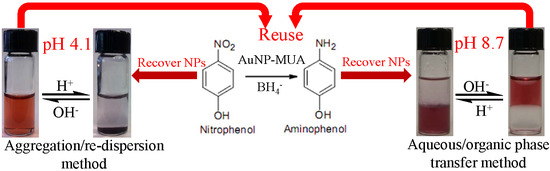
pH-Responsive Mercaptoundecanoic Acid Functionalized Gold Nanoparticles and Applications in Catalysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Equipment
2.2. Mercaptoundecanoic Acid (MUA) Functionalized AuNP Synthesis
2.3. Thermogravimetric (TGA) Analysis
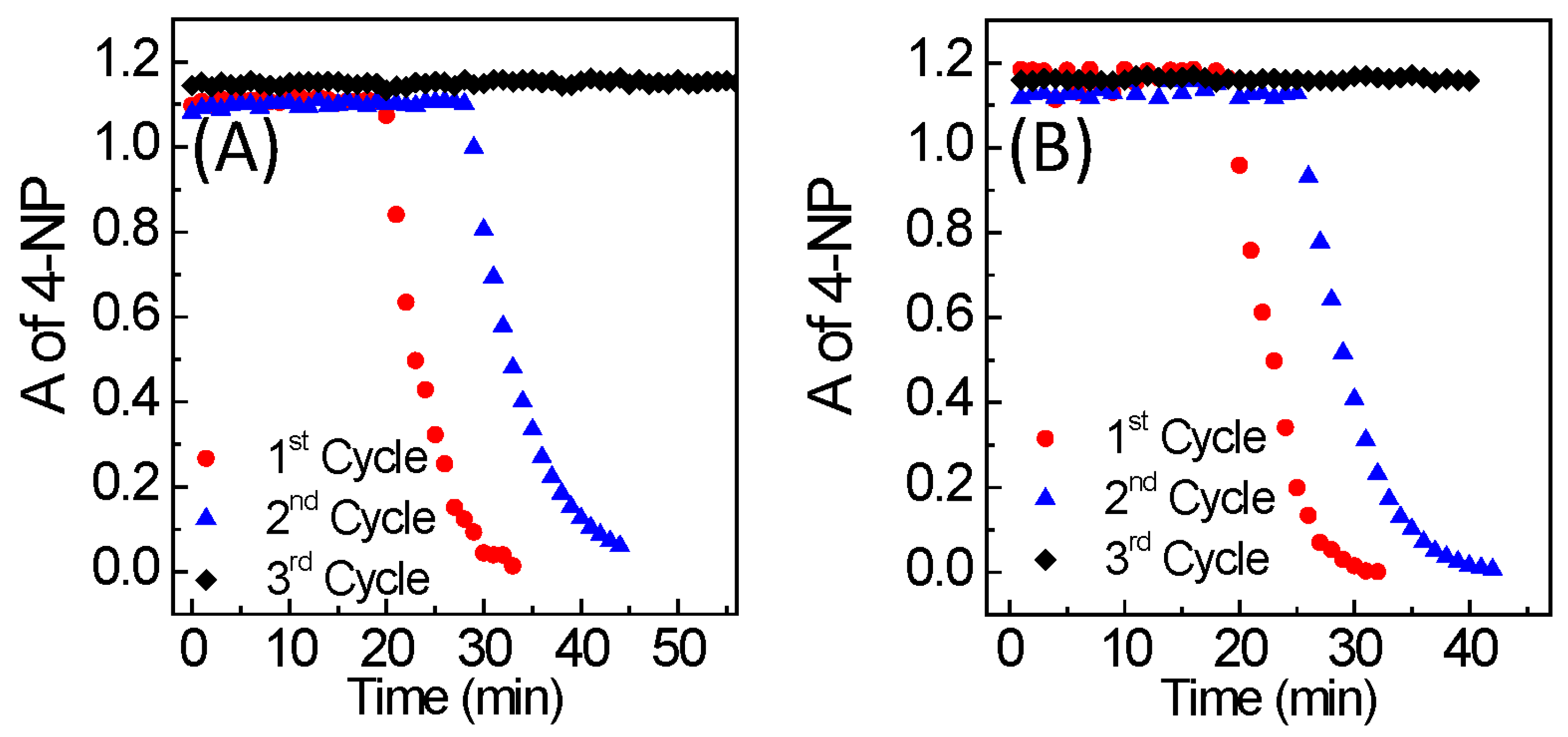
2.4. 4-Nitrophenol Reduction Catalysis
2.5. Transmission Electron Microscopy (TEM) Analysis
2.6. Dynamic Light Scattering (DLS) Measurements
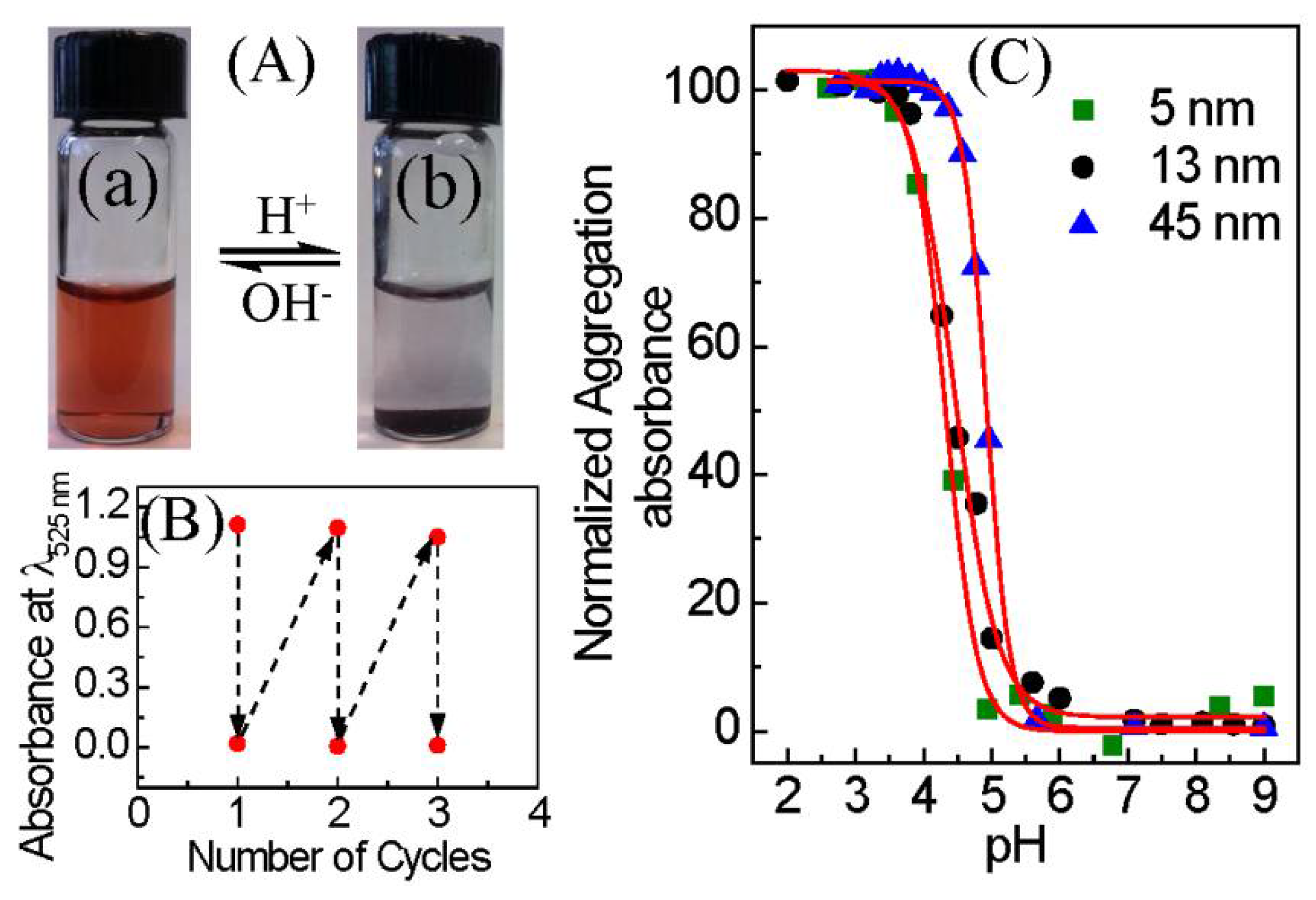
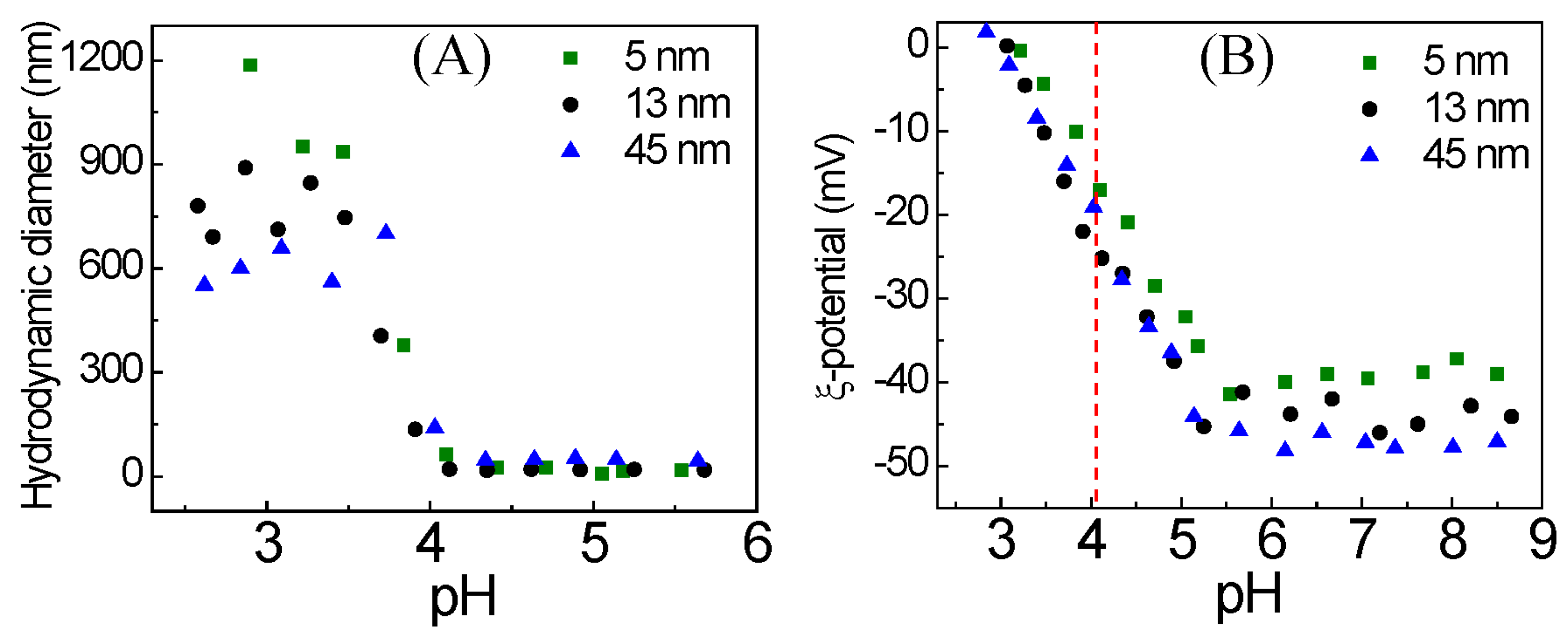
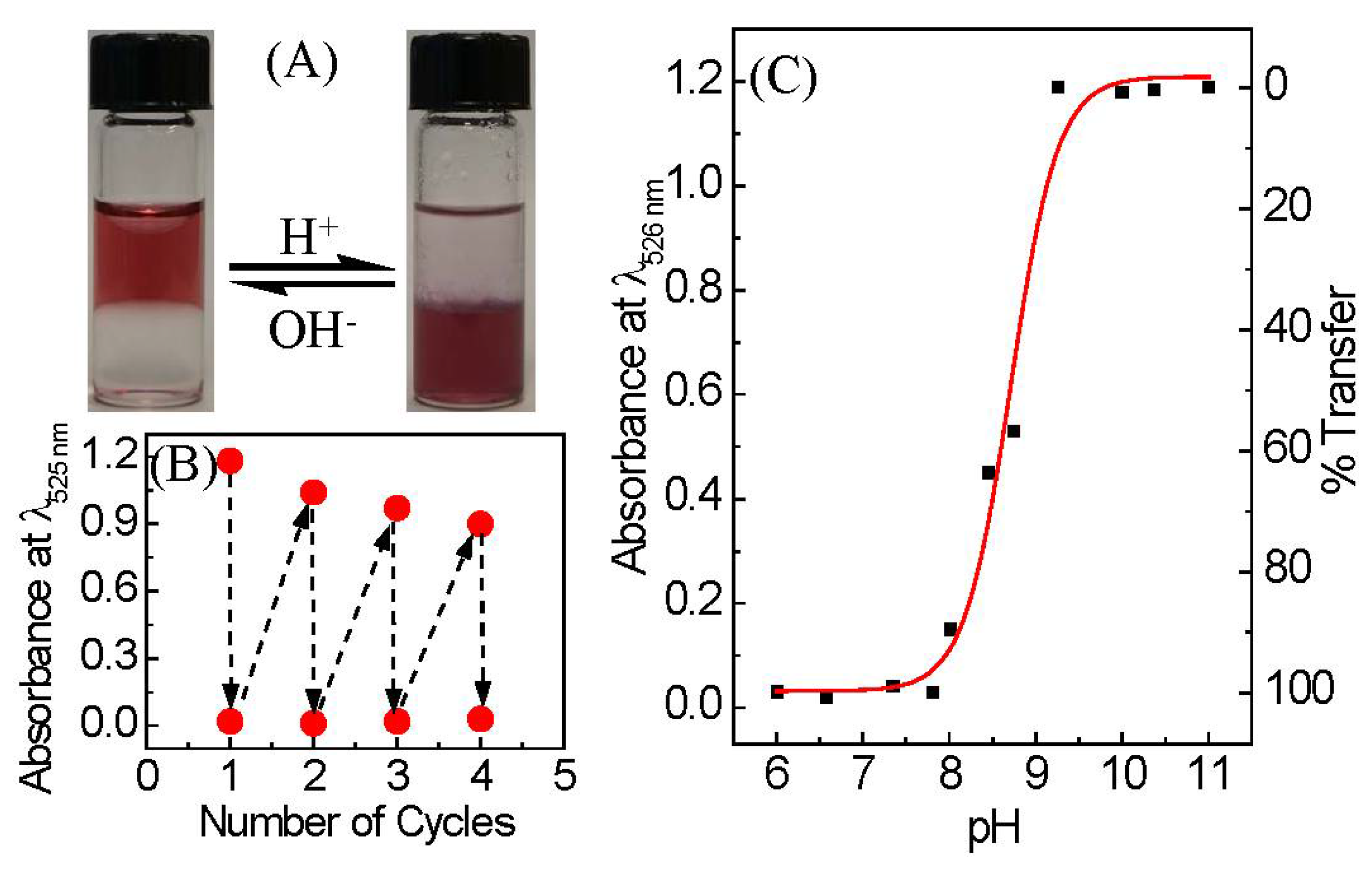
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kang, B.; Mackey, M.A.; El-Sayed, M.A. Nuclear Targeting of Gold Nanoparticles in Cancer Cells Induces DNA Damage, Causing Cytokinesis Arrest and Apoptosis. J. Am. Chem. Soc. 2010, 132, 1517–1519. [Google Scholar] [CrossRef] [PubMed]
- Shang, C.; Liu, Z.-P. Origin and Activity of Gold Nanoparticles as Aerobic Oxidation Catalysts in Aqueous Solution. J. Am. Chem. Soc. 2011, 133, 9938–9947. [Google Scholar] [CrossRef] [PubMed]
- Abadeer, N.S.; Murphy, C.J. Recent Progress in Cancer Thermal Therapy Using Gold Nanoparticles. J. Phys. Chem. C 2016, 120, 4691–4716. [Google Scholar] [CrossRef]
- Yuan, Y.; Yan, N.; Dyson, P.J. pH-Sensitive Gold Nanoparticle Catalysts for the Aerobic Oxidation of Alcohols. Inorg. Chem. 2011, 50, 11069–11074. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.; Won, N.; Jin, H.; Chung, H.; Kim, S. pH-Induced Aggregation of Gold Nanoparticles for Photothermal Cancer Therapy. J. Am. Chem. Soc. 2009, 131, 13639–13645. [Google Scholar] [CrossRef] [PubMed]
