Recent Advances in DNA Nanotechnology for Plasmonic Biosensor Construction
Abstract
:1. Introduction
2. Aptameric Structure-Based Plasmon Biosensor
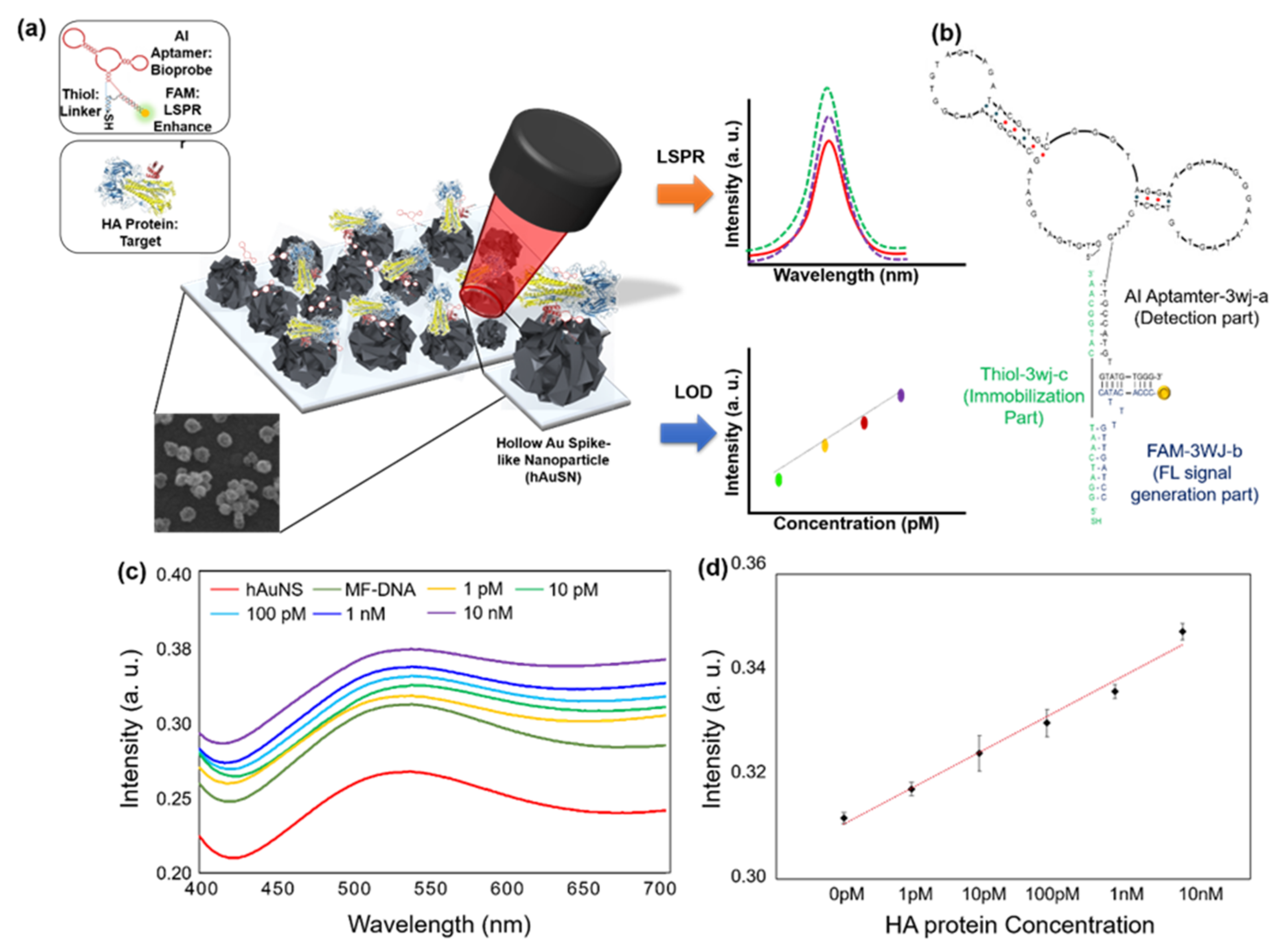
2.1. SPR/LSPR-Based Sensing
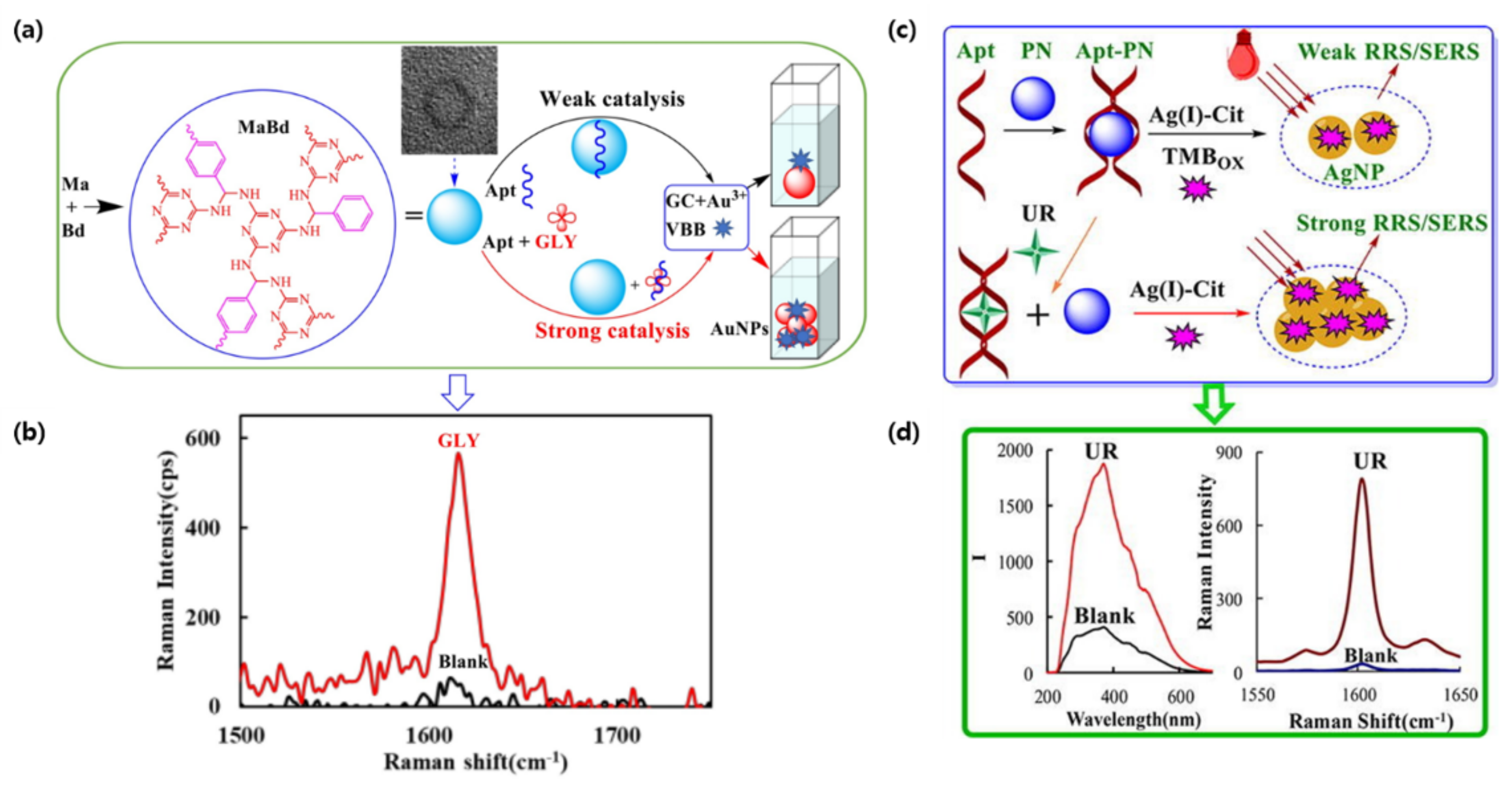
2.2. Surface-Enhanced Raman Scattering-Based Sensing
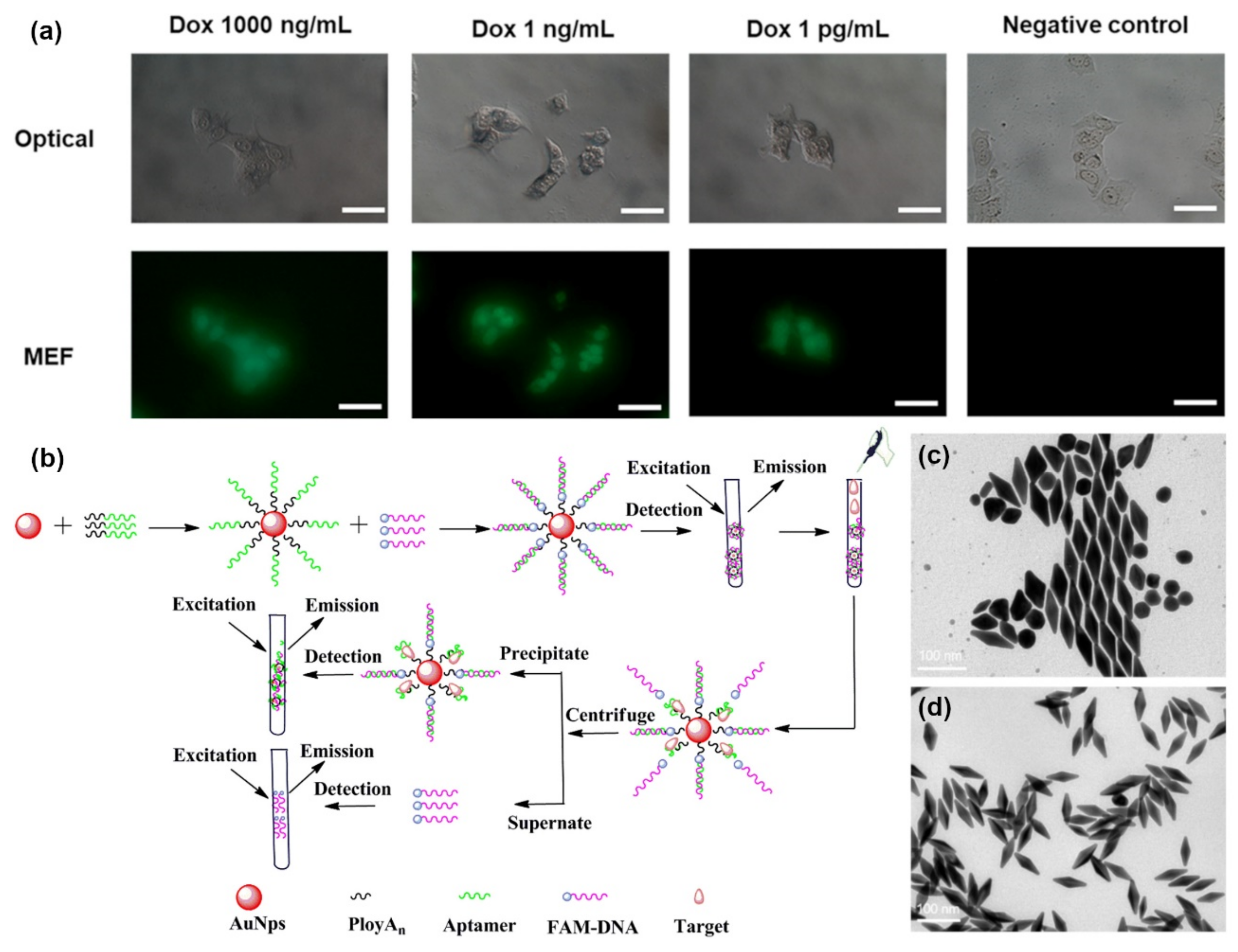
2.3. MEF-Based Sensing
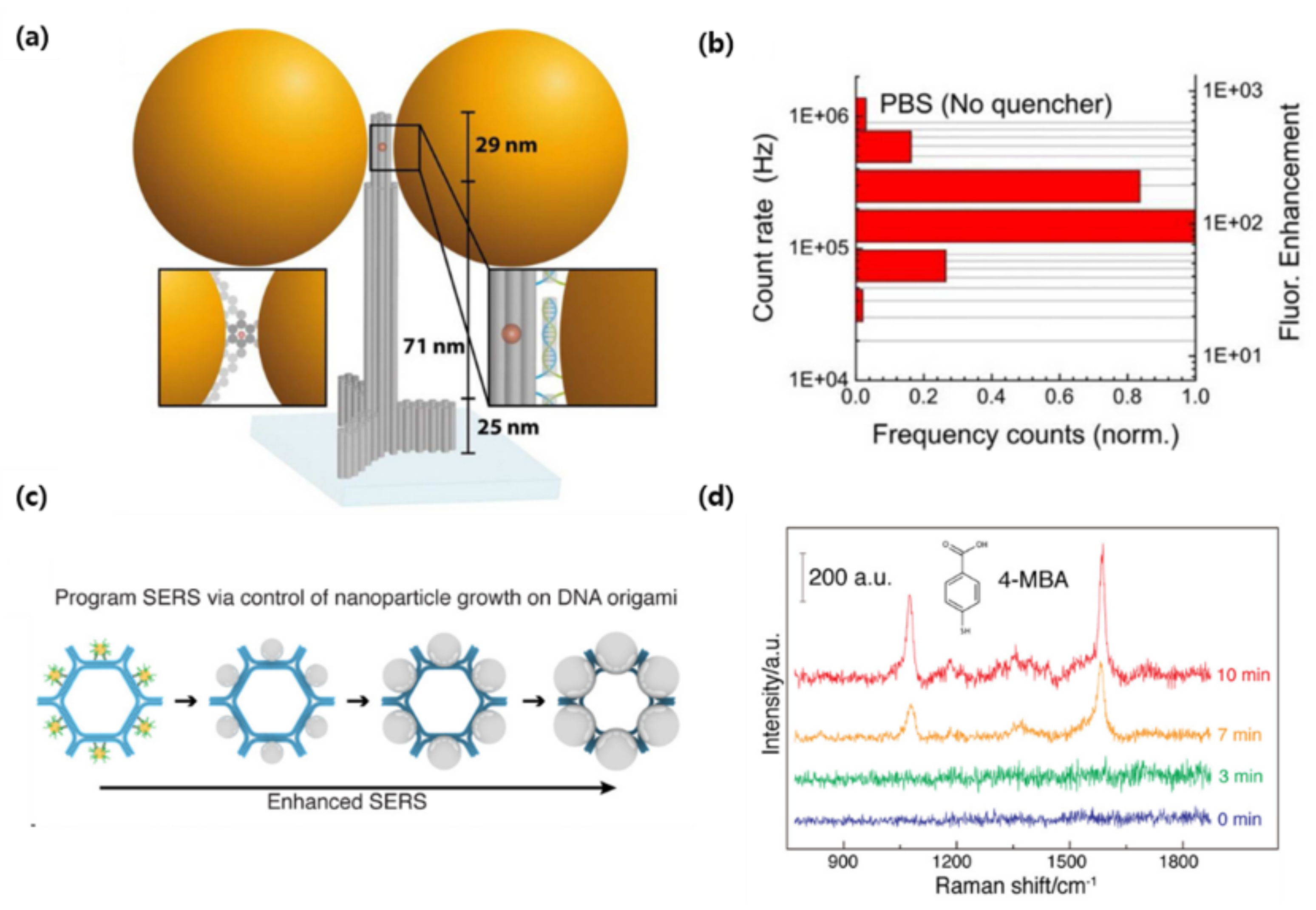
3. DNA Origami-Based Plasmonic Biosensor
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Albert, B.; Johnson, A.; Lewis, J.; Reff, M.; Roberts, K.; Walter, P. DNA, Chromosomes and Genomes. In Molecular Biology of the Cell, 6th ed.; Garland Pub: Spokane, WA, USA, 2014; Chapter 4; ISBN 970-0-8153-4432-2. [Google Scholar]
- Birney, E.; Stamatoyannopoulos, J.A.; Dutta, A.; Guigó, R.; Gingeras, T.R.; Margulies, E.H.; Weng, Z.; Snyder, M.; Dermitzakis, E.T.; Thurman, R.E. Identification and Analysis of Functional Elements in 1% of the Human Genome by the ENCODE Pilot Project. Nature 2007, 447, 799–816. [Google Scholar] [PubMed] [Green Version]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in Oligonucleotide Drug Delivery. Nat. Rev. Drug Discov. 2020, 19, 673–694. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, J.A.; Witzigmann, D.; Thomson, S.B.; Chen, S.; Leavitt, B.R.; Cullis, P.R.; van der Meel, R. The Current Landscape of Nucleic Acid Therapeutics. Nat. Nanotechnol. 2021, 16, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, D. Basics of Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2013; pp. 541–601. [Google Scholar]
- Fakruddin, M.; Hossain, Z.; Afroz, H. Prospects and Applications of Nanobiotechnology: A Medical Perspective. J. Nanobiotechnol. 2012, 10, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef]
- Nagamune, T. Biomolecular Engineering for Nanobio/Bionanotechnology. Nano Converg. 2017, 4, 1–56. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.J.; Groves, B.; Muscat, R.A.; Seelig, G. DNA Nanotechnology from the Test Tube to the Cell. Nat. Publ. Group 2015, 10, 748–760. [Google Scholar] [CrossRef]
- Keller, A.; Linko, V. Challenges and Perspectives of DNA Nanostructures in Biomedicine. Angew. Chem. Int. Ed. 2020, 59, 15818–15833. [Google Scholar] [CrossRef] [Green Version]
- Chao, J.; Zhu, D.; Zhang, Y.; Wang, L.; Fan, C. DNA Nanotechnology-Enabled Biosensors. Biosens. Bioelectron. 2016, 76, 15818–15833. [Google Scholar] [CrossRef]
- Shen, L.; Wang, P.; Ke, Y. DNA Nanotechnology-Based Biosensors and Therapeutics. Adv. Healthc. Mater. 2021, 10, 2002205. [Google Scholar] [CrossRef]
- Ke, Y.; Castro, C.; Choi, J.H. Structural DNA Nanotechnology: Artificial Nanostructures for Biomedical Research. Annu. Rev. Biomed. Eng. 2018, 20, 375–401. [Google Scholar] [CrossRef] [Green Version]
- Pinheiro, A.V.; Han, D.; Shih, W.M.; Yan, H. Challenges and Opportunities for Structural DNA Nanotechnology. Nat. Publ. Group 2011, 6, 763–772. [Google Scholar] [CrossRef]
