A Double-Deck Structure of Reduced Graphene Oxide Modified Porous Ti3C2Tx Electrode towards Ultrasensitive and Simultaneous Detection of Dopamine and Uric Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
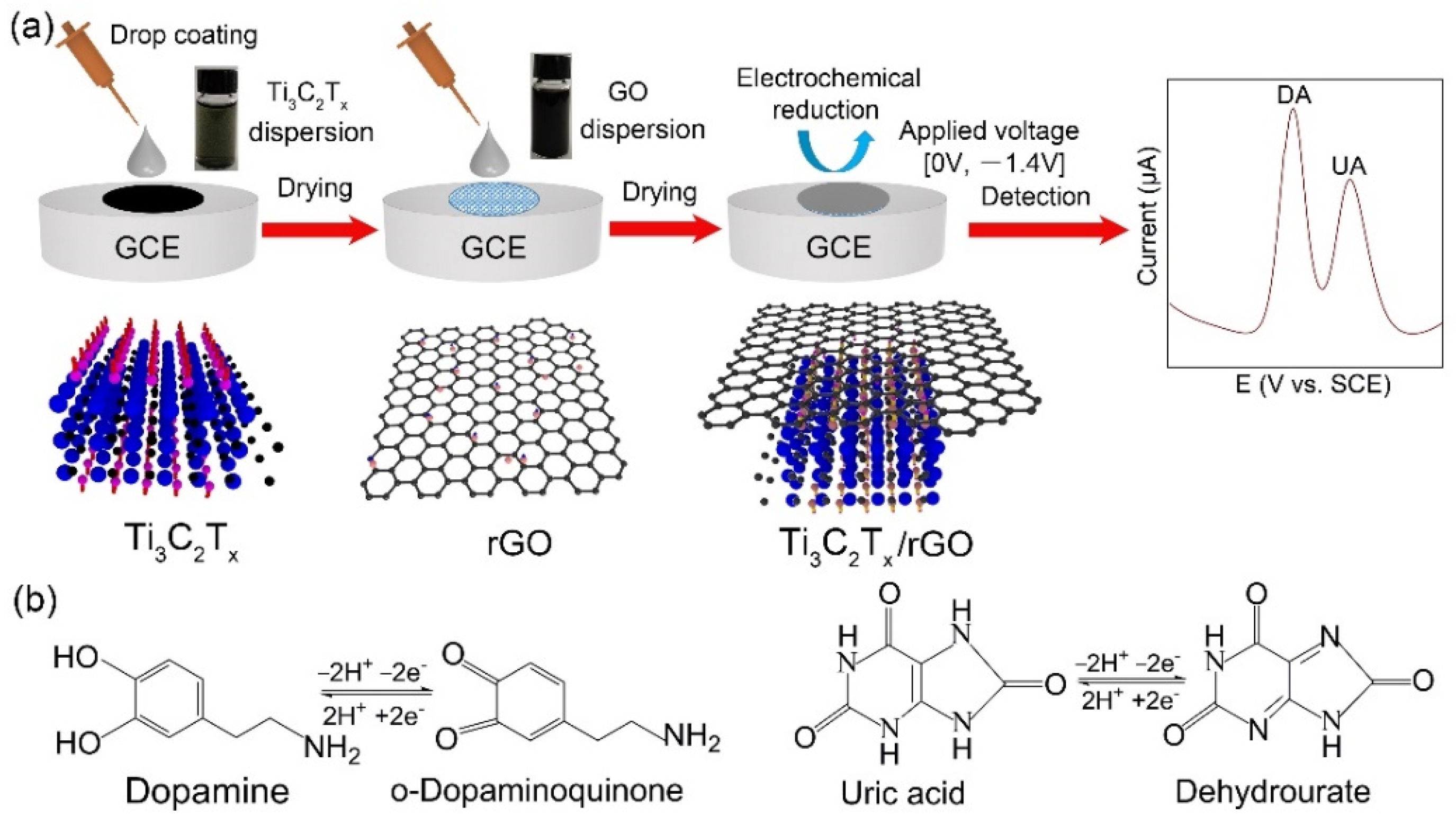
2.2. Fabrication of Ti3C2Tx/rGO Electrodes
2.3. Characterizations
2.4. Electrochemical Tests
3. Results and Discussion
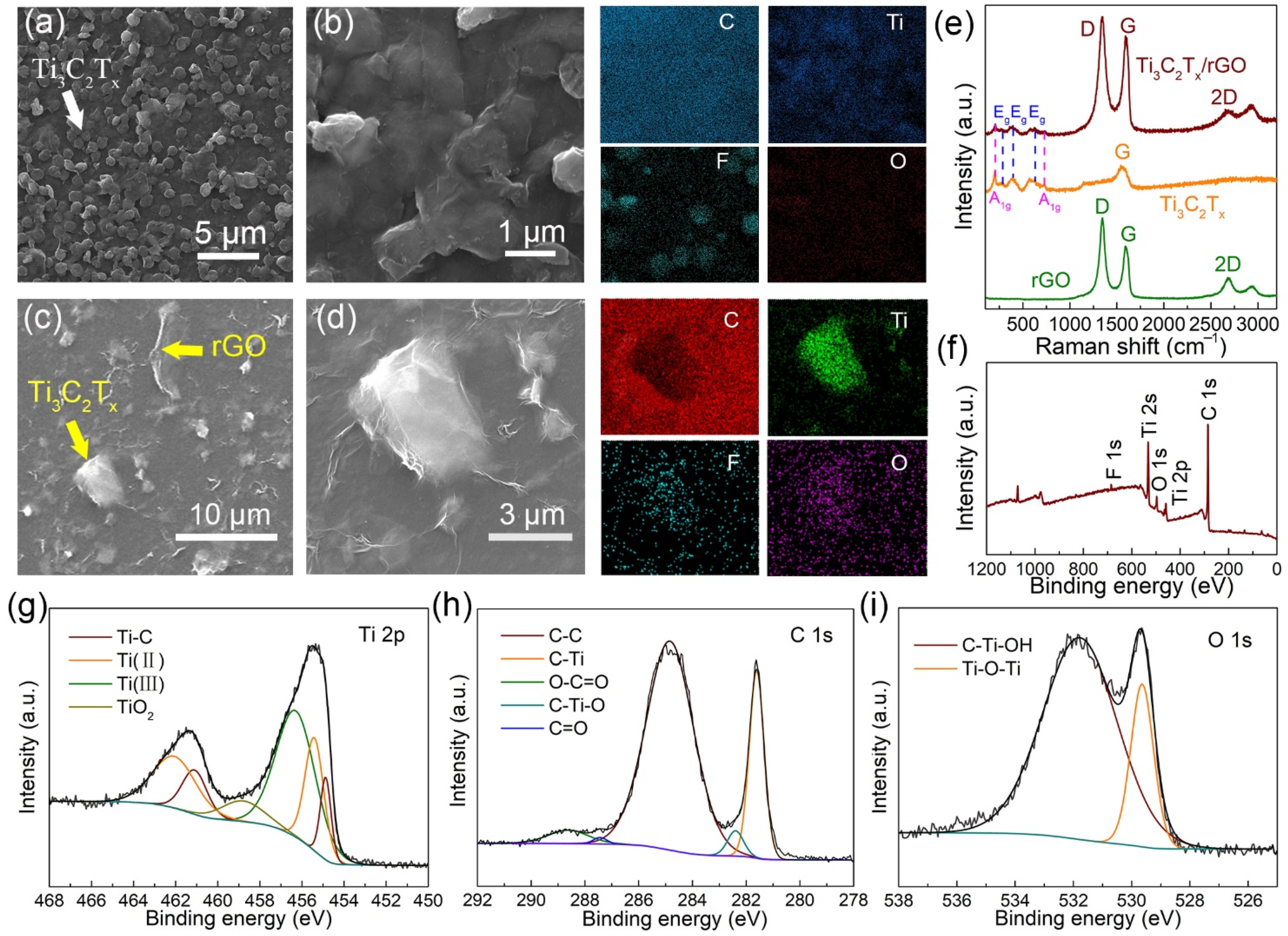
3.1. Characterization of Ti3C2Tx/rGO Nanocomposite
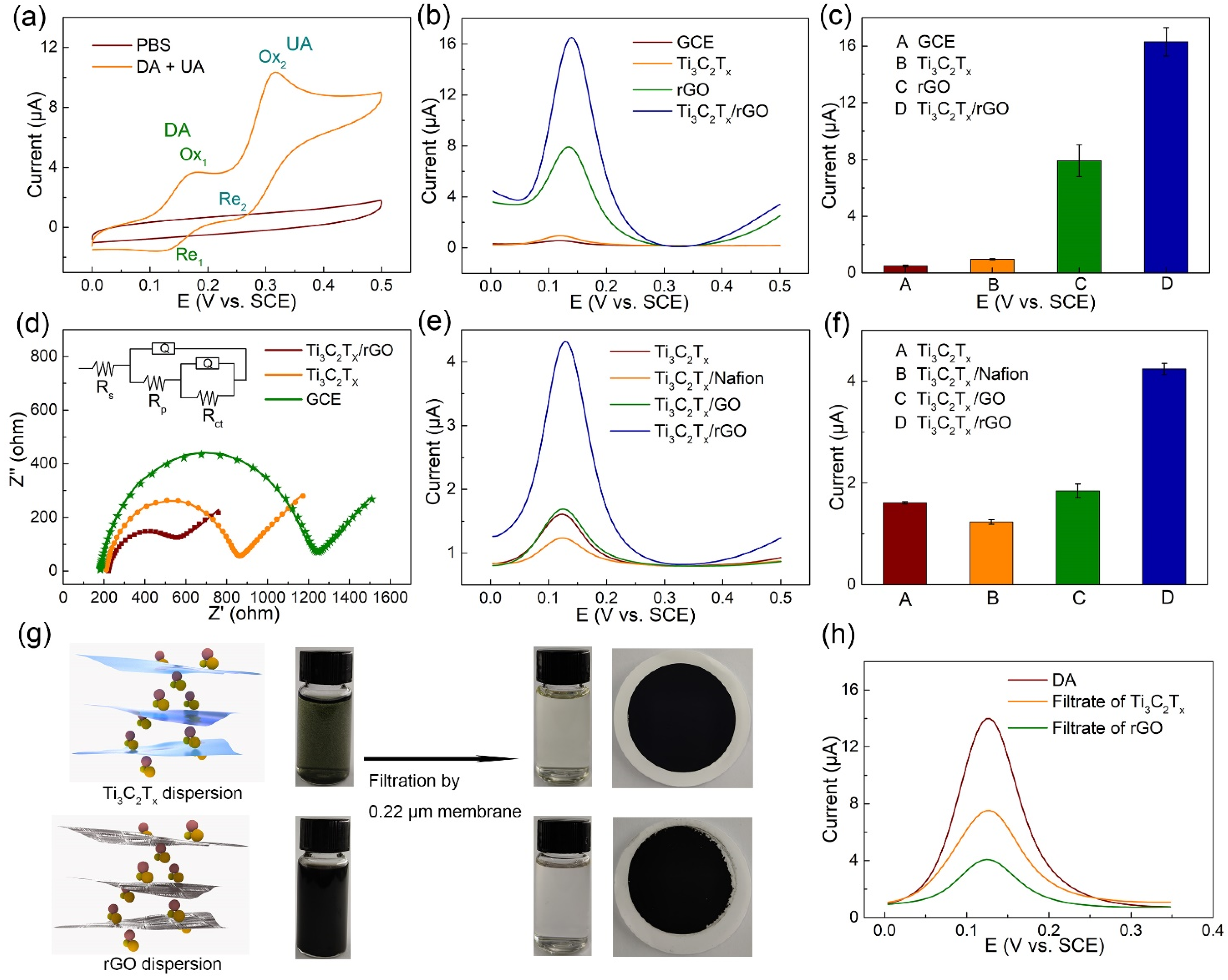
3.2. Electrochemical Collaboration Behavior of Ti3C2Tx/rGO towards DA
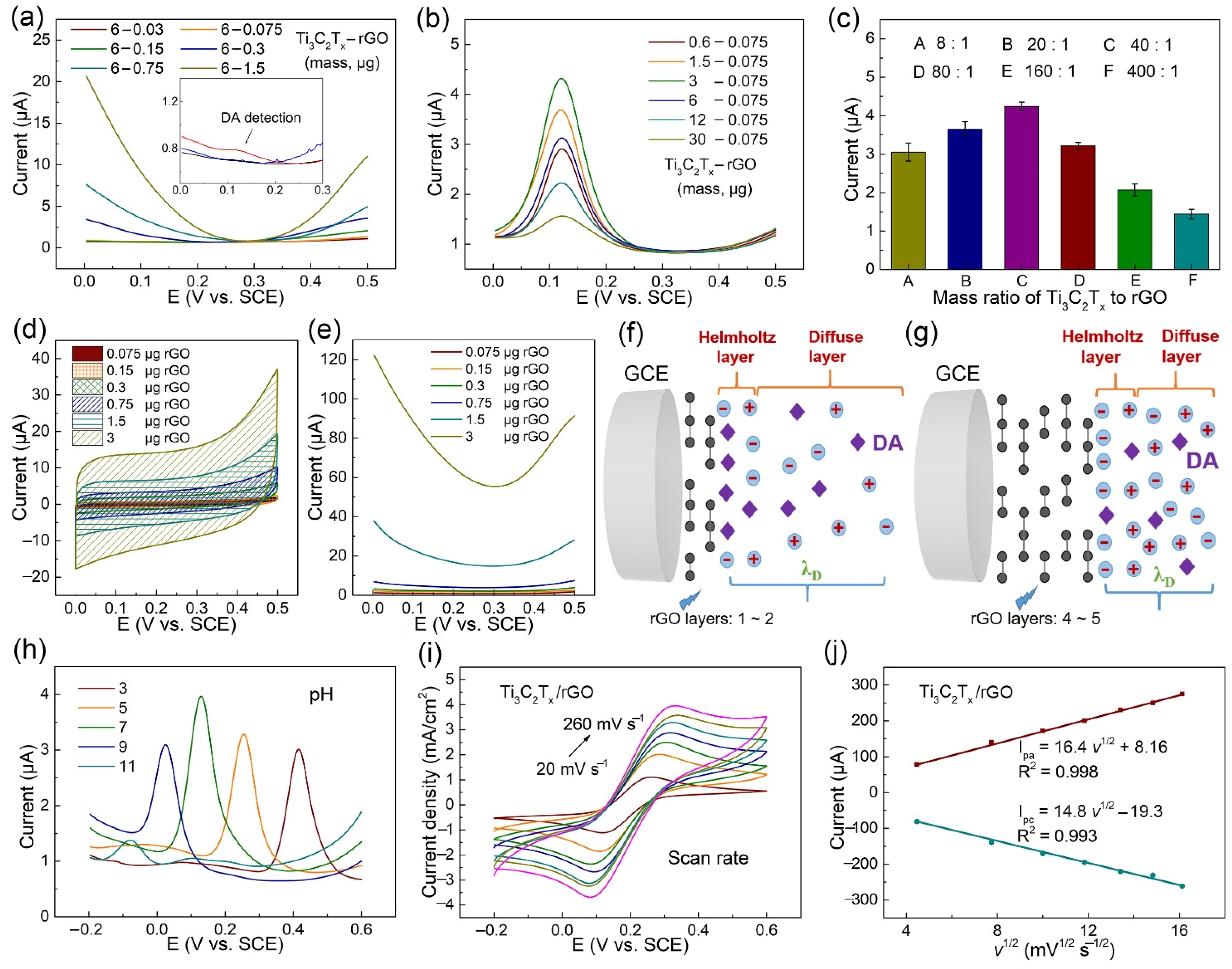
3.3. Ti3C2Tx/rGO Electrode Performance Optimization of DA Detection
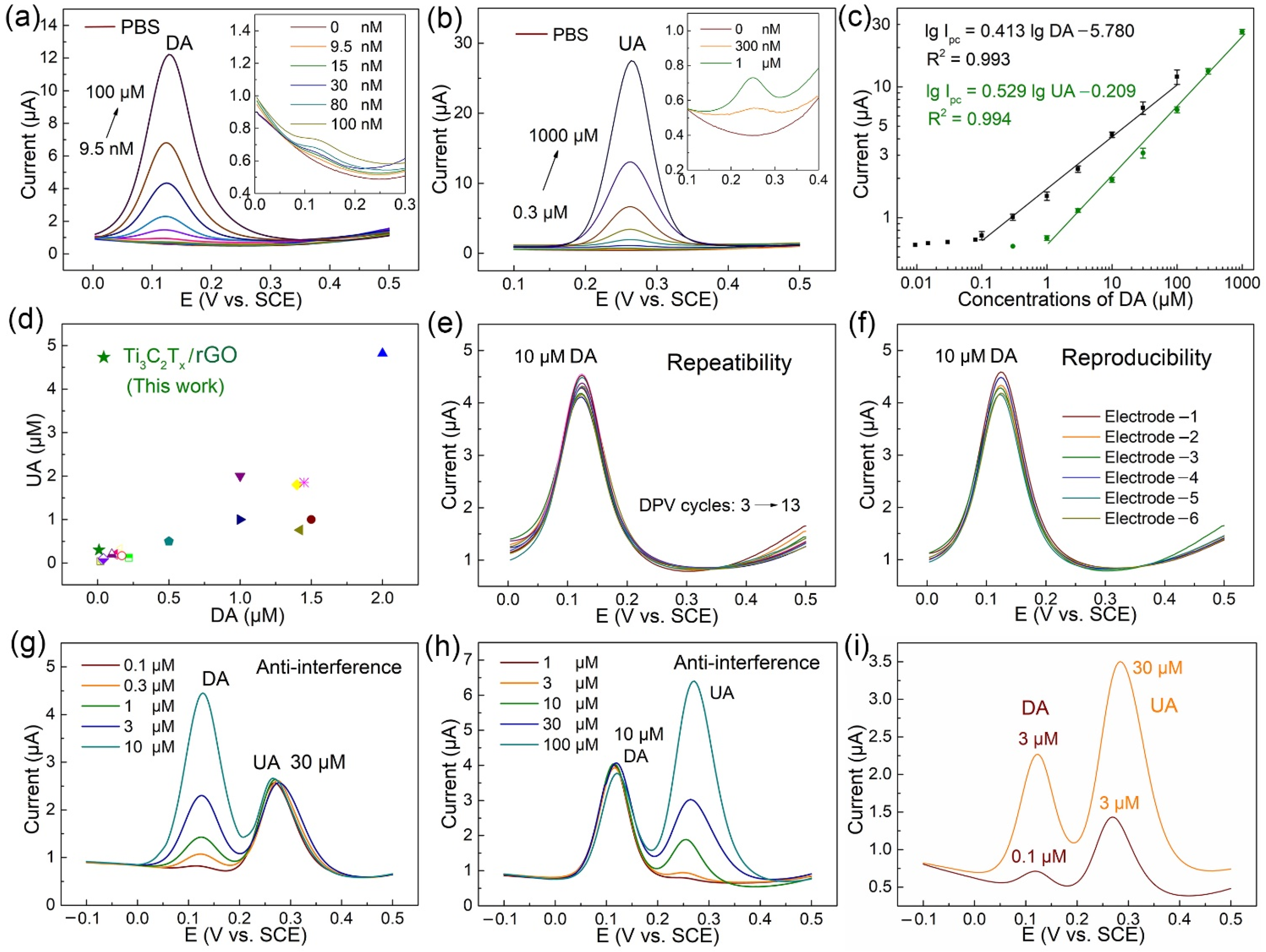
3.4. Electrochemical Determination of DA and UA with Different Concentrations
3.5. Repeatability, Reproducibility, Interference, and Real Sample Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, X.; Gu, S.Q.; Ding, Y.P. Simultaneous detection of roxithromycin and dopamine using a sensor platform based on poly(sulfosalicylic acid) and its application in human serum studies. Anal. Methods 2014, 6, 3316–3321. [Google Scholar] [CrossRef]
- Han, H.S.; Lee, H.K.; You, J.-M.; Jeong, H.; Jeon, S. Electrochemical biosensor for simultaneous determination of dopamine and serotonin based on electrochemically reduced GO-porphyrin. Sens. Actuators B Chem. 2014, 190, 886–895. [Google Scholar] [CrossRef]
- Meenakshi, S.; Pandian, K. Simultaneous voltammetry detection of dopamine and uric acid in pharmaceutical products and urine samples using ferrocene carboxylic acid primed nanoclay modified glassy carbon electrode. J. Electrochem. Soc. 2016, 163, B543–B555. [Google Scholar] [CrossRef]
- Ibrahim, H.; Temerk, Y. Sensitive electrochemical sensor for simultaneous determination of uric acid and xanthine in human biological fluids based on the nano-boron doped ceria modified glassy carbon paste electrode. J. Electroanal. Chem. 2016, 780, 176–186. [Google Scholar] [CrossRef]
- Ni, F.; Wang, Y.L.; Zhang, D.D.; Gao, F.; Li, M.G. Electrochemical oxidation of epinephrine and uric acid at a layered double hydroxide film modified glassy carbon electrode and its application. Electroanalysis 2010, 22, 1130–1135. [Google Scholar] [CrossRef]
- Yan, J.; Liu, S.; Zhang, Z.Q.; He, G.W.; Zhou, P.; Liang, H.Y.; Tian, L.L.; Zhou, X.M.; Jiang, H.J. Simultaneous electrochemical detection of ascorbic acid, dopamine and uric acid based on graphene anchored with Pd–Pt nanoparticles. Colloids Surf. B 2013, 111, 392–397. [Google Scholar] [CrossRef]
