Abstract
Novel carbon quantum dots (CQDs) modified with Bi2O2CO3 (CQDs/Bi2O2CO3) were prepared using a simple dynamic-adsorption precipitation method. X-ray diffractometry (XRD), transmission electron microscopy (TEM), energy dispersive X-ray spectroscopy (EDX), and scanning electron microscopy (SEM) were used to test the material composition, structure, and band structures of the as-prepared samples. Methylene blue (MB) and colorless phenol, as target organic pollutants, were used to evaluate the photocatalytic performance of the CQDs/Bi2O2CO3 hybrid materials under visible light irradiation. Experimental investigation shows that 2–5 nm CQDs were uniformly decorated on the surface of Bi2O2CO3; CQDs/Bi2O2CO3 possess an efficient photocatalytic performance, and the organic matter removal rate of methylene blue and phenol can reach up to 94.45% and 61.46% respectively, within 2 h. In addition, the degradation analysis of phenol by high performance liquid chromatography (HPLC) proved that there are no other impurities in the degradation process. Photoelectrochemical testing proved that the introduction of CQDs (electron acceptor) effectively suppresses the recombination of e−-h+, and promotes charge transfer. Quenching experiments and electron spin resonance (ESR) suggested that ·OH, h+, and ·O2− were involved in the photocatalytic degradation process. These results suggested that the up-conversion function of CQDs could improve the electron transfer and light absorption ability of photocatalysts and ·O2− formation. Furthermore, the up-conversion function of CQDs would help maintain photocatalytic stability. Finally, the photocatalytic degradation mechanism was proposed according to the above experimental result.
1. Introduction
In recent years, life and industrial sewage treatments have attracted increasing attention. Sewage is an important factor that affects environmental governance. Photocatalytic technology used for treating nocuous organic pollutants in water has recently received a lot of interest [1,2]. Pollutants can be effectively degraded via the generation of photoinduced electron-holes in photocatalysts under visible light irradiation. The above problems could be solved using semiconductor photocatalytic technology, which has been regarded as a promising green technology [3,4]. TiO2 has been a highly researched material because of its non-toxicity, chemical stability, commercial availability, high photoactivity, etc. [5,6,7] However, its practical application was limited by two main disadvantages: (1) poor visible light response ability, and (2) a low photogenerated charge separation efficiency [8]. Thus, many efforts have been made to explore new visible light photocatalytic materials which possess efficient photocatalytic activity.
In recent years, bismuth-based oxides which possess aurivillius-layered structures have been the focus of much research research, due to their hybridized valence band by O2p and Bi6s [9]. Some advantages of these photocatalysts are their good thermal stability, non-toxicity, etc. Some examples include BiVO4 [10,11,12], BiWO6 [13,14,15], BiOX (X = Br, Cl, I) [16,17,18], BiPO4 [19], and Bi2MoO6 [20]. Among these photocatalysts, the Bi2O2CO3 photocatalyst is a new type of semiconductor photocatalytic material which has a unique layer structure consisting of CO32− layers interwoven by [Bi2O2]2+ layers [21]. Thus, Bi2O2CO3 as a potential performance photocatalyst has gradually caught the attention of researchers. Thus far, the different morphological structures of Bi2O2CO3 have been studied, such as flower-like, sponge-like, porous ball, slice layer, etc. Various organic pollutants have also been used to investigate their photocatalytic performance [22,23]. However, because of its weak visible light response and wide band structure, modification research of Bi2O2CO3 is very important [24]. Hence, an effective way to improve the photocatalytic performance of Bi2O2CO3 may be to introduce a narrow band gap semiconductor, such as Bi2O2CO3/BiOI [25], Ag-AgBr/Bi2O2CO3 [26], Bi2O2CO3/Bi2S3 [27], etc.
Carbon quantum dots (CQDs), which are smaller than 10 nm in size, are well-dispersed spherical fluorescent nanocarbon materials. They gradually roused interest because of their easy functionalization, powerful chemical inertness, good photo-stability, excellent biocompatibility, and non-toxicity [28,29,30]. In addition, its photoluminescence (PL) up-conversion is a unique property [31]. Visible light emissions and shorter wavelength UV could be generated from near infrared (NIR) light radiation. The long wavelength of visible light makes CQDs a potential material for photocatalysis, solar cells, bioluminescence, etc., via the PL up-conversion effect [32,33,34].
CQDs can be either photoinduced electron donors or acceptors, due to their photoelectrochemical properties and their being richly fluorescent [28,35]. CQDs can either be used as a photocatalytic material or as functional components in composite photocatalyst designs to broaden the spectrum response range and promote the separation of photoinduced e−-h+ [32]. They can also inhibit semiconductor photolysis, such as in CQDs/Fe2O3 [36] and CQDs/ZnO [37].
In this research, we present a simple method to fabricate CQDs/Bi2O2CO3 composite with 3D flower micro-sphered structures. Structure and morphology characterizations, as well as the mechanism behind the efficiency promotion effect of CQDs/Bi2O2CO3, were carefully studied. The photocatalytic property of the CQDs/Bi2O2CO3 compound was investigated by removing methylene blue (MB) and colorless phenol, under simulated sunlight irradiation.
2. Experimental Methods
2.1. Photocatalyst Synthesis
All of the chemicals are analytical reagents without further purification.
2.1.1. Synthesis of the 3D Flower-Like Bi2O2CO3
The 3D flower-like Bi2O2CO3 precursor was prepared by using the classic hydrothermal method. Using a classic course to synthesize the Bi2O2CO3 flower-like precursor, 0.003 mol of Bi(NO3)3·5H2O was dissolved in 20 mL of 1 M HNO3, and then 0.002 mol of citric acid was added. After 10 min stirring, NaOH was added to adjust the pH of the above reaction solution to 4.0–4.2. Then the reaction solution was transferred in a Teflon-lined, stainless steel autoclave, and maintained at 160 °C for 24 h. After natural cooling to room temperature, the prepared precursor was handled by repeated washing and centrifugation with ethanol and distilled water, and subsequently dried at 80 °C for 8 h.
2.1.2. Preparation of Carbon Quantum Dots (CQDs)
A pair of graphite rods with a 7.5 cm separation were parallelly inserted into deionized water as the anode and counter-electrode (18.4 MΩ∙cm−1, 400 mL) [38]. A direct current (DC) supplies a 30 V static potential to the two electrodes. The solution then gradually became dark yellow, with corrosion being observed on the anode graphite rod. Finally, solution became a dark brown after a period of electrolytic process. The electrolyte was first filtered with filter paper. The filtrate was then centrifuged at 22,000 rpm for 30 min, which could remove the graphite particles and graphite oxide. The resultant solution is the ideal CQDs aqueous solution. (C(CQDs) = 2.01 g/L)
2.1.3. Preparation of CQD/Bi2O2CO3 Photocatalysts
CQD/Bi2O2CO3 composites were synthesized by a simple dynamic adsorption precipitation method. 0.3 g of Bi2O2CO3 photocatalyst were added to 50 mL of deionized water under ultrasonic treatment for 1 h. Next, a defined volume of CQD solution (10, 30, 50, 70 and 90 mL) was added to the above solution and stirred for 90 min at room temperature to create a clear dispersion of CQDs. The obtained samples were labeled as 10-CQDs/Bi2O2CO3, 30-CQDs/Bi2O2CO3, 50-CQDs/Bi2O2CO3, 70-CQDs/Bi2O2CO3, 90-CQDs/Bi2O2CO3, respectively. The CQD/Bi2O2CO3 photocatalysts were handled by repeated washing and centrifugation with ethanol and distilled water, and subsequently dried at 80 °C for 8 h.
