Treatment Modalities and Antimicrobial Stewardship Initiatives in the Management of Intra-Abdominal Infections
Abstract
:1. Introduction
2. Appendicitis
2.1. Epidemiology/Pathogenesis
2.2. Microbiology
2.3. Clinical Presentation/Diagnosis
2.4. Treatment
2.5. Conclusion
3. Diverticulitis
3.1. Epidemiology/Pathogenesis
3.2. Microbiology
3.3. Clinical Presentation/Diagnosis
3.4. Treatment
3.5. Conclusion
4. Cholecystitis
4.1. Epidemiology/Pathogenesis
4.2. Microbiology
4.3. Clinical Presentation/Diagnosis
4.4. Treatment
| Indication | SIS-IDSA [2] | WSES [3] | TG13 [4] |
|---|---|---|---|
| Community-acquired | Acute cholecystitis, mild-to-moderate severity
| Biliary IAI, stable, non-critical patients with no risk factors for ESBL pathogens a
| 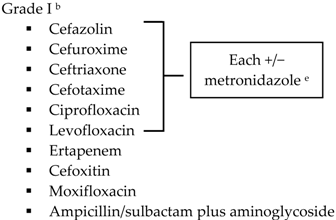 |
Biliary IAI, stable, non-critical patients with risk factors for ESBL pathogens a
| 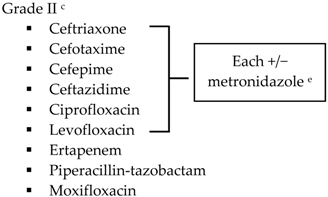 | ||
| Community-acquired | 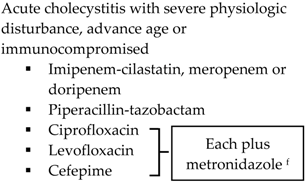 | Biliary IAI, critically ill patients with no risk factors for ESBL pathogens a
|  |
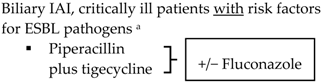 | |||
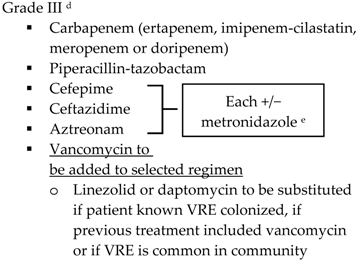
| Health-care associated | Biliary infection of any severity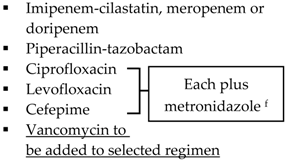 | Health-care associated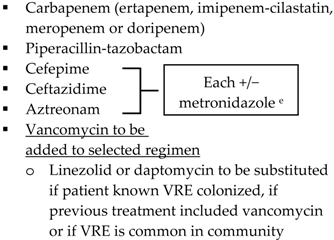 | |
| Other | 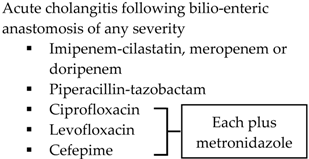 |
4.5. Conclusion
5. Antimicrobial Stewardship Initiatives
| Guideline | Indication | Treatment |
|---|---|---|
| SIS-IDSA [2] | Community-acquired IAIs of mild-moderate severity including perforated or abscessed appendicitis | Single agent
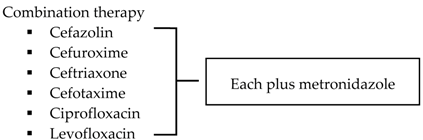 |
| Community-acquired IAIs of high risk or severity a | 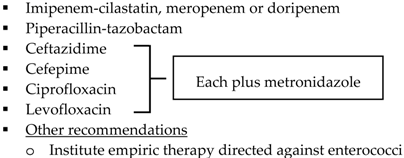 | |
| Hospital-acquired IAIs |  | |
| WSES [3] | Community-acquired extra-biliary IAIs | Stable, non-critical patients with no risk factors for ESBL pathogens b
|
Stable, non-critical patients
with risk factors for ESBL pathogens b
| ||
Critically ill patients with no risk factors for ESBL pathogens b
| ||
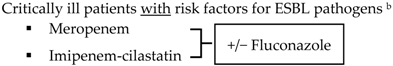 | ||
| Hospital-acquired extra-biliary IAIs | Stable, non-critical patients
with risk factors for MDR pathogens c
| |
Critically ill patients
with risk factors for MDR pathogens c
|
| Guideline | Recommendation | Strength of Recommendation a |
|---|---|---|
| SIS-IDSA [2] |
| A-II b |
| B-II c | |
| WSES [3] |
| 1A d |
| 2B e | |
| ACS [60] |
| - |
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Armstrong, C. Updated guideline on diagnosis and treatment of intra-abdominal infections. Am. Fam. Phys. 2010, 82, 694–709. [Google Scholar]
- Solomkin, J.S.; Mazuski, J.E.; Bradley, J.S.; Rodvold, K.A.; Goldstein, E.J.C.; Baron, E.J.; O’Neill, P.J.; Chow, A.W.; Dellinger, E.P.; Eachempati, S.R.; et al. Diagnosis and management of complicated intra-abdominal infections in adults and children: Guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin. Infect. Dis. 2010, 50, 133–164. [Google Scholar] [CrossRef] [PubMed]
- Sartelli, M.; Viale, P.; Catena, F.; Ansaloni, L.; Moore, E.; Malangoni, M.; Moore, F.A.; Velmahos, G.; Coimbra, R.; Ivatury, R.; et al. 2013 WSES guidelines for management of intra-abdominal infections. World J. Emerg. Surg. 2013, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Gomi, H.; Solomkin, J.S.; Takada, T.; Strasberg, S.M.; Pitt, H.A.; Yoshida, M.; Kusachi, S.; Mayumi, T.; Miura, F.; Kiriyama, S.; et al. TG13 antimicrobial therapy for acute cholangitis and cholecystitis. J. Hepatobiliary Pancreat. Sci. 2013, 20, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Sartelli, M.; Catena, F.; Coccolini, F.; Pinna, A.D. Antimicrobial management of intra-abdominal infections: Literature’s guidelines. World J. Gastroenterol. 