Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors
Abstract
:1. Introduction
2. Methods
2.1. Study Participants
2.2. Quantitative Measurements
2.2.1. Anthropometric Measurements
2.2.2. Capillary Blood Glucose and Continuous Interstitial Glucose Measurement
2.3. Assessment of Glycemic Variability
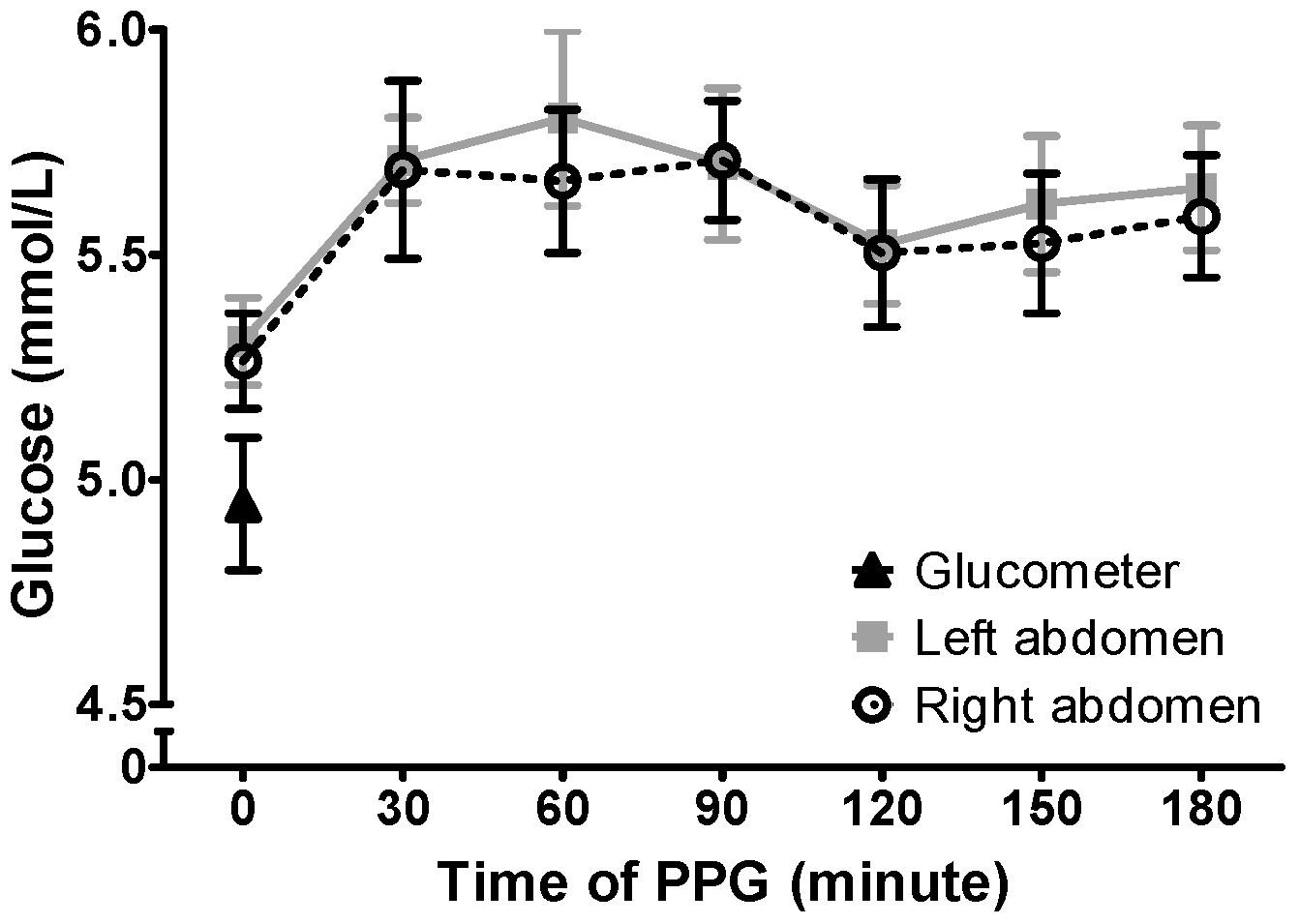
2.3.1. Postprandial Glucose (PPG) Excursion
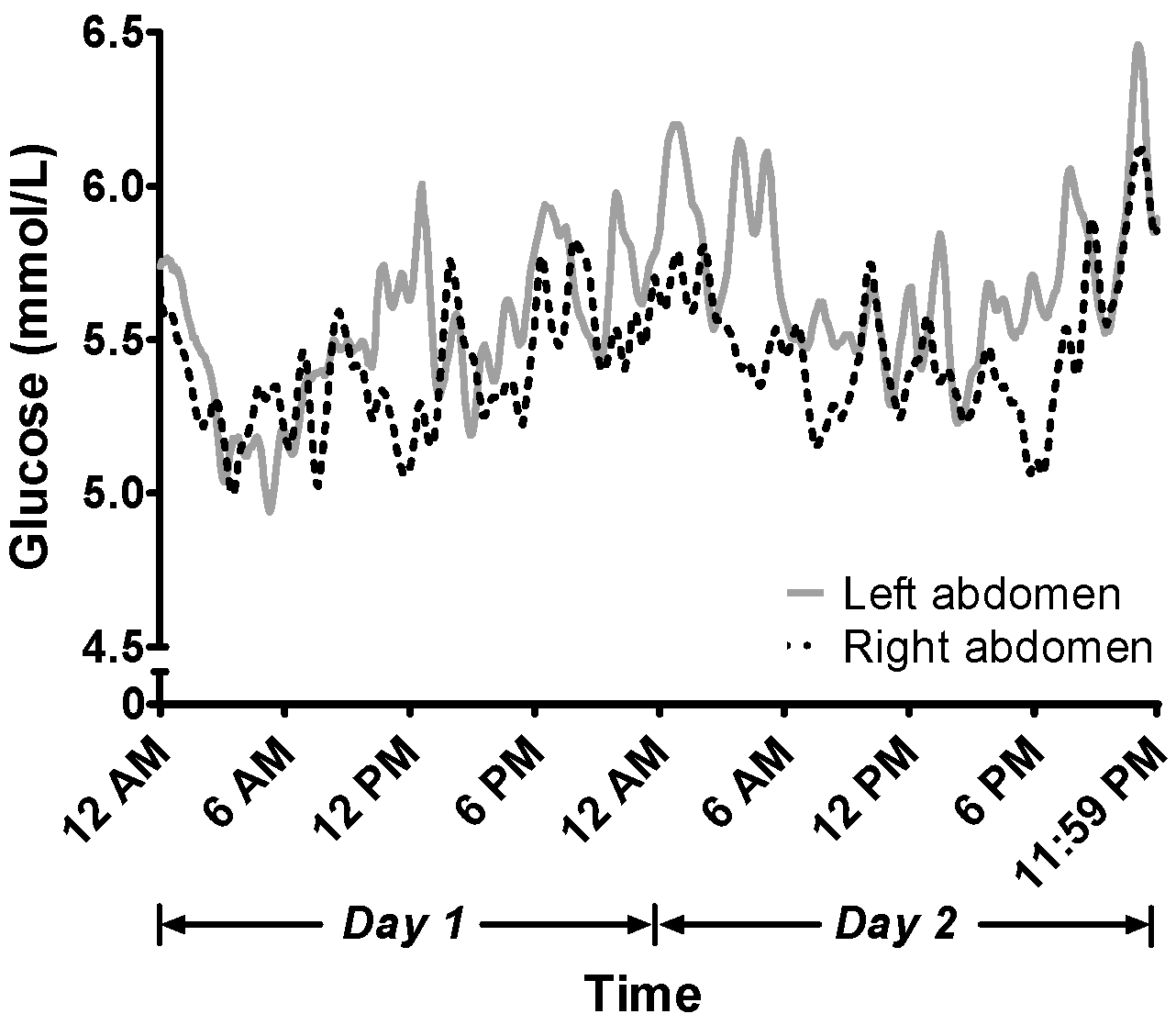
2.3.2. Dynamic, Continuous 48-h Interstitial Glucose Profile
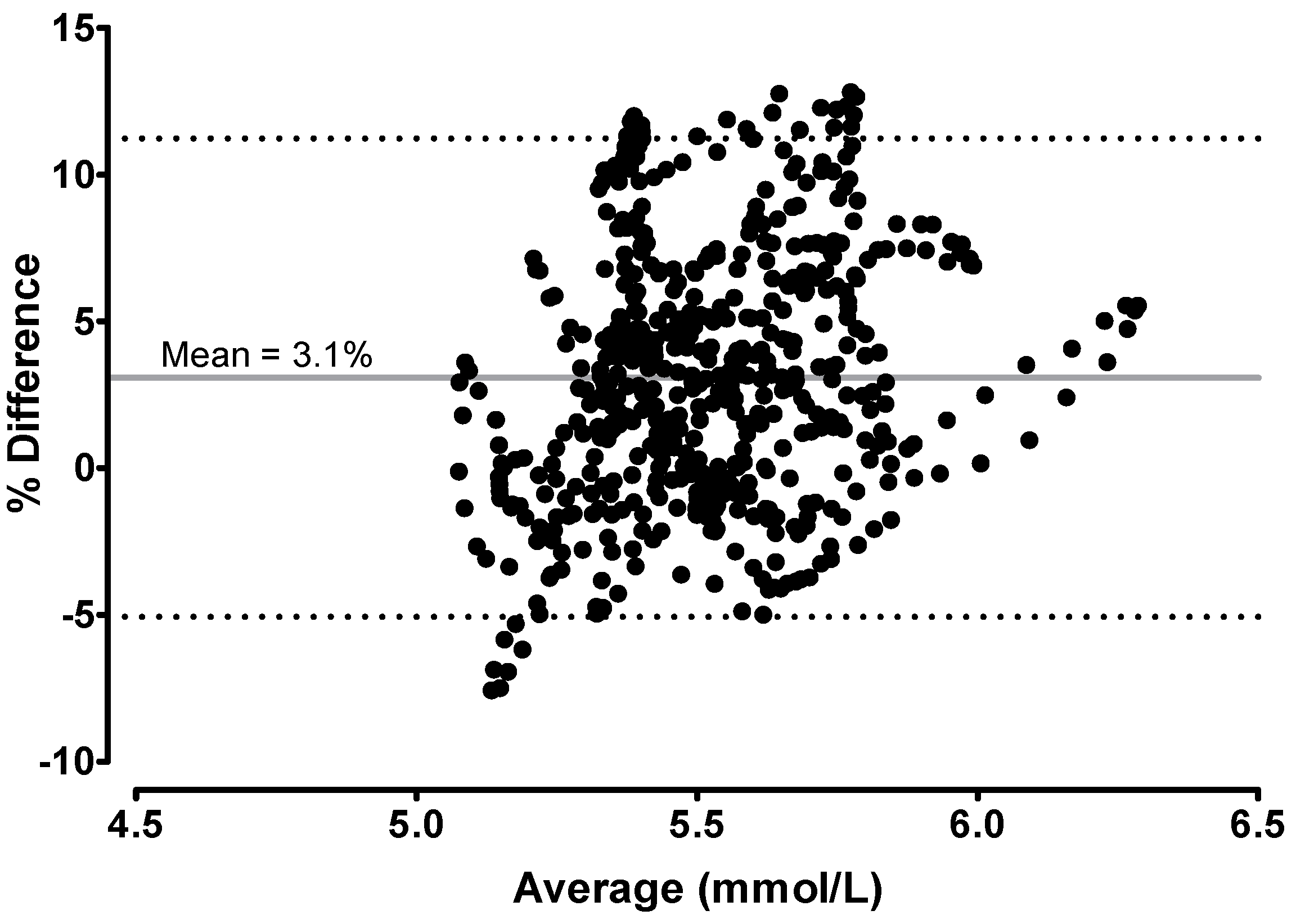
