Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective
Abstract
1. Introduction
2. Overview of Graphene-TiO2 Composite Photocatalyst
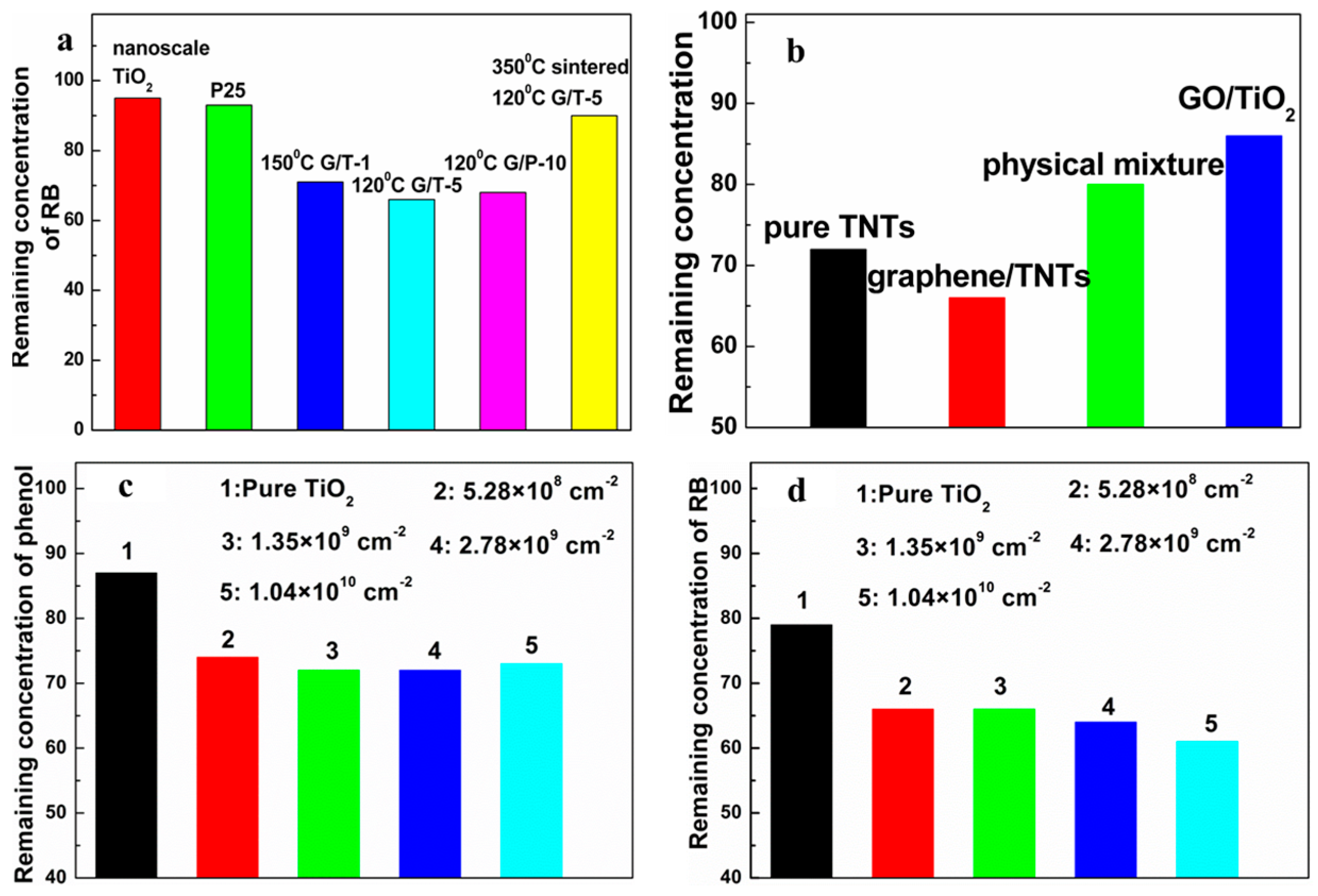
2.1. Photocatalytic Performances of Graphene-TiO2 Photocatalyst
2.2. Photocatalytic Mechanisms of the Graphene-TiO2 Photocatalyst
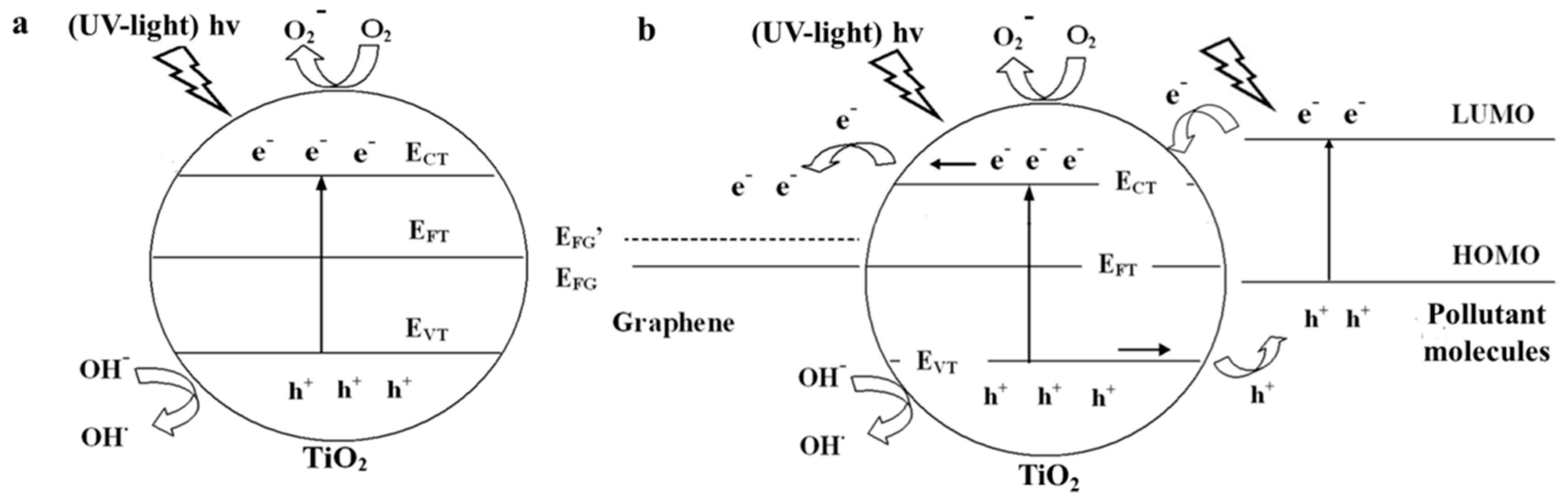
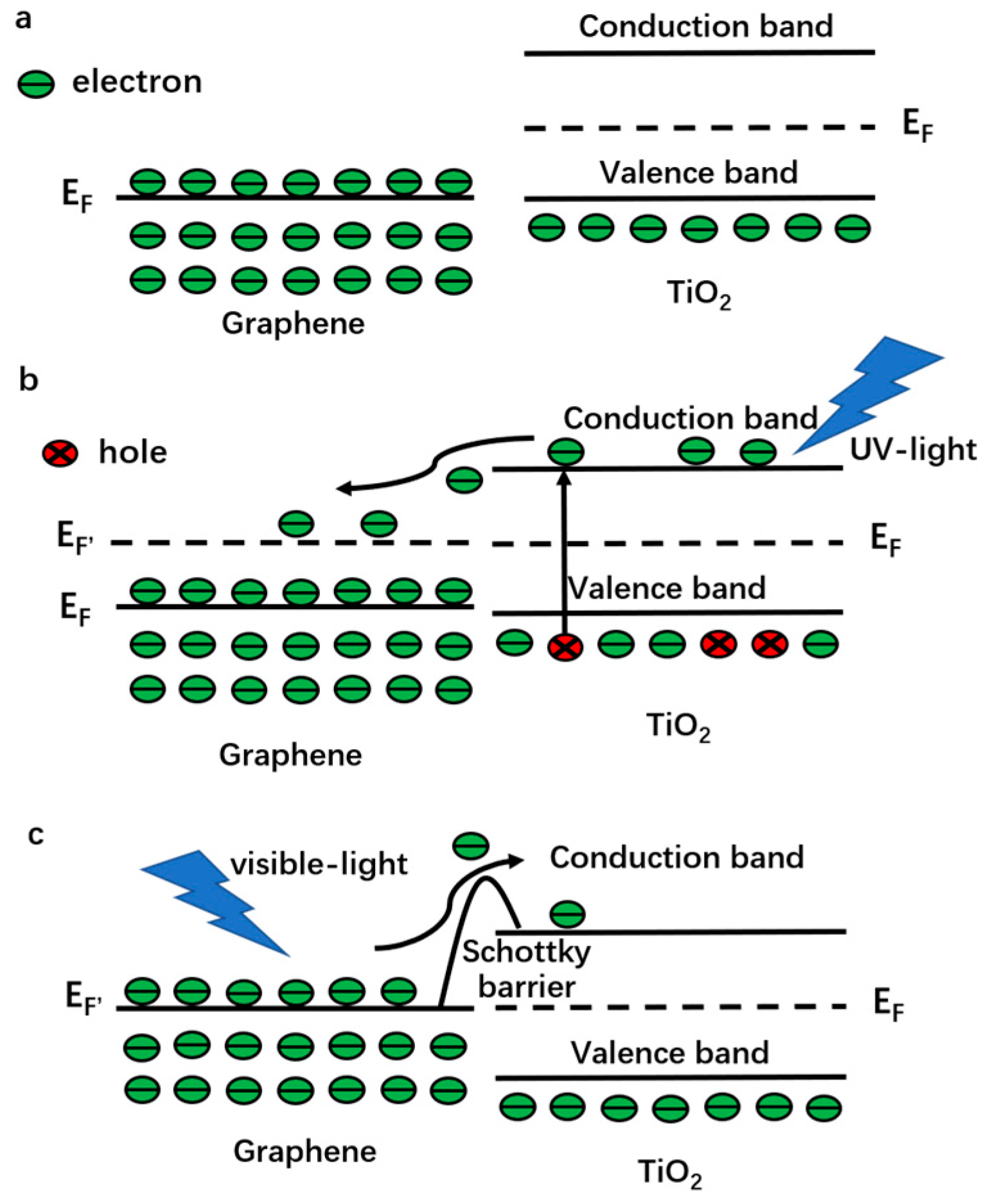
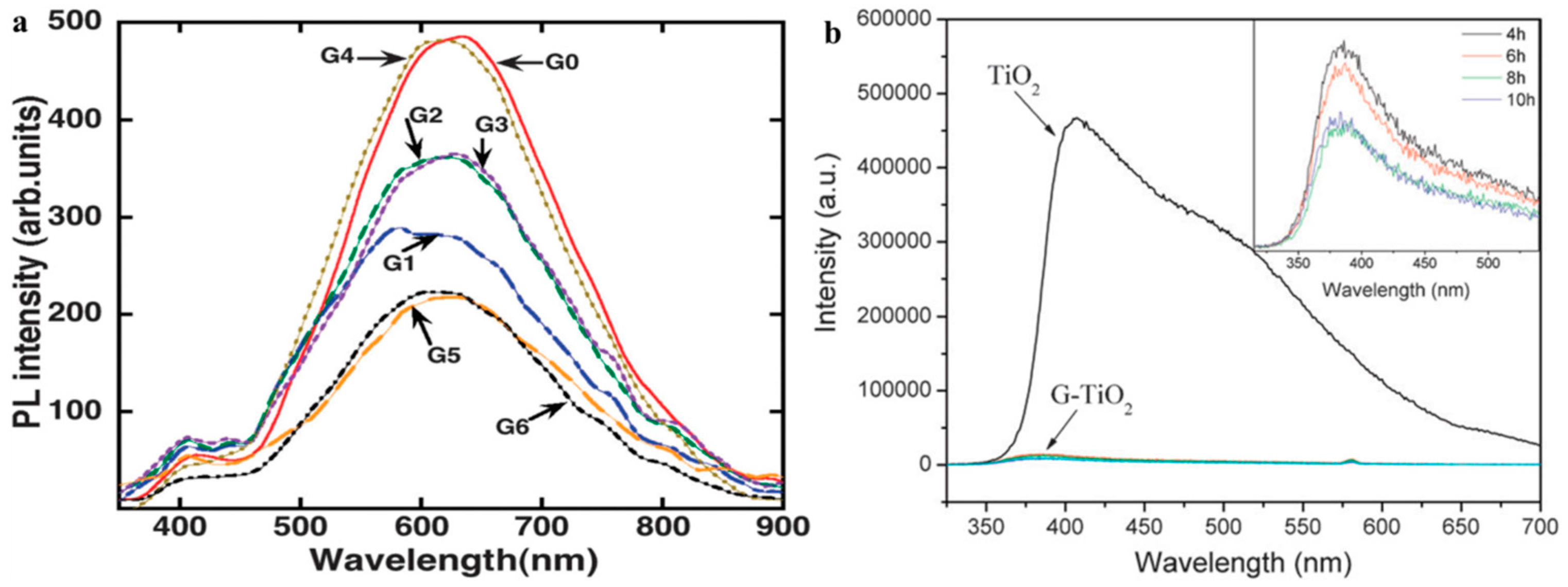
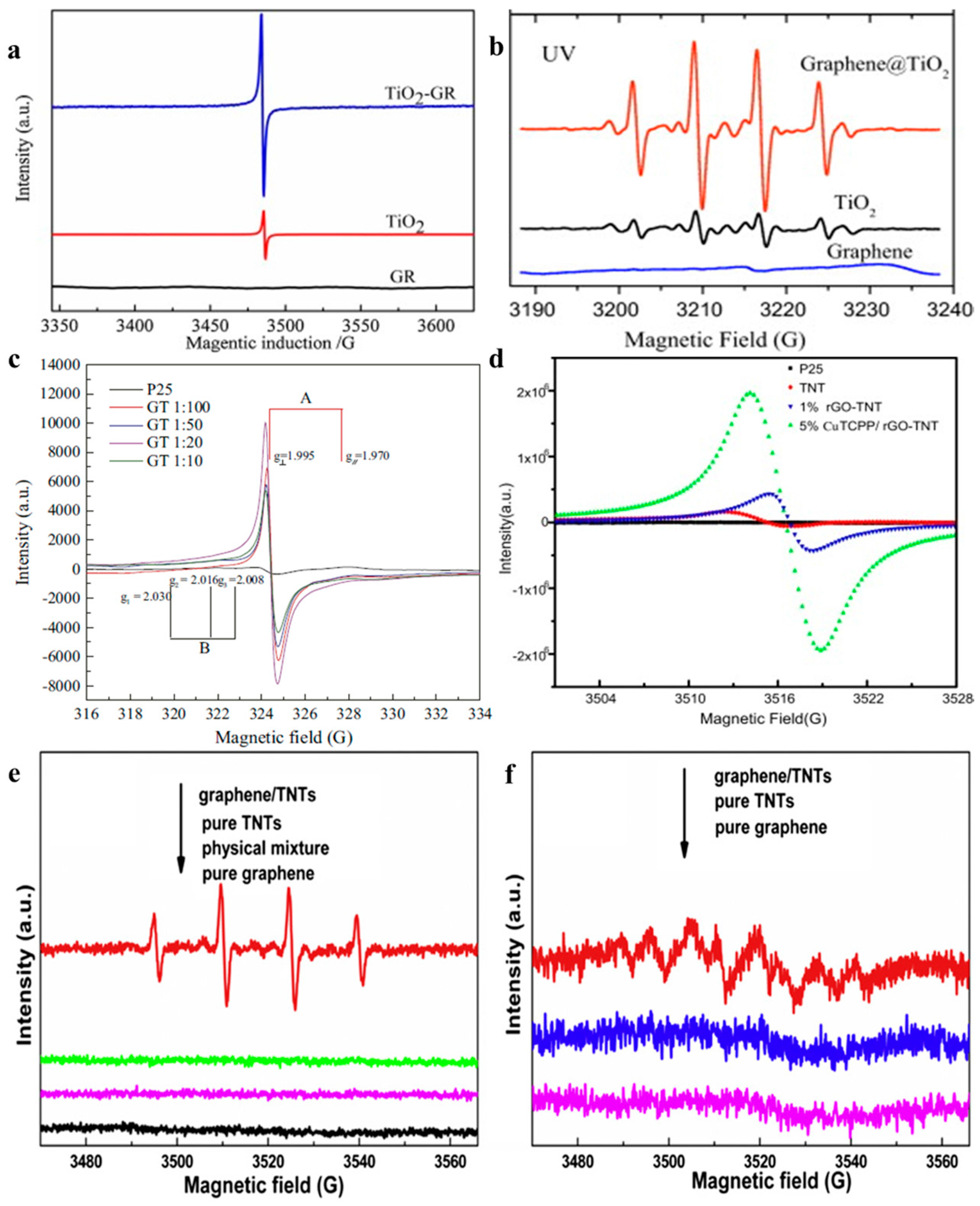
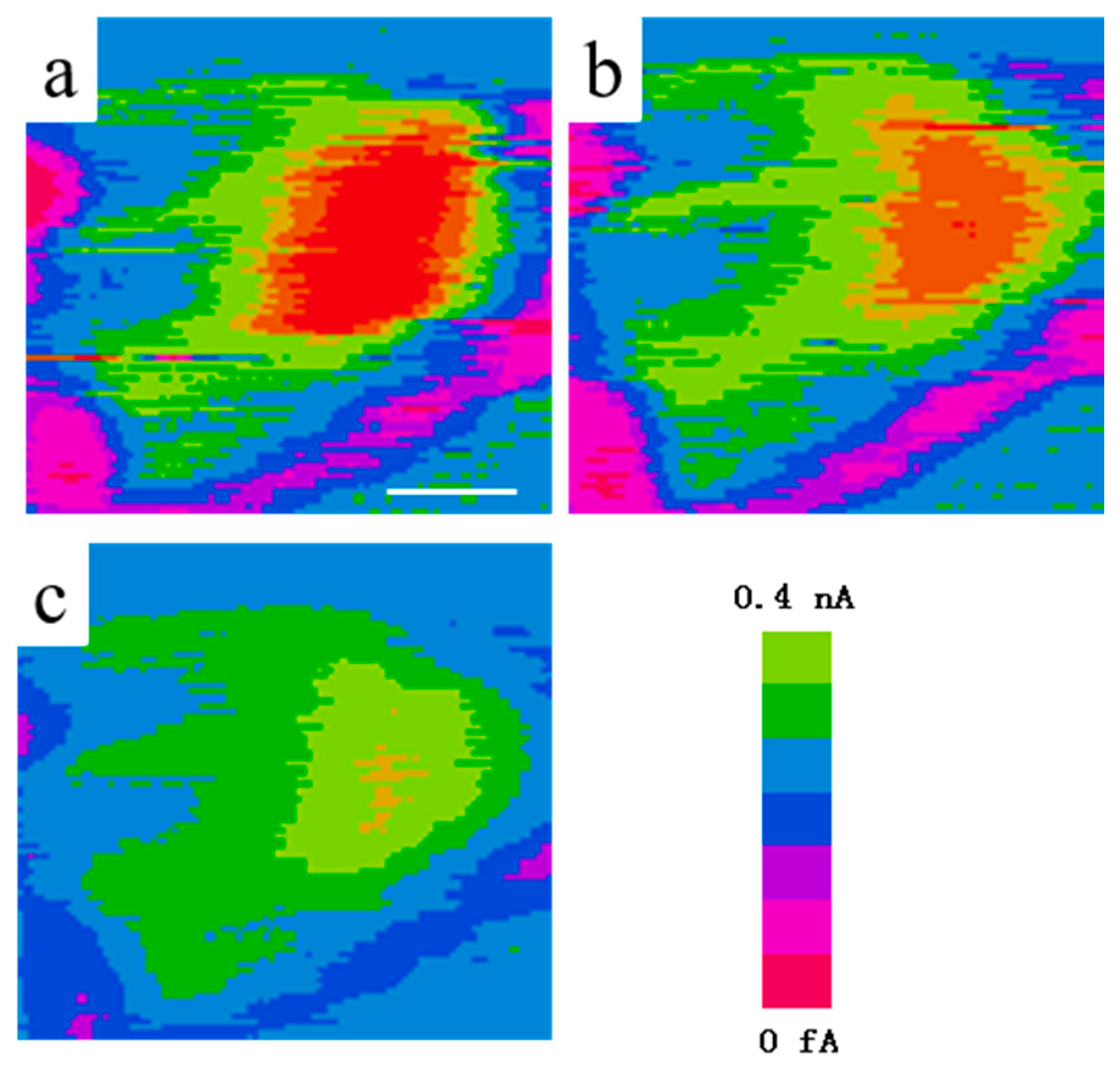
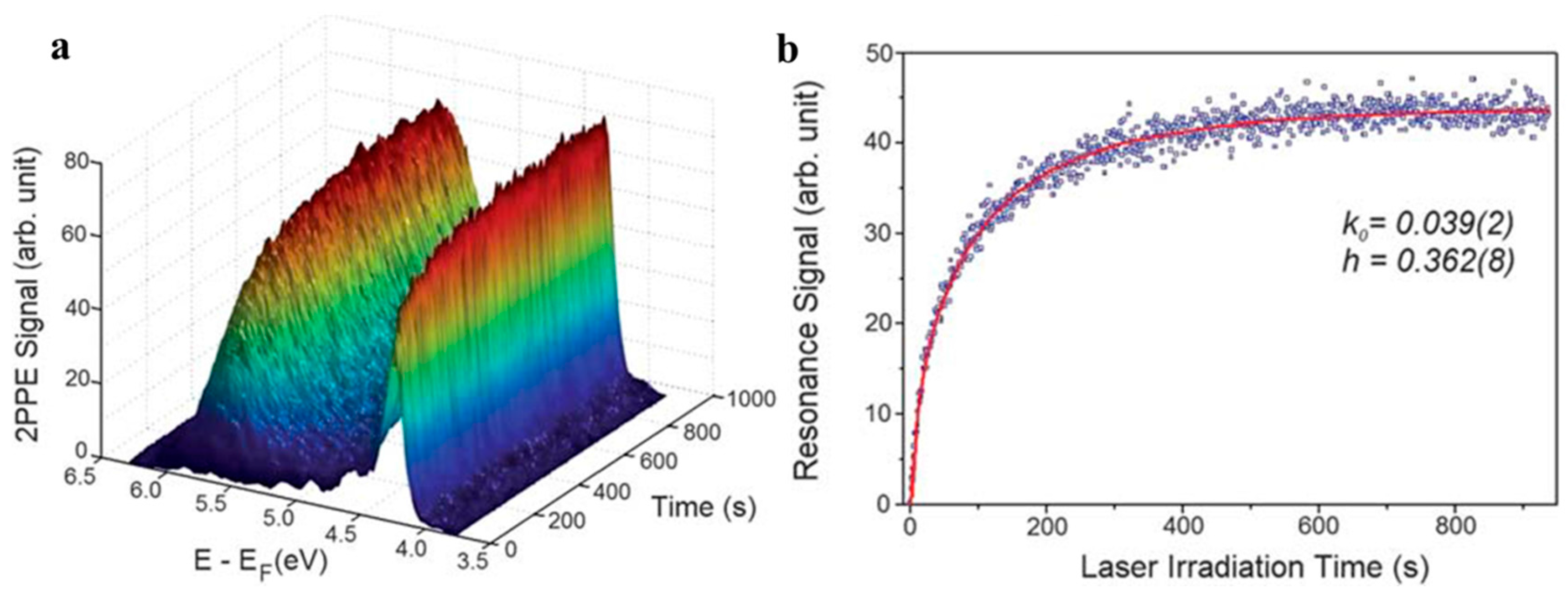
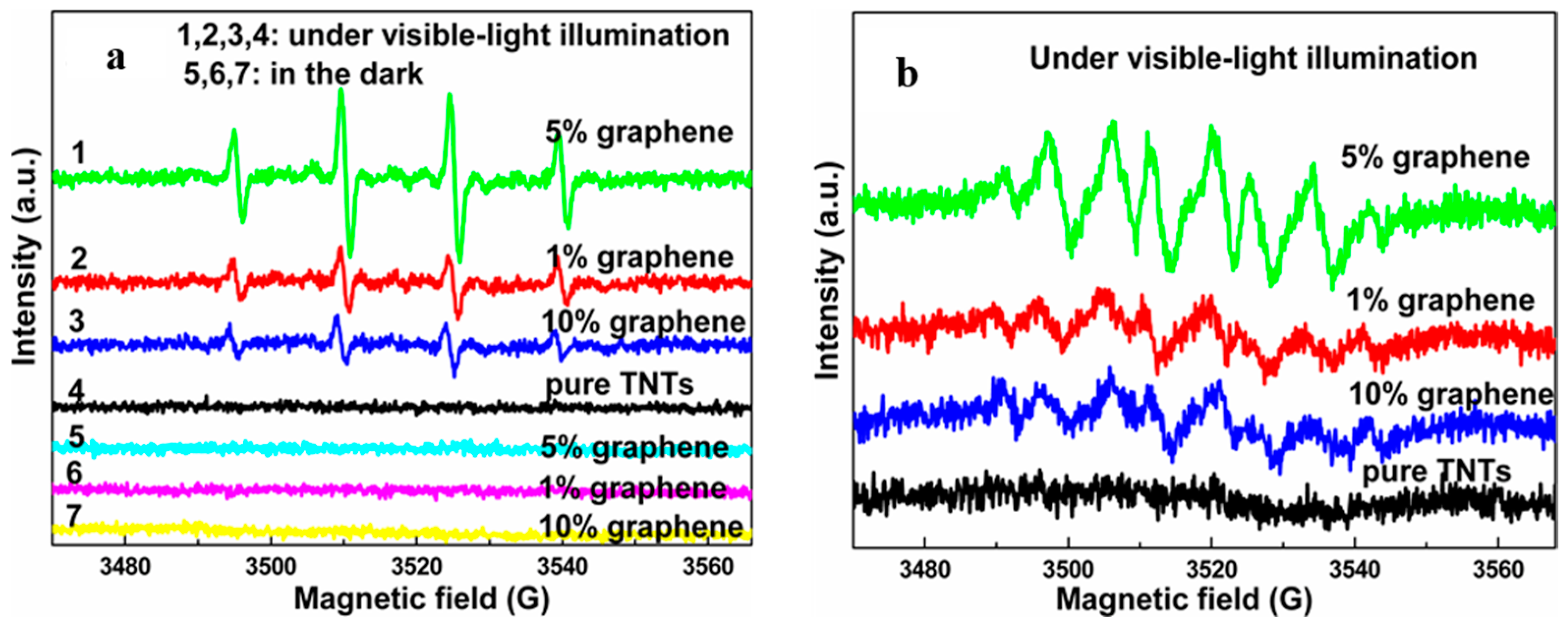
2.2.1. Under UV-Light Irradiation
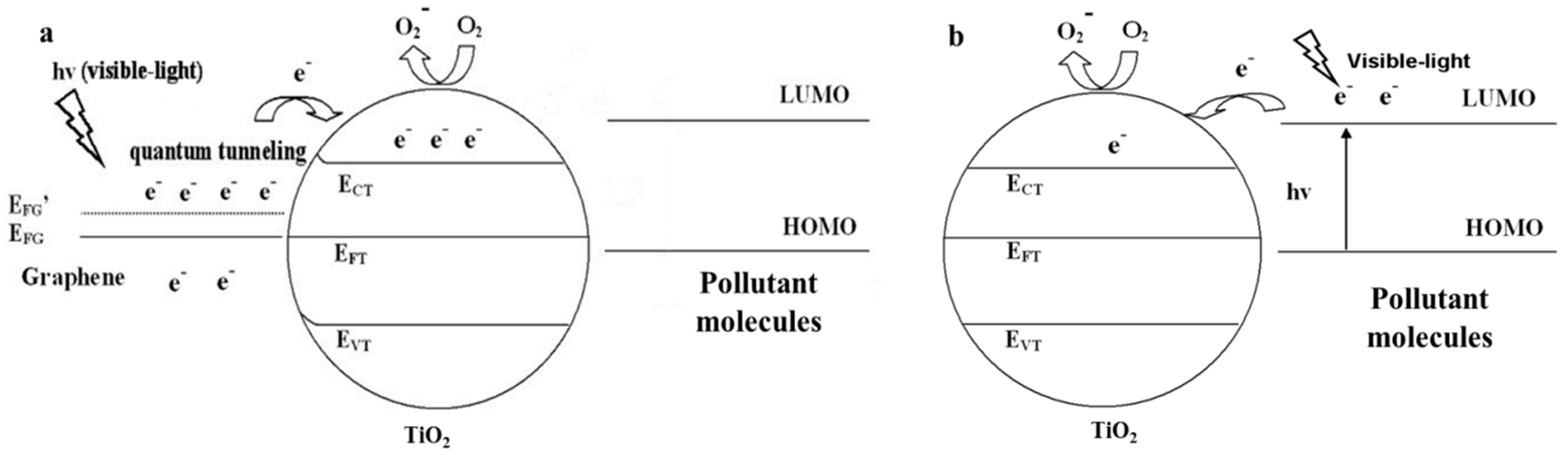
2.2.2. Visible light Irradiation
2.3. Characterization Approaches
3. Optimizing of the Graphene-TiO2 Composite Photocatalyst
3.1. Mass Fraction of Graphene
3.2. Morphologies of TiO2 and Graphene
3.3. Quality of Graphene
4. Prospective
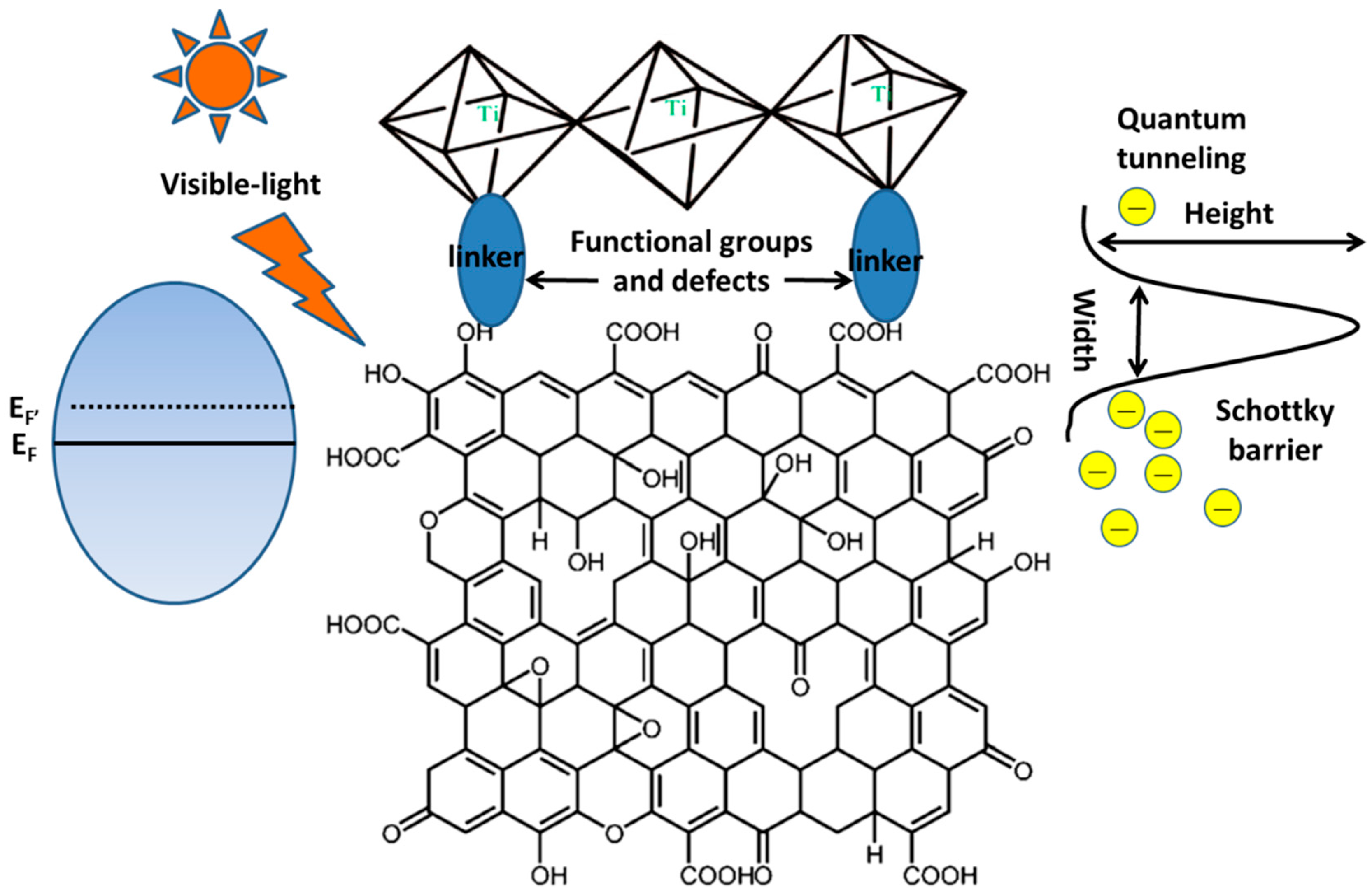
4.1. How to Enhance the Chemisorption Ability of the Graphene-TiO2 Photocatalysts?
4.2. How to Depress the Recombination of the Electron-Hole Pairs in TiO2 under UV-Light Irradiation?
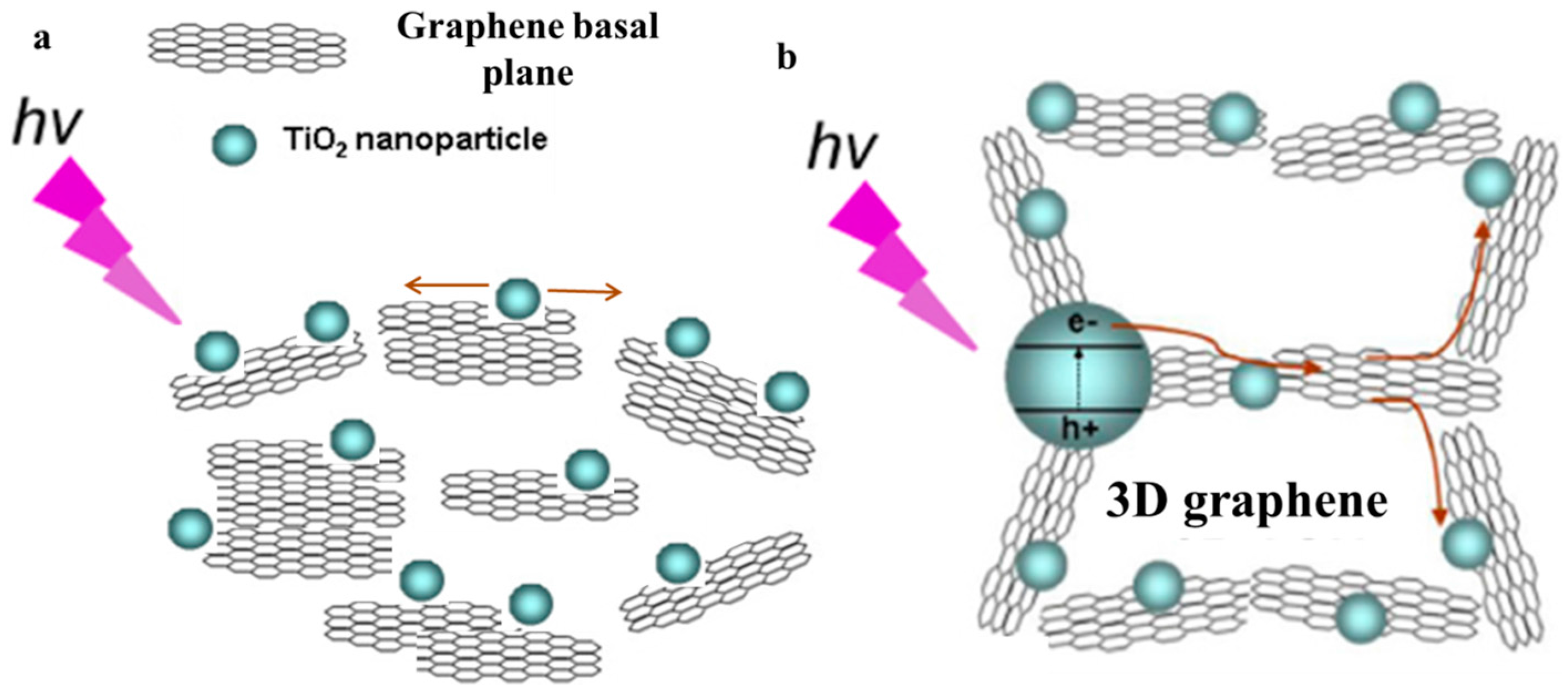
4.3. How to Promote Electron Transport at the Interface of the Graphene Basal Plane and TiO2 under Visible Light Irradiation?
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maeda, K.; Teramura, K.; Lu, D.; Takata, T.; Saito, N. Photocatalyst releasing hydrogen from water. Nature 2006, 440, 295. [Google Scholar] [CrossRef] [PubMed]
- Fujihira, M.; Satoh, Y.; Osa, T. Heterogeneous photocatalytic oxidation of aromatic compounds on TiO2. Nature 1981, 293, 206–208. [Google Scholar] [CrossRef]
- Tang, B.; Chen, H.Q.; He, Y.F.; Wang, Z.W. Influence from defects of three-dimensional graphene network on photocatalytic performance of composite photocatalyst. Compos. Sci. Technol. 2017, 150, 54–64. [Google Scholar] [CrossRef]
- Tang, B.; Wang, S.L.; Zhang, J.; Wang, Z.W.; He, Y.F.; Huang, W.Q. Three-dimensional graphene monolity-based composite: Superiority in properties and applications. Int. Mater. Rev. 2018, 63, 204–225. [Google Scholar] [CrossRef]
- Tian, C.G.; Zhang, Q.; Wu, A.P.; Jiang, M.J.; Liang, Z.L.; Jiang, B.J.; Fu, H.G. Cost-effective large-scale synthesis of ZnO photocatalyst with excellent performance for dye photodegradation. Chem. Commun. 2012, 48, 2858–2860. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.M.; Shahry, M.A.; Ingler, W.B. Efficient photochemical water splitting by a chemically modified n-TiO2. Science 2002, 297, 2243–2245. [Google Scholar] [CrossRef] [PubMed]
- Wahlstrom, E.; Vestergaard, E.K.; Schaub, R.; Rønnau, A.; Vestergaard, M.; Lægsgaard, E.; Stensgaard, I.; Besenbacher, F. Electron transfer-induced dynamics of oxygen molecules. Science 2004, 303, 511–513. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B.N.; Paul, L.F.; Cost, Í.A.; Machado, A.E.H.; Paterno, L.G.; Patrocinio, A.O.T. Layer-by-layer assembled photocatalysts for environmental remediation and solar energy conversion. J. Photochem. Photobiol. C 2017, 32, 1–20. [Google Scholar] [CrossRef]
- Kamat, P.V. Photochemistry on nonreactive and reactive (semiconductor) surfaces. Chem. Rev. 1993, 93, 267–300. [Google Scholar] [CrossRef]
- Etacheri, V.; Valentin, C.D.; Schneider, J.; Bahnemann, D.; Pillai, S.C. Visible-light activation of TiO2 photocatalysts: Advances in theory and experiments. J. Photochem. Photobiol. C 2015, 25, 1–29. [Google Scholar] [CrossRef]
- Drunka, R.; Grabis, J.; Jankovica, D.; Krumina, A.; Rasmane, D. Microwave-assisted synthesis and photocatalytic properties of sulphur and platinum modified TiO2 nanofibers. Mater. Sci. Eng. B 2015, 77, 012010–012014. [Google Scholar]
- Wang, S.B.; Pan, L.; Song, J.J. Titanium-defected undoped anatase TiO2 with p-type conductivity, room-temperature ferromagnetism, and remarkable photocatalytic performance. J. Am. Chem. Soc. 2015, 137, 2975–2983. [Google Scholar] [CrossRef] [PubMed]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.X.; Tang, B. Photocatalytic mechanism of graphene/titanate nanotubes photocatalyst under visible-light irradiation. Mater. Chem. Phys. 2013, 138, 608–614. [Google Scholar] [CrossRef]
- Zhai, Q.Q.; Tang, B.; Hu, G.X. High photoactive and visible-light responsive graphene/titanate nanotubes photocatalysts: Preparation and characterization. J. Hazard. Mater. 2011, 198, 78–86. [Google Scholar]
- Dette, C.; Pérez, M.A.; Kley, C.S.; Punke, P.; Patrick, C.E.; Jacobson, P.; Giustino, F.; Jung, S.J.; Kern, K. TiO2 anatase with a bandgap in the visible region. Nano Lett. 2014, 12, 6533–6538. [Google Scholar] [CrossRef] [PubMed]
- Khairy, M.; Zakaria, W. Effect of metal-doping of TiO2 nanoparticles on their photocatalytic activities toward removal of organic dyes. Egypt. J. Chem. 2014, 23, 419–426. [Google Scholar] [CrossRef]
- Davinder, S.S.; Bhachu, S.; Lu, Y.; Chadwick, N.; Althabaiti, S.A.; Alyoubi, A.O.; Basahel, S.N.; Carmalt, C.J.; Parkin, I.P. Tungsten doped TiO2 with enhanced photocatalytic and opt-electrical properties via aerosol assisted chemical vapor deposition. Sci. Rep. 2015, 4, 10952–10956. [Google Scholar]
