Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial
Abstract
:1. Introduction
2. Results
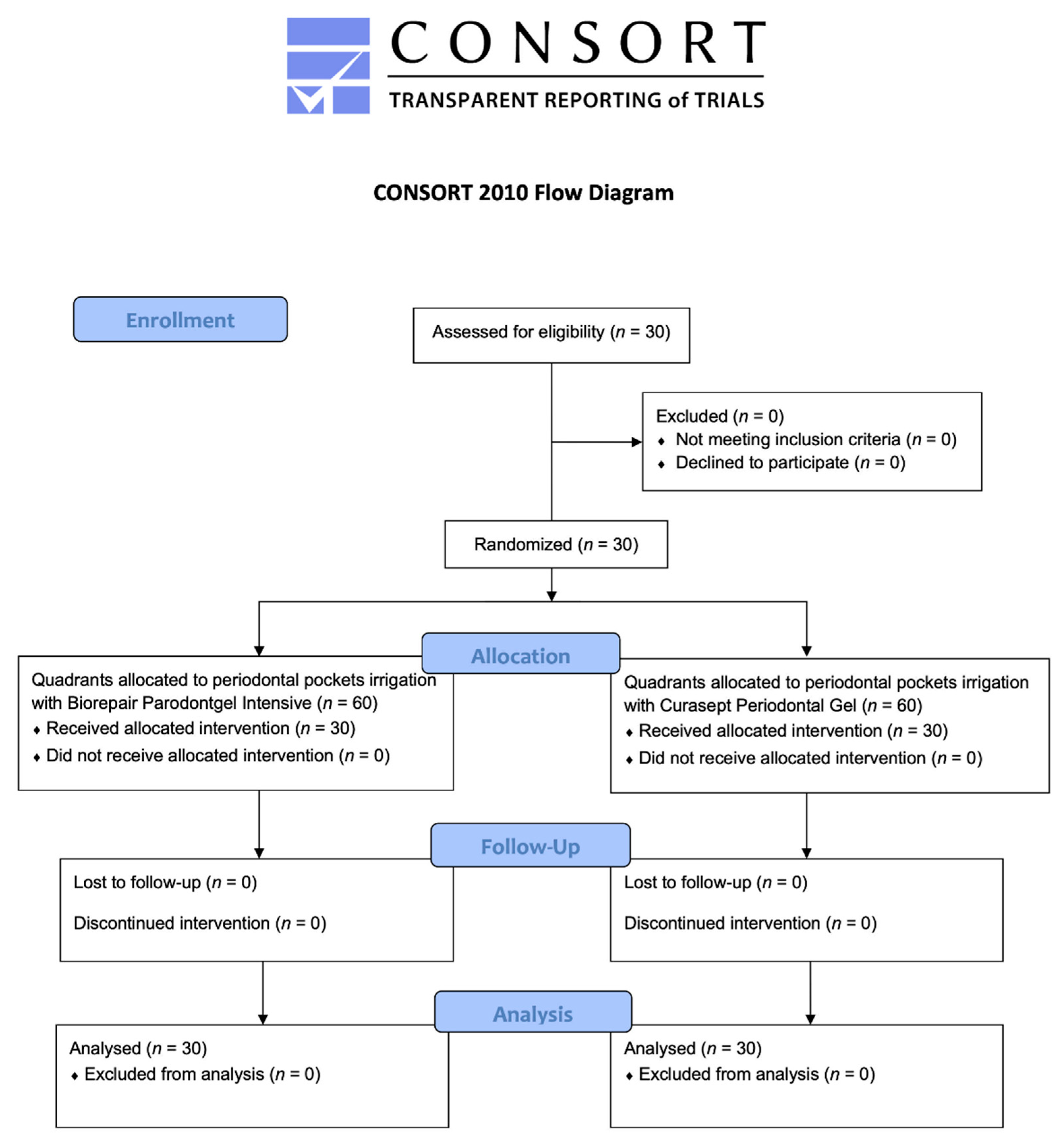
2.1. Participant Flow and Baseline Data
2.2. Probing Pocket Depth (PPD)
2.3. Recession (R)
2.4. Dental Mobility
2.5. Bleeding on Probing (BoP%)
2.6. Plaque Control Record (PCR%)
3. Discussion
4. Materials and Methods
4.1. Trial Design
4.2. Participants
4.3. Interventions and Outcomes
4.4. Sample Size
4.5. Randomization and Blinding
4.6. Statistical Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baelum, V.; Lopez, R. Periodontal disease epidemiology—Learned and unlearned? Periodontol. 2000 2013, 62, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Preshaw, P.M.; Alba, A.L.; Herrera, D.; Jepsen, S.; Konstantinidis, A.; Makrilakis, K.; Taylor, R. Periodontitis and diabetes: A two-way relationship. Diabetologia 2012, 55, 21–31. [Google Scholar] [CrossRef] [Green Version]
- Kinane, D.F.; Chestnutt, I.G. Smoking and periodontal disease. Crit. Rev. Oral Biol. Med. 2000, 11, 356–365. [Google Scholar] [CrossRef] [Green Version]
- Wilton, J.M.; Griffiths, G.S.; Curtis, M.A.; Maiden, M.F.; Gillett, I.R.; Wilson, D.T.; Sterne, J.A.; Johnson, N.W. Detection of high-risk groups and individuals for periodontal diseases. Systemic predisposition and markers of general health. J. Clin. Periodontol. 1988, 15, 339–346. [Google Scholar] [CrossRef]
- Barr, C.; Lopez, M.R.; Rua-Dobles, A. Periodontal changes by HIV serostatus in a cohort of homosexual and bisexual men. J. Clin. Periodontol. 1992, 19, 794–801. [Google Scholar] [CrossRef]
- Shapira, L.; Wilensky, A.; Kinane, D.F. Effect of genetic variability on the inflammatory response to periodontal infection. J. Clin. Periodontol. 2005, 32, 72–86. [Google Scholar] [CrossRef]
- Marsh, P.D. Dental plaque: Biological significance of a biofilm and community life-style. J. Clin. Periodontol. 2005, 32, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Berezow, A.B.; Darveau, R.P. Microbial shift and periodontitis. Periodontol. 2000 2011, 55, 36–47. [Google Scholar] [CrossRef]
- Mombelli, A. Microbial colonization of the periodontal pocket and its significance for periodontal therapy. Periodontol. 2000 2018, 76, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Feres, M. Antibiotics in the treatment of periodontal diseases: Microbiological basis and clinical applications. Ann. R Australas. Coll. Dent. Surg. 2008, 19, 37–44. [Google Scholar]
- Meimandi, M.; Talebi Ardakani, M.R.; Esmaeil Nejad, A.; Yousefnejad, P.; Saebi, K.; Tayeed, M.H. The Effect of Photodynamic Therapy in the Treatment of Chronic Periodontitis: A Review of Literature. J. Lasers Med. Sci. 2017, 8, S7–S11. [Google Scholar] [CrossRef] [PubMed]
- Butera, A.; Gallo, S.; Pascadopoli, M.; Luraghi, G.; Scribante, A. Ozonized Water Administration in Peri-Implant Mucositis Sites: A Randomized Clinical Trial. Appl. Sci. 2021, 11, 7812. [Google Scholar] [CrossRef]