- Lee, H.-C.; Chen, T.-H.; Tseng, W.-L.; Lin, C.-H. Novel core etching technique of gold nanoparticles for colorimetric dopamine detection. Analyst 2012, 137, 5352–5357. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.H.; Teshima, N.; Hasebe, T.; Kurihara, M.; Kawashima, T. Flow-injection determination of trace amounts of dopamine by chemiluminescence detection. Talanta 1999, 50, 677–683. [Google Scholar] [CrossRef]
- Zhao, H.X.; Mu, H.; Bai, Y.H.; Yu, H.; Hu, Y.M. A rapid method for the determination of dopamine in porcine muscle by pre-column derivatization and HPLC with fluorescence detection. J. Pharm. Anal. 2011, 1, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y.; Qv, S.F.; Wang, Z.L.; Cheng, J.K. Determination of dopamine in single rat pheochromocytoma cell by capillary electrophoresis with amperometric detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2003, 792, 381–385. [Google Scholar] [CrossRef]
- Li, Q.; Wu, J.T.; Liu, Y.; Qi, X.M.; Jin, H.G.; Yang, C.; Liu, J.; Li, G.L.; He, Q.G. Recent advances in black phosphorus-based electrochemical sensors: A review. Anal. Chim. Acta 2021, 1170, 338480. [Google Scholar] [CrossRef]
- Li, G.L.; Xia, Y.H.; Tian, Y.L.; Wu, Y.Y.; Liu, J.; He, Q.G.; Chen, D.C. Review—Recent developments on graphene-based electrochemical sensors toward nitrite. J. Electrochem. Soc. 2019, 166, B881–B895. [Google Scholar] [CrossRef]
- Fu, L.; Zheng, Y.H.; Zhang, P.C.; Zhang, H.Y.; Xu, Y.T.; Zhou, J.T.; Zhang, H.W.; Hassan, K.-M.; Lai, G.S.; Zhao, S.C.; et al. Development of an electrochemical biosensor for phylogenetic analysis of Amaryllidaceae based on the enhanced electrochemical fingerprint recorded from plant tissue. Biosens. Bioelectron. 2020, 159, 112212. [Google Scholar] [CrossRef]
- Ping, J.F.; Wu, J.; Wang, Y.X.; Ying, Y.B. Simultaneous determination of ascorbic acid, dopamine and uric acid using high-performance screen-printed graphene electrode. Biosens. Bioelectron. 2012, 34, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.; Yasmin, S.; Jeon, S. Effective electrochemical detection of dopamine with highly active molybdenum oxide nanoparticles decorated on 2,6 diaminopyridine/reduced graphene oxide. Microchem. J. 2020, 153, 104501. [Google Scholar] [CrossRef]
