Influence of Quantum Dot Concentration on Carrier Transport in ZnO:TiO2 Nano-Hybrid Photoanodes for Quantum Dot-Sensitized Solar Cells
Abstract
:1. Introduction
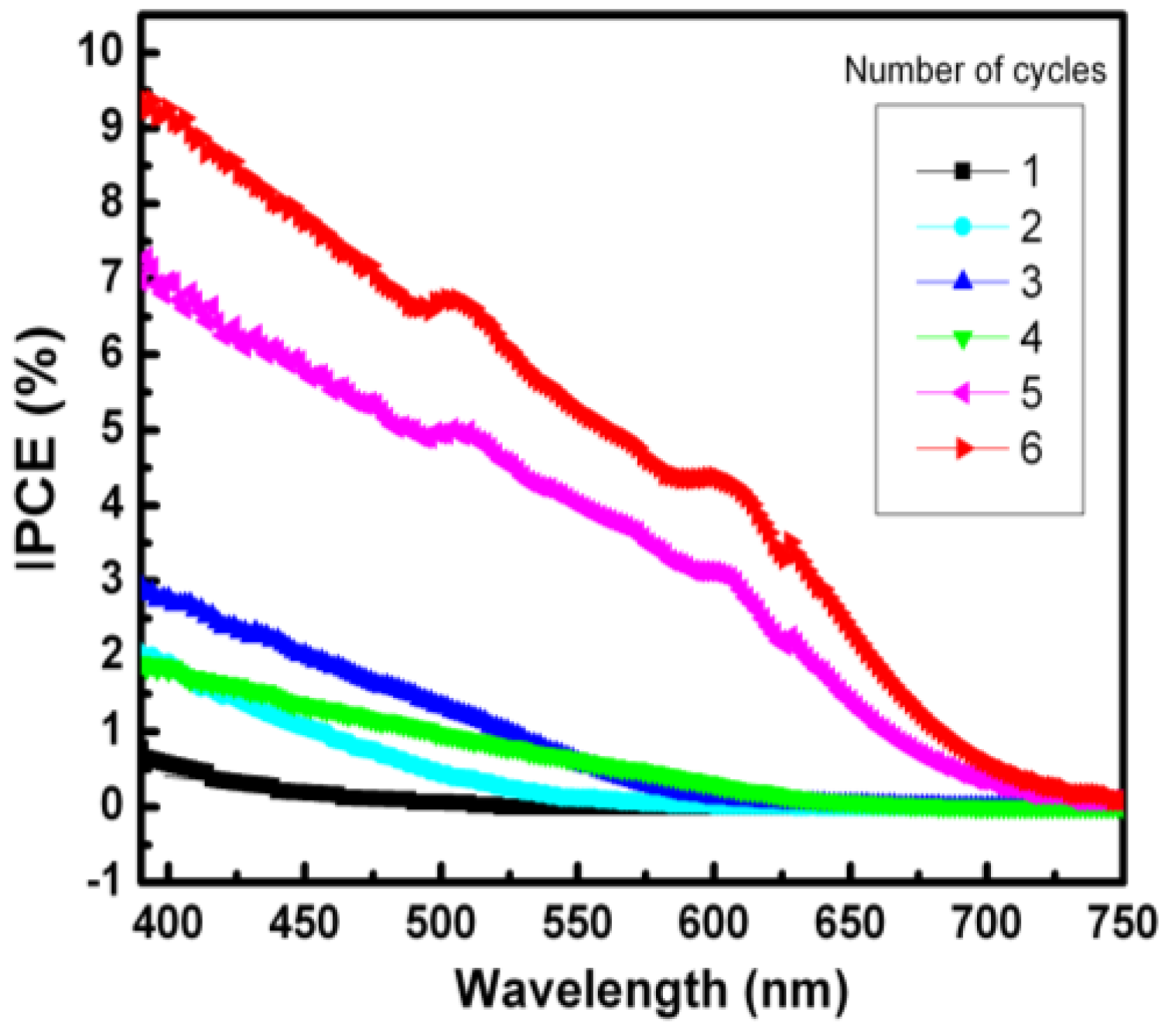
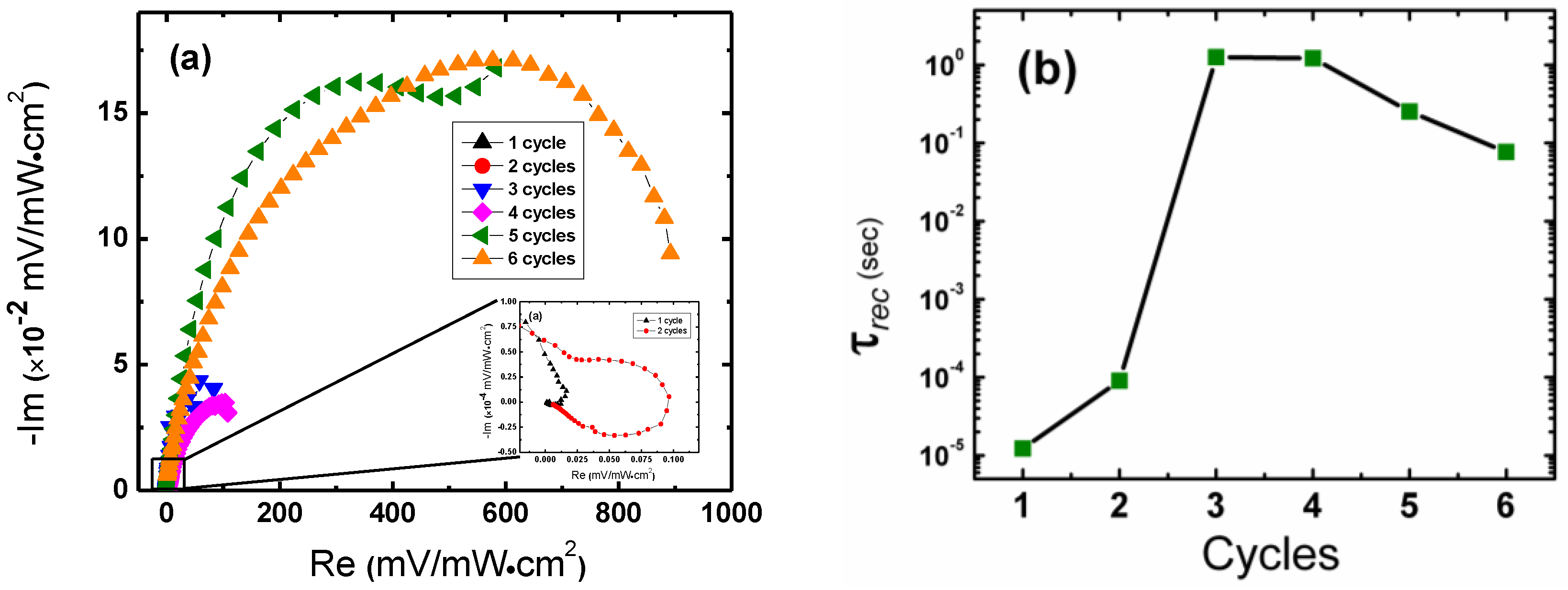
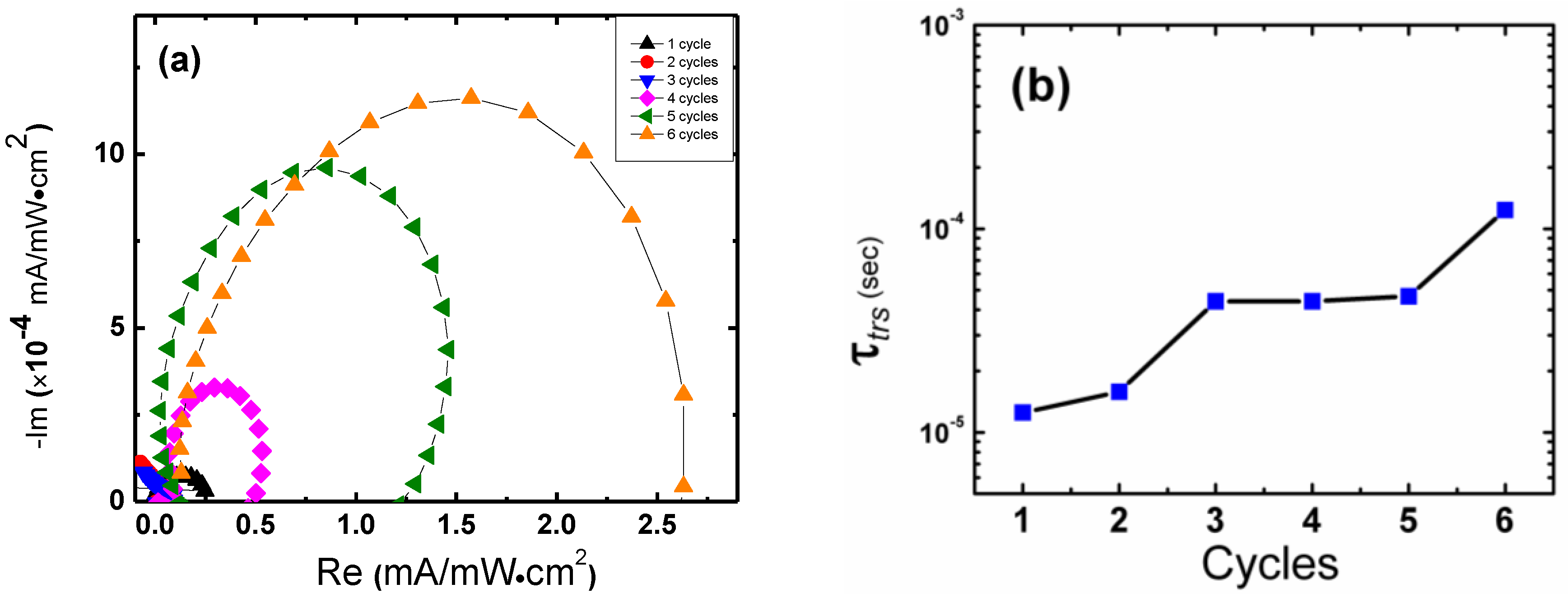
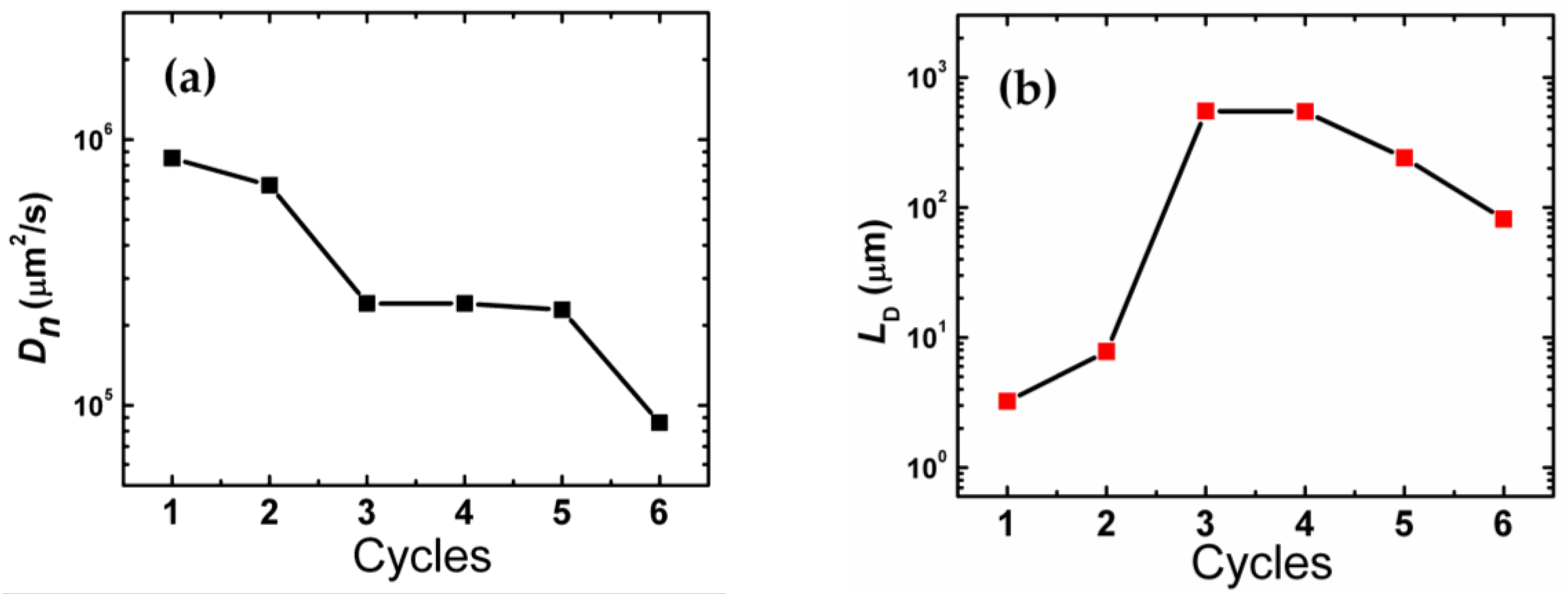
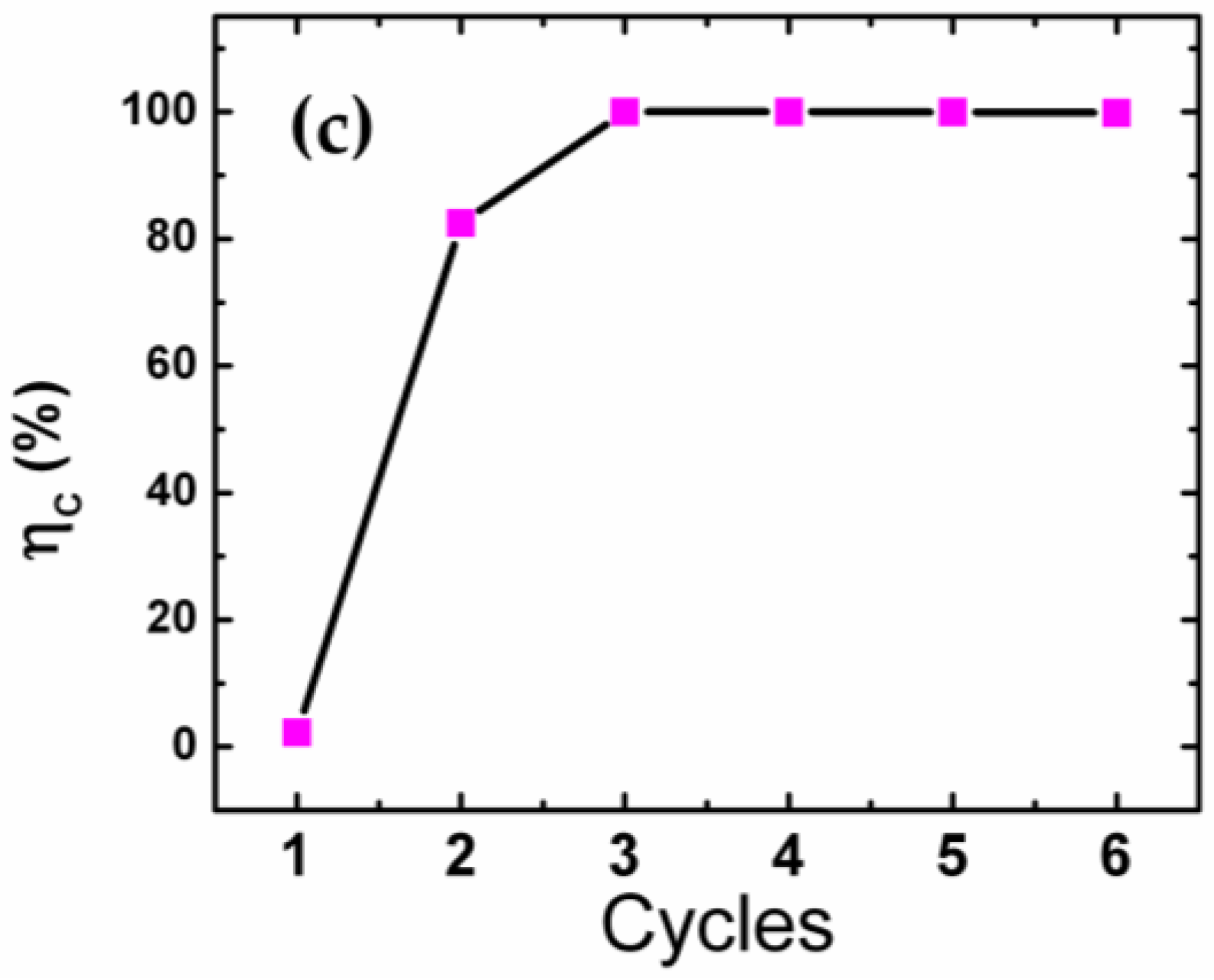
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- O’Regan, B.; Grätzel, M. Photochemical method for the conversion of light to chemical into energy. Nature 1991, 353, 737–747. [Google Scholar]
- Kamat, P.V. Quantum dot solar cells. Semiconductor nanocrystals as light harvesters. J. Phys. Chem. C 2008, 112, 18737–18753. [Google Scholar] [CrossRef]
- Diguna, L.J.; Shen, Q.; Kobayashi, J.; Toyoda, T. High efficiency of CdSe quantum-dot-sensitized TiO2 inverse opal solar cells. Appl. Phys. Lett. 2007, 91. [Google Scholar] [CrossRef]
- Dai, Q.; Sabio, E.M.; Wang, W.; Wang, J. Pulsed laser deposition of Mn doped CdSe quantum dots for improved solar cell performance. Appl. Phys. Lett. 2014, 104. [Google Scholar] [CrossRef]
- Lu, L.; Chen, J.; Wang, W. Wide bandgap Zn2GeO4 nanowires as photoanode for quantum dot sensitized solar cells. Appl. Phys. Lett. 2013, 103. [Google Scholar] [CrossRef]
