Promoting the Segregation of Sr2+ from the Perovskite Oxygen Catalyst La0.5Sr0.5Co3−δ via Quenching
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
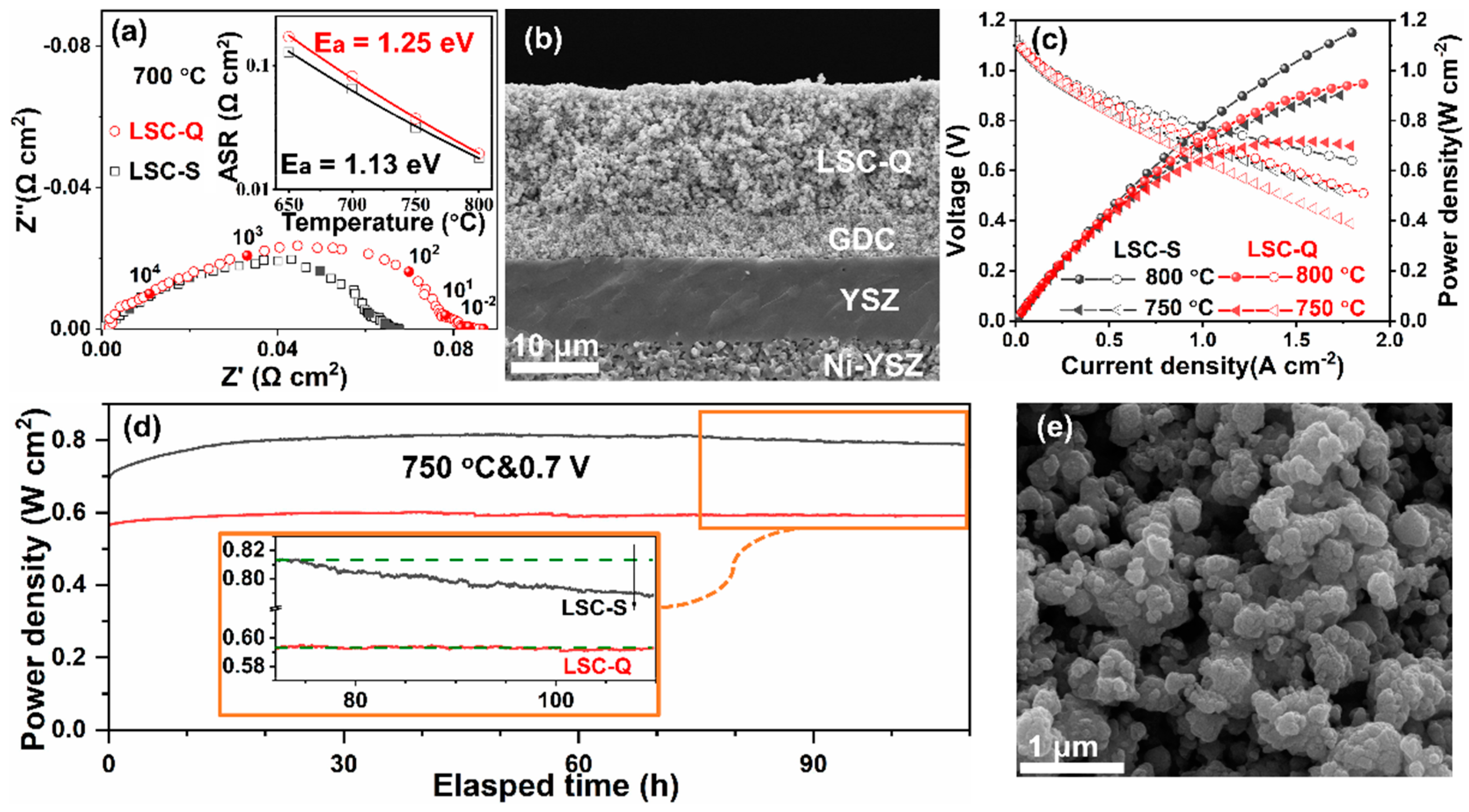
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Boldrin, P.; Brandon, N.P. Progress and outlook for solid oxide fuel cells for transportation applications. Nat. Catal. 2019, 2, 571–577. [Google Scholar] [CrossRef]
- Pirou, S.; Talic, B.; Brodersen, K.; Hauch, A.; Frandsen, H.L.; Skafte, T.L.; Persson, Å.H.; Høgh, J.V.T.; Henriksen, H.; Navasa, M.; et al. Production of a monolithic fuel cell stack with high power density. Nat. Commun. 2022, 13, 1263. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Liu, M.F.; Liu, Z.B.; Li, X.X.; Blinn, K.; Zhu, X.B.; Liu, M.L. Efficient electro-catalysts for enhancing surface activity and stability of SOFC cathodes. Adv. Energy Mater. 2013, 3, 1149–1154. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Zhu, Z. Perovskite cathode materials for low-temperature solid oxide fuel cells: Fundamentals to optimization. Electrochem. Energy Rev. 2022, 5, 263–311. [Google Scholar] [CrossRef]
- Zheng, F.; Pang, S. Progress in developing LnBaCo2O5+δ as an oxygen reduction catalyst for solid oxide fuel cells. Catalysts 2023, 13, 1288. [Google Scholar] [CrossRef]
- Hanif, M.B.; Motola, M.; Qayyum, S.; Rauf, S.; Khalid, A.; Li, C.-J.; Li, C.-X. Recent advancements, doping strategies and the future perspective of perovskite-based solid oxide fuel cells for energy conversion. Chem. Eng. J. 2022, 428, 132603. [Google Scholar] [CrossRef]
- Yatoo, M.A.; Habib, F.; Malik, A.H.; Qazi, M.J.; Ahmad, S.; Ganayee, M.A.; Ahmad, Z. Solid-oxide fuel cells: A critical review of materials for cell components. MRS Commun. 2023, 13, 378–384. [Google Scholar] [CrossRef]
- Yatoo, M.A.; Skinner, S.J. Ruddlesden-Popper phase materials for solid oxide fuel cell cathodes: A short review. Mater. Today Proc. 2022, 56, 3747–3754. [Google Scholar] [CrossRef]
- Pikalova, E.; Kolchugin, A.; Tsvinkinberg, V.; Sereda, V.; Khrustov, A.; Filonova, E. Comprehensive study of functional properties and electrochemical performance of layered lanthanum nickelate substituted with rare-earth elements. J. Power Sources 2023, 581, 233505. [Google Scholar] [CrossRef]
- Li, L.; Kong, Z.; Yao, B.; Yang, H.; Gao, Z.; Xu, L.; Dong, F.; Ni, M.; Lin, Z. An efficient and durable perovskite electrocatalyst for oxygen reduction in solid oxide fuel cells. Chem. Eng. J. 2020, 396, 125237. [Google Scholar] [CrossRef]
- Cai, C.; Xie, M.; Xue, K.; Shi, Y.; Li, S.; Liu, Y.; An, S.; Yang, H. Enhanced electrochemical performance of La0.6Sr0.4Co0.2Fe0.8O3−δ cathode via Ba-doping for intermediate-temperature solid oxide fuel cells. Nano Res. 2022, 15, 3264–3272. [Google Scholar] [CrossRef]
- Cai, Z.; Kubicek, M.; Fleig, J.; Yildiz, B. Chemical heterogeneities on La0.6Sr0.4CoO3−δ thin films-correlations to cathode surface activity and stability. Chem. Mater. 2012, 24, 1116–1127. [Google Scholar] [CrossRef]