- La, T.H.; Nguyen, T.T.T.; Pham, V.P.; Nguyen, T.M.H.; Le, Q.H. Using DNA Nanotechnology to Produce a Drug Delivery System. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013, 4, 015002. [Google Scholar] [CrossRef]
- Hu, Q.; Li, H.; Wang, L.; Gu, H.; Fan, C. DNA Nanotechnology-Enabled Drug Delivery Systems. Chem. Rev. 2019, 119, 6459–6506. [Google Scholar] [CrossRef]
- Song, T.; Eshra, A.; Shah, S.; Bui, H.; Fu, D.; Yang, M.; Mokhtar, R.; Reif, J. Fast and Compact DNA Logic Circuits Based on Single-Stranded Gates Using Strand-Displacing Polymerase. Nat. Nanotechnol. 2019, 14, 1075–1081. [Google Scholar] [CrossRef]
- Zhang, C.; Zhao, Y.; Xu, X.; Xu, R.; Li, H.; Teng, X.; Du, Y.; Miao, Y.; Lin, H.C.; Han, D. Cancer Diagnosis with DNA Molecular Computation. Nat. Nanotechnol. 2020, 15, 709–715. [Google Scholar] [CrossRef]
- Dey, S.; Fan, C.; Gothelf, K.V.; Li, J.; Lin, C.; Liu, L.; Liu, N.; Nijenhuis, M.A.D.; Saccà, B.; Simmel, F.C.; et al. DNA Origami. Nat. Rev. Methods Primers 2021, 1, 13. [Google Scholar] [CrossRef]
- Hong, F.; Zhang, F.; Liu, Y.; Yan, H. DNA Origami: Scaffolds for Creating Higher Order Structures. Chem. Rev. 2017, 117, 12584–12640. [Google Scholar] [CrossRef]
- Liu, W.; Duan, H.; Zhang, D.; Zhang, X.; Luo, Q.; Xie, T.; Yan, H.; Peng, L.; Hu, Y.; Liang, L.; et al. Concepts and Application of DNA Origami and DNA Self-Assembly: A Systematic Review. Appl. Bionics Biomech. 2021, 2021, 9112407. [Google Scholar] [CrossRef]
- Amir, Y.; Ben-Ishay, E.; Levner, D.; Ittah, S.; Abu-Horowitz, A.; Bachelet, I. Universal Computing by DNA Origami Robots in a Living Animal. Nat. Publ. Group 2014, 9, 353–357. [Google Scholar] [CrossRef] [Green Version]
- Saran, R.; Wang, Y.; Li, I.T.S. Mechanical Flexibility of Dna: A Quintessential Tool for Dna Nanotechnology. Sensors 2020, 20, 7019. [Google Scholar] [CrossRef] [PubMed]
- Raveendran, M.; Lee, A.J.; Sharma, R.; Wälti, C.; Actis, P. Rational Design of DNA Nanostructures for Single Molecule Biosensing. Nat. Commun. 2020, 11, 4384. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Jiang, Q.; Zhao, X.; Zhao, R.; Wang, Y.; Wang, Y.; Liu, J.; Shang, Y.; Zhao, S.; Wu, T.; et al. A DNA Nanodevice-Based Vaccine for Cancer Immunotherapy. Nat. Mater. 2021, 20, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Weiden, J.; Bastings, M.M.C. DNA Origami Nanostructures for Controlled Therapeutic Drug Delivery. Curr. Opin. Colloid Interface Sci. 2021, 52, 101411. [Google Scholar] [CrossRef]
- Adachi, T.; Nakamura, Y. Aptamers: A Review of Their Chemical Properties. Molecules 2019, 24, 4229. [Google Scholar] [CrossRef] [Green Version]
- Keefe, A.D.; Pai, S.; Ellington, A. Aptamers as Therapeutics. Nat. Rev. Drug Discov. 2010, 9, 537–550. [Google Scholar] [CrossRef]
- Afrasiabi, S.; Pourhajibagher, M.; Raoofian, R.; Tabarzad, M.; Bahador, A. Therapeutic Applications of Nucleic Acid Aptamers in Microbial Infections. J. Biomed. Sci. 2020, 27, 6. [Google Scholar] [CrossRef]
- Achenbach, J.; Chiuman, W.; Cruz, R.; Li, Y. DNAzymes: From Creation In Vitro to Application In Vivo. Curr. Pharm. Biotechnol. 2005, 5, 321–336. [Google Scholar] [CrossRef]