- Lee, H.; Wang, M.; Chen, P.; Gamelin, D.R.; Zakeeruddin, S.M.; Gratzel, M.; Nazeeruddin, M.K. Efficient CdSe quantum dot-sensitized solar cells prepared by an improved successive ionic layer adsorption and reaction process. Nano Lett. 2009, 9. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Zhu, Y. Synthesis of CdS nanocrystals based on low-temperature thermolysis of one single-source organometallic precursor. Nanotechnology 2006, 17, 845–851. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Battaglia, D.; Blackman, B.; Wang, Y.; Peng, X.G. Coupled and decoupled dual quantum systems in one semiconductor nanocrystal. J. Am. Chem. Soc. 2005, 127, 10889–10897. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Zhang, H.; Hu, S.; Li, J. Preparation and enhanced photoelectrochemical performance of coupled bicomponent ZnO-TiO2 nanocomposites. J. Phys. Chem. C 2008, 112, 117–122. [Google Scholar] [CrossRef]
- Marcí, G.; Augugliaro, V.; López-Muñoz, M.J.; Martín, C.; Palmisano, L.; Rives, V.; Shciavello, M.; Tilley, R.J.D.; Venezia, A.M. Preparation characterization and photocatalytic activity of polycrystalline ZnO/TiO2 systems. J. Phys. Chem. B 2001, 105, 1033–1040. [Google Scholar] [CrossRef]
- Conesa, J.C. Band structures and nitrogen doping effects in zinc titanate photocatalysts. Catal. Today 2013, 208, 11–18. [Google Scholar] [CrossRef]
