Isolation, Characterization and Chemical Synthesis of Large Spectrum Antimicrobial Cyclic Dipeptide (l-leu-l-pro) from Streptomyces misionensis V16R3Y1 Bacteria Extracts. A Novel 1H NMR Metabolomic Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Media
2.2. Microbial Strains Origin
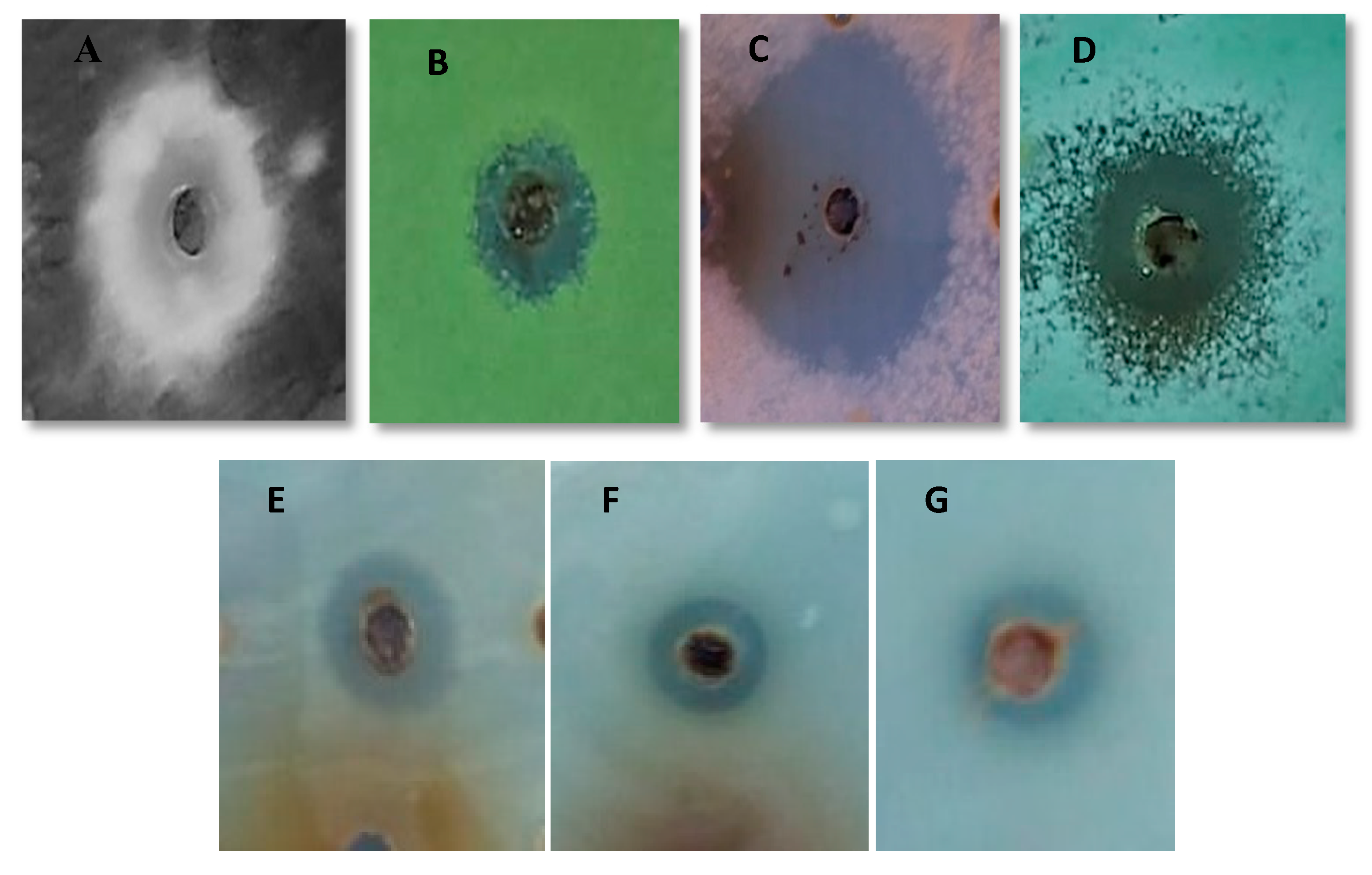
2.3. Antimicrobial Activity Tests
2.4. Minimum Inhibitory Concentration (MIC) Assay
2.5. Screening of Solvent for the Extraction of Antimicrobial Compound V16R3Y1
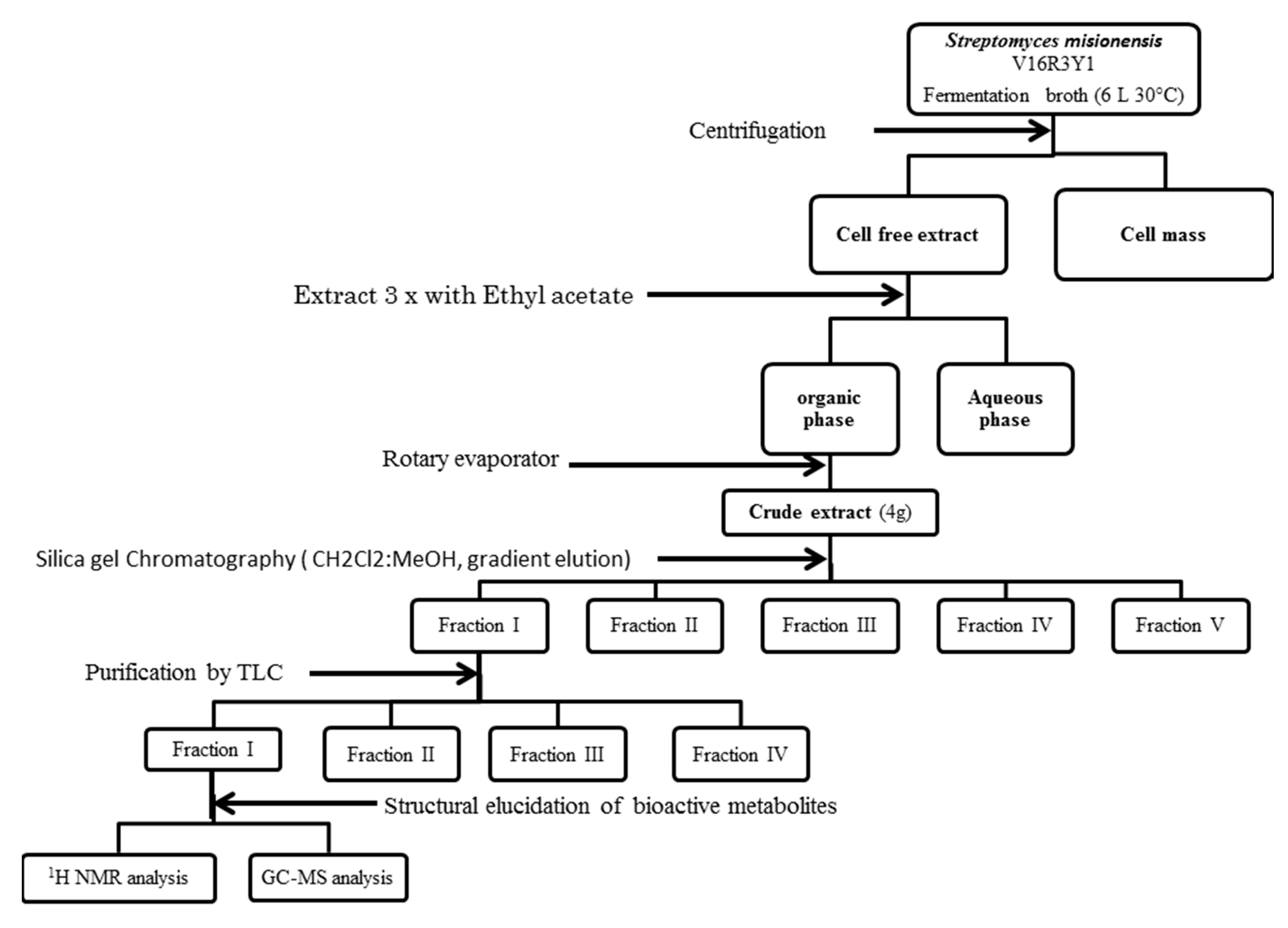
2.6. Purification of Bioactive Compounds
2.7. Thin Layer Chromatography (TLC) Procedure
2.8. Gas Chromatography–Mass Spectrometry (GC–MS) Analysis
2.9. 1H Nuclear Magnetic Resonance (NMR)
2.10. Application of Metabolomic Procedure for the Identification of the Extracted Metabolites
2.11. Synthesis of the Cyclo Dipeptide (Leu-Pro) in Solution
2.12. Liquid Chromatography–Mass Spectrometry (HPLC–MS) Analysis
3. Results
3.1. Extraction and purification of Bioactive Compounds
3.2. Gas Chromatography–Mass Spectrometry (GC–MS) Analysis
3.3. NMR Spectra Analysis with the New Procedure
3.4. Cyclo Dipeptide Synthesis
3.5. Biological Activity of Biological Fractionand Synthetic Peptide
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vu, H.-N.T.; Nguyen, D.T.; Nguyen, H.Q.; Chu, H.H.; Chu, S.K.; Chau, M.V.; Phi, Q.-T. Antimicrobial and Cytotoxic Properties of Bioactive Metabolites Produced by Streptomyces cavourensis YBQ59 Isolated from Cinnamomum cassia Prels in Yen Bai Province of Vietnam. Curr. Microbiol. 2018, 75, 1247–1255. [Google Scholar] [CrossRef]
- Demain, A.L.; Sanchez, S. Microbial drug discovery: 80 years of progress. J. Antibiot. 2009, 62, 5–16. [Google Scholar] [CrossRef]
- Strobel, G.; Daisy, B. Bioprospecting for Microbial Endophytes and Their Natural Products. Microbiol. Mol. Biol. Rev. 2003, 67, 491–502. [Google Scholar] [CrossRef]
- Solecka, J.; Zajko, J.; Postek, M.; Rajnisz, A. Biologically active secondary metabolites from Actinomycetes. Open Life Sci. 2012, 7, 373–390. [Google Scholar] [CrossRef]
- Bérdy, J. Bioactive Microbial Metabolites: A Personal View. J. Antibiot. 2005, 58, 1–26. [Google Scholar] [CrossRef]