- Ma, L.; Liu, J. Catalytic Nucleic Acids: Biochemistry, Chemical Biology, Biosensors, and Nanotechnology. Iscience 2020, 23, 100815. [Google Scholar] [CrossRef] [Green Version]
- Morrison, D.; Rothenbroker, M.; Li, Y. DNAzymes: Selected for Applications. Small Methods 2018, 2, 1700319. [Google Scholar] [CrossRef]
- Sun, H.; Zhu, X.; Lu, P.Y.; Rosato, R.R.; Tan, W.; Zu, Y. Oligonucleotide Aptamers: New Tools for Targeted Cancer Therapy. Am. Soc. Gene Cell Ther. 2014, 3, e182. [Google Scholar] [CrossRef]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 34, 818–822. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systematic Evolution of Ligands by Exponential Enrichment: RNA Ligands to Bacteriophage T4 DNA Polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Ng, E.W.M.; Shima, D.T.; Calias, P.; Cunningham, E.T.; Guyer, D.R.; Adamis, A.P. Pegaptanib, a Targeted Anti-VEGF Aptamer for Ocular Vascular Disease. Nat. Rev. Drug Discov. 2006, 5, 123–132. [Google Scholar] [CrossRef]
- Lee, M.; Park, S.J.; Kim, G.; Park, C.; Lee, M.H.; Ahn, J.H.; Lee, T. A Pretreatment-Free Electrical Capacitance Biosensor for Exosome Detection in Undiluted Serum. Biosens. Bioelectron. 2022, 199, 113872. [Google Scholar] [CrossRef]
- Kim, J.; Noh, S.W.; Park, C.; Lee, J.H.; Cho, H.Y.; Min, J.; Lee, T. Fabrication of Electrochemical Biosensor Composed of Multi-Functional DNA 4 Way Junction for TNF-α Detection in Human Serum. Bioelectrochemistry 2021, 142, 107939. [Google Scholar] [CrossRef]
- Pultar, J.; Sauer, U.; Domnanich, P.; Preininger, C. Aptamer-Antibody on-Chip Sandwich Immunoassay for Detection of CRP in Spiked Serum. Biosens. Bioelectron. 2009, 24, 1456–1461. [Google Scholar] [CrossRef]
- Abbaspour, A.; Norouz-Sarvestani, F.; Noori, A.; Soltani, N. Aptamer-Conjugated Silver Nanoparticles for Electrochemical Dual-Aptamer-Based Sandwich Detection of Staphylococcus Aureus. Biosens. Bioelectron. 2015, 68, 149–155. [Google Scholar] [CrossRef]
- Kaur, H.; Bruno, J.G.; Kumar, A.; Sharma, T.K. Aptamers in the Therapeutics and Diagnostics Pipelines. Theranostics 2018, 8, 4016. [Google Scholar] [CrossRef]
- Huo, W.; Li, X.; Wang, B.; Zhang, H.; Zhang, J.; Yang, X.; Jin, Y. Recent Advances of DNAzyme-Based Nanotherapeutic Platform in Cancer Gene Therapy. Biophys. Rep. 2020, 6, 256–265. [Google Scholar] [CrossRef]
- Wang, Y.; Nguyen, K.; Spitale, R.C.; Chaput, J.C. A Biologically Stable DNAzyme That Efficiently Silences Gene Expression in Cells. Nat. Chem. 2021, 13, 319–326. [Google Scholar] [CrossRef]
- McConnell, E.M.; Cozma, I.; Mou, Q.; Brennan, J.D.; Lu, Y.; Li, Y. Biosensing with DNAzymes. Chem. Soc. Rev. 2021, 50, 8954–8994. [Google Scholar] [CrossRef]
- Khan, S.; Burciu, B.; Filipe, C.D.M.; Li, Y.; Dellinger, K.; Didar, T.F. DNAzyme-Based Biosensors: Immobilization Strategies, Applications, and Future Prospective. ACS Nano 2021, 15, 13943–13969. [Google Scholar] [CrossRef]