- Kanaoka, S.; Yagi, N.; Fukuyama, Y.; Aoshima, S.; Tsunoyama, H.; Tsukuda, T.; Sakurai, H. Thermosensitive Gold Nanoclusters Stabilized by Well-Defined Vinyl Ether Star Polymers: Reusable and Durable Catalysts for Aerobic Alcohol Oxidation. J. Am. Chem. Soc. 2007, 129, 12060–12061. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Johnson, R.C.; Hupp, J.T. Gold Nanoparticle-Based Sensing of “Spectroscopically Silent” Heavy Metal Ions. Nano Lett. 2001, 1, 165–167. [Google Scholar] [CrossRef]
- Wei, Y.; Han, S.; Kim, J.; Soh, S.; Grzybowski, B.A. Photoswitchable Catalysis Mediated by Dynamic Aggregation of Nanoparticles. J. Am. Chem. Soc. 2010, 132, 11018–11020. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Heng, X.; Hu, J. Salt- and pH-Resistant Gold Nanoparticles Decorated with Mixed-Charge Zwitterionic Ligands, and Their Ph-Induced Concentration Behavior. RSC Adv. 2012, 2, 12648–12651. [Google Scholar] [CrossRef]
- Kairdolf, B.A.; Nie, S. Multidentate-Protected Colloidal Gold Nanocrystals: pH Control of Cooperative Precipitation and Surface Layer Shedding. J. Am. Chem. Soc. 2011, 133, 7268–7271. [Google Scholar] [CrossRef] [PubMed]
- Kalsin, A.M.; Kowalczyk, B.; Smoukov, S.K.; Klajn, R.; Grzybowski, B.A. Ionic-Like Behavior of Oppositely Charged Nanoparticles. J. Am. Chem. Soc. 2006, 128, 15046–15047. [Google Scholar] [CrossRef] [PubMed]
- Pillai, P.P.; Huda, S.; Kowalczyk, B.; Grzybowski, B.A. Controlled pH Stability and Adjustable Cellular Uptake of Mixed-Charge Nanoparticles. J. Am. Chem. Soc. 2013, 135, 6392–6395. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, T.; Ahonen, P.; Johans, C.; Kontturi, K. Stability and Electrostatics of Mercaptoundecanoic Acid-Capped Gold Nanoparticles with Varying Counterion Size. ChemPhysChem 2006, 7, 2143–2149. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Tejerina, B.; Lagzi, I.; Kowalczyk, B.; Grzybowski, B.A. Bridging Interactions and Selective Nanoparticle Aggregation Mediated by Monovalent Cations. ACS Nano 2011, 5, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Chia-Hao, S.; Pei-Lin, W.; Chen-Sheng, Y. pH Dependence of Interparticle Coupling for Gold Nanoparticle Assemblies Formation: Electrostatic Attraction and Hydrogen Bonding. Bull. Chem. Soc. Jpn. 2004, 77, 189–193. [Google Scholar] [CrossRef]
- Ansar, S.M.; Kitchens, C.L. Impact of Gold Nanoparticle Stabilizing Ligands on the Colloidal Catalytic Reduction of 4-Nitrophenol. ACS Catal. 2016, 6, 5553–5560. [Google Scholar] [CrossRef]
- Freeman, R.G.; Hommer, M.B.; Grabar, K.C.; Jackson, M.A.; Natan, M.J. Ag-Clad Au Nanoparticles: Novel Aggregation, Optical, and Surface-Enhanced Raman Scattering Properties. J. Phys. Chem. 1996, 100, 718–724. [Google Scholar] [CrossRef]
- Jana, N.R.; Gearheart, L.; Murphy, C.J. Seeding Growth for Size Control of 5–40 Nm Diameter Gold Nanoparticles. Langmuir 2001, 17, 6782–6786. [Google Scholar] [CrossRef]
- Liu, S.H.; Han, M.Y. Synthesis, Functionalization, and Bioconjugation of Monodisperse, Silica-Coated Gold Nanoparticles: Robust Bioprobes. Adv. Funct. Mater. 2005, 15, 961–967. [Google Scholar] [CrossRef]
- Siriwardana, K.; Gadogbe, M.; Ansar, S.M.; Vasquez, E.S.; Collier, W.E.; Zou, S.; Walters, K.B.; Zhang, D. Ligand Adsorption and Exchange on Pegylated Gold Nanoparticles. J. Phys. Chem. C. 2014, 118, 11111–11119. [Google Scholar] [CrossRef]
