Extended-Spectrum β-Lactamases (ESBL) Producing Bacteria in Animals
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
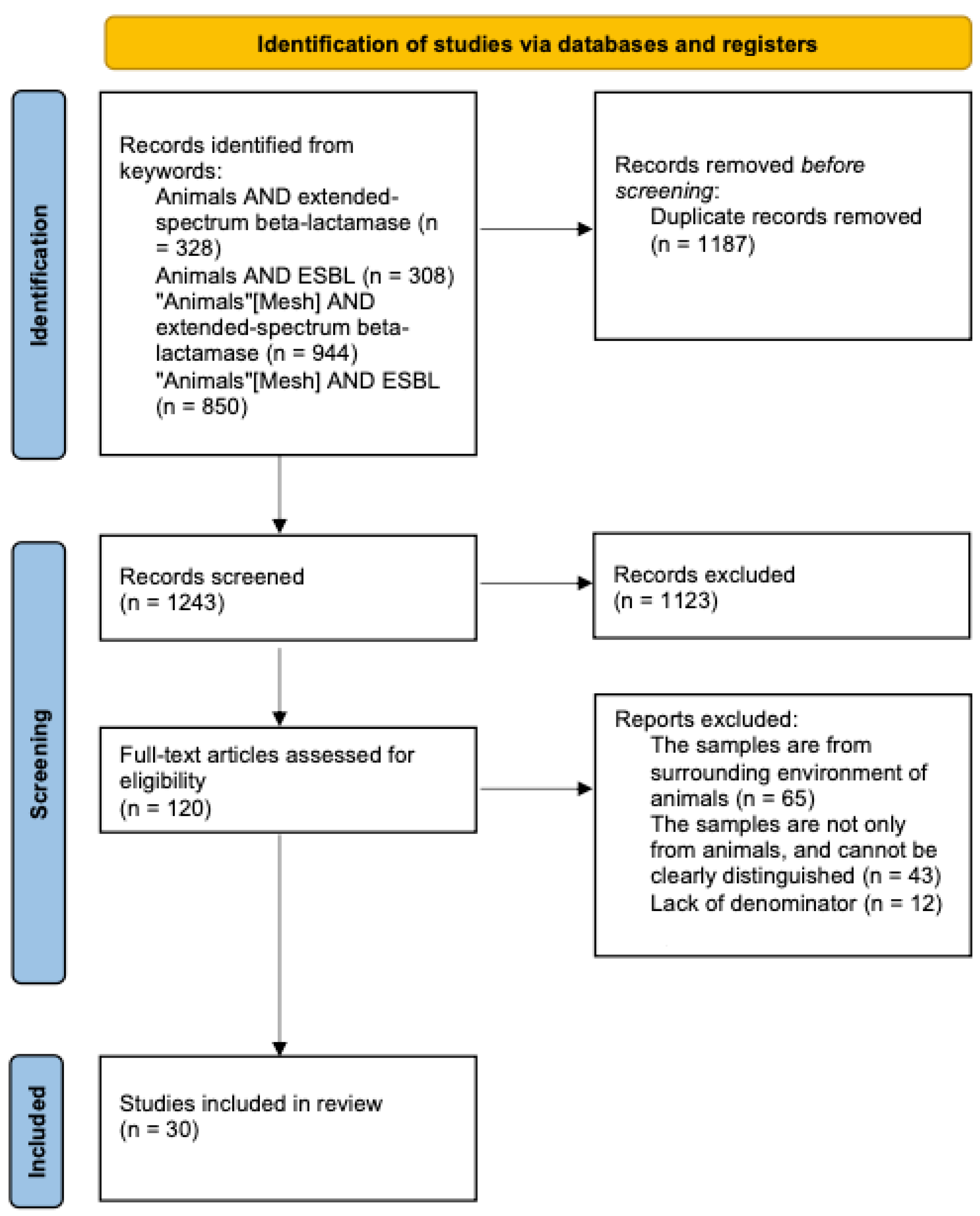
4.1. Literature Search Strategy
4.2. Inclusion and Exclusion Criteria
4.3. Study Selection
4.4. Data Extraction
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef] [PubMed]
- Darwich, L.; Vidal, A.; Seminati, C.; Albamonte, A.; Casado, A.; Lopez, F.; Molina-Lopez, R.A.; Migura-Garcia, L. High prevalence and diversity of extended-spectrum beta-lactamase and emergence of OXA-48 producing Enterobacterales in wildlife in Catalonia. PLoS ONE 2019, 14, e0210686. [Google Scholar] [CrossRef] [PubMed]
- Dandachi, I.; Sokhn, E.S.; Dahdouh, E.A.; Azar, E.; El-Bazzal, B.; Rolain, J.M.; Daoud, Z. Prevalence and Characterization of Multi-Drug-Resistant Gram-Negative Bacilli Isolated from Lebanese Poultry: A Nationwide Study. Front. Microbiol. 2018, 9, 550. [Google Scholar] [CrossRef]
- Tang, K.L.; Caffrey, N.P.; Nobrega, D.B.; Cork, S.C.; Ronksley, P.E.; Barkema, H.W.; Polachek, A.J.; Ganshorn, H.; Sharma, N.; Kellner, J.D.; et al. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: A systematic review and meta-analysis. Lancet Planet Health 2017, 1, e316–e327. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, M.; Simner, P.J.; Bradford, P.A. Extended-spectrum beta-lactamases: An update on their characteristics, epidemiology and detection. JAC Antimicrob. Resist. 2021, 3, dlab092. [Google Scholar] [CrossRef]
- Zhu, F.H.; Rodado, M.P.; Asmar, B.I.; Salimnia, H.; Thomas, R.; Abdel-Haq, N. Risk factors for community acquired urinary tract infections caused by extended spectrum beta-lactamase (ESBL) producing Escherichia coli in children: A case control study. Infect. Dis. 2019, 51, 802–809. [Google Scholar] [CrossRef]
- Tacconelli, E.; Gorska, A.; De Angelis, G.; Lammens, C.; Restuccia, G.; Schrenzel, J.; Huson, D.H.; Carevic, B.; Preotescu, L.; Carmeli, Y.; et al. Estimating the association between antibiotic exposure and colonization with extended-spectrum beta-lactamase-producing Gram-negative bacteria using machine learning methods: A multicentre, prospective cohort study. Clin. Microbiol. Infect. 2020, 26, 87–94. [Google Scholar] [CrossRef]
- Paterson, D.L.; Bonomo, R.A. Extended-spectrum beta-lactamases: A clinical update. Clin. Microbiol. Rev. 2005, 18, 657–686. [Google Scholar] [CrossRef]
- Johansson, V.; Nykasenoja, S.; Myllyniemi, A.L.; Rossow, H.; Heikinheimo, A. Genomic characterization of ESBL/AmpC-producing and high-risk clonal lineages of Escherichia coli and Klebsiella pneumoniae in imported dogs with shelter and stray background. J. Glob. Antimicrob. Resist. 2022, 30, 183–190. [Google Scholar] [CrossRef]
- Shafiq, M.; Rahman, S.U.; Bilal, H.; Ullah, A.; Noman, S.M.; Zeng, M.; Yuan, Y.; Xie, Q.; Li, X.; Jiao, X. Incidence and molecular characterization of ESBL-producing and colistin-resistant Escherichia coli isolates recovered from healthy food-producing animals in Pakistan. J. Appl. Microbiol. 2022, 133, 1169–1182. [Google Scholar] [CrossRef]
- Torres, R.T.; Cunha, M.V.; Araujo, D.; Ferreira, H.; Fonseca, C.; Palmeira, J.D. A walk on the wild side: Wild ungulates as potential reservoirs of multi-drug resistant bacteria and genes, including Escherichia coli harbouring CTX-M beta-lactamases. Environ. Pollut. 2022, 306, 119367. [Google Scholar] [CrossRef] [PubMed]
- Tello, M.; Ocejo, M.; Oporto, B.; Lavin, J.L.; Hurtado, A. Within-farm dynamics of ESBL-producing Escherichia coli in dairy cattle: Resistance profiles and molecular characterization by long-read whole-genome sequencing. Front. Microbiol. 2022, 13, 936843. [Google Scholar] [CrossRef] [PubMed]
- Mengistu, T.S.; Garcias, B.; Castellanos, G.; Seminati, C.; Molina-Lopez, R.A.; Darwich, L. Occurrence of multidrug resistant Gram-negative bacteria and resistance genes in semi-aquatic wildlife—Trachemys scripta, Neovison vison and Lutra lutra—As sentinels of environmental health. Sci. Total Environ. 2022, 830, 154814. [Google Scholar] [CrossRef]
- Aldea, I.; Gibello, A.; Hernandez, M.; Leekitcharoenphon, P.; Bortolaia, V.; Moreno, M.A. Clonal and plasmid-mediated flow of ESBL/AmpC genes in Escherichia coli in a commercial laying hen farm. Vet. Microbiol. 2022, 270, 109453. [Google Scholar] [CrossRef] [PubMed]
- Elkenany, R.; Eltaysh, R.; Elsayed, M.; Abdel-Daim, M.; Shata, R. Characterization of multi-resistant Shigella species isolated from raw cow milk and milk products. J. Vet. Med. Sci. 2022, 84, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Bello Gonzalez, T.D.J.; Kant, A.; Dijkstra, Q.; Marcato, F.; van Reenen, K.; Veldman, K.T.; Brouwer, M.S.M. Changes in Fecal Carriage of Extended-Spectrum Beta-Lactamase Producing Enterobacterales in Dutch Veal Calves by Clonal Spread of Klebsiella pneumoniae. Front. Microbiol. 2022, 13, 866674. [Google Scholar] [CrossRef]
- Gelalcha, B.D.; Ensermu, D.B.; Agga, G.E.; Vancuren, M.; Gillespie, B.E.; D’Souza, D.H.; Okafor, C.C.; Kerro Dego, O. Prevalence of Antimicrobial Resistant and Extended-Spectrum Beta-Lactamase-producing Escherichia coli in Dairy Cattle Farms in East Tennessee. Foodborne Pathog. Dis. 2022, 19, 408–416. [Google Scholar] [CrossRef]
- Gottling, J.; Heckel, J.O.; Hotzel, H.; Fruth, A.; Pfeifer, Y.; Henning, K.; Kopp, P.; Mertens-Scholz, K.; Rietschel, W.; Pfeffer, M. Zoonotic bacteria in clinically healthy goats in petting zoo settings of zoological gardens in Germany. Zoonoses Public Health 2022, 69, 333–343. [Google Scholar] [CrossRef]
