Abstract
Staphylococcus spp. have been associated with cases of healthcare associated infections due to their high incidence in isolates from the hospital environment and their ability to cause infections in immunocompromised patients; synthesize biofilms on medical instruments, in the case of negative coagulase species; and change in genetic material, thus making it possible to disseminate genes that code for the acquisition of resistance mechanisms against the action of antibiotics. This study evaluated the presence of blaZ, femA, and mecA chromosomal and plasmid genes of Staphylococcus spp. using the qPCR technique. The results were associated with the phenotypic expression of resistance to oxacillin and penicillin G. We found that the chromosomal femA gene was present in a greater proportion in S. intermedius when compared with the other species analyzed, while the plasmid-borne mecA gene was prevalent in the S. aureus samples. The binary logistic regression performed to verify the association among the expression of the genes analyzed and the acquisition of resistance to oxacillin and penicillin G were not significant in any of the analyses, p > 0.05.
1. Introduction
Healthcare-associated infections (HAIs) have been one of the main causes of morbidity and mortality not only in Brazil but worldwide, with a global estimate of 51.4% of morbidity and mortality in intensive care units (ICUs) [1]; about 3% to 15% of hospitalized patients in Brazil acquire nosocomial infections [2]. Such infections are transmitted during the provision of health care, and may occur during hospitalization or after discharge, when related to the procedures performed [3]. HAIs are related to biosafety failures such as the incorrect use of PPE (personal protection equipment) and EPCs (collective protection equipment) [4]. HAI cases are also associated with the patient’s long hospital stay, immunosuppression status, severity of the initial disease, inappropriate use of antibiotics [5] and surgical procedures [6].
For HAI control, much has been said about the importance of proper hand hygiene by health professionals—especially within ICUs, the most conducive place for the spread of pathogens. Correct hand hygiene by health professionals is essential to minimize cases of cross-transmission between patients. This transmission occurs when there are pathogens colonizing the skin of patients or hospital objects and the health professional is contaminated through direct contact with these surfaces. Thus, if there is no hand hygiene at the appropriate times and in the advisable way, this professional will disseminate the pathogens carried on their person when offering assistance to other patients [7].
The main etiological agents of HAIs comprise certain bacterial species of the genus Staphylococcus [8]. Staphylococcus species are a genus of Gram-positive bacteria characterized by the presence of the enzyme catalase, considered a virulence factor, since its function consists of the degradation of H2O2 into O2 + H2O and prevention of neutrophils from causing bacterial cell death [9].
Staphylococci are divided into two groups: producers and non-producers of the enzyme coagulase. In the coagulase-positive group, the species S. aureus stands out, which makes up the residual microbiota of the nasal passages of about 20% of the population, in addition to the individuals who occasionally carry it. This species is of great importance due to its high capacity to cause infections and adapt to different environmental conditions [10].
S. aureus is one of the main causes of healthcare-associated infections (HAIs) and, currently, of infections acquired in the community, resulting in serious consequences. This pathogen is involved in infections of the bloodstream, skin, soft tissues, and respiratory tract and can trigger diseases from simple to serious in nature such as pimples, boils, cellulitis, pneumonia, meningitis, endocarditis, and sepsis, among others [8]. Furthermore, strains of S. aureus are also related to methicillin resistance [11].
Coagulase-negative staphylococci are present on the surface of the skin and constitute about 90% of the body’s normal microbiota. They become pathogenic when the skin is broken or with the use of medical devices such as catheters. Infection through medical devices occurs due to the bacterial capacity to create biofilms on these surfaces, these biofilms serve as protection, since they are not susceptible to the action of disinfectants [12].
The species S. epidermidis and S. haemolyticus are related to nosocomial infections and are the most prevalent coagulase-negative species in individuals [13,14], being responsible for most nosocomial infections such as bacteremia and infective endocarditis [15], with the ability to synthesize biofilms on medical devices such as prosthetic devices and intravenous catheters [16]. Another important factor is that staphylococci are considered a reservoir of antimicrobial resistance genes due to their ability to exchange genetic material with other species [17].
Staphylococci have rapidly evolved to show resistance to beta-lactams: penicillins, cephalosporins, carbapenems, and monobactams [13]. The genes that code for resistance mechanisms can be located on plasmids and/or on the chromosome depending on the way that such a gene was obtained. Staphylococci have two resistance mechanisms that stand out: the production of beta-lactamases and the production of PBP2a [18].
The production of beta-lactamases, extracellular enzymes, occurs through the expression of the blaZ gene, usually located in the genomic material; however, it can also be present on plasmids. Beta-lactamase inhibits the action of beta-lactams by cleaving the beta-lactam ring, which provides the mechanism of action for this class of antimicrobials. The blaZ gene is regulated by two other genes, the blaR1 antirepressor and the blaI repressor. After exposure to beta-lactams, blaR1, a transmembrane sensor-transducer, undergoes autocatalytic cleavage, promoting cleavage of the repressor gene, blaI, thus allowing transcription of blaZ [18,19].
The PBP2a or PBP2’ proteins produced from the expression of the mecA gene are fully functional for the bacterial cell but have a low affinity for beta-lactams [20]. Expression of the mecA gene is constitutive or induced by beta-lactam antibiotics. The mecA gene is inserted into the staphylococcal chromosome, through a mobile genetic element called staphylococcal chromosomal cassette mec (SCCmec). The SCCmec is composed of several essential genetic elements: the mec complex, composed of the IS431 pathogenicity island, the mecA genes and their regulators mecI and mecR1, and the ccr complex (chromosome recombinase cassette), characterized by the presence of genes that encode recombinases. In all types of SCCmec, the mecA gene sequence is highly conserved in strains of S. aureus and Staphylococcus spp. negative coagulase [18].