- Karunakaran, C.; Dhanalakshmi, R.; Gomathisankar, P.; Manikandan, G. Enhanced phenol-photodegradation by particulate semiconductor mixtures: Interparticle electron-jump. J. Haz. Mat. 2010, 176, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, X.; Tai, Q.; Hu, H.; Chen, B.; Huang, N.; Sebo, B.; Zhao, X.Z. Efficiency enhancement in dye-sensitized solar cells by interfacial modification of conducting glass/mesoporous TiO2 using a novel ZnO compact blocking film. J. Power Sources 2011, 196, 475–481. [Google Scholar] [CrossRef]
- Zhang, Q.; Dandenau, C.S.; Zhou, X.; Cao, G. ZnO Nanostructures for Dye-Sensitized Solar Cells. Adv. Mater. 2009, 21, 4087–4108. [Google Scholar] [CrossRef]
- Law, M.; Greene, L.E.; Radenovic, A.; Kuykendall, T.; Liphardt, J.; Yang, P. ZnO-Al2O3 and ZnO-TiO2 core-shell nanowire dye-sensitized solar cells. J. Phys. Chem. B 2006, 110, 22652–22663. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Neale, N.R.; Miedaner, A.; Frank, A.J. Enhanced charge-collection efficiencies and light scattering in dye-sensitized solar cells using oriented TiO2 nanotube arrays. Nano Lett. 2007, 7, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, W. Carrier transport processes in dye sensitized solar cells based on Zn2SnO4 nanostructures studied by intensity modulated photocurrent/photovoltage spectroscopy. Appl. Phys. Lett. 2013, 102. [Google Scholar] [CrossRef]