- Feng, X.M.; Zhang, Y.; Zhou, J.H.; Li, Y.; Chen, S.F.; Zhang, L.; Ma, Y.W.; Wang, L.H.; Yan, X.H. Three-dimensional nitrogen-doped graphene as an ultrasensitive electrochemical sensor for the detection of dopamine. Nanoscale 2015, 7, 2427–2432. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.N.; Yu, L.Y.; Yan, M.X.; Ye, J.; Huang, J.S.; Yang, X.R. Holey nitrogen-doped graphene aerogel for simultaneously electrochemical determination of ascorbic acid, dopamine and uric acid. Talanta 2021, 224, 121851. [Google Scholar] [CrossRef]
- Yao, J.; Sun, Y.; Yang, M.; Duan, Y.X. Chemistry, physics and biology of graphene-based nanomaterials: New horizons for sensing, imaging and medicine. J. Mater. Chem. 2012, 22, 14313–14329. [Google Scholar] [CrossRef]
- Ma, X.Y.; Chao, M.Y.; Wang, Z.X. Electrochemical detection of dopamine in the presence of epinephrine, uric acid and ascorbic acid using a graphene-modified electrode. Anal. Methods 2012, 4, 1687–1692. [Google Scholar] [CrossRef]
- Lin, C.-T.; Lee, C.-Y.; Chiu, H.-T.; Chin, T.-S. Graphene Structure in Carbon Nanocones and Nanodiscs. Langmuir 2007, 23, 12806–12810. [Google Scholar] [CrossRef]
- Loan, P.T.K.; Wu, D.Q.; Ye, C.; Li, X.Q.; Tra, V.T.; Wei, Q.P.; Fu, L.; Yu, A.M.; Li, L.-J.; Lin, C.-T. Hall effect biosensors with ultraclean graphene film for improved sensitivity of label-free DNA detection. Biosens. Bioelectron. 2018, 99, 85–91. [Google Scholar] [CrossRef]
- Sun, H.Y.; Chen, D.; Ye, C.; Li, X.M.; Dai, D.; Yuan, Q.L.; Chee, K.W.A.; Zhao, P.; Jiang, N.; Lin, C.-T. Large-area self-assembled reduced graphene oxide/electrochemically exfoliated graphene hybrid films for transparent electrothermal heaters. Appl. Surf. Sci. 2018, 435, 809–814. [Google Scholar] [CrossRef]
- Kim, Y.R.; Bong, S.; Kang, Y.-J.; Yang, Y.; Mahajan, R.K.; Kim, J.S.; Kim, H. Electrochemical detection of dopamine in the presence of ascorbic acid using graphene modified electrodes. Biosens. Bioelectron. 2010, 25, 2366–2369. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.P.; Zhao, B.; Tang, H.Q.; Jiang, X.Q. Determination of ascorbic acid, dopamine, and uric acid by a novel electrochemical sensor based on pristine graphene. Electrochim. Acta 2015, 161, 395–402. [Google Scholar] [CrossRef]
- Gao, F.; Cai, X.L.; Wang, X.; Gao, C.; Liu, S.L.; Gao, F.; Wang, Q.X. Highly sensitive and selective detection of dopamine in the presence of ascorbic acid at graphene oxide modified electrode. Sens. Actuators B Chem. 2013, 186, 380–387. [Google Scholar] [CrossRef]