In addition to the product encoded by the expression of the mecA gene that can induce methicillin resistance in S. aureus, another gene may also be associated with the resistance level of these bacteria, the femA gene (essential factor for the expression of methicillin resistance). The femA gene produces a protein of a size of 48 kDa that can restore methicillin resistance in S. aureus [21].
The acquisition of resistance mechanisms by bacteria in the hospital environment has made the treatment of HAIs difficult, and has increased the morbidity and mortality rates of patients in a state of immunosuppression. Therefore, the objective of the present work is to investigate the blaZ, femA, and mecA resistance genes in bacterial samples isolated through material collected from the hands of ICU health professionals, since the hands are the main vector of cross-transmission of pathogens.
2. Results
In this study, 48 isolates of Staphylococcus spp. were obtained, and approximately 90% of them were classified as coagulase negative. The predominant species isolated from the material coming from the hands of health professionals in an intensive care unit (ICU) was S. haemolyticus (20 isolated), followed by S. intermedius (7 isolated), and the least common species were S. pasteuri and S. epidermidis (1 each isolated) (Figure 1).
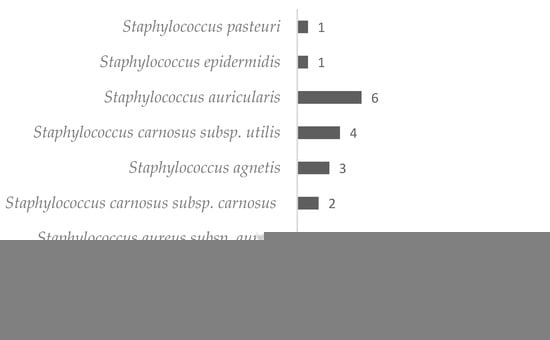
Figure 1.
Number and species of Staphylococci isolated from the hands of healthcare professionals.
Among the isolates tested for the blaZ gene of the chromosomal material, none of the samples, regardless of species, was positive. On the other hand, the femA gene was widespread among the isolates, being present in 100% of S. intermedius, S. epidermidis, S. carnosus subsp. utilis, and S. pasteuri tested; and positive in 50% of S. carnosus subsp. carnosus and S. auricularis and 30% of S. haemolyticus samples.
However, none of the S. agnetis and S. aureus subsp. aureus were positive for chromosomal femA. As for mecA, a single isolate of S. haemolyticus tested positive for the chromosomal gene (Table 1). The femA gene showed a higher proportion in S. intermedius when compared with S. haemolyticus, p = 0.003. When compared with all Staphylococcus species, the result showed a higher proportion in S. intermedius, p = 0.004; thus, the S. intermedius species is associated with the femA chromosomal gene.

Table 1.
Proportion of the presence of blaZ, femA, and mecA genes in the chromosomal and plasmid DNA of Staphylococcus spp. tested.
Regarding the analyzed plasmid DNA, the blaZ gene was present in 100% of S. agnetis and absent in S. aureus subsp. aureus, S. epidermidis, and S. pasteuri. The femA was present in S. auricularis (100%) and absent in S. agnetis, S. epidermidis, and S. pasteuri. The mecA gene was present in 100% of the S. agnetis tests and absent in S. haemolyticus, S. auricularis, S. epidermidis, and S. pasteuri (Table 1).
When considering the plasmid, the femA gene showed no difference in proportion of presence. When considering the comparison among S. haemolyticus and S. intermedius, p = 0.47; likewise, when comparing S. intermedius with all species, p = 0.41. However, the mecA gene showed a proportionally higher presence in S. aureus subsp. aureus when compared with other species, p = 0.04.
Results previously published by our group [22] showed that 39.4% and 42.4% of S. haemolyticus species were resistant to penicillin G and oxacillin, respectively. S. auricularis exhibited values of 15.2% for both tested antibiotics and S. intermedius of 12.1% for both antibiotics. Therefore, in this study, we associated species that were positive for the tested genes and that showed resistance to these antibiotics in the susceptibility test in the previous study in order to relate resistance to the acquisition of resistance mechanisms expressed by such genes, which act mainly in the synthesis of the bacterial cell wall.
Table 2 presents the data obtained. Of the S. haemolyticus isolates, two strains expressed from chromosomal femA, from chromosomal mecA (1), and from plasmid femA (2) and together with the expression of these genes also showed resistance to oxacillin (Table 2).

Table 2.
Isolates resistant to oxacillin and positive for the genes blaZ, femA, and mecA.
From the following isolates, only one chromosomal femA resistant gene for penicillin was found: S. intermedius (3) and S. pasteuri (1) (Table 3). Despite considering each sample and gene separately, some isolates were positive for more than one gene. In the Supplementary Material (Table S1), it is possible to observe the identification of each sample and its results for the presence of genes and positivity for isolated antibiotics—in addition to being able to visualize the samples that were not tested for some of the genes, due to the lack of growth at the time of their reactivation.

Table 3.
Isolates resistant to penicillin G and positive for the genes blaZ, femA, and mecA.
The binary logistic regression performed to verify the association between the presence of the investigated genes and the expression of resistance to the antibiotics penicillin G and oxacillin was not significant in any of the analyses p > 0.05 (Table S2).
