Miniemulsion RAFT Copolymerization of MMA with Acrylic Acid and Methacrylic Acid and Bioconjugation with BSA
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Reactions
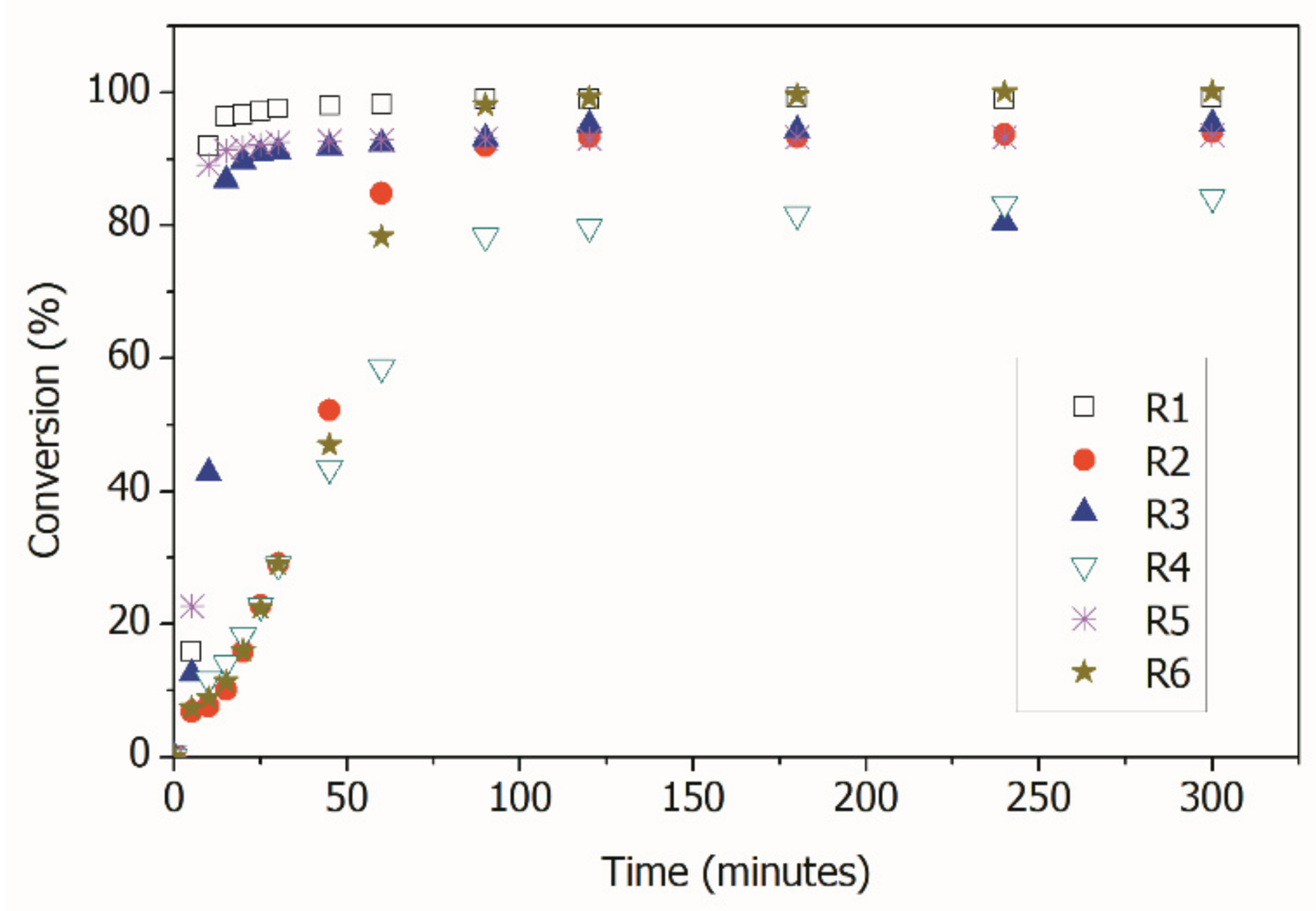
2.2.1. RAFT Polymerization
2.2.2. Bioconjugation
2.2.3. Labeling with Rodamine B
2.3. Characterization
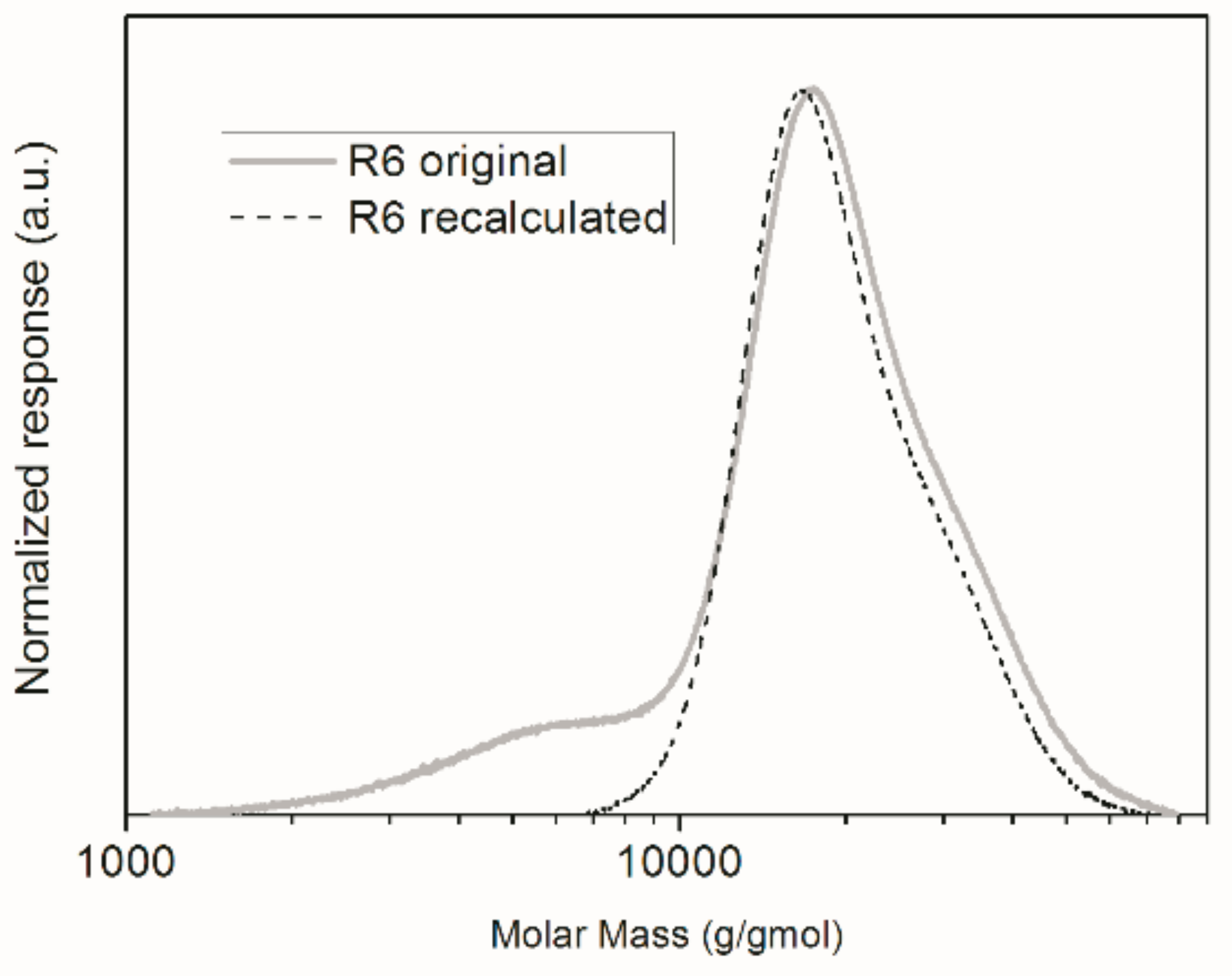
2.3.1. Gel Permeation Chromatography (GPC)
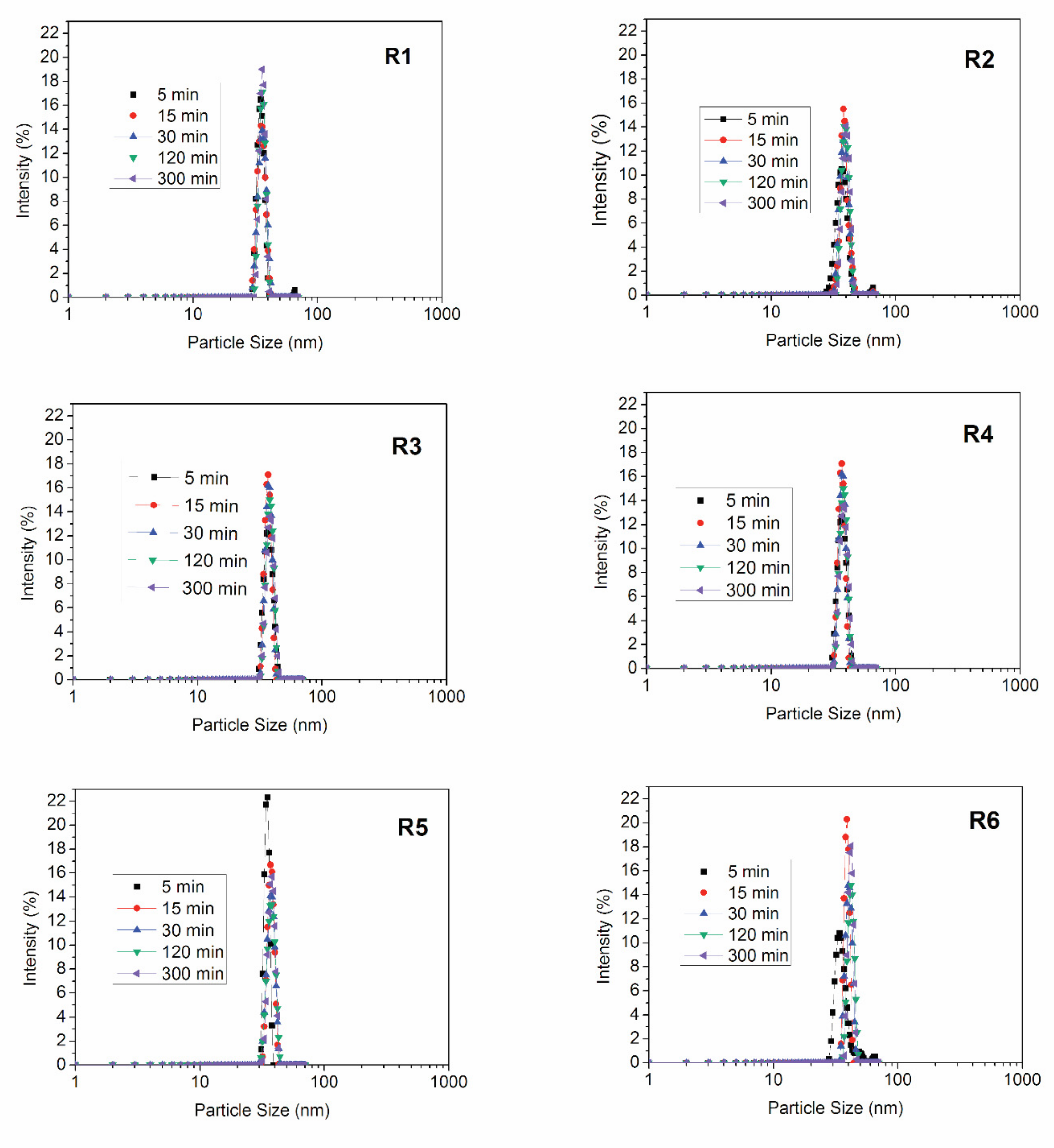
2.3.2. Dynamic Light Scattering (DLS)
2.3.3. Nuclear Magnetic Resonance (NMR)
2.3.4. Contact Angle
2.3.5. Bioconjugation Efficiency
2.3.6. Cellular Uptake
3. Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barner-Kowollik, C. Handbook of RAFT Polimerization; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Moad, G.; Rizzardo, E.; Thang, S.H. Living Radical Polymerization by the RAFT Process—A First Update. Aust. J. Chem. 2006, 59, 669–692. [Google Scholar] [CrossRef]
- Moad, G. RAFT polymerization to form stimuli-responsive polymers. Polym. Chem. 2017, 8, 177–219. [Google Scholar] [CrossRef]
- Alexis, F.; Pridgen, E.; Molnar, L.K.; Farokhzad, O.C. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol. Pharm. 2008, 5, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.F.; Ferreira, P.C.; Alves, P.; Cordeiro, R.; Fonseca, A.C.; Góis, J.; Gil, M.H. Drug delivery systems: Advanced technologies potentially applicable in personalized treatments. EPMA J. 2010, 1, 164–209. [Google Scholar] [CrossRef] [PubMed]
