Dosage Forms Suitability in Pediatrics: Acceptability of Antibiotics in a German Hospital
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Subjects
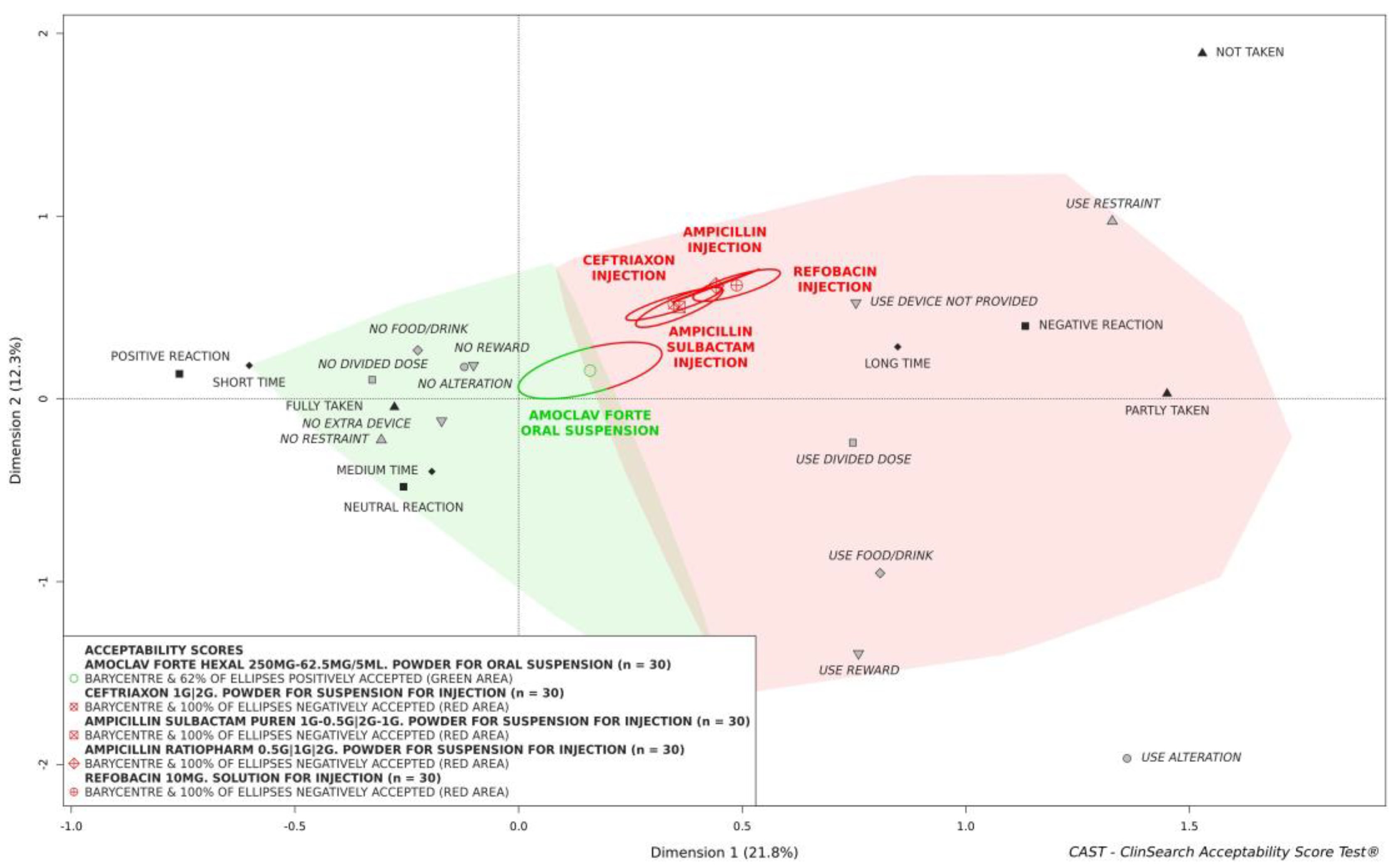
- Amoclav forte (co-amoxiclav) 250–62.5 mg/5 mL. Powder for oral suspension (Hexal®, Holzkirchen, Germany).
