Two-Dimensional Hybrid Composites of SnS2 Nanosheets Array Film with Graphene for Enhanced Photoelectric Performance
Abstract
1. Introduction
2. Experimental Methods
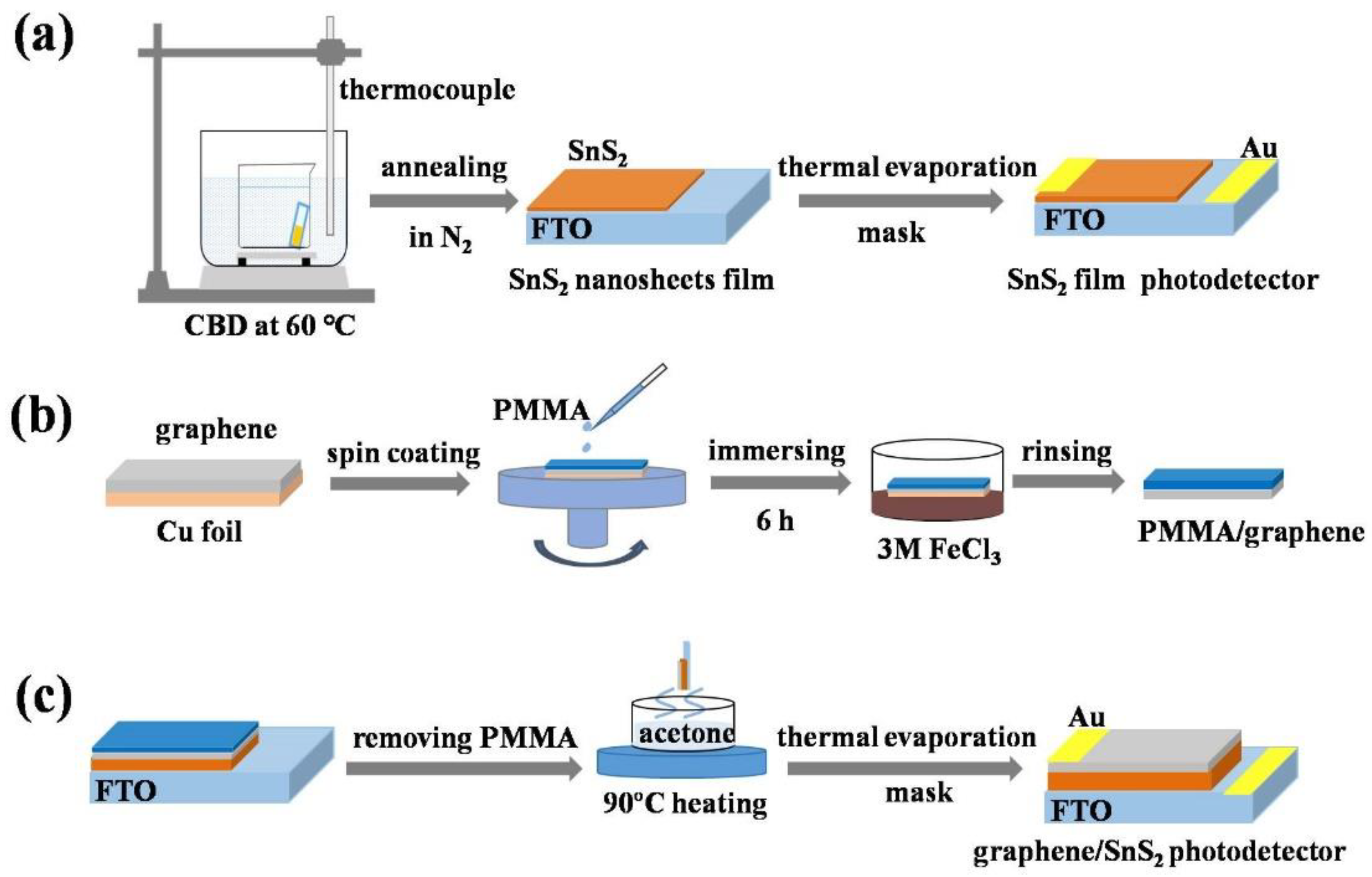
2.1. Synthesis of SnS2 Nanosheets Array Film
2.2. Preparation of Graphene/SnS2 Heterojunction
2.3. Characterization
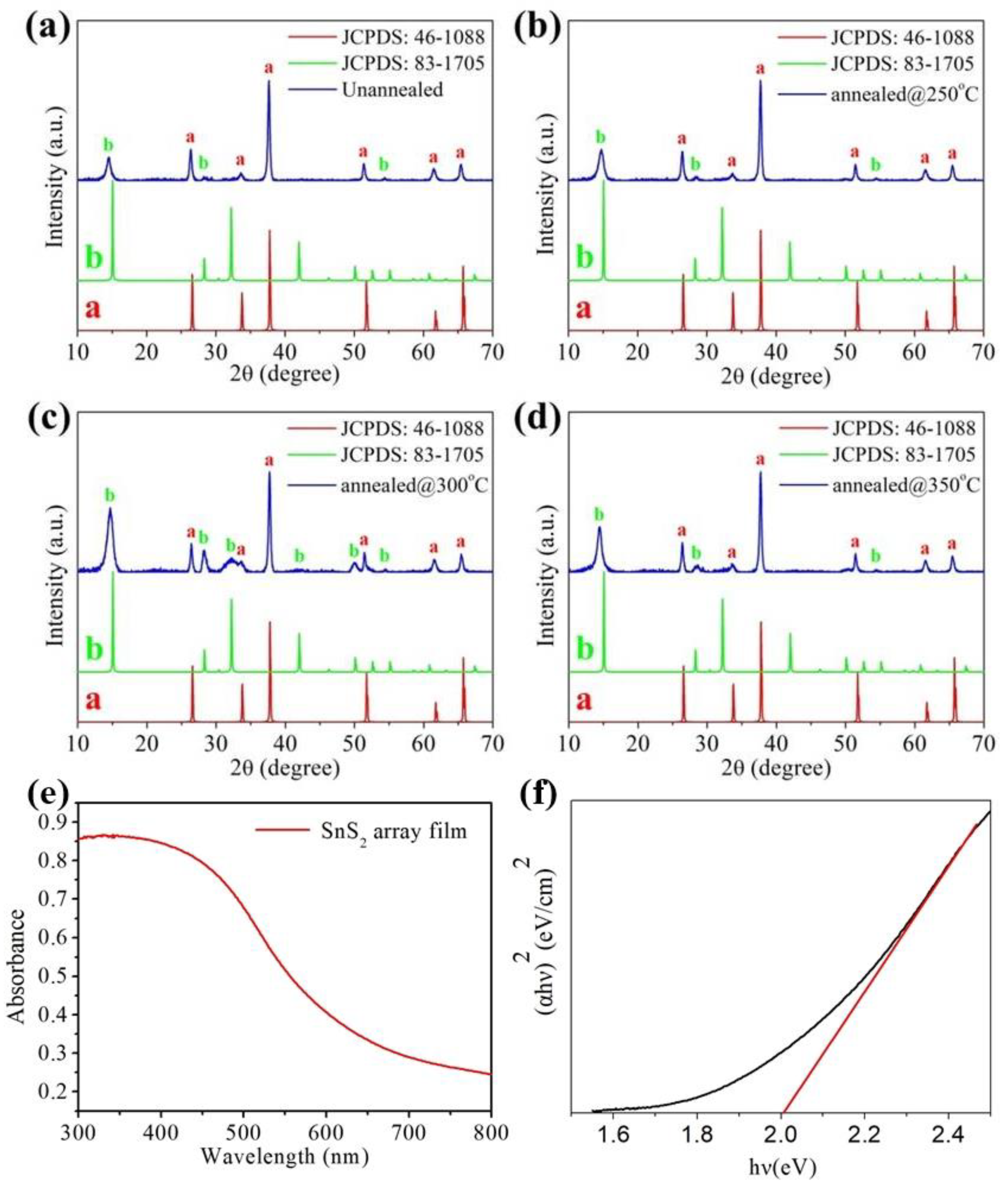
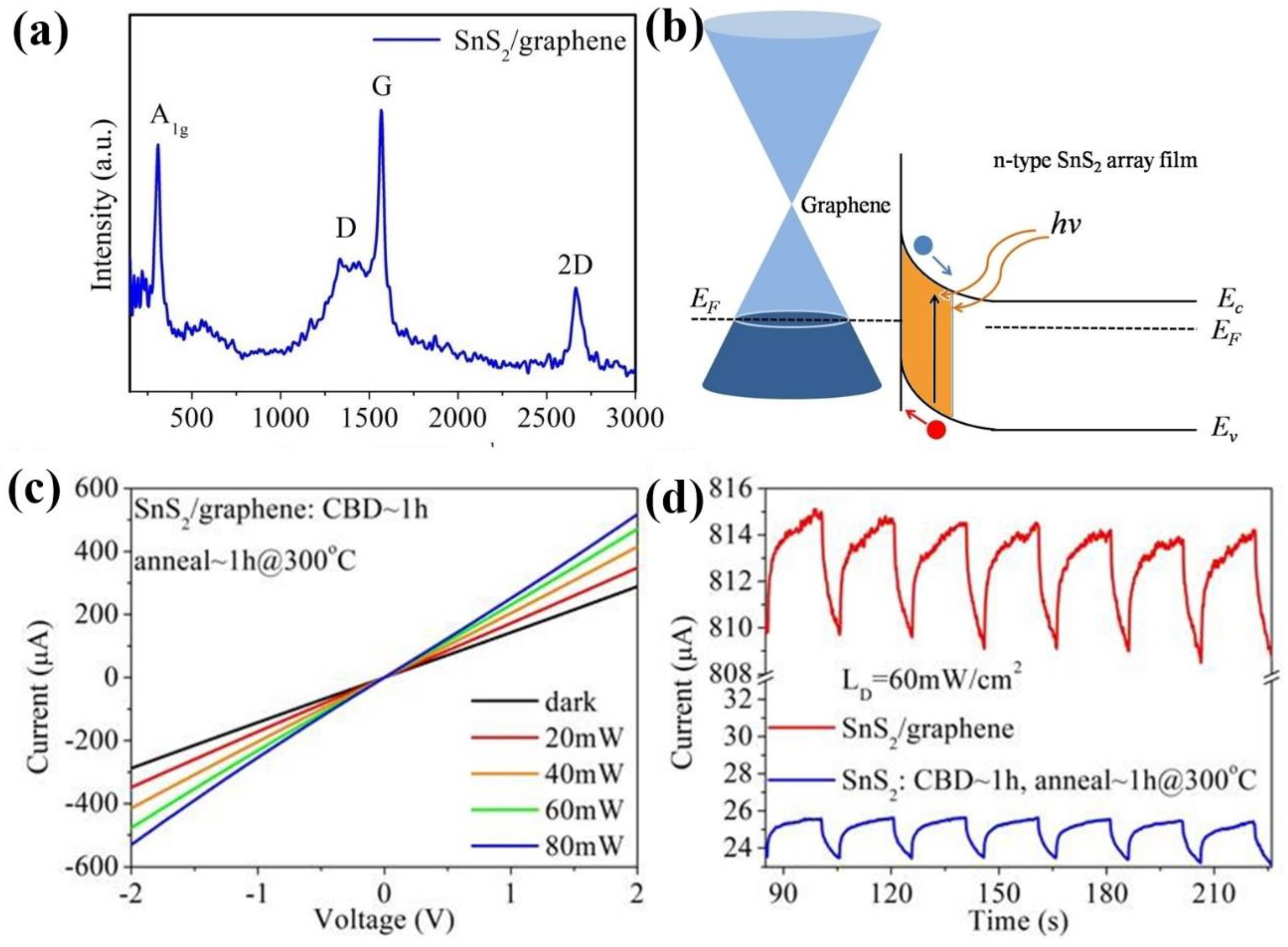
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Novoselov, K.S.; Fal’Ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Mueller, T.; Lin, Y.M.; Valdes-Garcia, A.; Avouris, P. Ultrafast graphene photodetector. Nat. Nanotechnol. 2009, 4, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Anguita, J.V.; Ahmad, M.; Haq, S.; Allam, J.; Silva, S.R.P. Nanotechnology: Ultra-broadband light trapping using nanotextured decoupled graphene multilayers. Sci. Adv. 2016, 2, e1501238. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Huang, H.; Ning, K.; Xu, X.; Xie, G.; Qian, L.; Loh, K.P.; Tang, D. Generation of 30 fs pulses from a diode-pumped graphene mode-locked Yb:CaYAlO4 laser. Opt. Lett. 2016, 41, 890–893. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yao, H.; Li, H.; Wang, Z.; Shi, Y. Recent advances of low-dimensional materials in lasing applications. FlatChem 2018, 10, 22–38. [Google Scholar] [CrossRef]
- Liu, B.; Chen, Y.; You, C.; Liu, Y.; Kong, X.; Li, J.; Li, S.; Deng, W.; Li, Y.; Yan, H.; et al. High performance photodetector based on graphene/MoS2/graphene lateral heterostrurcture with Schottky junctions. J. Alloys Compd. 2019, 779, 140–146. [Google Scholar] [CrossRef]
- Cakmakyapan, S.; Lu, P.K.; Navabi, A.; Jarrahi, M. Gold-patched graphene nano-stripes for high-responsivity and ultrafast photodetection from the visible to infrared regime. Light Sci. Appl. 2018, 7, 20. [Google Scholar] [CrossRef]