- Pang, S.; Xu, J.; Su, Y.; Yang, G.; Zhu, M.; Cui, M.; Shen, X.; Chen, C. The role of A-site cation size mismatch in tune the catalytic activity and durability of double perovskite oxides. Appl. Catal. B Environ. 2020, 270, 118868. [Google Scholar] [CrossRef]
- Koo, B.; Kim, K.; Kim, J.K.; Kwon, H.; Han, J.W.; Jung, W. Sr segregation in perovskite oxides: Why it happens and how it exists. Joule 2018, 2, 1476–1499. [Google Scholar] [CrossRef]
- Feng, Z.; Hong, W.T.; Fong, D.D.; Lee, Y.L.; Yacoby, Y.; Morgan, D.; Shao-Horn, Y. Catalytic activity and stability of oxides: The role of near-surface atomic structures and compositions. Acc. Chem. Res. 2016, 49, 966–973. [Google Scholar] [CrossRef]
- Wei, B.; Schroeder, M.; Martin, M. Surface cation segregation and chromium deposition on the double-perovskite oxide PrBaCo2O5+δ. ACS Appl. Mater. Interfaces 2018, 10, 8621–8629. [Google Scholar] [CrossRef]
- Chen, K.; Jiang, S.P. Surface segregation in solid oxide cell oxygen electrodes: Phenomena, mitigation strategies and electrochemical properties. Electrochem. Energy Rev. 2020, 3, 730–765. [Google Scholar] [CrossRef]
- Cao, Y.; Gadre, M.J.; Ngo, A.T.; Adler, S.B.; Morgan, D.D. Factors controlling surface oxygen exchange in oxides. Nat. Commun. 2019, 10, 1346. [Google Scholar] [CrossRef]
- Rupp, G.M.; Opitz, A.K.; Nenning, A.; Limbeck, A.; Fleig, J. Real-time impedance monitoring of oxygen reduction during surface modification of thin film cathodes. Nat. Mater. 2017, 16, 640–645. [Google Scholar] [CrossRef]
- Kubicek, M.; Limbeck, A.; Frömling, T.; Hutter, H.; Fleig, J. Relationship between cation segregation and the electrochemical oxygen reduction kinetics of La0.6Sr0.4CoO3−δ thin film electrodes. J. Electrochem. Soc. 2011, 158, B727–B734. [Google Scholar] [CrossRef]
- Crumlin, E.J.; Mutoro, E.; Liu, Z.; Grass, M.E.; Biegalski, M.D.; Lee, Y.-L.; Morgan, D.; Christen, H.M.; Bluhm, H.; Shao-Horn, Y. Surface strontium enrichment on highly active perovskites for oxygen electrocatalysis in solid oxide fuel cells. Energy Environ. Sci. 2012, 5, 6081–6088. [Google Scholar] [CrossRef]
- Feng, Z.; Crumlin, E.J.; Hong, W.T.; Lee, D.; Mutoro, E.; Biegalski, M.D.; Zhou, H.; Bluhm, H.; Christen, H.M.; Shao-Horn, Y. In situ studies of the temperature-dependent surface structure and chemistry of single-crystalline (001)-oriented La0.8Sr0.2CoO3−δ perovskite thin films. J. Phys. Chem. Lett. 2013, 4, 1512–1518. [Google Scholar] [CrossRef]
- Kubicek, M.; Rupp, G.M.; Huber, S.; Opitz, A.K.; Bernardi, J.; Stöger-Pollach, M.; Hutter, H.; Fleig, J. Cation diffusion in La0.6Sr0.4CoO3−δ below 800 °C and its relevance for Sr segregation. Phys. Chem. Chem. Phys. 2014, 16, 2715–2726. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, W.; Wu, T.; Zheng, Y.; Chen, J.; Yu, B.; Zhu, J.; Liu, M. Segregation induced self-assembly of highly active perovskite for rapid oxygen reduction reaction. Adv. Energy Mater. 2018, 8, 1801893. [Google Scholar] [CrossRef]