Therefore, X2 statistics were used for the association between two variables, verifying whether resistance to penicillin G or oxacillin in the genus Staphylococcus could be associated with the presence of one of the researched genes. There was no association between the presence of blaZ femA, or mecA genes (plasmid or chromosomal) and resistance to the antimicrobials penicillin G or oxacillin, p > 0.05 for all analyses. For the blaZ and mecA (chromosomal) genes, it was not possible to perform the association test due to low or no variables in the model. All results of the X2 association tests can be seen in Table S3.
3. Discussion
Among the staphylococci, the species S. aureus stands out in the genus due to its strong relationship both with nosocomial infections and antimicrobial resistance. Even at the beginning of the era of antibiotics, it was possible to identify the first strain resistant to penicillin, with the action of the blaZ gene that codes for β-lactamases; these enzymes cleave the β-lactam ring, preventing the action of the antibiotic. Initially, this was a problem found only in the hospital environment, but it is already possible to find strains of multidrug-resistant S. aureus in infections in the community.
Currently, both coagulase-positive and negative community staphylococci have a high rate of resistance to penicillin G [23]. In the present study, we obtained four S. aureus isolates. Of these samples, two were positive for the femA plasmid and showed resistance to penicillin G in the susceptibility test. One of the samples was positive for both the femA and mecA plasmid genes but remained sensitive to oxacillin. This finding may be related to the presence of silent genes that are carried in the bacterial genetic material but are not expressed and, therefore, there is no phenotypic resistance. This inactivation and activation of the gene can occur due to different conditions such as the culture medium [24]. Another factor that can interfere with phenotypic resistance is polygenic resistance [25].
S. haemolyticus and S. epidermidis have been the most frequently isolated staphylococci in hospital environments and are mainly associated with medical devices [26]. In one study [27], there was the identification of 11 isolates of coagulase-negative staphylococci, being 45% S. epidermidis and 27% S. haemolyticus. Of 200 strains of coagulase-negative staphylococci obtained from patients with nosocomial bacteremia in Turkey, 87 corresponded to S. epidermidis and 23 to S. haemolyticus, both of which are more prevalent among 12 other species also isolated [28].
Other studies also reported similar data [29], where 41% of isolates from ICU patients in Sri Lanka were identified as S. haemolyticus, with the total number of coagulase-negative staphylococcus (CNS) equal to 82 samples. Researchers [30] in Nepal obtained 123 CNS isolates from hospital materials in the ICU and wards; in this case, the most common species was S. epidermidis (42%), and S. haemolyticus represented 9% of isolates. Another study [31] collected nasal and hand samples from 125 health professionals from a hospital in Nepal. Of the 250 samples, 203 were identified as SCN and 38 as S. aureus.
According to the data, in our study, 41.6% of the isolates were S. haemolyticus, and in none of these was the blaZ chromosomal gene found. However, one sample was positive for chromosomal mecA and the femA plasmid and showed resistance to oxacillin, while samples positive for femA (plasmid or chromosomal) were resistant to penicillin G. In further consideration of the S. haemolyticus species, one of the isolates was positive for blaZ and plasmid femA genes; however, it did not show positive test for resistance to the two analyzed antibiotics.
S. intermedius took second place with approximately 14% of the samples. In one of the isolates, femA was found in chromosomal and plasmidial DNA, but the isolate did not show resistance to oxacillin or penicillin G. In contrast, a negative sample for the chromosomal genes blaZ and femA and for the plasmidial mecA was resistant to penicillin G. However, the chromosomal genes femA and mecA were not investigated and, therefore, the acquisition of resistance may be related to the presence of these genes, since samples that were positive for chromosomal femA alone or together with plasmid femA showed resistance to penicillin G.
The species S. agnetis represented 6% of the total samples isolated in this study. Despite not being significant in the area of human health, these staphylococci have been reported due to their veterinary importance as a potential etiological agent of infections in birds such as broilers and cattle, both of which have great commercial value [32,33]. As shown in the results section, S. agnetis tested positive for the plasmidial blaZ and mecA genes and showed resistance to both oxacillin and penicillin G in the susceptibility test. Suggesting that this species may share resistance genes with other species in the hospital environment, as well as hinder the treatment of infected birds and cattle.
S. carnosus was only classified as belonging to the genus of staphylococci in 1982 [34]; however, it has been used since 1950 to ferment sausages and is a non-pathogenic microorganism. It is considered as a contributor for food quality, providing flavor and fermentation control [35]. In the present study, when we consider all samples of S. carnosus, we have the presence of all the genes of the plasmidial material studied, while in the chromosomal DNA, only femA was present. However, none of the positive samples for such genes showed resistance to the tested antibiotics. This condition suggests the possibility that these strains, even without the potential to cause human infections, transfer such resistance genes to other species of greater clinical importance, increasing the difficulty in treating patients.
Even today, there are few reports on infections caused by S. pasteuri, but it is already known that it infects the gastrointestinal microbiota of children with active celiac disease and that it is a contaminant of platelet transfusions, with infective endocarditis [36]. The isolate from this study showed expression of the chromosomal femA gene and resistance to penicillin G but remained sensitive to oxacillin.
S. auricularis was discovered in 1983. It colonizes the external auditory canal, it has been the cause of rare community and nosocomial infections, and its relationship with prosthetic valve endocarditis was recently described [37]. Furthermore, this species was isolated from 10.7% of a total of 5447 low-birth-weight infants who participated in a survey that associated S. auricularis infection as a cause of early onset sepsis. According to an antibiogram test, only 17% of the isolated strains were susceptible to penicillin [38]. Here, we analyzed 6 isolates of S. auricularis, one sample was positive for blaZ and femA (plasmidial) and showed resistance to the two antimicrobials analyzed. Other samples were also resistant to oxacillin and penicillin G but it was not possible to investigate the presence of resistance genes.