- Invernici, M.M.; Salvador, S.L.; Silva, P.; Soares, M.; Casarin, R.; Palioto, D.B.; Souza, S.; Taba, M., Jr.; Novaes, A.B., Jr.; Furlaneto, F.; et al. Effects of Bifidobacterium probiotic on the treatment of chronic periodontitis: A randomized clinical trial. J. Clin. Periodontol. 2018, 45, 1198–1210. [Google Scholar] [CrossRef] [Green Version]
- Butera, A.; Pascadopoli, M.; Gallo, S.; Lelli, M.; Tarterini, F.; Giglia, F.; Scribante, A. SEM/EDS Evaluation of the Mineral Deposition on a Polymeric Composite Resin of a Toothpaste Containing Biomimetic Zn-Carbonate Hydroxyapatite (microRepair®) in Oral Environment: A Randomized Clinical Trial. Polymers 2021, 13, 2740. [Google Scholar] [CrossRef] [PubMed]
- Francino, M. Antibiotics and the human gut microbiome: Dysbioses and accumulation of resistances. Front. Microbiol. 2016, 6, 1543–1545. [Google Scholar] [CrossRef] [Green Version]
- Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Working Group. Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food; World Health Organization: London, ON, Canada, 2002. [Google Scholar]
- Ince, G.; Gürsoy, H.; Ipçi, S.D.; Cakar, G.; Emekli-Alturfan, E.; Yılmaz, S. Clinical and Biochemical Evaluation of Lozenges Containing Lactobacillus reuteri as an Adjunct to Non-Surgical Periodontal Therapy in Chronic Periodontitis. J. Periodontol. 2015, 86, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics-A Step Beyond Pre- and Probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef] [PubMed]
- Piepho, H.P. An Algorithm for a Letter-Based Representation of All-Pairwise Comparisons. J. Comput. Graph. Stat. 2004, 13, 456–466. [Google Scholar] [CrossRef]
- Scribante, A.; Poggio, C.; Gallo, S.; Riva, P.; Cuocci, A.; Carbone, M.; Arciola, C.R.; Colombo, M. In Vitro Re-Hardening of Bleached Enamel Using Mineralizing Pastes: Toward Preventing Bacterial Colonization. Materials 2020, 13, 818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belibasakis, G.N.; Mylonakis, E. Oral infections: Clinical and biological perspectives. Virulence 2015, 6, 173–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lutovac, M.; Popova, O.V.; Macanovic, G.; Kristina, R.; Lutovac, B.; Ketin, S.; Biocanin, R. Testing the Effect of Aggressive Beverage on the Damage of Enamel Structure. Open Access Maced. J. Med. Sci. 2017, 5, 987–993. [Google Scholar] [CrossRef] [Green Version]
- Colombo, M.; Poggio, C.; Lasagna, A.; Chiesa, M.; Scribante, A. Vickers Micro-Hardness of New Restorative CAD/CAM Dental Materials: Evaluation and Comparison after Exposure to Acidic Drink. Materials 2019, 12, 1246. [Google Scholar] [CrossRef] [Green Version]
- Deng, Z.L.; Szafrański, S.P.; Jarek, M.; Bhuju, S.; Wagner-Döbler, I. Dysbiosis in chronic periodontitis: Key microbial players and interactions with the human host. Sci. Rep. 2017, 7, 3703. [Google Scholar] [CrossRef] [Green Version]
- Ikram, S.; Hassan, N.; Raffat, M.A.; Mirza, S.; Akram, Z. Systematic review and meta- analysis of double- blind, placebo- controlled, randomized clinical trials using probiotics in chronic periodontitis. J. Investig. Clin. Dent. 2018, 9, e12338. [Google Scholar] [CrossRef]