- Pang, S.; Song, Y.; Cui, M.; Tang, X.; Long, C.; Ke, L.; Yang, G.; Fang, T.; Guan, Y.; Chen, C. Rapid and durable oxygen reduction reaction enabled by a perovskite oxide with self-cleaning surface. J. Energy Chem. 2023, 83, 333–340. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Zheng, Y.; Chen, J.; Yu, B.; Chen, Y.; Liu, M. Controlling cation segregation in perovskite-based electrodes for high electro-catalytic activity and durability. Chem. Soc. Rev. 2017, 46, 6345–6378. [Google Scholar] [CrossRef] [PubMed]
- Tsvetkov, N.; Lu, Q.; Sun, L.; Crumlin, E.J.; Yildiz, B. Improved chemical and electrochemical stability of perovskite oxides with less reducible cations at the surface. Nat. Mater. 2016, 15, 1010–1016. [Google Scholar] [CrossRef]
- Pang, S.; Yang, G.; Jiang, X.; Shen, X.; Rao, D.; Chen, C. Insight into tuning the surface and bulk microstructure of perovskite catalyst through control of cation non-stoichiometry. J. Catal. 2020, 381, 408–414. [Google Scholar] [CrossRef]
- Koo, B.; Kwon, H.; Kim, Y.; Seo, H.G.; Han, J.W.; Jung, W. Enhanced oxygen exchange of perovskite oxide surfaces through strain-driven chemical stabilization. Energy Environ. Sci. 2018, 11, 71–77. [Google Scholar] [CrossRef]
- Ke, L.; Pang, S.; Long, C.; Fang, T.; Yang, G.; Song, Y.; He, X.; Ma, S.; Qian, Y.; Shen, X.; et al. Quenching-induced surface reconstruction of perovskite oxide for rapid and durable oxygen catalysis. Chem. Eng. J. 2023, 463, 142509. [Google Scholar] [CrossRef]
- Fan, L.; Wang, J.; Huang, Z.; Yao, X.; Hou, N.; Gan, T.; Gan, J.; Zhao, Y.; Li, Y. Enhancement of the electrocatalytic activity of La0.6Sr0.4Co0.2Fe0.8O3−δ through surface modification by acid etching. Catal. Today 2021, 364, 97–103. [Google Scholar] [CrossRef]
- Siebenhofer, M.; Riedl, C.; Nenning, A.; Artner, W.; Rameshan, C.; Opitz, A.K.; Fleig, J.; Kubicek, M. Improving and degrading the oxygen exchange kinetics of La0.6Sr0.4CoO3−δ by Sr decoration. J. Mater. Chem. A 2023, 11, 12827–12836. [Google Scholar] [CrossRef]
- Choi, M.; Lee, W. Tuning the oxygen vacancy concentration in a heterostructured electrode for high chemical and electrochemical stabilities. Chem. Eng. J. 2022, 431, 134345. [Google Scholar] [CrossRef]
- Li, J.; Zhou, X.; Wu, C.; Zhao, L.; Dong, B.; Wang, S.; Chi, B. Self-stabilized hybrid cathode for solid oxide fuel cell: A-site deficient perovskite coating as solid solution for strontium diffusion. Chem. Eng. J. 2022, 438, 135446. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, J.; Ye, Y.; Sun, X.; Chen, H.; Zhou, D.; Yin, Y.; Zhang, N.; Ling, Y.; Ciucci, F.; et al. Enhancing the intrinsic activity and stability of perovskite cobaltite at elevated temperature through surface stress. Small 2021, 17, 2104144. [Google Scholar] [CrossRef]