- Karabasanavar, N.; Sivaraman, G.K.; S, P.S.; Nair, A.S.; Vijayan, A.; Rajan, V.; Girish, P.S. Non-diarrhoeic pigs as source of highly virulent and multidrug-resistant non-typhoidal Salmonella. Braz. J. Microbiol. 2022, 53, 1039–1049. [Google Scholar] [CrossRef]
- Islam, M.M.; Farag, E.; Hassan, M.M.; Enan, K.A.; Mohammad Sabeel, K.V.; Alhaddad, M.M.; Smatti, M.K.; Al-Marri, A.M.; Al-Zeyara, A.A.; Al-Romaihi, H.; et al. Diversity of bacterial pathogens and their antimicrobial resistance profile among commensal rodents in Qatar. Vet. Res. Commun. 2022, 46, 487–498. [Google Scholar] [CrossRef]
- Khawaskar, D.P.; Sinha, D.K.; Lalrinzuala, M.V.; Athira, V.; Kumar, M.; Chhakchhuak, L.; Mohanapriya, K.; Sophia, I.; Abhishek; Kumar, O.R.V.; et al. Pathotyping and antimicrobial susceptibility testing of Escherichia coli isolates from neonatal calves. Vet. Res. Commun. 2022, 46, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Haeili, M.; Salehzeinali, H.; Mirzaei, S.; Pishnian, Z.; Ahmadi, A. Molecular characterization of quinolone resistance and antimicrobial resistance profiles of Klebsiella pneumoniae and Escherichia coli isolated from human and broiler chickens. Int. J. Environ. Health Res. 2022, 32, 1382–1392. [Google Scholar] [CrossRef] [PubMed]
- Aklilu, E.; Harun, A.; Singh, K.K.B. Molecular characterization of bla(NDM), bla(OXA-48), mcr-1 and bla(TEM-52) positive and concurrently carbapenem and colistin resistant and extended spectrum beta-lactamase producing Escherichia coli in chicken in Malaysia. BMC Vet. Res. 2022, 18, 190. [Google Scholar] [CrossRef]
- Menck-Costa, M.F.; Baptista, A.A.S.; Gazal, L.E.S.; Justino, L.; Sanches, M.S.; de Souza, M.; Nishio, E.K.; Queiroz Dos Santos, B.; Cruz, V.D.; Berbert, J.V.M.; et al. High-Frequency Detection of fosA3 and bla (CTX-M-55) Genes in Escherichia coli From Longitudinal Monitoring in Broiler Chicken Farms. Front. Microbiol. 2022, 13, 846116. [Google Scholar] [CrossRef] [PubMed]
- Ndukui, J.G.; Gikunju, J.K.; Aboge, G.O.; Mwaniki, J.K.; Maina, J.N.; Mbaria, J.M. Molecular Characterization of ESBLs and QnrS Producers from Selected Enterobacteriaceae Strains Isolated from Commercial Poultry Production Systems in Kiambu County, Kenya. Microbiol. Insights 2022, 15, 11786361211063619. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Yan, X.; Li, Y.; Zhang, D.; Li, L.; Geng, Y.; Su, F.; Yue, C.; Hou, R.; Liu, S. Identification of extended-spectrum beta-lactamase (CTX-M)-producing Klebsiella pneumoniae belonging to ST37, ST290, and ST2640 in captive giant pandas. BMC Vet. Res. 2022, 18, 186. [Google Scholar] [CrossRef] [PubMed]
- Gruel, G.; Couvin, D.; Guyomard-Rabenirina, S.; Arlet, G.; Bambou, J.C.; Pot, M.; Roy, X.; Talarmin, A.; Tressieres, B.; Ferdinand, S.; et al. High Prevalence of bla (CTXM-1)/IncI1-Igamma/ST3 Plasmids in Extended-Spectrum Beta-Lactamase-Producing Escherichia coli Isolates Collected from Domestic Animals in Guadeloupe (French West Indies). Front. Microbiol. 2022, 13, 882422. [Google Scholar] [CrossRef]
- Mahmoud, S.F.; Fayez, M.; Swelum, A.A.; Alswat, A.S.; Alkafafy, M.; Alzahrani, O.M.; Alsunaini, S.J.; Almuslem, A.; Al Amer, A.S.; Yusuf, S. Genetic Diversity, Biofilm Formation, and Antibiotic Resistance of Pseudomonas aeruginosa Isolated from Cow, Camel, and Mare with Clinical Endometritis. Vet. Sci. 2022, 9, 239. [Google Scholar] [CrossRef]
- Islam, M.S.; Sobur, M.A.; Rahman, S.; Ballah, F.M.; Ievy, S.; Siddique, M.P.; Rahman, M.; Kafi, M.A.; Rahman, M.T. Detection of bla(TEM), bla(CTX-M), bla(CMY), and bla(SHV) Genes Among Extended-Spectrum Beta-Lactamase-Producing Escherichia coli Isolated from Migratory Birds Travelling to Bangladesh. Microb. Ecol. 2022, 83, 942–950. [Google Scholar] [CrossRef]
- Landolsi, S.; Selmi, R.; Hadjadj, L.; Ben Haj Yahia, A.; Ben Romdhane, K.; Messadi, L.; Rolain, J.M. First Report of Extended-Spectrum beta-Lactamase (bla(CTX-M1)) and Colistin Resistance Gene mcr-1 in E. coli of Lineage ST648 from Cockroaches in Tunisia. Microbiol. Spectr. 2022, 10, e0003621. [Google Scholar] [CrossRef]
- Garcia-Fierro, R.; Drapeau, A.; Dazas, M.; Saras, E.; Rodrigues, C.; Brisse, S.; Madec, J.Y.; Haenni, M. Comparative phylogenomics of ESBL-, AmpC- and carbapenemase-producing Klebsiella pneumoniae originating from companion animals and humans. J. Antimicrob. Chemother. 2022, 77, 1263–1271. [Google Scholar] [CrossRef]
- Belas, A.; Marques, C.; Menezes, J.; da Gama, L.T.; Cavaco-Silva, P.; Pomba, C. ESBL/pAmpC-Producing Escherichia coli Causing Urinary Tract Infections in Non-Related Companion Animals and Humans. Antibiotics 2022, 11, 559. [Google Scholar] [CrossRef] [PubMed]
- Aworh, M.K.; Ekeng, E.; Nilsson, P.; Egyir, B.; Owusu-Nyantakyi, C.; Hendriksen, R.S. Extended-Spectrum ss-Lactamase-Producing Escherichia coli among Humans, Beef Cattle, and Abattoir Environments in Nigeria. Front. Cell Infect. Microbiol. 2022, 12, 869314. [Google Scholar] [CrossRef]
- Nuesch-Inderbinen, M.; Hanni, C.; Zurfluh, K.; Hartnack, S.; Stephan, R. Antimicrobial resistance profiles of Escherichia coli and prevalence of extended-spectrum beta-lactamase-producing Enterobacteriaceae in calves from organic and conventional dairy farms in Switzerland. Microbiologyopen 2022, 11, e1269. [Google Scholar] [CrossRef]