- Wang, G.; Sun, W. Optical Limiting of Gold Nanoparticle Aggregates Induced by Electrolytes. J. Phys. Chem. B 2006, 110, 20901–20905. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Nap, R.J.; Lagzi, I.; Kowalczyk, B.; Han, S.; Grzybowski, B.A.; Szleifer, I. How and Why Nanoparticle’s Curvature Regulates the Apparent Pka of the Coating Ligands. J. Am. Chem. Soc. 2011, 133, 2192–2197. [Google Scholar] [CrossRef] [PubMed]
- Mulvaney, P. Surface Plasmon Spectroscopy of Nanosized Metal Particles. Langmuir 1996, 12, 788–800. [Google Scholar] [CrossRef]
- Kelly, K.L.; Coronado, E.; Zhao, L.L.; Schatz, G.C. The Optical Properties of Metal Nanoparticles: The Influence of Size, Shape, and Dielectric Environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar] [CrossRef]
- Parviz, D.; Das, S.; Ahmed, H.S.T.; Irin, F.; Bhattacharia, S.; Green, M.J. Dispersions of Non-Covalently Functionalized Graphene with Minimal Stabilizer. ACS Nano 2012, 6, 8857–8867. [Google Scholar] [CrossRef] [PubMed]
- Mefford, O.T.; Vadala, M.L.; Goff, J.D.; Carroll, M.R.J.; Mejia-Ariza, R.; Caba, B.L.; St Pierre, T.G.; Woodward, R.C.; Davis, R.M.; Riffle, J.S. Stability of Polydimethylsiloxane-Magnetite Nanoparticle Dispersions against Flocculation: Interparticle Interactions of Polydisperse Materials. Langmuir 2008, 24, 5060–5069. [Google Scholar] [CrossRef] [PubMed]
- Kalliola, S.; Repo, E.; Sillanpää, M.; Singh Arora, J.; He, J.; John, V.T. The Stability of Green Nanoparticles in Increased pH and Salinity for Applications in Oil Spill-Treatment. Colloids Surf. A Physicochem. Eng. Asp. 2016, 493, 99–107. [Google Scholar] [CrossRef]