- Xie, C.; Mak, C.; Tao, X.; Yan, F. Photodetectors Based on Two-Dimensional Layered Materials Beyond Graphene. Adv. Funct. Mater. 2017, 27, 1603886. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, L.; Zhang, Y.; Xue, Y.; Zhang, E.; Wang, H.; Kong, Z.; Xi, J.; Ji, Z. Controlled synthesis of graphene nanoribbons for field effect transistors. J. Alloys Compd. 2015, 649, 933–938. [Google Scholar] [CrossRef]
- Shi, Y.; Fang, W.; Zhang, K.; Zhang, W.; Li, L.J. Photoelectrical response in single-layer graphene transistors. Small 2009, 5, 2005–2011. [Google Scholar] [CrossRef]
- El-Kady, M.F.; Shao, Y.; Kaner, R.B. Graphene for batteries, supercapacitors and beyond. Nat. Rev. Mater. 2016, 1, 16033. [Google Scholar] [CrossRef]
- Liu, Z.; Lau, S.P.; Yan, F. Functionalized graphene and other two-dimensional materials for photovoltaic devices: Device design and processing. Chem. Soc. Rev. 2015, 44, 5638–5679. [Google Scholar] [CrossRef]
- Putri, L.K.; Ong, W.J.; Chang, W.S.; Chai, S.P. Heteroatom doped graphene in photocatalysis: A review. Appl. Surf. Sci. 2015, 358, 2–14. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Wageh, S.; Al-Ghamdi, A.A.; Xie, J. Graphene in Photocatalysis: A Review. Small 2016, 12, 6640–6696. [Google Scholar] [CrossRef]
- Li, M.; Chen, C.H.; Shi, Y.; Li, L.J. Heterostructures based on two-dimensional layered materials and their potential applications. Mater. Today 2016, 19, 322–335. [Google Scholar] [CrossRef]
- Ouyang, W.; Teng, F.; He, J.H.; Fang, X. Enhancing the Photoelectric Performance of Photodetectors Based on Metal Oxide Semiconductors by Charge-Carrier Engineering. Adv. Funct. Mater. 2019, 29, 1807672. [Google Scholar] [CrossRef]