- Samir, A.; Abdel-Moein, K.A.; Zaher, H.M. The Public Health Burden of Virulent Extended-Spectrum Beta-Lactamase-Producing Klebsiella pneumoniae Strains Isolated from Diseased Horses. Vector-Borne Zoonotic Dis. 2022, 22, 217–224. [Google Scholar] [CrossRef]
- Loucif, L.; Chelaghma, W.; Cherak, Z.; Bendjama, E.; Beroual, F.; Rolain, J.M. Detection of NDM-5 and MCR-1 antibiotic resistance encoding genes in Enterobacterales in long-distance migratory bird species Ciconia ciconia, Algeria. Sci. Total Environ. 2022, 814, 152861. [Google Scholar] [CrossRef] [PubMed]
- Benavides, J.A.; Godreuil, S.; Opazo-Capurro, A.; Mahamat, O.O.; Falcon, N.; Oravcova, K.; Streicker, D.G.; Shiva, C. Long-term maintenance of multidrug-resistant Escherichia coli carried by vampire bats and shared with livestock in Peru. Sci. Total Environ. 2022, 810, 152045. [Google Scholar] [CrossRef]
- Trongjit, S.; Assavacheep, P.; Samngamnim, S.; My, T.H.; An, V.T.T.; Simjee, S.; Chuanchuen, R. Plasmid-mediated colistin resistance and ESBL production in Escherichia coli from clinically healthy and sick pigs. Sci. Rep. 2022, 12, 2466. [Google Scholar] [CrossRef] [PubMed]
- Dallenne, C.; Da Costa, A.; Decre, D.; Favier, C.; Arlet, G. Development of a set of multiplex PCR assays for the detection of genes encoding important beta-lactamases in Enterobacteriaceae. J. Antimicrob. Chemother. 2010, 65, 490–495. [Google Scholar] [CrossRef]
- Randall, L.P.; Cooles, S.W.; Osborn, M.K.; Piddock, L.J.; Woodward, M.J. Antibiotic resistance genes, integrons and multiple antibiotic resistance in thirty-five serotypes of Salmonella enterica isolated from humans and animals in the UK. J. Antimicrob. Chemother. 2004, 53, 208–216. [Google Scholar] [CrossRef]
- Strauss, L.M.; Dahms, C.; Becker, K.; Kramer, A.; Kaase, M.; Mellmann, A. Development and evaluation of a novel universal beta-lactamase gene subtyping assay for blaSHV, blaTEM and blaCTX-M using clinical and livestock-associated Escherichia coli. J. Antimicrob. Chemother. 2015, 70, 710–715. [Google Scholar] [CrossRef][Green Version]
- Gootz, T.D.; Lescoe, M.K.; Dib-Hajj, F.; Dougherty, B.A.; He, W.; Della-Latta, P.; Huard, R.C. Genetic organization of transposase regions surrounding blaKPC carbapenemase genes on plasmids from Klebsiella strains isolated in a New York City hospital. Antimicrob. Agents Chemother. 2009, 53, 1998–2004. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Sen, M.R.; Anupurba, S.; Prakash, P.; Nath, G. Detection of OXA-2 group extended-spectrum-beta-lactamase-producing clinical isolates of Escherichia coli from India. J. Antimicrob. Chemother. 2007, 60, 703–704. [Google Scholar] [CrossRef]
- Christophy, R.; Osman, M.; Mallat, H.; Achkar, M.; Ziedeh, A.; Moukaddem, W.; Dabboussi, F.; Hamze, M. Prevalence, antibiotic susceptibility and characterization of antibiotic resistant genes among carbapenem-resistant Gram-negative bacilli and yeast in intestinal flora of cancer patients in North Lebanon. J. Infect. Public Health 2017, 10, 716–720. [Google Scholar] [CrossRef]
- Grobner, S.; Linke, D.; Schutz, W.; Fladerer, C.; Madlung, J.; Autenrieth, I.B.; Witte, W.; Pfeifer, Y. Emergence of carbapenem-non-susceptible extended-spectrum beta-lactamase-producing Klebsiella pneumoniae isolates at the university hospital of Tubingen, Germany. J. Med. Microbiol. 2009, 58, 912–922. [Google Scholar] [CrossRef]
- Ly, T.D.A.; Hadjadj, L.; Hoang, V.T.; Louni, M.; Dao, T.L.; Badiaga, S.; Tissot-Dupont, H.; Raoult, D.; Rolain, J.M.; Gautret, P. Low prevalence of resistance genes in sheltered homeless population in Marseille, France, 2014–2018. Infect. Drug Resist. 2019, 12, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Hosu, M.C.; Vasaikar, S.D.; Okuthe, G.E.; Apalata, T. Detection of extended spectrum beta-lactamase genes in Pseudomonas aeruginosa isolated from patients in rural Eastern Cape Province, South Africa. Sci. Rep. 2021, 11, 7110. [Google Scholar] [CrossRef]
- Pitout, J.D.; Thomson, K.S.; Hanson, N.D.; Ehrhardt, A.F.; Moland, E.S.; Sanders, C.C. beta-Lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob. Agents Chemother. 1998, 42, 1350–1354. [Google Scholar] [CrossRef]
- Fang, H.; Ataker, F.; Hedin, G.; Dornbusch, K. Molecular epidemiology of extended-spectrum beta-lactamases among Escherichia coli isolates collected in a Swedish hospital and its associated health care facilities from 2001 to 2006. J. Clin. Microbiol. 2008, 46, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Hasman, H.; Mevius, D.; Veldman, K.; Olesen, I.; Aarestrup, F.M. Beta-Lactamases among extended-spectrum beta-lactamase (ESBL)-resistant Salmonella from poultry, poultry products and human patients in The Netherlands. J. Antimicrob. Chemother. 2005, 56, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Rahman, S.U.; Zhang, L.; Shahid, M.; Han, D.; Gao, J.; Zhang, S.; Ruegg, P.L.; Saddique, U.; Han, B. Characteristics and genetic diversity of multi-drug resistant extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli isolated from bovine mastitis. Oncotarget 2017, 8, 90144–90163. [Google Scholar] [CrossRef] [PubMed]
- Weill, F.X.; Demartin, M.; Tande, D.; Espie, E.; Rakotoarivony, I.; Grimont, P.A. SHV-12-like extended-spectrum-beta-lactamase-producing strains of Salmonella enterica serotypes Babelsberg and Enteritidis isolated in France among infants adopted from Mali. J. Clin. Microbiol. 2004, 42, 2432–2437. [Google Scholar] [CrossRef] [PubMed]