In turn, S. epidermidis has been considered an opportunistic pathogen of medical relevance due to its ability to produce biofilms and act as a reservoir of resistance genes, rendering it the main cause of nosocomial infections [39], along with S. aureus causing infections in both orthopedic and breast catheters and implants [40]. As a result of its high rate of antimicrobial resistance, infections are difficult to treat. In this study, S. epidermidis corresponded to 2% of the isolates from the hands of ICU professionals, and this sample was positive for chromosomal femA and showed resistance to penicillin G and oxacillin. However, for this sample, only the chromosomal and plasmid femA gene was investigated, the latter being negative in the analyses.
One study [41] isolated and identified 12 strains of S. epidermidis from 150 smear samples and tracked the blaZ and mecA genes in 91.7% of the studied samples; in addition to these genes, this same author investigated the presence of other genes that appeared with lesser incidence and 75% of the samples were resistant to penicillin and 66.7% to oxacillin.
Widely described in the literature, the mecA gene was present in only one chromosomal DNA sample from S. haemolyticus in the present study. Researchers [42] have described that of 129 isolates from the ICU and from health professionals, 86% were identified as S. aureus and 20% were positive for MRSA, highlighting the influence of health professionals in the cross-transmission of infections. In another study, detection of mecA was reported in 70% (n = 87) of blood, endotracheal tube, and central venous catheter isolates [30], reaching a total of 41% (n = 34) in S. haemolyticus isolated from patients with catheter-related bloodstream infection and colonized central venous catheter [29].
In another study [43], the mecA gene was isolated in 95% of samples from different surfaces of an ICU bed, and all isolates grew in MRSA medium, even though none of them were of the species S. aureus; based on sensitivity tests, it was found that 100% of the samples were resistant to oxacillin.
In one work [44], 15 CNS isolates were identified, belonging to S. epidermidis, S. haemolyticus, S. warningeri, S. hominis, and S. capitis. The researchers found that 13 strains were resistant to oxacillin and β-lactams. Molecular tests of all samples were positive for the mecA gene. Another study [45] reported that of 89 SCN isolates, 63 had the mecA gene and all isolates were resistant to penicillin and other antibiotics in different proportions.
The association among the isolation of blaZ, femA, and mecA genes with the acquisition of mechanisms of resistance, mainly against β-lactams, is already known and widely reported. In the present study, statistical evaluation was carried out to relate these variables, the results obtained did not attest to such associations in any of the analyses performed (p > 0.05). It is suggested that the small number of samples and events in each of the analyzed variables has negatively interfered in the statistical model.
In our study, some events were null and most of them were less than ten (Table S1). Furthermore, when performing the X2 statistic for association among these variables, verifying whether resistance to penicillin G or oxacillin of the Staphylococcus spp. may be associated with one of the genes investigated, no association was found among the presence of the genes blaZ, femA, mecA, and blaZ (chromosomal and plasmid) and resistance to the antimicrobials penicillin G and oxacillin, p > 0.05 for all analyses.
The CDC (Centers for Disease Control) reports that with the use of preventive measures such as the correct hygiene of hands and the hospital environment, it would be possible to avoid 20 to 30% of HAIs. However, adherence to HAI prevention measures by health professionals is still very low. The incidence of HAI cases associated with increased resistance to antimicrobials by infecting pathogens has further aggravated the situation, thus becoming a global public health problem [46].
The acquisition of resistance mechanisms by bacteria has been aggravated, in part, by the indiscriminate use of antimicrobials; approximately 90% of patients who acquire such drugs use them in a period equal to or less than 3 days. About 50% of antimicrobials are prescribed inappropriately, and in some countries the purchase of these drugs without a prescription reaches around 2/3. In addition to the increase in bacterial resistance achieved by the incorrect use of antimicrobials, this error can lead to other problems such as an increase in more serious diseases, an increased risk of complications, an increase in mortality, and an increase in health costs [47].
Another factor is the difficulty in developing new antibiotics. The production process of a drug is very long, about 10 years. Furthermore, this includes a high financial investment in research and there is no guarantee of developing a molecule that is effective for the treatment of infections and viable for commercialization. Another reason that has discouraged investors is the possibility that a new molecule launched on the market may become infeasible for use in the first few years due to the rapid acquisition of resistance by bacteria. Due to these limitations, the pharmaceutical industry has invested less and less in this field, prioritizing the production of drugs for the treatment of chronic diseases. Therefore, it is necessary to maintain supervision, sales control, prescription, and correct use; otherwise, cases of HAI associated with resistant and multidrug-resistant species will become increasingly recurrent and severe [48].
Based on published and obtained data, the hands of health professionals can be vehicles for cross-transmission of pathogens between immunosuppressed patients, increasing cases of HAI. These pathogens, in turn, can carry genotypic resistance, resulting or not resulting in phenotypic resistance to antibiotics commonly used to treat infections, thus bringing more complications to public health.
4. Materials and Methods
The isolates used in this study were collected, identified, and tested for antibiotic susceptibility in the study conducted by Rodrigues and collaborators [22].