2012, 18, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E., Jr.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, W.C.; Paterson, D.L.; Fishman, N.O.; Carpenter, C.F.; et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Sartelli, M. A focus on intra-abdominal infections. World J. Emerg. Surg. 2010, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Sifri, C.D.; Madoff, L.C. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 7th ed.; Churchill Livingstone Elsevier: Philadelphia, PA, USA, 2009; Volume 75, pp. 1059–1062. [Google Scholar]
- Livingston, E.H.; Fomby, T.B.; Woodward, W.A.; Haley, R.W. Epidemiological similarities between appendicitis and diverticulitis suggesting a common underlying pathogenesis. Arch. Surg. 2011, 146, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Ramdass, M.J.; Young Sing, Q.; Milne, D.; Mooteeram, J.; Barrow, S. Association between the appendix and fecalith in adults. Can. J. Surg. 2015, 58, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Bouassria, A.; Ibn Majdoub, K.; Yazough, I.; Ousadden, A.; Mazaz, K.; Taleb, K.A. Traumatic appendicitis: A case report and literature review. Would J. Emerg. Surg. 2013, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Lob, S.H.; Badal, R.E.; Bouchillon, S.K.; Hawser, S.P.; Hackel, M.A.; Hoban, D.J. Epidemiology and susceptibility of Gram-negative appendicitis pathogens: SMART 2008–2010. Surg. Infect. 2013, 14, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Foo, F.J.; Beckingham, I.J.; Ahmed, I. Intra-operative culture swabs in acute appendicitis: A waste of resources. Surgeon 2008, 6, 278–281. [Google Scholar] [CrossRef]
- Krajewski, S.; Brown, J.; Phang, P.T.; Raval, M.; Brown, C.J. Impact of computed tomography of the abdomen on clinical outcomes in patients with acute right lower quadrant pain: A meta-analysis. Can. J. Surg. 2011, 54, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Pinto Leite, N.; Pereira, J.M.; Cunha, R.; Pinto, P.; Sirlin, C. CT evaluation of appendicitis and its complications: Imaging techniques and key diagnostic findings. AJR Am. J. Roentgenol. 2005, 185, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Terasawa, T.; Blackmore, C.C.; Bent, S.; Kohlwes, R.J. Systematic review: Computed tomography and ultrasonography to detect acute appendicitis in adults and adolescents. Ann. Intern. Med. 2004, 141, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Prosenz, J.; Hirtler, L. Rovsing sign revisited-effects of an erroneous translation on medical teaching and research. J. Surg. Educ. 2014, 71, 738–742. [Google Scholar] [CrossRef] [PubMed]
- Jaschinski, T.; Mosch, C.; Eikermann, M.; Neugebauer, E.A. Laparoscopic versus open appendectomy in patients with suspected appendicitis: A systematic review of meta-analyses of randomised controlled trials. BMC Gastroenterol. 2015, 15, 48. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.G.; Ahn, E.J.; Kim, S.Y.; Chung, I.Y.; Park, J.M.; Park, S.H.; Choi, K.W. A clinical comparison of laparoscopic versus open appendectomy for complicated appendicitis. J. Korean Soc. Coloproctol. 2011, 27, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Flum, D.R. Clinical practice. Acute appendicitis—Appendectomy or the “antibiotics first” strategy. N. Engl. J. Med. 2015, 372, 1937–1943. [Google Scholar] [PubMed]
- Vons, C.; Barry, C.; Maitre, S.; Pautrat, K.; Leconte, M.; Costaglioli, B.; Karoui, M.; Alves, A.; Dousset, B.; Valleur, P.; et al. Amoxicillin plus clavulanic acid versus appendicectomy for treatment of acute uncomplicated appendicitis: An open-label, non-inferiority, randomised controlled trial. Lancet 2011, 377, 1573–1579. [Google Scholar] [CrossRef]