2.2. Photocatalyst Characterization
The crystal structures and phase data of samples could be determined by X-ray diffractometry (XRD) (D/MAX2500 PC, Rigaku Corporation, Tokyo, Japan). The morphologies and composition of the samples could be investigated by transmission electron microscopy (TEM) (JEM-2010, JEOL Ltd., Akishima, Japan), energy dispersive X-ray spectroscopy (EDX) (s-4800, Hitachi, Chiyoda, Japan), and scanning electron microscopy (SEM) (s-4800, Hitachi, Chiyoda, Japan). The spectrofluorometer (f7000, Hitachi, Chiyoda, Japan) could be used to investigate the separation efficiency of photo-induced charge for powdered samples. The Perkin Elmer System 2000 infrared spectrometer provided the Fourier transform infrared (FTIR) spectra (Perkin Elmer, Shanghai, China), with KBr as the reference sample. The spin trapping electron spin resonance (ESR) measurements were performed on a Bruker JES FA200 ESR spectrometer (Oubeier, Beijing, China). The electrochemical and photoelectrochemical measurements were performed via a three-electrode quartz cell system. A CHI 660B electrochemical system (Shanghai Chenhua Instrument Corp., Shanghai, China) was used to record the photoelectrochemical results.
2.3. Photocatalytic Activity
The degradation rate of MB (or colorless phenol) was used to test the photocatalytic performance of CQDs/Bi2O2CO3 samples under the simulated sunlight (a 400 W metal halide lamp, λ > 400 nm, transmittance > 90%) in a multi-tube agitated reactor (XPA-7) (Xujiang, Nanjing, China). Thermostatic water provided cooling to control the reaction temperature (25 ± 2 °C). Firstly, 0.05 g of CQDs/Bi2O2CO3 powder was added to 50 mL of a 10 mg/L MB solution. Before light source irradiation, it was necessary to reach adsorption equilibrium for CQDs/Bi2O2CO3 in the absence of light about 30 min. During metal halide lamp irradiation, 3 mL reaction solution was measured out every 15 min in 120 min and stored in a dark environment. Finally, the collected reaction solutions were centrifuged for 6 min at 10,000 rpm, to separate the sample particles. A UV-vis spectrophotometer (UV-1901, Puxi, Beijing, China) was used to analyze the supernatant solutions (MB). (The determining wavelength: 400 nm < λ < 800 nm)
The degradation efficiency (%) was calculated as follows:
where C and C0 were the t min and initial concentration of MB, respectively.
Additionally, blank and contrast tests were also performed. However, the high-performance liquid chromatography (HPLC) was used to analyze the concentration of phenol. The HPLC column was a 15 cm × 4.6 mm C18 column with a particle size of 5 μm. The mobile phase consisted of methanol/water = 70:30. The flow rate was 1 mL/min and column temperature of 35 °C.
2.4. Photoelectrocatalytic Activity
A photoelectrocatalytic test was used to evaluate the charge separation efficiency of the CQDs/Bi2O2CO3. In the test, CHI 660E Electrochemical Workstation (Shanghai Chenhua Instrument Corp., Shanghai, China) carried out all photoelectrochemical experiments results via a conventional three-electrode quartz cell system in a homemade quartz reactor (electrolyte: 0.1 M Na2SO4). A 500-W Xe lamp purchased (Beijing Changtuo Co. Ltd., Beijing, China) with a UV cut-off filter (420 nm) was used for light source. The prepared photoelectrode was used as a working electrode, while Pt was used as a counter electrode, and a saturated calomel electrode (SCE) was used as the reference electrode.
3. Results and Discussion
3.1. Catalyst Characterization
Figure 1 shows the XRD characteristic peak of the Bi2O2CO3 and different CQDs/Bi2O2CO3 composites, with various modified quantities of CQDs. The composition and crystalline properties of the photocatalysts can be identified using XRD. As shown, the curve of pure Bi2O2CO3 possesses many characteristic and distinct diffraction peaks, which perfectly match the diffraction peaks of tetragonal phase Bi2O2CO3 (JCPDS: 41-1488), suggesting satisfactory composition and purity. The main characteristic peak positions of samples appeared at 12.93°, 23.90°, 26.03°, 30.25°, 32.73°, 35.31°, 39.51°, 42.30°, 46.97°, 48.93°, 52.23°, 53.41°, 54.51° and 56.90°. This corresponds to the (002), (011), (004), (013), (110), (112), (006), (114), (020), (022), (116), (121), (024) and (123) crystal faces of Bi2O2CO3, respectively. In addition, the sharp and intense characteristic peaks suggested that the samples possessed good crystallinity. The XRD patterns of CQDs/Bi2O2CO3 photocatalysts were largely similar to those of Bi2O2CO3. The diffraction peaks of CQDs were not obvious, even at highly modified amounts, due to high dispersion and the small amount of the CQDs in the CQDs/Bi2O2CO3 composite.

Figure 1.
XRD patterns of the synthesized photocatalysts.
An ultraviolet-visible (UV-vis) diffuse reflection spectrum can be used to measure the light absorption ability of photocatalysts. Additionally, the band gap energies of semiconductor photocatalysts could be calculated according to their electronic structure features on the UV-vis absorption curve. As shown in Figure S1a, less than 370 nm of UV light can be absorbed by the pure Bi2O2CO3 sample. The absorption ability of Bi2O2CO3 was weak in the visible region. After the introduction of CQDs, the light absorption ability of Bi2O2CO3 was markedly enhanced, and the absorption edges of the CQDs/Bi2O2CO3 exhibited a slight systematic red-shift. Furthermore, the light absorption intensity of CQDs/Bi2O2CO3 composites were obviously enhanced with increases of the CQD content. Additionally, the absorption spectrum of CQDs further confirms that the up-conversion function of CQDs could promote full spectrum absorption. Figure S1b shows the band gap energies of the prepared photocatalysts. The forbidden band of a crystalline semiconductor could be calculated using the following formula [39]:
where α is the absorption coefficient, h is the Plank constant, ν is the light frequency, Eg is the band gap and A is a constant [40].
The value of n is 1 or 4, depending on the photocatalyst type for a direct or indirect transition, respectively. Therefore, the n value of Bi2O2CO3 is 4 due to the indirect transition band gaps [41,42]. The plot of (αhν)1/2 versus the photon energy (hν) could calculated the band gap energy of Bi2O2CO3 and CQDs/Bi2O2CO3 through the intercept of the tangent to the x-axis. As shown in Figure S1b, the band gap of pure Bi2O2CO3 was approximately 3.4 eV. The above results indicated that CQDs/Bi2O2CO3 composites have significant visible light absorption ability, due to the deposition and up-conversion behavior of CQDs. The introduction of CQDs effectively promotes the charge-transfer between CQDs and Bi2O2CO3. It broadens the optical absorption edges and enhanced the optical absorption intensity of CQDs/Bi2O2CO3 composites. In summary, the up-conversion behavior of CQDs effectively improved the photocatalytic degradation ability of CQDs/Bi2O2CO3 photocatalysts under visible light irradiation.
