A Recyclable Cu-MOF-74 Catalyst for the Ligand-Free O-Arylation Reaction of 4-Nitrobenzaldehyde and Phenol
Abstract
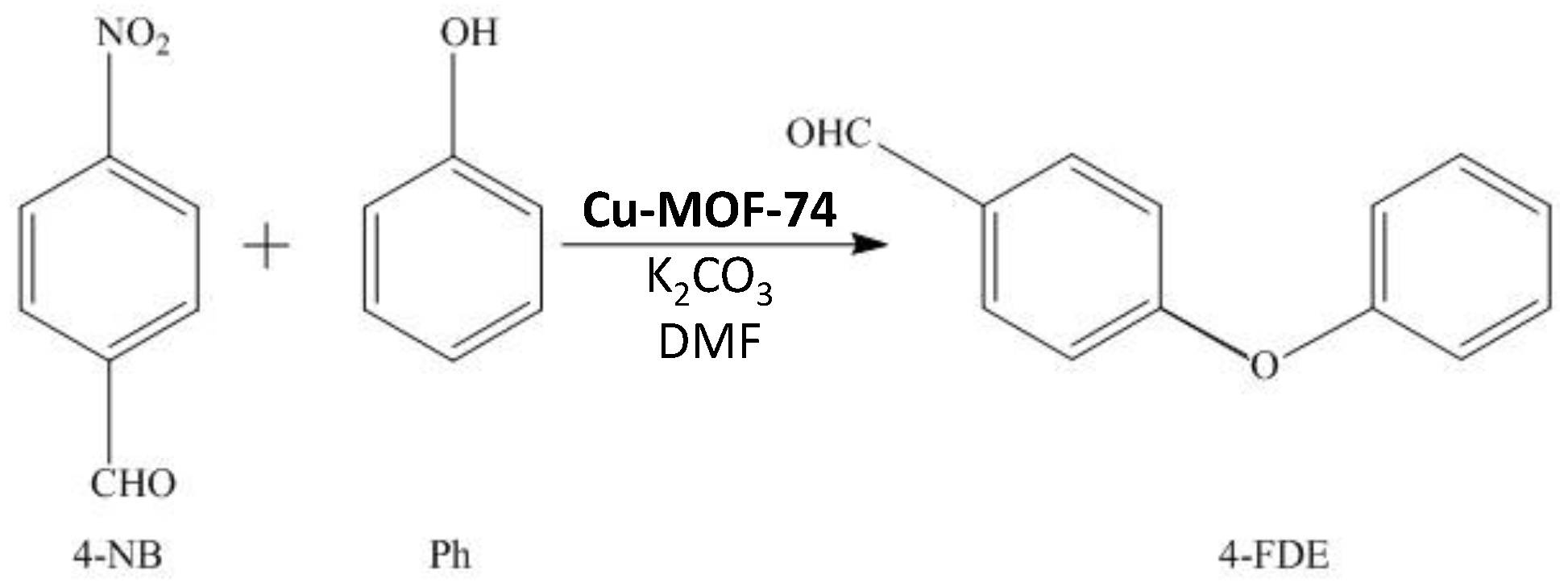
:1. Introduction
2. Results and Discussion
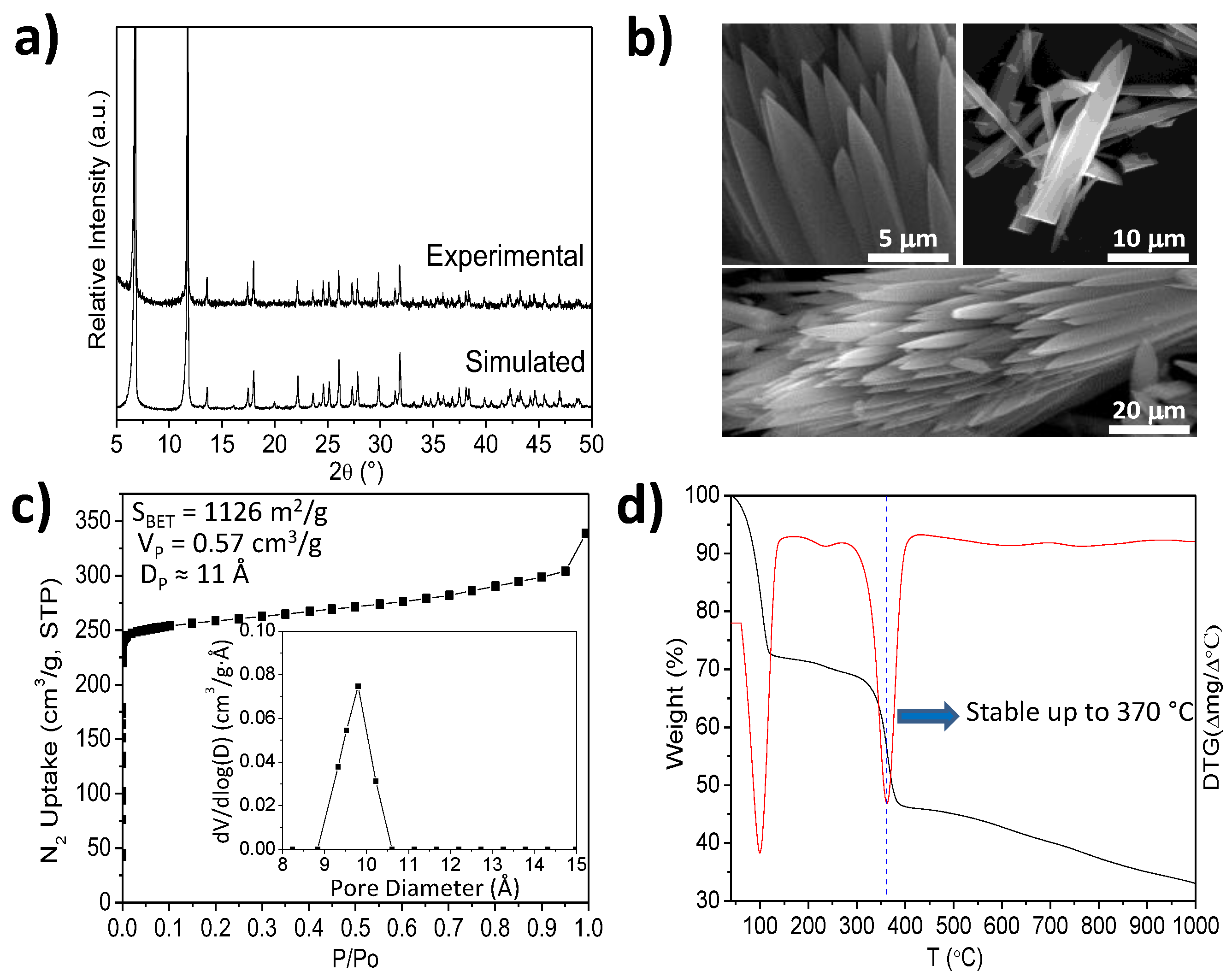
2.1. Characterization of Cu-MOF-74
2.2. Catalytic Study
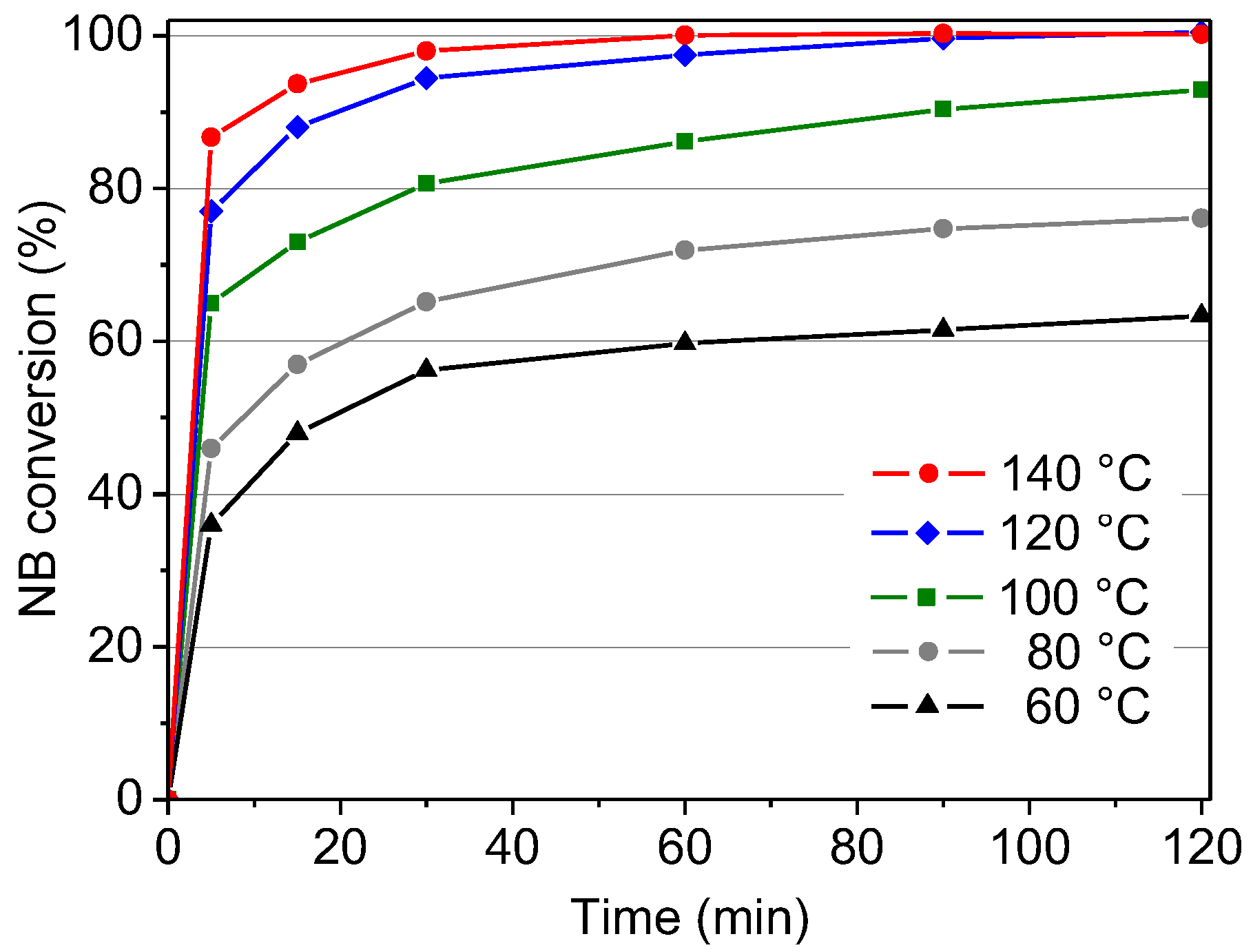
2.2.1. Influence of Temperature
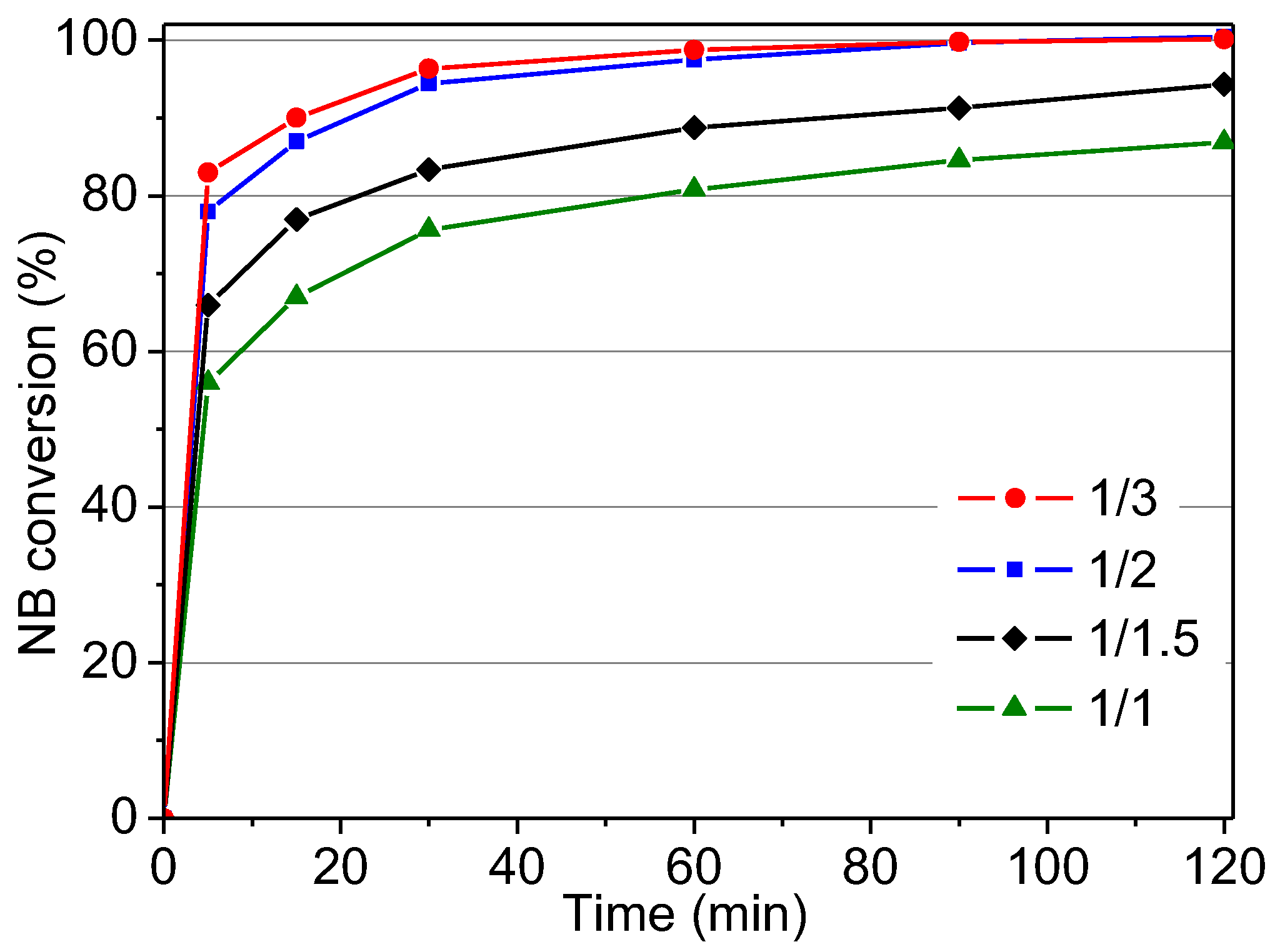
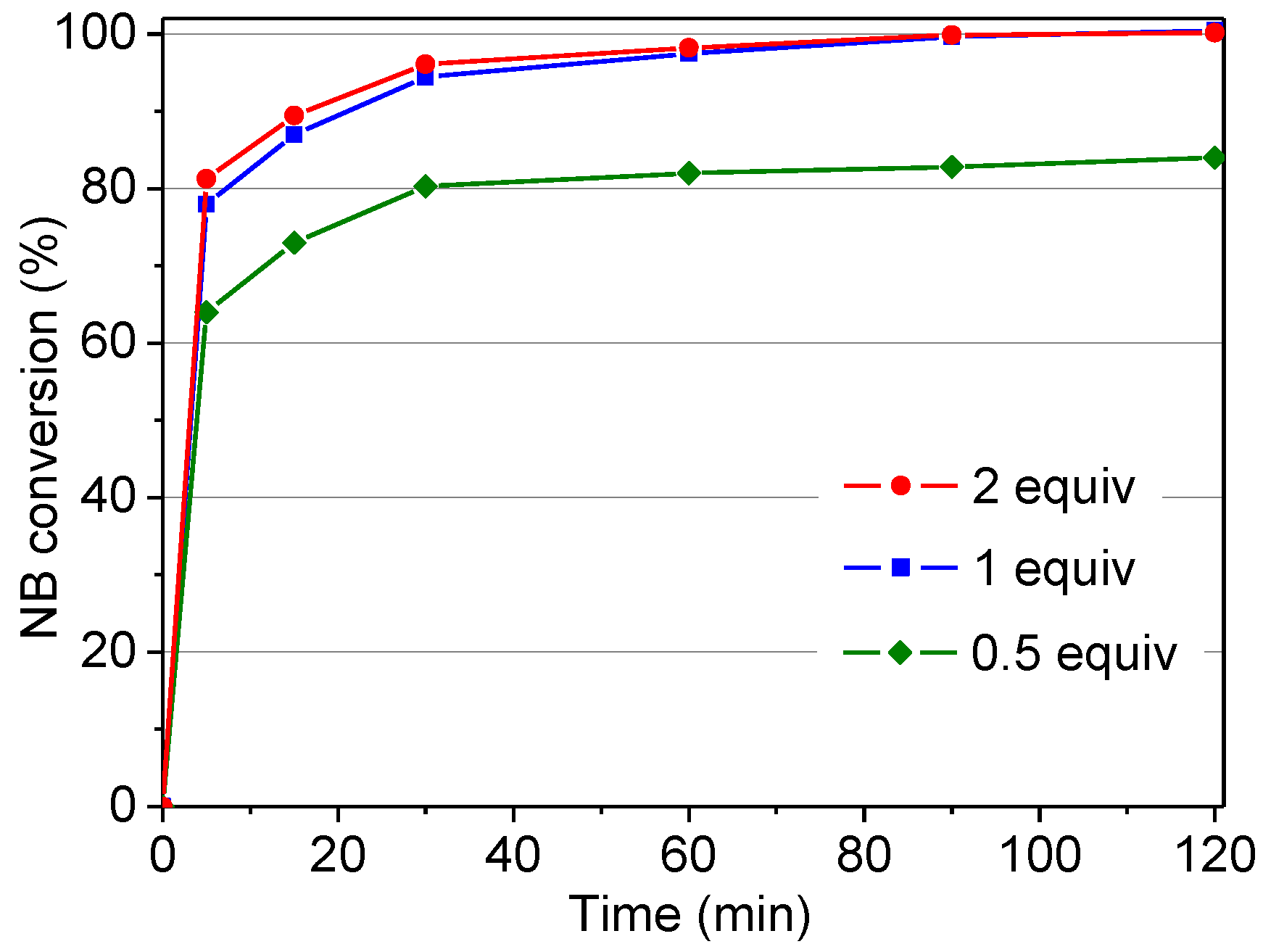
2.2.2. Influence of 4-Nitrobenzaldehyde/Phenol Molar Ratio
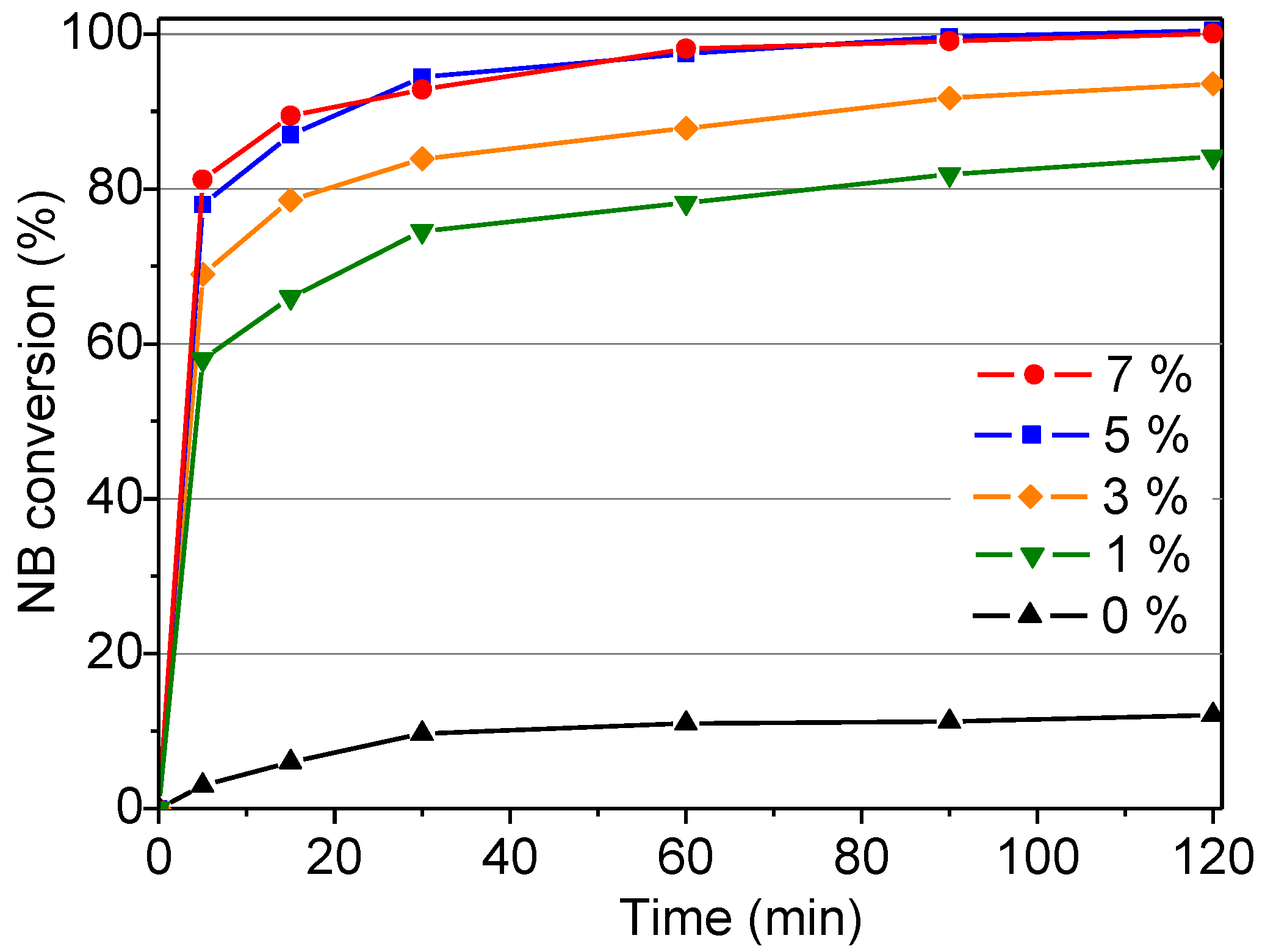
2.2.3. Influence of Catalyst Concentration
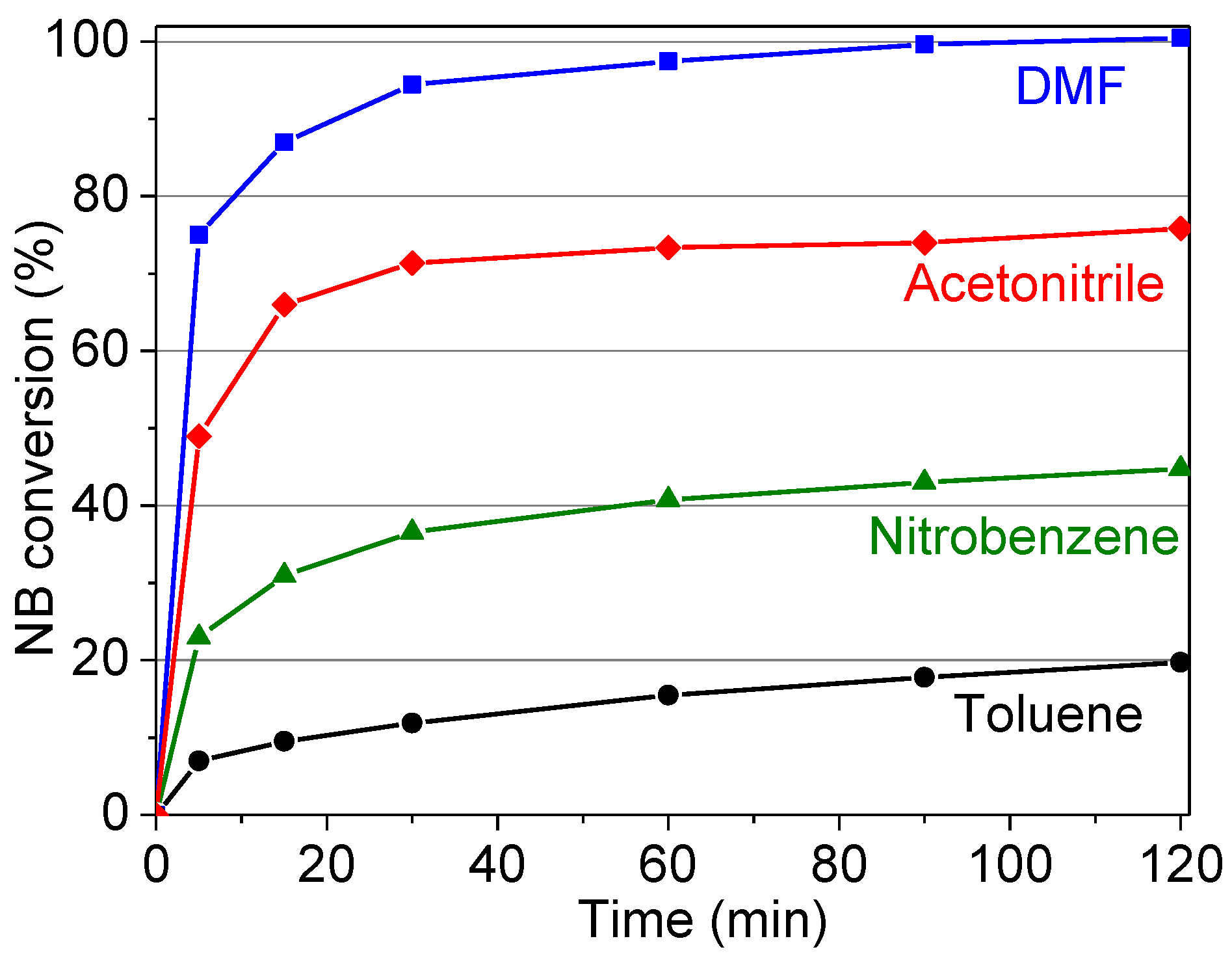
2.2.4. Influence of the Solvent
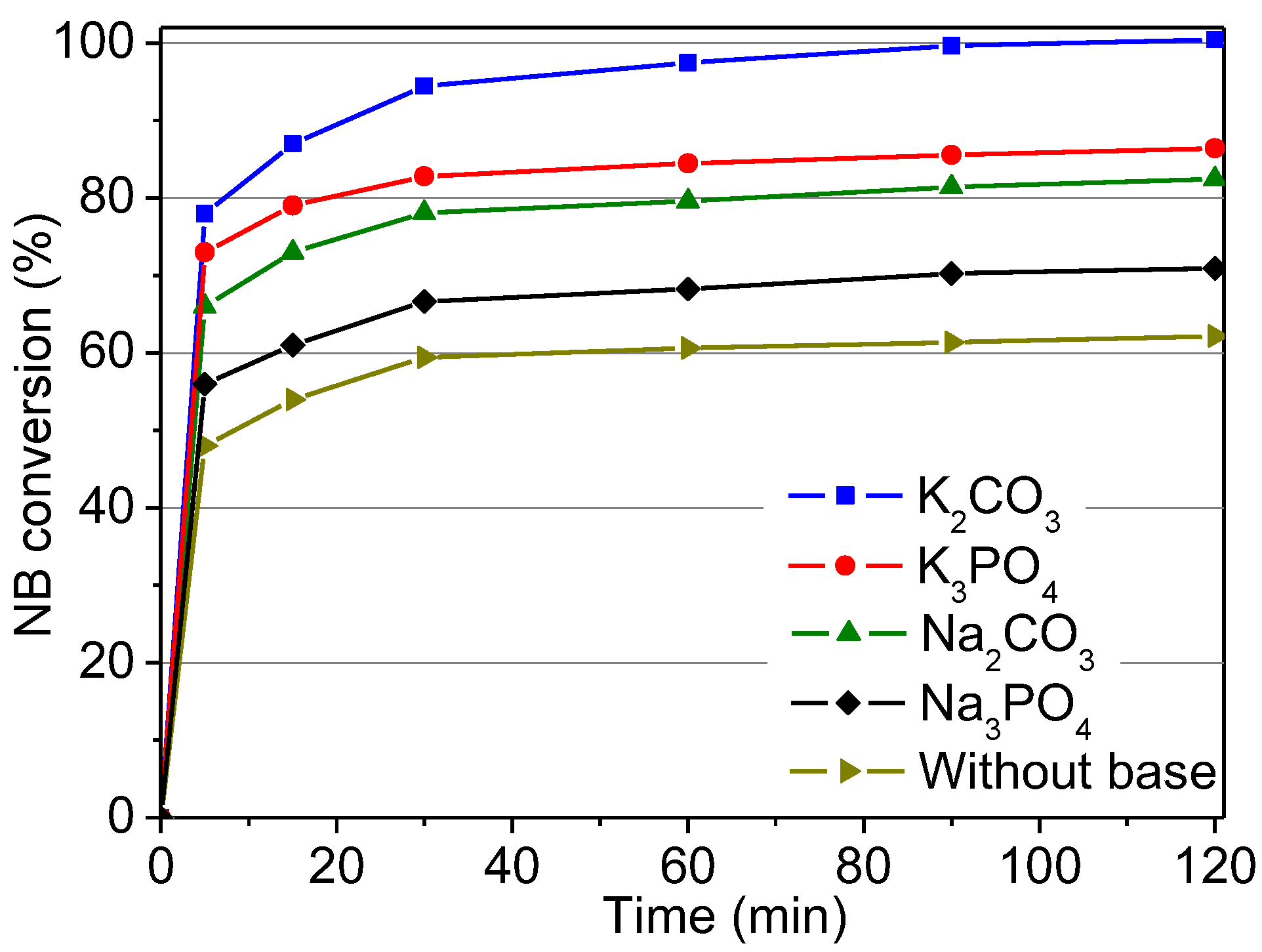
2.2.5. Influence of the Base
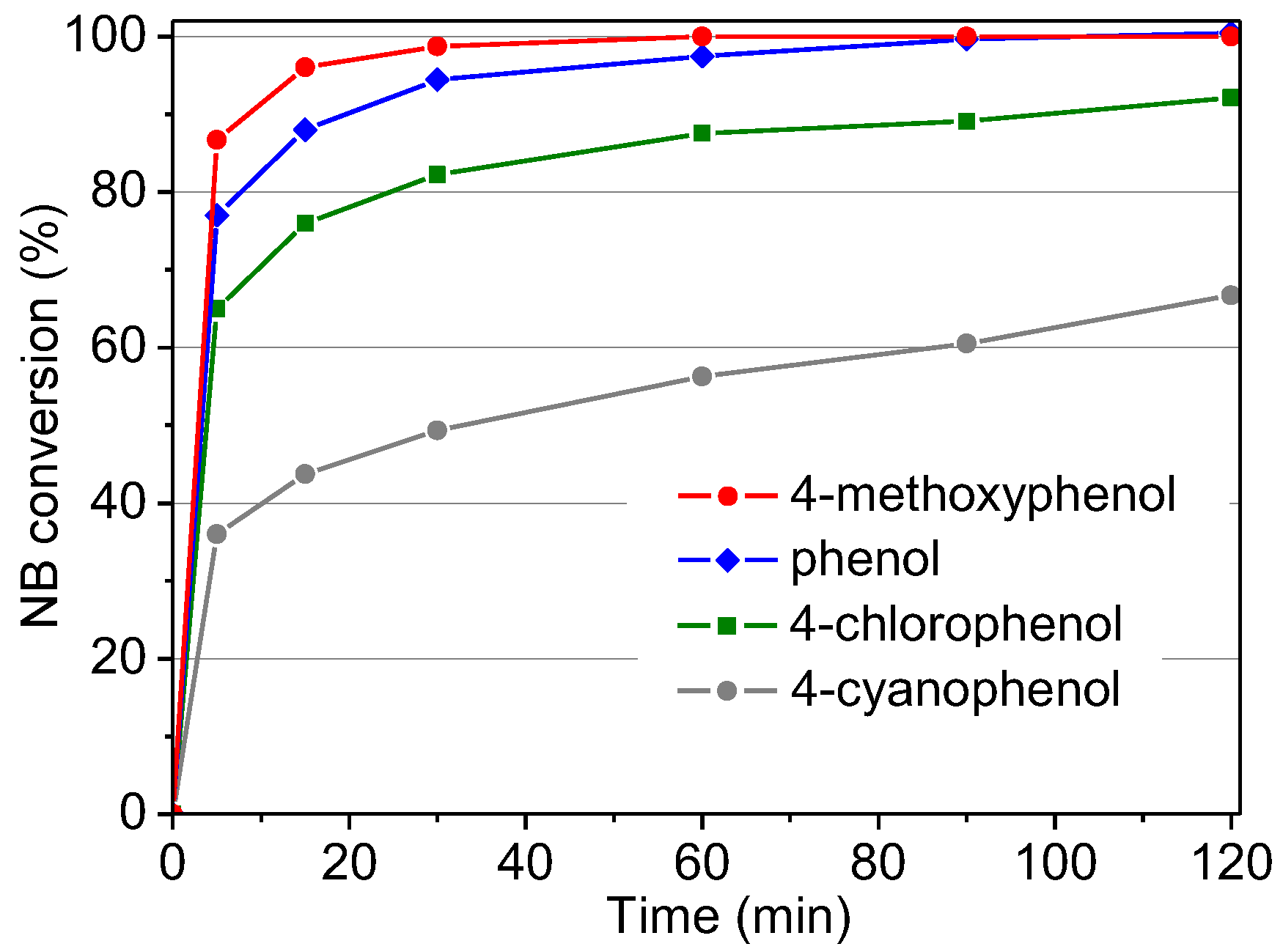
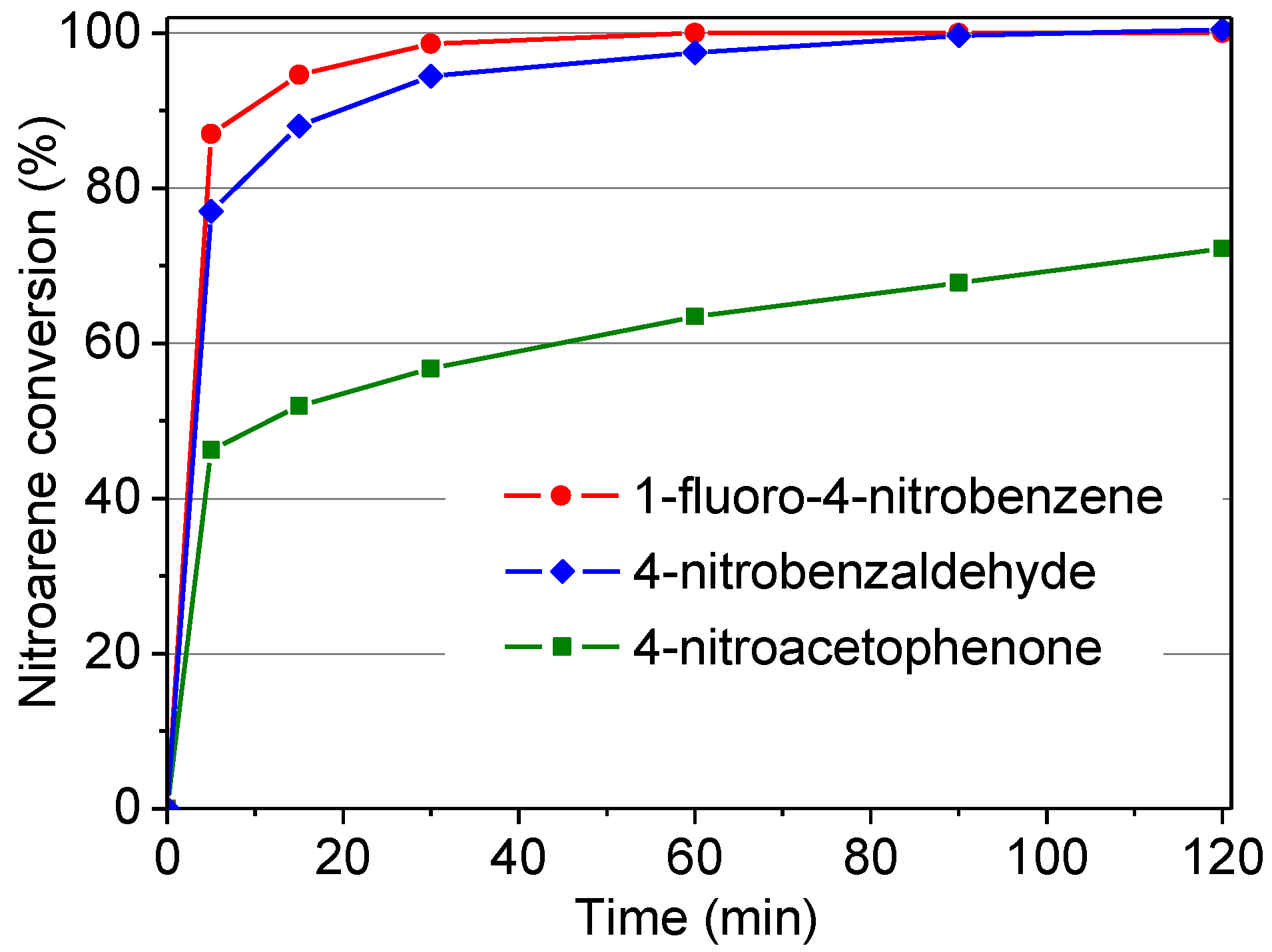
2.2.6. Influence of Different Substrates
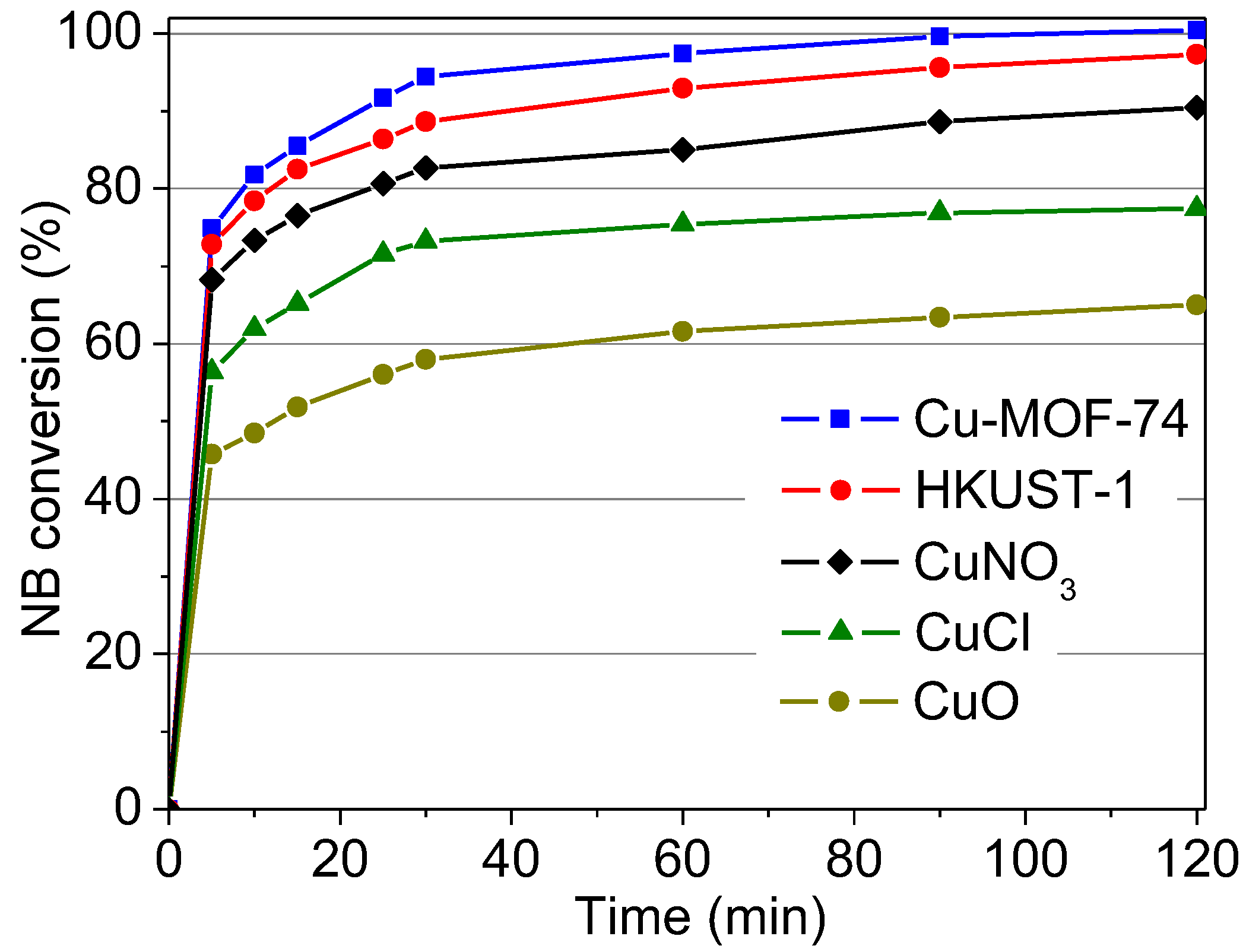
2.2.7. Comparison with Other Cu-Catalysts
2.2.8. Effect of Nanostructured Cu-MOF-74
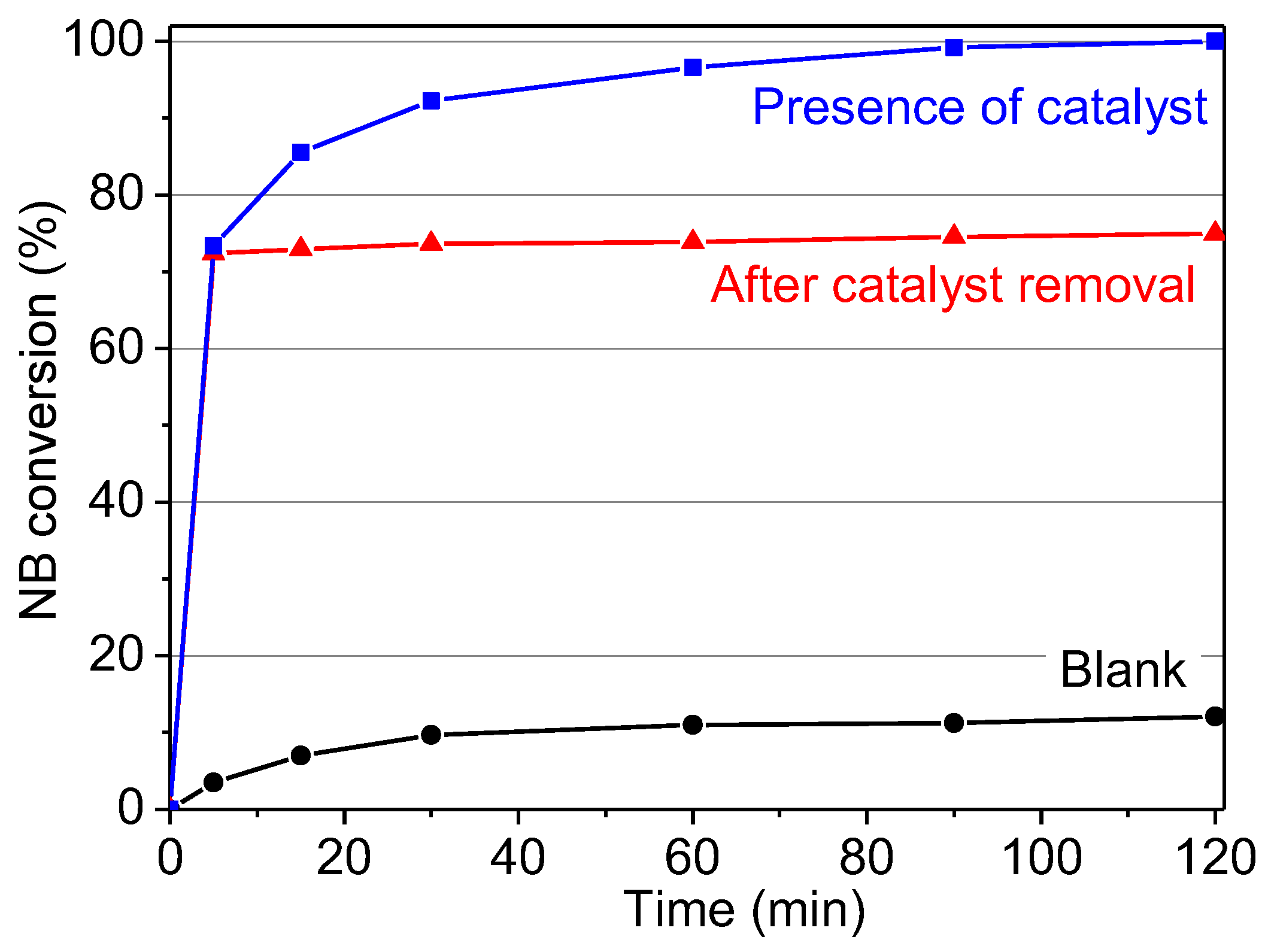
2.2.9. Reuse of Cu-MOF-74
3. Materials and Methods
3.1. Catalysts Preparation
3.2. Catalyst Characterization
3.3. Reaction Procedure
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lindley, J. Copper assisted nucleophilic substitution of aryl halogen. Tetrahedron 1984, 40, 1433–1456. [Google Scholar] [CrossRef]