- Vivekananda, M.R.; Vandana, K.L.; Bhat, K.G. Effect of the probiotic Lactobacilli reuteri (Prodentis) in the management of periodontal disease: A preliminary randomized clinical trial. J. Oral Microbiol. 2010, 2, 5344. [Google Scholar] [CrossRef]
- Teughels, W.; Durukan, A.; Ozcelik, O.; Pauwels, M.; Quirynen, M.; Haytac, M.C. Clinical and microbiological effects of Lactobacillus reuteri probiotics in the treatment of chronic periodontitis: A randomized placebo- controlled study. J. Clin. Periodontol. 2013, 40, 1025–1035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szkaradkiewicz, A.K.; Stopa, J.; Karpiński, T.M. Effect of oral administration involving a probiotic strain of Lactobacillus reuteri on pro- inflammatory cytokine response in patients with chronic periodontitis. Arch. Immunol. Ther. Exp. 2014, 62, 495–500. [Google Scholar] [CrossRef] [Green Version]
- Varoni, E.; Tarce, M.; Lodi, G.; Carrassi, A. Chlorhexidine (CHX) in dentistry: State of the art. Minerva Stomatol. 2012, 61, 399–419. [Google Scholar] [PubMed]
- Ishikawa, K.H.; Bueno, M.R.; Kawamoto, D.; Simionato, M.R.L.; Mayer, M.P.A. Lactobacilli postbiotics reduce biofilm formation and alter transcription of virulence genes of Aggregatibacter actinomycetemcomitans. Mol. Oral Microbiol. 2021, 36, 92–102. [Google Scholar] [CrossRef]
- Park, E.; Ha, J.; Lim, S.; Kim, G.; Yoon, Y. Development of postbiotics by whey bioconversion with Enterococcus faecalis M157 KACC81148BP and Lactococcus lactis CAU2013 KACC81152BP for treating periodontal disease and improving gut health. J. Dairy Sci. 2021, 104, 12321–12331. [Google Scholar] [CrossRef] [PubMed]
- Izuddin, W.I.; Humam, A.M.; Loh, T.C.; Foo, H.L.; Samsudin, A.A. Dietary Postbiotic Lactobacillus plantarum Improves Serum and Ruminal Antioxidant Activity and Upregulates Hepatic Antioxidant Enzymes and Ruminal Barrier Function in Post-Weaning Lambs. Antioxidants 2020, 9, 250. [Google Scholar] [CrossRef] [Green Version]
- Osman, A.; El-Gazzar, N.; Almanaa, T.N.; El-Hadary, A.; Sitohy, M. Lipolytic Postbiotic from Lactobacillus paracasei Manages Metabolic Syndrome in Albino Wistar Rats. Molecules 2021, 26, 472. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Maiorani, C.; Preda, C.; Chiesa, A.; Esposito, F.; Pascadopoli, M.; Scribante, A. Management of Gingival Bleeding in Periodontal Patients with Domiciliary Use of Toothpastes Containing Hyaluronic Acid, Lactoferrin, or Paraprobiotics: A Randomized Controlled Clinical Trial. Appl. Sci. 2021, 11, 8586. [Google Scholar] [CrossRef]
- Yadav, N.; Lamba, A.K.; Thakur, A.; Faraz, F.; Tandon, S.; Pahwa, P. Effect of periodontal therapy on lactoferrin levels in gingival crevicular fluid. Aust. Dent. J. 2014, 59, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Berlutti, F.; Pilloni, A.; Pietropaoli, M.; Polimeni, A.; Valenti, P. Lactoferrin and oral diseases: Current status and perspective in periodontitis. Ann. Stomatol. 2011, 2, 10–18. [Google Scholar]