- Salminen, P.; Paajanen, H.; Rautio, T.; Nordström, P.; Aarnio, M.; Rantanen, T.; Tuominen, R.; Hurme, S.; Virtanen, J.; Mecklin, J.P.; et al. Antibiotic therapy vs. appendectomy for treatment of uncomplicated acute appendicitis: The APPAC randomized clinical trial. JAMA 2015, 313, 2340–2348. [Google Scholar] [CrossRef] [PubMed]
- Schug-Pass, C.; Geers, P.; Hugel, O.; Lippert, H.; Köckerling, F. Prospective randomized trial comparing short-term antibiotic therapy versus standard therapy for acute uncomplicated sigmoid diverticulitis. Int. J. Colorectal Dis. 2010, 25, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Varadhan, K.K.; Humes, D.J.; Neal, K.R.; Lobo, D.N. Antibiotic therapy versus appendectomy for acute appendicitis: A meta-analysis. World J. Surgery 2010, 34, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Ansaloni, L.; Catena, F.; Coccolini, F.; Ercolani, G.; Gazzotti, F.; Pasqualini, E.; Pinna, A.D. Surgery versus conservative antibiotic treatment in acute appendicitis: A systematic review and meta-analysis of randomized controlled trials. Dig. Surg. 2011, 28, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Fogg, L. Use of antibiotics alone for treatment of uncomplicated acute appendicitis: A systematic review and meta-analysis. Surgery 2011, 150, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Wilms, I.M.; de Hoog, D.E.; de Visser, D.C.; Janzing, H.M. Appendectomy versus antibiotic treatment for acute appendicitis. Cochrane Database Syst. Rev. 2011, CD008359. [Google Scholar] [CrossRef]
- Varadhan, K.K.; Neal, K.R.; Lobo, D.N. Safety and efficacy of antibiotics compared with appendicectomy for treatment of uncomplicated acute appendicitis: Meta-analysis of randomised controlled trials. BMJ 2012, 344, e2156. [Google Scholar] [CrossRef] [PubMed]
- Murphy, T.; Hunt, R.H.; Fried, M.; Krabshuis, J.H. World Gastroenterology Organization Practice Guidelines: Diverticular Disease; World Gastroenterology Organisation: Milwaukee, WI, USA, 2007. [Google Scholar]
- Sifri, C.D.; Madoff, L.C. Diverticulitis and Typhlitis. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 7th ed.; Churchill Livingstone Elsevier: Philadelphia, PA, USA, 2009; Volume 76, pp. 1063–1065. [Google Scholar]
- Wieghard, N.; Geltzeiler, C.B.; Tsikitis, V.L. Trends in surgical management of diverticulitis. Ann. Gastroenterol. 2015, 28, 25–30. [Google Scholar] [PubMed]
- Tursi, A.; Papagrigoriadis, S. Review article: The current and evolving treatment of colonic diverticular disease. Ailment Pharmcol. Ther. 2009, 30, 532–546. [Google Scholar] [CrossRef] [PubMed]
- Brook, I.; Frazier, E.H. Aerobic and anaerobic microbiology in intra-abdominal infections associated with diverticular disease. J. Med. Microbiol. 2000, 49, 827–830. [Google Scholar] [CrossRef] [PubMed]
- Bauer, V.P. Emergency management of diverticulitis. Clin. Colon Rectal Surg. 2009, 22, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Klarenbeek, B.R.; de Korte, N.; van der Peet, D.L.; Cuesta, M.A. Review of current classifications for diverticular disease and a translation into clinical practice. Int. J. Colorectal Dis. 2012, 27, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Wasvary, H.; Turfah, F.; Kadro, O.; Beauregard, W. Same hospitalization resection for acute diverticulitis. Am. Surg. 1999, 65, 632–635. [Google Scholar] [PubMed]
- Bontemps, E.; Pardoll, P.M. Diverticulosis and Diverticulitis; Center for Digestive Diseases: South Pasadena, FL, USA, 2004; Updated September 2008; Available online: http://patients.gi.org/topics/diverticulosis-and-diverticulitis/ (accessed on 11 September 2015).