- Ceftriaxone 1 g (Hexal®, Holzkirchen, Germany) or 2 g (Hikma®, London, UK). Powder for suspension for injection.
- Ampicillin/sulbactam 1–0.5 g or 2–1 g. Powder for suspension for injection (Puren®).
- Ampicillin 0.5 g, 1 g or 2 g. Powder for suspension for injection (Ratiopharm®, Ulm, Germany).
- Refobacin (Gentamicin) 10 mg. Solution for injection (Merck®, Darmstadt, Germany).
2.3. Data Collection
2.4. Data Analysis
3. Results
3.1. Study Subject
3.2. Acceptability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EMA. Guideline on Pharmaceutical Development of Medicines for Paediatric Use; European Medicines Agency (EMA) CfMPfHU: London, UK, 2013. [Google Scholar]
- EMA. The European Paediatric Initiative: History of the Paediatric Regulation; EMA: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Klingmann, V.; Linderskamp, H.; Meissner, T.; Mayatepek, E.; Moeltner, A.; Breitkreutz, J.; Bosse, H.M. Acceptability of Multiple Uncoated Minitablets in Infants and Toddlers: A Randomized Controlled Trial. J. Pediatr. 2018, 201, 202–207.e201. [Google Scholar] [CrossRef]
- Münch, J.; Meissner, T.; Mayatepek, E.; Wargenau, M.; Breitkreutz, J.; Bosse, H.M.; Klingmann, V. Acceptability of Small-Sized Oblong tablets in Comparison to Syrup and Mini-tablets in Infants and Toddlers: A Randomized Controlled Trial. Eur. J. Pharm. Biopharm. 2021, 166, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Vallet, T.; Bensouda, Y.; Saito, J.; Mathiesen, L.; Pokharkar, V.; Klingmann, V.; Peak, M.; Elhamdaoui, O.; Yamatani, A.; Ivanovic, I.; et al. Exploring Acceptability Drivers of Oral Antibiotics in Children: Findings from an International Observational Study. Pharmaceutics 2021, 13, 1721. [Google Scholar] [CrossRef] [PubMed]
- Klingmann, V.; Vallet, T.; Münch, J.; Stegemann, R.; Wolters, L.; Bosse, H.-M.; Ruiz, F. Dosage Forms Suitability in Pediatrics: Acceptability of Analgesics and Antipyretics in a German Hospital. Pharmaceutics 2022, 14, 337. [Google Scholar] [CrossRef]
- Li, G.; Jackson, C.; Bielicki, J.; Ellis, S.; Hsia, Y.; Sharland, M. Global sales of oral antibiotics formulated for children. Bull. World Health Organ. 2020, 98, 458–466. [Google Scholar] [CrossRef]
- WHO. Global Action Plan on Antimicrobial Resistance; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Tuleu, C.; Hughes, D.A.; Clapham, D.; Vallet, T.; Ruiz, F. Acceptability of generic versus innovator oral medicines: Not only a matter of taste. Drug Discov. Today 2020, 26, 329–343. [Google Scholar] [CrossRef]
- Ruiz, F.; Nunn, A.; Gill, A.; Clapham, D.; Fotaki, N.; Salunke, S.; Cram, A.; O’Brien, F. A review of paediatric injectable drug delivery to inform the study of product acceptability–An introduction. Eur. J. Pharm. Biopharm. 2023, 188, 265–270. [Google Scholar] [CrossRef]
- Ruiz, F.; Vallet, T.; Pense-Lheritier, A.M.; Aoussat, A. Standardized method to assess medicines’ acceptability: Focus on paediatric population. J. Pharm. Pharmacol. 2017, 69, 406–416. [Google Scholar] [CrossRef]
- European Medicines Agency. Letter of Support for an Acceptability Score Test in Relative Acceptability Testing for Oral Medicines in Children under 12 Years of Age. Available online: https://www.ema.europa.eu/en/documents/other/letter-support-acceptability-score-test-relative-acceptability-testing-oral-medicines-children-under_en.pdf (accessed on 3 February 2023).
