; Hlil, E.K.; Mounkachi, O. Experimental and first-principles study of the origin of the magnetic properties of CoFe2O4 spinel ferrite. Appl. Phys. A 2020, 126, 940. [Google Scholar] [CrossRef]
- Manohar, A.; Geleta, D.D.; Krishnamoorthi, C.; Lee, J. Synthesis, characterization and magnetic hyperthermia properties of nearly monodisperse CoFe2O4 nanoparticles. Ceram. Int. 2020, 46, 28035–28041. [Google Scholar] [CrossRef]
- Velho-Pereira, S.; Noronha, A.; Mathias, A.; Zakane, R.; Naik, V.; Naik, P.; Salker, A.V.; Naik, S.R. Antibacterial action of doped CoFe2O4 nanocrystals on multidrug resistant bacterial strains. Mater. Sci. Eng. C 2015, 52, 282–287. [Google Scholar] [CrossRef]
- Shakil, M.S.; Hasan, M.A.; Uddin, M.F.; Islam, A.; Nahar, A.; Das, H.; Khan, M.N.I.; Dey, B.P.; Rokeya, B.; Hoque, S.M. In vivo toxicity studies of chitosan-coated cobalt ferrite nanocomplex for its application as MRI contrast dye. ACS Appl. Bio Mater. 2020, 3, 7952–7964. [Google Scholar] [CrossRef]
- Mindru, I.; Gingasu, D.; Diamandescu, L.; Patron, L.; Marinescu, G.; Culita, D.C.; Calderon Moreno, J.M.; Preda, S.; Oprea, O.; Parvulescu, V. CoFe2−xCrxO4 ferrites: Synthesis, characterization and their catalytic activity. Chem. Pap. 2018, 72, 3203–3213. [Google Scholar] [CrossRef]
- Imanipour, P.; Hasani, S.; Seifoddini, A.; Nabiałek, M. Synthesis and characterization of zinc and vanadium Co-substituted CoFe2O4 nanoparticles synthesized by using the sol-gel auto-combustion method. Nanomaterials 2022, 12, 752. [Google Scholar] [CrossRef]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Nguyen, N.T.T.; Nguyen, L.M.; Nguyen, T.T.T.; Nguyen, T.T.; Nguyen, D.T.C.; Van Tran, T. Formation, antimicrobial activity, and biomedical performance of plant-based nanoparticles: A review. Environ. Chem. Lett. 2022, 20, 2531–2571. [Google Scholar] [CrossRef]
- Bukhari, A.; Ijaz, I.; Gilani, E.; Nazir, A.; Zain, H.; Saeed, R.; Alarfaji, S.S.; Hussain, S.; Aftab, R.; Naseer, Y. Green synthesis of metal and metal oxide nanoparticles using different plants’ parts for antimicrobial activity and anticancer activity: A review article. Coatings 2021, 11, 1374. [Google Scholar] [CrossRef]
- Sila, J.M.; Kiio, I.; Mwaura, F.B.; Michira, I.; Abongo, D.; Iwuoha, E.; Kamau, G.N. Green synthesis of silver nanoparticles using Eucalyptus corymbia leaves extract and antimicrobial applications. Nano Hybrids Compos. 2019, 25, 32–45. [Google Scholar] [CrossRef]
- Shah, M.; Fawcett, D.; Sharma, S.; Tripathy, S.K.; Poinern, G.E.J. Green synthesis of metallic nanoparticles via biological entities. Materials 2015, 8, 7278–7308. [Google Scholar] [CrossRef] [PubMed]
- Routray, K.L.; Saha, S.; Behera, D. Green synthesis approach for nano sized CoFe2O4 through aloe vera mediated sol-gel auto combustion method for high frequency devices. Mater. Chem. Phys. 2019, 224, 29–35. [Google Scholar] [CrossRef]
- Mahajan, P.; Sharma, A.; Kaur, B.; Goyal, N.; Gautam, S. Green synthesized (Ocimum sanctum and Allium sativum) Ag-doped cobalt ferrite nanoparticles for antibacterial application. Vacuum 2019, 161, 389–397. [Google Scholar] [CrossRef]