- Sakthivel, S.; Kisch, H. Daylight photocatalysis by carbon-modified titanium dioxide. Angew. Chem. Int. Ed. 2003, 42, 4908–4911. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bahnemann, D.W.; Dohrmann, J.K. A novel preparation of iron-doped TiO2 nanoparticles with enhanced photocatalytic activity. Chem. Commun. 2000, 16, 1539–1540. [Google Scholar] [CrossRef]
- Livraghi, S.; Paganini, M.C.; Giamello, E.; Selloni, A.; Valentin, C.D.; Pacchioni, G. Origin of photoactivity of nitrogen-doped titanium dioxide under visible light. J. Am. Chem. Soc. 2006, 128, 15666–15671. [Google Scholar] [CrossRef] [PubMed]
- Valentin, C.D.; Pacchioni, G.; Selloni, A. Theory of carbon doping of titanium dioxide. Chem. Mater. 2005, 17, 6656–6665. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Chen, C.; Wu, F.Q.; Zou, B.; Zhao, M.; Wang, J.X.; Feng, C.H. Photodegradation of rhodamine B under visible light by bimetal co-doped TiO2 nanocrystals. J. Hazard. Mater. 2009, 164, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.P.; Lo, S.L.; Ou, H.H. Microwave-assisted hydrothermal synthesis of N doped titanate nanotubes for visible-light-responsive photocatalysis. J. Hazard. Mater. 2010, 183, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.L.; Zheng, G.; Wang, X.C.; He, K.H.; Chen, Q.L.; Yu, L.; Wang, Q.B. First-principles study on the structural and electronic properties of double N atoms doped-rutile TiO2. J. At. Mol. Sci. 2010, 1, 177–184. [Google Scholar] [CrossRef]
- Zhang, X.T.; Zhou, G.W.; Zhang, H.Y.; Wu, C.C.; Song, H.B. Characterization and activity of visible light–driven TiO2 photocatalysts co-doped with nitrogen and lanthanum. Trans. Metal Chem. 2011, 36, 217–222. [Google Scholar] [CrossRef]
- Subramanian, V.; Wolf, E.; Kamat, P.V. Catalysis with TiO2/gold nanocomposites, Effect of metal particle size on the fermi level equilibration. J. Am. Chem. Soc. 2004, 126, 4943–4950. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, E.; Remita, H.; Colbeau, J.C.; Hupka, J.; Belloni, J. Modification of titanium dioxide with platinum ions and clusters: Application in photocatalysis. J. Phys. Chem. C 2008, 112, 1124–1131. [Google Scholar] [CrossRef]
- Ansari, M.O.; Khan, M.M.; Ansaria, S.A.; Cho, M.H. Electrically conductive polyaniline sensitized defective-TiO2 for improved visible light photocatalytic and photoelectrochemical performance: A synergistic effect. New J. Chem. 2015, 39, 8381–8388. [Google Scholar] [CrossRef]
- Jing, L.; Yue, E.; Lian, L.S.; Ma, W.H. Visible light induced dye-sensitized photocatalytic hydrogen production over platinized TiO2 derived from decomposition of platinum complex precursor. Int. J. Hydrogen Energy 2013, 38, 10746–10753. [Google Scholar]
- Camarillo, R.; Tostón, S.; Martínez, F.; Jiménez, C.; Rincón, J. Enhancing the photocatalytic reduction of CO2 through engineering of catalysts with high pressure technology: Pd/TiO2 photocatalysts. J. Supercrit. Fluids 2017, 123, 18–27. [Google Scholar] [CrossRef]
- Tostón, S.; Camarillo, R.; Martínez, F.; Jiménez, C.; Rincón, J. Supercritical synthesis of platinum-modified titanium dioxide for solar fuel production from carbon dioxide. Chin. J. Catal. 2017, 38, 636–650. [Google Scholar] [CrossRef]
- Camarillo, R.; Tostón, S.; Martínez, F.; Jiménez, C.; Rincón, J. Preparation of TiO2-based catalysts with supercritical fluid technology: Characterization and photocatalytic activity in CO2 reduction. J. Chem. Technol. Biotechnol. 2017, 92, 1710–1720. [Google Scholar] [CrossRef]
- Camarillo, R.; Tostón, S.; Martínez, F.; Jiménez, C.; Rincón, J. Improving the photo-reduction of CO2 to fuels with catalysts synthesized under high pressure: Cu/TiO2. J. Chem. Technol. Biotechnol. 2017. [Google Scholar] [CrossRef]
- Chowdhury, P.; Moreira, J.; Gomaa, H.; Ray, A.K. Visible-solar-light-driven photocatalytic degradation of phenol with dye-sensitized TiO2: Parametric and kinetic study. Ind. Eng. Chem. Res. 2012, 51, 4523–4532. [Google Scholar] [CrossRef]
- Győri, Z.; Kóny, Z.; Kukovecz, Á. Visible light activation photocatalytic performance of PbSe quantum dot sensitized TiO2 nanowires. Appl. Catal. B Environ. 2015, 179, 583–588. [Google Scholar] [CrossRef]
- Sampaio, M.J.; Silva, C.G.; Marques, R.R.N.; Silva, A.M.T.; Faria, J.L. Carbon nanotube-TiO2 thin films for photocatalytic applications. Catal. Today 2011, 161, 91–96. [Google Scholar] [CrossRef]
- Yu, J.; Ma, T.; Liu, G.; Cheng, B. Enhanced photocatalytic activity of bimodal mesoporous titania powders by C60 modification. Dalton Trans. 2011, 40, 6635–6644. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, K. Physicochemical and photocatalytic activities of self-assembling TiO2 nanoparticles on nanocarbons surface. Curr. Appl. Phys. 2012, 12, 346–352. [Google Scholar] [CrossRef]