Valence band X-ray photoelectron spectroscopy (VB XPS) could be used to calculate the valence band (VB) position of photocatalysts. As shown in Figure S2a, the valence band of Bi2O2CO3 is about 3.56 eV. According to the result of Figure S1b, the conduction band (CB) edge of Bi2O2CO3 should be located at 0.16 eV. This result is supported by the Mott-Schottky (MS) plots experiment (Figure S2b). The MS plots for Bi2O2CO3 indicate typical n-type semiconductors with a typical shape [43]. The conduction band is about 0.15 eV. The measured value was close to the calculated value. The above results could provide a strong theoretical basis for the discussion of the photocatalytic degradation mechanism.
The TEM analysis was used to characterize the microstructure and morphology of Bi2O2CO3 and CQDs/Bi2O2CO3. Figure 2a shows the TEM image of the prepared Bi2O2CO3 sample. The average diameter of the flower-like microsphere structure was about 1 to 2 µm, which consists of 2D nanosheet structures. Figure 2b,c show the TEM images of CQDs/Bi2O2CO3 samples. It is obvious that a mass of CQDs are evenly attached to the surface of the Bi2O2CO3 “flower-like” nanosheets. The results demonstrated that the CQDs modified on the surface of Bi2O2CO3 were homodisperse, with a 2–5 nm spherical diameter. The modifications of CQDs can significantly increase the number of photocatalytic reaction sites and the surface roughness of the Bi2O2CO3 flower structure [44], thereby significantly improving the photocatalytic degradation efficiency. The HRTEM image (Figure 2d) showed that the interplanar spacing of CQDs and Bi2O2CO3 was 0.33 and 0.30 nm, corresponding to the (002) and (013) crystallographic planes, respectively. Figure 2e shows the selected-area electron diffraction (SAED) pattern of CQDs/Bi2O2CO3. The high-crystallinity of CQDs/Bi2O2CO3 photocatalysts was obvious according to the clear lattice fringe. The elemental composition of the CQDs/Bi2O2CO3 photocatalyst was estimated using EDS analysis. The EDS spectrum shown in Figure 2f revealed that the CQDs/Bi2O2CO3 composite photocatalyst contained only Bi, C, and O. The measured value of the C element was more than predicted in the theoretical value of C in Bi2O2CO3, which confirmed the existence of CQDs on the surface of Bi2O2CO3. The above results indicate the effective compounding of CQDs and Bi2O2CO3.

Figure 2.
TEM images of as-prepared samples: (a) Bi2O2CO3; (b,c) CQDs/Bi2O2CO3; (d–f) HRTEM, SAED, EDX images of CQDs/Bi2O2CO3.
Figure 3a shows the photoluminescence (PL) spectra of Bi2O2CO3 and CQDs/Bi2O2CO3. The PL spectra were dependent on the 260 nm excitation wavelengths. It is known that the recombination degree of the photogenerated e−-h+ could be reflected by the PL technique. Therefore, to a certain extent, the PL spectra could reflect the photocatalytic properties of semiconductors. Clearly, the shapes and peak positions of Bi2O2CO3 were similar to those of CQDs/Bi2O2CO3. However, the peak intensity of CQDs/Bi2O2CO3 decreased, suggesting the high separation efficiency of the carrier charge when CQDs are decorated on the surface of Bi2O2CO3. Figure 3b shows the fluorescence spectrum peaks of CQDs excited by long wavelengths. The up-conversion PL spectra show excitation-dependent PL feature spectra. With the excitation wavelengths increasing from 600 nm to 900 nm, the up-converted emission peaks exhibit a red shift from 430 nm to 520 nm. The above results show that CQDs have strong fluorescence up-conversion properties.

Figure 3.
(a) Photoluminescence (PL) spectra of Bi2O2CO3 and CQDs/Bi2O2CO3; (b) PL curve of CQDs excited by different wavelengths of light.
The presence of CQDs in CQDs/Bi2O2CO3 photocatalysts could be further determined using a Fourier transform of the infrared (FTIR) spectra. As shown in Figure S3, by comparing the FTIR spectrums of CQDs, Bi2O2CO3 and CQDs/Bi2O2CO3, no obvious impurity absorption peaks were detected. For the CQDs/Bi2O2CO3 material, the stretching vibrations of the Bi-O in Bi2O2CO3 could be obtained according to the intensive peaks at about 560–850 cm−1. The stretching vibrations of C–O and C=O were assigned to the peaks at approximately 1400 cm−1 [45], respectively. The absorption band at approximately 1040 cm−1 attributes to C–C or C–O stretching vibrations. The existence of COO− in CQDs could be proven by the presence of 1452 cm−1 absorption peaks [46]. The peaks at 1705–3454 cm−1 are due to the bending vibrations of water molecules absorbed on CQDs/Bi2O2CO3, and stretching vibrations of the –OH group. The peaks at about 2950 cm−1 are attributed to C–H stretching and rocking band vibrations, respectively [47]. All these results successfully prove the existence of CQDs and Bi2O2CO3, and suggest the combination and an interface interaction between CQDs and Bi2O2CO3. The XPS analysis (Figure 4) could be further used to prove the results.
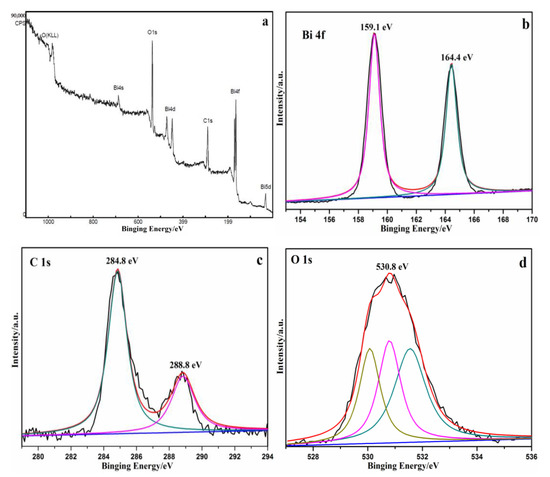
Figure 4.
XPS characteristic peak of the CQDs/Bi2O2CO3 sample. (a) Survey of the sample; (b) Bi 4f; (c) C 1s; (d) O 1s.
The surface properties and chemical compositions of the samples could be determined by using an X-ray photoelectron spectrometer (XPS). Figure 4a displays the full survey spectrum of CQDs/Bi2O2CO3, which suggested the existence of oxygen (O 1s), carbon (C 1s) and bismuth (Bi 4f). The high-resolution XPS spectra of Bi 4f appeared at about 159.1 eV (Bi 4f5/2) and 164.4 eV (Bi 4f7/2) (Figure 4b), which belong to a crystal structure of Bi3+ [48]. In the narrow scan of the C 1s (Figure 4c), the main peak at 284.8 eV is ascribed to the C–C sp2-hybridizedcarbon of CQDs [49]. The characteristic peaks of the C 1s, which appeared at 288.8 eV, was due to the presence of C–O–C and/or C=O [50]. The peak of O 1s was shown in Figure 4d. The characteristic peak could be split into three peaks. The peaks at 531.5, 530.8 and 530.0 eV are ascribed to adsorption oxygen, hydroxyl oxygen, and lattice oxygen, respectively. The presence of CQDs in CQDs/Bi2O2CO3 samples could be confirmed according to the C 1s and O 1s patterns, which are consistent with the FTIR analysis results (Figure S4).