The sample collection took place in a public hospital of high complexity specialized in traumatology, in urgent and emergency care in the city of Goiânia, capital of the State of Goiás. This hospital has multi and interdisciplinary teams, with 470 beds for the Unified Health System (SUS), among which, 57 beds are in the ICU, where the samples were collected (05/19 to 09/19), for bacteriological identification in the laboratory.
Volunteers for data collection were nursing professionals such as technicians, nurses, and physiotherapists; over 18 years old; and who agreed to participate in the research. Those nursing professionals who did not agree to participate in the research were excluded from the research. Samples were collected from eight health professionals, and from each professional, samples were collected from the left and right hands, isolating 48 bacteria.
After submission to the Teaching and Research Ethics Committee of the Hospital de Urgências de Goiânia and approval of the study, in accordance with current legislation in Brazil, the Free and Informed Consent Form (TCLE) was applied to health professionals who volunteered to participate in this study. The collection was standardized by approaching the employee at random and inviting them to the survey. The research was approved on 10 May 2019 with CAAE: 08689018.6.0000.0033.
4.1. Data Collection
The hands of the professionals who participated in the research were placed separately in a sterile plastic bag containing distilled water and the fingertips were rubbed together for five minutes. At the end of the collections, the samples were placed in an isothermal box and transported to the Microorganism Biotechnology Laboratory (LBMIC/IPTSP) for microbiological analysis.
4.2. Bacterial Identification
A volume of 1 mL was pipetted for each sample, the material was inoculated in BHI broth (brain heart infusion), and then these media were incubated at 37 °C for 24 h. The samples that showed turbidity were seeded by exhaustion with three different types of culture media in Petri dishes: blood agar (AS) for the identification of Acinetobacter spp., MacConkey agar (AMC) for the identification of Escherichia coli and other enterobacteria, and agar salted mannitol (AMS) for the identification of Staphylococcus spp., before incubation at 37 °C for 24 h. After the growth of the colonies, isolation and observation of the macro and microscopic aspects of the colonies followed. Another step used was the Gram stain.
As an additional exam to the Gram-staining result, the KOH (potassium hydroxide) test was performed. For identification, catalase and oxidase tests were performed. After confirming the growth and morphology by the Gram technique, those microorganisms with positive catalase tests were seeded again and isolated in salty mannitol agar for S. aureus research. For the isolates that presented negative catalase tests, we proceeded to seeding in blood agar, with the purpose of verifying Streptococcus spp., Micrococcus spp., and negative Staphylococcus coagulase.
After the analyses, all samples were kept in a freezer at −80 °C in tubes containing casein-soy broth and 10% glycerol until the moment of reactivation for the molecular analysis of the present study. Thus, in order to complement the results previously published by our group [22], we evaluated the molecular expression of the blaZ, femA, and mecA genes that are associated with the acquisition of resistance mechanisms against the action of beta-lactams.
4.3. DNA Extraction and Quantification
For molecular evaluation, the samples were seeded in BHI medium and incubated at 37 °C for 24 h. At this stage, some samples did not show growth. For the viable samples, the extraction of plasmid and chromosomal DNA was performed separately, following the instructions for the Pharmacia® FLEXIPREP (Stockholm, Sweden) and Qiagen® DNA minikit extraction kit (Venlo, Netherlands), respectively.
After carrying out the DNA extraction protocol, the genetic material was quantified using the Thermo Scientific NanoDrop® 2000 Spectrophotometer (Waltham, MA, USA). The quantification was performed individually and sterile milliQ water was used as a blank for proper calibration between the quantifications of each sample.
4.4. qPCR
The identified samples were subjected to the characterization of resistance genes through real-time PCR assays with the Sybr Green Real Time PCR kit (Sybr Green qPCR master mix LOW ROX—100 reactions × 25 µL), adding DNA and specific primers for amplification of each gene. For the internal control, a C protein reactive specific primer was used; for the positive control of the reaction, primers were used to amplify the 16S RNA; and for the negative control, water was added instead of DNA.
The conditions of the real time PCR technique were standardized following the manufacturer’s instructions. A cycling protocol was used under the following conditions: initial denaturation at 95 °C for 2 min; maintenance of denaturation at 95 °C for 15 s; annealing and extension of the oligonucleotides at 60 °C for 60 s; melting curve at 65 °C for 30 s.
For the amplification of Staphylococcus spp., the following primers were used: mecA (310 bp), (forward) 5′ GTA GAA ATG ACT GAA CGT CCG ATAA3′ and (reverse) 5′ CCA ATT CCA CAT TGT TTC GGT CTA A 3′ [49]; blaZ (228 bp), (forward) 5′ AAG AGA TTT GCC TAT GCT TC 3′ and (reverse) 5′ GCT TGA CCA CTT TTA TCA GC 3′ (Araújo, 2019); femA (forward) 5′ AAAAAAGCACATAACAAGCG 3′ and (reverse) 5′ GATAAAGAAGAAACCAGCAG 3′ [50].
4.5. Statistical Analysis
For association of Staphylococcus spp. with resistance genes, a comparison among species was performed, considering the binomial test for two proportions. Statistical analyses were performed using the BioEstat 5.3 software, considering a significance limit of 5%.