- Le, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Josse, J.; Husson, F. missMDA: A package for handling missing values in multivariate data analysis. J. Stat. Softw. 2016, 70, 1–31. [Google Scholar] [CrossRef]
- Vallet, T.; Elhamdaoui, O.; Berraho, A.; Cherkaoui, L.O.; Kriouile, Y.; Mahraoui, C.; Mouane, N.; Pense-Lheritier, A.-M.; Ruiz, F.; Bensouda, Y. Medicines Acceptability in Hospitalized Children: An Ongoing Need for Age-Appropriate Formulations. Pharmaceutics 2020, 12, 766. [Google Scholar] [CrossRef]
- Leung, A.K.; Wong, A.H. Acute otitis media in children. Recent. Pat. Inflamm. Allergy Drug Discov. 2017, 11, 32–40. [Google Scholar] [CrossRef]
- Youngster, I.; Avorn, J.; Belleudi, V.; Cantarutti, A.; Díez-Domingo, J.; Kirchmayer, U.; Park, B.-J.; Peiró, S.; Sanfélix-Gimeno, G.; Schröder, H. Antibiotic use in children–a cross-national analysis of 6 countries. J. Pediatr. 2017, 182, 239–244.e231. [Google Scholar] [CrossRef] [PubMed]
- Pottegård, A.; Broe, A.; Aabenhus, R.; Bjerrum, L.; Hallas, J.; Damkier, P. Use of antibiotics in children: A Danish nationwide drug utilization study. Pediatr. Infect. Dis. J. 2015, 34, e16–e22. [Google Scholar] [CrossRef] [PubMed]
- Hsia, Y.; Sharland, M.; Jackson, C.; Wong, I.C.; Magrini, N.; Bielicki, J.A. Consumption of oral antibiotic formulations for young children according to the WHO Access, Watch, Reserve (AWaRe) antibiotic groups: An analysis of sales data from 70 middle-income and high-income countries. Lancet Infect. Dis. 2019, 19, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Mistry, P.; Stirling, H.; Callens, C.; Hodson, J.; Batchelor, H. Evaluation of patient-reported outcome measurements as a reliable tool to measure acceptability of the taste of paediatric medicines in an inpatient paediatric population. BMJ Open 2018, 8, e021961. [Google Scholar] [CrossRef] [PubMed]
- Wollner, A.; Lecuyer, A.; De La Rocque, F.; Sedletzki, G.; Derkx, V.; Boucherat, M.; Elbez, A.; Gelbert-Baudino, N.; Levy, C.; Corrard, F.; et al. Acceptability, compliance and schedule of administration of oral antibiotics in outpatient children. Arch. Pediatr. 2011, 18, 611–616. [Google Scholar] [CrossRef]
- Matsui, D.; Lim, R.; Tschen, T.; Rieder, M.J. Assessment of the Palatability of β-Lactamase—Resistant Antibiotics in Children. Arch. Pediatr. Adolesc. Med. 1997, 151, 599–602. [Google Scholar] [CrossRef]
- Powers, J.L.; Gooch, W.M., 3rd; Oddo, L.P. Comparison of the palatability of the oral suspension of cefdinir vs. amoxicillin/clavulanate potassium, cefprozil and azithromycin in pediatric patients. Pediatr. Infect. Dis. J. 2000, 19, S174–S180. [Google Scholar] [CrossRef]
- Gooch, W.; Philips, A.; Rhoades, R.; Rosenberg, R.; Schaten, R.; Starobin, S. Comparison of the efficacy, safety and acceptability of cefixime and amoxicillin/clavulanate in acute otitis media. Pediatr. Infect. Dis. J. 1997, 16, S21–S24. [Google Scholar] [CrossRef]
- Toscani, M.; Drehobl, M.; Freed, J.; Stool, S. A multicenter, randomized, comparative assessment in healthy pediatric volunteers of the palatability of oral antibiotics effective in the therapy of otitis media. Curr. Ther. Res. 2000, 61, 278–285. [Google Scholar] [CrossRef]
- Angelilli, M.L.; Toscani, M.; Matsui, D.M.; Rieder, M.J. Palatability of oral antibiotics among children in an urban primary care center. Arch. Pediatr. Adolesc. Med. 2000, 154, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Ubaka, C.M.; Udeogaranya, O.P.; Ezeugwu, N. Palatability of oral paediatric antibiotics commonly prescribed in a Nigerian specialist hospital. Eur. J. Hosp. Pharm.: Sci. Pract. 2013, 20, 122–124. [Google Scholar] [CrossRef]