- Saha, B.; Mondal, A.C.; Basu, S.; Dasgupta, P.S. Circulating dopamine level, in lung carcinoma patients, inhibits proliferation and cytotoxicity of CD4+ and CD8+ T cells by D1 dopamine receptors: An in vitro analysis. Int. Immunopharmacol. 2001, 1, 1363–1374. [Google Scholar] [CrossRef]
- Wang, C.Q.; Du, J.; Wang, H.W.; Zou, C.; Jiang, F.X.; Yang, P.; Du, Y.K. A facile electrochemical sensor based on reduced gra phene oxide and Au nanoplates modified glassy carbon electrode for simultaneous detection of ascorbic acid, dopamine and uric acid. Sens. Actuators B Chem. 2014, 204, 302–309. [Google Scholar] [CrossRef]
- Sun, C.-L.; Lee, H.-H.; Yang, J.-M.; Wu, C.-C. The simultaneous electrochemical detection of ascorbic acid, dopamine, and uric acid using graphene/size-selected Pt nanocomposites. Biosens. Bioelectron. 2011, 26, 3450–3455. [Google Scholar] [CrossRef]
- Yang, Y.J.; Li, W.K. CTAB functionalized graphene oxide/multiwalled carbon nanotube composite modified electrode for the simultaneous determination of ascorbic acid, dopamine, uric acid and nitrite. Biosens. Bioelectron. 2014, 56, 300–306. [Google Scholar] [CrossRef]
- Kang, R.Y.; Zhang, Z.Y.; Guo, L.C.; Cui, J.F.; Chen, Y.P.; Hou, X.; Wang, B.; Lin, C.-T.; Jiang, N.; Yu, J.H. Enhanced thermal conductivity of epoxy composites filled with 2D transition metal carbides (MXenes) with ultralow loading. Sci. Rep. 2019, 9, 9135. [Google Scholar] [CrossRef]
- Naguib, M.; Come, J.; Dyatkin, B.; Presser, V.; Taberna, P.-L.; Simon, P.; Barsoum, M.W.; Gogotsi, Y. MXene: A promising transition metal carbide anode for lithium-ion batteries. Electrochem. Commun. 2012, 16, 61–64. [Google Scholar] [CrossRef]
- Rasheed, P.A.; Pandey, R.P.; Rasool, K.; Mahmoud, K.A. Ultra-sensitive electrocatalytic detection of bromate in drinking water based on Nafion/Ti3C2Tx (MXene) modified glassy carbon electrode. Sens. Actuators B Chem. 2018, 265, 652–659. [Google Scholar] [CrossRef]
- Shan, Q.M.; Mu, X.P.; Alhabeb, M.; Shuckb, C.E.; Panga, D.; Zhao, X.; Chu, X.-F.; Wei, Y.J.; Du, F.; Chen, G.; et al. Two-dimensional vanadium carbide (V2C) MXene as electrode for supercapacitors with aqueous electrolytes. Electrochem. Commun. 2018, 96, 103–107. [Google Scholar] [CrossRef]
- Rakhi, R.B.; Nayak, P.; Xia, C.; Alshareef, H.N. Novel amperometric glucose biosensor based on MXene nanocomposite. Sci. Rep. 2016, 6, 36422. [Google Scholar] [CrossRef] [PubMed]