- Peter, L.M.; Wijayantha, K.G.U. Intensity dependence of the electron diffusion length in dye-sensitized nanocrystalline TiO2 photovoltaic cells. Electrochem. Comm. 1999, 1, 576–580. [Google Scholar] [CrossRef]
- van de Lagemaat, J.; Park, N.G.; Frank, A.J. Influence of electrical potential distribution, charge transport, and recombination on the photopotential and photocurrent conversion efficiency of dye-sensitized nanocrystalline TiO2 solar cells: A study by electrical impedance and optical modulation techniques. J. Phys. Chem. B 2000, 104, 2044–2052. [Google Scholar]
- Pesika, N.S.; Stebe, K.J.; Searson, P.C. Determination of the particle size distribution of quantum nanocrystals from absorbance spectra. Adv. Mater. 2003, 15, 1289–1291. [Google Scholar] [CrossRef]
- Hagfeldt, A.; Grätzel, M. Light-induced redox reaction in nanocrystalline systems. Chem. Rev. 1995, 95, 49–68. [Google Scholar] [CrossRef]
- Zhang, Q.; Chou, T.P.; Russo, B.; Jenekhe, S.A.; Cao, G. Aggregation of ZnO Nanocrystallites for High Conversion Efficiency in Dye-Sensitized Solar Cells. Agnew. Chem. 2008, 120, 2436–2440. [Google Scholar] [CrossRef]