- Sugihara, K.; Teranishi, T.; Shimazu, K.; Uosaki, K. Structure Dependence of the Surface Pka of Mercaptoundecanoic Acid Sam on Gold. Electrochemistry 1999, 67, 1172–1174. [Google Scholar]
- Charron, G.; Hühn, D.; Perrier, A.; Cordier, L.; Pickett, C.J.; Nann, T.; Parak, W.J. On the Use of pH Titration to Quantitatively Characterize Colloidal Nanoparticles. Langmuir 2012, 28, 15141–15149. [Google Scholar] [CrossRef] [PubMed]
- Edwards, E.W.; Chanana, M.; Wang, D.; Möhwald, H. Stimuli-Responsive Reversible Transport of Nanoparticles across Water/Oil Interfaces. Angew. Chem. Int. Ed. 2008, 47, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Zhao, Z.; Song, R.; Shanbhag, S.; Tang, Z. A Temperature-Driven Reversible Phase Transfer of 2-(Diethylamino)Ethanethiol-Stabilized Cdte Nanoparticles. Angew. Chem. Int. Ed. 2008, 47, 9875–9878. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; You, M.; Wu, C.; Han, D.; Öçsoy, I.; Chen, T.; Chen, Z.; Tan, W. Reversible Phase Transfer of Nanoparticles Based on Photoswitchable Host–Guest Chemistry. ACS Nano 2014, 8, 2555–2561. [Google Scholar] [CrossRef] [PubMed]
- Dorokhin, D.; Tomczak, N.; Han, M.; Reinhoudt, D.N.; Velders, A.H.; Vancso, G.J. Reversible Phase Transfer of (Cdse/Zns) Quantum Dots between Organic and Aqueous Solutions. ACS Nano 2009, 3, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Luo, Z.; Zhang, Q.; Zhang, X.; Zheng, Y.; Lee, J.Y.; Xie, J. Synthesis of Highly Fluorescent Metal (Ag, Au, Pt, and Cu) Nanoclusters by Electrostatically Induced Reversible Phase Transfer. ACS Nano 2011, 5, 8800–8808. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-W.; Schadt, M.J.; Young, K.; Luo, J.; Zhong, C.-J. Determination of Ion Pairing on Capping Structures of Gold Nanoparticles by Phase Extraction. Analyst 2015, 140, 6239–6244. [Google Scholar] [CrossRef] [PubMed]