- Colom, K.; Perez, J.; Alonso, R.; Fernandez-Aranguiz, A.; Larino, E.; Cisterna, R. Simple and reliable multiplex PCR assay for detection of blaTEM, bla(SHV) and blaOXA-1 genes in Enterobacteriaceae. FEMS Microbiol. Lett. 2003, 223, 147–151. [Google Scholar] [CrossRef]
- Rayamajhi, N.; Kang, S.G.; Lee, D.Y.; Kang, M.L.; Lee, S.I.; Park, K.Y.; Lee, H.S.; Yoo, H.S. Characterization of TEM-, SHV- and AmpC-type beta-lactamases from cephalosporin-resistant Enterobacteriaceae isolated from swine. Int. J. Food Microbiol. 2008, 124, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Lob, S.H.; Kazmierczak, K.M.; Badal, R.E.; Hackel, M.A.; Bouchillon, S.K.; Biedenbach, D.J.; Sahm, D.F. Trends in susceptibility of Escherichia coli from intra-abdominal infections to ertapenem and comparators in the United States according to data from the SMART program, 2009 to 2013. Antimicrob. Agents Chemother. 2015, 59, 3606–3610. [Google Scholar] [CrossRef]
- El-Shazly, S.; Dashti, A.; Vali, L.; Bolaris, M.; Ibrahim, A.S. Molecular epidemiology and characterization of multiple drug-resistant (MDR) clinical isolates of Acinetobacter baumannii. Int. J. Infect. Dis. 2015, 41, 42–49. [Google Scholar] [CrossRef]
- Van, T.T.; Chin, J.; Chapman, T.; Tran, L.T.; Coloe, P.J. Safety of raw meat and shellfish in Vietnam: An analysis of Escherichia coli isolations for antibiotic resistance and virulence genes. Int. J. Food Microbiol. 2008, 124, 217–223. [Google Scholar] [CrossRef]
- Archambault, M.; Petrov, P.; Hendriksen, R.S.; Asseva, G.; Bangtrakulnonth, A.; Hasman, H.; Aarestrup, F.M. Molecular characterization and occurrence of extended-spectrum beta-lactamase resistance genes among Salmonella enterica serovar Corvallis from Thailand, Bulgaria, and Denmark. Microb. Drug Resist. 2006, 12, 192–198. [Google Scholar] [CrossRef]
- Ahmed, O.I.; El-Hady, S.A.; Ahmed, T.M.; Ahmed, I.Z. Detection of bla SHV and bla CTX-M genes in ESBL producing Klebsiella pneumoniae isolated from Egyptian patients with suspected nosocomial infections. Egypt. J. Med. Hum. Genet. 2013, 14, 277–283. [Google Scholar] [CrossRef]
- Gundran, R.S.; Cardenio, P.A.; Villanueva, M.A.; Sison, F.B.; Benigno, C.C.; Kreausukon, K.; Pichpol, D.; Punyapornwithaya, V. Prevalence and distribution of bla(CTX-M), bla(SHV), bla(TEM) genes in extended- spectrum beta- lactamase- producing E. coli isolates from broiler farms in the Philippines. BMC Vet. Res. 2019, 15, 227. [Google Scholar] [CrossRef]
- Batchelor, M.; Hopkins, K.; Threlfall, E.J.; Clifton-Hadley, F.A.; Stallwood, A.D.; Davies, R.H.; Liebana, E. Bla(CTX-M) genes in clinical Salmonella isolates recovered from humans in England and Wales from 1992 to 2003. Antimicrob. Agents Chemother. 2005, 49, 1319–1322. [Google Scholar] [CrossRef]
- Edelstein, M.; Pimkin, M.; Palagin, I.; Edelstein, I.; Stratchounski, L. Prevalence and molecular epidemiology of CTX-M extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae in Russian hospitals. Antimicrob. Agents Chemother. 2003, 47, 3724–3732. [Google Scholar] [CrossRef] [PubMed]
- Horton, R.A.; Randall, L.P.; Snary, E.L.; Cockrem, H.; Lotz, S.; Wearing, H.; Duncan, D.; Rabie, A.; McLaren, I.; Watson, E.; et al. Fecal carriage and shedding density of CTX-M extended-spectrum beta-lactamase-producing Escherichia coli in cattle, chickens, and pigs: Implications for environmental contamination and food production. Appl. Environ. Microbiol. 2011, 77, 3715–3719. [Google Scholar] [CrossRef] [PubMed]
- Woodford, N.; Fagan, E.J.; Ellington, M.J. Multiplex PCR for rapid detection of genes encoding CTX-M extended-spectrum (beta)-lactamases. J. Antimicrob. Chemother. 2006, 57, 154–155. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, Y.; Matten, J.; Rabsch, W. Salmonella enterica serovar Typhi with CTX-M beta-lactamase, Germany. Emerg. Infect. Dis. 2009, 15, 1533–1535. [Google Scholar] [CrossRef]
- Wetzker, W.; Pfeifer, Y.; Wolke, S.; Haselbeck, A.; Leistner, R.; Kola, A.; Gastmeier, P.; Salm, F. Extended-Spectrum Beta-Lactamase (ESBL)-Producing Escherichia coli Isolated from Flies in the Urban Center of Berlin, Germany. Int. J. Environ. Res. Public Health 2019, 16, 1530. [Google Scholar] [CrossRef]
- Carattoli, A. Animal reservoirs for extended spectrum beta-lactamase producers. Clin. Microbiol. Infect. 2008, 14 (Suppl. S1), 117–123. [Google Scholar] [CrossRef]
- Dantas Palmeira, J.; Ferreira, H.M.N. Extended-spectrum beta-lactamase (ESBL)-producing Enterobacteriaceae in cattle production—A threat around the world. Heliyon 2020, 6, e03206. [Google Scholar] [CrossRef]
- Mesbah Zekar, F.; Granier, S.A.; Marault, M.; Yaici, L.; Gassilloud, B.; Manceau, C.; Touati, A.; Millemann, Y. From Farms to Markets: Gram-Negative Bacteria Resistant to Third-Generation Cephalosporins in Fruits and Vegetables in a Region of North Africa. Front. Microbiol. 2017, 8, 1569. [Google Scholar] [CrossRef]
- De Boeck, H.; Miwanda, B.; Lunguya-Metila, O.; Muyembe-Tamfum, J.J.; Stobberingh, E.; Glupczynski, Y.; Jacobs, J. ESBL-positive Enterobacteria isolates in drinking water. Emerg. Infect. Dis. 2012, 18, 1019–1020. [Google Scholar] [CrossRef]
- World Health Organization. WHO Integrated Global Surveillance on ESBL-Producing E. coli Using a “One Health” Approach: Implementation and Opportunities; World Health Organization: Geneva, Switzerland, 2021.
