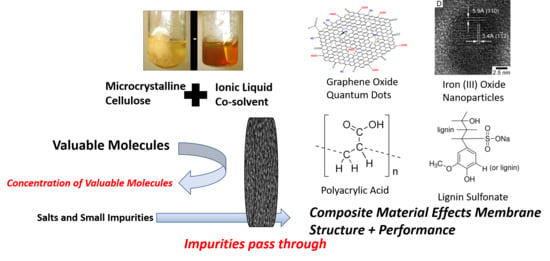
Composite Membranes Derived from Cellulose and Lignin Sulfonate for Selective Separations and Antifouling Aspects
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cellulose Composite Membranes
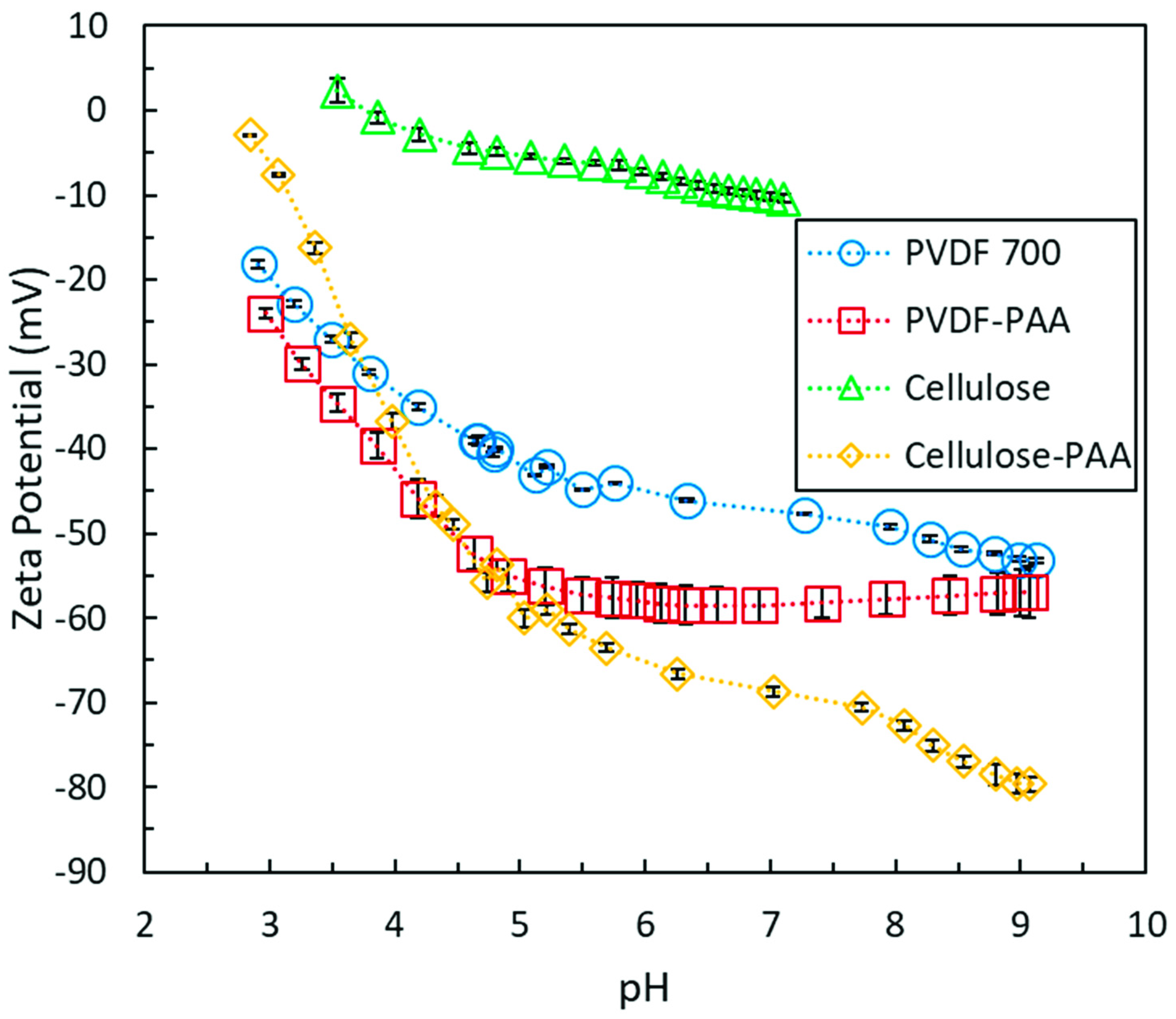
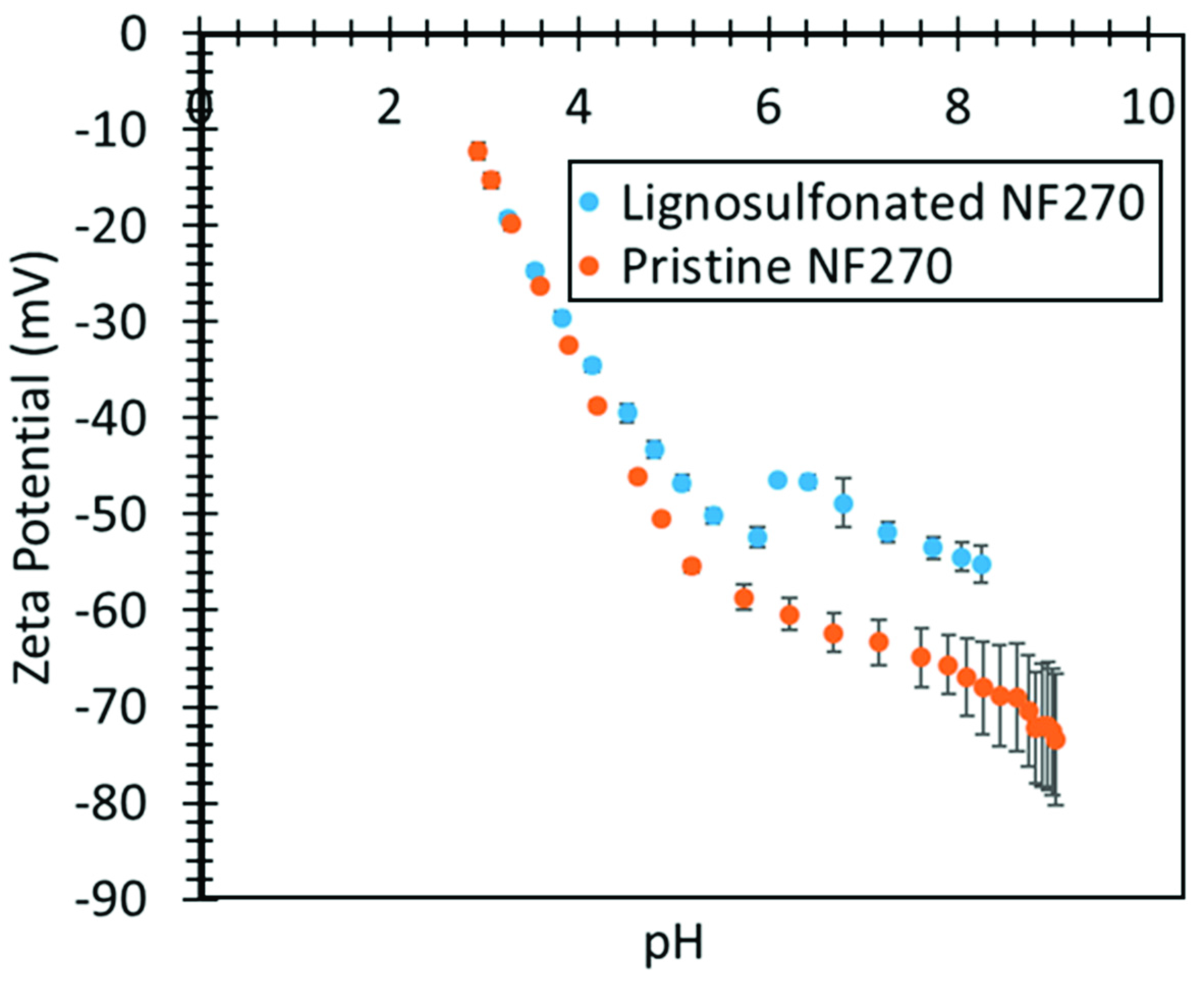
2.3. Zeta Potential Characterization
2.4. Contact Angle Characterization
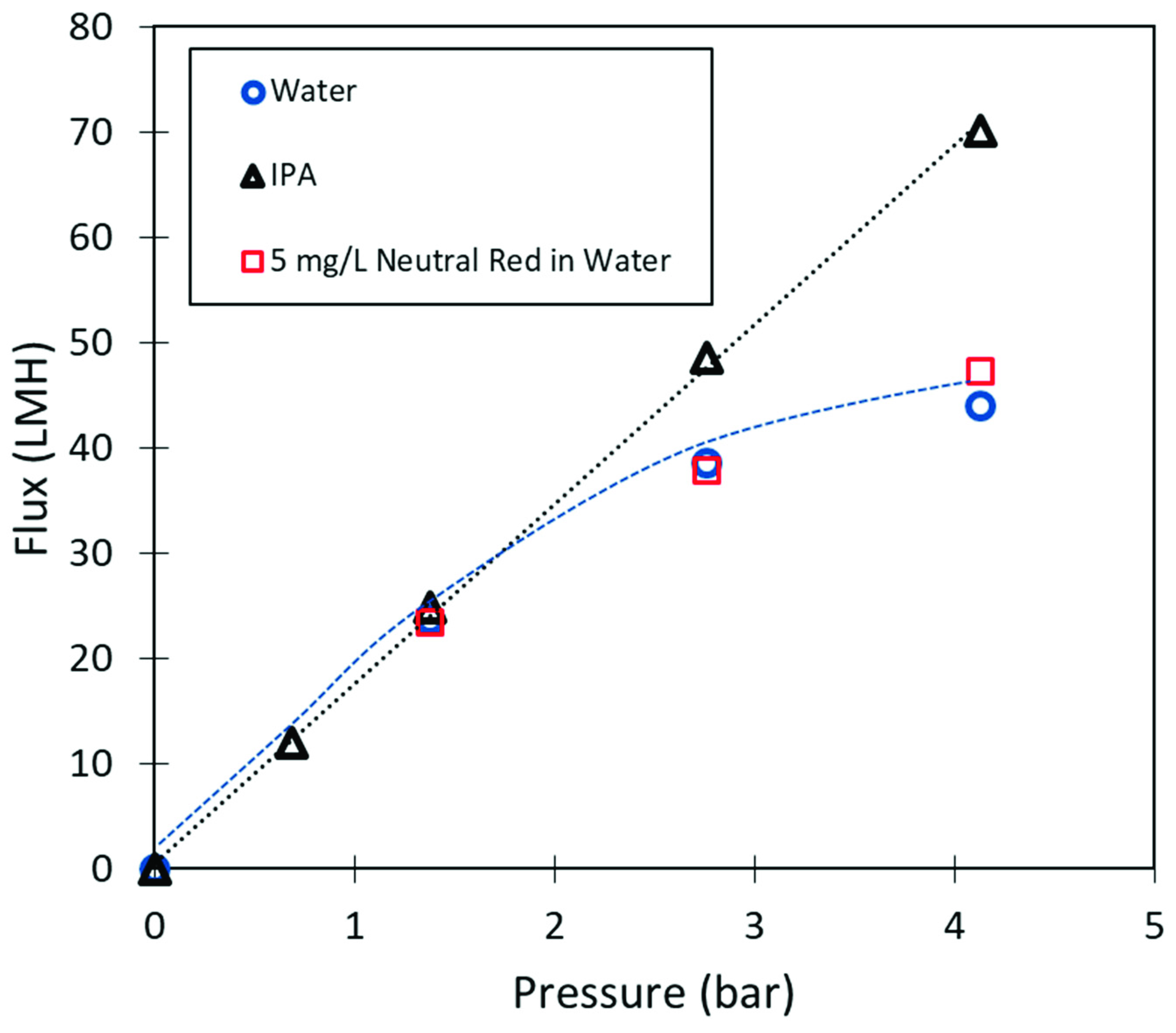
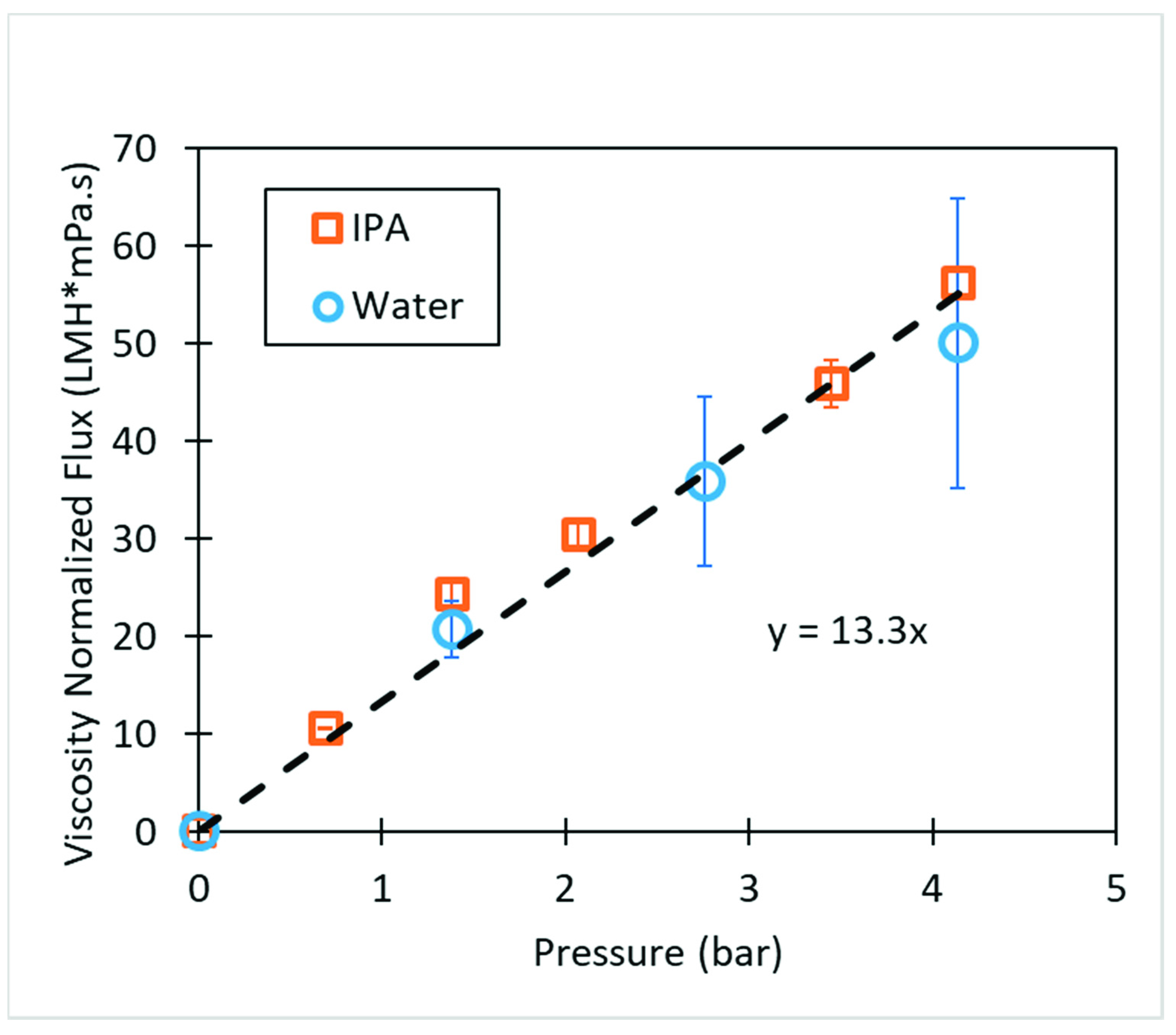
2.5. Membrane Performance
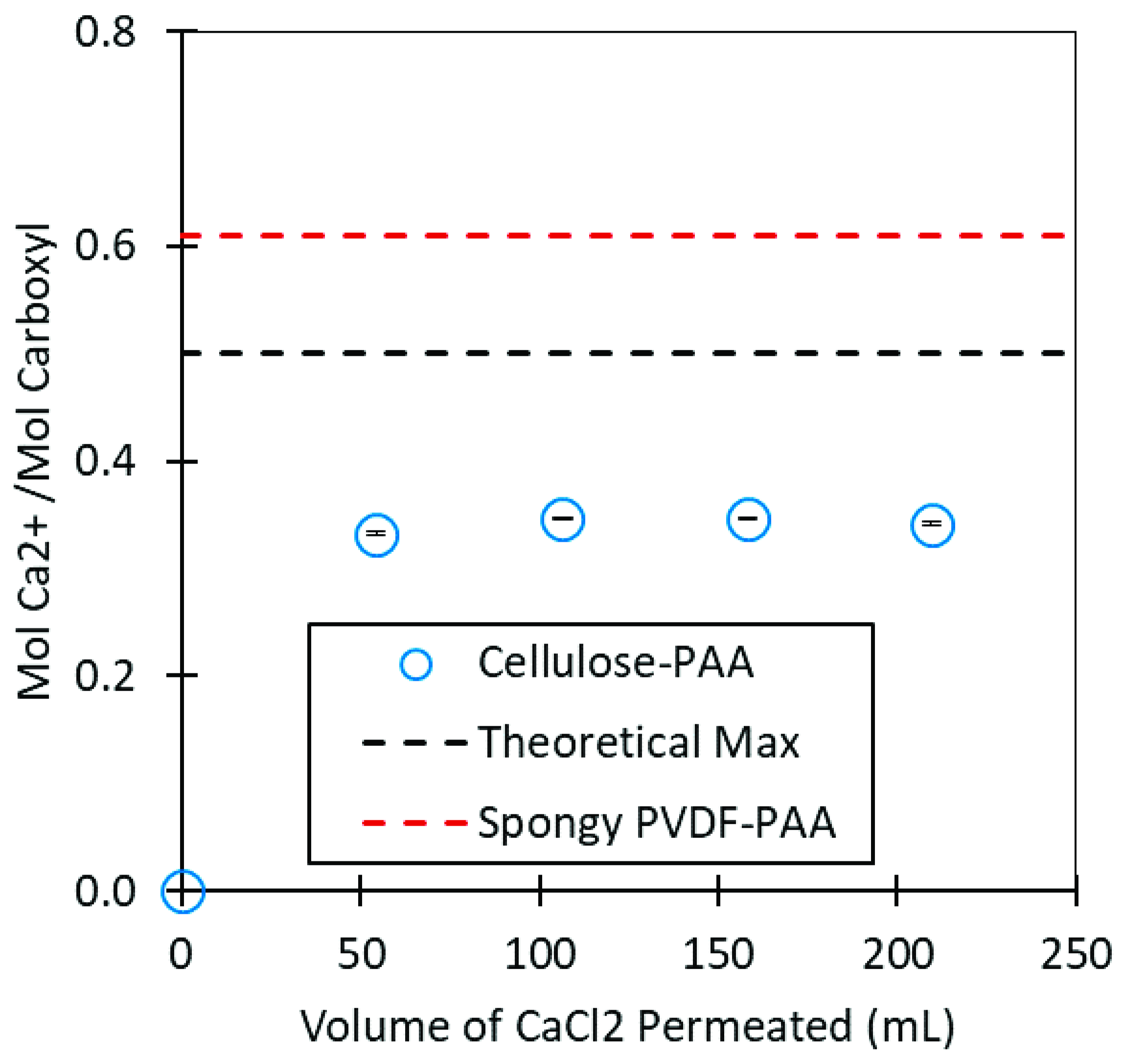
2.6. Divalent Ion Capture by Cellulose-PAA Membranes
2.7. Lignin Sulfonate Functionalized Nanofiltration Membrane
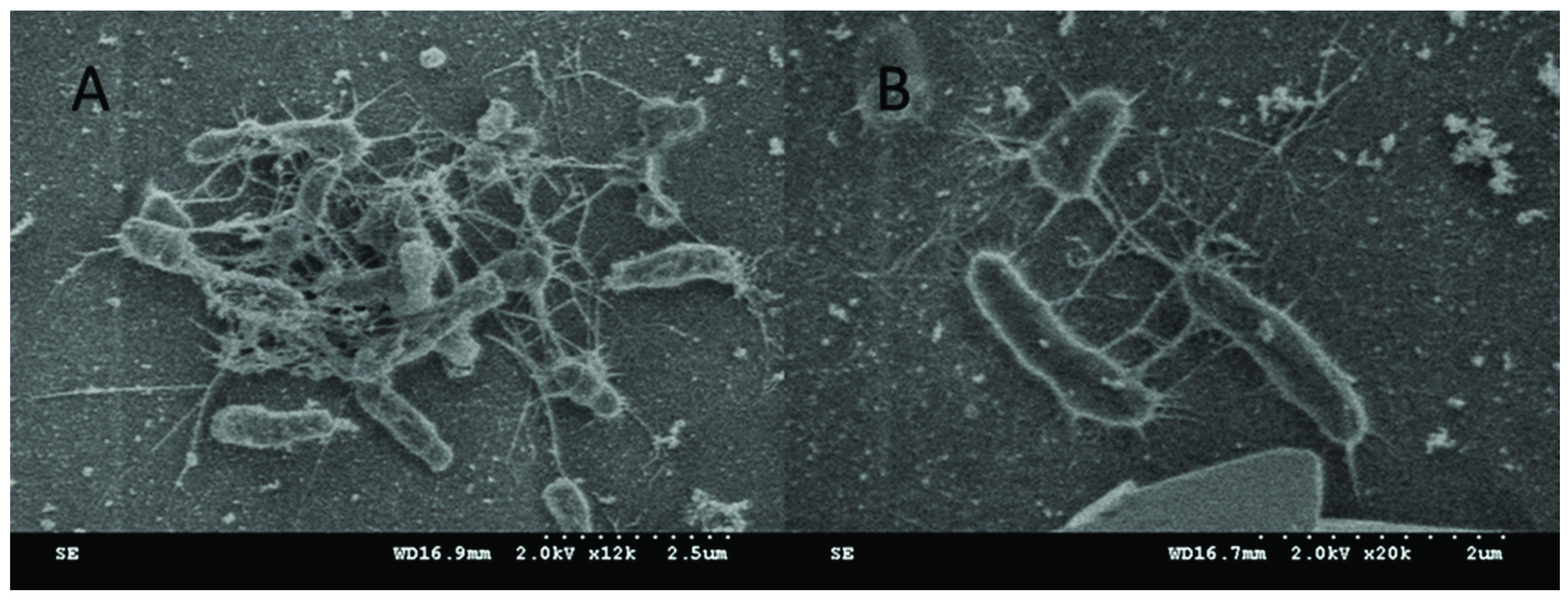
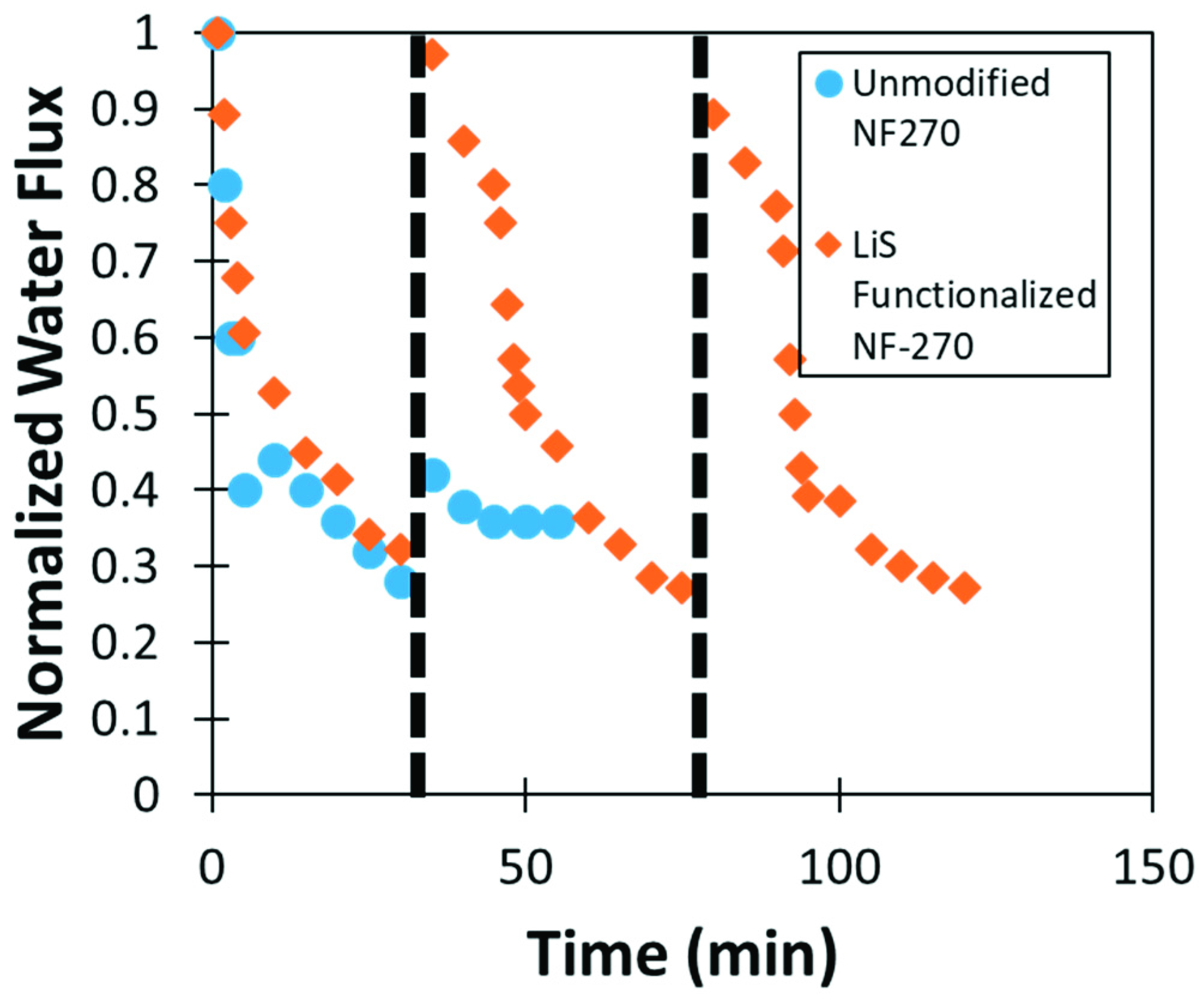
2.8. Bacteria Fouling Studies
3. Results and Discussion
3.1. Summary of Membranes
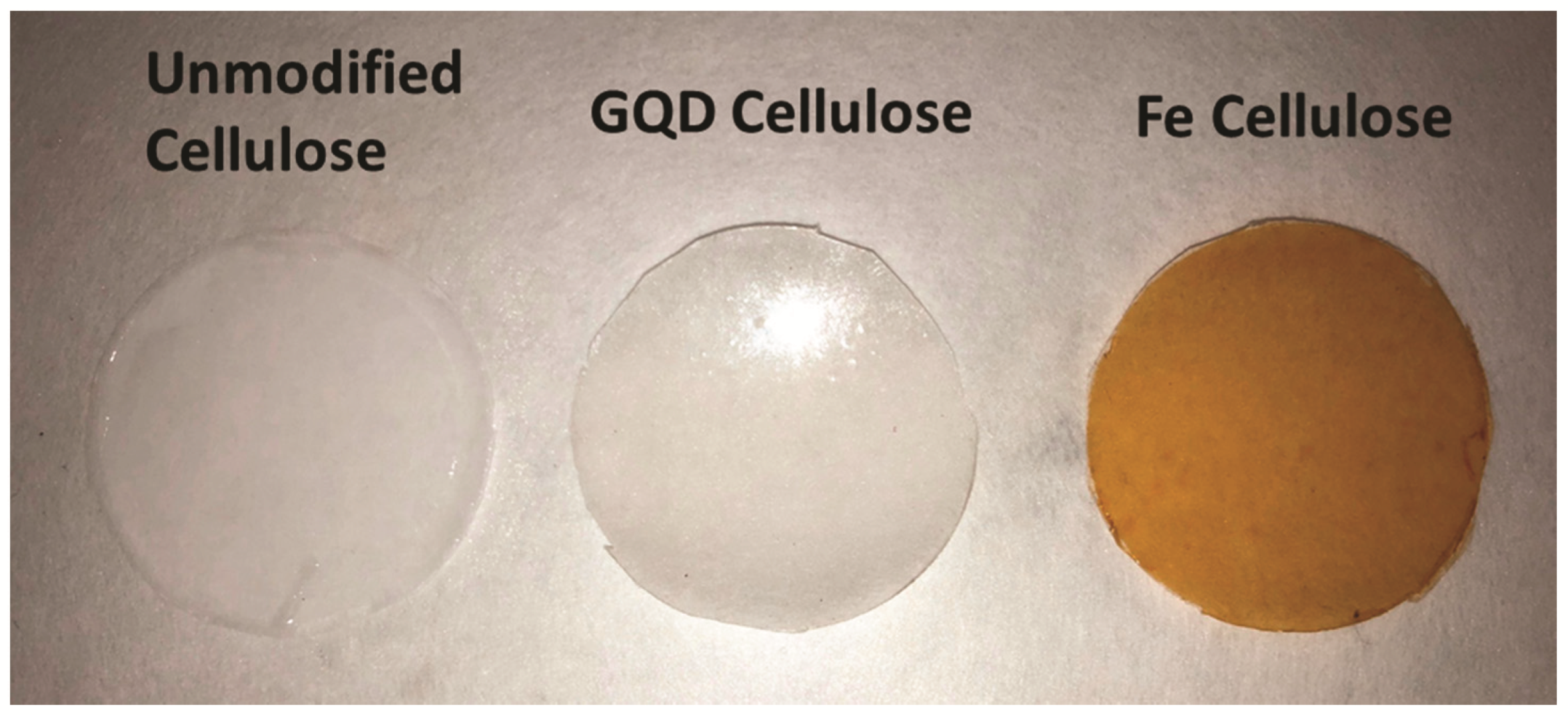
3.2. Iron Cellulose Composite Membranes
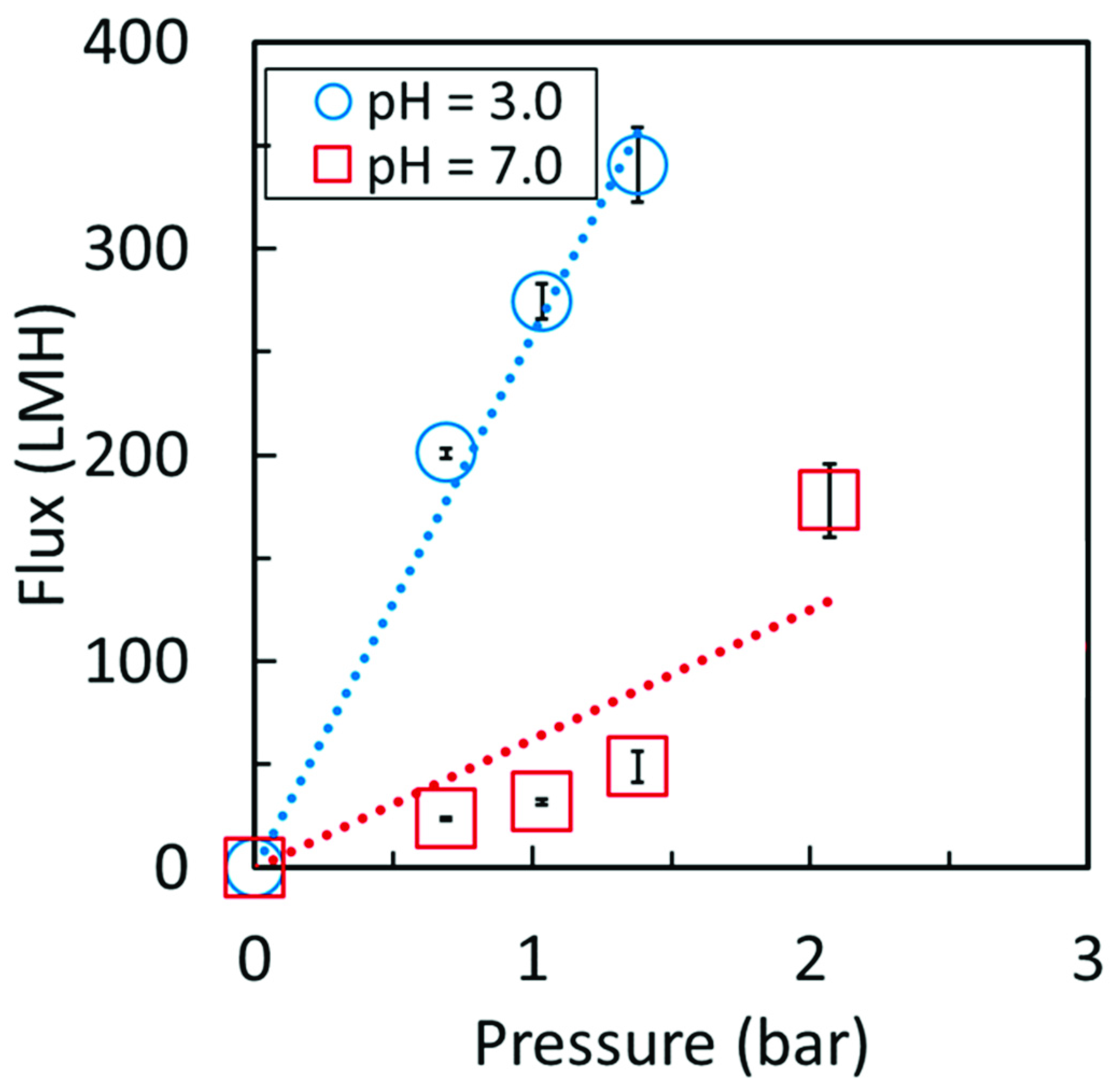
3.3. Polyacrylic Acid Cellulose Composite Membranes
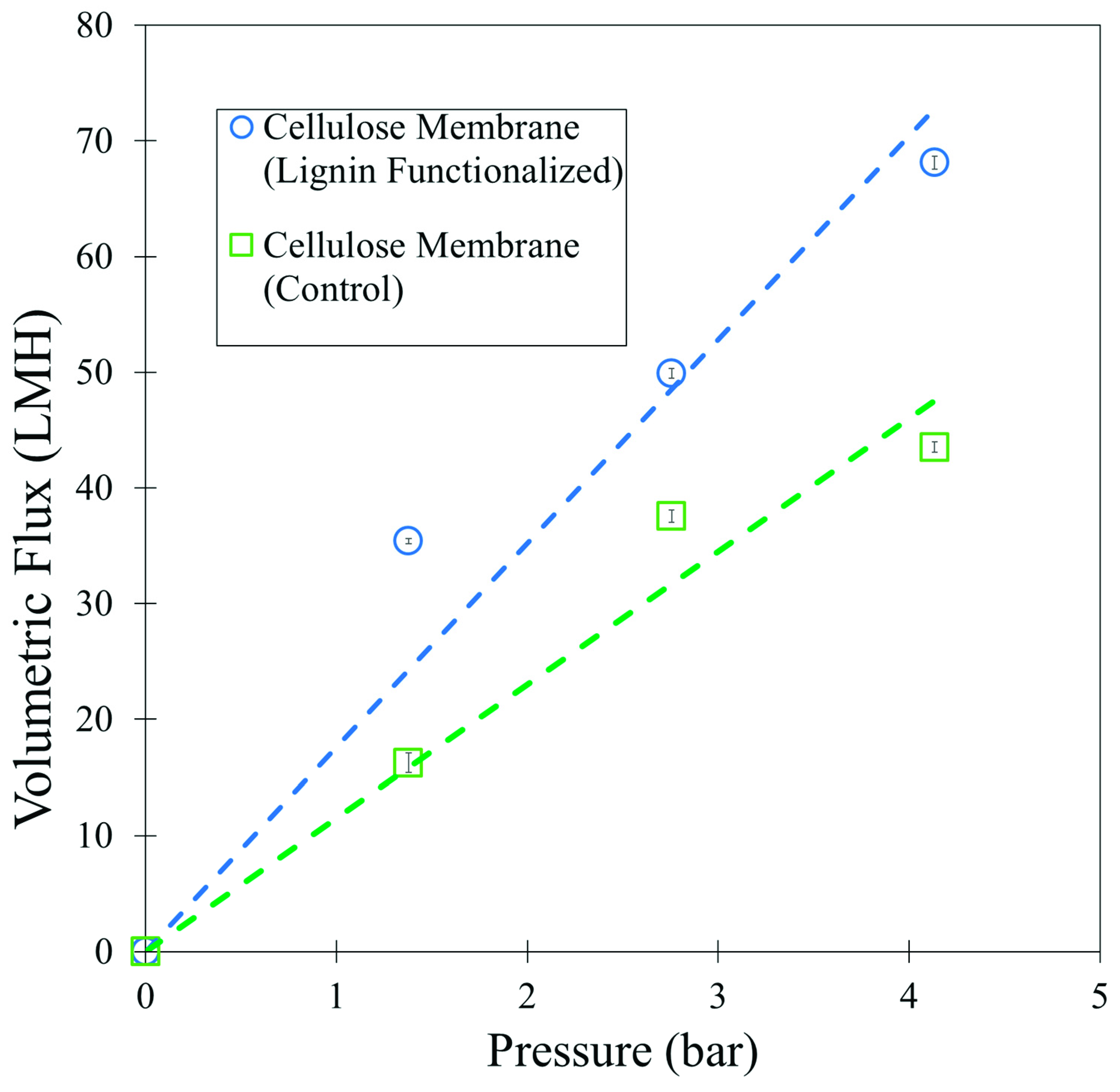
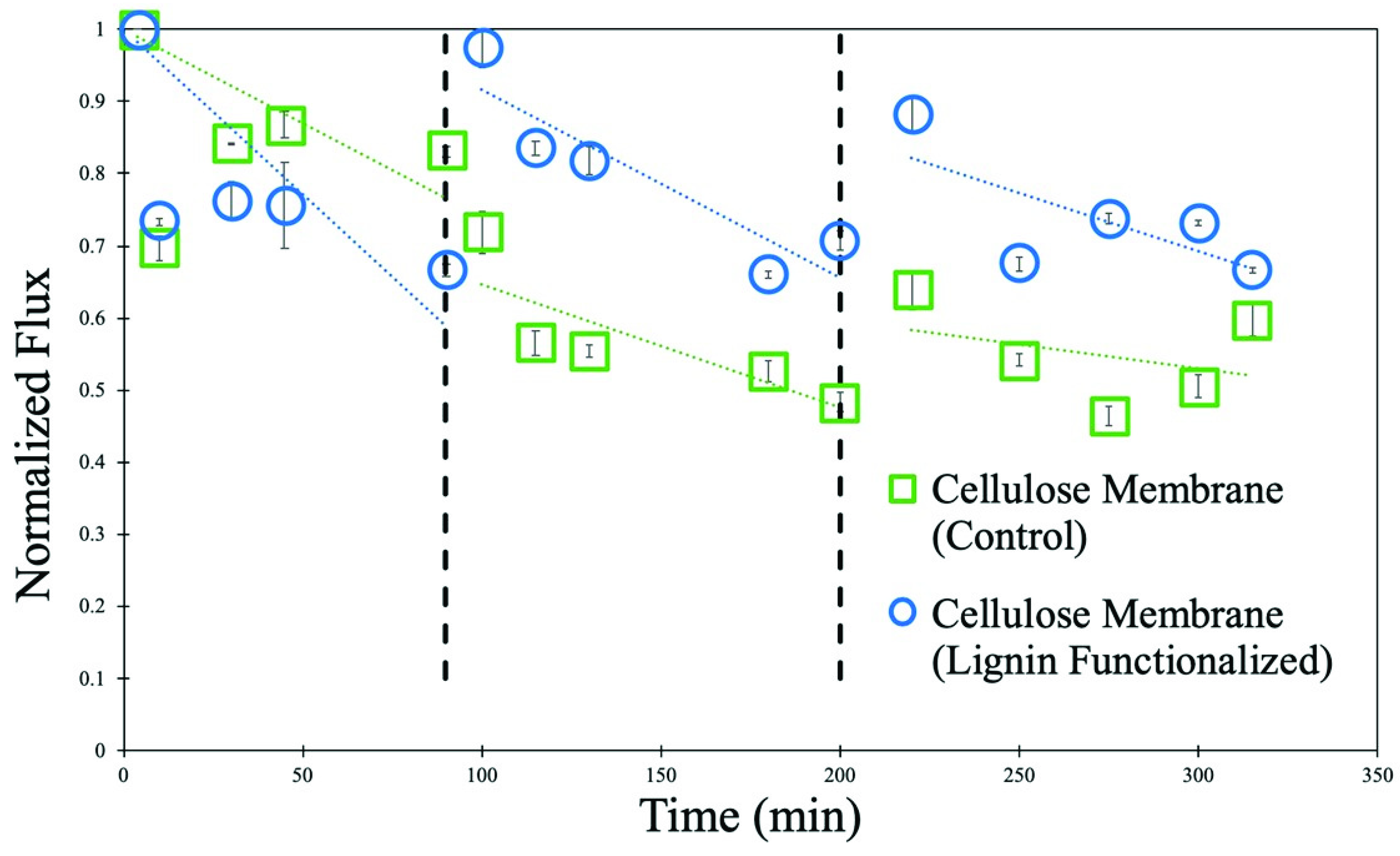
3.4. Lignin Sulfonate Cellulose Composite Membranes
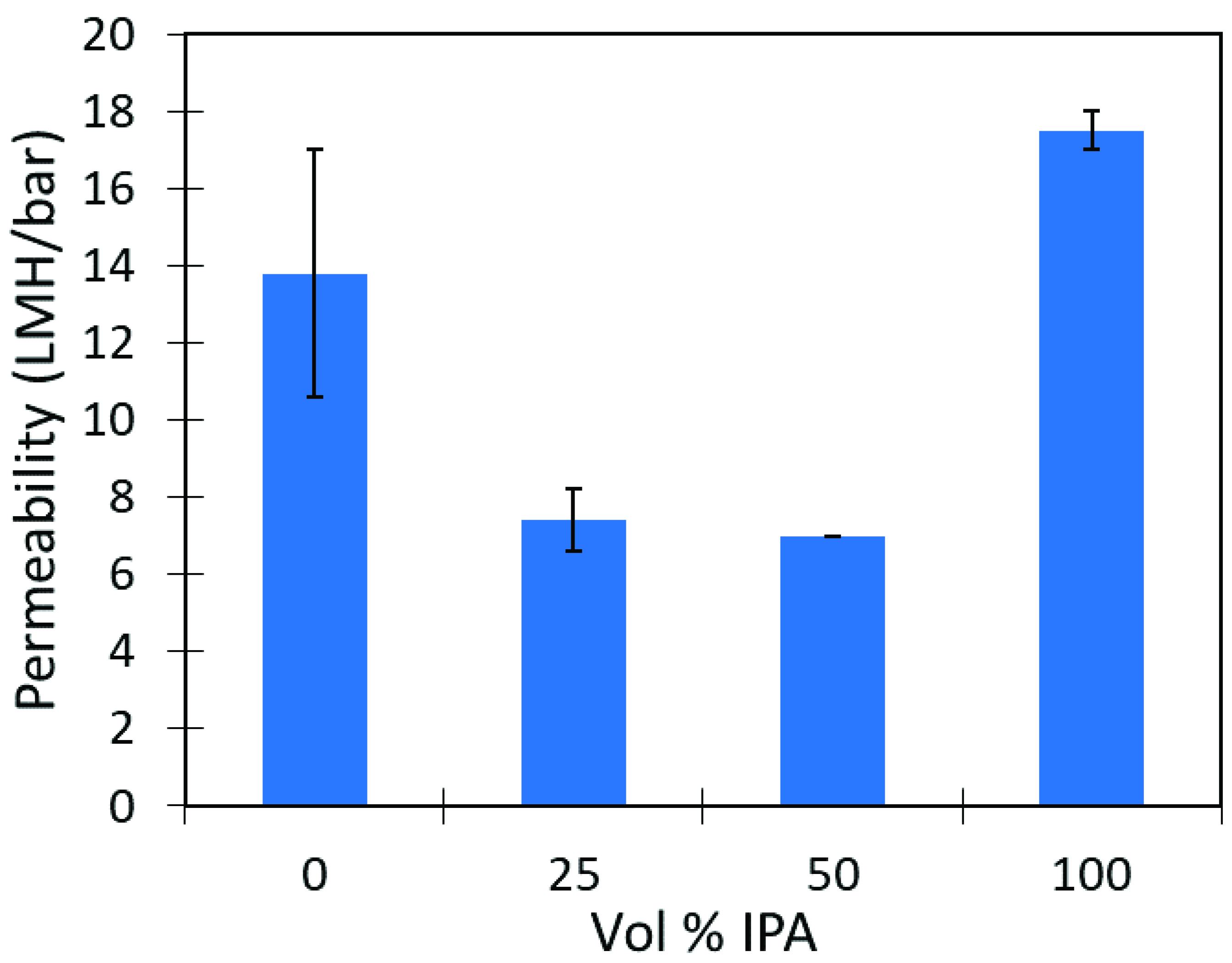