- Owens, D.E., III; Peppas, N.A. Opsonization, biodistribution, and pharmacokinectics of polymeric nanoparticles. Int. J. Pharm. 2006, 307, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Mitsukami, Y.; Donovan, M.S.; Lowe, A.B.; McCormick, C.M. Water-Soluble Polymers. 81. Direct Synthesis of Hydrophilic Styrenic-Based Homopolymers and Block Copolymers in Aqueous Solution via RAFT. Macromolecules 2001, 34, 2248–2256. [Google Scholar] [CrossRef]
- Oliveira, M.A.M.; Nele, M.; Pinto, J.C. Polimerização RAFT em Miniemulsão. Polímeros 2013, 6, 784–797. [Google Scholar] [CrossRef][Green Version]
- Benaglia, M.; Rizzardo, E.; Alberti, A.; Guerra, M. Searching for More Effective Agents and Conditions for RAFT Polymerization of MMA: Influence of Dithioester Substituents, Solvents, and Temperature. Macromolecules 2005, 138, 3129–3140. [Google Scholar] [CrossRef]
- Chiefari, J.; Chong, Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.; Moad, G.; et al. Living free-radical polymerization by reversible addition-fragmentation chain transfer: The RAFT process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Sontakke, T.K.; Jagtap, R.N. Synthesis of AA-MMA Block Copolymer by RAFT Polymerization and Used as Emulsifier cum Macroinitiator and Its Influence on the Film Properties. J. Disper. Sci. Technol. 2013, 34, 1575–1584. [Google Scholar] [CrossRef]