- Danilishin, S.L.; Knyazev, E.; Voronchev, N.V.; Khalili, F.Y.; Gräf, C.; Steinlechner, S.; Hennig, J.S.; Hild, S. A new quantum speed-meter interferometer: Measuring speed to search for intermediate mass black holes article. Light Sci. Appl. 2018, 7, 11. [Google Scholar] [CrossRef]
- Liu, X.; Gu, L.; Zhang, Q.; Wu, J.; Long, Y.; Fan, Z. All-printable band-edge modulated ZnO nanowire photodetectors with ultra-high detectivity. Nat. Commun. 2014, 5, 4007. [Google Scholar] [CrossRef]
- Mueller, T.; Xia, F.; Avouris, P. Graphene photodetectors for high-speed optical communications. Nat. Photonics 2010, 4, 297–301. [Google Scholar] [CrossRef]
- Zhang, J.; Itzler, M.A.; Zbinden, H.; Pan, J.W. Advances in InGaAs/InP single-photon detector systems for quantum communication. Light Sci. Appl. 2015, 4, e286. [Google Scholar] [CrossRef]
- Nair, R.R.; Blake, P.; Grigorenko, A.N.; Novoselov, K.S.; Booth, T.J.; Stauber, T.; Peres, N.M.R.; Geim, A.K. Fine structure constant defines visual transparency of graphene. Science 2018, 320, 1308. [Google Scholar] [CrossRef]
- Chen, X.; Huang, Z.; Ren, X.; Xu, G.; Zhou, J.; Tao, Y. Photodetectors Based on SnS2/Graphene Heterostructure on Rigid and Flexible Substrates. ChemNanoMat 2018, 4, 373–378. [Google Scholar] [CrossRef]
- Li, H.; Shi, Y.; Li, L.J. Synthesis and optoelectronic applications of graphene/transition metal dichalcogenides flat-pack assembly. Carbon N. Y. 2018, 127, 602–610. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Sun, S.H.; Sun, X.J.; Zhao, Y.R.; Chen, L.; Yang, Y.; Lü, W.; Li, D.B. Plasma-induced, nitrogen-doped graphene-based aerogels for high-performance supercapacitors. Light Sci. Appl. 2016, 5, e16130. [Google Scholar] [CrossRef]