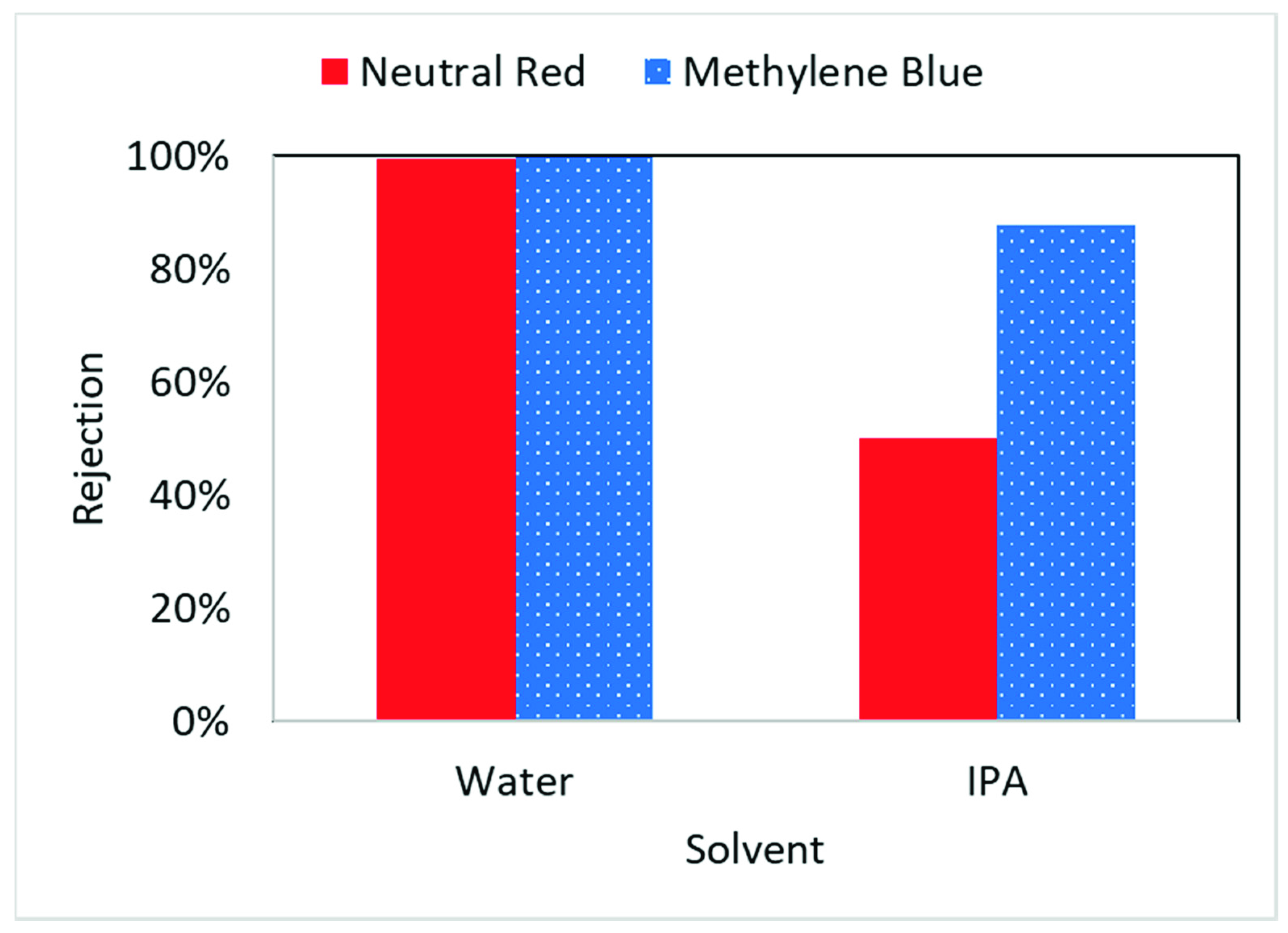
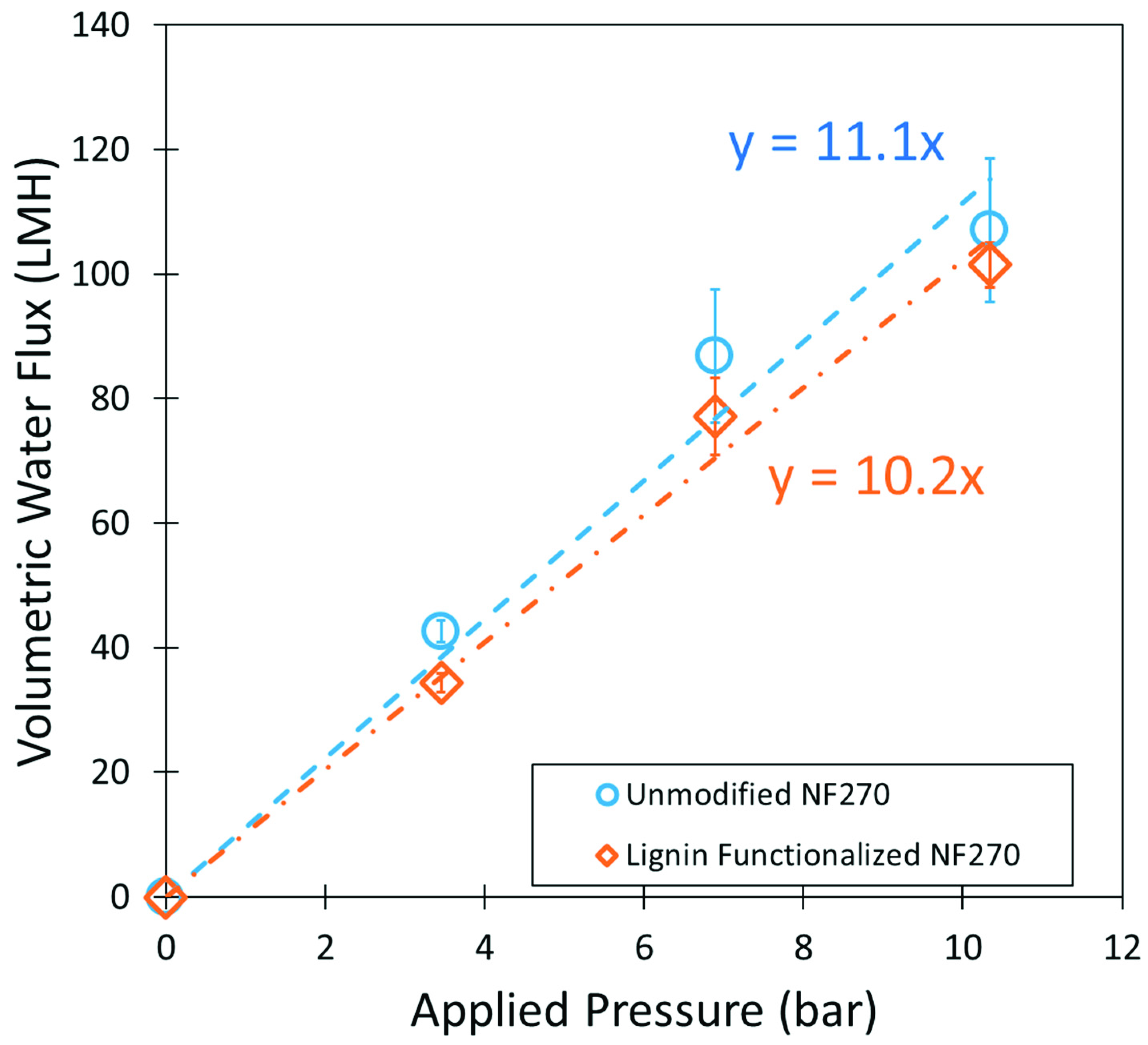
3.5. Lignin Sulfonate Functionalized Commercial Nanofiltration Membrane
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Adani, F.; Papa, G.; Schievano, A.; Cardinale, G.; D’Imporzano, G.; Tambone, F. Nanoscale structure of the cell wall protecting cellulose from enzyme attack. Environ. Sci. Technol. 2011, 45, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Moulherat, C.; Tengberg, M.; Haquet, J.-F.; Mille, B.T. First evidence of cotton at Neolithic Mehrgarh, Pakistan: Analysis of mineralized fibres from a copper bead. J. Archaeol. Sci. 2002, 29, 1393–1401. [Google Scholar] [CrossRef]
- Colwell, R.R.; Huq, A.; Islam, M.S.; Aziz, K.M.A.; Yunus, M.; Khan, N.H.; Mahmud, A.; Sack, R.B.; Nair, G.B.; Chakraborty, J.; et al. Reduction of cholera in Bangladeshi villages by simple filtration. Proc. Natl. Acad. Sci. USA 2003, 100, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Huq, A.; Yunus, M.; Sohel, S.S.; Bhuiya, A.; Emch, M.; Luby, S.P.; Russek-Cohen, E.; Nair, G.B.; Sack, R.B.; Colwell, R.R. Simple sari cloth filtration of water is sustainable and continues to protect villagers from cholera in Matlab, Bangladesh. MBio 2010, 1, e00034-10. [Google Scholar] [CrossRef] [PubMed]