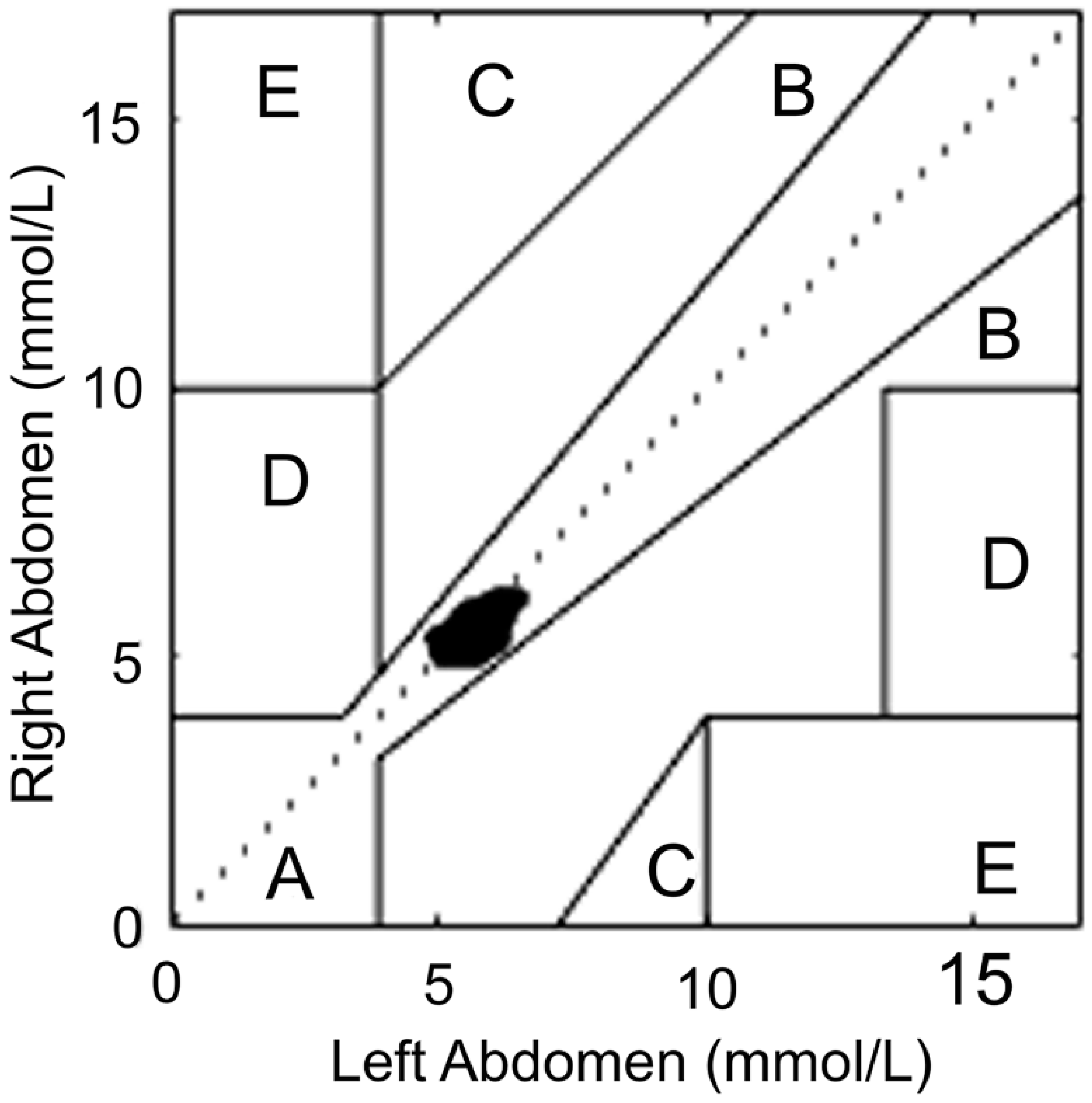
2.4. Statistical Analyses
3. Results
3.1. Postprandial Glucose Level
3.2. Continuous 48-h Glucose Level
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- US Department of Health and Human Services. Continuous Glucose Monitoring. National Institute of Diabetes and Digestice and Kidney Diseases Website. Available online: http://www.niddk.nih.gov/health-information/health-topics/Diabetes/continuous-glucose-monitoring/Pages/index.aspx (accessed on 3 July 2016).