- Kobylinska, N.; Klymchuk, D.; Shakhovsky, A.; Khainakova, O.; Ratushnyak, Y.; Duplijc, V.; Matvieieva, N. Biosynthesis of magnetite and cobalt ferrite nanoparticles using extracts of “hairy” roots: Preparation, characterization, estimation for environmental remediation and biological applications. RSC Adv. 2021, 11, 26974. [Google Scholar] [CrossRef]
- Gingasu, D.; Mindru, I.; Mocioiu, O.C.; Preda, S.; Stanica, N.; Patron, L.; Ianculescu, A.; Oprea, O.; Nita, S.; Paraschiv, I.; et al. Synthesis of nanocrystalline cobalt ferrite through soft chemistry methods: A green chemistry approach using sesame seed extract. Mater. Chem. Phys. 2016, 182, 219–230. [Google Scholar] [CrossRef]
- Gingasu, D.; Mindru, I.; Preda, S.; Calderon Moreno, J.M.; Culita, D.C.; Patron, L.; Diamandescu, L. Green synthesis of cobalt ferrite nanoparticles using plant extract. Rev. Roum. Chim. 2017, 62, 647–655. [Google Scholar]
- Gingasu, D.; Mindru, I.; Culita, D.C.; Calderon Moreno, J.M.; Bartha, C.; Greculeasa, S.; Iacob, N.; Preda, S.; Oprea, O. Structural, morphological and magnetic investigations on cobalt ferrite nanoparticles obtained through green synthesis routes. Appl. Phys. A 2021, 127, 892. [Google Scholar] [CrossRef]
- Chandorkar, N.; Tambe, S.; Amin, P.; Madankar, C. A systematic and comprehensive review on current understanding of the pharmacological actions, molecular mechanisms, and clinical implications of the genus Eucalyptus. Phytomed. Plus 2021, 1, 100089. [Google Scholar] [CrossRef]
- Avidzba, Y.N.; Koshovyi, O.M.; Komisarenko, A.M. Research of eucalyptus leaves dry extract obtained after essential oil extraction. Pharma Inov. J. 2014, 3, 18–22. [Google Scholar]
- Shala, A.Y.; Gururan, M.A. Phytochemical properties and diverse beneficial roles of Eucalyptus globulus Labill.: A review. Horticulturae 2021, 7, 450. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Pinto, R.J.B.; Rocha, S.M.; Marques, P.A.A.P.; Neto, C.P.; Silvestre, A.J.D.; Freire, C.S.R. Unveiling the chemistry behind the green synthesis of metal nanoparticles. Chem. Sus. Chem. 2014, 7, 2704–2711. [Google Scholar] [CrossRef] [PubMed]
- El-Seedi, H.R.; El-Shabasy, R.M.; Khalifa, S.A.M.; Saeed, A.; Shah, A.; Shah, R.; Iftikhar, F.J.; Daim, M.M.A.; Omri, A.; Hajrahand, N.H.; et al. Metal nanoparticles fabricated by green chemistry using natural extracts: Biosynthesis, mechanisms, and applications. RSC Adv. 2019, 9, 24539. [Google Scholar] [CrossRef] [PubMed]
- Balciunaitiene, A.; Liaudanskas, M.; Puzeryte, V.; Viškelis, J.; Janulis, V.; Viškelis, P.; Griškonis, E.; Jankauskaite, V. Eucalyptus globulus and Salvia officinalis extracts mediated green synthesis of silver nanoparticles and their application as an antioxidant and antimicrobial agent. Plants 2022, 11, 1085. [Google Scholar] [CrossRef]
- Munir, H.; Mumtaz, A.; Rashid, R.; Najeeb, J.; Zubair, M.T.; Munir, S.; Bilal, M.; Cheng, H. Eucalyptus camaldulensis gum as a green matrix to fabrication of zinc and silver nanoparticles: Characterization and novel prospects as antimicrobial and dye-degrading agents. J. Mater. Res. Technol. 2020, 9, 15513–15524. [Google Scholar] [CrossRef]