- Lorencova, L.; Bertok, T.; Filip, J.; Jerigova, M.; Velic, D.; Kasak, P.; Mahmoud, K.A.; Tkac, J. Highly stable Ti3C2Tx (MXene)/Pt nanoparticles-modified glassy carbon electrode for H2O2 and small molecules sensing applications. Sens. Actuators B Chem. 2018, 263, 360–368. [Google Scholar] [CrossRef]
- Zhu, X.L.; Liu, B.C.; Hou, H.J.; Huang, Z.Y.; Zeinu, K.M.; Huang, L.; Yuan, X.Q.; Guo, D.B.; Hua, J.P.; Yang, J.K. Alkaline intercalation of Ti3C2 MXene for simultaneous electrochemical detection of Cd(II), Pb(II), Cu(II) and Hg(II). Electrochim. Acta 2017, 248, 46–57. [Google Scholar] [CrossRef]
- Murugan, N.; Jerome, R.; Preethika, M.; Sundaramurthy, A.; Sundramoorthy, A.K. 2D-titanium carbide (MXene) based selective electrochemical sensor for simultaneous detection of ascorbic acid, dopamine and uric acid. J. Mater. Sci. Technol. 2021, 72, 122–131. [Google Scholar] [CrossRef]
- Zhou, T.Z.; Wu, C.; Wang, Y.L.; Tomsia, A.P.; Li, M.Z.; Saiz, E.; Fang, S.L.; Baughman, R.H.; Jiang, L.; Cheng, Q.F. Super-tough MXene-functionalized graphene sheets. Nat. Commun. 2020, 11, 2077. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhao, X.S. Carbon-based materials as supercapacitor electrodes. Chem. Soc. Rev. 2009, 38, 2520–2531. [Google Scholar] [CrossRef]
- Huang, W.G.; Diallo, A.K.; Dailey, J.L.; Besar, K.; Katz, H.E. Electrochemical processes and mechanistic aspects of field-effect sensors for biomolecules. J. Mater. Chem. C 2015, 3, 6445–6470. [Google Scholar] [CrossRef]
- Yue, H.Y.; Huang, S.; Chang, J.; Heo, C.; Yao, F.; Adhikari, S.; Gunes, F.; Liu, L.C.; Lee, T.H.; Oh, E.S.; et al. ZnO nanowire arrays on 3D hierachical graphene foam: Biomarker detection of Parkinson’s disease. ACS Nano 2014, 8, 1639–1646. [Google Scholar] [CrossRef]
- Shi, K.M.; Sun, B.; Huang, X.Y.; Jiang, P.K. Synergistic effect of graphene nanosheet and BaTiO3 nanoparticles on performance enhancement of electrospun PVDF nanofiber mat for flexible piezoelectric nanogenerators. Nano Energy 2018, 52, 153–162. [Google Scholar] [CrossRef]