- Li, D.; Müller, M.B.; Gilje, S.; Kaner, R.B.; Wallace, G.G. Processable aqueous dispersions of graphene nanosheets. Nat. Nanotechnol. 2008, 3, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Niyogi, S.; Bekyarova, E.; Itkis, M.E.; McWilliams, J.L.; Hamon, M.A.; Haddon, R.C. Solution properties of graphite and graphene. J. Am. Chem. Soc. 2006, 128, 7720–7721. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.C.; Ngoc, M.P.; Nguyen, M.V. Enhanced photocatalytic activity of nanohybrids TiO2/CNTs materials. Mater. Lett. 2016, 165, 247–251. [Google Scholar] [CrossRef]
- Djokića, V.R.; Marinkovića, A.; Mitrićb, M.; Uskokovića, P.S.; Petrovića, R.D.; Radmilovićc, V.R.; Janaćković, D.T. Preparation of TiO2/carbon nanotubes photocatalysts: The influence of the method of oxidation of the carbon nanotubes on the photocatalytic activity of the nanocomposites. Ceram. Int. 2012, 38, 6123–6129. [Google Scholar] [CrossRef]
- Liu, J.; Xu, Z.B. Preparation and photocatalytic activity of Ag-TiO2/CNT photocatalyst. Adv. Mater. Res. 2012, 472, 157–160. [Google Scholar]
- Woan, K.; Pyrgiotakis, G.; Sigmund, W. Photocatalytic carbon-nanotube–TiO2 composites. Adv. Mater. 2009, 21, 2233–2239. [Google Scholar] [CrossRef]
- Vajda, K.; Mogyorosi, K.; Nemeth, Z.; Hernad, K.; Forro, L.; Magrez, A.; Domb, A. Photocatalytic activity of TiO2/SWCNT and TiO2/MWCNT nanocomposites with different carbon nanotube content. Phys. Status Solidi B 2011, 248, 2496–2499. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Hu, G.X.; Gao, H.Y. Raman spectroscopic characterization of graphene. Appl. Spectrosc. Rev. 2010, 45, 369–407. [Google Scholar] [CrossRef]
- Tang, B.; Hu, G.X.; Gao, H.Y. Application of graphene as filler to improve thermal transport property of epoxy resin for thermal interface materials. Int. J. Heat Mass Transf. 2015, 85, 420–429. [Google Scholar] [CrossRef]
- Tang, B.; Hu, G.X. Three-dimensional graphene network assisted high performance dye sensitized solar cells. J. Power Sources 2013, 234, 60–68. [Google Scholar] [CrossRef]
- Tang, B.; Hu, G.X. Two kinds of graphene-based composites for photoanode applying in dye-sensitized solar cell. J. Power Sources 2012, 220, 95–102. [Google Scholar] [CrossRef]
- Tang, B.; Hu, G.X. Preparation of few layers three-dimensional graphene networks by CVD for energy storage application. Chem. Vapor Depos. 2014, 20, 14–22. [Google Scholar] [CrossRef]
- Tang, B.; Wang, Z.W.; Huang, W.Q.; Li, S.; Ma, T.T.; Yu, H.G.; Li, X.F. RGO and Three-dimensional graphene networks co-modified TIMs with high performances. Nanoscale Res. Lett. 2017, 12, 527–533. [Google Scholar]
- Sun, Y.F.; Wang, X.B.; Tang, B.; Ban, J.M.; He, Y.F.; Huang, W.Q.; Tao, C.B.; Luo, H. Three-dimensional graphene networks modified photocatalyst with high performance under visible light irradiation. Mater. Lett. 2017, 189, 54–57. [Google Scholar] [CrossRef]
- Tang, B.; Hu, G.X. Growth mechanism and influences from kinetic factors on carbon materials with Cu and silica substrates during atmospheric pressure chemical vapor deposition. J. Phys. Chem. C 2013, 117, 25175–25184. [Google Scholar] [CrossRef]
- Neto, A.H.C.; Guinea, F.; Peres, N.M.R.; Novoselov, K.S.; Geim, A.K. The electronic properties of graphene. Rev. Mod. Phys. 2009, 81, 109–162. [Google Scholar] [CrossRef]
- Maka, K.F.; Sfeir, M.Y.; Misewich, J.A.; Heinz, T.F. The evolution of electronic structure in few-layer graphene revealed by optical spectroscopy. PNAS 2010, 107, 14999–15004. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.W.; Fu, H.B.; Zhu, Y.F. Efficient TiO2 photocatalysts from surface hybridization of TiO2 particles with graphite-like carbon. Adv. Funct. Mater. 2008, 18, 2180–2189. [Google Scholar] [CrossRef]
- Xu, Z.X.; Gao, H.Y.; Hu, G.X. Solution-based synthesis and characterization of a silver nanoparticle–graphene hybrid film. Carbon 2011, 49, 4731–4738. [Google Scholar] [CrossRef]
- Kamat, P.V. Graphene-based nanoassemblies for energy conversion. J. Phys. Chem. Lett. 2010, 2, 242–251. [Google Scholar] [CrossRef]
- Shen, J.F.; Yan, B.; Shi, M.; Ma, H.W.; Li, N.; Ye, M.X. One step hydrothermal synthesis of TiO2-reduced graphene oxide sheets. J. Mater. Chem. 2011, 21, 3415–3421. [Google Scholar] [CrossRef]
- Anjusree, G.S.; Nair, A.S.; Naira, S.V.; Vadukumpully, S. One-pot hydrothermal synthesis of TiO2/graphene nanocomposites for enhanced visible light photocatalysis and photovoltaics. RSC Adv. 2013, 3, 12933–12938. [Google Scholar] [CrossRef]
- Shi, M.; Shen, J.F.; Ma, H.W.; Li, Z.Q.; Lu, X.; Li, N.; Ye, M.X. Preparation of graphene–TiO2 composite by hydrothermal method from peroxotitanium acid and its photocatalytic properties. Colloid Surf. A Physicochem. Eng. Asp. 2012, 405, 30–37. [Google Scholar] [CrossRef]
- Liang, D.Y.; Cui, C.; Hu, H.H.; Wang, Y.P.; Xu, S.; Ying, B.L.; Li, P.G.; Lu, B.Q.; Shen, H.L. One-step hydrothermal synthesis of anatase TiO2/reduced graphene oxide nanocomposites with enhanced photocatalytic activity. J. Alloys Compd. 2014, 582, 236–240. [Google Scholar] [CrossRef]
- Bai, X.J.; Wang, L.; Zong, R.L.; Lv, Y.L.; Sun, Y.Q.; Zhu, Y.F. Performance enhancement of ZnO photocatalyst via synergic effect of surface oxygen defect and graphene hybridization. Langmuir 2013, 29, 3097–3105. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Moon, G.; Satoca, D.M.; Park, Y.; Choi, W.S. Photoconversion using graphene/TiO2 composites: Nanographene shell on TiO2 core versus TiO2 nanoparticles on graphene sheet. J. Phys. Chem. C 2012, 116, 1535–1543. [Google Scholar] [CrossRef]