- Hill, M.R.; Carmean, R.N.; Sumerlin, B.S. Expanding the Scope of RAFT Polymerization: Recent Advances and New Horizons. Macromolecules 2015, 48, 5459–5469. [Google Scholar]
- Rizzardo, E.; Chiefari, J.; Mayadunne, R.; Moad, G.; Thang, S. Tailored polymer architectures by reversible addition-fragmentation chain transfer. Macromol. Symp. 2001, 174, 209–212. [Google Scholar] [CrossRef]
- Vollrath, A.; Schallon, A.; Pietsch, C.; Schubert, S.; Nomoto, T.; Matsumoto, Y.; Kataoka, K.; Schubert, U.S. A toolbox of differently sized and labeled PMMA nanoparticles for cellular uptake investigations. Soft Matter 2013, 9, 99–108. [Google Scholar] [CrossRef]
- Elsabahy, M.; Wooley, K.L. Design of polymeric nanoparticles for biomedical delivery applications. Chem. Soc. Rev. 2012, 41, 2545–2561. [Google Scholar] [CrossRef]
- Odian, G. Principles of Polymerization, 4th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- Li, J.; Jiang, T.; Shen, J.; Ruan, H. Preparation and Characterization of PMMA and its Derivative via RAFT Technique in the Presence of Disulfide as a Source of Chain Transfer Agent. J. Membr. Sep. Technol. 2012, 1, 117–128. [Google Scholar]
- Willcock, H.; O’Reilly, R.K. End group removal and modification of RAFT polymers. Polym. Chem. 2010, 1, 149–157. [Google Scholar] [CrossRef]