- Evano, G.; Blanchard, N.; Toumi, M. Copper-Mediated Coupling Reactions and Their Applications in Natural Products and Designed Biomolecules Synthesis. Chem. Rev. 2008, 108, 3054–3131. [Google Scholar] [CrossRef] [PubMed]
- Maiti, D.; Buchwald, S.L. Cu-Catalyzed Arylation of Phenols: Synthesis of Sterically Hindered and Heteroaryl Diaryl Ethers. J. Org. Chem. 2010, 75, 1791–1794. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, V.R.; Dumbre, D.K.; Yadav, P.N.; Bhargava, S.K. Thermally decomposed Cu-Fe-hydrotalcite: A novel highly active catalyst for o-arylation of naphthol and phenols by aryl halides. Catal. Commun. 2012, 29, 132–136. [Google Scholar] [CrossRef]
- Benyahya, S.; Monnier, F.; Taillefer, M.; Man, M.W.C.; Bied, C.; Ouazzani, F. Efficient and Versatile Sol–Gel Immobilized Copper Catalyst for Ullmann Arylation of Phenols. Adv. Synth. Catal. 2008, 350, 2205–2208. [Google Scholar] [CrossRef]
- Antilla, J.C.; Klapars, A.; Buchwald, S.L. The copper-catalyzed N-arylation of indoles. J. Am. Chem. Soc. 2002, 124, 11684–11688. [Google Scholar] [CrossRef] [PubMed]
- Naidu, A.B.; Jaseer, E.A.; Sekar, G. General, Mild, and Intermolecular Ullmann-Type Synthesis of Diaryl and Alkyl Aryl Ethers Catalyzed by Diol–Copper(I) Complex. J. Org. Chem. 2009, 74, 3675–3679. [Google Scholar] [CrossRef] [PubMed]
- Gujadhur, R.; Venkataraman, D.; Kintigh, J.T. Formation of arylnitrogen bonds using a soluble copper(I) Catalyst. Tetrahedron Lett. 2001, 42, 4791–4793. [Google Scholar] [CrossRef]