OR was calculated by means of binary logistic regression, in order to estimate the probability of the outcome of resistance to the tested antibiotic or sensitivity of the sample analyzed. For regression, the predictors were blaZ, femA, and mecA (chromosomal and plasmid), and the outcome was resistance to penicillin G or oxacillin. Regression was performed for all grouped and individual species. Pearson’s chi-square (X2) test [51] for association was calculated for all species grouped, considering genes and resistance to antibiotics already described All confidence intervals were 95% (95% CI) and p < 0.05 was considered statistically significant. Statistical analysis was performed using Minitab® software version 19.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/antibiotics12040671/s1, Table S1: Staphylococcus spp. tested for chromosomal and plasmid blaZ, femA, and mecA genes and oxacillin and penicillin G resistance profile; Table S2: Logistic regression to verify the association between the presence of the gene and resistance to penicillin G and oxacillin; Table S3: X2 test for association between the presence of genes and resistance to penicillin G and oxacillin.
Author Contributions
Conceptualization: L.K.L.P., C.A.R. and L.C.; data curation: L.K.L.P. and L.C.; formal analysis: L.K.L.P., A.R.G.F. and L.C. funding acquisition: A.R.G.F., C.J.C., V.G., M.E., P.d.S. and L.C.; investigation: L.K.L.P., C.A.R. and L.C.; methodology: L.K.L.P., A.R.G.F., C.A.R. and L.C.; project administration: L.K.L.P., C.J.C., A.R.G.F., M.A.G.A. and L.C.; resources: L.K.L.P. and C.A.R.; supervision: C.J.C., A.R.G.F., J.D.G.V. and L.C.; validation: J.D.G.V. and L.C.; visualization: L.K.L.P. and L.C.; writing—original draft: L.K.L.P., J.D.G.V. and L.C.; writing—review and editing: C.J.C., J.D.G.V. and L.C. All authors have read and agreed to the published version of the manuscript.
Funding
FAPEG—6° PPSUS, 2020.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Braga, I.; Campos, P.; Gontijo-Filho, P.; Rjjohi, R. Multi-hospital point prevalence study of healthcare-associated infections in 28 adult intensive care units in Brazil. J. Hosp. Infect. 2018, 99, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Lorenzini, E.; Costa, T.C.D.; Silva, E.F.D. Prevenção e controle de infecção em unidade de terapia intensiva neonatal. Rev. Gaúcha Enferm. 2013, 34, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.C.; Paula, A.O.; Iquiapaza, R.A.; Lacerda, A.C.D.S. Infecções relacionadas à assistência em saúde e gravidade clínica em uma unidade de terapia intensiva. Rev. Gaúcha Enferm. 2012, 33, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, A.L.P.; Junior, A.C.S.S. Resistência bacteriana aos antibióticos e Saúde Pública: Uma breve revisão de literatura. Estação Científica 2017, 7, 45–57. [Google Scholar] [CrossRef]
- Amaral, L.d.S.; Godinho, S.M. Principais Fatores Causais de Infecções Relacionadas à Assistência à Saúde em Unidades de Terapia Intensiva: Uma Revisão Integrativa. 2020. Available online: https://dspace.uniceplac.edu.br/bitstream/123456789/289/1/Luisa%20Amaral_0003714_Silma%20Godinho_0003022.pdf (accessed on 19 February 2023).
- Andrew, E.F.; Friday, U.N.; Andrew, E.K.; Isaiah, L.N.; Silas, E.E.; Unah, U.V. Prevalence and antimicrobial susceptibility profile of bacterial isolates from infected caesarean sites in three federal capital territory hospitals, abuja nigeria. Am. J. Biomed. Life Sci. 2018, 6, 90–95. [Google Scholar] [CrossRef]
- Pittet, D.; Allegranzi, B.; Sax, H.; Dharan, S.; Pessoa-Silva, C.L.; Donaldson, L.; Boyce, J.M. Evidence-based model for hand transmission during patient care and the role of improved practices. Lancet Infect. Dis. 2006, 6, 641–652. [Google Scholar] [CrossRef]
- Santos, A.L.D.; Santos, D.O.; Freitas CCd Ferreira, B.L.A.; Afonso, I.F.; Rodrigues, C.R.; Castro, H.C. Staphylococcus aureus: Visiting a strain of clinical importance. J. Bras. Patol. Med. Lab. 2007, 43, 413–423. [Google Scholar] [CrossRef]
- Mandell, G.L. Catalase, superoxide dismutase, and virulence of Staphylococcus aureus. In vitro and in vivo studies with emphasis on staphylococcal--leukocyte interaction. J. Clin. Investig. 1975, 55, 561–566. [Google Scholar] [CrossRef]
- Bes, T.M.; Martins, R.R.; Perdigão, L.; Mongelos, D.; Moreno, L.; Moreno, A.; De Oliveira, G.S.; Costa, S.F.; Levin, A. Prevalence of methicillin-resistant Staphylococcus aureus colonization in individuals from the community in the city of Sao Paulo, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2018, 60, e58. [Google Scholar] [CrossRef]
- Appelbaum, P.C.; Bozdogan, B. Vancomycin resistance in Staphylococcus aureus. Clin. Lab. Med. 2004, 24, 381–402. [Google Scholar] [CrossRef]
- Foster, T. Staphylococcus. In Medical Microbiology, 4th ed.; Baron, S., Ed.; Chapter 12; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996. Available online: https://pubmed.ncbi.nlm.nih.gov/21413338/ (accessed on 19 February 2023).