- Sunasee, R.; Narain, R. Covalent and Noncovalent Bioconjugation Strategies. In Chemistry of Bioconjugates: Synthesis, Characterization, and Biomedical Applications; Narain, R., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 3–78. [Google Scholar]
- Ciécwierz, K.W.; Wisniewiski, M.; Terzyk, A.P.; Furmaniak, S. The Chemistry of Bioconjugation in Nanoparticles-Based Drug Delivery System. Adv. Condens. Matter Phys. 2015, 2015, 198175. [Google Scholar]
- Gelamo, E.L.; Silva, C.H.T.P.; Iasato, H.; Tabak, M. Interaction of bovine (BSA) and human (HSA) serum albumins with ionic surfactants: Spectroscopy and modelling. Biochim. Biophys. Acta 2002, 1594, 84–99. [Google Scholar] [CrossRef]
- Li, S.; Hu, J.; Liu, B. A study on the adsorption behavior of protein onto functional microspheres. J. Chem. Technol. Biotechnol. 2005, 80, 531–536. [Google Scholar] [CrossRef]
- Zimmerman, Z.L.; Nicolaus, T.; Neuert, G.; Blank, K. Thiol-based, site-specific and covalent immobilization of biomolecules for single-molecule experiments. Nat. Protoc. 2010, 5, 975–985. [Google Scholar]
- Schlüter, D.; Hawker, C.; Sakamoto, J. Synthesis of Polymers: New Structures and Methods; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar]
- Nair, D.P.; Podgórski, M.; Chatani, S.; Gong, T.; Xi, W.; Fenoli, C.; Bowman, C. The Thiol-Michael Addition Click Reaction: A Powerful and Widely Used Tool in Materials Chemistry. Chem. Mater 2014, 26, 724–744. [Google Scholar] [CrossRef]
