Dealloying of Cu-Based Metallic Glasses in Acidic Solutions: Products and Energy Storage Applications
Abstract
:1. Introduction
2. Fabrication of NPC

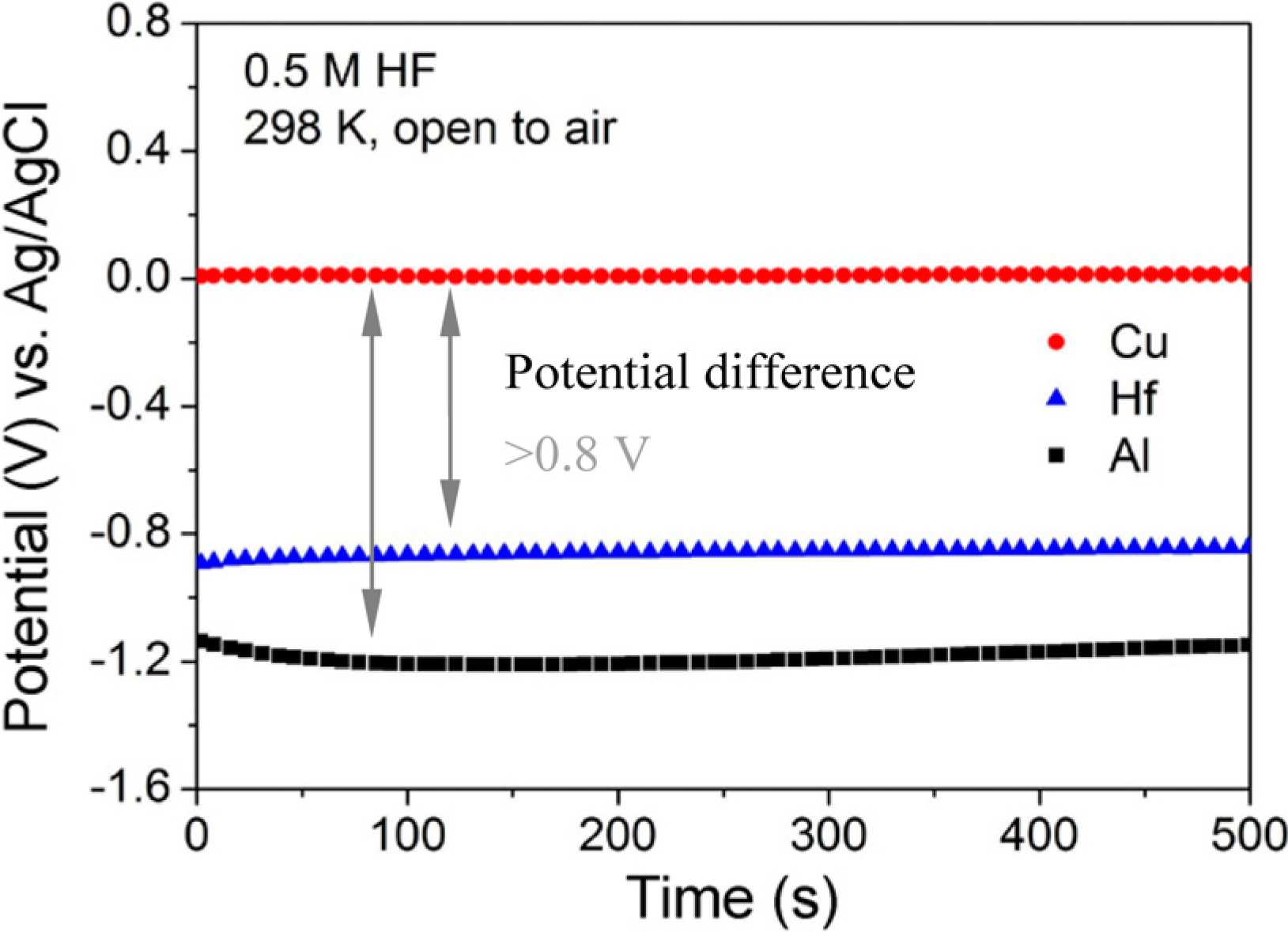
| Dealloying Solution | Precursors | References |
|---|---|---|
| HCl | Al70Cu18Mg12, (Al75Cu17Mg8)97Ni3 | [52,62] |
| H2SO4 | Mg90−xCuxY10 (x = 20, 25, 30, 40 at.%) | [49,56] |
| Mg65Cu25Gd10 | [50] | |
| H2SO4 + PVP | Mg65Cu25Y10 | [12] |
| HF | Cu52.5Hf40Al7.5 | [30] |
| Cu50Zr50−xAlx (x = 0, 5 at.%) | [31] | |
| Cu60Zr30Ti10 | [51] | |
| Ti100−xCux (x = 33, 40, 50, 60, 70 at.%) | [47,63,64] | |
| Ti–Cu–(Ag, Au, Ni, Pd, Pt) | [48,53,57,65,66,67] | |