- Lee, D.; Jacobs, R.; Jee, Y.; Seo, A.; Sohn, C.; Ievlev, A.V.; Ovchinnikova, O.S.; Huang, K.; Morgan, D.; Lee, H.N. Stretching epitaxial La0.6Sr0.4CoO3−δ for fast oxygen reduction. J. Phys. Chem. C 2017, 121, 25651–25658. [Google Scholar] [CrossRef]
- Kubicek, M.; Cai, Z.; Ma, W.; Yildiz, B.; Hutter, H.; Fleig, J. Tensile lattice strain accelerates oxygen surface exchange and diffusion in La1−xSrxCoO3−δ thin films. ACS Nano 2013, 7, 3276–3286. [Google Scholar] [CrossRef]
- Zhuang, Z.; Li, Y.; Yu, R.; Xia, L.; Yang, J.; Lang, Z.; Zhu, J.; Huang, J.; Wang, J.; Wang, Y.; et al. Reversely trapping atoms from a perovskite surface for high-performance and durable fuel cell cathodes. Nat. Catal. 2022, 5, 300–310. [Google Scholar] [CrossRef]
- Lee, W.; Han, J.W.; Chen, Y.; Cai, Z.; Yildiz, B. Cation size mismatch and charge interactions drive dopant segregation at the surfaces of manganite perovskites. J. Am. Chem. Soc. 2013, 135, 7909–7925. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.; Su, Y.; Yang, G.; Shen, X.; Zhu, M.; Wu, X.; Li, S.; Yang, X.; Xi, X. Enhanced electrochemical performance of Ca-doped NdBa1−xCaxCoCuO5+δ as cathode material for intermediate-temperature solid oxide fuel cells. Ceram. Int. 2018, 44, 21902–21907. [Google Scholar] [CrossRef]
- Druce, J.; Téllez, H.; Burriel, M.; Sharp, M.D.; Fawcett, L.J.; Cook, S.N.; McPhail, D.S.; Ishihara, T.; Brongersma, H.H.; Kilner, J.A. Surface termination and subsurface restructuring of perovskite-based solid oxide electrode materials. Energy Environ. Sci. 2014, 7, 3593–3599. [Google Scholar] [CrossRef]
- Li, W.; Chu, X.-S.; Wang, F.; Dang, Y.-Y.; Liu, X.-Y.; Wang, X.-C.; Wang, C.-Y. Enhanced cocatalyst-support interaction and promoted electron transfer of 3D porous g-C3N4/GO-M (Au, Pd, Pt) composite catalysts for hydrogen evolution. Appl. Catal. B Environ. 2021, 288, 120034. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, C.; Zhang, S.; Song, X.; Tang, Y.; Cheng, H.-M. Reversible calcium alloying enables a practical room-temperature rechargeable calcium-ion battery with a high discharge voltage. Nat. Chem. 2018, 10, 667–672. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, F.; Qian, Y.; Pang, S. Promoting the Segregation of Sr2+ from the Perovskite Oxygen Catalyst La0.5Sr0.5Co3−δ via Quenching. Coatings 2023, 13, 2032. https://doi.org/10.3390/coatings13122032
Zheng F, Qian Y, Pang S. Promoting the Segregation of Sr2+ from the Perovskite Oxygen Catalyst La0.5Sr0.5Co3−δ via Quenching. Coatings. 2023; 13(12):2032. https://doi.org/10.3390/coatings13122032
Chicago/Turabian StyleZheng, Fa, Yaozheng Qian, and Shengli Pang. 2023. "Promoting the Segregation of Sr2+ from the Perovskite Oxygen Catalyst La0.5Sr0.5Co3−δ via Quenching" Coatings 13, no. 12: 2032. https://doi.org/10.3390/coatings13122032
APA StyleZheng, F., Qian, Y., & Pang, S. (2023). Promoting the Segregation of Sr2+ from the Perovskite Oxygen Catalyst La0.5Sr0.5Co3−δ via Quenching. Coatings, 13(12), 2032. https://doi.org/10.3390/coatings13122032