- Kornsakulkarn, J.; Saepua, S.; Srijomthong, K.; Rachtawee, P.; Thongpanchang, C. Quinazolinone alkaloids from actinomycete Streptomyces sp. BCC 21795. Phytochem. Lett. 2015, 12, 6–8. [Google Scholar] [CrossRef]
- Borthwick, A.D. 2,5-Diketopiperazines: Synthesis, Reactions, Medicinal Chemistry, and Bioactive Natural Products. Chem. Rev. 2012, 112, 3641–3716. [Google Scholar] [CrossRef]
- Ortiz, A.; Sansinenea, E. Cyclic Dipeptides: Secondary Metabolites Isolated from Different Microorganisms with Diverse Biological Activities. CMC 2017, 24, 2773–2780. [Google Scholar] [CrossRef]
- Giessen, T.W.; Marahiel, M.A. Rational and combinatorial tailoring of bioactive cyclic dipeptides. Front. Microbiol. 2015, 6, 785. [Google Scholar] [CrossRef]
- Ferjani, R.; Marasco, R.; Rolli, E.; Cherif, H.; Cherif, A.; Gtari, M.; Boudabous, A.; Daffonchio, D.; Ouzari, H.-I. The Date Palm Tree Rhizosphere Is a Niche for Plant Growth Promoting Bacteria in the Oasis Ecosystem. BioMed Res. Int. 2015, 2015, 153851. [Google Scholar] [CrossRef]
- NCCLS; NCFCLS. Performance Standards for Antimicrobial Disk Susceptibility Tests: Approved Standard; NCCLS Doc. M2-A7; National Committee for Clinical Laboratory Standards: Wayne, PA, USA, 2000; p. 20. [Google Scholar]
- Cappuccino, J.; Sherman, N. Microbiology: A Laboratory Manual; Pearson/Benjamin Cummings: San Francisco, CA, USA, 2005; p. 507. [Google Scholar]
- El Euch, I.Z.; Frese, M.; Sewald, N.; Smaoui, S.; Shaaban, M.; Mellouli, L. Bioactive secondary metabolites from new terrestrial Streptomyces sp. TN82 strain: Isolation, structure elucidation and biological activity. Med. Chem. Res. 2018, 27, 1085–1092. [Google Scholar] [CrossRef]
- Weljie, A.M.; Newton, J.; Mercier, P.; Carlson, E.; Slupsky, C.M. Targeted Profiling: Quantitative Analysis of 1H NMR Metabolomics Data. Anal. Chem. 2006, 78, 4430–4442. [Google Scholar] [CrossRef]
- Chang, D.; Weljie, A.; Newton, J. Leveraging latent information in nmr spectra for robust predictive models. Biocomputing 2007, 2007, 115–126. [Google Scholar] [CrossRef]