- Yeturu, S.K.; Acharya, S.; Urala, A.S.; Pentapati, K.C. Effect of Aloe vera, chlorine dioxide, and chlorhexidine mouth rinses on plaque and gingivitis: A randomized controlled trial. J. Oral Biol. Craniofac. Res. 2016, 6, 55–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moghaddam, A.A.; Radafshar, G.; Jahandideh, Y.; Kakaei, N. Clinical evaluation of effects of local application of Aloe vera gel as an adjunct to scaling and root planning in patients with chronic periodontitis. J. Dent. 2017, 18, 165–172. [Google Scholar]
- Ipshita, S.; Kurian, I.G.; Dileep, P.; Kumar, S.; Singh, P.; Pradeep, A.R. One percent alendronate and Aloe vera gel local host modulating agents in chronic periodontitis patients with class II furcation defects: A randomized, controlled clinical trial. J. Investig. Clin. Dent. 2018, 9, e12334. [Google Scholar] [CrossRef]
- Kurian, I.G.; Dileep, P.; Ipshita, S.; Pradeep, A.R. Comparative evaluation of subgingivally-delivered 1% metformin and Aloe vera gel in the treatment of intrabony defects in chronic periodontitis patients: A randomized, controlled clinical trial. J. Investig. Clin. Dent. 2018, 9, e12324. [Google Scholar] [CrossRef]
- Sánchez, M.; González-Burgos, E.; Iglesias, I.; Gómez-Serranillos, M.P. Pharmacological Update Properties of Aloe vera and its Major Active Constituents. Molecules 2020, 25, 1324. [Google Scholar] [CrossRef] [Green Version]
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Figuero, E.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S286–S291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lang, N.P.; Joss, A.; Orsanic, T.; Gusberti, F.A.; Siegrist, B.E. Bleeding on probing. A predictor for the progression of periodontal disease? J. Clin. Periodontol. 1986, 13, 590–596. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.J.; Drake, R.B.; Naylor, J.E. The Plaque Control Record. J. Periodontol. 1972, 43, 38. [Google Scholar] [CrossRef]
- Purkait, S.; Bandyopadhyaya, P.; Mallick, B.; Das, I. Classification of tooth mobility—Concept Revisited. Int. J. Rec. Adv. Multidiscip. Res. 2016, 3, 1510–1522. [Google Scholar]
- Sreedhar, A.; Sarkar, I.; Rajan, P.; Pai, J.; Malagi, S.; Kamath, V.; Barmappa, R. Comparative evaluation of the efficacy of curcumin gel with and without photo activation as an adjunct to scaling and root planing in the treatment of chronic periodontitis: A split mouth clinical and microbiological study. J. Nat. Sci. Biol. Med. 2015, 6, S102–S109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Group | Time | Mean (*) | St Dev | Min | Median | Max |
|---|---|---|---|---|---|---|
| Control (CHX) | T0 | 6.37 a | 1.53 | 4.00 | 6.00 | 10.00 |
| T1 | 5.17 a | 1.38 | 2.00 | 5.00 | 9.00 | |
| T2 | 4.28 a,b,c | 1.31 | 2.00 | 4.00 | 9.00 | |
| T3 | 3.87 c | 1.11 | 2.00 | 4.00 | 7.00 | |
| Test (BPI) | T0 | 6.40 a | 1.53 | 4.00 | 6.00 | 10.00 |
| T1 | 5.15 a,b | 1.44 | 3.00 | 5.00 | 9.00 | |
| T2 | 4.24 b,c | 1.30 | 2.00 | 4.00 | 9.00 | |
| T3 | 3.76 c | 1.21 | 2.00 | 4.00 | 7.00 |
| Group | Time | Mean (*) | St Dev | Min | Median | Max |
|---|---|---|---|---|---|---|
| Control (CHX) | T0 | 1.78 a | 1.55 | 0.00 | 2.00 | 6.00 |
| T1 | 2.07 a,b | 1.76 | 0.00 | 2.00 | 6.00 | |