- Renkin, E.M. Filtration, diffusion, and molecular sieving through porous cellulose membranes. J. Gen. Physiol. 1954, 38, 225–243. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, H.K.; Merten, U.; Riley, R.L. Transport properties of cellulose acetate osmotic membranes. J. Appl. Polym. Sci. 1965, 9, 1341–1362. [Google Scholar] [CrossRef]
- Yang, G.; Xiong, X.; Zhang, L. Microporous formation of blend membranes from cellulose/konjac glucomannan in NaOH/thiourea aqueous solution. J. Membr. Sci. 2002, 201, 161–173. [Google Scholar] [CrossRef]
- Anokhina, T.S.; Yushkin, A.A.; Makarov, I.S.; Ignatenko, V.Y.; Kostyuk, A.V.; Antonov, S.V.; Volkov, A.V. Cellulose composite membranes for nanofiltration of aprotic solvents. Pet. Chem. 2016, 56, 1085–1092. [Google Scholar] [CrossRef]
- Mao, Z.; Cao, Y.; Jie, X.; Kang, G.; Zhou, M.; Yuan, Q. Dehydration of isopropanol–water mixtures using a novel cellulose membrane prepared from cellulose/N-methylmorpholine-N-oxide/H2O solution. Sep. Purif. Technol. 2010, 72, 28–33. [Google Scholar] [CrossRef]