- Alves Puiatti, G.; Passos de Carvalho, J.; Teixeira de Matos, A.; Pereira Lopes, R. Green synthesis of Fe0 nanoparticles using Eucalyptus grandis leaf extract: Characterization and application for dye degradation by a (Photo) Fenton-like process. J. Environ. Manag. 2022, 311, 114828. [Google Scholar] [CrossRef]
- Torres-Limiñana, J.; Feregrino-Pérez, A.A.; Vega-González, M.; Escobar-Alarcón, L.; Cervantes-Chávez, J.A.; Esquivel, K. Green synthesis via Eucalyptus globulus L. extract of Ag-TiO2 catalyst: Antimicrobial activity evaluation toward water disinfection process. Nanomaterials 2022, 12, 1944. [Google Scholar] [CrossRef]
- Barzinjy, A.A.; Azeez, H.H. Green synthesis and characterization of zinc oxide nanoparticles using Eucalyptus globulus Labill. leaf extract and zinc nitrate hexahydrate salt. SN Appl. Sci. 2020, 2, 991. [Google Scholar] [CrossRef]
- Maheshwaran, G.; Muneeswari, R.S.; Bharathi, A.N.; Kumar, M.K.; Sudhahar, S. Eco-friendly synthesis of lanthanum oxide nanoparticles by Eucalyptus globulus leaf extracts for effective biomedical applications. Mater. Lett. 2021, 283, 128799. [Google Scholar] [CrossRef]
- Iconaru, S.L.; Ciobanu, C.S.; Predoi, D.; Motelica-Heino, M.; Negrilă, C.C.; Badea, M.L.; Predoi, M.V.; Chifiriuc, C.M.; Popa, M. Nitrogen and bromide Co-doped hydroxyapatite thin films with antimicrobial properties. Coatings 2021, 11, 1505. [Google Scholar] [CrossRef]
- Marinescu, G.; Culita, D.C.; Mocanu, T.; Mitran, R.-A.; Petrescu, S.; Stan, M.S.; Chifiriuc, M.C.; Popa, M. New nanostructured materials based on mesoporous silica loaded with Ru(II)/Ru(III) complexes with anticancer and antimicrobial properties. Pharmaceutics 2023, 15, 1458. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, M.; Kruszka, D.; Kachlicki, P.; Mondal, D.; Franklin, G. Uncovering the phytochemical basis and the mechanism of plant extract-mediated eco-friendly synthesis of silver nanoparticles using ultra-performance liquid chromatography coupled with a photodiode array and high-resolution mass spectrometry. ACS Sustain. Chem. Eng. 2022, 10, 562–571. [Google Scholar] [CrossRef]
- Banifatemi, S.S.; Davar, F.; Aghabarari, B.; Segura, J.A.; Alonso, F.J.; Ghoreishi, S.M. Green synthesis of CoFe2O4 nanoparticles using olive leaf extract and characterization of their magnetic properties. Ceram. Int. 2021, 47, 19198–19204. [Google Scholar] [CrossRef]
- Hosterman, B.D. Raman Spectroscopic Study of Solid Solution Spinel Oxides. Ph.D. Dissertation, University of Nevada, Las Vegas, NV, USA, August 2011. [Google Scholar] [CrossRef]
- Slotznick, S.P.; Shim, S.-H. In situ Raman spectroscopy measurements of MgAl2O4 spinel up to 1400 °C. Am. Mineral. 2008, 93, 470–476. [Google Scholar] [CrossRef]
- D’Ippolito, V.; Andreozzi, G.B.; Bersani, D.; Lottici, P.P. Raman fingerprint of chromate, aluminate and ferrite spinels. J. Raman Spectrosc. 2015, 46, 1255–1264. [Google Scholar] [CrossRef]
- Chandramohan, P.; Srinivasan, M.P.; Velmurugan, S.; Narasimhan, S.V. Cation distribution and particle size effect on Raman spectrum of CoFe2O4. J. Solid State Chem. 2011, 184, 89–96. [Google Scholar] [CrossRef]