- Liu, H.H.; Zhu, D.B.; Shi, H.; Shao, X. Fabrication of a contamination-free interface between graphene and TiO2 single crystals. ACS Omega 2016, 1, 168–176. [Google Scholar] [CrossRef]
- Li, J.H.; Wang, G.; Geng, H.; Zhu, H.Q.; Zhang, M.; Di, Z.F.; Liu, X.Y.; Chu, P.K.; Wang, X. CVD growth of graphene on NiTi alloy for enhanced biological activity. ACS Appl. Mater. Interfaces 2015, 7, 19876–19881. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Cai, W.M.; Long, M.C.; Zhou, B.X.; Wu, Y.H.; Wu, D.Y.; Feng, Y.J. Synthesis of visible-light responsive graphene oxide/TiO2 composites with p/n heterojunction. ACS Nano 2010, 4, 6425–6432. [Google Scholar] [CrossRef] [PubMed]
- Fitri, M.A.; Ota, M.; Hirota, Y.; Uchida, Y.; Nishiyama, N. Fabrication of TiO2-graphene photocatalyst by direct chemical vapor deposition and its anti-fouling property. Mater. Chem. Phys. 2017, 198, 42–48. [Google Scholar] [CrossRef]
- Yang, L.X.; Li, Z.Y.; Jiang, H.M.; Jiang, W.J.; Luo, Y. Photoelectrocatalytic oxidation of bisphenol A over mesh of TiO2/graphene/Cu2O. Appl. Catal. B Environ. 2016, 183, 75–85. [Google Scholar] [CrossRef]
- Iglesias, D.; Atienzar, P.; Vázquez, E.; Herrero, M.A.; García, H. Carbon nanohorns modified with conjugated terthienyl/terthiophene structures: Additives to enhance the performance of dye-sensitized solar cells. Nanomaterials 2017, 7, 294. [Google Scholar] [CrossRef] [PubMed]
- Nica, I.C.; Stan, M.S.; Popa, M.; Chifiriuc, M.C.; Pircalabioru, G.G.; Lazar, V.; Dumitrescu, I.; Diamandescu, L.; Feder, M.; Baibarac, M.; et al. Development and biocompatibility evaluation of photocatalytic TiO2/reduced graphene oxide-based nanoparticles designed for self-cleaning purposes. Nanomaterials 2017, 7, 279. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, C.; Shi, G.Q. Graphene materials for electrochemical capacitors. J. Phys. Chem. Lett. 2013, 4, 1244–1253. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.J.; Yu, J.G. Graphene-based photocatalysts for hydrogen generation. J. Phys. Chem. Lett. 2013, 4, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Li, Y.M.; Liu, S.M.; Zhang, L. N-doped TiO2 nanotubes as an effective additive to improve the catalytic capability of methanol oxidation for Pt/graphene nanocomposites. Nanomaterials 2016, 6, 40. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.C.; Yao, Z.H.; Wang, X.Q. Graphene-based nanomaterials for catalysis. Ind. Eng. Chem. Res. 2017, 56, 3477–3502. [Google Scholar] [CrossRef]
- Mishra, Y.K.; Adelung, R. ZnO tetrapod materials for functional applications. Mater. Today 2018. [Google Scholar] [CrossRef]
- Garlof, S.; Mecklenburg, M.; Smazna, D.; Mishra, Y.K.; Adelung, R.; Schulte, K.; Fiedler, B. 3D carbon networks and their polymer composites: Fabrication and electromechanical investigations of neat aerographite and aerographite-based PNCs under compressive load. Carbon 2017, 111, 103–112. [Google Scholar] [CrossRef]
- TorresLuisa, S.M.; MartínezJosé, M.; Joaquim, L.; Adrián, L.F.; Silva, M.T. Design of graphene-based TiO2 photocatalysts—A review. Environ. Sci. Pollut. Res. 2012, 19, 3676–3687. [Google Scholar]
- Zhang, Q.; He, Y.Q.; Gang, X.; Hu, C.D.; Lin, H.; Li, J.; Yin, T.; Ji, L.L. Structure and photocatalytic properties of TiO2-graphene oxide intercalated composite. Chin. Sci. Bull. 2011, 56, 331–339. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Li, H.P.; Cui, X.L.; Lin, Y.H. Graphene/TiO2 nanocomposites: Synthesis, characterization and application in hydrogen evolution from water photocatalytic splitting. J. Mater. Chem. 2010, 20, 2801–2806. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.J.; Li, Y.M.; Wang, Y.; Li, J.H. P25–graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.G.; Xiao, P.; Tian, J.; Wang, F.Z.; Yu, J.G. Phenylamine-functionalized rGO/TiO2 photocatalysts: Spatially separated adsorption sites and tunable photocatalytic selectivity. ACS Appl. Mater. Interfaces 2016, 8, 29470–29477. [Google Scholar] [CrossRef] [PubMed]
- Li, L.H.; Yu, L.L.; Lin, Z.Y.; Yang, G.W. Reduced TiO2-graphene oxide heterostructure as broad spectrum-driven efficient water-splitting photocatalysts. ACS Appl. Mater. Interfaces 2016, 8, 8536–8545. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.W.; Guo, H.L.; Sun, H.Q.; Zeng, R.C. Hydrothermal synthesis and photoelectrochemical performance enhancement of TiO2/graphene composite in photo-generated cathodic protection. Appl. Surf. Sci. 2016, 382, 128–134. [Google Scholar] [CrossRef]
- Min, S.X.; Wang, F.; Lu, G.X. Graphene-induced spatial charge separation for selective water splitting over TiO2 photocatalyst. Catal. Commun. 2016, 80, 28–32. [Google Scholar] [CrossRef]
- Štengl, V.; Popelková, D.; Václav, P. TiO2–graphene nanocomposite as high performance photocatalysts. J. Phys. Chem. C 2011, 115, 25209–25218. [Google Scholar] [CrossRef]