- Shabanzadeh, D.M.; Wille-Jorgenson, P. Antibiotics for uncomplicated diverticulitis. Cochrane Database Syst. Rev. 2012, 11. [Google Scholar] [CrossRef]
- Chabok, A.; Pahlman, L.; Hjern, F.; Haapaniemi, S.; Smedh, K.; AVOD Study Group. Randomized clinical trial of antibiotics in acute uncomplicated diverticulitis. Br. J. Surg. 2012, 99, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Ribas, Y.; Bombardo, J.; Aguilar, F.; Jovell, E.; Alcantara-Moral, M.; Campillo, F.; Lleonart, X.; Serra-Aracil, X. Prospective randomized clinical trial assessing the efficacy of a short course of intravenously administered amoxicillin plus clavulanic acid followed by oral antibiotic in patients with uncomplicated acute diverticulitis. Int. J. Colorectal Dis. 2010, 25, 1363–1370. [Google Scholar] [CrossRef] [PubMed]
- Kellum, J.M.; Sugerman, H.J.; Coppa, G.F.; Way, L.R.; Fine, R.; Herz, B.; Speck, E.L.; Jackson, D.; Duma, R.J. Randomized, prospective comparison of cefoxitin and gentamicin-clindamycin in the treatment of acute colonic diverticulitis. Clin. Ther. 1992, 14, 376–384. [Google Scholar] [PubMed]
- de Korte, N.; Kuyvenhoven, J.P.; van der Peet, D.L.; Felt-Bersma, R.J.; Cuesta, M.A.; Stockmann, H.B. Mild colonic diverticulitis can be treated without antibiotics. A case control study. Colorectal Dis. 2012, 14, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Tazuma, S. Gallstone disease: Epidemiology, pathogenesis, and classification of biliary stones (common bile duct and intrahepatic). Best Pract. Res. Clin. Gastroenterol. 2006, 20, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Sifri, C.D.; Madoff, L.C. Infections of the liver and biliary system. In Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases, 7th ed.; Churchill Livingstone Elsevier: Philadelphia, PA, USA, 2009; Volume 72, pp. 1035–1044. [Google Scholar]
- Kimura, Y.; Takada, T.; Strasberg, S.M.; Pitt, H.A.; Gouma, D.J.; Garden, O.J.; Büchler, M.W.; Windsor, J.A.; Mayumi, T.; Yoshida, M.; et al. TG13 current terminology, etiology, and epidemiology of acute cholangitis and cholecystitis. J. Hepatobiliary Pancreat. Sci. 2013, 20, 8–23. [Google Scholar] [CrossRef] [PubMed]
- Trendle, M.C.; Moertel, C.G.; Kvols, L.K. Incidence and morbidity of cholelithiasis in patients receiving chronic octreotide for metastatic carcinoid and malignant islet cell tumors. Cancer 1997, 79, 830–834. [Google Scholar] [CrossRef]
- Huffman, J.L.; Schenker, S. Acute acalculous cholecystitis: A review. Clin. Gastroenterol. Hepatol. 2010, 8, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Bagley, S.J.; Sehgal, A.R.; Gill, S.; Frey, N.V.; Hexner, E.O.; Loren, A.W.; Mangan, J.K.; Porter, D.L.; Stadtmauer, E.A.; Reshef, R.; et al. Acute cholecystitis is a common complication after allogeneic stem cell transplantation and is associated with the use of total parenteral nutrition. Biol. Blood Marrow Transplant. 2015, 21, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Darkahi, B.; Sandblom, G.; Liljeholm, H.; Videhult, P.; Melhus, Å.; Rasmussen, I.C. Biliary microflora in patients undergoing cholecystectomy. Surg. Infect. 