- Lee, T.; Park, S.Y.; Jang, H.; Kim, G.H.; Lee, Y.; Park, C.; Mohammadniaei, M.; Lee, M.H.; Min, J. Fabrication of Electrochemical Biosensor Consisted of Multi-Functional DNA Structure/Porous Au Nanoparticle for Avian Influenza Virus (H5N1) in Chicken Serum. Mater. Sci. Eng. C 2019, 99, 511–519. [Google Scholar] [CrossRef]
- Ramakrishnan, S.; Krainer, G.; Grundmeier, G.; Schlierf, M.; Keller, A. Structural Stability of DNA Origami Nanostructures in the Presence of Chaotropic Agents. Nanoscale 2016, 8, 10398–10405. [Google Scholar] [CrossRef] [Green Version]
- Rutten, I.; Daems, D.; Lammertyn, J. Boosting Biomolecular Interactions through DNA Origami Nano-Tailored Biosensing Interfaces. R. Soc. Chem. 2020, 8, 3606–3615. [Google Scholar] [CrossRef]
- Assenberg, R.; Weston, A.; Cardy, D.L.N.; Fox, K.R. Sequence-Dependent Folding of DNA Three-Way Junctions. Nucleic Acids Res. 2002, 30, 5142–5150. [Google Scholar] [CrossRef] [Green Version]
- Lee, T.; Lee, Y.; Park, S.Y.; Hong, K.; Kim, Y.; Park, C.; Chung, Y.H.; Lee, M.H.; Min, J. Fabrication of Electrochemical Biosensor Composed of Multi-Functional DNA Structure/Au Nanospike on Micro-Gap/PCB System for Detecting Troponin I in Human Serum. Colloids Surf. B Biointerfaces 2019, 175, 343–350. [Google Scholar] [CrossRef]
- Li, J.; Liu, Q.; Xi, H.; Wei, X.; Chen, Z. Y-Shaped DNA Duplex Structure-Triggered Gold Nanoparticle Dimers for Ultrasensitive Colorimetric Detection of Nucleic Acid with the Dark-Field Microscope. Anal. Chem. 2017, 89, 12850–12856. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, Y.; Yang, C.; Xu, H.; Luo, J.; Zhang, W.; Tang, X.; Yang, S.; Fu, W.; Chang, K.; et al. A Label-Free Electrochemical Biosensor for MicroRNAs Detection Based on DNA Nanomaterial by Coupling with Y-Shaped DNA Structure and Non-Linear Hybridization Chain Reaction. Biosens. Bioelectron. 2019, 126, 657–663. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.A.; Yim, G.; Jang, H.; Kim, T.H.; Sohn, H.; Lee, T. Fabrication of an Electrochemical Biosensor Composed of Multi-Functional Ag Ion Intercalated DNA Four-Way Junctions/Rhodium Nanoplate Heterolayer on a Micro-Gap for C-Reactive Protein Detection in Human Serum. R. Soc. Chem. 2021, 146, 2085–2402. [Google Scholar] [CrossRef] [PubMed]
- Xi, H.; Juhas, M.; Zhang, Y. G-Quadruplex Based Biosensor: A Potential Tool for SARS-CoV-2 Detection. Biosens. Bioelectron. 2020, 167, 112494. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Wang, X.; Xiao, Y.; Feng, L.; Zhao, C.; Ren, J.; Qu, X. I-Motif Quadruplex DNA-Based Biosensor for Distinguishing Single- and Multiwalled Carbon Nanotubes. J. Am. Chem. Soc 2009, 131, 13813–13818. [Google Scholar] [CrossRef] [PubMed]
- Mochán, W.L. Plasmons. Ref. Modul. Mater. Sci. Mater. Eng. 2016, 1–13. [Google Scholar]
- Li, H.; Zhang, L. Photocatalytic Performance of Different Exposed Crystal Facets of BiOCl. Curr. Opin. Green Sustain. Chem. 2017, 6, 48–56. [Google Scholar] [CrossRef]
- Petryayeva, E.; Krull, U.J. Localized Surface Plasmon Resonance: Nanostructures, Bioassays and Biosensing—A Review. Anal. Chim. Acta 2011, 706, 8–24. [Google Scholar] [CrossRef]