- Williams, G.; Seger, B.; Kamat, P.V. TiO2-graphene nanocomposites. UV-assisted photocatalytic reduction of graphene oxide. ACS Nano 2008, 2, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Anandan, S.; Rao, T.N.; Sathish, M.; Rangappa, D.; Honma, I.; Miyauchi, M. Superhydrophilic graphene-loaded TiO2 thin film for self-cleaning applications. ACS Appl. Mater. Interfaces 2013, 5, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.Y.; Yang, M.Q.; Tang, Z.R.; Xu, Y.J. Noncovalently functionalized graphene-directed synthesis of ultralarge graphene-based TiO2 nanosheet composites: Tunable morphology and photocatalytic applications. J. Phys. Chem. C 2014, 118, 27325–27335. [Google Scholar] [CrossRef]
- Jiang, G.J.; Tian, C.G.; Pan, Q.J.; Jiang, Z.; Wang, J.Q.; Yan, W.S.; Fu, H.G. Enhanced photocatalytic activity and electron transfer mechanisms of graphene/TiO2 with exposed {001} facets. J. Phys. Chem. C 2011, 115, 23718–23725. [Google Scholar] [CrossRef]
- Wang, W.S.; Wang, D.H.; Qu, W.G.; Lu, L.G.; Xu, A.W. Large ultrathin anatase TiO2 nanosheets with exposed {001} facets on graphene for enhanced visible light photocatalytic activity. J. Phys. Chem. C 2012, 116, 19893–19901. [Google Scholar] [CrossRef]
- Gu, L.A.; Wang, J.Y.; Cheng, H.; Zhao, Y.Z.; Liu, L.F.; Han, X.J. One-step preparation of graphene-supported anatase TiO2 with exposed {001} facets and mechanism of enhanced photocatalytic properties. ACS Appl. Mater. Interfaces 2013, 5, 3085–3093. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Luo, L.J.; Xiao, M.; Li, H.N.; Pan, X.J.; Jiang, F.Z. One-step hydrothermal synthesis of surface fluorinated TiO2/reduced graphene oxide nanocomposites for photocatalytic degradation of estrogens. Mater. Sci. Semicond. Process. 2015, 40, 183–193. [Google Scholar] [CrossRef]
- Pham, T.T.; Huy, C.N.; Lee, H.J.; Phan, T.D.; Son, T.H.; Kim, C.K.; Shin, E.W. Cu-doped TiO2/reduced graphene oxide thin-film photocatalysts: Effect of Cu content upon methylene blue removal in water. Ceram. Int. 2015, 41, 11184–11193. [Google Scholar] [CrossRef]
- Safarpour, M.; Khataee, A.; Vatanpour, V. Preparation of a novel polyvinylidene fluoride (PVDF) ultrafiltration membrane modified with reduced graphene oxide/titanium dioxide (TiO2) nanocomposite with enhanced hydrophilicity and antifouling properties. Ind. Eng. Chem. Res. 2014, 53, 13370–13382. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, L.Q.; Liu, R.; Gao, Z.F.; Yang, X.P.; Tu, Z.Q.; Yang, F.; Ye, Z.Z.; Cui, L.S.; Xu, C.M.; et al. Hydrothermal synthesis of N-doped TiO2 nanowires and N-doped graphene heterostructures with enhanced photocatalytic properties. J. Alloys Compd. 2016, 656, 24–32. [Google Scholar] [CrossRef]
- Mou, Z.G.; Wu, Y.J.; Sun, J.H.; Yang, P.; Du, Y.K.; Lu, C. TiO2 nanoparticles-functionalized N-doped graphene with superior interfacial contact and enhanced charge separation for photocatalytic hydrogen generation. ACS Appl. Mater. Interfaces 2014, 6, 13798–13806. [Google Scholar] [CrossRef] [PubMed]
- Mamba, G.; Mamo, M.A.; Mbianda, X.Y.; Mishra, A.K. Nd, N, S-TiO2 Decorated on reduced graphene oxide for a visible light active photocatalyst for dye degradation: Comparison to its MWCNT/Nd, N, S-TiO2 analogue. Ind. Eng. Chem. Res. 2014, 53, 14329–14338. [Google Scholar] [CrossRef]
- Jaiswal, V.; Umrao, K.S.; Rastogi, R.B.; Kumar, R.; Srivastava, A. Synthesis, characterization, and tribological evaluation of TiO2-reinforced boron and nitrogen co-doped reduced graphene oxide based hybrid nanomaterials as efficient antiwear lubricant additives. ACS Appl. Mater. Interfaces 2016, 8, 11698–11710. [Google Scholar] [CrossRef] [PubMed]
- Ao, Y.H.; Xu, L.Y.; Wang, P.F.; Wang, C.; Hou, J.; Qian, J.; Li, Y. Graphene and TiO2 co-modified flower-like Bi2O2CO3: A novel multi-heterojunction photocatalyst with enhanced photocatalytic activity. Appl. Surf. Sci. 2015, 355, 411–418. [Google Scholar] [CrossRef]
- Perera, S.D.; Mariano, R.G.; Vu, K.; Nour, N.; Seitz, O.; Chabal, Y.; Balkus, K.J. Hydrothermal synthesis of graphene-TiO2 nanotube composites with enhanced photocatalytic activity. ACS Catal. 2012, 2, 949–956. [Google Scholar] [CrossRef]
- Li, Y.R.; Yan, J.; Su, Q.; Xie, E.Q.; Lan, W. Preparation of graphene–TiO2 nanotubes/nanofibers composites as an enhanced visible light photocatalyst using a hybrid synthetic strategy. Mater. Sci. Semicond. Process. 2014, 27, 695–701. [Google Scholar] [CrossRef]
- Kim, H.; Kim, S.; Kang, J.; Choi, W. Graphene oxide embedded into TiO2 nanofiber: Effective hybrid photocatalyst for solar conversion. J. Catal. 2014, 309, 49–57. [Google Scholar] [CrossRef]
- Qiu, B.C.; Xing, M.Y.; Zhang, J.L. Mesoporous TiO2 nanocrystals grown in situ on graphene aerogels for high photocatalysis and lithium-ion batteries. J. Am. Chem. Soc. 2014, 136, 5852–5855. [Google Scholar] [CrossRef] [PubMed]
- Haldorai, Y.; Rengaraj, A.; Kwak, C.H.; Huh, Y.S.; Han, Y.K. Fabrication of nano TiO2@graphene composite: Reusable photocatalyst for hydrogen production, degradation of organic and inorganic pollutants. Synth. Met. 2014, 198, 10–18. [Google Scholar] [CrossRef]