- Motelica, L.; Ficai, D.; Oprea, O.; Ficai, A.; Trusca, R.-D.; Andronescu, E.; Holban, A.M. Biodegradable alginate films with ZnO nanoparticles and Citronella essential oil—A novel antimicrobial structure. Pharmaceutics 2021, 13, 1020. [Google Scholar] [CrossRef]
- Grigorova, M.; Blythe, H.J.; Blaskov, V.; Rusanov, V.; Petkov, V.; Masheva, V.; Nihtianova, D.; Martinez, L.; Munoz, J.S.; Mikhov, M. Magnetic properties and Mössbauer spectra of nanosized CoFe2O4 powders. J. Magn. Magn. Mater. 1998, 183, 163–172. [Google Scholar] [CrossRef]
- Palade, P.; Comanescu, C.; Kuncser, A.; Berger, D.; Matei, C.; Iacob, N.; Kuncser, V. Mesoporous cobalt ferrite nanosystems obtained by surfactant-assisted hydrothermal method: Tuning morpho-structural and magnetic properties via pH-variation. Nanomaterials 2020, 10, 476. [Google Scholar] [CrossRef]
- Hunpratub, S.; Phokha, S.; Kidkhunthod, P.; Chanlek, N.; Chindaprasirt, P. The effect of cation distribution on the magnetic properties of CoFe2O4 nanoparticles. Results Phys. 2021, 24, 104112. [Google Scholar] [CrossRef]
- Stoner, E.C.; Wohlfarth, E.P. A mechanism of magnetic hysteresis in heterogeneous alloys. Philos. Trans. R. Soc. A 1948, 240, 599. [Google Scholar] [CrossRef]
- Chikazumi, S. Physics of Ferromagnetism; Oxford University Press: Oxford, UK, 1997. [Google Scholar]




| Eucalyptus Leaves Dry Extract | N | N-800 | Assignement |
|---|---|---|---|
| 3382 vs, br | 3419 vs, br | - | νOH/H2O |
| 2915 m, sh | - | - | νCH2 asym |
| 2850 w, sh | - | - | νCH2 sym |
| - | 1635 s | - | νOH in water |
| 1616 vs | - | - | νCOO− asym |
| 1396 m | 1384 m | - | νCOO− sym/νNO3− |
| 1072 s | - | - | νC-O-C sym |
| - | 1031 w | 987 w | - |
| - | 578 s | 573 vs | νM-O spinel |
| 486 w | 490 m | νM-O spinel |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gingasu, D.; Culita, D.C.; Calderon Moreno, J.M.; Marinescu, G.; Bartha, C.; Oprea, O.; Preda, S.; Chifiriuc, M.C.; Popa, M. Synthesis of CoFe2O4 through Wet Ferritization Method Using an Aqueous Extract of Eucalyptus Leaves. Coatings 2023, 13, 1250. https://doi.org/10.3390/coatings13071250
Gingasu D, Culita DC, Calderon Moreno JM, Marinescu G, Bartha C, Oprea O, Preda S, Chifiriuc MC, Popa M. Synthesis of CoFe2O4 through Wet Ferritization Method Using an Aqueous Extract of Eucalyptus Leaves. Coatings. 2023; 13(7):1250. https://doi.org/10.3390/coatings13071250
Chicago/Turabian StyleGingasu, Dana, Daniela C. Culita, Jose Maria Calderon Moreno, Gabriela Marinescu, Cristina Bartha, Ovidiu Oprea, Silviu Preda, Mariana Carmen Chifiriuc, and Marcela Popa. 2023. "Synthesis of CoFe2O4 through Wet Ferritization Method Using an Aqueous Extract of Eucalyptus Leaves" Coatings 13, no. 7: 1250. https://doi.org/10.3390/coatings13071250
APA StyleGingasu, D., Culita, D. C., Calderon Moreno, J. M., Marinescu, G., Bartha, C., Oprea, O., Preda, S., Chifiriuc, M. C., & Popa, M. (2023). Synthesis of CoFe2O4 through Wet Ferritization Method Using an Aqueous Extract of Eucalyptus Leaves. Coatings, 13(7), 1250. https://doi.org/10.3390/coatings13071250