- Klapars, A.; Huang, X.; Buchwald, S.L. A General and Efficient Copper Catalyst for the Amidation of Aryl Halides. J. Am. Chem. Soc. 2002, 124, 7421–7428. [Google Scholar] [CrossRef] [PubMed]
- Burgos, C.H.; Barder, T.E.; Huang, X.; Buchwald, S.L. Significantly Improved Method for the Pd-Catalyzed Coupling of Phenols with Aryl Halides: Understanding Ligand Effects. Angew. Chem. Int. Ed. 2006, 118, 4427–4432. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, J.; Liu, M.; Zheng, X.; Ding, J.; Wu, H. Ligand-free copper-catalyzed coupling of nitroarenes with arylboronic acids. Green Chem. 2012, 14, 912–916. [Google Scholar] [CrossRef]
- Chen, J.; Wang, X.; Zheng, X.; Ding, J.; Liu, M.; Wu, H. Ligand-free copper-catalyzed O-arylation of nitroarenes with phenols. Tetrahedron 2012, 68, 8905–8907. [Google Scholar] [CrossRef]
- Satish, G.; Harsha, K.; Reddy, V.; Ramesh, K.; Anil Kulmar, B.S.P.; Nageswar, Y.V.D. An elegant protocol for the synthesis of N-substituted pyrroles through C–N cross coupling/aromatization process using CuFe2O4 nanoparticles as catalyst under ligand-free conditions. Tetrahedron Lett. 2014, 55, 2596–2599. [Google Scholar] [CrossRef]
- Zhou, H.; Long, J.; Yaghi, O. Introduction to Metal–Organic Frameworks. Chem. Rev. 2012, 112, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Corma, A.; García, H.; Llabrés i Xamena, F.X. Engineering Metal Organic Frameworks for Heterogeneous Catalysis. Chem. Rev. 2010, 110, 4606–4655. [Google Scholar] [CrossRef] [PubMed]