- Zulak, K.G.; Weljie, A.M.; Vogel, H.J.; Facchini, P.J. Quantitative 1H NMR metabolomics reveals extensive metabolic reprogramming of primary and secondary metabolism in elicitor-treated opium poppy cell cultures. BMC Plant Biol. 2008, 8, 5. [Google Scholar] [CrossRef]
- Mercier, P.; Lewis, M.J.; Chang, D.; Baker, D.; Wishart, D.S. Towards automatic metabolomic profiling of high-resolution one-dimensional proton NMR spectra. J. Biomol. NMR 2011, 49, 307–323. [Google Scholar] [CrossRef]
- He, R.; Wang, B.; Wakimoto, T.; Wang, M.; Zhu, L.; Abe, I. Cyclodipeptides from metagenomic library of a japanese marine sponge. J. Braz. Chem. Soc. 2013, 24, 1926–1932. [Google Scholar] [CrossRef]
- O’Neill, J.C.; Lin, Q.; Geske, G.D.; Blackwell, H.E. New and Unexpected Insights into the Modulation of LuxR-type Quorum Sensing by Cyclic Dipeptides. ACS Chem. Biol. 2009, 4, 1051–1059. [Google Scholar]
- Thirumurugan, D.; Vijayakumar, R.; Vadivalagan, C.; Karthika, P.; Alam Khan, M.K. Isolation, structure elucidation and antibacterial activity of methyl-4,8-dimethylundecanate from the marine actinobacterium Streptomyces albogriseolus ECR64. Microb. Pathog. 2018, 121, 166–172. [Google Scholar] [CrossRef]
- Baskaran, R.; Mohan, P.M.; Sivakumar, K.; Kumar, A. Antimicrobial Activity and Phylogenetic Analysis of Streptomyces parvulus Dosmb-D105 Isolated from the Mangrove Sediments of Andaman Islands. Acta Microbiol. Immunol. Hung. 2016, 63, 27–46. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Lindon, J.C.; Holmes, E. “Metabonomics”: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 1999, 29, 1181–1189. [Google Scholar] [CrossRef]
- Beckonert, O.; Keun, H.C.; Ebbels, T.M.D.; Bundy, J.; Holmes, E.; Lindon, J.C.; Nicholson, J.K. Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat. Protoc. 2007, 2, 2692–2703. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Wilson, I.D. High resolution proton magnetic resonance spectroscopy of biological fluids. Prog. Nucl. Magn. Reson. Spectrosc. 1989, 21, 449–501. [Google Scholar] [CrossRef]
- Keun, H.C.; Athersuch, T.J. Nuclear Magnetic Resonance (NMR)-Based Metabolomics. In Metabolic Profiling; Humana Press: Totowa, NJ, USA, 2011; pp. 321–334. [Google Scholar]