- Szunerits, S.; Boukherroub, R. Sensing Using Localised Surface Plasmon Resonance Sensors. Chem. Commun. 2012, 48, 8999–9010. [Google Scholar] [CrossRef]
- Duan, Q.; Liu, Y.; Chang, S.; Chen, H.; Chen, J.H. Surface Plasmonic Sensors: Sensing Mechanism and Recent Applications. Sensors 2021, 21, 5262. [Google Scholar] [CrossRef]
- Chatterjee, B.; Kalyani, N.; Das, S.; Anand, A.; Sharma, T.K. Nano-Realm for Point-of-Care (POC) Bacterial Diagnostics. Methods Microbiol. 2019, 46, 19–42. [Google Scholar]
- Langer, J.; de Aberasturi, D.J.; Aizpurua, J.; Alvarez-Puebla, R.A.; Auguié, B.; Baumberg, J.J.; Bazan, G.C.; Bell, S.E.J.; Boisen, A.; Brolo, A.G.; et al. Present and Future of Surface-Enhanced Raman Scattering. ACS Nano 2020, 14, 28–117. [Google Scholar] [CrossRef] [Green Version]
- Bodelón, G.; Montes-García, V.; Pérez-Juste, J.; Pastoriza-Santos, I. Surface-Enhanced Raman Scattering Spectroscopy for Label-Free Analysis of P. Aeruginosa Quorum Sensing. Front. Cell. Infect. Microbiol. 2018, 8, 143. [Google Scholar] [CrossRef]
- Jeong, Y.; Kook, Y.M.; Lee, K.; Koh, W.G. Metal Enhanced Fluorescence (MEF) for Biosensors: General Approaches and a Review of Recent Developments. Biosens. Bioelectron. 2018, 111, 102–116. [Google Scholar] [CrossRef]
- Fothergill, S.M.; Joyce, C.; Xie, F. Metal-Enhanced Fluorescence Biosensing: From Ultra-Violet towards Second near-Infrared Window. Nanoscale 2018, 10, 20914–20929. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Li, J.; Pan, S.; Wang, H.; Liang, A.; Jiang, Z. A Novel Small Molecular Liquid Crystal Catalytic Amplification-Nanogold SPR Aptamer Absorption Assay for Trace Oxytetracycline. Talanta 2021, 233, 122528. [Google Scholar] [CrossRef]
- Écija-Arenas, Á.; Kirchner, E.M.; Hirsch, T.; Fernández-Romero, J.M. Development of an Aptamer-Based SPR-Biosensor for the Determination of Kanamycin Residues in Foods. Anal. Chim. Acta 2021, 1169, 338631. [Google Scholar] [CrossRef]
- Chen, R.; Kan, L.; Duan, F.; He, L.; Wang, M.; Cui, J.; Zhang, Z.; Zhang, Z. Surface Plasmon Resonance Aptasensor Based on Niobium Carbide MXene Quantum Dots for Nucleocapsid of SARS-CoV-2 Detection. Microchim. Acta 2021, 188, 316. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Lei, K.N.; Li, C.N.; Luo, Y.H.; Jiang, Z.L. A New and Facile Nanosilver SPR Colored Method for Ultratrace Arsenic Based on Aptamer Regulation of Au-Doped Carbon Dot Catalytic Amplification. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2020, 232, 118174. [Google Scholar] [CrossRef]
- Sodhi, K.K.; Kumar, M.; Agrawal, P.K.; Singh, D.K. Perspectives on Arsenic Toxicity, Carcinogenicity and Its Systemic Remediation Strategies. Environ. Technol. Innov. 2019, 16, 100462. [Google Scholar] [CrossRef]
- Liu, B.; Huang, R.; Yu, Y.; Su, R.; Qi, W.; He, Z. Gold Nanoparticle-Aptamer-Based LSPR Sensing of Ochratoxin A at a Widened Detection Range by Double Calibration Curve Method. Front. Chem. 2018, 6, 94. [Google Scholar] [CrossRef]