- Zou, R.Q.; Sakurai, H.; Xu, Q. Preparation, adsorption properties, and catalytic activity of 3D porous metal-organic frameworks composed of cubic building blocks and alkali-metal ions. Angew. Chem. Int. Ed. Engl. 2006, 45, 2542–2546. [Google Scholar] [CrossRef] [PubMed]
- Gándara, F.; Gómez-Lor, B.; Gutiérrez-Puebla, E.; Iglesias, M.; Monge, M.A.; Proserpio, D.M.; Snejkoal, N. An Indium Layered MOF as Recyclable Lewis Acid Catalyst. Chem. Mater. 2008, 20, 72–76. [Google Scholar] [CrossRef]
- Valvekens, P.; Vermoortele, F.; De Vos, D. Metal–organic frameworks as catalysts: The role of metal active sites. Catal. Sci. Technol. 2013, 3, 1435–1445. [Google Scholar] [CrossRef]
- Schröder, F.; Esken, D.; Cokoja, M.; van den Berg, M.W.E.; Lebedev, O.I.; van Ten-deloo, G.; Walaszek, B. Ruthenium Nanoparticles inside Porous [Zn4O(bdc)3] by Hydrogenolysis of Adsorbed [Ru(cod)(cot)]: A Solid-State Reference System for Surfactant-Stabilized Ruthenium Colloids. J. Am. Chem. Soc. 2008, 130, 6119–6130. [Google Scholar] [CrossRef] [PubMed]
- Phan, N.T.S.; Nguyen, T.T.; Nguyen, V.T.; Nguyen, K.D. Ligand-Free Copper-Catalyzed Coupling of Phenols with Nitroarenes by using a Metal–Organic Framework as a Robust and Recoverable Catalyst. ChemCatChem 2013, 5, 2374–2381. [Google Scholar] [CrossRef]