2014, 15, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Galili, O.; Eldar, S., Jr.; Matter, I.; Madi, H.; Brodsky, A.; Galis, I.; Eldar, S., Sr. The effect of bactibilia on the course and outcome of laparoscopic cholecystectomy. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Carrascosa, M.F.; Salcines-Caviedes, J.R. Emphysematous cholecystitis. CMAJ 2012, 184, E81. [Google Scholar] [CrossRef] [PubMed]
- Coccolini, F.; Sartelli, M.; Catena, F.; Montori, G.; Di Saverio, S.; Sugrue, M.; Ceresoli, M.; Manfredi, R.; Ansaloni, L.; CIAO and CIAOW Study Groups. Antibiotic resistance pattern and clinical outcomes in acute cholecystitis: 567 consecutive worldwide patients in a prospective cohort study. Int. J. Surg. 2015, 21, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Yokoe, M.; Takada, T.; Strasberg, S.M.; Solomkin, J.S.; Mayumi, T.; Gomi, H.; Pitt, H.A.; Garden, O.J.; Kiriyama, S.; Hata, J.; et al. TG13 diagnostic criteria and severity grading of acute cholecystitis (with videos). J. Hepatobiliary Pancreat. Sci. 2013, 20, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Miura, F.; Takada, T.; Strasberg, S.M.; Solomkin, J.S.; Pitt, H.A.; Gouma, D.J.; Garden, O.J.; Büchler, M.W.; Yoshida, M.; Mayumi, T.; et al. TG13 flowchart for the management of acute cholangitis and cholecystitis. J. Hepatobiliary Pancreat. Sci. 2013, 20, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Bang, C.S.; Yoon, J.H.; Kim, Y.J.; Kim, J.B.; Baik, G.H.; Suk, K.T.; Kim, Y.S.; Kim, D.J. Clinical impact of body mass index on bactibilia and bacteremia. BMC Gastroenterol. 2014, 14, 104. [Google Scholar] [CrossRef] [PubMed]
- Gutt, C.N.; Encke, J.; Koninger, J.; Harnoss, J.C.; Weigand, K.; Kipfmüller, K.; Schunter, O.; Götze, T.; Golling, M.T.; Menges, M.; et al. Acute cholecystitis: Early versus delayed cholecystectomy, a multicenter randomized trial (ACDC study, NCT00447304). Ann. Surg. 2013, 258, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Minutolo, V.; Licciardello, A.; Arena, M.; Nicosia, A.; Di Stefano, B.; Calì, G.; Arena, G. Laparoscopic cholecystectomy in the treatment of acute cholecystitis: Comparison of outcomes and costs between early and delayed cholecystectomy. Eur. Rev. Med. Pharmacol. Sci. 2014, 18 (2 Suppl.), 40–46. [Google Scholar] [PubMed]
- FDA Drug Safety Communication. FDA Warns of Increased Risk of Death with IV Antibacterial Tygacil (Tigecycline) and Approves New Boxed Warning. Published 27 September 2013. Available online: http://www.fda.gov/Drugs/DrugSafety/ucm369580.htm (accessed on 25 September 2015).
- Herzog, T.; Chromik, A.M.; Uhl, W. Treatment of complicated intra-abdominal infections in the era of multi-drug resistant bacteria. Eur. J. Med. Res. 2010, 15, 525–532. [Google Scholar] [CrossRef] [PubMed]
- American College of Surgeons. Appendectomy, Surgical Removal of the Appendix Brochure. Reviewed 2014. Available online: https://www.facs.org/~/media/files/education/patient%20ed/app.ashx (accessed on 25 September 2015).