- Chen, H.Z.; Wang, N.; Liu, L.Y. Regenerated cellulose membrane prepared with ionic liquid 1-butyl-3-methylimidazolium chloride as solvent using wheat straw. J. Chem. Technol. Biotechnol. 2012, 87, 1634–1640. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, Y.X.; Yu, H.P.; Sun, Q.F. Preparation, characterization and biocompatibility of regenerated cellulose/PVA blend membranes in 1-allyl-3-methyimidazolium chloride. In Materials Science and Engineering, Pts 1–2; Zhu, G., Ed.; Trans Tech Publications: Zurich, Switzerland, 2011; Volume 179, pp. 179–180. [Google Scholar]
- Lei, L.; Lindbråthen, A.; Sandru, M.; Gutierrez, M.; Zhang, X.; Hillestad, M.; He, X. Spinning cellulose hollow fibers using 1-ethyl-3-methylimidazolium acetate–dimethylsulfoxide co-solvent. Polymers 2018, 10, 972. [Google Scholar] [CrossRef] [PubMed]
- Sukma, F.M.; Çulfaz-Emecen, P.Z. Cellulose membranes for organic solvent nanofiltration. J. Membr. Sci. 2018, 545, 329–336. [Google Scholar] [CrossRef]
- Kim, D.; Livazovic, S.; Falca, G.; Nunes, S.P. Oil–water separation using membranes manufactured from cellulose/ionic liquid solutions. ACS Sustain. Chem. Eng. 2019, 7, 5649–5659. [Google Scholar] [CrossRef]