3.2. Photocatalytic Activity
MB was used as target pollutants to evaluate the photocatalytic degradation performance of the prepared samples under metal halide lamp irradiation. The 30 min absorption process was used to ensure the absorption-desorption balance between the photocatalyst and pollutants. Firstly, the degradation of MB by the composite without light irradiation, as well as the direct photolysis of MB without a photocatalyst, was investigated; the results are presented in Figure 5a. In the dark or without a photocatalyst, only a slight degradation of MB was observed. However, it is obvious that the photocatalytic property of Bi2O2CO3 improved significantly with the introduction of CQDs. 50-CQDs/Bi2O2CO3 showed the highest degradation efficiency (94.45%) within a 2 h irradiation, which was higher than that of Bi2O2CO3 (54.43%) for the same time period. CQDs could promote the photocatalytic process by improving the visible light responsiveness, or enhancing the charge separation efficiency of the system. Specifically, Bi2O2CO3 could be excited to generate the electrons-hole pairs by short wavelengths (ultraviolet light) by up-converting the long wavelength (visible light) via CQDs on the surface of Bi2O2CO3. Meanwhile, CQDs function as an excellent electron mediator and acceptor in CQDs/Bi2O2CO3 to effectively separate e−-h+ pairs, and improve degradation efficiency. The function mechanism was similar to that of previous studies [51,52,53]. Figure 5b shows the UV-vis absorption spectra temporal changes for MB solutions in the presence of 50-CQDs/Bi2O2CO3. It is obvious that the arresting characteristic peak (664 nm) weakened in intensity as the reaction time increased. Therefore, the final solution almost became colorless from the initial blue after approximately 120 min of irradiation, which further implies the complete destruction of the conjugated structure of MB. Figure 5c shows the total organic carbon (TOC) removal of MB as a function of reaction time. After 120 min illumination, the TOC removal by Bi2O2CO3 and 50-CQDs/Bi2O2CO3 were 4.67% and 80.64%, respectively. Additionally, High performance liquid chromatography (HPLC) was used to further verify the degradation process.
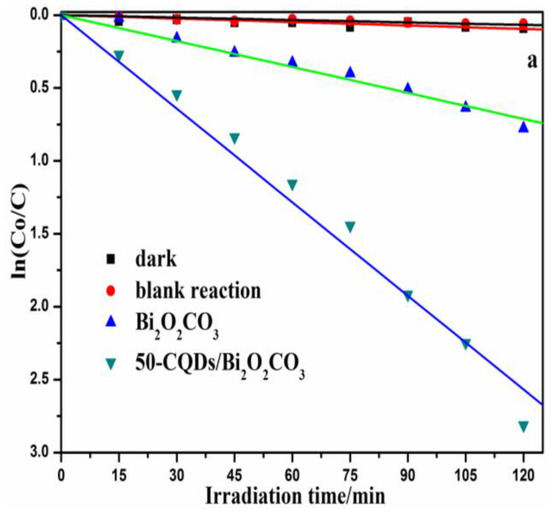

Figure 5.
(a) Comparison of the photocatalytic degradation efficiency of MB by different photocatalysts (b) UV-vis spectral absorption changes of MB solution degraded by the 50-CQDs/Bi2O2CO3 composite; (c) TOC removal of MB.
As is known, pseudo-first-order kinetics is suitable for the photocatalytic degradation of organic pollutants. The kapp values of CQDs/Bi2O2CO3 composites with different amounts of CQDs are shown in Figure S4. More CQDs resulted in higher kinetic constants for the CQDs/Bi2O2CO3, and the highest kinetic constant (0.0214 min−1) appeared when using 50 mL of CQDs. The kinetic constant did not increase with a further increase of CQDs, and even decreased sharply when 90 mL of CQDs was used. This may be due to the fact that when the CQD content is sufficiently high, there is an associated decrease in the number of available active sites, and thus, lower photoelectrocatalytic activity [54]. In such a situation, excess CQDs could act as recombination sites for electron-hole pairs. A suitable quantity of CQDs could effectively improve the synergetic effects of CQDs and Bi2O2CO3, which largely contributes to the separation of electron-hole pairs.
In order to verify the role of active species in the organic pollutants degradation process over CQDs/Bi2O2CO3 hybrid materials in detail, quenching experiments were performed. As shown in Figure 6, isopropanol (IPA, 0.01 M) and ethylenediamine tetraacetic acid disodium (EDTA-2Na, 0.01 M) were used to quench the hydroxyl radicals (·OH) and holes (h+), Nitrogen (N2) was used as the superoxide radical (·O2−) scavenger. The addition of IPA, EDTA-2Na and N2 made the kinetic constants decrease from 0.0214 min−1 to 0.0086 min−1, 0.0033 min−1 and 0.0017 min−1, respectively. As the kinetic constants decreased, a greater effect was observed for the corresponding active species. Therefore, these above results suggested that the ·O2−, ·OH and h+ were involved in the photocatalytic degradation process. Moreover, the ·O2− was the main active species in degradation process.

Figure 6.
Different photocatalytic degradation performances after adding different quenching agents ((a) IPA, (b)EDTA-2Na, (c) N2); (d) The corresponding degradation kinetic constant.
To further confirm the generation of ·O2− and ·OH species, the ESR spin-trap measurement based on CQDs/Bi2O2CO3 photocatalysts under dark and visible light irradiation was conducted. DMPO (5, 5-dimethyl-1-pyrroline N-oxide), a nitrone spin trapping reagent, was utilized to capture the superoxide (·O2−) and hydroxyl radicals (·OH). As shown in Figure 7a,b, negligible ESR signals were observed under dark conditions. However, the four-line characteristic ESR signal for DMPO-·O2−, a signal for the DMPO-·OH spin adduct were found under visible light irradiation. Furthermore, in the ESR spectra of DMPO-·OH for CQDs/Bi2O2CO3, four sets of antisymmetric peaks with intensities of 1:2:2:1 were observed, which are the characteristic signals of DMPO-·OH adducts. ESR demonstrates that ·O2− and ·OH are the main active species of CQDs/Bi2O2CO3 in the photocatalytic degradation process under visible light illumination.
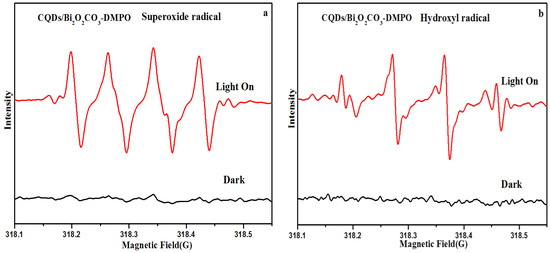
Figure 7.
ESR spectra of radical adducts trapped by DMPO: (a) superoxide radical (·O2−); (b) hydroxyl radical (·OH).
The stability of the photocatalysts is an important factor in its industrial application. Recycling reactions were used to evaluate the stability of the CQDs/Bi2O2CO3 samples. As shown in Figure 8, the degradation rate of CQDs/Bi2O2CO3 samples decrease from 94.45% (1st) to 82.91% (5th); the photocatalytic efficiency was only slightly lower. Considering the loss of catalyst in each cycling process and the test error, the experimental results imply that the CQDs/Bi2O2CO3 possessed good stability. This suggests that the oxygen vacancies of CQDs/Bi2O2CO3 could effectively be refreshed by the up-conversion effect of CQDs, and therefore, CQDs/Bi2O2CO3 possess good stability [55]. CQDs/Bi2O2CO3 materials have great potential and prospects for practical application in the future.