- Fan, Z.M.; Wang, Y.S.; Xie, Z.M.; Wang, D.L.; Yuan, Y.; Kang, H.J.; Su, B.L.; Cheng, Z.J.; Liu, Y.Y. Modified MXene/holey graphene films for advanced supercapacitor electrodes with superior energy storage. Adv. Sci. 2018, 5, 1800750. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Liu, N.S.; Ma, Y.N.; Wang, S.L.; Liu, W.J.; Luo, C.; Zhang, H.; Cheng, F.; Rao, J.Y.; Hu, X.K.; et al. Highly selfhealable 3D microsupercapacitor with MXene-graphene composite aerogel. ACS Nano 2018, 12, 4224–4232. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Mathis, T.S.; Kurra, N.; Sarycheva, A.; Xiao, X.; Hedhili, M.N.; Jiang, Q.; Alshareef, H.N.; Xu, B.M.; Pan, F.; et al. Tuning the electrochemical performance of titanium carbide MXene by controllable in situ anodic oxidation. Angew. Chem. 2019, 131, 18013–18019. [Google Scholar] [CrossRef]
- Xu, C.; Yang, F.; Deng, B.J.; Che, S.; Yang, W.; Zhang, G.; Sun, Y.K.; Li, Y.F. RGO-wrapped Ti3C2/TiO2 nanowires as a highly efficient photocatalyst for simultaneous reduction of Cr(VI) and degradation of RhB under visible light irradiation. J. Alloys Compd. 2021, 874, 159865. [Google Scholar] [CrossRef]
- Tran, N.M.; Ta, Q.T.H.; Noh, J.-S. rGO/Ti3C2Tx heterostructures for the efficient, room-temperature detection of multiple toxic gases. Mater. Chem. Phys. 2021, 273, 125087. [Google Scholar] [CrossRef]
- Borowiec, J.; Wang, R.; Zhu, L.; Zhang, J.D. Synthesis of nitrogen-doped graphene nanosheets decorated with gold nanoparticles as an improved sensor for electrochemical determination of chloramphenicol. Electrochim. Acta 2013, 99, 138–144. [Google Scholar] [CrossRef]
- Yang, L.; Liu, D.; Huang, J.S.; You, T.Y. Simultaneous determination of dopamine, ascorbic acid and uric acid at electrochemically reduced graphene oxide modified electrode. Sens. Actuators B Chem. 2014, 193, 166–172. [Google Scholar] [CrossRef]
- Oldham, K.B. A Gouy–Chapman–Stern model of the double layer at a (metal)/(ionic liquid) interface. J. Electroanal. Chem. 2008, 613, 131–138. [Google Scholar] [CrossRef]
- Allagui, A.; Benaoum, H.; Olendski, O. On the Gouy–Chapman–Stern model of the electrical double-layer structure with a generalized Boltzmann factor. Physica A 2021, 582, 126252. [Google Scholar] [CrossRef]
- Guo, Z.; Huang, G.Q.; Li, J.; Wang, Z.Y.; Xu, X.F. Graphene oxide-Ag/poly-l-lysine modified glassy carbon electrode as an electrochemical sensor for the determination of dopamine in the presence of ascorbic acid. J. Electroanal. Chem. 2015, 759, 113–121. [Google Scholar] [CrossRef]
- Erdogdu, G.; Mutlu, M.M. Selective detection of dopamine in the presence of ascorbic acid at poly(m-aminobenzene sulfonic acid). Am. J. Anal. Chem. 2011, 2, 582–588. [Google Scholar] [CrossRef][Green Version]