- Leone, S.; Stefani, S.; Venditti, M.; Grossi, P.; Colizza, S.; De Gasperi, A.; Scaglione, F.; Sganga, G.; Esposito, S.; Italian Intra-abdominal Infections Working Group. Intra-abdominal infections: Model of antibiotic stewardship in an era with limited antimicrobial options. Int. J. Antimicrob. Agents 2011, 38, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Dubrovskaya, Y.; Papadopoulos, J.; Scipione, M.R.; Altshuler, J.; Phillips, M.; Mehta, S.A. Antibiotic stewardship for intra-abdominal infections: Early impact on antimicrobial use and patient outcome. Infect. Control. Hosp. Epidemiol. 2012, 33, 427–428. [Google Scholar] [CrossRef] [PubMed]
- Popovski, Z.; Mercuri, M.; Main, C.; Sne, N.; Walsh, K.; Sung, M.; Rice, T.; Mertz, D. Multifaceted intervention to optimize antibiotic use for intra-abdominal infections. J. Antimicrob. Chemother. 2015, 70, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; Fish, D.N.; Napolitano, L.M.; Sawyer, R.G.; Slain, D.; et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am. J. Health Syst. Pharm. 2013, 70, 195–283. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Prasad, P.; Lin, M.; Garritson, S.; Nichols, A.; Liu, C. Ertapenem prophylaxis associated with an increased risk of Clostridium difficile infection among surgical patients. Infect. Control. Hosp. Epidemiol. 2015, 36, 1351–1354. [Google Scholar] [CrossRef] [PubMed]
- Stollman, N.; Smalley, W.; Hirano, I.; AGA Institute Clinical Guidelines Committee. American Gastroenterological Association Institute guideline for the management of acute diverticulitis. Gastroenterology 2015, 149, 1944–1949. [Google Scholar] [CrossRef] [PubMed]
- Blot, S.; De Waele, J.J.; Vogelaers, D. Essentials for selecting antimicrobial therapy for intra-abdominal infections. Drugs 2012, 72, e17–e32. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sanjuán, J.C.; Casella, G.; Antolín, F.; Castillo, F.; Fernández-Santiago, R.; Riaño, M.; Herrera, L.A.; Gómez-Fleitas, M. How long is antibiotic therapy necessary after urgent cholecystectomy for acute cholecystitis? J. Gastrointest. Surg. 2013, 17, 1947–1952. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, R.G.; Claridge, J.A.; Nathens, A.B.; Rotstein, O.D.; Duane, T.M.; Evans, H.L.; Cook, C.H.; O'Neill, P.J.; Mazuski, J.E.; Askari, R.; et al. Trial of short-course antimicrobial therapy for intraabdominal infection. N. Engl. J. Med. 2015, 372, 1996–2005. [Google Scholar] [CrossRef] [PubMed]
- Catena, F.; Vallicelli, C.; Ansaloni, L.; Sartelli, M.; Di Saverio, S.; Schiavina, R.; Pasqualini, E.; Amaduzzi, A.; Coccolini, F.; Cucchi, M.; et al. T.E.A. study: Three-day ertapenem versus three-day ampicillin-sulbactam. BMC Gastroenterol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoffmann, C.; Zak, M.; Avery, L.; Brown, J. Treatment Modalities and Antimicrobial Stewardship Initiatives in the Management of Intra-Abdominal Infections. Antibiotics 2016, 5, 11. https://doi.org/10.3390/antibiotics5010011
Hoffmann C, Zak M, Avery L, Brown J. Treatment Modalities and Antimicrobial Stewardship Initiatives in the Management of Intra-Abdominal Infections. Antibiotics. 2016; 5(1):11. https://doi.org/10.3390/antibiotics5010011
Chicago/Turabian StyleHoffmann, Charles, Matthew Zak, Lisa Avery, and Jack Brown. 2016. "Treatment Modalities and Antimicrobial Stewardship Initiatives in the Management of Intra-Abdominal Infections" Antibiotics 5, no. 1: 11. https://doi.org/10.3390/antibiotics5010011
APA StyleHoffmann, C., Zak, M., Avery, L., & Brown, J. (2016). Treatment Modalities and Antimicrobial Stewardship Initiatives in the Management of Intra-Abdominal Infections. Antibiotics, 5(1), 11. https://doi.org/10.3390/antibiotics5010011