| T2 | 2.18 a,b,c | 1.84 | 0.00 | 2.00 | 7.00 | |
| T3 | 2.15 c,d | 1.85 | 0.00 | 2.00 | 7.00 | |
| Test (BPI) | T0 | 1.54 a | 1.38 | 0.00 | 1.00 | 5.00 |
| T1 | 1.74 a,b,c | 1.44 | 0.00 | 2.00 | 6.00 | |
| T2 | 1.76 c,d | 1.49 | 0.00 | 2.00 | 6.00 | |
| T3 | 1.72 d | 1.45 | 0.00 | 2.00 | 6.00 |
| Group | Time | Mean (*) | St Dev | Min | Median | Max |
|---|---|---|---|---|---|---|
| Control (CHX) | T0 | 0.50 a | 0.80 | 0.00 | 0.00 | 3.00 |
| T1 | 0.50 a | 0.80 | 0.00 | 0.00 | 3.00 | |
| T2 | 0.38 a | 0.55 | 0.00 | 0.00 | 2.00 | |
| T3 | 0.39 a | 0.56 | 0.00 | 0.00 | 2.00 | |
| Test (BPI) | T0 | 0.47 a | 0.76 | 0.00 | 0.00 | 2.00 |
| T1 | 0.41 a | 0.67 | 0.00 | 0.00 | 2.00 | |
| T2 | 0.31 a | 0.47 | 0.00 | 0.00 | 1.00 | |
| T3 | 0.28 a | 0.46 | 0.00 | 0.00 | 1.00 |
| Group | Time | Mean (*) | St Dev | Min | Median | Max |
|---|---|---|---|---|---|---|
| Control (CHX) | T0 | 0.35 a | 0.25 | 0.08 | 0.28 | 1.00 |
| T1 | 0.20 a,b | 0.16 | 0.00 | 0.18 | 0.55 | |
| T2 | 0.14 b,c | 0.12 | 0.01 | 0.09 | 0.40 | |
| T3 | 0.12 c,d | 0.11 | 0.01 | 0.07 | 0.36 | |
| Test (BPI) | T0 | 0.36 a | 0.26 | 0.04 | 0.34 | 1.00 |
| T1 | 0.17 b,c | 0.13 | 0.00 | 0.14 | 0.48 | |
| T2 | 0.11 c,d | 0.09 | 0.01 | 0.08 | 0.37 | |
| T3 | 0.09 d | 0.08 | 0.00 | 0.05 | 0.29 |
| Group | Time | Mean (*) | St Dev | Min | Median | Max |
|---|---|---|---|---|---|---|
| Control (CHX) | T0 | 0.85 a | 0.19 | 0.36 | 0.95 | 1.00 |
| T1 | 0.52 a,b | 0.19 | 0.13 | 0.50 | 1.00 | |
| T2 | 0.40 b,c | 0.18 | 0.05 | 0.40 | 0.88 | |
| T3 | 0.30 d | 0.15 | 0.04 | 0.31 | 0.75 | |
| Test (BPI) | T0 | 0.85 a | 0.19 | 0.36 | 0.94 | 1.00 |
| T1 | 0.50 b,c | 0.17 | 0.10 | 0.50 | 0.88 | |
| T2 | 0.37 c,d | 0.18 | 0.00 | 0.41 | 0.75 | |
| T3 | 0.28 d | 0.16 | 0.06 | 0.27 | 0.70 |
| Inclusion criteria | Age between 18 and 70 years Presence of periodontal disease according to the recent 2017 classification (2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions) [42]: severity from grade I onwards (grade I: clinical attachment loss of 1–2 mm, bone loss in the cervical third greater than 15%, no elements loss for periodontitis); complexity from grade II onwards (grade II: maximum probing depth of 5 mm, bone loss mainly horizontally) Presence of bilateral periodontal probes, both to the right and to the left of the midline incisal line Electric toothbrush and a floss/brush according to the dimensions of the interdental spaces. Written informed consent to take part of the study |
| Exclusion criteria | Cardiac pacemaker Psychological, neurological or psychiatric disorders Pregnancy or breastfeeding within the last 12 months Low compliance or inconsistent motivation Use of drugs or alcohol Antibiotic therapy in the previous 6 months Presence of periodontal probes exclusively on the right or left side compared to the midline incisal line |
| Gel | Manufacturer | Composition |
|---|---|---|