- Deiss, D.; Bolinder, J.; Riveline, J.P.; Battelino, T.; Bosi, E.; Tubiana-Rufi, N.; Kerr, D.; Phillip, M. Improved glycemic control in poorly controlled patients with type 1 diabetes using real-time continuous glucose monitoring. Diabetes Care 2006, 29, 2730–2732. [Google Scholar] [CrossRef] [PubMed]
- Battelino, T.; Phillip, M.; Bratina, N.; Nimri, R.; Oskarsson, P.; Bolinder, J. Effect of continuous glucose monitoring on hypoglycemia in type 1 diabetes. Diabetes Care 2011, 34, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Iscoe, K.E.; Davey, R.J.; Fournier, P.A. Is the response of continuous glucose monitors to physiological changes in blood glucose levels affected by sensor life? Diabetes Technol. Ther. 2011, 14, 135–142. [Google Scholar] [CrossRef] [PubMed]
- King, A.B.; Armstrong, D.; Chu, C.; Sharma, M.; Price, D. Agreement between Glucose Trends Derived from Three Simultaneously Worn Continuous Glucose Sensors. J. Diabetes Sci. Technol. 2008, 2, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, R.L.; Schwartz, S.L.; Brazg, R.L.; Bugler, J.R.; Peyser, T.A.; McGarraugh, G.V. Accuracy of the 5-Day FreeStyle Navigator Continuous Glucose Monitoring System. Comparison with frequent laboratory reference measurements. Diabetes Care 2007, 30, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
- Medtronic MiniMed. Insertion Site Management. Medtronic Website. Available online: http://www.medtronicdiabetes.com/customer-support/insertion-site-management/site-rotation (accessed on 21 October 2016).
- Hardy, K.J.; Gill, G.V.; Bryson, J.R. Severe insulin-induced lipohypertrophy successfully treated by liposuction. Diabetes Care 1993, 16, 929–930. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.S.; Ahmann, A.; Brazg, R.; Christiansen, M.; Garg, S.; Elaine, W. Accuracy and acceptability of the 6-day Enlite continuous subcutaneous glucose sensor. Diabetes Technol. Ther. 2014, 16, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Medtronic MiniMed. iPro2 Professional CGM-User Guide. Medtronic Website. Available online: http://www.medtronicdiabetes.com/download-library/ipro-2 (accessed on 30 October 2016).
- Gross, T.M.; Bode, B.W.; Einhorn, D.; Kayne, D.M.; Reed, J.H.; White, N.H.; Mastrototaro, J.J. Performance evaluation of the MiniMed continuous glucose monitoring system during patient home use. Diabetes Technol. Ther. 2000, 2, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Clarke, W.L.; Cox, D.; Gonder-Frederick, L.A.; Carter, W.; Pohl, S.L. Evaluating clinical accuracy of systems for self-monitoring of blood glucose. Diabetes Care 1987, 10, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Guerci, B.; Floriot, M.; Bohme, P.; Durain, D.; Benichou, M.; Jellimann, S.; Drouin, D. Clinical performance of CGMS in type 1 diabetic patients treated by continuous subcutaneous insulin infusion using insulin analogs. Diabetes Care 2003, 26, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.S.; Zhang, Y. Introduction to the Glucose Sensing Problem. In Vivo Glucose Sensing; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 1–27. [Google Scholar]
- Klonoff, D.C. Continuous glucose Monitoring: Roadmap for 21st century diabetes therapy. Diabetes Care 2005, 28, 1231–1239. [Google Scholar] [PubMed]
- US Food and Drug Administration. Summary of Safety and Effectiveness Data. US Food and Drug Administration Website. Available online: http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpma/pma.cfm?id=P150029 (accessed on 10 January 2017).