- Bradford, P.A. Extended-spectrum beta-lactamases in the 21st century: Characterization, epidemiology, and detection of this important resistance threat. Clin. Microbiol. Rev. 2001, 14, 933–951. [Google Scholar] [CrossRef] [PubMed]
- Brazelton de Cardenas, J.N.; Garner, C.D.; Su, Y.; Tang, L.; Hayden, R.T. Comparative Evaluation of Assays for Broad Detection of Molecular Resistance Mechanisms in Enterobacterales Isolates. J. Clin. Microbiol. 2021, 59, e0103321. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Stephan, R.; Karczmarczyk, M.; Yan, Q.; Hachler, H.; Fanning, S. Molecular characterization of bla ESBL-harboring conjugative plasmids identified in multi-drug resistant Escherichia coli isolated from food-producing animals and healthy humans. Front. Microbiol. 2013, 4, 188. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Stephan, R.; Power, K.; Yan, Q.; Hachler, H.; Fanning, S. Nucleotide sequences of 16 transmissible plasmids identified in nine multidrug-resistant Escherichia coli isolates expressing an ESBL phenotype isolated from food-producing animals and healthy humans. J. Antimicrob. Chemother. 2014, 69, 2658–2668. [Google Scholar] [CrossRef]
- Anes, J.; Nguyen, S.V.; Eshwar, A.K.; McCabe, E.; Macori, G.; Hurley, D.; Lehner, A.; Fanning, S. Molecular characterisation of multi-drug resistant Escherichia coli of bovine origin. Vet. Microbiol. 2020, 242, 108566. [Google Scholar] [CrossRef]
- Saidani, M.; Messadi, L.; Mefteh, J.; Chaouechi, A.; Soudani, A.; Selmi, R.; Daaloul-Jedidi, M.; Ben Chehida, F.; Mamlouk, A.; Jemli, M.H.; et al. Various Inc-type plasmids and lineages of Escherichia coli and Klebsiella pneumoniae spreading bla(CTX-M-15,)bla(CTX-M-1) and mcr-1 genes in camels in Tunisia. J. Glob. Antimicrob. Resist. 2019, 19, 280–283. [Google Scholar] [CrossRef]
- Pepin-Puget, L.; El Garch, F.; Bertrand, X.; Valot, B.; Hocquet, D. Genome analysis of enterobacteriaceae with non-wild type susceptibility to third-generation cephalosporins recovered from diseased dogs and cats in Europe. Vet. Microbiol. 2020, 242, 108601. [Google Scholar] [CrossRef]
- Rozwandowicz, M.; Brouwer, M.S.M.; Fischer, J.; Wagenaar, J.A.; Gonzalez-Zorn, B.; Guerra, B.; Mevius, D.J.; Hordijk, J. Plasmids carrying antimicrobial resistance genes in Enterobacteriaceae. J. Antimicrob. Chemother. 2018, 73, 1121–1137. [Google Scholar] [CrossRef]
- Wiegand, I.; Geiss, H.K.; Mack, D.; Sturenburg, E.; Seifert, H. Detection of extended-spectrum beta-lactamases among Enterobacteriaceae by use of semiautomated microbiology systems and manual detection procedures. J. Clin. Microbiol. 2007, 45, 1167–1174. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- University of Alabama. A Research Guide for Systematic Literature Reviews. University Libraries. Available online: https://guides.lib.ua.edu/c.php?g=1135040&p=8286355 (accessed on 24 May 2022).
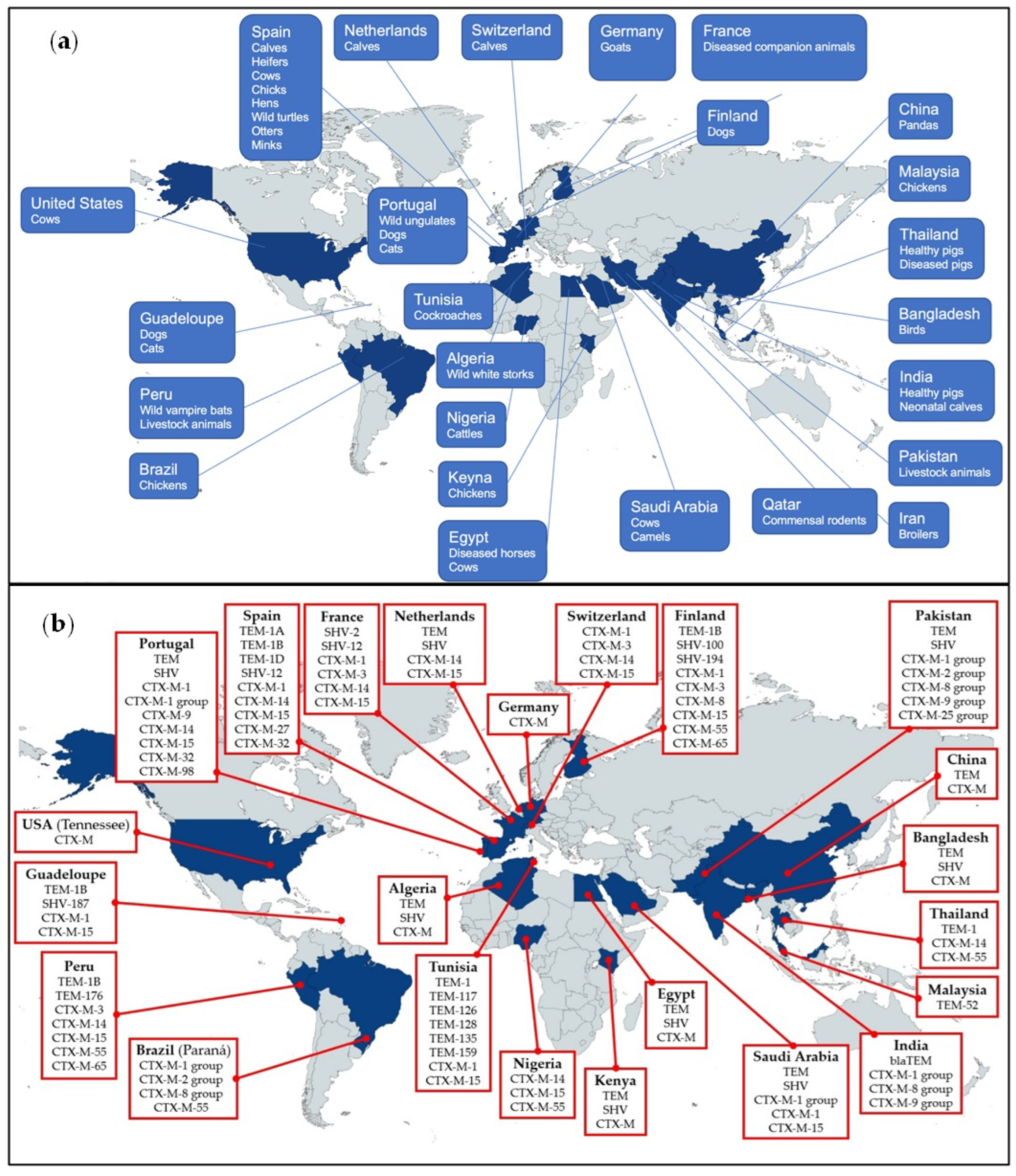
| Article | Country | Location | Sample | Animals | Date of Isolation | Bacteria |
|---|---|---|---|---|---|---|
| Venla Johansson et al., 2022 [9] | Finland | airport, animal clinics, animal shelters | rectal swabs or fresh feces | dogs | 2017–2018 | Escherichia coli and Klebsiella pneumoniae |
| Muhammad Shafiq et al., 2022 [10] | Pakistan | farms | rectal swabs or fresh droppings | buffaloes, cattle, sheep, goats, and broilers | no details | Escherichia coli |
| Rita Tinoco Torres et al., 2022 [11] | Portugal | hunting grounds | rectal swabs | Wild ungulates | October 2018–February 2020 | Enterobacterales |
| Maitane Tello et al., 2022 [12] | Spain | dairy cattle farms | rectal swabs | calves, heifers, and cows | February 2019–October 2020 | Escherichia coli |
| Tilaye Shibbiru Mengistu et al., 2022 [13] | Spain | a highly populated and intensive farming region | cloacal/rectal swabs | turtles, minks, and otters | January 2018–July 2021 | Enterobacterales, and some other Gram-negative bacteria |
| Irene Aldea et al., 2022 [14] | Spain | a commercial laying hen farm | fresh meconium droppings, feces | chicks and hens | March 2016–October 2018 | Escherichia coli |
| Rasha Elkenany et al., 2022 [15] | Egypt | dairy farms | raw cow milk | cows | 2018 | Shigella species |
| Teresita d.J. Bello Gonzalez et al., 2022 [16] | Netherlands | dairy farms | rectal swabs | calves | March 2019–May 2020 | Escherichia coli and Klebsiella pneumoniae |
| Benti D Gelalcha et al., 2022 [17] | USA | dairy farms | bulk tank milk | cows | no details | Escherichia coli |
| Jannis Göttling et al., 2022 [18] | Germany | petting zoo | rectal swabs | healthy goats | August 2016–June 2017 | Enterobacterales |
| Nagappa Karabasanavar et al., 2022 [19] | India | pig farms | rectal swabs | healthy pigs | April 2019–April 2020 | Salmonella species |
| Md Mazharul Islam et al., 2021 [20] | Qatar | livestock farms, agricultural farms | blood and visceral samples | commensal rodents | August 2019–February 2020 | Gram-negative bacilli |
| Damini P. Khawaskar et al., 2021 [21] | India | dairy farms | rectal swabs | neonatal calves | no details | Escherichia coli |