- Liu, C.; Qui, Z.; Yue, W.; Zhou, Z.; Wang, M. Characterization of Photocurrent Generation Dynamics in Polymer Solar Cells Based on ZnO/CdS-Core/Shell Nanoarrays by Intensity Modulated Photocurrent Spectroscopy: Theoretical Modeling. J. Phys. Chem. C 2014, 118, 28462–28473. [Google Scholar] [CrossRef]
- Halme, J.; Miettunen, K.; Lund, P. Effect of nonuniform generation and inefficient collection of electrons on the dynamic photocurrent and photovoltage response of nanostructured photoelectrodes. J. Phys. Chem. C 2008, 112, 20491–20504. [Google Scholar] [CrossRef]
- Halme, J.; Boschloo, G.; Hagfeldt, A.; Lund, P. Spectral characteristics of light harvesting, electron injection, and steady-state charge collection in pressed TiO2 dye solar cells. J. Phys. Chem. C 2008, 112, 5623–5637. [Google Scholar] [CrossRef]
- Jun, H.K.; Careem, M.A.; Arof, A.K. Quantum dot-sensitized solar cells–perspective and recent developments: A review of Cd chalcogenide quantum dots as sensitizers. Renew. Sust. Energ. Rev. 2013, 22, 148–167. [Google Scholar] [CrossRef]
- Hossain, M.A.; Jennings, J.R.; Koh, Z.Y.; Wang, Q. Carrier generation and collection in CdS/CdSe-sensitized SnO2 solar cells exhibiting unprecedented photocurrent densities. Am. Chem. Soc. Nano 2011, 5, 3172–3181. [Google Scholar]