- Pan, D.Y.; Jiao, J.K.; Li, Z.; Guo, Y.T.; Feng, C.Q.; Liu, Y.; Wang, L.; Wu, M.H. Efficient separation of electron–hole pairs in graphene quantum dots by TiO2 heterojunctions for dye degradation. ACS Sustain. Chem. Eng. 2015, 3, 2405–2413. [Google Scholar] [CrossRef]
- Ding, Y.B.; Bai, W.; Sun, J.H.; Wu, Y.; Memon, M.A.; Wang, C.; Liu, C.B.; Huang, Y.; Geng, J.X. Cellulose tailored anatase TiO2 nanospindles in three-dimensional graphene composites for high-performance supercapacitors. ACS Appl. Mater. Interfaces 2016, 8, 12165–12175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Xiao, F.; Guo, Y.L.; Wang, S.; Liu, Y.Q. One-pot self-assembled three-dimensional TiO2-graphene hydrogel with improved adsorption capacities and photocatalytic and electrochemical activities. ACS Appl. Mater. Interfaces 2013, 5, 2227–2233. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Jin, T.; Liu, C.; Li, G.H.; Yan, M.D. Three-dimensional graphene–TiO2 nanocomposite photocatalyst synthesized by covalent attachment. ACS Omega 2016, 1, 351–356. [Google Scholar] [CrossRef]
- Linsebigler, L.A.; Lu, G.G.; Yates, J.T. Photocatalysis on TiO2 surfaces: Principles, mechanisms and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.L.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Bointon, T.H.; Jones, G.F.; Sanctis, A.D.; Pearce, R.H.; Craciun, M.F.; Russo, S. Large-area functionalized CVD graphene for work function matched transparent electrodes. Sci. Rep. 2015, 5, 16464–16469. [Google Scholar] [CrossRef] [PubMed]
- Zahid, F.S.; Saad, P.S.M.; Zahidi, M.M.; Mahmood, M.R. Influence of cathode work functions on the photovoltaic properties of MEH-PPV: TiO2 bulk heterojunction solar cell. Adv. Mater. Res. 2014, 832, 399–403. [Google Scholar] [CrossRef]
- Park, J.; Ann, Y.H.; Ruiz, C. Imaging of photocurrent generation and collection in single-layer graphene. Nano Lett. 2009, 9, 1742–1746. [Google Scholar] [CrossRef] [PubMed]
- Mai, S.; Syzranov, S.V.; Efetov, K.B. Photocurrent in a visible-light graphene photodiode. Phys. Rev. B 2011, 83, 033402–033406. [Google Scholar] [CrossRef]
- Curran, K.S.; Lamouche, D. Transport and kinetics in photoelectrolysis by semiconductor particles in suspension. J. Phys. Chem. 1983, 87, 5405–5411. [Google Scholar] [CrossRef]
- Gerischer, H. A mechanism of electron hole pair separation in illuminated semiconductor particles. J. Phys. Chem. 1984, 88, 6096–6097. [Google Scholar] [CrossRef]
- Fuentes, G.G.; Elizalde, E.; Yubero, F.; Sanz, J.M. Electron inelastic mean free paths have been estimated for Ti, TiC, TiN and TiO2 for energies between 250 and 2000 eV. Surf. Interface Anal. 2002, 33, 230–237. [Google Scholar] [CrossRef]
- Tang, B.; Ji, G.J.; Wang, Z.W.; Chen, H.Q.; Li, X.F.; Yu, H.G.; Li, S.; Liu, H. Three-dimensional graphene networks and reduced graphene oxide nanosheets co-modified dye-sensitized solar cells. RSC Adv. 2017, 7, 45280–45286. [Google Scholar] [CrossRef]
- Abazović, N.D.; Cÿomor, M.I.; Dramicanin, M.D.; Jovanović, D.J.; Ahrenkiel, S.P.; Nedeljkovic, J.M. Photoluminescence of anatase and rutile TiO2 particles. J. Phys. Chem. B 2006, 110, 25366–25370. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Berger, H.; Schmid, P.E.; Levy, F.; Burri, G. Photoluminescence in TiO2 anatase single crystals. Solid State Commun. 1993, 87, 847–850. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Ding, C.X. Investigation on the surface state of TiO2, ultrafine particles by luminescence. J. Solid State Chem. 1999, 145, 711–715. [Google Scholar] [CrossRef]
- Anpo, M.; Aikava, N.; Kubokawa, Y. Photoluminescence and photocatalytic activity of highly dispersed titanium oxide anchored onto porous vycor glass. J. Phys. Chem. 1985, 89, 5017–5021. [Google Scholar] [CrossRef]
- Melnyk, V.; Shymanovska, V.; Puchkovska, G.; Bezrodna, T.; Klishevich, G. Low-temperature luminescence of different TiO2 modifications. J. Mol. Struct. 2005, 744, 573–576. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Yamaguchi, Y.; Sekiya, T.; Kurita, S. Time-resolved luminescence spectra in colorless anatase TiO2 single crystal. J. Lumin. 2005, 112, 50–53. [Google Scholar] [CrossRef]
- Haart, L.G.J.; DeVries, A.J.; Blasse, G. On the photoluminescence of semiconducting titanates applied in photoelectrochemical cells. J. Solid State Chem. 1985, 58, 291–300. [Google Scholar] [CrossRef]
- Watanabe, M.; Hayashi, T.; Yagasaki, H.; Sasaki, S. Luminescence process in anatase TiO2 studied by time-resolved spectroscopy. Int. J. Mod. Phys. B 2001, 15, 3997–4000. [Google Scholar] [CrossRef]
- Sellappan, R.; Sun, J.; Galeckas, A.; Lindvall, N.; Yurgens, A.; Kuznetsovc, A.Y.; Chakarov, D. Influence of graphene synthesizing techniques on the photocatalytic performance of graphene–TiO2 nanocomposites. Phys. Chem. Chem. Phys. 2013, 15, 15528–15537. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.F.; Zhu, Y.H.; Yang, X.L.; Xin, J.G.; Li, C.Z. Preparation of graphene–TiO2 composites with enhanced photocatalytic activity. New J. Chem. 2011, 35, 353–359. [Google Scholar] [CrossRef]
