An Immunosensor for the Determination of Cathepsin S in Blood Plasma by Array SPRi—A Comparison of Analytical Properties of Silver–Gold and Pure Gold Chips
Abstract
1. Introduction
2. Apparatus, Chip Manufacture, Materials and Methods
2.1. SPRi Apparatus
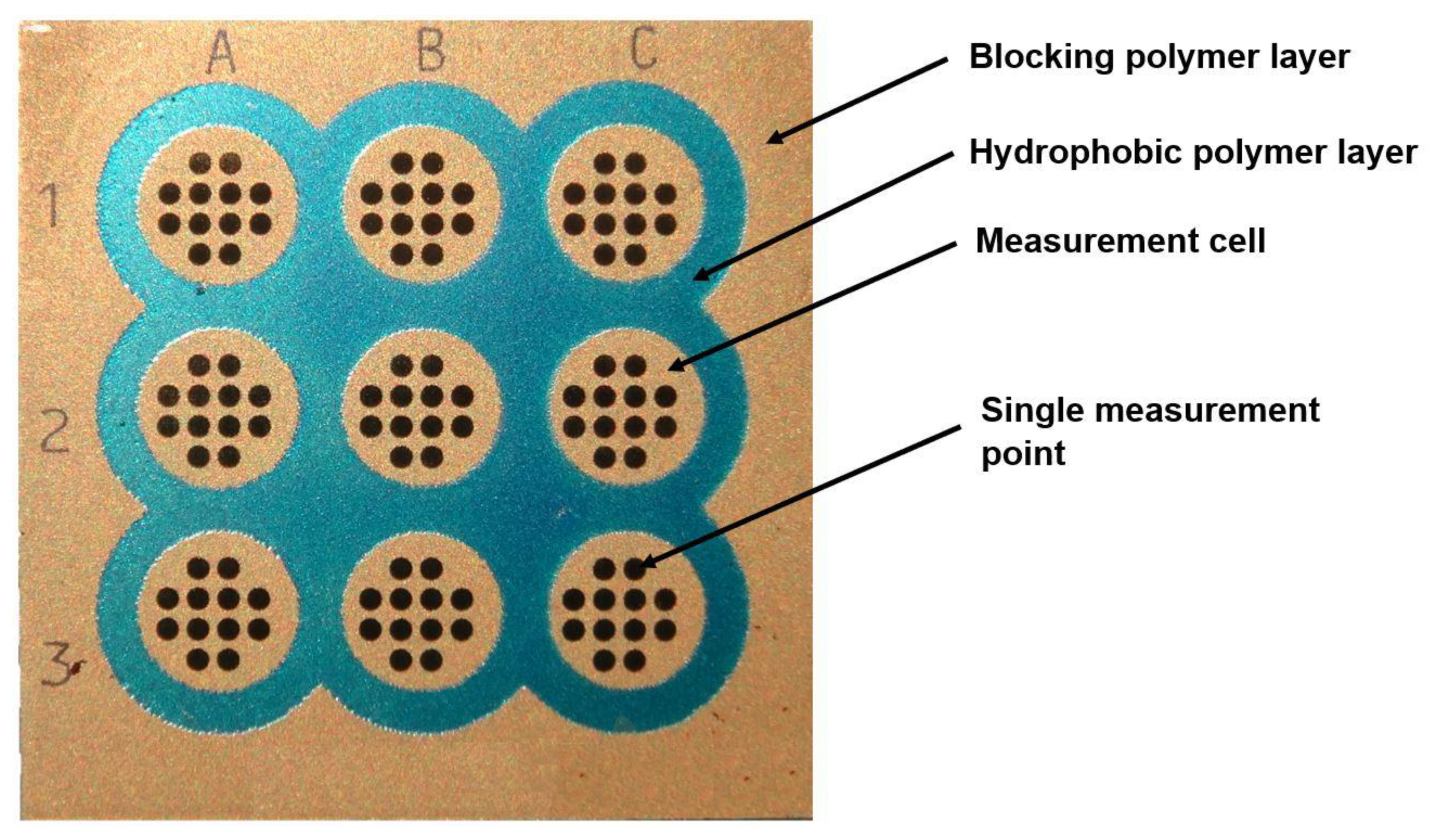
2.2. Chip Manufacture
Deposition of Metallic Cr-Ag-Au Layers onto Glass Substrate
2.3. Preparation of Separating Paint Layers
2.4. Reagents
2.5. Biological Material
2.6. Procedure of Antibody Immobilization
2.7. SPRi Measurement
3. Results and Discussion
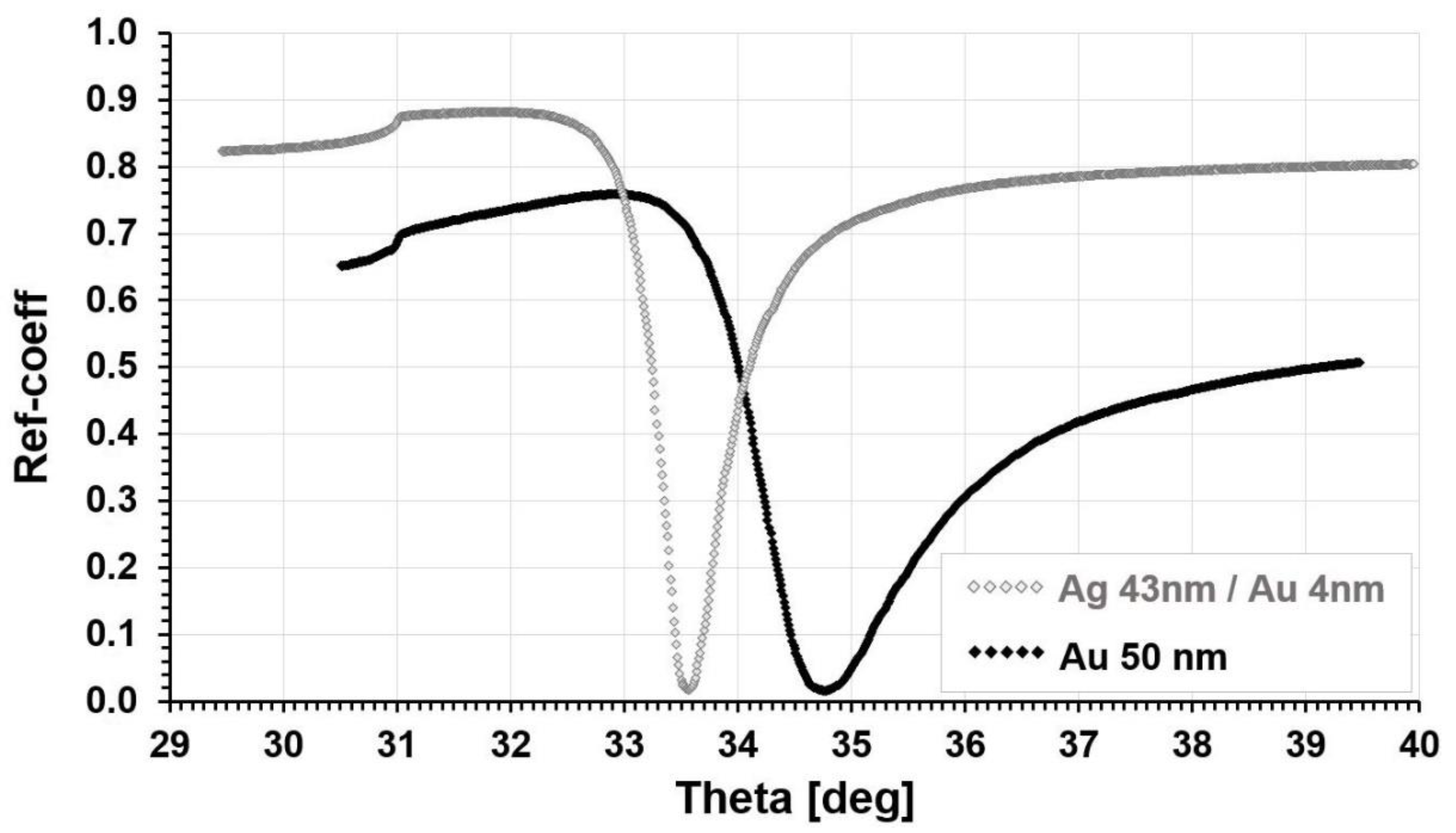
Analytical Characteristics of the Biosensor for the Determination of Cathepsin S Formed to the Ag/Au Chip Substrate
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Laudański, P.; Gorodkiewicz, E.; Ramotowska, B.; Charkiewicz, R.; Kuzmicki, M.; Szamatowicz, J. Determination of cathepsins B, D and G concentration in eutopicproliferative endometrium of women with endometriosis by surface plasmon resonance imaging (SPRI) technique. Eur. J. Obstetr. Gynecol. Reprod. Biol. 2013, 169, 80–83. [Google Scholar] [CrossRef]
- Oldak, L.; Sankiewicz, A.; Żelazowska-Rutkowska, B.; Cylwik, B.; Lukaszewski, Z.; Skoczylas, M.; Gorodkiewicz, E. Two SPRi biosensors for the determination of cathepsin S in blood plasma. Talanta 2020, 225, 121900. [Google Scholar] [CrossRef] [PubMed]
- Sankiewicz, A.; Romanowicz, L.; Laudanski, P.; Zelazowska-Rutkowska, B.; Puzan, B.; Cylwik, B.; Gorodkiewicz, E. SPR imaging biosensor for determination of laminin-5 as a potential cancer marker in biological material. Anal. Bioanal. Chem. 2016, 408, 5269–5276. [Google Scholar] [CrossRef]