- Maity, T.; Saha, D.; Das, S.; Bhunia, S.; Koner, S. Ligand free copper-catalyzed heterogeneous O-arylation reaction under green condition. Catal. Commun. 2015, 58, 141–148. [Google Scholar] [CrossRef]
- Llabres i Xamena, F. Metal Organic Frameworks as Heterogeneous Catalysts; Gascón, J.F., Ed.; Royal Society of Chemistry: Cambridge, UK, 2013; ISBN 978-1-84973-572-8. [Google Scholar]
- Calleja, G.; Sanz, R.; Orcajo, G.; Briones, D.; Leo, P.; Martínez, F. Copper-based MOF-74 material as effective acid catalyst in Friedel–Crafts acylation of anisole. Catal. Today 2014, 227, 130–137. [Google Scholar] [CrossRef]
- Sanz, R.; Martínez, F.; Orcajo, G.; Wojtas, L.; Briones, D. Synthesis of a honeycomb-like Cu-based metal–organic framework and its carbon dioxide adsorption behavior. Dalton Trans. 2013, 42, 2392–2398. [Google Scholar] [CrossRef] [PubMed]
- Rosi, N.; Kim, J.; Eddaoudi, M.; Chen, B.; O’Keeffe, M.; Yaghi, O. Rod Packings and Metal–Organic Frameworks Constructed from Rod-Shaped Secondary Building Units. J. Am. Chem. Soc. 2005, 127, 1504–1518. [Google Scholar] [CrossRef] [PubMed]
- Boger, D.L.; Nomoto, Y.; Teegarden, B.R. Vancomycin and ristocetin models: Synthesis via the Ullmann macrocyclization reaction. J. Org. Chem. 1993, 58, 1425–1433. [Google Scholar] [CrossRef]