Figure 8.
Cycling runs for the photocatalytic degradation of MB by CQDs/Bi2O2CO3 hybrid materials under visible light irradiation. (MB: 50 mL, 10 mg/L; CQDs/Bi2O2CO3: 1 g/L).
The transient photocurrent response could be used to verify the separation efficiency of photoinduced charges in the samples, as shown in Figure 9. The photocurrent intensity of Bi2O2CO3 was 0.04 mA/cm2. The photocurrent intensity of CQDs/Bi2O2CO3 was higher than that of Bi2O2CO3 (0.11 mA/cm2). The results demonstrated that CQDs in CQDs/Bi2O2CO3 effectively improved the separation of the photogenesis charge, and increased the numbers of generated electrons [55], thus improving the degradation efficiency. These results were consistent with PL analysis (Figure 3); the electrochemical impedance spectra (EIS) (Figure 10) further confirmed this fact.

Figure 9.
the photocurrent of Bi2O2CO3 and CQDs/Bi2O2CO3 under visible light.

Figure 10.
(a) Electrochemical impedance spectra (EIS) of Bi2O2CO3 and CQDs/Bi2O2CO3 under visible light; (b) The local amplification of (a).
The most critical drawback in photocatalysis is the recombination process. EIS is quite a useful tool for investigating the charge transfer and recombination processes at the semiconductor/electrolyte interface [56]. Impedance spectra could be fitted by the ZsimpWin software according to the corresponding circuit. As shown in Figure 10a,b, the series resistance (R1) of the Pt counter electrode corresponds to the intercept on the real axis (high frequency). The charge-transfer resistance (R2) of the Pt counter electrode/electrolyte interface corresponds to the first semi-circle (high frequency). The charge-transfer resistance (R3) of the prepared samples anode/electrolyte interface corresponds to the second semi-circle (middle frequency) [57]. Compared with Bi2O2CO3, the smaller charge transfer resistance could be obtained from CQDs/Bi2O2CO3 using the smaller arc radius [58]. Thus, the performance of CQDs/Bi2O2CO3 contributed to the fast transfer and effective separation of e−-h+ pairs. The above results indicated that the introduction of CQDs could improve the electron transfer of photocatalysts and promote the photocatalytic property of CQDs/Bi2O2CO3.
In order to prove the degradation ability of the CQDs/Bi2O2CO3 photocatalyst for organic pollutants, and explore the organic molecule changes in the degradation process, phenol was degraded as the target pollutant under visible light irradiation. Then, this was tested by using high performance liquid chromatography (HPLC). As shown in Figure 11a, the result revealed that the CQDs/Bi2O2CO3 sample had a higher photocatalytic property than pure Bi2O2CO3 for the phenol; 61.96% phenol was degraded for CQDs/Bi2O2CO3 within 120 min. As Figure 11b–d shows, the characteristic peak of phenol, located at 3.35 min retention time (RT), gradually weakens with increasing reaction time. No obvious impurity peak appeared in the degradation process. The results imply that phenol degradation was the mineralization process, and that CQDs could effectively promote the photocatalytic property of CQDs/Bi2O2CO3. Additionally, based on the degradation of two types of model organic pollutants (MB and phenol), the above results imply that the CQDs/Bi2O2CO3 has great potential for application in the treatment of organic pollutants.
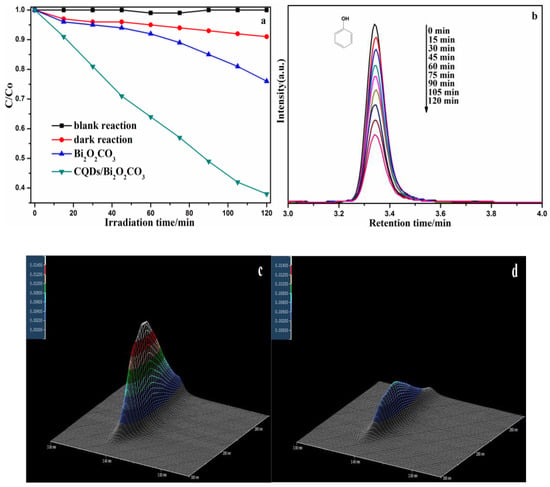
Figure 11.
(a) The photocatalytic activity comparison of phenol degradation; (b) HPLC chromatograms of phenol solutions with CQDs/Bi2O2CO3 photocatalyst; (c,d) 3D HPLC spectra of phenol degradation at 0 min and at 120 min.
According to the characterization and experimental results above, it was found that the CQDs/Bi2O2CO3 exhibited an efficient photocatalytic degradation ability for organic contaminants. The obviously enhanced photocatalytic property of CQDs/Bi2O2CO3 may be attributed to its unique structure. The flower-like Bi2O2CO3 functions as a good stabilizer and substrate for CQDs, and the up-conversion effect of CQDs could improve the electron transfer and light absorption ability of photocatalysts and improve ·O2− formation. The degradation schematic of CQDs/Bi2O2CO3 is presented in Figure 12. According to the literature and the experimental tests, the energy gap (Eg) of Bi2O2CO3 is about 3.4 eV, and its valence band (VB) and conduction band (CB) potentials are 3.56 and 0.15 eV, respectively. Therefore, there is no obvious photocatalytic activity under visible light irradiation for Bi2O2CO3. However, CQDs, which possess the up-conversion effect and function as an excellent electron mediator and acceptor in CQDs/Bi2O2CO3, modify the surface of Bi2O2CO3, and enhance the visible light response of the photocatalyst due to the electronic coupling between conduction band states of Bi2O2CO3 and π states of CQDs [38]. As shown in Figure 12, the up-converted PL property of CQDs could take advantage of harnessing the full spectrum of sunlight. This, in turn, excites Bi2O2CO3 to form electron-hole pairs, thereby improving photocatalytic activity. Bi2O2CO3 could be excited to generate the electron-hole pairs by short wavelength ultraviolet light through up-converting the long wavelength visible light via CQDs on the surface of Bi2O2CO3. When CQDs/Bi2O2CO3 was irradiated by simulated sunlight, the electrons could be excited from the VB to the CB of Bi2O2CO3, leaving the holes on the VB of Bi2O2CO3. According to the PL spectra, CQDs facilitates the electron transfer from the CB of Bi2O2CO3 to oxygen across CQDs, retarding the recombination [59]. The electrons could then reduce the adsorbed O2 to ·O2−. Meanwhile the h+ on the VB of Bi2O2CO3 could react with the OH− and/or H2O to produce ·OH, or a portion of the holes could also oxidize the organic pollutant directly [60]. The generated reactive oxygen species played an important role in the photocatalytic degradation process. These effects would guarantee the high photocatalytic activities of the CQDs modified Bi2O2CO3 samples.