- Sankiewicz, A.; Romanowicz, L.; Pyc, M.; Hermanowicz, A.; Gorodkiewicz, E. SPR imaging biosensor for the quantitation of fibronectin concentration in blood samples. J. Pharm. Biomed. Anal. 2018, 150, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sankiewicz, A.; Lukaszewski, Z.; Trojanowska, K.; Gorodkiewicz, E. Determination of collagen type IV by Surface Plasmon Resonance Imaging using a specific biosensor. Anal. Biochem. 2016, 515, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Tokarzewicz, A.; Romanowicz, L.; Sveklo, I.; Gorodkiewicz, E. The development of a matrix metalloproteinase-1 biosensor based on the surface plasmon resonance imaging technique. Anal. Methods 2016, 8, 6428–6435. [Google Scholar] [CrossRef]
- Sankiewicz, A.; Laudanski, P.; Romanowicz, L.; Hermanowicz, A.; Roszkowska-Jakimiec, W.; Debek, W.; Gorodkiewicz, E. Development of surface plasmon resonance imaging biosensors for detection of ubiquitin carboxyl-terminal hydrolase L1. Anal. Biochem. 2015, 469, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Tokarzewicz, A.; Romanowicz, L.; Matuszczak, E.; Hermanowicz, A.; Gorodkiewicz, E. SPRI biosensors for quantitative de-termination of matrix metalloproteinase-2. Anal. Method. 2017, 9, 2407–2414. [Google Scholar] [CrossRef]
- Falkowski, P.; Lukaszewski, Z.; Gorodkiewicz, E. Potential of surface plasmon resonance biosensors in cancer detection. J. Pharm. Biomed. Anal. 2020, 194, 113802. [Google Scholar] [CrossRef]
- Sankiewicz, A.; Guszcz, T.; Mena-Hortelano, R.; Zukowski, K.; Gorodkiewicz, E. Podoplanin serum and urine concen-tration in transitional bladder cancer. Cancer Biomark. 2016, 16, 343–350. [Google Scholar] [CrossRef]
- Guszcz, T.; Szymańska, B.; Kozlowski, R.; Lukaszewski, Z.; Laskowski, P.; Gorodkiewicz, E. Plasma aromatase as a sensitive and selective potential biomarker of bladder cancer and its role in tumorigenesis. Oncol. Lett. 2019, 19, 562–568. [Google Scholar] [CrossRef]