- Sun, D.; Wu, Y.; Chang, S.J.; Chen, C.J.; Liu, J.T. Investigation of the Recognition Interaction between Glycated Hemoglobin and Its Aptamer by Using Surface Plasmon Resonance. Talanta 2021, 222, 121466. [Google Scholar] [CrossRef]
- Hu, R.; Huang, X.; Huang, J.; Li, Y.; Zhang, C.; Yin, Y.; Chen, Z.; Jin, Y.; Cai, J.; Cui, F. Long- and Short-Term Health Effects of Pesticide Exposure: A Cohort Study from China. PLoS ONE 2015, 10, e0128766. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, R.; Yu, B.; Liang, A.; Jiang, Z. A Highly Sensitive Gold Nanosol SERS Aptamer Assay for Glyphosate with a New COF Nanocatalytic Reaction of Glycol-Au(III). Sens. Actuators B Chem. 2021, 344, 130288. [Google Scholar] [CrossRef]
- Austin, J. Urea Toxicity and Its Prevention. In Urea as a Protein Supplement; Briggs, M.H., Ed.; Elsevier: Amsterdam, The Netherlands, 1967; Chapter 9; pp. 173–184. [Google Scholar]
- Yao, D.; Wang, H.; Lu, S.; Li, C.; Liang, A.; Wen, G.; Jiang, Z. On-Signal Amplification of Silver Nanosol RRS/SERS Aptamer Detection of Ultratrace Urea by Polystyrene Nanosphere Catalyst. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2022, 265, 120353. [Google Scholar] [CrossRef] [PubMed]
- Bhamidipati, M.; Cho, H.Y.; Lee, K.B.; Fabris, L. SERS-Based Quantification of Biomarker Expression at the Single Cell Level Enabled by Gold Nanostars and Truncated Aptamers. Bioconjug. Chem. 2018, 29, 2970–2981. [Google Scholar] [CrossRef] [PubMed]
- Ning, C.F.; Wang, L.; Tian, Y.F.; Yin, B.C.; Ye, B.C. Multiple and Sensitive SERS Detection of Cancer-Related Exosomes Based on Gold-Silver Bimetallic Nanotrepangs. Analyst 2020, 145, 2795–2804. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, S.; Song, X.; Wang, H.; Wang, J.; Wang, Y.; Huang, J.; Yu, J. Robust and Universal SERS Sensing Platform for Multiplexed Detection of Alzheimer’s Disease Core Biomarkers Using PAapt-AuNPs Conjugates. ACS Sens. 2019, 4, 2140–2149. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, C.; Zhang, G.; Peng, Z.; Yao, L.; Wang, Q.; Cao, Z.; Mu, Q.; Xuan, L. Distance-Dependent Metal Enhanced Fluorescence by Flowerlike Silver Nanostructures Fabricated in Liquid Crystalline Phase. Opt. Mater. 2017, 72, 289–294. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, J.W. Metal-Enhanced Fluorescence by Bifunctional Au Nanoparticles for Highly Sensitive and Simple Detection of Proteolytic Enzyme. Nano Lett. 2020, 20, 7100–7107. [Google Scholar] [CrossRef]
- Jiang, L.; Hang, X.; Zhang, P.; Zhang, J.; Wang, Y.; Wang, W.; Ren, L. A Highly Sensitive Fluorescence-Enhanced Aptasensor Based on PolyAn-Aptamer Nanostructure. Microchem. J. 2019, 148, 285–290. [Google Scholar] [CrossRef]
- Zheng, M.; Kang, Y.; Liu, D.; Li, C.; Zheng, B.; Tang, H. Detection of ATP from “Fluorescence” to “Enhanced Fluorescence” Based on Metal-Enhanced Fluorescence Triggered by Aptamer Nanoswitch. Sens. Actuators B Chem. 2020, 319, 128263. [Google Scholar] [CrossRef]