- Nicholson, J.K.; Buckingham, M.J.; Sadler, P.J. High resolution 1H n.m.r. studies of vertebrate blood and plasma. Biochem. J. 1983, 211, 605–615. [Google Scholar] [CrossRef]
- Nicholson, J.K.; O’Flynn, M.P.; Sadler, P.J.; Macleod, A.F.; Juul, S.M.; Sönksen, P.H. Proton-nuclear-magnetic-resonance studies of serum, plasma and urine from fasting normal and diabetic subjects. Biochem. J. 1984, 217, 365–375. [Google Scholar] [CrossRef]
- Kumar, S.N.; Mohandas, C.; Siji, J.V.; Rajasekharan, K.N.; Nambisan, B. Identification of antimicrobial compound, diketopiperazines, from a Bacillus sp. N strain associated with a rhabditidentomopathogenic nematode against major plant pathogenic fungi. J. Appl. Microbiol. 2012, 113, 914–924. [Google Scholar] [CrossRef]
- Mangamuri, U.K.; Muvva, V.; Poda, S.; Manavathi, B.; Bhujangarao, C.; Yenamandra, V. Chemical characterization & bioactivity of diketopiperazine derivatives from the mangrove derived Pseudonocardiaendophytica. Egypt. J. Aquat. Res. 2016, 42, 169–175. [Google Scholar]
- Brandl, C.J.; Deber, C.M. Hypothesis about the function of membrane-buried proline residues in transport proteins. Proc. Natl. Acad. Sci. USA 1986, 83, 917–921. [Google Scholar] [CrossRef]
- Balagurunathan, R.; Subramanian, A. Antagonistic streptomycetes from marine sediments. Adv. Biosci. 2001, 20, 71–76. [Google Scholar]
- Dal Bello, F.; Clarke, C.I.; Ryan, L.A.M.; Ulmer, H.; Schober, T.J.; Ström, K.; Sjögren, J.; van Sinderen, D.; Schnürer, J.; Arendt, E.K. Improvement of the quality and shelf life of wheat bread by fermentation with the antifungal strain Lactobacillus plantarum FST 1.7. J. Cereal Sci. 2007, 45, 309–318. [Google Scholar] [CrossRef]
- Johnson, J.L.; Jackson, W.G.; Eble, T.E. Isolation of L-Leucyl-L-proline Anhydride from Microbiological Fermentations. J. Am. Chem. Soc. 1951, 73, 2874–2948. [Google Scholar] [CrossRef]
- Yang, M.-L.; Kuo, P.-C.; Hwang, T.-L.; Wu, T.-S. Anti-inflammatory Principles from Cordycepssinensis. J. Nat. Prod. 2011, 74, 1996–2000. [Google Scholar] [CrossRef]