- Du, Y.; Yin, Z.; Rui, X.; Zeng, Z.; Wu, X.J.; Liu, J.; Zhu, Y.; Zhu, J.; Huang, X.; Yan, Q.; et al. A facile, relative green, and inexpensive synthetic approach toward large-scale production of SnS2 nanoplates for high-performance lithium-ion batteries. Nanoscale 2013, 5, 1456–1459. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, X.; Yan, B.; Li, D.; Lawes, S.; Sun, X. Significant impact of 2D graphene nanosheets on large volume change tin-based anodes in lithium-ion batteries: A review. J. Power Sources 2015, 274, 869–884. [Google Scholar] [CrossRef]
- Balogun, M.S.; Qiu, W.; Jian, J.; Huang, Y.; Luo, Y.; Yang, H.; Liang, C.; Lu, X.; Tong, Y. Vanadium Nitride Nanowire Supported SnS2 Nanosheets with High Reversible Capacity as Anode Material for Lithium Ion Batteries. ACS Appl. Mater. Interfaces 2015, 7, 23205–23215. [Google Scholar] [CrossRef]
- Haghighi, M.; Minbashi, M.; Taghavinia, N.; Kim, D.H.; Mahdavi, S.M.; Kordbacheh, A.A. A modeling study on utilizing SnS2 as the buffer layer of CZT(S, Se) solar cells. Sol. Energy 2018, 167, 165–171. [Google Scholar] [CrossRef]
- Zhao, E.; Gao, L.; Yang, S.; Wang, L.; Cao, J.; Ma, T. In situ fabrication of 2D SnS2 nanosheets as a new electron transport layer for perovskite solar cells. Nano Res. 2018, 11, 5913–5923. [Google Scholar] [CrossRef]
- Di, T.; Zhu, B.; Cheng, B.; Yu, J.; Xu, J. A direct Z-scheme g-C3N4/SnS2 photocatalyst with superior visible-light CO2 reduction performance. J. Catal. 2017, 352, 532–541. [Google Scholar] [CrossRef]
- Dashairya, L.; Sharma, M.; Basu, S.; Saha, P. SnS2/RGO based nanocomposite for efficient photocatalytic degradation of toxic industrial dyes under visible-light irradiation. J. Alloys Compd. 2019, 774, 625–636. [Google Scholar] [CrossRef]
- Jianzhe, L.; Congxin, X.; Honglai, L.; Anlian, P. High on/off ratio photosensitive field effect transistors based on few layer SnS2. Nanotechnology 2016, 27, 34LT01. [Google Scholar]