- Czekaj, T.; Ciszewski, M.; Szewczyk, E.M. Staphylococcus haemolyticus—An emerging threat in the twilight of the antibiotics age. Microbiology 2015, 161, 2061–2068. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, C.; Ziebuhr, W.; Becker, K. Are coagulase-negative staphylococci virulent? Clin. Microbiol. Infect. 2019, 25, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.Y.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef]
- Sizar, O.; Unakal, C.G. Gram Positive Bacteria; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Rossi, C.C.; Pereira, M.F.; Giambiagi-deMarval, M.J.G.; Biology, M. Underrated Staphylococcus species and their role in antimicrobial resistance spreading. Genet. Mol. Biol. 2020, 43, e20190065. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, E.C.; Marques, V.F.; Melo, D.A.; Alencar, T.A.; Coelho, I.d.S.; Coelho, S.M.; Souza, M. Caracterização fenogenotípica da resistência antimicrobiana em Staphylococcus spp. isolados de mastite bovina. Pesqui. Veterinária Bras. 2012, 32, 859–864. [Google Scholar] [CrossRef]
- El Feghaly, R.E.; Stamm, J.E.; Fritz, S.A.; Burnham, C.A.D. Presence of the blaZ beta-lactamase gene in isolates of Staphylococcus aureus that appear penicillin susceptible by conventional phenotypic methods. Diagn. Microbiol. Infect. Dis. 2012, 74, 388–393. [Google Scholar] [CrossRef]
- Gelatti, L.C.; Bonamigo, R.R.; Becker, A.P.; d’Azevedo, P.A. Staphylococcus aureus resistentes à meticilina: Disseminação emergente na comunidade. An. Bras. Dermatol. 2009, 84, 501–506. [Google Scholar] [CrossRef]
- Berger-Bächi, B.; Barberis-Maino, L.; Strässle, A.; Kayser, F.H. FemA, a host-mediated factor essential for methicillin resistance in Staphylococcus aureus: Molecular cloning and characterization. Mol. Gen. Genet. MGG 1989, 219, 263–269. [Google Scholar] [CrossRef]
- Andrade, C.R.; Filho, A.R.G.; Costa, A.C.M.; De Oliveira, T.A.; Carneiro, L.C.; Avelino, M.A.G. Identificação de Bactérias Causadoras de Infecção Hospitalar Utilizando Fenotipagem Clássica / Identification of Hospital Infectious Bacteria Using Classical Phenotyping. Braz. J. Dev. 2021, 7, 54446–54463. [Google Scholar] [CrossRef]
- Aguayo-Reyes, A.; Quezada-Aguiluz, M.; Mella, S.; Riedel, G.; Opazo-Capurro, A.; Bello-Toledo, H.; Domínguez, M.; González-Rocha, G. Bases moleculares de la resistencia a meticilina en Staphylococcus aureus. Rev. Chil. Infectol. 2018, 35, 7–14. [Google Scholar] [CrossRef]
- Deekshit, V.K.; Srikumar, S. ‘To be, or not to be’—The dilemma of ‘silent’ antimicrobial resistance genes in bacteria. J. Appl. Microbiol. 2022, 133, 2902–2914. [Google Scholar] [CrossRef] [PubMed]
- Laura, D.M.; Scott, N.L.; Vanner, E.A.; Miller, D.; Flynn, H.W., Jr. Genotypic and phenotypic antibiotic resistance in Staphylococcus epidermidis endophthalmitis. Ophthalmic Surg. Lasers Imaging Retin. 2020, 51, S13–S16. [Google Scholar] [CrossRef] [PubMed]
- Becker, K.; Heilmann, C.; Peters, G. Coagulase-negative staphylococci. Clin. Microbiol. Rev. 2014, 27, 870–926. [Google Scholar] [CrossRef] [PubMed]
- d’Azevedo, P.A.; Secchi, C.; Antunes, A.L.; Sales, T.; Silva, F.M.; Tranchesi, R.; Pignatari, A.C.C. Oxacilin-resistant Coagulase-negative staphylococci (CoNS) bacteremia in a general hospital at São Paulo city, Brasil. Braz. J. Microbiol. 2008, 39, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Koksal, F.; Yasar, H.; Samasti, M. Antibiotic resistance patterns of coagulase-negative staphylococcus strains isolated from blood cultures of septicemic patients in Turkey. Microbiol. Res. 2009, 164, 404–410. [Google Scholar] [CrossRef]
- Medis, S.; Dissanayake, T.; Kottahachchi, J.; Namali, D.; Gunasekara, S.; Wijesinghe, G.; Dilrukshi, N.; Weerasekera, M. Biofilm formation and antibiotic resistance among Coagulase Negative Staphylococcus species isolated from central venous catheters of intensive care unit patients. Indian J. Med. Microbiol. 2023, 42, 71–76. [Google Scholar] [CrossRef]
- Shrestha, L.B.; Bhattarai, N.R.; Rai, K.; Khanal, B. Antibiotic resistance and mecA gene characterization of coagulase-negative staphylococci isolated from clinical samples in Nepal. Infect. Drug Resist. 2020, 13, 3163–3169. [Google Scholar] [CrossRef]
- Shah, P.; Dhungel, B.; Bastola, A.; Banjara, M.R.; Rijal, K.R.; Ghimire, P. Methicillin resistant Staphylococcus aureus in health care workers of a tertiary care infectious disease hospital in Nepal. Tribhuvan Univ. J. Microbiol. 2020, 7, 19–30. [Google Scholar] [CrossRef]
- Shwani, A.; Adkins, P.R.F.; Ekesi, N.S.; Alrubaye, A.; Calcutt, M.J.; Middleton, J.R.; Rhoads, D.D. Whole-Genome Comparisons of Staphylococcus agnetis Isolates from Cattle and Chickens. Appl. Environ. Microbiol. 2020, 86, e00484-20. [Google Scholar] [CrossRef]