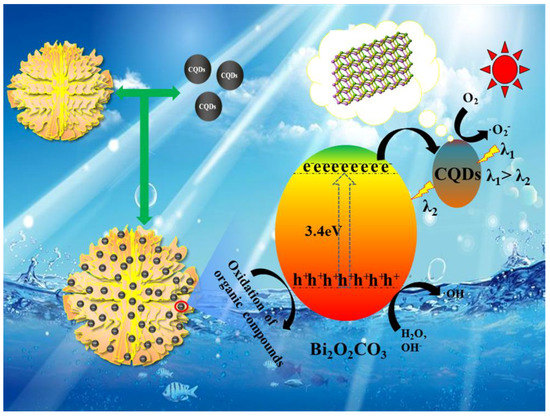
Figure 12.
Schematic of the possible reaction mechanism for organic pollutants degradation by CQDs/Bi2O2CO3 under the simulated sunlight irradiation.
4. Conclusions
In summary, CQDs modified Bi2O2CO3 (CQDs/Bi2O2CO3) was prepared by a simple dynamic adsorption precipitation method at room temperature. With CQDs approximately 2–5 nm in size evenly dispersed on the surface of Bi2O2CO3, CQDs/Bi2O2CO3 has efficient photocatalytic activity and long term stability under visible light. The photocatalytic degradation efficiency of methylene blue and phenol rate can reach up to 94.45% and 61.46% within 2 h, respectively. Photoelectrochemical testing proved that the introduction of CQDs (as electron acceptors) effectively suppresses the recombination of e−-h+ and promotes charge transfer. The quenching results suggested that ·O2−, h+ and ·OH were involved in the photocatalytic degradation process. These results demonstrated that the excellent photocatalytic performance for CQDs/Bi2O2CO3 can be attributed to the up-conversion effect of CQDs. The up-conversion effect of CQDs could improve ·O2− formation and the electron transfer and light absorption ability of photocatalysts. Furthermore, the up-conversion effect of CQDs could help maintain photocatalytic stability. The above results can provide important inspiration for the development of other CQDs-based photocatalytic materials.
Supplementary Materials
The following are available online at http://www.mdpi.com/2079-4991/8/5/330/s1, Figure S1: (a) UV-vis diffuse reflectance spectrum (DRS) and (b) the band gap energies (Eg) of Bi2O2CO3 and different CQDs/Bi2O2CO3 samples, Figure S2: (a) Valence band X-ray photoelectron spectroscopy (VB XPS) spectrum of Bi2O2CO3. (b) Mott-Schottky (MS) plots of Bi2O2CO3 photoelectrode, Figure S3: FTIR spectra of CQDs, Bi2O2CO3 and CQDs/Bi2O2CO3 samples, Figure S4: kinetic constant for the degradation of MB with the CQDs/Bi2O2CO3 composites prepared with different amounts of CQDs.
Author Contributions
S.L. and Z.Z. conceived and designed the experiments; S.L. and T.Z. performed the experiments; S.L. and H.L. analyzed the data; X.L. and W.C. contributed reagents/materials/analysis tools; S.L. wrote the paper.
Funding
This work was funded by the National Natural Science Foundation of China (grant No. 21476161), the Hebei Provincial Foundation for International Cooperation (No. 15391403D) and the Natural Sciences and Engineering Research Council of Canada (Discovery). Z.Z. and S.L. both contributed equally to this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Que, Q.H.; Xing, Y.L.; He, Z.L.; Yang, Y.W.; Yin, X.T.; Que, W.X. Bi2O3/Carbon quantum dots heterostructured photocatalysts with enhanced photocatalytic activity. Mater. Lett. 2017, 209, 220–223. [Google Scholar] [CrossRef]
- Kian, M.L.; Chin, W.L.; Koh, S.N.; Joon, C.J. Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 2016, 88, 428–448. [Google Scholar]
- Yogendra, K.M.; Rainer, A. ZnO tetrapod materials for functional applications. Mater. Today 2018. [Google Scholar] [CrossRef]
- Wang, H.L.; Zhang, L.S.; Chen, Z.G.; Hu, J.Q.; Li, S.J.; Wang, Z.H.; Liu, J.S.; Wang, X.C. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances. Chem. Soc. Rev. 2014, 43, 5234–5244. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.M.; Sun, J.; Xing, S.M.; Liu, D.J.; Zhang, G.J.; Bai, L.J.; Jiang, B.L. Enhanced Raman scattering and photocatalytic activity of TiO2 films with embedded Ag nanoparticles deposited by magnetron sputtering. J. Alloys Compd. 2016, 679, 88–93. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, C.Y.; Ma, Z.B.; Ren, Z.F.; Fan, H.J.; Yang, X.M. Elementary photocatalytic chemistry on TiO2 surfaces. Chem. Soc. Rev. 2016, 45, 3701–3730. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.X.; Han, S.C.; Liu, H.; Yu, P.P.; Fang, X.S. Hierarchical MoS2 nanosheet@TiO2 nanotube array composites with enhanced photocatalytic and photocurrent performances. Small 2016, 12, 1527–1536. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Han, R.R.; Ji, H.M.; Sun, T.; Zhao, J.; Chen, N.N.; Chen, J.; Guo, X.F.; Hou, W.H.; Ding, W.P. S-doped mesoporous nanocomposite of HTiNbO5 nanosheets and TiO2 nanoparticles with enhanced visible light photocatalytic activity. Phys. Chem. Chem. Phys. 2016, 18, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.P.; Xu, L.J.; Liu, C.L.; Wang, Y. Magnetic composite ZnFe2O4/SrFe12O19: Preparation, characterization, and photocatalytic activity under visible light. Appl. Surf. Sci. 2013, 273, 684–691. [Google Scholar] [CrossRef]
- García-Pérez, U.M.; Sepúlveda-Guzmán, S.; Martínezde la Cruz, A. Nanostructured BiVO4 photocatalysts synthesized via a polymer-assisted coprecipitation method and their photocatalytic properties under visible-light irradiation. Solid State Sci. 2012, 14, 293–298. [Google Scholar] [CrossRef]
- Zhu, Z.F.; Zhang, L.; Li, J.Q.; Du, J.; Zhang, Y.B.; Zhou, J.Q. Synthesis and photocatalytic behavior of BiVO4 with decahedral structure. Ceram. Int. 2013, 39, 7461–7465. [Google Scholar] [CrossRef]
- Dong, S.Y.; Feng, J.L.; Li, Y.K.; Hu, L.M.; Liu, M.L.; Wang, Y.F.; Pi, Y.Q.; Sun, J.Y.; Sun, J.H. Shape-controlled synthesis of BiVO4 hierarchical structures with unique natural-sunlight-driven photocatalytic activity. Appl. Catal. B 2014, 152–153, 413–424. [Google Scholar] [CrossRef]
- Zhuo, Y.Q.; Huang, J.F.; Cao, L.Y.; Ouyang, H.B.; Wu, J.P. Photocatalytic activity of snow-like Bi2WO6 microcrystalline for decomposition of Rhodamine B under natural sunlight irradiation. Mater. Lett. 2013, 90, 107–110. [Google Scholar] [CrossRef]