| Mehri Haeili et al., 2021 [22] | Iran | a chicken slaughterhouse | cloacal swabs | slaughtered broilers | no details | Escherichia coli and Klebsiella pneumoniae |
| Erkihun Aklilu et al., 2022 [23] | Malaysia | farms | cloacal swabs | broiler chickens | no details | Escherichia coli |
| Maísa Fabiana Menck-Costa et al., 2022 [24] | Brazil | broiler farms | cloacal swabs | broiler chickens | March 2019–July 2020 | Escherichia coli |
| James G Ndukui et al., 2021 [25] | Kenya | poultry production centers | cloacal swabs | chickens | November 2020–February 2021 | Enterobacterales |
| Xiaoyan Su et al., 2022 [26] | China | Chengdu Research Base of Giant Panda Breeding | fresh feces | captive giant pandas | 2018–2019 | Klebsiella pneumoniae |
| Gaëlle Gruel et al., 2022 [27] | Guadeloupe (French West Indies) | animal shelters and veterinary clinics | rectal swabs | dogs and cats | June 2019–September 2019 | Enterobacterales |
| Samy F. Mahmoud et al., 2022 [28] | Saudi Arabia | farms | uterine swabs | diseased cow, camel, and mare | May 2020–February 2021 | Pseudomonas aeruginosa |
| Md Saiful Islam et al., 2021 [29] | Bangladesh | no details | fresh feces | migratory birds | November 2019–November 2020 | Escherichia coli |
| Sarrah Landolsi et al., 2022 [30] | Tunisia | a collective catering, houses, and a hospital | external surface and gut homogenates | cockroaches | July 2017–June 2018 | Enterobacterales |
| Raquel Garcia-Fierro et al., 2022 [31] | France | no details | Urine, pus, and respiratory pathological specimens | diseased dogs, cats, horses, cattle, and birds | 2010–2018 | Klebsiella pneumoniae |
| Adriana Belas et al., 2022 [32] | Portugal | faculty of veterinary medicine | urine | dogs and cats | 1999–2015 | Escherichia coli |
| Mabel Kamweli Aworh et al., 2022 [33] | Nigeria | abattoirs | Cecal contents from the cecum | slaughtered beef cattle | May 2020–December 2020 | Escherichia coli |
| Magdalena Nüesch-Inderbinen et al., 2022 [34] | Switzerland | organic and conventional dairy farms | fresh feces | calves | September 2020 | Enterobacterales |
| Ahmed Samir et al., 2022 [35] | Egypt | equine farms | rectal swabs and nasal swabs | diseased adult horses | August 2020–March 2021 | Klebsiella pneumoniae |
| Lotfi Loucif et al., 2021 [36] | Algeria | nests and a colony | fresh feces | white stork | May 2019 | Enterobacterales |
| Julio A Benavides et al., 2021 [37] | Peru | colonies | rectal swabs | vampire bats | 2015 (October), 2017 (March to May), and 2018 (February and March) | Escherichia coli |
| farms located nearby vampire bat colonies | fresh feces | cows, pigs, goats, sheep, and donkeys | 2015 | Escherichia coli | ||
| Suthathip Trongjit et al., 2022 [38] | Thailand | farms | rectal swabs | pigs | 2007–2018 | Escherichia coli |
| Article | Selective Media | Target Identification | Total Number | ESBL Number | % | ESBL Test | ESBL Genes Test | ESBL Genes and Number |
|---|---|---|---|---|---|---|---|---|
| Venla Johansson et al., 2022 [9] | MacConkey agar with cefotaxime (1 mg/L) | MALDI-TOF | 60 | 47 | 78.3% | double-disc synergy test | WGS | blaTEM (25), blaSHV (2), blaCTX-M-1 (7), blaCTX-M-3 (1), blaCTX-M-15 (36), blaCTX-M-55 (2), blaCTX-M-8 (1), blaCTX-M-65 (1) |
| Muhammad Shafiq et al., 2022 [10] | MacConkey agar | PCR (uidA gene) | 153 | 75 | 49.0% | double-disc synergy test | PCR | blaTEM (37), blaSHV (32), blaCTX-M-1 group (35), blaCTX-M-2 group (5), blaCTX-M-8 group (1), blaCTX-M-9 group (32), blaCTX-M-25 group (3) |
| Rita Tinoco Torres et al., 2022 [11] | MacConkey agar with antibiotic 1 | biochemical reaction (API20E galleries) | 151 | 4 | 2.6% | Characteristic phenotypic synergism with ESBL genes | PCR | blaTEM (60), blaSHV (3), blaCTX-M (4) [CTX-M-14 (2), CTX-M-15 (1), CTX-M-98 (1)] |
| Maitane Tello et al., 2022 [12] | MacConkey agar with cefotaxime (1 mg/L) | PCR (uidA gene) | 41 | 39 | 95.1% | ESBL genes | WGS | blaTEM (17), blaSHV (1), blaCTX-M-1 (9), blaCTX-M-14 (12), blaCTX-M-15 (9), blaCTX-M-27 (3), blaCTX-M-32 (5) |
| Tilaye Shibbiru Mengistu et al., 2022 [13] | MacConkey agar with ceftriaxone (1 mg/L) | API® biochemical test strips or automated system (VITEK 2) | 131 | 4 | 3.1% | ESBL genes | PCR | blaTEM (0), blaCTX-M (4) [CTX-M-15 (4)] |
| Irene Aldea et al., 2022 [14] | MacConkey agar with cefotaxime (1 mg/L) | PCR, API 20-E kit, or whole genome sequencing | 47 | 29 | 61.7% | ESBL genes 8 | WGS | blaTEM (19), blaSHV (9), blaCTX-M-1 (19), blaCTX-M-14 (1) |
| Rasha Elkenany et al., 2022 [15] | Salmonella-Shigella agar, MacConkey agar, and xylose-lysine-deoxycholate agar | Biochemical reaction 4 | 16 | 4 | 25.0% | double-disc synergy test | PCR | blaTEM (16), blaSHV (0), blaCTX-M (4) |
| Teresita d.J. Bello Gonzalez et al., 2022 [16] | MacConkey agar with cefotaxime (1 mg/L) | MALDI-TOF | 254 | 254 | 100.0% | ESBL genes | PCR | blaTEM (254), blaSHV (174), blaCTX-M-14 (174), blaCTX-M-15 (80) |
| Benti D Gelalcha et al., 2022 [17] | CHROMagar™ E. coli agar | PCR (uidA gene) | 14 | 4 | 28.6% | ESBL genes | PCR | blaTEM (0), blaSHV (0), blaCTX-M (4) |