- Northrop, B.H.; Frayne, S.H.; Choudhary, U. Thiol–maleimide “click” chemistry: Evaluating the influence of solvent, initiator, and thiol on the reaction mechanism, kinetics, and selectivity. Polym. Chem. 2015, 6, 3415–3430. [Google Scholar] [CrossRef]
- Hermanson, G. Bioconjugate Techniques, 3rd ed.; Thermo Fisher Scientific: Rockford, IL, USA, 2013. [Google Scholar]
- Moreira, T.S.; Oliveira, M.A.M.; Nele, M.; Pinto, J.C.; Lima, L.M.T.R. Effect of tamoxifen in RAFT miniemulsion polymerization during the synthesis of polymer nanoparticles. Polímeros 2014, 24, 25–30. [Google Scholar] [CrossRef][Green Version]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Fonseca, L.B.; Nele, M.; Volpato, N.M.; Seiceira, R.; Pinto, J.C. Production of PMMA Nanoparticles Loaded with Praziquantel through “in situ” Miniemulsion Polymerization. Macromol. React. Eng. 2012, 7, 54–63. [Google Scholar] [CrossRef]
- Peixoto, A.C.B.; Campos, I.M.F.; Ferraz, H.C.; Pinto, J.C. Use of Hydrophilic Monomers to Avoid Secondary Particle Nucleation in Miniemulsion Polymerizations of Methyl Methacrylate. J. Res. Updates Polym. Sci. 2016, 5, 60–71. [Google Scholar]
- Binks, B.P. Modern Characterization Methods of Surfactant Systems; CRC Press: New York, NY, USA, 1999. [Google Scholar]