- Iftekhar, S.; Srivastava, V.; Sillanpää, M. Synthesis and application of LDH intercalated cellulose nanocomposite for separation of rare earth elements (REEs). Chem. Eng. J. 2017, 309, 130–139. [Google Scholar] [CrossRef]
- Colburn, A.; Wanninayake, N.; Kim, D.Y.; Bhattacharyya, D. Cellulose-graphene quantum dot composite membranes using ionic liquid. J. Membr. Sci. 2018, 556, 293–302. [Google Scholar] [CrossRef]
- Islam, M.S.; Hernández, S.; Wan, H.; Ormsbee, L.; Bhattacharyya, D. Role of membrane pore polymerization conditions for pH responsive behavior, catalytic metal nanoparticle synthesis, and PCB degradation. J. Membr. Sci. 2018, 555, 348–361. [Google Scholar] [CrossRef] [PubMed]
- Chitpong, N.; Husson, S.M. Nanofiber ion-exchange membranes for the rapid uptake and recovery of heavy metals from water. Membranes 2016, 6, 59. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Li, Z.; Kong, Y.; Song, Q.; Wang, K. Heavy metal ions retention by bi-functionalized lignin: Synthesis, applications, and adsorption mechanisms. J. Ind. Eng. Chem. 2014, 20, 4429–4436. [Google Scholar] [CrossRef]