| Biorepair Parodontgel Intensive | Coswell S.p.A., Funo di Argelato, BO, Italy | Aqua, Propylene Glycol, Peg-40 Hydrogenated Castor Oil, Xylitol, Xanthan Gum, Silica, Zinc Hydroxyapatite, Zinc PCA, Aloe Barbadensis Leaf Juice Powder, Lactobacillus Ferment, Sodium Hyaluronate, Lactoferrin, Solidago Virgaurea Extract, Aroma, Sodium Benzoate, Phenylpropanol, Benzyl Alcohol, Hydroxyacetophenone, Sodium Saccharin, O-Cymen-5-ol, Mannitol, Decylene Glycol, Sodium Myristoyl Sarcosinate, Sodium Methyl Cocoyl Taurate, Citric Acid, Potassium Sorbate, Phenoxyethanol, Linalool, Benzyl Benzoate, Limonene. |
| Curasept Periodontal Gel (with 1% chlorhexidine) | Curasept S.p.A, Saronno, VA, Italy | Purified water, Propylene glycol, Hydroxy Ethyl Cellulose, PVP/VA copolymer, PEG-40 hydrogenated castor oil, Chlorhexidine digluconate, Sodium acetate, Aroma, Acetic acid, Sodium metabisulfite, Ascorbic acid |
| Appointment | Procedures |
|---|---|
| Signature to the informed consent for the study | |
| Assessment of periodontal clinical indices | |
| Professional supragingival and subgingival oral hygiene | |
| Baseline (T0) | Supragingival and subgingival decontamination with glycine powders. Group 1: application in periodontal pockets of quadrants Q1 and Q3 of Biorepair Parodontgel Intensive; irrigation of periodontal pockets of quadrants Q2 and Q4 with chlorhexidine 1% gel. Group 2: irrigation of periodontal pockets of quadrants Q1 and Q3 with Curasept Periodontal Gel with 1% chlorhexidine; application in periodontal pockets of quadrants Q2 and Q4 of Biorepair Parodontgel Intensive. Domiciliary use of the two gels for the same quadrants until the 14 following days from the visit. |
| After 1 month (T1) After 3 months (T2) After 6 months (T3) | Assessment of periodontal clinical indices Professional supragingival and subgingival oral hygiene Group 1: application in periodontal pockets of quadrants Q1 and Q3 of Biorepair Parodontgel Intensive; irrigation of periodontal pockets of quadrants Q2 and Q4 with chlorhexidine 1% gel. Group 2: irrigation of periodontal pockets of quadrants Q1 and Q3 with Curasept Periodontal Gel with 1% chlorhexidine; application in periodontal pockets of quadrants Q2 and Q4 of Biorepair Parodontgel Intensive. |
| Domiciliary use of the two gels for the same quadrants until the 14 following days from the visit. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butera, A.; Gallo, S.; Pascadopoli, M.; Taccardi, D.; Scribante, A. Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial. Antibiotics 2022, 11, 118. https://doi.org/10.3390/antibiotics11010118
Butera A, Gallo S, Pascadopoli M, Taccardi D, Scribante A. Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial. Antibiotics. 2022; 11(1):118. https://doi.org/10.3390/antibiotics11010118
Chicago/Turabian StyleButera, Andrea, Simone Gallo, Maurizio Pascadopoli, Damiano Taccardi, and Andrea Scribante. 2022. "Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial" Antibiotics 11, no. 1: 118. https://doi.org/10.3390/antibiotics11010118
APA StyleButera, A., Gallo, S., Pascadopoli, M., Taccardi, D., & Scribante, A. (2022). Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial. Antibiotics, 11(1), 118. https://doi.org/10.3390/antibiotics11010118