- Yan, P.-S.; Song, Y.; Sakuno, E.; Nakajima, H.; Nakagawa, H.; Yabe, K. Cyclo(l-Leucyl-l-Prolyl) Produced by Achromobacterxylosoxidans Inhibits Aflatoxin Production by Aspergillusparasiticus. Appl. Environ. Microbiol. 2004, 70, 7466–7473. [Google Scholar] [CrossRef]
- Song, G.C.; Choi, H.K.; Kim, Y.S.; Choi, J.S.; Ryu, C.-M. Seed defense biopriming with bacterial cyclo-dipeptides triggers immunity in cucumber and pepper. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Narasaiah, B. Structural elucidation and antimicrobial activity of secondary metabolites from Streptomyces albus CN-4. Int. J. Res. Eng. Appl. Sci. 2016, 6, 77–89. [Google Scholar]
- Tangjitjaroenkun, J. Evaluation of antioxidant, antibacterial and gas chromatography-mass spectrometry analysis of ethyl acetate extract of Streptomyces omiyaensis sch2. Evaluation 2018, 11. [Google Scholar] [CrossRef]

| Retention Time; (Peak Area %) | m/z | Name of the Compound | Chemical Structure | Activity a |
|---|---|---|---|---|
| 7.614 (10.088) | 134.89; 91.88; 90.92; 64.61 | Phenylacetamide | MF: C8H9NO MW: 135.166 g/mol 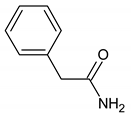 | Drug, antimalarial |
| 9.247 (2.880) | 84.91; 70.88; 56.74; 54.73 | Hexyl-(2-ethylhexyl)sulfate | MF: C14H30O3S MW: 278.451 g/mol  | Non active |
| 10.859 (28.241) | 153.88; 69.59; 56.66 | N-Valeryl-l-proline decyl ester | MF: C20H37NO3 MW: 339.5 g/mol 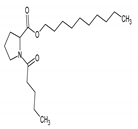 | Non active |
| 11.004 (1.758) | 153.96; 54.57; 69.67 | 5-Isopropylidene-3,3-dimethyl-dihydrofuran-2-one | MF: C9H14O MW: 154.206 g/mol 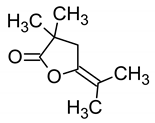 | Non active |
| 11.669 (57.033) | 154.04; 124.91; 70.88; 69.59 | cyclo-(l-prolyl-l-leucine); cyclo-(l-leu-l-pro) | MF: C11H18N2O2 MW: 210.272 g/mol  | -Antimicrobial -Antitumoral -Antiviral -Cytotoxic and neuroprotective effects |
| Test Microbes | Inhibition Zone (mm) BF (400 μg) | Inhibition Zone (mm) SP (300μg) | SP MIC (μg/mL) ± SD |
|---|---|---|---|
| Fusarium oxysporum | 37 | 40 | 16 ± 1.00 |
| Aspergillus flavus | 13 | 15 | 16 ± 1.70 |
| Penicillium expansum | 24 | 27 | 18 ± 1.00 |
| Aspergillus niger | 13 | 15 | 17 ± 0.00 |
| Candida albicans | 10 | 12 | 50 ± 1.00 |
| Candida metapsilosis | 9 | 10 | 32 ± 1.00 |