- Ye, J.; Cheng, Y.; Sun, L.; Ding, M.; Wu, C.; Yuan, D.; Zhao, X.; Xiang, C.; Jia, C. A green SPEEK/lignin composite membrane with high ion selectivity for vanadium redox flow battery. J. Membr. Sci. 2019, 572, 110–118. [Google Scholar] [CrossRef]
- Zhao, J.; Baba, T.; Mori, H.; Shimizu, K. Effect of zwf gene knockout on the metabolism of Escherichia coli grown on glucose or acetate. Metab. Eng. 2004, 6, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Chao, Y.; Zhang, T. Optimization of fixation methods for observation of bacterial cell morphology and surface ultrastructures by atomic force microscopy. Appl. Microbiol. Biotechnol. 2011, 92, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Davenport, D.M.; Gui, M.; Ormsbee, L.R.; Bhattacharyya, D. Development of PVDF membrane nanocomposites via various functionalization approaches for environmental applications. Polymers 2016, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Vanholme, R.; Demedts, B.; Morreel, K.; Ralph, J.; Boerjan, W. Lignin biosynthesis and structure. Plant Physiol. 2010, 153, 895. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Spigno, G.; Labidi, J. Antioxidant and biocide behaviour of lignin fractions from apple tree pruning residues. Ind. Crops Prod. 2017, 104, 242–252. [Google Scholar] [CrossRef]