| Jannis Göttling et al., 2022 [18] | Oxoid Brilliance ESBL agar | automated system (VITEK 2) | 300 | 1 | 0.3% | Commercial disc test system (D68C ESBL/AmpC ID, MAST group Diagnostics) | PCR | blaTEM (0), blaSHV (0), blaCTX-M-1 (1), blaCTX-M-2 (0), blaCTX-M-9 (0) |
| Nagappa Karabasanavar et al., 2022 [19] | Xylose-lysine-deoxycholate agar, Brilliant green agar, Bismuth sulfite agar, Hektoen Enteric agar | Biochemical reaction 5 | 22 | 12 | 54.5% | ESBL genes | PCR | blaTEM (12), blaSHV (0), blaCTX-M-1 (0), blaCTX-M-2 (0), blaCTX-M-9 (0) |
| Md Mazharul Islam et al., 2021 [20] | MacConkey agar, Hektoen enteric agar, Eosin methylene blue agar | automated system (VITEK 2) | 68 | 9 | 13.2% | VITEK 2 AST-GN cards | no test | no test |
| Damini P. Khawaskar et al., 2021 [21] | MacConkey agar and Eosin methylene blue agar | biochemical reaction (IMViC Test) | 280 | 120 | 42.9% | combination disk method | PCR | blaTEM (10), blaSHV (0), blaCTX-M-1 group (34), blaCTX-M-2 group (0), blaCTX-M-8 group (2), blaCTX-M-9 group (1), blaCTX-M-25 group (0) |
| Mehri Haeili et al., 2021 [22] | no details | no details | 21 | 0 | 0.0% | combination disk method | no test | no test |
| Erkihun Aklilu et al., 2022 [23] | MacConkey and Eosine Methylene Blue agars | PCR (E. coli specific gene) | 49 | 12 | 24.5% | ESBL genes | PCR | blaTEM (12), blaCTX-M (0) |
| Maísa Fabiana Menck-Costa et al., 2022 [24] | MacConkey agar with/without antibiotics 2 | biochemical reaction 6 | 360 | 198 | 55.0% | double-disc synergy test | PCR | blaCTX-M-1 group (153), blaCTX-M-2 group (61), blaCTX-M-8 group (5), blaCTX-M-9 group (0), blaCTX-M-25 group (0) |
| James G Ndukui et al., 2021 [25] | no details | biochemical reaction | 544 | 30 | 5.5% | phenotypic resistance profiles and then ESBL genes | PCR | blaTEM (14), blaSHV (5), blaCTX-M (11) |
| Xiaoyan Su et al., 2022 [26] | no details | 16 s rDNA and biochemical reaction | 211 | 3 | 1.4% | double-disc synergy test | PCR | blaTEM (2), blaSHV (0), blaCTX-M (3), blaGES (0), blaPER (0), blaVEB (0) |
| Gaëlle Gruel et al., 2022 [27] | CHROMagar™ CCA with ceftriaxone (4 mg/L) | API 20-E kit | 185 | 14 | 7.6% | double-disk synergy test | WGS | blaTEM (1), blaSHV (1), blaCTX-M-1 (11), blaCTX-M-15 (3) |
| Samy F. Mahmoud et al., 2022 [28] | Pseudomonas cetrimide agar | automated system (VITEK 2) | 44 | 20 | 45.5% | double-disk synergy test | PCR | blaTEM (18), blaSHV (8), blaCTX-M (11) |
| Md Saiful Islam et al., 2021 [29] | Eosin methylene blue agar | biochemical reaction 7 | 55 | 21 | 38.2% | double-disk synergy test | PCR | blaTEM (20), blaSHV (9), blaCTX-M (18) |
| Sarrah Landolsi et al., 2022 [30] | MacConkey agar with cefotaxime (1 mg/L) | MALDI-TOF | 144 | 22 | 15.3% | double-disk synergy test | PCR | blaTEM (9), blaSHV (0), blaCTX-M (15) [blaCTX-M-1 (7), blaCTX-M-15 (8)] |
| Raquel Garcia-Fierro et al., 2022 [31] | no details, but cefoxitin- and/or ceftiofur-resistant | MALDI-TOF | 105 | 52 | 49.5% | ESBL genes | WGS | blaSHV (2), blaCTX-M-1 (3), blaCTX-M-3 (1), blaCTX-M-14 (4), blaCTX-M-15 (42) |
| Adriana Belas et al., 2022 [32] | no details, but Third-generation cephalosporin-resistant | PCR (gadA gene) | 35 | 14 | 40.0% | ESBL genes | PCR | blaCTX-M-1 (2), blaCTX-M-1-like (2), blaCTX-M-9 (1), blaCTX-M-15 (7), blaCTX-M-32 (3) |
| Mabel Kamweli Aworh et al., 2022 [33] | MacConkey agar with cefotaxime (1 mg/L) | biochemical reaction (commercially Microbact GNB 24E kit) | 272 | 44 | 16.2% | combination disk method | WGS | blaCTX-M-14 (1), blaCTX-M-15 (41), blaCTX-M-55 (1) |
| Magdalena Nüesch-Inderbinen et al., 2022 [34] | Rapid’ E. coli two agar plates | MALDI-TOF | 196 | 21 | 10.7% | Brilliance ESBL agar plates | PCR | blaTEM (0), blaSHV (0), blaCTX-M-1 (7), blaCTX-M-3 (4), blaCTX-M-14 (2), blaCTX-M-15 (8) |
| Ahmed Samir et al., 2022 [35] | MacConkey agar with cefotaxime (2 mg/L) | PCR (Klebsiella gyrA gene, ITS gene) | 100 | 13 | 13.0% | double-disc synergy test | PCR | blaTEM (13), blaSHV (13), blaCTX-M (12) |
| Lotfi Loucif et al., 2021 [36] | MacConkey agar with antibiotics 3 | MALDI-TOF | 42 | 8 | 19.0% | double-disc synergy test | PCR | blaTEM (20), blaSHV (4), blaCTX-M (19) |
| Julio A Benavides et al., 2021 [37] | ChromID ESBL agar | MALDI-TOF | 388 | 20 | 5.2% | ChromID ESBL agar | WGS | blaTEM (17), blaCTX-M-3 (2), blaCTX-M-14 (0), blaCTX-M-15 (7), blaCTX-M-55 (8), blaCTX-M-65 (1) |
| 134 | 65 | 48.5% | ChromID ESBL agar | WGS | blaTEM (14), blaCTX-M-3 (1), blaCTX-M-14 (3), blaCTX-M-15 (2), blaCTX-M-55 (7), blaCTX-M-65 (3) | |||
| Suthathip Trongjit et al., 2022 [38] | no details | no details | 454 | 112 | 24.7% | combination disk method | PCR | blaTEM (81), blaSHV (0), blaCTX-M-14 (61), blaCTX-M-55 (48) |
| Target | Forward Primer (5′-3′) | Reverse Primer (5′-3′) | Articles | Reference |
|---|---|---|---|---|
| blaTEM | CATTTCCGTGTCGCCCTTATTC | CGTTCATCCATAGTTGCCTGAC | [11,19,26] | Dallenne et al., 2010 [39] |
| CATTTCCGTGTCGCCCTTATTC | TCCATAGTTGCCTGACTCCC | [29] | Randall et al., 2004 [40] | |
| CATTTCCGTGTCGCCCTTATTC | CCAATGCTTAATCAGTGAGGC | [17] | Strauss et al., 2015 [41] | |
| ATGAGTATTCAACATTTCCG | CCAATGCTTAATCAGTGAGGC | [25] | Gootz et al., 2009 [42] | |