- Dumrongrojthanath, P.; Thongtem, T.; Phuruangrat, A.; Thongtem, S. Hydrothermal synthesis of Bi2WO6 hierarchical flowers with their photonic and photocatalytic properties. Superlattices Microstruct. 2013, 54, 71–77. [Google Scholar] [CrossRef]
- Zhang, L.S.; Wang, H.L.; Chen, Z.G.; Wong, P.K.; Liu, J.S. Bi2WO6 micro/nanostructures: Synthesis modifications and visible-light-driven photocatalytic applications. Appl. Catal. B 2011, 106, 1–13. [Google Scholar]
- Hu, J.L.; Fan, W.J.; Ye, W.Q.; Huang, C.J.; Qiu, X.Q. Insights into the photosensitivity activity of BiOCl under visible light irradiation. Appl. Catal. B 2014, 158–159, 182–189. [Google Scholar] [CrossRef]
- Chai, B.; Zhou, H.; Zhang, F.; Liao, X.; Ren, M.X. Visible light photocatalytic performance of hierarchical BiOBr microspheres synthesized via a reactable ionic liquid. Mater. Sci. Semicond. Process. 2014, 23, 151–158. [Google Scholar] [CrossRef]
- Li, T.T.; Luo, S.L.; Yang, L.X. Three-dimensional hierarchical Ag/AgI/BiOI microspheres with high visible-light photocatalytic activity. Mater. Lett. 2013, 109, 247–252. [Google Scholar] [CrossRef]
- Zhang, Q.Y.; Tian, H.; Li, N.; Chen, M.D.; Teng, F. Controllable growth of novel BiPO4 dendrites by an innovative approach and high energy facets-dependent photocatalytic activity. CrystEngComm 2014, 16, 8334–8339. [Google Scholar] [CrossRef]
- Lin, X.; Liu, D.; Guo, X.Y.; Sun, N.; Zhao, S.; Chang, L.M.; Zhai, H.J.; Wang, Q.W. Fabrication and efficient visible light-induced photocatalytic activity of Bi2MoO6/BiPO4 composite. J Phys. Chem. Solids 2015, 76, 170–176. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, W.; Zhang, Q.; Cao, J.J.; Huang, R.J.; Ho, W.K.; Lee, S.C. In situ fabrication of a-Bi2O3/BiO,2CO3 nanoplate heterojunctions with tunable optical property and photocatalytic activity. Sci. Rep. 2016, 6, 23435. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Duan, F.; Chen, M.Q.; Xie, Y. Synthetic Bi2O2CO3 nanostructures: Novel photocatalyst with controlled special surface exposed. J. Mol. Catal. A Chem. 2010, 317, 34–40. [Google Scholar] [CrossRef]
- Yang, L.L.; Han, Q.F.; Zhu, J.W.; Wang, X. Synthesis of egg-tart shaped Bi2O2CO3 hierarchical nanostructures from single precursor and its photocatalytic performance. Mater. Lett. 2015, 138, 235–237. [Google Scholar] [CrossRef]
- Cai, G.Y.; Xu, L.L.; Wei, B.; Che, J.X.; Gao, H.; Sun, W.J. Facile synthesis of β-Bi2O3/Bi2O2CO3 nanocomposite with high visible-light photocatalytic activity. Mater. Lett. 2014, 120, 1–4. [Google Scholar] [CrossRef]
- Chen, L.; Yin, S.F.; Luo, S.L.; Huang, R.; Zhang, Q.; Hong, T.; Au, P.C.T. Bi2O2CO3/BiOI Photocatalysts with Heterojunctions Highly Efficient for Visible-Light Treatment of Dye-Containing Wastewater. Ind. Eng. Chem. Res. 2012, 51, 6760–6768. [Google Scholar] [CrossRef]
- Jin, L.; Zhu, G.Q.; Hojamberdiev, M.; Luo, X.C.; Tan, C.W.; Peng, J.H.; Wei, X.M.; Li, J.P.; Liu, P. A plasmonic AgAgBr/Bi2O2CO3 composite photocatalyst with enhanced visible-light photocatalytic activity. Ind. Eng. Chem. Res. 2014, 53, 13718–13727. [Google Scholar] [CrossRef]
- Wang, W.J.; Cheng, H.F.; Huang, B.B.; Lin, X.J.; Qin, X.Y.; Zhang, X.Y.; Dai, Y. Synthesis of Bi2O2CO3/Bi2S3 hierarchical microspheres with heterojunctions and their enhanced visible light-driven photocatalytic degradation of dye pollutants. J Colloid. Interface Sci. 2013, 402, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.N.; Baker, G.A. Luminescent Carbon Nanodots: Emergent Nanolights. Angew. Chem. Int. Ed. 2010, 49, 6726–6744. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.H.; Zhu, Y.H.; Yang, X.L.; Li, C.Z. Graphene quantum dots: Emergent nanolights for bioimaging, sensors, catalysis and photovoltaic devices. Chem. Commun. 2012, 48, 3686–3699. [Google Scholar] [CrossRef] [PubMed]
- Li, H.T.; Kang, Z.H.; Liu, Y.; Lee, S.T. Carbon nanodots: Synthesis, properties and applications. J. Mater. Chem. 2012, 22, 24230–24253. [Google Scholar] [CrossRef]
- Tang, D.; Zhang, H.; Huang, H.; Liu, R.; Han, Y.; Liu, Y.; Tong, C.; Kang, Z.H. Carbon quantum dots enhance the photocatalytic performance of BiVO4 with different exposed facets. Dalton Trans. 2013, 42, 6285–6289. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; He, X.; Kang, Z.; Huang, H.; Liu, Y.; Liu, J.; Lian, S.; Tsang, C.; Yang, X.; Lee, S.T. Water-Soluble Fluorescent Carbon Quantum Dots and Photocatalyst Design. Angew. Chem. Int. Ed. 2010, 49, 4430–4434. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Chaudhary, N.; Srivastava, R.; Sharma, G.D.; Bhardwaj, R.; Chand, S. Luminscent Graphene Quantum Dots for Organic Photovoltaic Devices. J. Am. Chem. Soc. 2011, 133, 9960–9963. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Meng, Q.; Wang, L.; Zhang, J.; Song, Y.; Jin, H.; Zhang, K.; Sun, H.; Wang, H.; Yang, B. Highly Photoluminescent Carbon Dots for Multicolor Patterning, Sensors, and Bioimaging. Angew. Chem. Int. Ed. 2013, 52, 3953–3957. [Google Scholar] [CrossRef] [PubMed]
- Li, H.T.; Liu, R.H.; Lian, S.Y.; Liu, Y.; Huang, H.; Kang, Z.H. Near-infrared light controlled photocatalytic activity of carbon quantum dots for highly selective oxidation reaction. Nanoscale 2013, 5, 3289–3297. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.C.; Ming, H.; Lian, S.Y.; Huang, H.; Li, H.T.; Zhang, L.L.; Liu, Y.; Kang, Z.H.; Lee, S.T. Fe2O3/carbon quantum dots complex photocatalysts and their enhanced photocatalytic activity under visible light. Dalton Trans. 2011, 40, 10822–10825. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Zhang, H.C.; Huang, H.; Liu, Y.; Li, H.T.; Ming, H.; Kang, Z.H. ZnO/carbon quantum dots nanocomposites: One-step fabrication and superior photocatalytic ability for toxic gas degradation under visible light at room temperature. New J. Chem. 2012, 36, 1031–1035. [Google Scholar] [CrossRef]