- Houtman, C.J.; Atalla, R.H. Cellulose-lignin interactions (a computational study). Plant Physiol. 1995, 107, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and sulfonated lignin. Chem. Sustain. Chem. 2017, 10, 1861–1877. [Google Scholar] [CrossRef] [PubMed]
- Zemek, J.; Košíková, B.; Augustín, J.; Joniak, D. Antibiotic properties of lignin components. Folia Microbiol. 1979, 24, 483–486. [Google Scholar] [CrossRef]
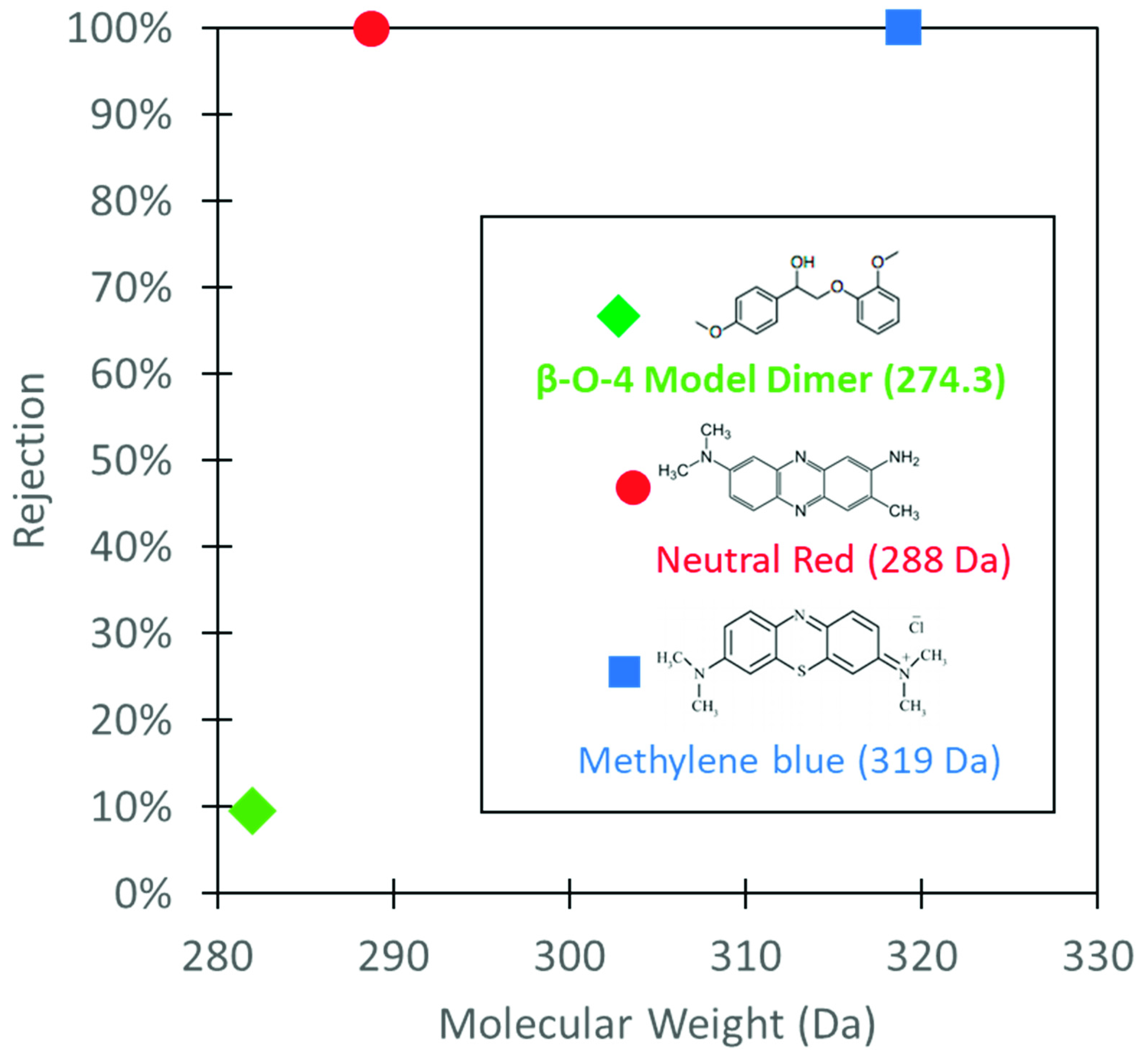

| Model Solute | Molecular Wt. (Da) | Structure |
|---|---|---|
| β-O-4 Model Dimer | 282 | 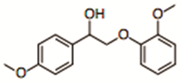 |
| Neutral Red | 289 | 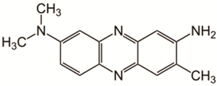 |
| Methylene Blue | 320 | 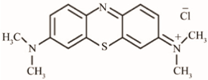 |
| Composite Material | Wt% Composite | Wt% Cellulose | Casting Solution Viscosity (Pa*s) | Water Permeability (LMH/bar) (pH=7) | Rejection (%) 5kDa Blue Dextran |
|---|---|---|---|---|---|
| Iron | 4 | 5 | 6.8 | 17.4 | >99 |
| PAA | 2 | 5 | 44 | 267 | 44 |
| Lignin | 5 | 10 | 96 | 17.5 | 59 |