- Zhou, X.; Zhang, Q.; Gan, L.; Li, H.; Zhai, T. Large-Size Growth of Ultrathin SnS2 Nanosheets and High Performance for Phototransistors. Adv. Funct. Mater. 2016, 26, 4405–4413. [Google Scholar] [CrossRef]
- Jia, X.; Tang, C.; Pan, R.; Long, Y.; Gu, C.; Li, J. Thickness-Dependently Enhanced Photodetection Performance of Vertically Grown SnS2 Nanoflakes with Large Size and High Production. ACS Appl. Mater. Interfaces 2018, 10, 18073–18081. [Google Scholar] [CrossRef]
- Gao, L.; Chen, C.; Zeng, K.; Ge, C.; Yang, D.; Song, H.; Tang, J. Broadband, sensitive and spectrally distinctive SnS2 nanosheet/PbS colloidal quantum dot hybrid photodetector. Light Sci. Appl. 2016, 5, e16126. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Li, Y.; Lu, F.; Deng, H.X.; Wei, Z.; Li, J. Wavelength dependent UV-Vis photodetectors from SnS2 flakes. RSC Adv. 2015, 6, 422–427. [Google Scholar] [CrossRef]
- Su, G.; Hadjiev, V.G.; Loya, P.E.; Zhang, J.; Lei, S.; Maharjan, S.; Dong, P.; Ajayan, P.M.; Lou, J.; Peng, H. Chemical vapor deposition of thin crystals of layered semiconductor SnS2 for fast photodetection application. Nano Lett. 2015, 15, 506–513. [Google Scholar] [CrossRef]
- Tao, Y.; Wu, X.; Wang, W.; Wang, J. Flexible photodetector from ultraviolet to near infrared based on a SnS2 nanosheet microsphere film. J. Mater. Chem. C 2015, 3, 1347–1353. [Google Scholar] [CrossRef]
- Gedi, S.; Reddy, V.R.M.; Pejjai, B.; Park, C.; Jeon, C.W.; Kotte, T.R.R. Studies on chemical bath deposited SnS2 films for Cd-free thin film solar cells. Ceram. Int. 2017, 43, 3713–3719. [Google Scholar] [CrossRef]
- Ham, G.; Shin, S.; Park, J.; Lee, J.; Choi, H.; Lee, S.; Jeon, H. Engineering the crystallinity of tin disulfide deposited at low temperatures. RSC Adv. 2016, 6, 54069–54075. [Google Scholar] [CrossRef]