- Santra, P.K.; Kamat, P.V. Mn-doped quantum dot sensitized solar cells: A strategy to boost efficiency over 5%. J. Am. Chem. Soc. 2012, 134, 2508–2511. [Google Scholar] [CrossRef] [PubMed]
- Tvrdy, K.; Frantsuzov, P.; Kamat, P.V. Photoinduced electron transfer from semiconductor quantum dots to metal oxide nanoparticles. Proc. Nat. Acad. Sci. USA 2011, 108, 29–30. [Google Scholar] [CrossRef] [PubMed]
- Guijarro, N.; Shen, Q.; Giménez, S.; Mora-Seró, I.; Bisquert, J.; Lana-Villarreal, T.; Toyodo, T.; Gómez, R. Direct correlation between ultrafast injection and photoanode performance in quantum dot sensitized solar cells. J. Phys. Chem. C 2010, 114, 22352–22360. [Google Scholar] [CrossRef]
- Hodes, G. Comparison of dye-and semiconductor-sensitized porous nanocrystalline liquid junction solar cells. J. Phys. Chem. C 2008, 112, 17778–17787. [Google Scholar] [CrossRef]
- Gujarro, N.; Lana-Villareal, T.; Shen, Q.; Toyoda, T.; Gómez, R. Sensitization of titanium dioxide photoanodes with cadmium selenide quantum dots prepared by SILAR: Photoelectrochemical and carrier dynamics. J. Phys. Chem. C 2010, 114, 21928–21937. [Google Scholar] [CrossRef]
- Yeh, M.H.; Lee, C.P.; Chou, C.Y.; Lin, L.Y.; Wei, H.Y.; Chu, C.W. Conducting polymer-based counter electrode for a quantum-dot-sensitized solar cell (QDSSC) with a polysulfide electrolyte. Electrochim. Acta 2011, 57, 277–284. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, W. A new polymer electrolyte for solid-state quantum dot sensitized solar cells. J. Power Sources 2015, 285, 70–75. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maloney, F.S.; Poudyal, U.; Chen, W.; Wang, W. Influence of Quantum Dot Concentration on Carrier Transport in ZnO:TiO2 Nano-Hybrid Photoanodes for Quantum Dot-Sensitized Solar Cells. Nanomaterials 2016, 6, 191. https://doi.org/10.3390/nano6110191
Maloney FS, Poudyal U, Chen W, Wang W. Influence of Quantum Dot Concentration on Carrier Transport in ZnO:TiO2 Nano-Hybrid Photoanodes for Quantum Dot-Sensitized Solar Cells. Nanomaterials. 2016; 6(11):191. https://doi.org/10.3390/nano6110191
Chicago/Turabian StyleMaloney, Francis S., Uma Poudyal, Weimin Chen, and Wenyong Wang. 2016. "Influence of Quantum Dot Concentration on Carrier Transport in ZnO:TiO2 Nano-Hybrid Photoanodes for Quantum Dot-Sensitized Solar Cells" Nanomaterials 6, no. 11: 191. https://doi.org/10.3390/nano6110191
APA StyleMaloney, F. S., Poudyal, U., Chen, W., & Wang, W. (2016). Influence of Quantum Dot Concentration on Carrier Transport in ZnO:TiO2 Nano-Hybrid Photoanodes for Quantum Dot-Sensitized Solar Cells. Nanomaterials, 6(11), 191. https://doi.org/10.3390/nano6110191