- Nesterenko, D.V.; Rehman, S.; Sekkat, Z. Surface plasmon sensing with different metals in single and double layer configu-rations. Appl. Opt. 2012, 27, 6673–6682. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-T.; Lo, K.-C.; Chang, H.-Y.; Wu, H.-T.; Ho, J.H.; Yen, T.-J. Ag/Au bi-metallic film based color surface plasmon resonance biosensor with enhanced sensitivity, color contrast and great linearity. Biosens. Bioelectron. 2012, 36, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cheng, Z.; Singh, V.; Zheng, Z.; Wang, Y.; Li, S.; Song, L.; Zhu, J. Stable and Sensitive Silver Surface Plasmon Resonance Imaging Sensor Using Trilayered Metallic Structures. Anal. Chem. 2014, 86, 1430–1436. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Wang, Z.; Gillespie, D.E.; Lausted, C.; Zheng, Z.; Yang, M.; Zhu, J. Plain Silver Surface Plasmon Resonance for Microarray Application. Anal. Chem. 2015, 87, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Baburin, A.S.; Merzlikin, A.M.; Baryshev, A.V.; Ryzhikov, I.A.; Panfilov, Y.V.; Rodionov, I.A. Silver-based plasmonics: Golden material platform and application challenges [Invited]. Opt. Mater. Express 2019, 9, 611–642. [Google Scholar] [CrossRef]
- Mrozek, P.; Gorodkiewicz, E.; Falkowski, P.; Hoscilo, B. Sensitivity analysis of single- and bimetallic Surface Plasmon Reasons. Sensors 2021, 21, 4348. [Google Scholar] [CrossRef]
- Liu, W.-L.; Liu, D.; Cheng, K.; Liu, Y.-J.; Xiang, S.; Chi, P.-D.; Liu, X.-H.; Xue, N.; Lai, Y.-Z.; Guo, L.; et al. Evaluating the diagnostic and prognostic value of circulating cathepsin S in gastric cancer. Oncotarget 2016, 7, 28124–28138. [Google Scholar] [CrossRef]
- Kos, J.; Sekirnik, A.; Kopitar, G.; Cimerman, N.; Kayser, K.; Stremmer, A.; Fiehn, W.; Werle, B. Cathepsin S in tumours, regional lymph nodes and sera of patients with lung cancer: Relation to prognosis. Br. J. Cancer 2001, 85, 1193–1200. [Google Scholar] [CrossRef]