- Jin, F.; Li, H.; Xu, D. Enzyme-Free Fluorescence Microarray for Determination of Hepatitis B Virus DNA Based on Silver Nanoparticle Aggregates-Assisted Signal Amplification. Anal. Chim. Acta 2019, 1077, 297–304. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, Z.; Yang, S.; Li, K.; Teng, C.; Ke, Y.; Tian, Y. DNA Origami-Enabled Biosensors. Sensors 2020, 20, 6899. [Google Scholar] [CrossRef]
- Bellassai, N.; D’Agata, R.; Spoto, G. Novel Nucleic Acid Origami Structures and Conventional Molecular Beacon–Based Platforms: A Comparison in Biosensing Applications. Anal. Bioanal. Chem. 2021, 413, 6063–6077. [Google Scholar] [CrossRef]
- Lu, J.; Wang, J.; Hu, X.; Gyimah, E.; Yakubu, S.; Wang, K.; Wu, X.; Zhang, Z. Electrochemical Biosensor Based on Tetrahedral DNA Nanostructures and G-Quadruplex-Hemin Conformation for the Ultrasensitive Detection of MicroRNA-21 in Serum. Anal. Chem. 2019, 91, 7353–7359. [Google Scholar] [CrossRef]
- Lee, T.; Mohammadniaei, M.; Zhang, H.; Yoon, J.; Choi, H.K.; Guo, S.; Guo, P.; Choi, J.W. Single Functionalized PRNA/Gold Nanoparticle for Ultrasensitive MicroRNA Detection Using Electrochemical Surface-Enhanced Raman Spectroscopy. Adv. Sci. 2020, 7, 1902477. [Google Scholar] [CrossRef]
- Lee, T.; Kim, G.H.; Kim, S.M.; Hong, K.; Kim, Y.; Park, C.; Sohn, H.; Min, J. Label-Free Localized Surface Plasmon Resonance Biosensor Composed of Multi-Functional DNA 3 Way Junction on Hollow Au Spike-like Nanoparticles (HAuSN) for Avian Influenza Virus Detection. Colloids Surf. B Biointerfaces 2019, 182, 110341. [Google Scholar] [CrossRef]
- Puchkova, A.; Vietz, C.; Pibiri, E.; Wünsch, B.; Sanz Paz, M.; Acuna, G.P.; Tinnefeld, P. DNA Origami Nanoantennas with over 5000-Fold Fluorescence Enhancement and Single-Molecule Detection at 25 μM. Nano Lett. 2015, 15, 8354–8359. [Google Scholar] [CrossRef]
- Thacker, V.V.; Herrmann, L.O.; Sigle, D.O.; Zhang, T.; Liedl, T.; Baumberg, J.J.; Keyser, U.F. DNA Origami Based Assembly of Gold Nanoparticle Dimers for Surface-Enhanced Raman Scattering. Nat. Commun. 2014, 5, 3448. [Google Scholar] [CrossRef] [Green Version]
- Zhou, C.; Yang, Y.; Li, H.; Gao, F.; Song, C.; Yang, D.; Xu, F.; Liu, N.; Ke, Y.; Su, S.; et al. Programming Surface-Enhanced Raman Scattering of DNA Origami-Templated Metamolecules. Nano Lett. 2020, 20, 3155–3159. [Google Scholar] [CrossRef]
- Jung, J.K.; Archuleta, C.M.; Alam, K.K.; Lucks, J.B. Programming Cell-Free Biosensors with DNA Strand Displacement Circuits. Nat. Chem. Biol. 2022, 18, 385–393. [Google Scholar] [CrossRef]
- Dass, M.; Gür, F.N.; Kołątaj, K.; Urban, M.J.; Liedl, T. DNA Origami-Enabled Plasmonic Sensing. J. Phys. Chem. 2021, 125, 5969–5981. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chu, X.; Yang, B. Electrochemical Behavior of Hemin Binding with Human Centrin 3. Bioelectrochemistry 2017, 117, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Newbigging, A.M.; Wang, Z.; Tao, J.; Deng, W.; Le, X.C.; Zhang, H. DNAzyme-Mediated Assays for Amplified Detection of Nucleic Acids and Proteins. Anal. Chem. 2018, 90, 190–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasick, N.J.; Ramadas, R.; Todd, A.V. Subzymes: Regulating DNAzymes for Point of Care Nucleic Acid Sensing. Sens. Actuators B Chem. 2019, 297, 126704. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Li, M.; Tang, L.; He, J.J. DNAzyme-Integrated Plasmonic Nanosensor for Bacterial Sample-to-Answer Detection. Biosens. Bioelectron. 2017, 89, 880–885. [Google Scholar] [CrossRef]