| ATGAGTATTCAACATTTCCG | CTGACAGTTACCAATGCTTA | [21,30] | Bhattacharjee et al., 2007 [43], Christophy et al., 2017 [44] | |
| ATGAGTATTCAACATTTCCG | TTAATCAGTGAGGCACCTAT | [18] | Grobner et al., 2009 [45] | |
| TTCTGCTATGTGGTGCGGTA | GTCCTCCGATCGTTGTCAGA | [36] | Ly et al., 2019 [46] | |
| GCATCTTACGGATGGCATGA | GTCCTCCGATCGTTGTCAGA | [28] | Hosu et al., 2021 [47] | |
| TCGGGGAAATGTGCGCG | TGCTTAATCAGTGAGGCACC | [34] | Pitout et al., 1998 [48] | |
| CGCCGCATACACTATTCTCAGAATGA | ACGCTCACCGGCTCCAGATTTAT | [35] | Fang et al., 2008 [49] | |
| GCGGAACCCCTATTTG | TCTAAAGTATATATGAGTAAACTTGGTCTGAC | [13,38] | Darwich et al., 2019 [2], Hasman et al., 2005 [50] | |
| ATAAAATTCTTGAAGACGAAA | GACAGTTACCAATGCTTAATC | [10,23] | Ali et al., 2017 [51], Weill et al., 2004 [52] | |
| ATCAGCAATAAACCAGC | CCCCGAAGAACGTTTTC | [15] | Colom et al., 2003 [53] | |
| blaSHV | CACTCAAGGATGTATTGTG | TTAGCGTTGCCAGTGCTCG | [34] | Pitout et al., 1998 [48] |
| TTCGCCTGTGTATTATCTCCCTG | TTAGCGTTGCCAGTGYTCG | [38] | Hasman et al., 2005 [50] | |
| GGGTTATTCTTATTTGTCGC | TTAGCGTTGCCAGTGCTC | [10] | Ali et al., 2017 [51] | |
| CTTTATCGGCCCTCACTCAA | AGGTGCTCATCATGGGAAAG | [35] | Fang et al., 2008 [49] | |
| TCCCATGATGAGCACCTTTAAA | TCCTGCTGGCGATAGTGGAT | [28,36] | Hosu et al., 2021 [47], Ly et al., 2019 [46] | |
| AGCCGCTTGAGCAAATTAAAC | ATCCCGCAGATAAATCACCAC | [11,19,26] | Dallenne et al., 2010 [39] | |
| AGGATTGACTGCCTTTTTG | ATTTGCTGATTTCGCTCG | [15] | Colom et al., 2003 [53] | |
| GCCGGGTTATTCTTATTTGTCGC | ATGCCGCCGCCAGTCA | [17] | Rayamajhi et al., 2008 [54] | |
| GCAAAACGCCGGGTTATTC | GGTTAGCGTTGCCAGTGCT | [18] | Grobner et al., 2009 [45] | |
| CCTTTAAAGTAGTGCTCTGC | TTCGCTGACCGGCGAGTAGT | [21] | Lob et al., 2015 [55] | |
| GGTTATGCGTTATATTCGCC | TTAGCGTTGCCAGTGCTC | [30] | Christophy et al., 2017 [44] | |
| ATGCGTTATWTTCGCCTGTGT | TTAGCGTTGGCAGTGCTCG | [25] | El-Shazly et al., 2015 [56] | |
| TCGCCTGTGTATTATCTCCC | CGCAGATAAATCACCACAATG | [29] | Van et al., 2008 [57] | |
| blaCTX-M | ATGTGCAGYACCAGTAARGTKATGGC | TGGGTRAARTARGTSACCAGAAYCAGCGG | [13,15,25,26,29,35,38] | Darwich et al., 2019 [2], Archambault et al., 2006 [58], Ahmed et al., 2013 [59], Su et al., 2022 [26], Gundran et al., 2019 [60], Fang et al., 2008 [49], Hasman et al., 2005 [50] |
| ATGTGCAGYACCAGTAARGT | TGGGTRAARTARGTSACCAGA | [30] | Christophy et al., 2017 [44] | |
| CGATGTGCAGTACCAGTAA | TTAGTGACCAGAATCAGCGG | [38] | Batchelor et al., 2005 [61] | |
| TTTGCGATGTGCAGTACCAGTAA | CGATATCGTTGGTGGTGCCATA | [17,32] | Edelstein et al., 2003 [62] | |
| CGCTTTGCGATGTGCAG | ACCGCGATATCGTTGGT | [10,18] | Ali et al., 2017 [51], Grobner et al., 2009 [45] | |
| CCCATGGTTAAAAAACACTGC | CAGCGCTTTTGCCGTCTAAG | [23] | Horton et al., 2011 [63] | |
| ATGAGYACCAGTAARGTKATGGC | ATCACKCGGRTCGCCIGGRAT | [28] | Hosu et al., 2021 [47] | |
| blaCTX-M group 1 | AAAAATCACTGCGCCAGTTC | AGCTTATTCATCGCCACGTT | [11,21,24,32,34] | Woodford et al., 2006 [64] |
| TTAGGAARTGTGCCGCTGYA | CGATATCGTTGGTGGTRCCAT | [19,38] | Dallenne et al., 2010 [39] | |
| GTTCGTCTCTTCCAGAATAAGG | CAGCACTTTTGCCGTCTAAG | [18] | Pfeifer et al., 2009 [65] | |
| blaCTX-M group 2 | CGACGCTACCCCTGCTATT | CCAGCGTCAGATTTTTCAGG | [11,21,24,32,34] | Woodford et al., 2006 [64] |
| CGTTAACGGCACGATGAC | CGATATCGTTGGTGGTRCCAT | [19,38] | Dallenne et al., 2010 [39] | |
| blaCTX-M group 9 | CAAAGAGAGTGCAACGGATG | ATTGGAAAGCGTTCATCACC | [11,21,24,32,34] | Woodford et al., 2006 [64] |
| TCAAGCCTGCCGATCTGGT | TGATTCTCGCCGCTGAAG | [19,38] | Dallenne et al., 2010 [39] | |
| ACACGGATTGACCGTATTGG | TGATTCTCGCCGCTGAAG | [18] | Wetzker et al., 2019 [66] | |
| GCAGTACAGCGACAATACCG | TATCATTGGTGGTGCCGTAG | [18] | Grobner et al., 2009 [45] | |
| blaCTX-M group 8 | TCGCGTTAAGCGGATGATGC | AACCCACGATGTGGGTAGC | [11,21,24,32,34] | Woodford et al., 2006 [64] |
| AACRCRCAGACGCTCTAC | TCGAGCCGGAASGTGTYAT | [38] | Dallenne et al., 2010 [39] | |
| blaCTX-M group 25 | GCACGATGACATTCGGG | AACCCACGATGTGGGTAGC | [11,21,24,32,34] | Woodford et al., 2006 [64] |
| AACRCRCAGACGCTCTAC | TCGAGCCGGAASGTGTYAT | [38] | Dallenne et al., 2010 [39] | |
| blaGES | AGTCGGCTAGACCGGAAAG | TTTGTCCGTGCTCAGGAT | [26] | Su et al., 2022 [26] |
| blaPER | GCTCCGATAATGAAAGCG | TTCGGCTTGACTCGGCTGA | [26] | Su et al., 2022 [26] |
| blaVEB | CATTTCCCGATGCAAAGCGT | CGAAGTTTCTTTGGACTCTG | [26] | Su et al., 2022 [26] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseng, C.-H.; Liu, C.-W.; Liu, P.-Y. Extended-Spectrum β-Lactamases (ESBL) Producing Bacteria in Animals. Antibiotics 2023, 12, 661. https://doi.org/10.3390/antibiotics12040661
Tseng C-H, Liu C-W, Liu P-Y. Extended-Spectrum β-Lactamases (ESBL) Producing Bacteria in Animals. Antibiotics. 2023; 12(4):661. https://doi.org/10.3390/antibiotics12040661
Chicago/Turabian StyleTseng, Chien-Hao, Chia-Wei Liu, and Po-Yu Liu. 2023. "Extended-Spectrum β-Lactamases (ESBL) Producing Bacteria in Animals" Antibiotics 12, no. 4: 661. https://doi.org/10.3390/antibiotics12040661
APA StyleTseng, C.-H., Liu, C.-W., & Liu, P.-Y. (2023). Extended-Spectrum β-Lactamases (ESBL) Producing Bacteria in Animals. Antibiotics, 12(4), 661. https://doi.org/10.3390/antibiotics12040661