| Candida parapsilosis | 8 | 9 | 30 ± 2.60 |
| Escherichia fergusonii | 5 | 6 | 230 ± 1.00 |
| Salmonella enterica | 7 | 8 | 11 ± 0.00 |
| Enterococcus faecalis | 6 | 7 | 12 ± 1.70 |
| Bacillus cereus | 5 | 7 | 16 ± 1.00 |
| Staphylococcus aureus | 5 | 6 | 30 ± 1.00 |
| Pseudomonas aeruginosa | 5 | 6 | 34 ± 1.70 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saadouli, I.; Zendah El Euch, I.; Trabelsi, E.; Mosbah, A.; Redissi, A.; Ferjani, R.; Fhoula, I.; Cherif, A.; Sabatier, J.-M.; Sewald, N.; et al. Isolation, Characterization and Chemical Synthesis of Large Spectrum Antimicrobial Cyclic Dipeptide (l-leu-l-pro) from Streptomyces misionensis V16R3Y1 Bacteria Extracts. A Novel 1H NMR Metabolomic Approach. Antibiotics 2020, 9, 270. https://doi.org/10.3390/antibiotics9050270
Saadouli I, Zendah El Euch I, Trabelsi E, Mosbah A, Redissi A, Ferjani R, Fhoula I, Cherif A, Sabatier J-M, Sewald N, et al. Isolation, Characterization and Chemical Synthesis of Large Spectrum Antimicrobial Cyclic Dipeptide (l-leu-l-pro) from Streptomyces misionensis V16R3Y1 Bacteria Extracts. A Novel 1H NMR Metabolomic Approach. Antibiotics. 2020; 9(5):270. https://doi.org/10.3390/antibiotics9050270
Chicago/Turabian StyleSaadouli, Ilhem, Imène Zendah El Euch, Emna Trabelsi, Amor Mosbah, Alaeddine Redissi, Raoudha Ferjani, Imene Fhoula, Ameur Cherif, Jean-Marc Sabatier, Norbert Sewald, and et al. 2020. "Isolation, Characterization and Chemical Synthesis of Large Spectrum Antimicrobial Cyclic Dipeptide (l-leu-l-pro) from Streptomyces misionensis V16R3Y1 Bacteria Extracts. A Novel 1H NMR Metabolomic Approach" Antibiotics 9, no. 5: 270. https://doi.org/10.3390/antibiotics9050270
APA StyleSaadouli, I., Zendah El Euch, I., Trabelsi, E., Mosbah, A., Redissi, A., Ferjani, R., Fhoula, I., Cherif, A., Sabatier, J.-M., Sewald, N., & Ouzari, H.-I. (2020). Isolation, Characterization and Chemical Synthesis of Large Spectrum Antimicrobial Cyclic Dipeptide (l-leu-l-pro) from Streptomyces misionensis V16R3Y1 Bacteria Extracts. A Novel 1H NMR Metabolomic Approach. Antibiotics, 9(5), 270. https://doi.org/10.3390/antibiotics9050270