- Fernandez, P.L.; Farre, X.; Nadal, A.; Fernandez, E.; Peiro, N.; Sloane, B.F.; Shi, G.P.; Chapman, H.A.; Campo, E.; Cardesa, A. Expression of cathepsins B and S in the progression of prostate carcinoma. Int. J. Cancer 2001, 95, 51–55. [Google Scholar] [CrossRef]
- Xu, J.; Li, D.; Ke, Z.; Liu, R.; Maubach, G.; Zhuo, L. Cathepsin S is aberrantly overexpressed in human heptacellular carcinoma. Mol. Med. Rep. 2009, 2, 713–718. [Google Scholar] [PubMed]
- Flannery, T.; Gibson, D.; Mirakhur, M.; McQuaid, S.; Greenan, C.; Trimble, A.; Walker, B.; McCormick, D.; Johnston, P.G. The Clinical Significance of Cathepsin S Expression in Human Astrocytomas. Am. J. Pathol. 2003, 163, 175–182. [Google Scholar] [CrossRef][Green Version]
- Burden, R.E.; Gormley, J.A.; Jaquin, T.J.; Small, D.; Quinn, D.J.; Hegarty, S.M.; Ward, C.; Walker, B.; Johnston, J.A.; Olwill, S.A.; et al. Antibody-Mediated Inhibition of Cathepsin S Blocks Colorectal Tumor Invasion and Angiogenesis. Clin. Cancer Res. 2009, 15, 6042–6051. [Google Scholar] [CrossRef] [PubMed]
- Jobs, E. Cathepsin S as a Biomarker of Low-Grade Inflammation, Insulin Resistance and Cardiometabolic Disease Risk. Master’s Thesis, Faculty of Medicine, Acta Universitatis Upsaliensis, Uppsala, Sweden, 2014; p. 48. [Google Scholar]




| (A) | |||
| Spike [ng mL−1] | Found [ng mL−1] | Recovery [%] | RSD [%] |
| 0.100 | 0.106 | 105.9 | 9.4 |
| 0.500 | 0.519 | 103.8 | 3.8 |
| 1.000 | 1.015 | 101.5 | 7.3 |
| (B) | |||
| Spike [ng mL−1] | Found [ng mL−1] | Recovery [%] | RSD [%] |
| 0.100 | 0.090 | 90.2 | 24.0 |
| 0.500 | 0.512 | 102.3 | 2.0 |
| 1.000 | 1.049 | 104.9 | 1.9 |
| Sample No. | Chip Ag/Au | Chip Au | ||
|---|---|---|---|---|
| Cath S [ng mL−1] | SD [ng mL−1] | Cath S [ng mL−1] | SD [ng mL−1] | |
| 1 | 0.48 | 0.05 | 0.39 | 0.03 |
| 2 | 0.98 | 0.04 | 0.93 | 0.01 |
| 3 | 2.90 | 0.07 | 2.60 | 0.03 |
| 4 | 1.90 | 0.04 | 1.90 | 0.03 |
| 5 | 0.77 | 0.04 | 0.48 | 0.03 |
| 6 | 1.40 | 0.04 | 1.20 | 0.03 |
| 7 | 2.80 | 0.11 | 2.40 | 0.04 |