- Baikeli, Y.; Mamat, X.; He, F.; Xin, X.L.; Li, Y.T.; Aisa, H.A.; Hu, G.Z. Electrochemical determination of chloramphenicol and metronidazole by using a glassy carbon electrode modified with iron, nitrogen co-doped nanoporous carbon derived from a metal-organic framework (type Fe/ZIF-8). Ecotoxicol. Environ. Saf. 2020, 204, 111066. [Google Scholar] [CrossRef] [PubMed]
- Han, D.X.; Han, T.T.; Shan, C.S.; Ivaska, A.; Niu, L. Simultaneous determination of ascorbic acid, dopamine and uric acid with Chitosan-graphene modified electrode. Electroanalysis 2010, 22, 2001–2008. [Google Scholar] [CrossRef]
- Liu, Y.; She, P.; Gong, J.; Wu, W.P.; Xu, S.M.; Li, J.G.; Zhao, K.; Deng, A.P. A novel sensor based on electrodeposited Au–Pt bimetallic nano-clusters decorated on graphene oxide (GO)–electrochemically reduced GO for sensitive detection of dopamine and uric acid. Sens. Actuators B Chem. 2015, 221, 1542–1553. [Google Scholar] [CrossRef]
- Nayak, S.P.; Ramamurthy, S.S.; Kiran Kumar, J.K. Green synthesis of silver nanoparticles decorated reduced graphene oxide nanocomposite as an electrocatalytic platform for the simultaneous detection of dopamine and uric acid. Mater. Chem. Phys. 2020, 252, 123302. [Google Scholar] [CrossRef]
- Vinodhkumar, G.; Jose, S.P.; Lokeswarareddy, S.; Sekar, C.; Potheher, I.V.; Peter, A.C. Sensitivity enhancement in rGO/Mn3O4 hybrid nanocomposites: A modified glassy carbon electrode for the simultaneous detection of dopamine and uric acid. Synth. Met. 2021, 280, 116859. [Google Scholar] [CrossRef]
- Zou, H.L.; Li, B.L.; Luo, H.Q.; Li, N.B. A novel electrochemical biosensor based on hemin functionalized graphene oxide sheets for simultaneous determination of ascorbic acid, dopamine and uric acid. Sens. Actuators B Chem. 2015, 207, 535–541. [Google Scholar] [CrossRef]
- Feng, J.; Li, Q.; Cai, J.P.; Yang, T.; Chen, J.H.; Hou, X.M. Electrochemical detection mechanism of dopamine and uric acid on titanium nitride-reduced graphene oxide composite with and without ascorbic acid. Sens. Actuators B Chem. 2019, 298, 126872. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Liu, S. Electrochemical sensors based on nitrogen-doped reduced graphene oxide for the simultaneous detection of ascorbic acid, dopamine and uric acid. J. Alloys Compd. 2020, 842, 155873. [Google Scholar] [CrossRef]
| Modified Electrodes | Measurements | Linear Range (μM) | LOD (μM) | Ref. | ||