- Jogdand, N.R.; Shingate, B.B.; Shingare, M.S. Selective reduction of 4h-1,3-thiazine-4-ones: Easy access to substituted 6h-1,3-thiazines. Tetrahedron Lett. 2009, 50, 4019–4021. [Google Scholar] [CrossRef]
- Kidwai, M.; Mishra, N.K.; Bansal, V.; Kumar, A.; Mozumdar, S. Cu-nanoparticle catalyzed O-arylation of phenols with aryl halides via Ullmann coupling. Tetrahedron Lett. 2007, 48, 8883–8887. [Google Scholar] [CrossRef]
- Bistri, O.; Correa, A.; Bolm, C. Iron-Catalyzed C–O Cross-Couplings of Phenols with Aryl Iodides. Angew. Chem. Int. Ed. 2008, 47, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.W.W.; Chee, S.; Mak, S.; Buranaprasertsuk, P.; Chavasiri, W.; Chan, P.W.H. Approaches towards the total synthesis of carolacton: Synthesis of C1–C16 fragment. Tetrahedron Lett. 2008, 49, 2018–2022. [Google Scholar] [CrossRef]
- Huang, W.H.; Luan, X.J.; Zhou, X.; Chen, J.; Wang, Y.Y.; Shi, Q.Z. The influence of ligand configuration, solvent size and solvent polarity on the porous shape and void volume in a series of isomeric or isomorphic porous MOFs. CrystEngComm 2013, 15, 10389–10398. [Google Scholar] [CrossRef]
- Mann, G.; Incarvito, C.; Rheingold, A.L.; Hartwig, J.F. Palladium-Catalyzed C–O Coupling Involving Unactivated Aryl Halides. Sterically Induced Reductive Elimination To Form the C–O Bond in Diaryl Ethers. J. Am. Chem. Soc. 1999, 121, 3224–3225. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, Z.; Ding, Y. Proposal for halogen atom transfer mechanism for Ullmann O-arylation of phenols with aryl halides. Dalton Trans. 2012, 41, 13832–13840. [Google Scholar] [CrossRef] [PubMed]