- Mayya, K.S.; Caruso, F. Phase Transfer of Surface-Modified Gold Nanoparticles by Hydrophobization with Alkylamines. Langmuir 2003, 19, 6987–6993. [Google Scholar] [CrossRef]
- McMahon, J.M.; Emory, S.R. Phase Transfer of Large Gold Nanoparticles to Organic Solvents with Increased Stability. Langmuir 2007, 23, 1414–1418. [Google Scholar] [CrossRef] [PubMed]
- Dong, A.; Ye, X.; Chen, J.; Kang, Y.; Gordon, T.; Kikkawa, J.M.; Murray, C.B. A Generalized Ligand-Exchange Strategy Enabling Sequential Surface Functionalization of Colloidal Nanocrystals. J. Am. Chem. Soc. 2011, 133, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.-Q.; Yung, L.-Y.L. Ethylenediamine-Assisted Ligand Exchange and Phase Transfer of Oleophilic Quantum Dots: Stripping of Original Ligands and Preservation of Photoluminescence. Chem. Mater. 2013, 25, 2193–2201. [Google Scholar] [CrossRef]
- Frankamp, B.L.; Fischer, N.O.; Hong, R.; Srivastava, S.; Rotello, V.M. Surface Modification Using Cubic Silsesquioxane Ligands. Facile Synthesis of Water-Soluble Metal Oxide Nanoparticles. Chem. Mater. 2006, 18, 956–959. [Google Scholar] [CrossRef]
- Wang, X.; Tilley, R.D.; Watkins, J.J. Simple Ligand Exchange Reactions Enabling Excellent Dispersibility and Stability of Magnetic Nanoparticles in Polar Organic, Aromatic, and Protic Solvents. Langmuir 2014, 30, 1514–1521. [Google Scholar] [CrossRef] [PubMed]