- Christiansen, M.; Bailey, T.; Watkins, E.; Liljenquist, D.; Price, D.; Boock, R.; Peyser, T. A New-Generation Continuous Glucose Monitoring System: Improved Accuracy and Reliability Compared with a Previous-Generation System. Diabetes Technol. Ther. 2013, 15, 881–888. [Google Scholar] [CrossRef] [PubMed]
- El-Khatib, F.H.; Jiang, J.; Damiano, E.R. Automated blood-glucose regulation in diabetic swine using bi-hormonal and insulin-only closed-loop control systems. In Proceedings of the 69th Scientific Sessions of the American Diabetes Association, New Orleans, LA, USA, 5–9 June 2009. [Google Scholar]
- Breton, M.D.; Patek, S.D.; Farret, A.; Place, J.; Demartini, S.; Brown, S.; Anderson, S.; Kovatchev, B.P.; Renard, E. Safety supervision System: First clinical trials. In Proceedings of the Fourth Advanced Technologies and Treatments for Diabetes Annual Meeting, London, UK, 16–19 Feburary 2011. [Google Scholar]
- Helton, K.L.; Ratner, B.D.; Wisniewski, N.A. Biomechanics of the Sensor–Tissue Interface—Effects of Motion, Pressure, and Design on Sensor Performance and Foreign Body Response—Part II: Examples and Application. J. Diabetes Sci. Technol. 2011, 5, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.S.; McLellan, K.; Prowse, M.; Bains, G.; Berk, L. The effect of body fat, aging, and diabetes on vertical and shear pressure in and under a waist belt and its effect on skin blood flow. Diabetes Technol. Ther. 2010, 12, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Mensh, B.D.; Wisniewski, N.A.; Neil, B.M.; Burnett, D.R. Susceptibility of interstitial continuous glucose monitor performance to sleeping position. J. Diabetes Sci. Technol. 2013, 7, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, E.; Tamborlane, W.V. A Tale of Two Compartments: Interstitial versus blood glucose monitoring. Diabetes Technol. Ther. 2009, 11, S-11–S-16. [Google Scholar] [CrossRef] [PubMed]
- Ardilouze, J.L.; Karpe, F.; Currie, J.M.; Frayn, K.N.; Fielding, B.A. Subcutaneous adipose tissue blood flow varies between superior and inferior levels of the anterior abdominal wall. Int. J. Obes. 2004, 28, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Gerritsen, M.; Jansen, J.A.; Kros, A.; Vriezema, D.M.; Sommerdijk, N.A.; Nolte, R.J.M.; Lutterman, J.A.; Van Hövell, S.W.F.M.; Van der Gaag, A. Influence of inflammatory cells and serum on the performance of implantable glucose sensors. J. Biomed. Mater. Res. 2001, 54, 69–75. [Google Scholar] [CrossRef]
- Clark, L.C., Jr.; Spokane, R.B.; Homan, M.M.; Sudan, R.; Miller, M. Long-term stability of electroenzymatic glucose sensors implanted in mice. An update. ASAIO Trans. 1988, 34, 259–265. [Google Scholar] [PubMed]