- Orenius, T.; LicPsych; Säilä, H.; Mikola, K.; Ristolainen, L. Fear of injections and needle phobia among children and adolescents: An overview of psychological, behavioral, and contextual factors. SAGE Open Nurs. 2018, 4, 2377960818759442. [Google Scholar] [CrossRef] [PubMed]
- Kozarewicz, P. Regulatory perspectives on acceptability testing of dosage forms in children. Int. J. Pharm. 2014, 469, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Usach, I.; Martinez, R.; Festini, T.; Peris, J.-E. Subcutaneous injection of drugs: Literature review of factors influencing pain sensation at the injection site. Adv. Ther. 2019, 36, 2986–2996. [Google Scholar] [CrossRef]
| Observational Variables | Observed Measures (Categories) |
|---|---|
| Result intake The results of intake of the required dose | Fully, Partly, or Not taken |
| Patient reaction The patient’s reaction to the intake on a 3-point facial hedonic scale | Positive, Neutral, or Negative reaction |
| Preparation and administration time The time hospital staff needed to prepare (from opening the packaging to having the required dose of medication ready to use, including all handling and modifications) and administer the required dose of medication (from the required dose of medication ready to use to the end of the intake), pooled and recoded | Short (≤1′), Medium (from 1′ to 2′30″), or Long (>2′30″) time |
| Divided dose Dividing the intake of a required dose which cannot be taken as a whole (e.g., successive sips of an oral liquid preparation, several tablets or pieces of tablets swallowed successively) | No divided dose, or Use divided dose |
| Food/drink Using food/drink (e.g., mixing with the drug or taking before/after to mask the taste or ease swallowing) | No food/drink, or Use food/drink |
| Alteration Altering the use, such as modifying the dosage form (e.g., prescribed dose of tablet split into fractions or crushed into powder) or using another route/mode of administration (e.g., oral administration of an injectable solution) | No alteration, or Use alteration |
| Extra device Using a device not provided with the medication (e.g., disposable spoon or oral syringe provided with another medication) | No extra device, or Use extra device |
| Reward Promising a reward | No reward, or Use reward |
| Restraint The child was made to take it | No restraint, or Use restraint |
| Characteristics | Amoclav Forte 250–62.5 mg/5 mL. Powder for Oral Suspension (n = 30) | Ceftriaxone 1 g|2 g. Powder for Suspension for Injection (n = 30) | Ampicillin/Sulbactam 1–0.5 g|2 g–1 g. Powder for Suspension for Injection (n = 30) | Ampicillin 0.5 g|1 g|2 g. Powder for Suspension for Injection (n = 30) | Refobacin 10 mg. Solution for Injection (n = 30) |
|---|---|---|---|---|---|
| Sex | |||||
| Female | 16 (53) a | 18 (60) | 16 (53) | 9 (30) | 9 (30) |
| Male | 14 (47) | 12 (40) | 14 (47) | 21 (70) | 21 (70) |
| Age group | |||||
| 0–2 years | 17 (57) | 13 (43) | 15 (50) | 24 (80) | 24 (80) |
| 3–5 years | 7 (23) | 4 (13) | 4 (13) | 3 (10) | 2 (7) |
| 6–11 years | 3 (10) | 5 (17) | 2 (7) | 1 (3) | 2 (7) |
| 12–17 years | 3 (10) | 8 (27) | 9 (30) | 2 (7) | 2 (7) |
| Parents Geographical regions of origin | |||||
| Western Europe | 9 (30) | 13 (43) | 11 (37) | 11 (37) | 13 (43) |
| Central and Eastern Europe | 7 (23) | 4 (13) | 7 (23) | 6 (20) | 6 (20) |
| Mixed origin including Western | 5 (17) | 5 (17) | 4 (13) | 4 (13) | 3 (10) |
| Europe | 3 (10) | 4 (13) | 1 (3) | 4 (13) | 2 (7) |