| 8 | 2.00 | 0.03 | 2.40 | 0.02 |
| 9 | 0.72 | 0.09 | 0.40 | 0.02 |
| average | 1.9 | 1.5 | 1.4 | 0.9 |
| Sample No. | Chip Ag/Au | Chip Au | ||
|---|---|---|---|---|
| Cath S [ng mL−1] | SD [ng mL−1] | Cath S [ng mL−1] | SD [ng mL−1] | |
| 18A | 16.0 | 0.32 | 16.0 | 0.68 |
| 19A | 17.0 | 0.24 | 19.0 | 0.80 |
| 21A | 12.0 | 0.65 | 13.0 | 0.49 |
| 23A | 12.0 | 0.24 | 11.0 | 0.27 |
| 24A | 15.0 | 0.22 | 17.0 | 0.67 |
| 25A | 9.8 | 0.41 | 10.0 | 0.17 |
| 26A | 10.0 | 0.12 | 8.2 | 0.13 |
| 27A | 13.0 | 0.30 | 13.0 | 0.54 |
| 28A | 7.5 | 0.13 | 7.0 | 0.11 |
| average | 12.5 | 3.1 | 12.7 | 4.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Falkowski, P.; Mrozek, P.; Lukaszewski, Z.; Oldak, L.; Gorodkiewicz, E. An Immunosensor for the Determination of Cathepsin S in Blood Plasma by Array SPRi—A Comparison of Analytical Properties of Silver–Gold and Pure Gold Chips. Biosensors 2021, 11, 298. https://doi.org/10.3390/bios11090298
Falkowski P, Mrozek P, Lukaszewski Z, Oldak L, Gorodkiewicz E. An Immunosensor for the Determination of Cathepsin S in Blood Plasma by Array SPRi—A Comparison of Analytical Properties of Silver–Gold and Pure Gold Chips. Biosensors. 2021; 11(9):298. https://doi.org/10.3390/bios11090298
Chicago/Turabian StyleFalkowski, Pawel, Piotr Mrozek, Zenon Lukaszewski, Lukasz Oldak, and Ewa Gorodkiewicz. 2021. "An Immunosensor for the Determination of Cathepsin S in Blood Plasma by Array SPRi—A Comparison of Analytical Properties of Silver–Gold and Pure Gold Chips" Biosensors 11, no. 9: 298. https://doi.org/10.3390/bios11090298
APA StyleFalkowski, P., Mrozek, P., Lukaszewski, Z., Oldak, L., & Gorodkiewicz, E. (2021). An Immunosensor for the Determination of Cathepsin S in Blood Plasma by Array SPRi—A Comparison of Analytical Properties of Silver–Gold and Pure Gold Chips. Biosensors, 11(9), 298. https://doi.org/10.3390/bios11090298