- Mishra, M.K.; Yagci, Y. Handbook of Vinyls Polymers: Radical Polymerization, Process, and Technology, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Svenson, S.; Prud’homme, R.K. Multifunctional Nanoparticles for Drug Delivery Applications: Imaging, Targeting and Delivery, 1st ed.; Springer: Boston, MA, USA, 2012. [Google Scholar]
- Che Man, S.H.; Yusof, N.Y.; Whittaker, M.R.; Thickett, S.C.; Zetterlund, P.B. Influence of monomer type on miniemulsion polymerization systems stabilized by graphene oxide as sole surfactante. J. Polym. Sci. A Polym. Chem. 2013, 51, 5153–5162. [Google Scholar] [CrossRef]
- Mazurek, N.M.; Ma, B.; Javakhishvili, I.; Hanse, J.H.; Hvilsted, S.; Skov, A.L. Controlled release in hard to access places by poly(methyl methacrylate) microcapsules triggered by gamma irradiation. Polym. Adv. Technol. 2015, 26, 1059–1064. [Google Scholar]
- Strandman, S.; Hietala, S.; Aseyev, V.; Koli, B.; Butcher, S.J.; Tenhu, H. Supramolecular assemblies of amphiphilic PMMA-block-PAA stars in aqueous solutions. Polymer 2006, 47, 6524–6535. [Google Scholar] [CrossRef]
- Hong, S.H.; McHugh, V.M. Review of Preparation and Properties of Polymers from Copolymerization of Aprotic Acrylic Monomers with Protic Acrylic Monomers; US Army Armament Munitions Chemical Command: Maryland, MD, USA, 1988. [Google Scholar]
- Moad, G.; Rizzardo, E.; Thang, S. Radical addition–fragmentation chemistry in polymer synthesis. Polymer 2008, 49, 1079–1131. [Google Scholar] [CrossRef]