- Woehrle, G.H.; Brown, L.O.; Hutchison, J.E. Thiol-Functionalized, 1.5-Nm Gold Nanoparticles through Ligand Exchange Reactions: Scope and Mechanism of Ligand Exchange. J. Am. Chem. Soc. 2005, 127, 2172–2183. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Yamada, M.; Park, D.-W.; Miyake, M. Particle Size Control of 11-Mercaptoundecanoic Acid-Protected Au Nanoparticles by Using Heat-Treatment Method. Chem. Lett. 2004, 33, 344–345. [Google Scholar] [CrossRef]
- Gangula, A.; Podila, R.M.R.; Karanam, L.; Janardhana, C.; Rao, A.M. Catalytic Reduction of 4-Nitrophenol Using Biogenic Gold and Silver Nanoparticles Derived from Breynia Rhamnoides. Langmuir 2011, 27, 15268–15274. [Google Scholar] [CrossRef] [PubMed]
- Wunder, S.; Polzer, F.; Lu, Y.; Mei, Y.; Ballauff, M. Kinetic Analysis of Catalytic Reduction of 4-Nitrophenol by Metallic Nanoparticles Immobilized in Spherical Polyelectrolyte Brushes. J. Phys. Chem. C 2010, 114, 8814–8820. [Google Scholar] [CrossRef]
- Zeng, J.; Zhang, Q.; Chen, J.; Xia, Y. A Comparison Study of the Catalytic Properties of Au-Based Nanocages, Nanoboxes, and Nanoparticles. Nano Lett. 2010, 10, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.A.; Makis, J.J.; Marvin, K.A.; Rodenbusch, S.E.; Stevenson, K.J. Size-Dependent Hydrogenation of P-Nitrophenol with Pd Nanoparticles Synthesized with Poly(Amido)Amine Dendrimer Templates. J. Phys. Chem. C 2013, 117, 22644–22651. [Google Scholar] [CrossRef]
- Hinterwirth, H.; Kappel, S.; Waitz, T.; Prohaska, T.; Lindner, W.; Lämmerhofer, M. Quantifying Thiol Ligand Density of Self-Assembled Monolayers on Gold Nanoparticles by Inductively Coupled Plasma–Mass Spectrometry. ACS Nano 2013, 7, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, K.; Ishida, T.; Haruta, M. Reduction of 4-Nitrophenol to 4-Aminophenol over Au Nanoparticles Deposited on Pmma. J. Mol. Catal. A Chem. 2009, 298, 7–11. [Google Scholar] [CrossRef]