- Wang, Y.A.; Zhang, S.L.; Zeng, Y.Q.; Ou, M.; Zhong, Q. Photocatalytic oxidation of NO over TiO2-Graphene catalyst by UV/H2O2 process and enhanced mechanism analysis. J. Mol. Catal. A Chem. 2016, 423, 339–346. [Google Scholar] [CrossRef]
- Zhao, D.L.; Sheng, G.D.; Chen, C.L.; Wang, X.K. Enhanced photocatalytic degradation of methylene blue under visible irradiation on graphene@TiO2 dyade structure. Appl. Catal. B Environ. 2012, 111, 303–308. [Google Scholar] [CrossRef]
- Wei, M.; Wan, J.M.; Hu, Z.W.; Peng, Z.Q.; Wang, B. Enhanced photocatalytic degradation activity over TiO2 nanotubes co-sensitized by reduced graphene oxide and copper(II) meso-tetra(4-carboxyphenyl)porphyrin. Appl. Surf. Sci. 2016, 377, 149–158. [Google Scholar] [CrossRef]
- Chen, C.J. Introduction to Scanning Tunneling Microscopy; Oxford University Press: Oxford, UK, 1993. [Google Scholar]
- Binnig, G.; Rohrer, H. Scanning tunneling microscopy. IBM J. Res. Dev. 1986, 30, 355–369. [Google Scholar]
- Zhou, C.Y.; Ren, Z.F.; Tan, S.J.; Ma, Z.B.; Mao, X.C.; Dai, D.X.; Fan, H.J.; Yang, X.M.; LaRue, J.; Cooper, R.; et al. Site-specific photocatalytic splitting of methanol on TiO2 (110). Chem. Sci. 2010, 1, 575–580. [Google Scholar] [CrossRef]
- Dai, K.; Lu, L.H.; Liu, Q.; Zhu, G.P.; Liu, Q.Z.; Liu, Z.L. Graphene oxide capturing surface-fluorinated TiO2 nanosheets for advanced photocatalysis and the reveal of synergism reinforce mechanism. Dalton Trans. 2014, 43, 2202–2210. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Tang, Z.R.; Fu, X.Z.; Xu, Y.J. TiO2−graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2−graphene truly different from other TiO2−carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.X.; Zhang, P.; Ling, M.; Li, S.; Liu, P.R.; Zhao, H.J.; Zhang, S.Q. Photocatalytic synthesis of TiO2 and reduced graphene oxide nanocomposite for lithium ion battery. ACS Appl. Mater. Interfaces 2012, 4, 3636–3642. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Fan, M.; Li, B.; Dong, L.; Zhang, F. Preparation and photocatalytic activity of mixed phase TiO2-graphene composites. ACTA Phys. Chim. Sin. 2015, 31, 519–526. [Google Scholar]
- Zhang, Y.X.; Zhou, Z.Y.; Chen, T.; Wang, H.T.; Lu, W.J. Graphene TiO2 nanocomposites with high photocatalytic activity for the degradation of sodium pentachlorophenol. J. Environ. Sci. 2014, 26, 2114–2122. [Google Scholar] [CrossRef] [PubMed]
- Sordello, F.; Zeb, G.; Hu, K.; Calza, P.; Minero, C.; Szkopekc, T.; Cerruti, M. Tuning TiO2 nanoparticle morphology in graphene–TiO2 hybrids by graphene surface modification. Nanoscale 2014, 6, 6710–6719. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cao, M.H.; Wang, P.F.; Ao, Y.H.; Hou, J.; Qian, J. Preparation of graphene–carbon nanotube–TiO2 composites with enhanced photocatalytic activity for the removal of dye and Cr (VI). Appl. Catal. A Gen. 2014, 473, 83–89. [Google Scholar] [CrossRef]
- Zhang, J.J.; Wu, Y.H.; Mei, J.Y.; Zheng, G.P.; Yan, T.T.; Zheng, X.C.; Liu, P.; Guan, X.X. Synergetic adsorption and photocatalytic degradation of pollutants over 3D TiO2–graphene aerogel composites synthesized via a facile one-pot route. Photochem. Photobiol. Sci. 2016, 15, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Han, W.J.; Zang, C.; Huang, Z.Y.; Zhong, J.X. Enhanced photocatalytic activities of three-dimensional graphene-based aerogel embedding TiO2 nanoparticles and loading MoS2 nanosheets as co-catalyst. Int. J. Hydrogen Energy 2014, 39, 19502–19512. [Google Scholar] [CrossRef]
- Sun, Y.F.; Cao, Y.C.; Huang, W.Q.; Zhu, Y.J.; Heng, L.; Tang, B. High-performance photoanode for dye sensitized solar cells with graphene modified two-layer construction. Mater. Lett. 2016, 165, 178–180. [Google Scholar] [CrossRef]
- Park, J.; Yan, M.D. Three-dimensional graphene-TiO2 hybrid nanomaterial for high efficient photocatalysis. Nanotechnol. Rev. 2016, 5, 417–423. [Google Scholar] [CrossRef]
- Yu, M.; Ma, Y.X.; Liu, J.H.; Li, X.J.; Li, S.M.; Liu, S.Y. Sub-coherent growth of ZnO nanorod arrays on three-dimensional graphene framework as one-bulk high-performance photocatalyst. Appl. Surf. Sci. 2016, 390, 266–272. [Google Scholar] [CrossRef]
- Liu, X.Y.; Cong, R.D.; Cao, L.F.; Liu, S.; Cui, H.N. The structure, morphology and photocatalytic activity of graphene–TiO2 multilayer films and charge transfer at the interface. New J. Chem. 2014, 38, 2362–2367. [Google Scholar] [CrossRef]
- Ohta, T.; Bostwick, A.; Seyller, T.; Horn, K.; Rotenberg, E. Controlling the electronic structure of bilayer graphene. Science 2006, 313, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Zhang, N.; Tang, Z.R.; Xu, Y.J. Improving the photocatalytic performance of graphene–TiO2 nanocomposites via a combined strategy of decreasing defects of graphene and increasing interfacial contact. Phys. Chem. Chem. Phys. 2012, 14, 9167–9175. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.T.; Vijayan, B.K.; Gray, K.A.; Hersam, M.C. Minimizing graphene defects enhances titania nanocomposite-based photocatalytic reduction of CO2 for improved solar fuel production. Nano Lett. 2011, 11, 2865–2870. [Google Scholar] [CrossRef] [PubMed]