| Cellulose only | 0 | 10 | 22.8 | 9.6 | 75 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colburn, A.; Vogler, R.J.; Patel, A.; Bezold, M.; Craven, J.; Liu, C.; Bhattacharyya, D. Composite Membranes Derived from Cellulose and Lignin Sulfonate for Selective Separations and Antifouling Aspects. Nanomaterials 2019, 9, 867. https://doi.org/10.3390/nano9060867
Colburn A, Vogler RJ, Patel A, Bezold M, Craven J, Liu C, Bhattacharyya D. Composite Membranes Derived from Cellulose and Lignin Sulfonate for Selective Separations and Antifouling Aspects. Nanomaterials. 2019; 9(6):867. https://doi.org/10.3390/nano9060867
Chicago/Turabian StyleColburn, Andrew, Ronald J. Vogler, Aum Patel, Mariah Bezold, John Craven, Chunqing Liu, and Dibakar Bhattacharyya. 2019. "Composite Membranes Derived from Cellulose and Lignin Sulfonate for Selective Separations and Antifouling Aspects" Nanomaterials 9, no. 6: 867. https://doi.org/10.3390/nano9060867
APA StyleColburn, A., Vogler, R. J., Patel, A., Bezold, M., Craven, J., Liu, C., & Bhattacharyya, D. (2019). Composite Membranes Derived from Cellulose and Lignin Sulfonate for Selective Separations and Antifouling Aspects. Nanomaterials, 9(6), 867. https://doi.org/10.3390/nano9060867