- Roe, J.N. Biosensor development. Pharm. Res. 1992, 9, 835–844. [Google Scholar] [CrossRef] [PubMed]
| Participant Characteristics (N = 9) | |
|---|---|
| Age (years) | 32.6 ± 4.1 |
| Gender (N) | 4 men |
| Weight (kg) | 71.5 ± 3.5 |
| Height (cm) | 168.7 ± 2.9 |
| BMI (kg/m2) | 25.0 ± 0.6 |
| Waist Circumference (cm) | 81.7 ± 3.8 |
| Fasting Glucose (mmol/L) | 5.0 ± 0.2 |
| Dominant Hand (N) | |
| Right | 8 |
| Left | 0 |
| Dominant Leg (N) | |
| Right | 8 |
| Left | 0 |
| Sleep Position (N) | |
| Right side | 4 |
| Left side | 2 |
| Back | 1 |
| Stomach | 1 |
| Variables | Left Site | Right Site |
|---|---|---|
| Peak PPG2h (mmol/L) | 6.26 ± 0.19 | 6.27 ± 0.18 |
| Peak PPG3h (mmol/L) | 6.38 ± 0.21 | 6.39 ± 0.21 |
| ∆PPG2h (mmol/L) | 0.34 ± 0.11 | 0.34 ± 0.08 |
| ∆PPG3h (mmol/L) | 0.33 ± 0.11 | 0.32 ± 0.08 |
| CV2h (%) | 7.03 ± 1.12 | 7.37 ± 1.06 |
| CV3h (%) | 7.47 ± 1.16 | 8.26 ± 1.12 |
| PPG AUC2h (min·mmol/L) | 707.33 ± 15.30 | 701.53 ± 15.79 |
| PPGAUC3h (min·mmol/L) | 1015.03 ± 22.91 | 1005.96 ± 22.68 |
| CGM-Glucometer (mmol/L) | 0.08 ± 0.04 | 0.03 ± 0.07 |
| MARD (%) | 2.0 ± 0.5 | 2.9 ± 1.1 |
| Variables | Left Site | Right Site |
|---|---|---|
| Average Glucose (mmol/L) | 5.58 ± 0.29 | 5.48 ± 0.08 |
| Min Glucose (mmol/L) | 4.14 ± 0.17 | 3.84 ± 0.18 |
| Max Glucose (mmol/L) | 7.20 ± 0.29 | 7.00 ± 0.24 |
| Number of Excursion above 140 mmol/L | 0.04 ± 0.04 | 0.02 ± 0.02 |
| Number of Excursion below 72 mmol/L | 0.03 ± 0.01 | 0.04 ± 0.01 |
| Duration of Excursion above 140 mmol/L(min) | 1.63 ± 1.47 | 0.62 ± 0.62 |
| Duration of Excursion below 72 mmol/L(min) | 1.02 ± 0.55 | 1.98 ± 0.91 |
| CV (%) | 10.34 ± 1.34 | 10.98 ± 0.71 |
| AUC48h (min·mmol/L) | 15,974.05 ± 251.73 | 15,740.91 ± 241.12 |
| CGM-Glucose meter (mmol/L) | −0.006 ± 0.02 | −0.008 ± 0.03 |
| MARD (%) | 6.9 ± 1.2 | 8.1 ± 0.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, P.T.; Segovia, D.E.; Lee, C.C.; Nguyen, K.-L. Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors. Biosensors 2018, 8, 49. https://doi.org/10.3390/bios8020049
Wu PT, Segovia DE, Lee CC, Nguyen K-L. Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors. Biosensors. 2018; 8(2):49. https://doi.org/10.3390/bios8020049
Chicago/Turabian StyleWu, Pei T., David E. Segovia, Cathy C. Lee, and Kim-Lien Nguyen. 2018. "Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors" Biosensors 8, no. 2: 49. https://doi.org/10.3390/bios8020049
APA StyleWu, P. T., Segovia, D. E., Lee, C. C., & Nguyen, K.-L. (2018). Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors. Biosensors, 8(2), 49. https://doi.org/10.3390/bios8020049