- Ming, H.; Ma, Z.; Liu, Y.; Pan, K.M.; Yu, H.; Wang, F.; Kang, Z.H. Large scale electrochemical synthesis of high quality carbon nanodots and their photocatalytic property. Dalton Trans. 2012, 41, 9526–9531. [Google Scholar] [CrossRef] [PubMed]
- Tian, N.; Huang, H.W.; Guo, Y.X.; He, Y.; Zhang, Y.H. A g-C3N4/Bi2O2CO3 composite with high visible-light-driven photocatalytic activity for rhodamine B degradation. Appl. Surf. Sci. 2014, 322, 249–254. [Google Scholar] [CrossRef]
- Huang, H.W.; Wang, S.B.; Tian, N.; Hang, Y.H. One-step hydrothermal preparation strategy for layered BiIO4/Bi2WO6 heterojunctions with enhanced visible light photocatalytic activities. RSC Adv. 2014, 4, 5561–5567. [Google Scholar] [CrossRef]
- Huang, H.W.; Li, X.W.; Wang, J.J.; Dong, F.; Chu, P.K.; Zhang, T.R.; Zhang, Y.H. Anionic Group Self-Doping as a Promising Strategy: Band-Gap Engineering and Multi-Functional Applications of High-Performance CO32− -Doped Bi2O2CO3. ACS Catal. 2015, 5, 4094–4103. [Google Scholar] [CrossRef]
- Huang, H.W.; Yao, J.Y.; Lin, Z.S.; Wang, X.Y.; He, R.; Yao, W.J.; Zhai, N.X.; Chen, C.T. NaSr3Be3B3O9F4: A promising deep-ultraviolet nonlinear optical material resulting from the cooperative alignment of the [Be3B3O12F]10− anionic group. Angew. Chem. Int. Ed. 2011, 50, 9141–9144. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liang, Y.H.; Liu, L.; Hu, J.S.; Cui, W.Q. Reduced graphene oxide wrapped Bi2WO6 hybrid with ultrafast charge separation and improved photoelectrocatalytic performance. Appl. Surf. Sci. 2017, 392, 51–60. [Google Scholar] [CrossRef]
- Yu, J.C.; Yu, J.G.; Ho, W.K.; Jiang, Z.T.; Zhang, L.Z. Effects of F-doping on the photocatalytic activity and microstructures of nanocrystalline TiO2 powders. Chem. Mater. 2002, 14, 3808–3816. [Google Scholar] [CrossRef]
- Tobon-Zapata, G.E.; Etcheverry, S.B.; Baran, E.J. Vibrational spectrum of bismuth subcarbonate. J. Mater. Sci. Lett. 1997, 16, 656–657. [Google Scholar] [CrossRef]
- Li, J.Z.; Wang, N.Y.; Tran, T.T.; Huang, C.A.; Chen, L.; Yuan, L.J.; Zhou, L.P.; Shen, R.; Cai, Q.Y. Electrogenerated chemiluminescence detection of trace level pentachlorophenol using carbon quantum dots. Analyst 2013, 138, 2038–2043. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Sharma, S.; Kansal, S.K. Synthesis of ZnS/CQDs nanocomposite and its application as a photocatalyst for the degradation of an anionic dye. Superlattice Microstruct. 2016, 98, 86–95. [Google Scholar] [CrossRef]
- Zhang, P.; Shao, C.L.; Zhang, M.Y.; Guo, Z.C.; Mu, J.B.; Zhang, Z.Y.; Zhang, X.; Liu, Y.C. Bi2MoO6 ultrathin nanosheets on ZnTiO3 nanofibers: A 3D open hierarchical heterostructures synergistic system with enhanced visible-light-driven photocatalytic activity. J. Hazard. Mater. 2012, 217–218, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.F.; Yue, D.T.; Tian, Z.Y.; Reng, M.; Zhu, Y.; Kan, M.; Zhang, T.Y.; Zhao, Y.X. Carbon quantum dots decorated Bi2WO6 nanocomposite with enhanced photocatalytic oxidation activity for VOCs. Appl. Catal. B 2016, 193, 16–21. [Google Scholar] [CrossRef]
- Yu, B.Y.; Kwak, S.Y. Carbon quantum dots embedded with mesoporous hematite nanospheres as efficient visible light-active photocatalysts. J. Mater. Chem. 2012, 22, 8345–8353. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, S.; Zhao, Z.Y.; Dong, F.; Dong, X.A.; Zhou, Y. N-Doped Bi2O2CO3/Graphene Quantum Dot Composite Photocatalyst: Enhanced Visible-Light Photocatalytic NO Oxidation and In Situ DRIFTS Studies. J. Phys. Chem. C 2017, 121, 12168–12177. [Google Scholar] [CrossRef]
- Yu, X.J.; Liu, J.J.; Yu, Y.C.; Zuo, S.L.; Li, B.S. Preparation and visible light photocatalytic activity of carbon quantum dots/TiO2 nanosheet composites. Carbon 2014, 68, 718–724. [Google Scholar] [CrossRef]
- Donat, F.; Corbel, S.; Alem, H.; Pontvianne, S.; Balan, L.; Medjahdi, G.; Schneider, R. ZnO nanoparticles sensitized by CuInZnxS2+x quantum dots as highly efficient solar light driven photocatalysts. Beilstein J. Nanotechnol. 2017, 8, 1080–1093. [Google Scholar] [CrossRef] [PubMed]
- Candal, R.J.; Zeltner, W.A.; Anderson, M.A. Effects of pH and Applied Potential on Photocurrent and Oxidation Rate of Saline Solutions of Formic Acid in a Photoelectrocatalytic Reactor. Environ. Sci. Technol. 2000, 34, 3443–3451. [Google Scholar] [CrossRef]
- Duo, F.F.; Wang, Y.W.; Fan, C.M.; Zhang, X.C.; Wang, Y.F. Enhanced visible light photocatalytic activity and stability of CQDs/BiOBr composites: The upconversion effect of CQDs. J Alloys Compd. 2016, 685, 34–41. [Google Scholar] [CrossRef]
- Hasan, M.R.; Hamid, S.B.A.; Basirun, W.J.; Chowdhury, Z.Z.; Kandjani, A.E.; Bhargava, S.K. Ga doped RGO–TiO2 composite on an ITO surface electrode for investigation of photoelectrocatalytic activity under visible light irradiation. New J. Chem. 2015, 39, 369–376. [Google Scholar] [CrossRef]
- Liang, Y.H.; Lin, S.L.; Liu, L.; Hu, J.S.; Cui, W.Q. Oil-in-water self-assembled Ag@AgCl QDs sensitized Bi2WO6: Enhanced photocatalytic degradation under visible light irradiation. Appl. Catal. B 2015, 164, 192–203. [Google Scholar] [CrossRef]
- Cong, Y.Q.; Ji, Y.; Ge, Y.H.; Jin, H.; Zhang, Y.; Wang, Q. Fabrication of 3D Bi2O3-BiOI heterojunction by a simple dipping method: Highly enhanced visible-light photoelectrocatalytic activity. Chem. Eng. J. 2017, 307, 572–582. [Google Scholar] [CrossRef]
- Fang, S.; Xia, Y.; Lv, K.L.; Li, Q.; Sun, J.; Li, M. Effect of carbon-dots modification on the structure and photocatalyticactivity of g-C3N4. Appl. Catal. B 2016, 185, 225–232. [Google Scholar] [CrossRef]
- Hu, C.; Lan, Y.Q.; Qu, J.H.; Hu, X.X.; Wang, A.M. Ag/AgBr/TiO2 Visible Light Photocatalyst for Destruction of Azodyes and Bacteria. J. Phys. Chem. B 2006, 110, 4066–4072. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).