- Perreault, F.; Fariaa, A.F.; Elimelech, M. Environmental applications of graphene-based nanomaterials. Chem. Soc. Rev. 2015, 44, 5861–5896. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y. Hydrothermal synthesis of TiO2–RGO composites and their improved photocatalytic activity in visible light. RSC Adv. 2014, 4, 36040–36045. [Google Scholar] [CrossRef]
- Zhu, Y.Y.; Wang, Y.J.; Yao, W.Q.; Zong, R.L.; Zhu, Y.F. New insights into the relationship between photocatalytic activity and TiO2–GR composites. RSC Adv. 2015, 5, 29201–29208. [Google Scholar] [CrossRef]
- Štengl, V.; Bakardjieva, S.; Grygar, T.M.; Bludská, J.; Kormunda, M. TiO2-graphene oxide nanocomposite as advanced photocatalytic materials. Chem. Cent. J. 2013, 7, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Wu, J.; Sun, L.; Liu, Y.; Fang, T.; Zhu, S.; Li, S.; Wang, Y. 3D graphene/ZnO composite with enhanced photocatalytic activity. Mater. Des. 2016, 90, 839–844. [Google Scholar] [CrossRef]
- Gao, W.Y.; Wang, M.Q.; Ran, C.X.; Yao, X.; Yang, H.H.; Liu, J.; He, D.L.; Bai, J.B. One-pot synthesis of Ag/r-GO/TiO2 nanocomposites with high solar absorption and enhanced anti-recombination in photocatalytic applications. Nanoscale 2014, 6, 5498–5508. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhao, Y.; Liu, S.; Korzeniewski, C.L.; Wang, S.; Fan, Z.Y. Comparing graphene-TiO2 nanowire and graphene-TiO2 nanoparticle composite photocatalysts. ACS Appl. Mater. Interfaces 2012, 4, 3944–3950. [Google Scholar] [CrossRef] [PubMed]
- Domýngu, K.A.; Kamat, P.V. Single wall carbon nanotube scaffolds for photoelectrochemical solar cells: Capture and transport of photogenerated electrons. Nano Lett. 2007, 7, 676–680. [Google Scholar]
- Kongkanand, A.; Kamat, P.V. Electron storage in single wall carbon nanotubes. Fermi level equilibration in semiconductor–SWCNT suspensions. ACS Nano 2007, 1, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.C.; Zhang, W.H.; Fan, X.D.; Wu, P.; Zeng, C.G.; Li, Z.Y.; Zhai, X.F.; Yang, J.L.; Hou, J.G. Graphene thickness control via gas-phase dynamics in chemical vapor deposition. J. Phys. Chem. C 2012, 116, 10557–10562. [Google Scholar] [CrossRef]
- Murdock, A.T.; Koos, A.; Britton, T.B.; Houben, L.; Batten, T.; Zhang, T.; Wilkinson, A.J.; Borkowski, R.E.D.; Lekka, C.E.; Grobert, N. Controlling the o orientation, edge geometry, and thickness of chemical vapor deposition graphene. ACS Nano 2013, 7, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
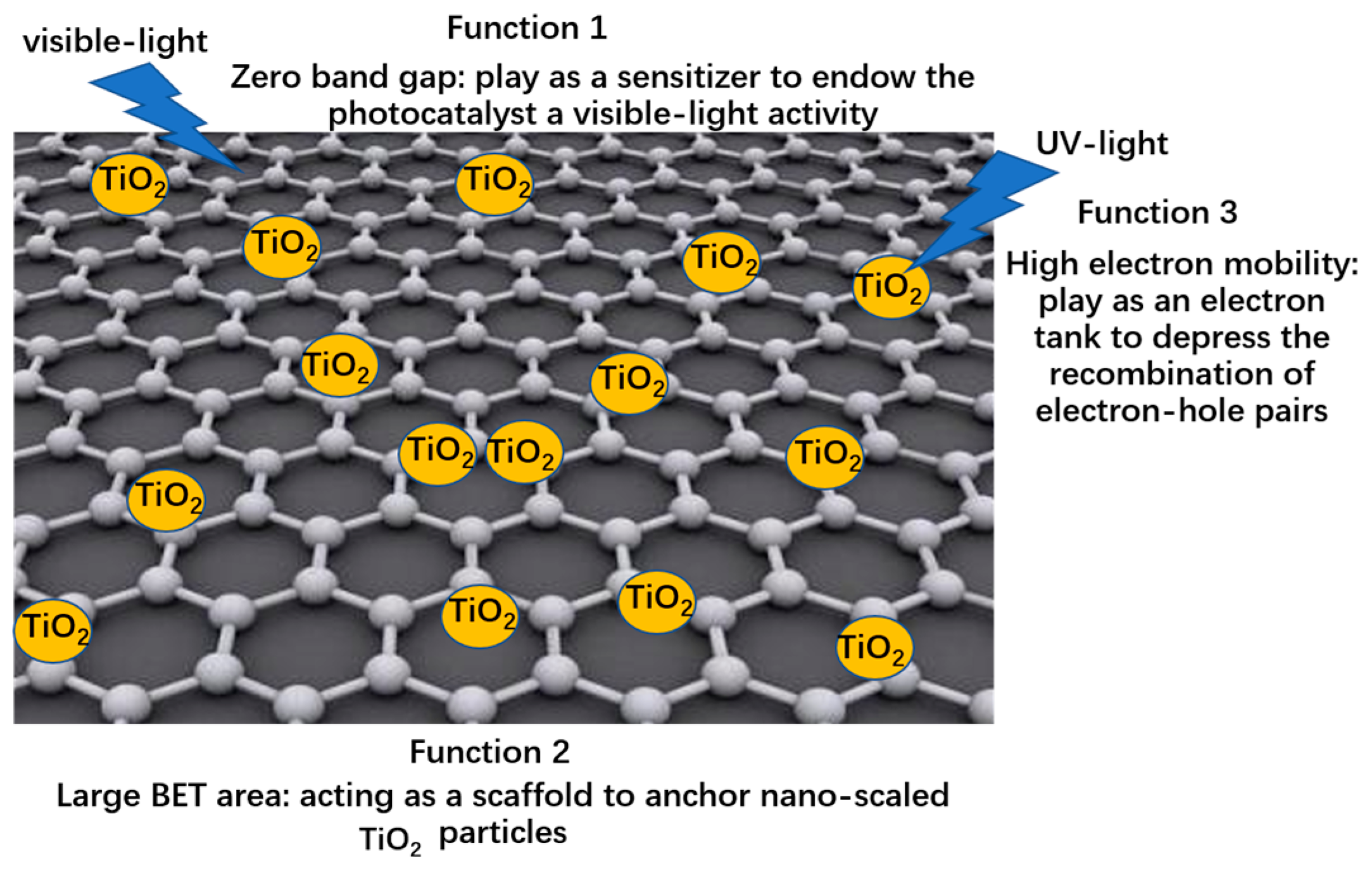



| Bottlenecks of Pure TiO2 | Solution | Essential Reason | Specific Optimizing | Relationship |
|---|---|---|---|---|
| Lacking visible light activity | Sensitization of graphene | Exciting photo-induced electron under visible light irradiation | Optimizing thickness of graphene | Thinner graphene brings about a higher quantum tunneling probability |
| Optimizing functional group amount and types of the RGO | A higher defect density or functional group amount is beneficial to a higher tunneling probability | |||
| Providing electron transport channels from graphene to TiO2 | Achieving the transfer of photo-induced electrons | Optimizing defect density of the 3DGNs | ||
| High electron-hole recombination rate | Improving quality of graphene | Enhancing the mean free path of electron | Optimizing the preparation process of graphene and resulting composite photocatalysts | A higher quality brings about a higher mean free path |
| Achieving the fast electron transport between graphene and TiO2 | Prolong the lifetime of electrons | Optimizing the morphology and defect density (or functional group amount) of graphene | A higher defect density or functional group amount provides more transport channels | |
| Low chemical adsorption ability | Increasing the BET area | Providing more adsorption sites | Optimizing the morphology of graphene and TiO2 | Graphene and TiO2 with 3D construction is beneficial to a higher BET area |
| Increasing chemisorption active sites | Promote the formation of chemical bond between graphene and TiO2 | Optimizing defect density (or functional group amount and type) of graphene modifier | A higher defect density or functional group amount provides more chemisorption active sites |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, B.; Chen, H.; Peng, H.; Wang, Z.; Huang, W. Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective. Nanomaterials 2018, 8, 105. https://doi.org/10.3390/nano8020105
Tang B, Chen H, Peng H, Wang Z, Huang W. Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective. Nanomaterials. 2018; 8(2):105. https://doi.org/10.3390/nano8020105
Chicago/Turabian StyleTang, Bo, Haiqun Chen, Haoping Peng, Zhengwei Wang, and Weiqiu Huang. 2018. "Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective" Nanomaterials 8, no. 2: 105. https://doi.org/10.3390/nano8020105
APA StyleTang, B., Chen, H., Peng, H., Wang, Z., & Huang, W. (2018). Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective. Nanomaterials, 8(2), 105. https://doi.org/10.3390/nano8020105