- Ruano, D.; Diaz-Garcia, M.; Alfayate, A.; Sanchez-Sanchez, M. Nanocrystalline M–MOF-74 as Heterogeneous Catalysts in the Oxidation of Cyclohexene: Correlation of the Activity and Redox Potential. ChemCatChem 2015, 7, 674–681. [Google Scholar] [CrossRef]
- Diaz-Garcia, M.; Mayoral, A.; Díaz, I.; Sánchez-Sánchez, M. Nanoscaled M-MOF-74 Materials Prepared at Room Temperature. Cryst. Growth Des. 2014, 14, 2479–2487. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.A.; Alvaro, M.; Hwang, Y.K.; Seo, Y.K.; Corma, A.; Garcia, H. Intracrystalline diffusion in Metal Organic Framework during heterogeneous catalysis: Influence of particle size on the activity of MIL-100 (Fe) for oxidation reactions. Dalton Trans. 2011, 40, 10719–10724. [Google Scholar] [CrossRef] [PubMed]
- Brunauer, S.; Emmett, P.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Jagiello, J.; Thommes, M. Comparison of DFT characterization methods based on N2, Ar, CO2, and H2 adsorption applied to carbons with various pore size distributions. Carbon 2004, 42, 1227–1232. [Google Scholar] [CrossRef]
- Lippens, B.C.; de Boer, J.H. Studies on pore systems in catalysts: V. The t method. J. Catal. 1965, 4, 319–323. [Google Scholar] [CrossRef]
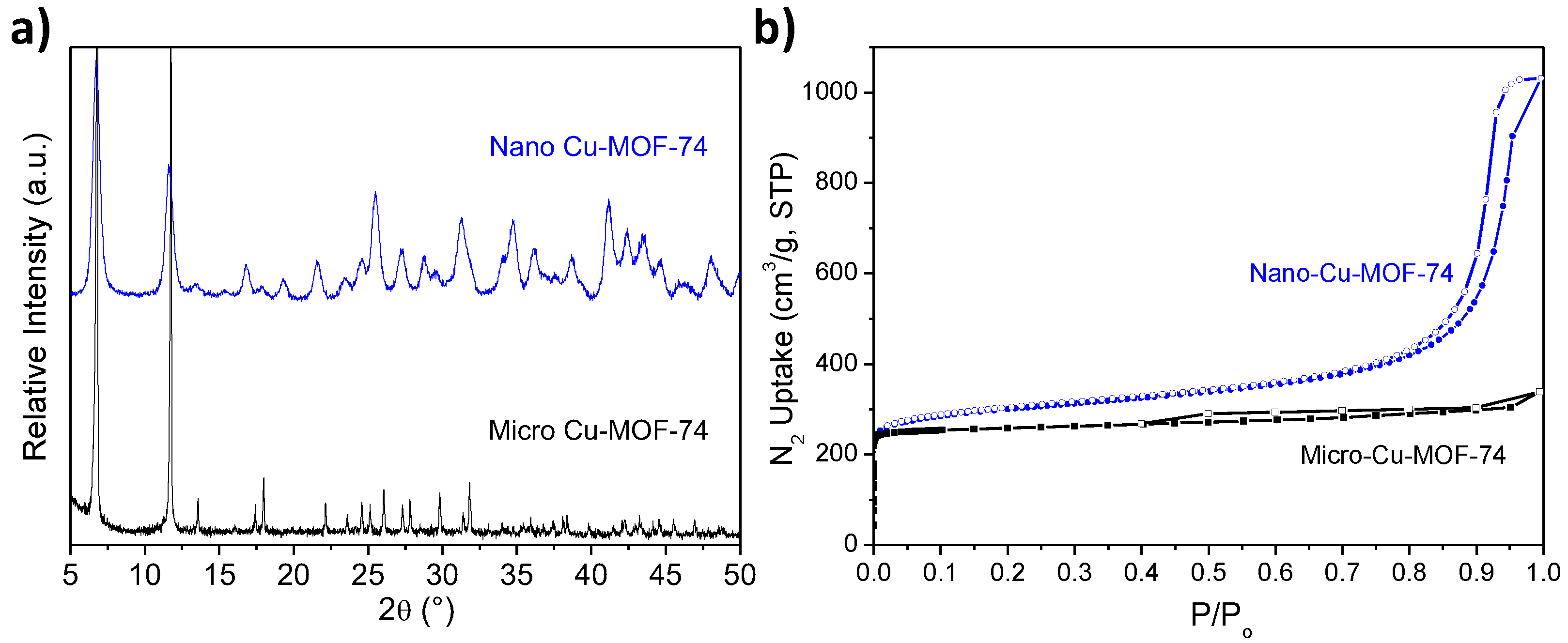