| Western Europe (no information for other parent) | 3 (10) | 1 (3) | 2 (7) | 1 (3) | 1 (3) |
| Other <5% b | 3 (10) | 3 (10) | 5 (17) | 4 (13) | 5 (17) |
| Person in charge of the medicine administration | |||||
| The patient | 6 (20) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| A caregiver c | 15 (50) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| A healthcare professional | 9 (30) | 30 (100) | 30 (100) | 30 (100) | 30 (100) |
| Observer-Reported Outcomes | Amoclav Forte 250–62.5 mg/5 mL. Powder for Oral Suspension (n = 30) | Ceftriaxone 1 g|2 g. Powder for Suspension for Injection (n = 30) | Ampicillin/Sulbactam 1–0.5 g|2–1 g. Powder for Suspension for Injection (n = 30) | Ampicillin 0.5 g|1 g|2 g. Powder for Suspension for Injection (n = 30) | Refobacin 10 mg. Solution for Injection (n = 30) |
|---|---|---|---|---|---|
| Result intake | |||||
| Fully taken | 28 (93) a | 30 (100) | 30 (100) | 30 (100) | 29 (97) |
| Partly taken | 1 (3) | 0 (0) | 0 (0) | 0 (0) | 1 (3) |
| Not taken | 1 (3) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Patient reaction | |||||
| Positive | 5 (17) | 1 (3) | 0 (0) | 0 (0) | 0 (0) |
| Neutral | 13 (43) | 7 (23) | 7 (23) | 5 (17) | 4 (13) |
| Negative | 12 (40) | 22 (73) | 23 (77) | 25 (83) | 26 (87) |
| Preparation and administration time | |||||
| Short | 4 (13) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Medium | 5 (17) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Long | 21 (70) | 30 (100) | 30 (100) | 30 (100) | 30 (100) |
| Divided dose | |||||
| No divided dose | 24 (80) | 30 (100) | 30 (100) | 30 (100) | 29 (97) |
| Use divided dose | 6 (20) | 0 (0) | 0 (0) | 0 (0) | 1 (3) |
| Food/drink | |||||
| No food/drink | 24 (80) | 30 (100) | 30 (100) | 30 (100) | 30 (100) |
| Use food/drink | 6 (20) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Alteration | |||||
| No alteration | 26 (87) | 30 (100) | 30 (100) | 30 (100) | 30 (100) |
| Use alteration | 4 (13) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Extra device | |||||
| No extra device | 16 (53) | 0 (0) | 1 (3) | 0 (0) | 0 (0) |
| Use extra device | 14 (47) | 30 (100) | 29 (97) | 30 (100) | 30 (100) |
| Reward | |||||
| No reward | 30 (100) | 29 (97) | 28 (93) | 30 (100) | 29 (97) |
| Use reward | 0 (0) | 1 (3) | 2 (7) | 0 (0) | 1 (3) |
| Restraint | |||||
| No restraint | 22 (73) | 18 (60) | 18 (60) | 12 (40) | 11 (37) |
| Use restraint | 8 (27) | 12 (40) | 12 (40) | 18 (60) | 19 (63) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klingmann, V.; Vallet, T.; Münch, J.; Wolters, L.; Stegemann, R.; Bosse, H.M.; Ruiz, F. Dosage Forms Suitability in Pediatrics: Acceptability of Antibiotics in a German Hospital. Antibiotics 2023, 12, 1709. https://doi.org/10.3390/antibiotics12121709
Klingmann V, Vallet T, Münch J, Wolters L, Stegemann R, Bosse HM, Ruiz F. Dosage Forms Suitability in Pediatrics: Acceptability of Antibiotics in a German Hospital. Antibiotics. 2023; 12(12):1709. https://doi.org/10.3390/antibiotics12121709
Chicago/Turabian StyleKlingmann, Viviane, Thibault Vallet, Juliane Münch, Lena Wolters, Robin Stegemann, Hans Martin Bosse, and Fabrice Ruiz. 2023. "Dosage Forms Suitability in Pediatrics: Acceptability of Antibiotics in a German Hospital" Antibiotics 12, no. 12: 1709. https://doi.org/10.3390/antibiotics12121709
APA StyleKlingmann, V., Vallet, T., Münch, J., Wolters, L., Stegemann, R., Bosse, H. M., & Ruiz, F. (2023). Dosage Forms Suitability in Pediatrics: Acceptability of Antibiotics in a German Hospital. Antibiotics, 12(12), 1709. https://doi.org/10.3390/antibiotics12121709