- Lee, W.; Hwang, B.H. Plasmonic Biosensor Controlled by DNAzyme for On-Site Genetic Detection of Pathogens. Biotechnol. J. 2020, 15, 1900329. [Google Scholar] [CrossRef]
- Wu, H.; Wang, S.; Li, S.F.Y.; Bao, Q.; Xu, Q. A label-free lead(II) ion sensor based on surface plasmon resonance and DNAzyme-gold nanoparticle conjugates. Anal. Bioanal. Chem. 2020, 412, 7525–7533. [Google Scholar] [CrossRef]


| Detection Method | Oligonucleotide Component | Plasmonic Component | Target | LOD | Reference |
|---|---|---|---|---|---|
| SPR 1 | Aptamer | AuNPs | Oxytetracycline | 0.50 ng/mL | [66] |
| Graphene coated Au chip | Kanamycin | 285 nmol/L | [67] | ||
| niobium carbide MXene quantum dots coated Au chip | N-gene of SARS-CoV-2 | 4.9 pg/mL | [68] | ||
| Ag nanosols | Arsenic (As3+) | 0.01 μg/L | [69] | ||
| LSPR 2 | Aptamer | AuNPs | Ochratoxin A, triphosphate, 17β-estradiol and oxytetracycline hydrochloride | 10−10.5 g/mL | [71] |
| DNA 3 Way Junction | Hollow Au nanospike | Avian influenza | 1 pM | [89] | |
| SERS 3 | Aptamer | Au nanosols | Glyphosate | 0.002 nmol/L | [74] |
| Ag nanosols | Urea | 0.03 nM | [76] | ||
| Au nanostars | Epithelial cell adhesion molecule (EpCAM) | 100 nM to 500 nM. | [77] | ||
| Au–Ag bimetallic nanotrepangs | PSMA, Her2 and AFP proteins expressing exosomes derived from LNCaP, SKBR3 and HepG2 cell lines | 6 particles/μL, 72 particles/μL and 35 particles/μL, respectively | [78] | ||
| AuNPs | Tau protein and Aβ(1–42) | 3.7 × 10−2 nM | [79] | ||
| DNA origami | AuNPs | N/A | N/A | [90] | |
| N/A | N/A | [91] | |||
| AuNPs and AgNPs | N/A | N/A | [92] | ||
| MEF 4 | Aptamer | AuNPs | Caspase-3 | 10 pg/mL | [81] |
| ATP | 200 pM | [82] | |||
| Au nanobipyramids | ATP | 35 nM | [83] | ||
| AgNPs aggregates | Hepatitis B virus DNA | 50 fM | [84] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.A.; Amri, C.; Kwon, Y.; Lee, J.-H.; Lee, T. Recent Advances in DNA Nanotechnology for Plasmonic Biosensor Construction. Biosensors 2022, 12, 418. https://doi.org/10.3390/bios12060418
Park JA, Amri C, Kwon Y, Lee J-H, Lee T. Recent Advances in DNA Nanotechnology for Plasmonic Biosensor Construction. Biosensors. 2022; 12(6):418. https://doi.org/10.3390/bios12060418
Chicago/Turabian StylePark, Jeong Ah, Chaima Amri, Yein Kwon, Jin-Ho Lee, and Taek Lee. 2022. "Recent Advances in DNA Nanotechnology for Plasmonic Biosensor Construction" Biosensors 12, no. 6: 418. https://doi.org/10.3390/bios12060418
APA StylePark, J. A., Amri, C., Kwon, Y., Lee, J.-H., & Lee, T. (2022). Recent Advances in DNA Nanotechnology for Plasmonic Biosensor Construction. Biosensors, 12(6), 418. https://doi.org/10.3390/bios12060418