- Voznyi, A.; Kosyak, V.; Grase, L.; Vecstaudža, J.; Onufrijevs, P.; Yeromenko, Y.; Medvid, A.; Opanasyuk, A. Formation of SnS phase obtained by thermal vacuum annealing of SnS2 thin films and its application in solar cells. Mater. Sci. Semicond. Process. 2018, 79, 32–39. [Google Scholar] [CrossRef]
- Kang, J.; Shin, D.; Hee, B. Graphene transfer: Key for applications. Nanoscale 2012, 4, 5527–5537. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Fang, X. Synthesis and Development of Graphene–Inorganic Semiconductor Nanocomposites. Chem. Rev. 2015, 115, 8294–8343. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Kim, K.K.; Reina, A.; Hofmann, M.; Li, L.; Kong, J. Work Function Engineering of Graphene Electrode via Chemical Doping. ACS Nano 2010, 4, 2689–2694. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, P.U.; Babu, G.S.; Kumar, Y.B.K.; Jayasree, Y.; Raja, V.S. Effect of bath concentration, temperature on the growth and properties of chemical bath deposited ZnS films. Mater. Chem. Phys. 2012, 134, 1106–1112. [Google Scholar] [CrossRef]
- Kavasoglu, A.S.; Yakuphanoglu, F.; Kavasoglu, N.; Pakma, O.; Birgi, O.; Oktik, S. The analysis of the charge transport mechanism of n-Si/MEH-PPV device structure using forward bias I–V–T characteristics. J. Alloys Compd. 2010, 492, 421–426. [Google Scholar] [CrossRef]
- Gu, P.; Zhu, X.; Wu, H.; Li, J.; Yang, D. Influence of oxygen vacancy on the response properties of TiO2 ultraviolet detectors. J. Alloys Compd. 2019, 779, 821–830. [Google Scholar] [CrossRef]
- Hwang, H.J.; Chang, E.; Yoo, B.; Shim, H. A graphene barristor using nitrogen profile controlled ZnO Schottky contacts. Nanoscale 2017, 9, 2442–2448. [Google Scholar] [CrossRef]
- Sheng, J.; Fan, K.; Wang, D.; Han, C.; Fang, J.; Gao, P.; Ye, J. Improvement of the SiOx Passivation Layer for High-Efficiency Si/PEDOT: PSS Heterojunction Solar Cells. ACS Appl. Mater. Interfaces 2014, 6, 16027–16034. [Google Scholar] [CrossRef]