|---|---|---|---|---|---|---|
| DA | UA | DA | UA | |||
| rGO | DPV | 0.5–60 | 0.5–60 | 0.5 | 0.5 | [49] |
| Graphene | Amperometric | 5.0–710 | 6.0–1330 | 2.0 | 4.8 | [24] |
| Graphene | DPV | 0.5–2000 | 0.8–2500 | 0.12 | 0.2 | [14] |
| Graphene aerogel | DPV | 0.65–75 | 0.4–50 | 0.22 | 0.12 | [17] |
| CNTs/GO | DPV | 5.0–500 | 3.0–60 | 1.5 | 1.0 | [29] |
| Chitosan/Graphene | DPV | 1.0–24 | 2.0–45 | 1.0 | 2.0 | [55] |
| Au/rGO | DPV | 6.8–41 | 8.8–53 | 1.4 | 1.8 | [27] |
| Au/Pt/GO/rGO | DPV | 0.07–49,800 | 0.13–82,800 | 0.02 | 0.04 | [56] |
| Pt NPs/Graphene | DPV | 0.03–8.13 | 0.05–11.9 | 0.03 | 0.05 | [28] |
| Ag/rGO | DPV | 10–70 | 10–130 | 1.0 | 1.0 | [57] |
| Pd/Pt/rGO | DPV | 4–200 | 4–400 | 0.04 | 0.1 | [6] |
| Mn3O4/rGO | SWV a | 1–600 | 1–600 | 1.42 | 0.76 | [58] |
| Hemin/GO | DPV | 0.5–40 | 0.5–50 | 0.17 | 0.17 | [59] |
| TiN/rGO | DPV | 5–175 | 30–215 | 0.16 | 0.35 | [60] |
| N-doped rGO | DPV | 1–60 | 1–30 | 0.1 | 0.2 | [61] |
| Ti3C2Tx/rGO | DPV | 0.1–100 | 1–1000 | 0.0095 | 0.3 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Y.; Tian, Q.; Li, X.; Wu, L.; Yu, A.; Lai, G.; Fu, L.; Wei, Q.; Dai, D.; Jiang, N.; et al. A Double-Deck Structure of Reduced Graphene Oxide Modified Porous Ti3C2Tx Electrode towards Ultrasensitive and Simultaneous Detection of Dopamine and Uric Acid. Biosensors 2021, 11, 462. https://doi.org/10.3390/bios11110462
Zhu Y, Tian Q, Li X, Wu L, Yu A, Lai G, Fu L, Wei Q, Dai D, Jiang N, et al. A Double-Deck Structure of Reduced Graphene Oxide Modified Porous Ti3C2Tx Electrode towards Ultrasensitive and Simultaneous Detection of Dopamine and Uric Acid. Biosensors. 2021; 11(11):462. https://doi.org/10.3390/bios11110462
Chicago/Turabian StyleZhu, Yangguang, Qichen Tian, Xiufen Li, Lidong Wu, Aimin Yu, Guosong Lai, Li Fu, Qiuping Wei, Dan Dai, Nan Jiang, and et al. 2021. "A Double-Deck Structure of Reduced Graphene Oxide Modified Porous Ti3C2Tx Electrode towards Ultrasensitive and Simultaneous Detection of Dopamine and Uric Acid" Biosensors 11, no. 11: 462. https://doi.org/10.3390/bios11110462
APA StyleZhu, Y., Tian, Q., Li, X., Wu, L., Yu, A., Lai, G., Fu, L., Wei, Q., Dai, D., Jiang, N., Li, H., Ye, C., & Lin, C.-T. (2021). A Double-Deck Structure of Reduced Graphene Oxide Modified Porous Ti3C2Tx Electrode towards Ultrasensitive and Simultaneous Detection of Dopamine and Uric Acid. Biosensors, 11(11), 462. https://doi.org/10.3390/bios11110462