- Szafraniec, G.; Szeleszczuk, P.; Dolka, B. A Review of Current Knowledge on Staphylococcus agnetis in Poultry. Animals 2020, 10, 1421. [Google Scholar] [CrossRef]
- Schleifer, K.H.; Fischer, U. Description of a New Species of the Genus Staphylococcus: Staphylococcus carnosus. Int. J. Syst. Evol. Microbiol. 1982, 32, 153–156. [Google Scholar] [CrossRef]
- Löfblom, J.; Rosenstein, R.; Nguyen, M.-T.; Ståhl, S.; Götz, F. Staphylococcus carnosus: From starter culture to protein engineering platform. Appl. Microbiol. Biotechnol. 2017, 101, 8293–8307. [Google Scholar] [CrossRef]
- Ramnarain, J.; Yoon, J.; Runnegar, N. Staphylococcus pasteuri infective endocarditis: A case report. IDCases 2019, 18, e00656. [Google Scholar] [CrossRef]
- Ha, E.T.; Heitner, J.F. Staphylococcus Auricularis Endocarditis: A Rare Cause of Subacute Prosthetic Valve Endocarditis with Severe Aortic Stenosis. Cureus 2021, 13, e2738. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.J.; Brown, G.D.; A Lombardo, F. Early-onset sepsis with Staphylococcus auricularis in an extremely low-birth weight infant–an uncommon pathogen. J. Perinatol. 2007, 27, 519–520. [Google Scholar] [CrossRef] [PubMed]
- Vuong, C.; Otto, M. Staphylococcus epidermidis infections. Microbes Infect. 2002, 4, 481–489. [Google Scholar] [CrossRef]
- Oliveira, W.; Silva, P.; Silva, R.; Silva, G.; Machado, G.; Coelho, L.; Correia, M. Staphylococcus aureus and Staphylococcus epidermidis infections on implants. J. Hosp. Infect. 2018, 98, 111–117. [Google Scholar] [CrossRef]
- Al-Amara, S.S. Comparison between phenotype and molecular resistance characteristic in Staphylococcus epidermidis isolates from wound infections in Al-Basrah province, Iraq. Period. Eng. Nat. Sci. (PEN) 2021, 9, 897–903. [Google Scholar] [CrossRef]
- Al-Essa, L.; Abunaja, M.H.; Hamadneh, L.; Abu-Sini, M. Mapping the intensive care unit environment and health care workers for methicillinresistant Staphylococcus aureus with mecA gene confirmation and antibacterial resistance pattern identification in a district hospital in Amman. Kuwait Med. J. 2021, 53, 265–270. [Google Scholar]
- Fernandes, L.F.; Souza, G.A.D.; De Almeida, A.C.; Cardoso, L.; Xavier, M.A.D.S.; Pinheiro, T.P.P.; Da Cruz, G.H.S.; Dourado, H.F.S.; Silva, W.S.; Xavier, A.R.E.D.O. Identification and characterization of methicillin-resistant Staphylococcus spp. isolated from surfaces near patients in an intensive care unit of a hospital in southeastern Brazil. Rev. Soc. Bras. Med. Trop. 2020, 53, e20200244. [Google Scholar] [CrossRef]
- Al-Sultany, Z.K.; Al-Charrakh, A.H. Antibiotic resistance patterns of coagulase negative Staphylococcus (CoNS) strains isolated from blood stream infections in Babylon province, Iraq. Ann. Trop. Med. Public Health 2020, 23. [Google Scholar] [CrossRef]
- Asante, J.; Hetsa, B.A.; Amoako, D.G.; Abia, A.L.K.; Bester, L.A.; Essack, S.Y. Multidrug-resistant coagulase-negative staphylococci isolated from bloodstream in the uMgungundlovu District of KwaZulu-Natal Province in South Africa: Emerging pathogens. Antibiotics 2021, 10, 198. [Google Scholar] [CrossRef] [PubMed]
- Lozano, C.; Torres, C. Actualización en la resistencia antibiótica en Gram positivos. Inf. Artículo 2017, 35, 2–8. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Monteiro, R.F.; dos Santos, V.R.R.; Ferreira, A.A.C.T.; de Abreu, J.R.G. O uso indiscriminado de antimicrobianos para o desenvolvimento de micro-organismos resistentes. Rev. Eletrônica Acervo Saúde 2020, 53, e3597. [Google Scholar] [CrossRef]
- Oliveira, M.; Pereira, K.D.S.P.S.; Zamberlam, C.R. Resistência Bacteriana Pelo Uso Indiscriminado De Antibióticos: Uma Questão De Saúde Pública. Rev. Ibero-Am. Humanid. Ciênc. Educ. 2020, 6, 183–201. [Google Scholar] [CrossRef]
- Bannoehr, J.; Ben Zakour, N.L.; Waller, A.S.; Guardabassi, L.; Thoday, K.L.; van den Broek, A.H.; Fitzgerald, J.R. Population genetic structure of the Staphylococcus intermedius group: Insights into agr diversification and the emergence of methicillin-resistant strains. J. Bacteriol. 2007, 189, 8685–8692. [Google Scholar] [CrossRef]
- Teixeira, J.P.; Silva, N.; da Fonseca, L.M.; da Costa, G.M. Uso de PCR Duplex para detecção dos genes femA e mecA e determinação da concentração inibitória mínima (CIM) em Staphylococcus aureus isolados de leite cru. Rev. Inst. Adolfo Lutz 2014, 73, 272–279. [Google Scholar] [CrossRef]
- Pearson, K. X. On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1900, 50, 157–175. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).