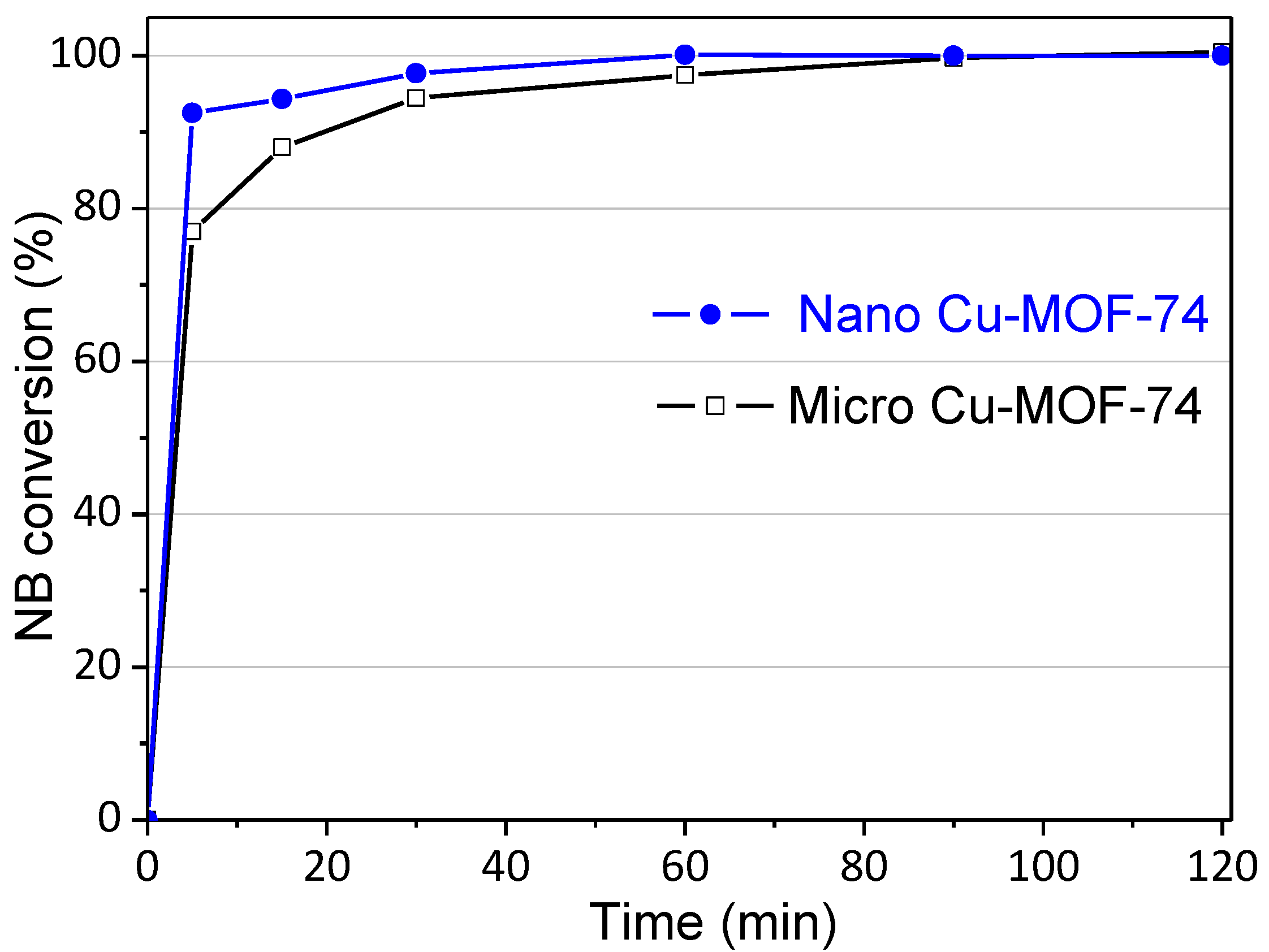
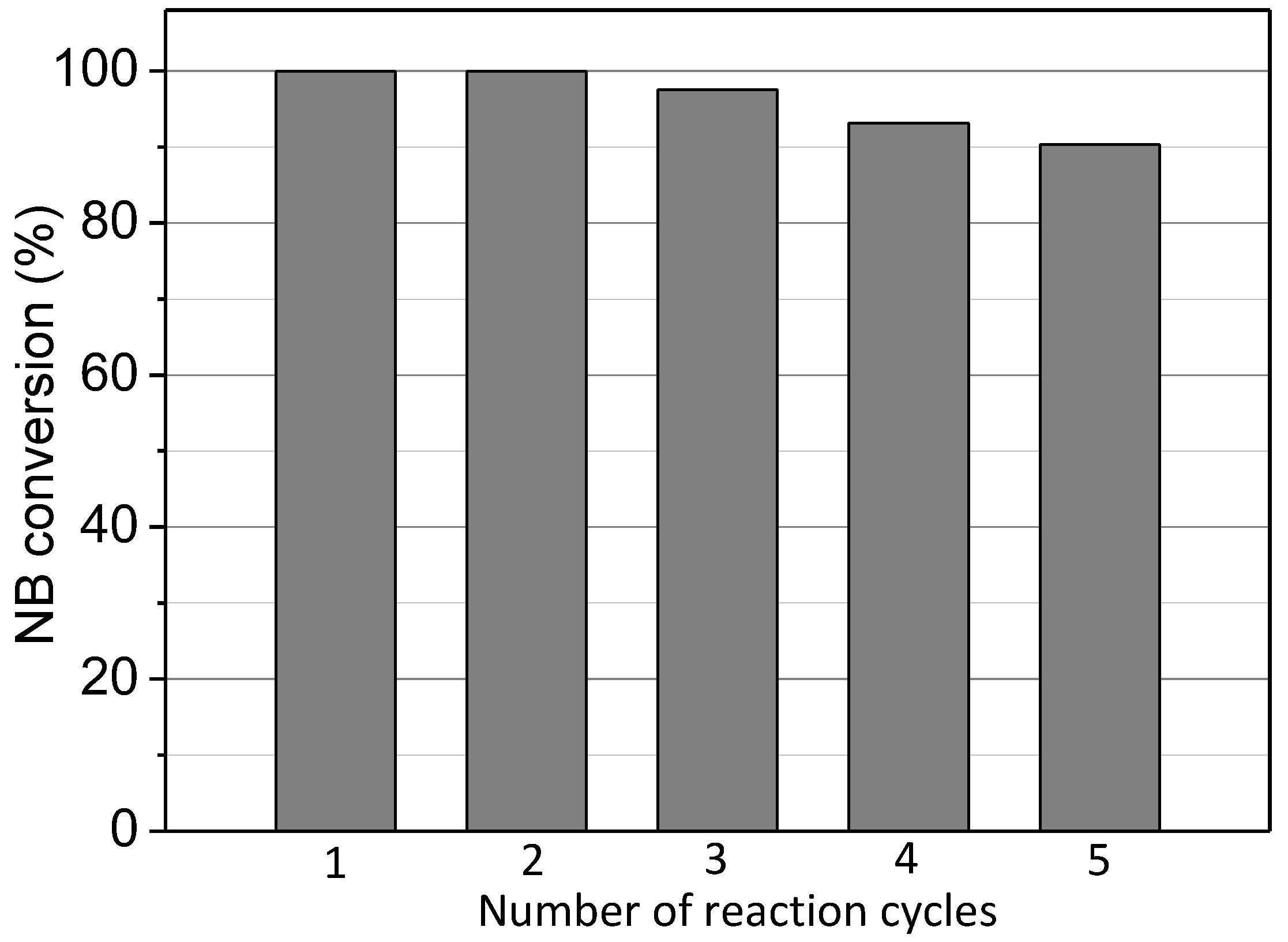
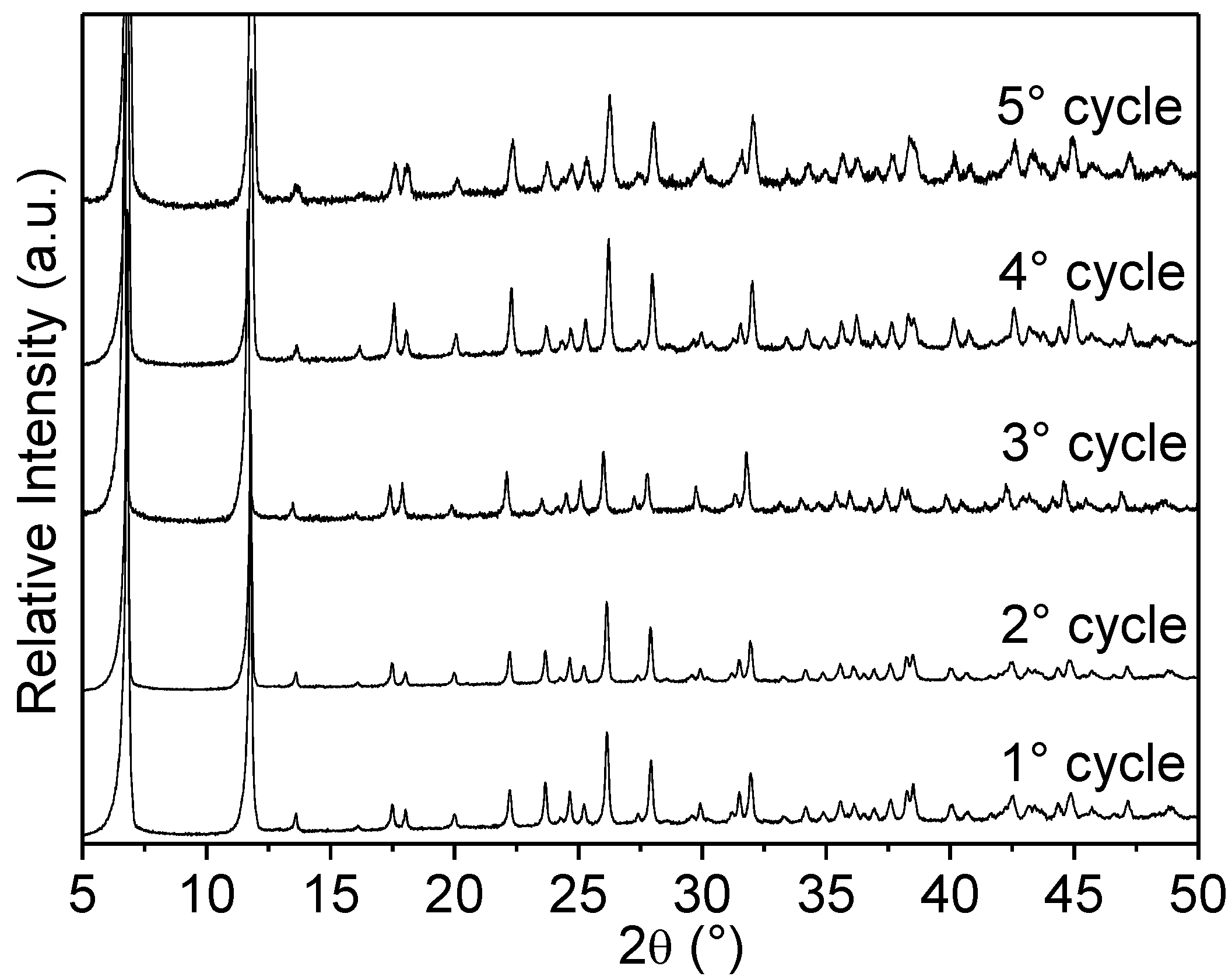
| Cu-MOF-74 | Crystal Size (nm) | SBET (m2/g) | SExternal c (m2/g) |
|---|---|---|---|
| Nanocrystalline | 14 a | 1103 | 286 |
| Microcrystalline | 10,000 b | 1126 | 124 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leo, P.; Orcajo, G.; Briones, D.; Calleja, G.; Sánchez-Sánchez, M.; Martínez, F. A Recyclable Cu-MOF-74 Catalyst for the Ligand-Free O-Arylation Reaction of 4-Nitrobenzaldehyde and Phenol. Nanomaterials 2017, 7, 149. https://doi.org/10.3390/nano7060149
Leo P, Orcajo G, Briones D, Calleja G, Sánchez-Sánchez M, Martínez F. A Recyclable Cu-MOF-74 Catalyst for the Ligand-Free O-Arylation Reaction of 4-Nitrobenzaldehyde and Phenol. Nanomaterials. 2017; 7(6):149. https://doi.org/10.3390/nano7060149
Chicago/Turabian StyleLeo, Pedro, Gisela Orcajo, David Briones, Guillermo Calleja, Manuel Sánchez-Sánchez, and Fernando Martínez. 2017. "A Recyclable Cu-MOF-74 Catalyst for the Ligand-Free O-Arylation Reaction of 4-Nitrobenzaldehyde and Phenol" Nanomaterials 7, no. 6: 149. https://doi.org/10.3390/nano7060149
APA StyleLeo, P., Orcajo, G., Briones, D., Calleja, G., Sánchez-Sánchez, M., & Martínez, F. (2017). A Recyclable Cu-MOF-74 Catalyst for the Ligand-Free O-Arylation Reaction of 4-Nitrobenzaldehyde and Phenol. Nanomaterials, 7(6), 149. https://doi.org/10.3390/nano7060149