- Xu, W.; Kong, J.S.; Yeh, Y.-T.E.; Chen, P. Single-Molecule Nanocatalysis Reveals Heterogeneous Reaction Pathways and Catalytic Dynamics. Nat. Mater. 2008, 7, 992–996. [Google Scholar] [CrossRef] [PubMed]
- Wunder, S.; Lu, Y.; Albrecht, M.; Ballauff, M. Catalytic Activity of Faceted Gold Nanoparticles Studied by a Model Reaction: Evidence for Substrate-Induced Surface Restructuring. ACS Catal. 2011, 1, 908–916. [Google Scholar] [CrossRef]
- Zhou, X.; Xu, W.; Liu, G.; Panda, D.; Chen, P. Size-Dependent Catalytic Activity and Dynamics of Gold Nanoparticles at the Single-Molecule Level. J. Am. Chem. Soc. 2010, 132, 138–146. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ansar, S.M.; Chakraborty, S.; Kitchens, C.L. pH-Responsive Mercaptoundecanoic Acid Functionalized Gold Nanoparticles and Applications in Catalysis. Nanomaterials 2018, 8, 339. https://doi.org/10.3390/nano8050339
Ansar SM, Chakraborty S, Kitchens CL. pH-Responsive Mercaptoundecanoic Acid Functionalized Gold Nanoparticles and Applications in Catalysis. Nanomaterials. 2018; 8(5):339. https://doi.org/10.3390/nano8050339
Chicago/Turabian StyleAnsar, Siyam M., Saptarshi Chakraborty, and Christopher L. Kitchens. 2018. "pH-Responsive Mercaptoundecanoic Acid Functionalized Gold Nanoparticles and Applications in Catalysis" Nanomaterials 8, no. 5: 339. https://doi.org/10.3390/nano8050339
APA StyleAnsar, S. M., Chakraborty, S., & Kitchens, C. L. (2018). pH-Responsive Mercaptoundecanoic Acid Functionalized Gold Nanoparticles and Applications in Catalysis. Nanomaterials, 8(5), 339. https://doi.org/10.3390/nano8050339