- Rao, C.E.E.; Sood, A.E.; Subrahmanyam, K.E.; Govindaraj, A. Graphene: The new two-dimensional nanomaterial. Angew. Chem. Int. Ed. 2009, 48, 7752–7777. [Google Scholar] [CrossRef]
- Yan, F.; Wei, Z.; Wei, X.; Lv, Q.; Zhu, W.; Wang, K. Toward High-Performance Photodetectors Based on 2D Materials: Strategy on Methods. Small Methods 2018, 2, 1700349. [Google Scholar] [CrossRef]
- Lu, J.; Xu, C.; Dai, J.; Li, J.; Wang, Y.; Lin, Y.; Li, P. Improved UV photoresponse of ZnO nanorod arrays by resonant coupling with surface plasmons of Al nanoparticles. Nanoscale 2015, 7, 3396–3403. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, F.; Li, H.; Yao, H.; Jiang, K.; Liu, Z.; Lin, C.; Chen, F.; Wang, Y.; Liu, L. Two-Dimensional Hybrid Composites of SnS2 Nanosheets Array Film with Graphene for Enhanced Photoelectric Performance. Nanomaterials 2019, 9, 1122. https://doi.org/10.3390/nano9081122
Fang F, Li H, Yao H, Jiang K, Liu Z, Lin C, Chen F, Wang Y, Liu L. Two-Dimensional Hybrid Composites of SnS2 Nanosheets Array Film with Graphene for Enhanced Photoelectric Performance. Nanomaterials. 2019; 9(8):1122. https://doi.org/10.3390/nano9081122
Chicago/Turabian StyleFang, Feier, Henan Li, Huizhen Yao, Ke Jiang, Zexiang Liu, Congjian Lin, Fuming Chen, Ye Wang, and Lai Liu. 2019. "Two-Dimensional Hybrid Composites of SnS2 Nanosheets Array Film with Graphene for Enhanced Photoelectric Performance" Nanomaterials 9, no. 8: 1122. https://doi.org/10.3390/nano9081122
APA StyleFang, F., Li, H., Yao, H., Jiang, K., Liu, Z., Lin, C., Chen, F., Wang, Y., & Liu, L. (2019). Two-Dimensional Hybrid Composites of SnS2 Nanosheets Array Film with Graphene for Enhanced Photoelectric Performance. Nanomaterials, 9(8), 1122. https://doi.org/10.3390/nano9081122