- Ramos, I.M.F.C. Nanopartículas Poliméricas para Liberação de Fármaco Sítio-Dirigida para o Tratamento de Doenças Neurodegenerativas. Master’s Thesis, PEQ/COPPE Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil, 2018. [Google Scholar]
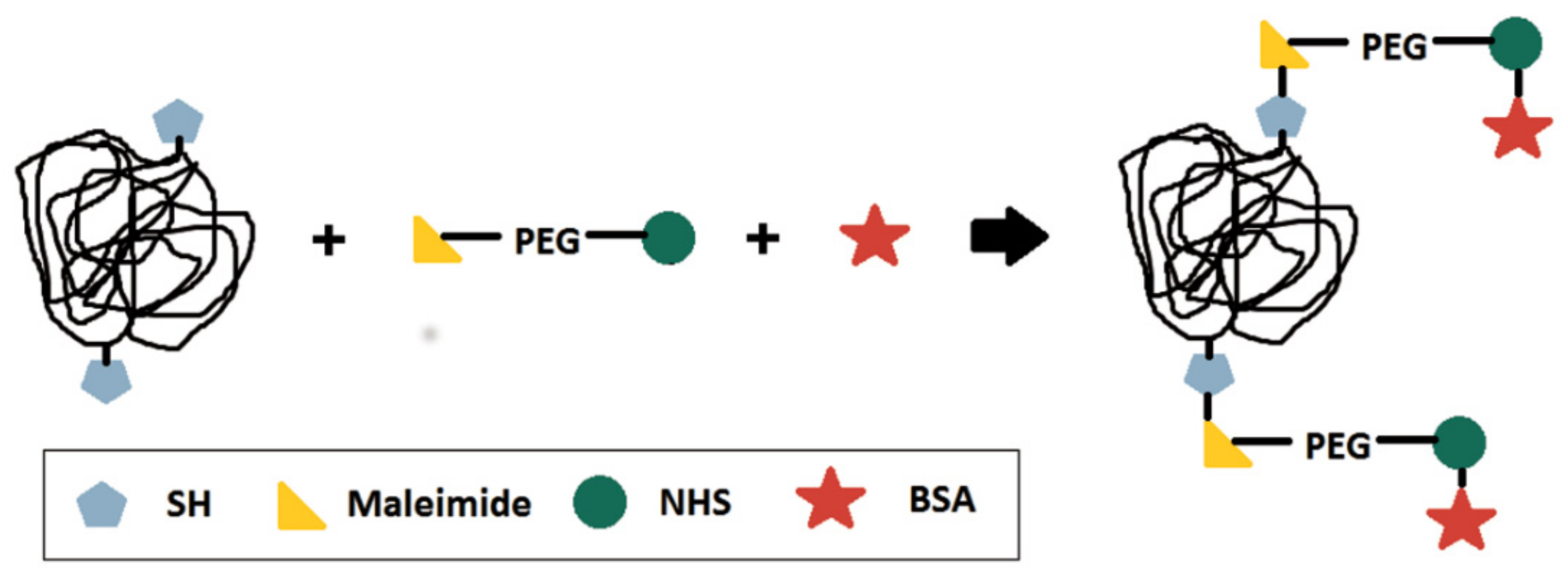

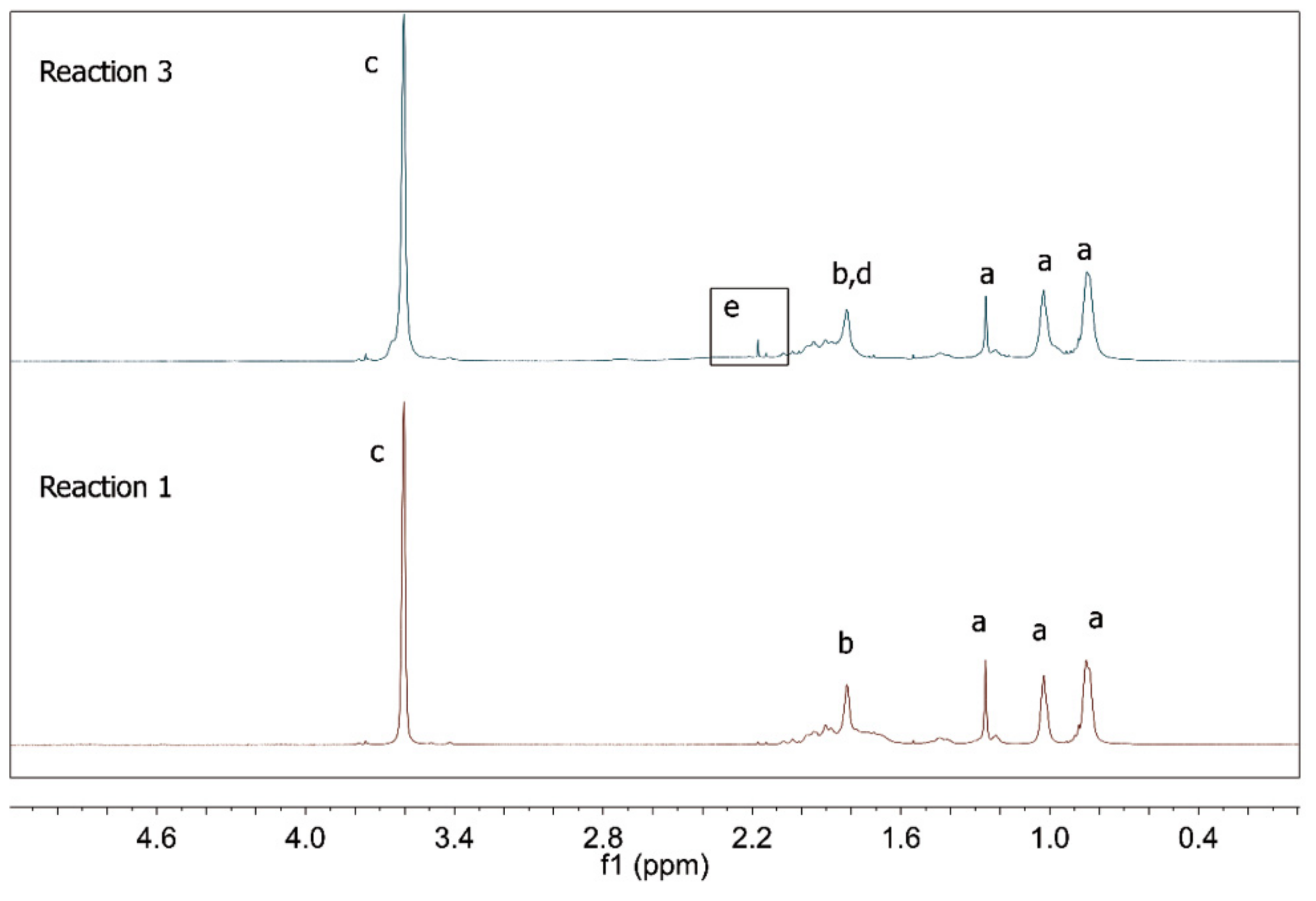
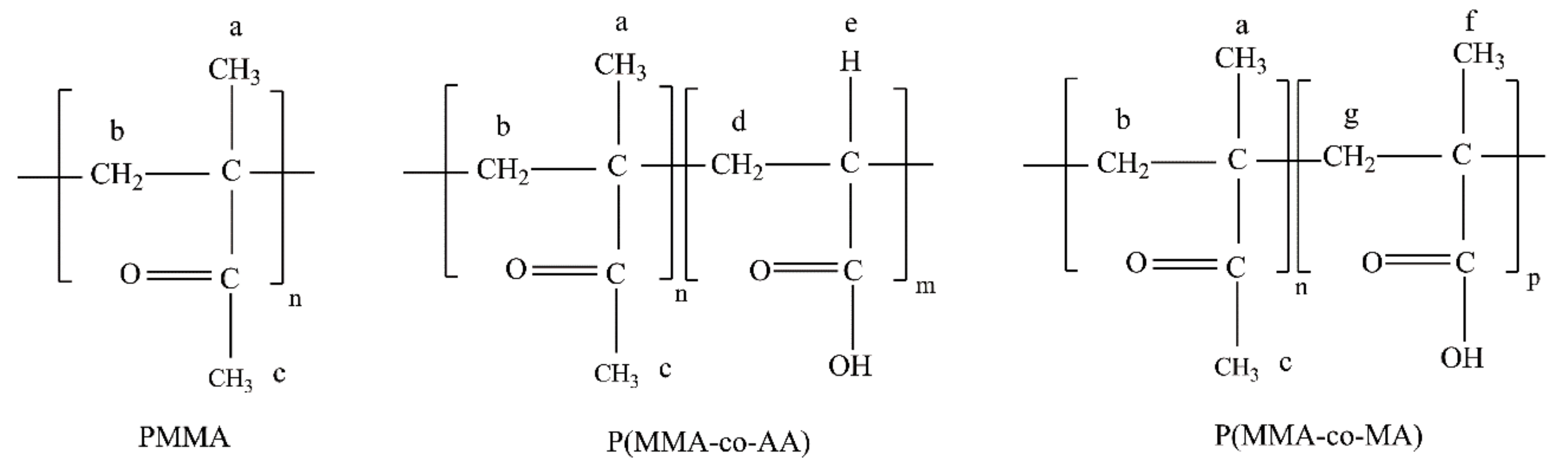
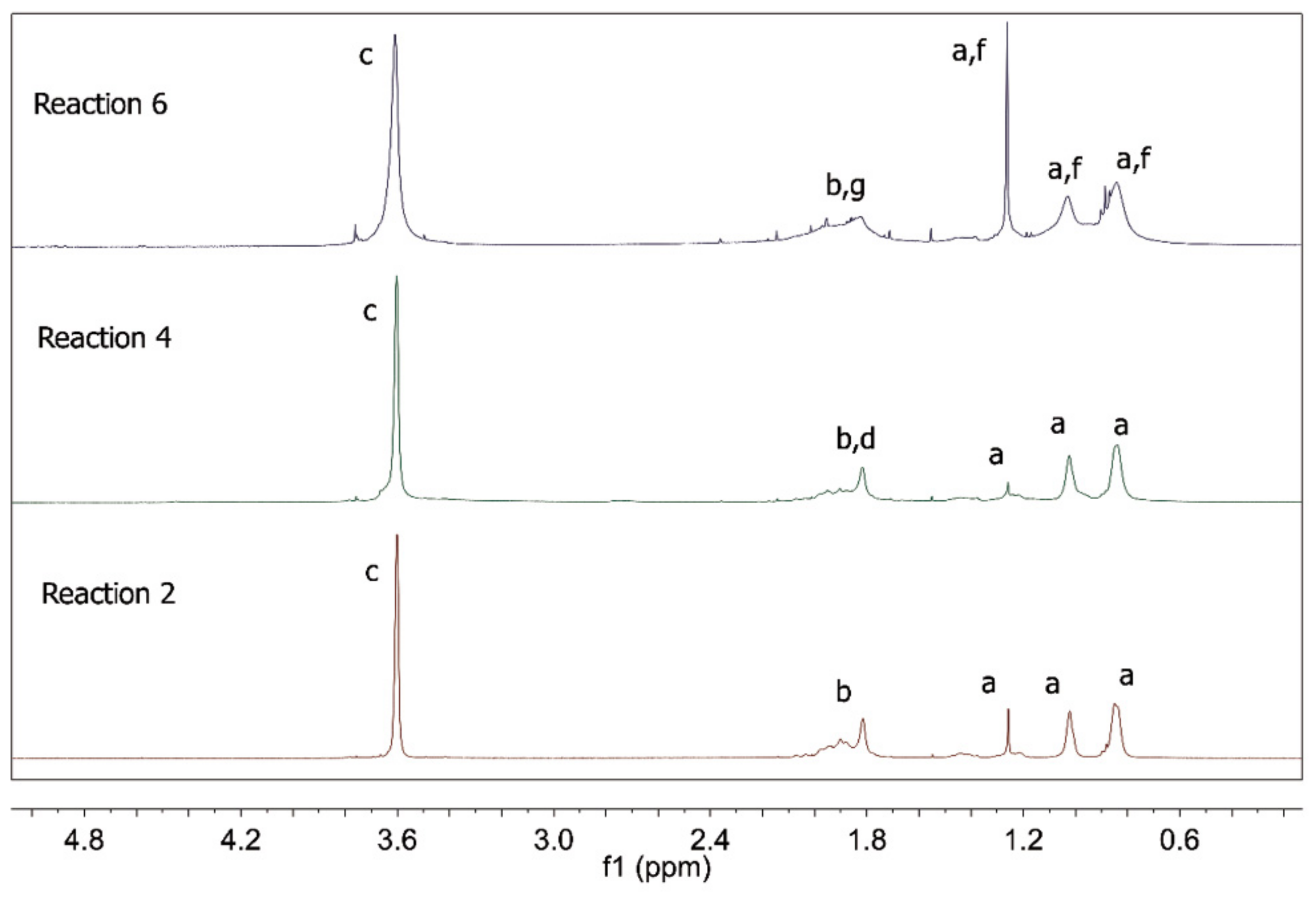
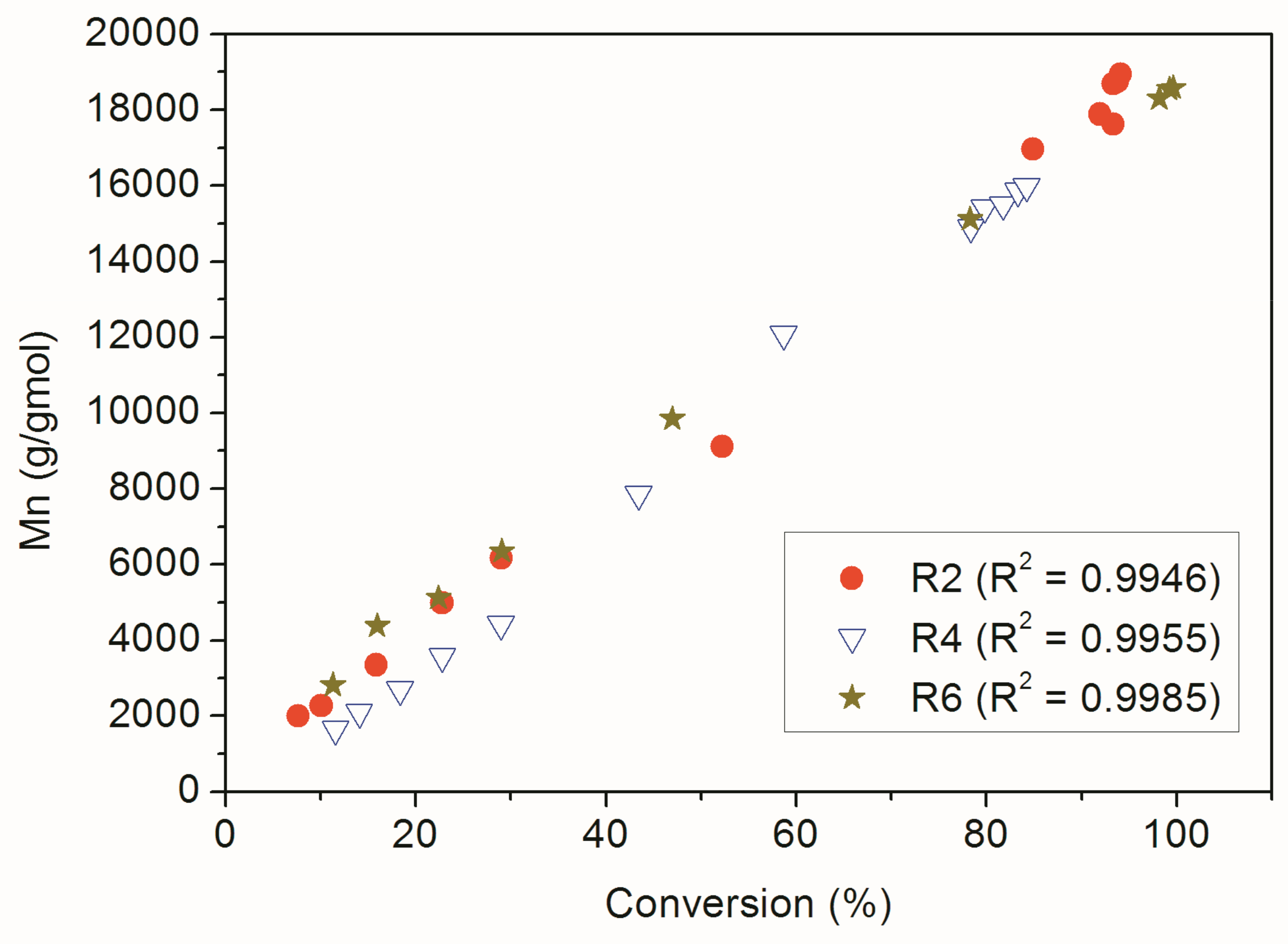
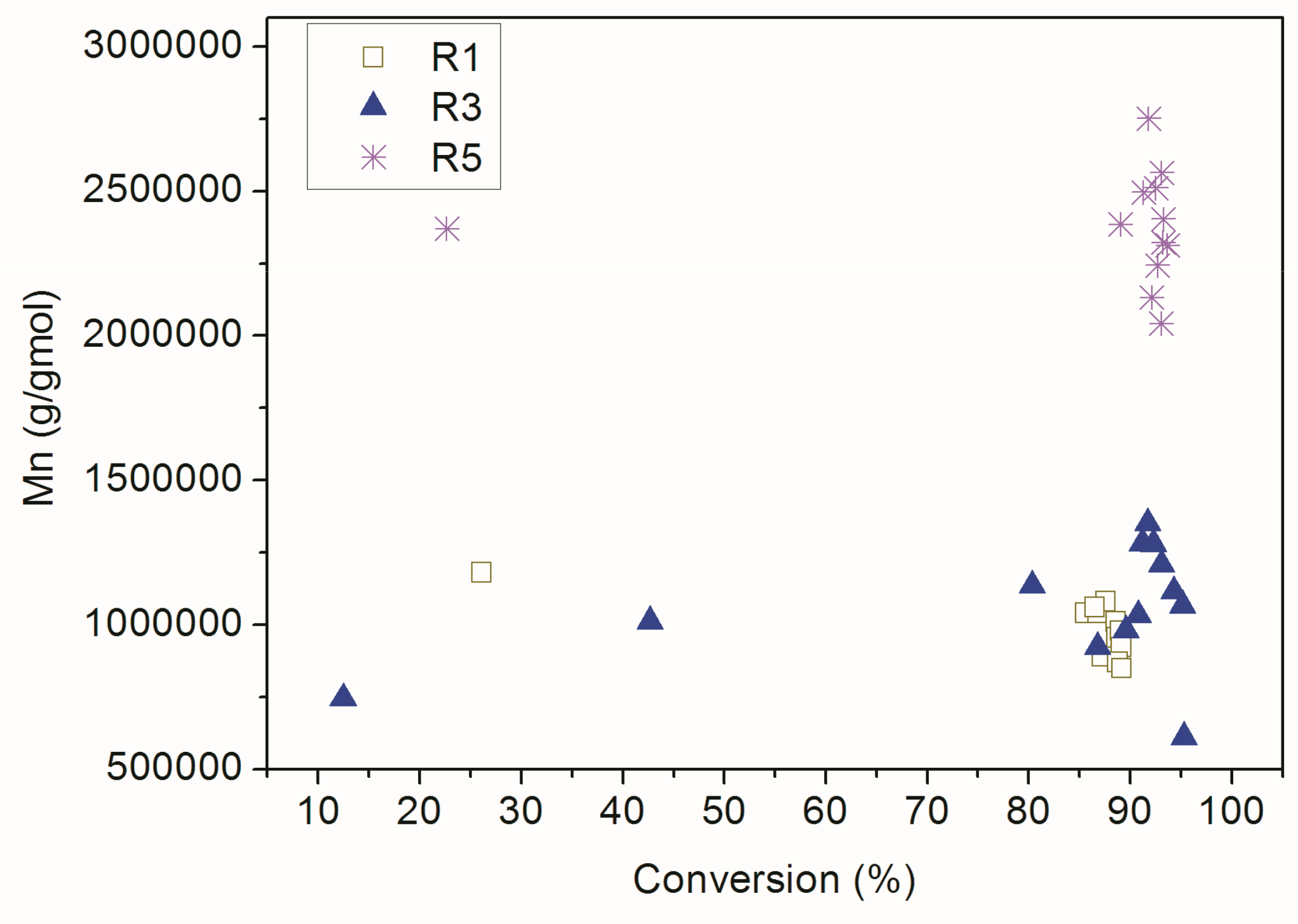
| Reaction | Comonomer * | RAFT Agent |
|---|---|---|
| R1 | - | No |
| R2 | - | Yes |
| R3 | Acrylic acid | No |
| R4 | Acrylic acid | Yes |
| R5 | Methacrylic acid | No |
| R6 | Methacrylic acid | Yes |
| Reaction | Conversion (%) | Theoretical Mn (g/gmol) | Mn (g/gmol) | PDI | Mean Particle Size (nm) |
|---|---|---|---|---|---|
| R1 | 99 | - | 1.13 × 106 | 1.93 | 72.6 ± 0.4 |
| R2 | 94 | 1.98 × 104 | 1.89 × 104 | 1.13 | 131.1± 0.7 |
| R3 | 95 | - | 6.11 × 106 | 3.33 | 103.6 ± 1.4 |
| R4 | 84 | 1.67 × 104 | 1.60 × 104 | 1.57 | 104.6 ± 1.5 |
| R5 | 94 | - | 2.31 × 106 | 1.62 | 94.1 ± 0.8 |
| R6 | 100 | 2.00 × 104 | 1.34 × 104 | 1.50 | 166.1 ± 0.3 |
| Reaction | Contact Angle |
|---|---|
| R1 | 77.2° ± 4.4 |
| R2 | 74.6° ± 1.1 |
| R3 | 39.4° ± 2.7 |
| R4 | 52.8° ± 1.6 |
| R5 | 39.3° ± 1.2 |
| R6 | 35.2° ± 2.1 |
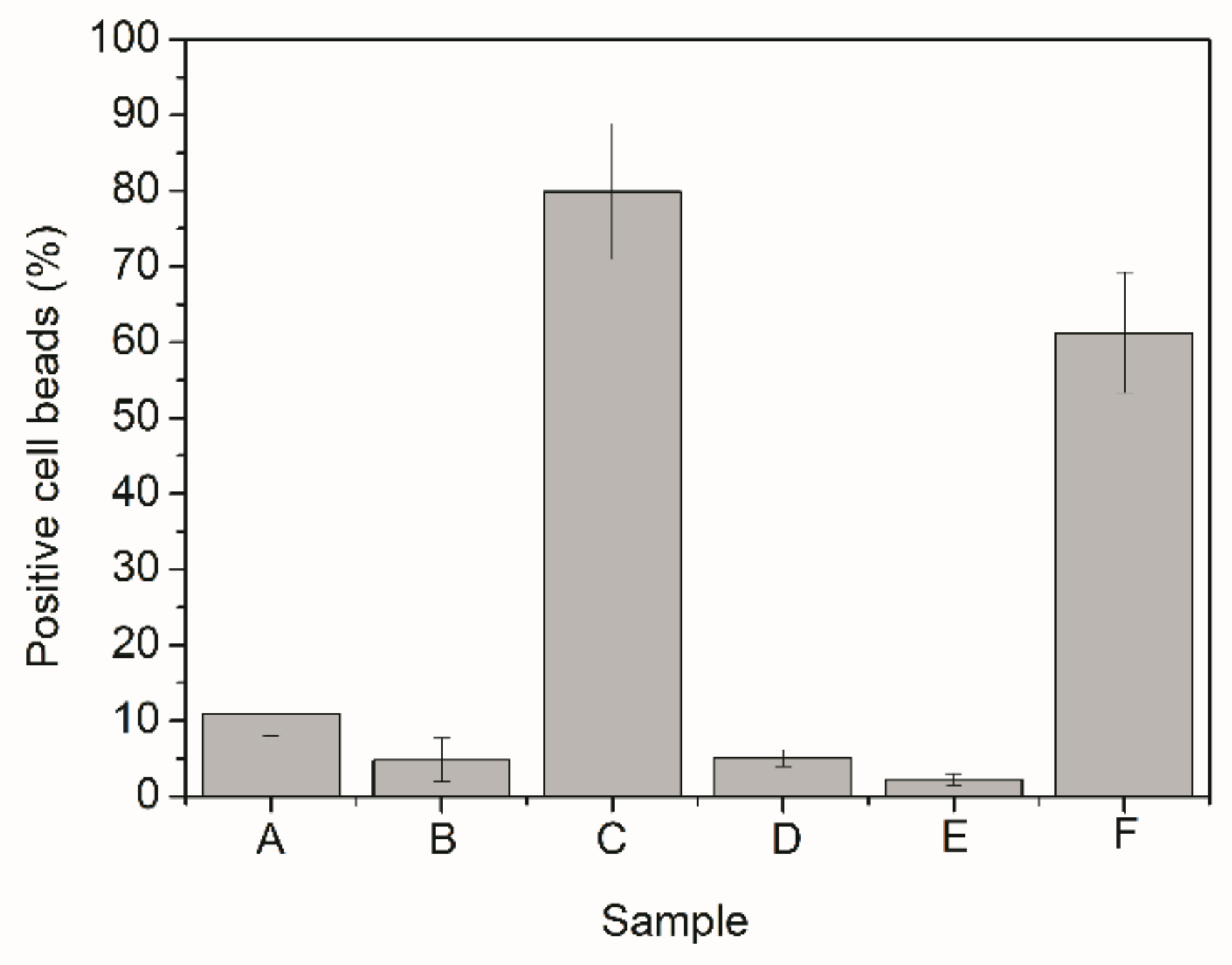
| Sample | Description |
|---|---|
| A | R2 + maleimide-PEG-NHS + BSA |
| B | R4 + maleimide-PEG-NHS + BSA |
| C | R6 + maleimide-PEG-NHS + BSA |
| D | R2 + BSA |
| E | R4 + BSA |
| F | R6 + BSA |
| Sample | Bioconjugation Efficiency |
|---|---|
| A | 93% |
| B | 85% |
| C | 85% |
| D 1 | 77% |
| E, F 1 | 70% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Way, D.V.; Braido, R.S.; dos Reis, S.A.; Lara, F.A.; Pinto, J.C. Miniemulsion RAFT Copolymerization of MMA with Acrylic Acid and Methacrylic Acid and Bioconjugation with BSA. Nanomaterials 2019, 9, 828. https://doi.org/10.3390/nano9060828
Way DV, Braido RS, dos Reis SA, Lara FA, Pinto JC. Miniemulsion RAFT Copolymerization of MMA with Acrylic Acid and Methacrylic Acid and Bioconjugation with BSA. Nanomaterials. 2019; 9(6):828. https://doi.org/10.3390/nano9060828
Chicago/Turabian StyleWay, Débora Vieira, Rayany Stôcco Braido, Sabrina Alves dos Reis, Flávio Alves Lara, and José Carlos Pinto. 2019. "Miniemulsion RAFT Copolymerization of MMA with Acrylic Acid and Methacrylic Acid and Bioconjugation with BSA" Nanomaterials 9, no. 6: 828. https://doi.org/10.3390/nano9060828
APA StyleWay, D. V., Braido, R. S., dos Reis, S. A., Lara, F. A., & Pinto, J. C. (2019). Miniemulsion RAFT Copolymerization of MMA with Acrylic Acid and Methacrylic Acid and Bioconjugation with BSA. Nanomaterials, 9(6), 828. https://doi.org/10.3390/nano9060828