| HF + PVP | Ti60Cu40 | [68] |
3. Synthesis of NPC/Metal Oxides Composites
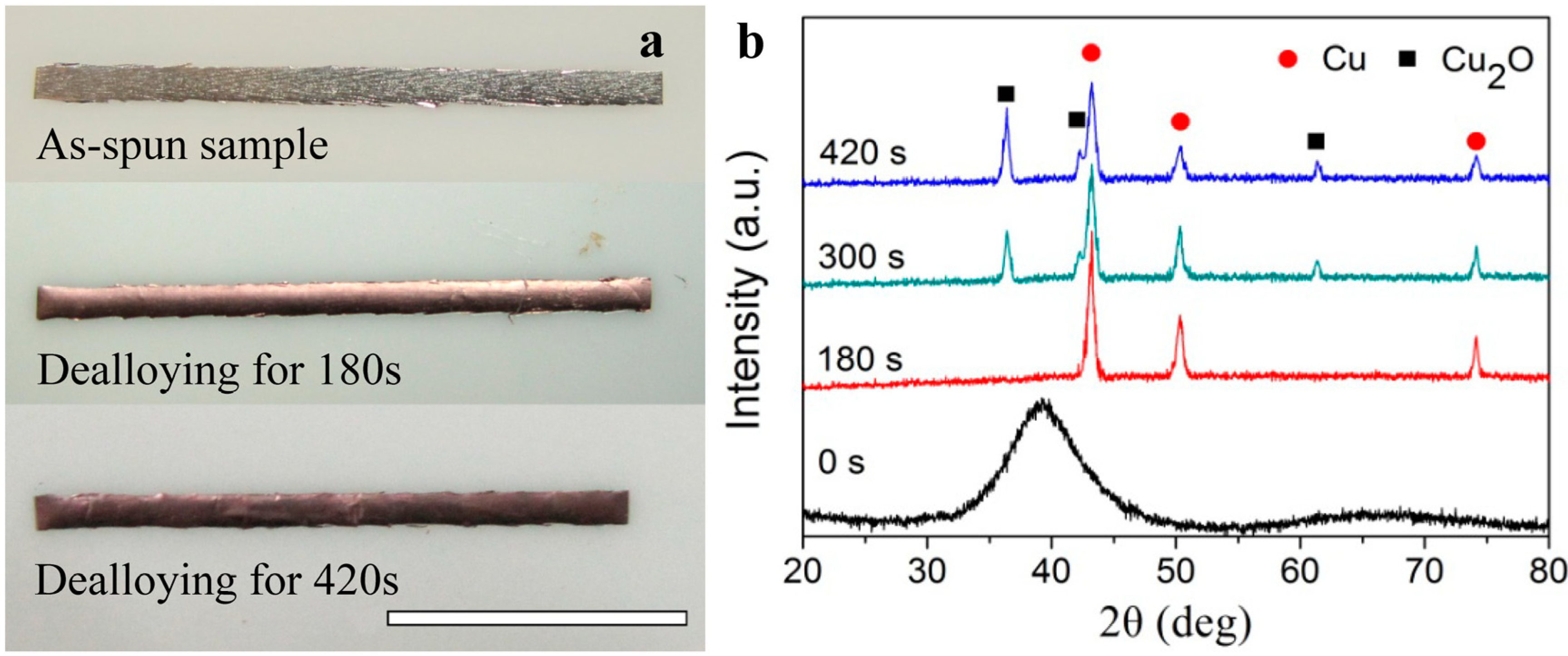


4. Synthesis of Cu2O Particles on Surface of MGs
4.1. Synthesis of Regular Cu2O Particles on Surface of MGs
| Dealloying Time/h | 4 | 5 | 6 | 8 | 14 | 20 | 24 |
| Morphology |  |  |  |  |  |  |  |
| Truncated tetrahedron | Cube | Cuboctahedron | Truncated octahedron | Octahedron | Hexapods | Octahedron-detached hexapods | |
| Edge length/nm | ~300 | ~300 | ~300 | ~150 | ~450 | ~500 | ~1100 |
| Volume fraction/% | 10.6 | 13.3 | 12.2 | 13.9 | 15.8 | 14.4 | 19.8 (mixed with other shapes) |
4.2. Preparation of Cu2O Micro-flowers on Surface of MGs

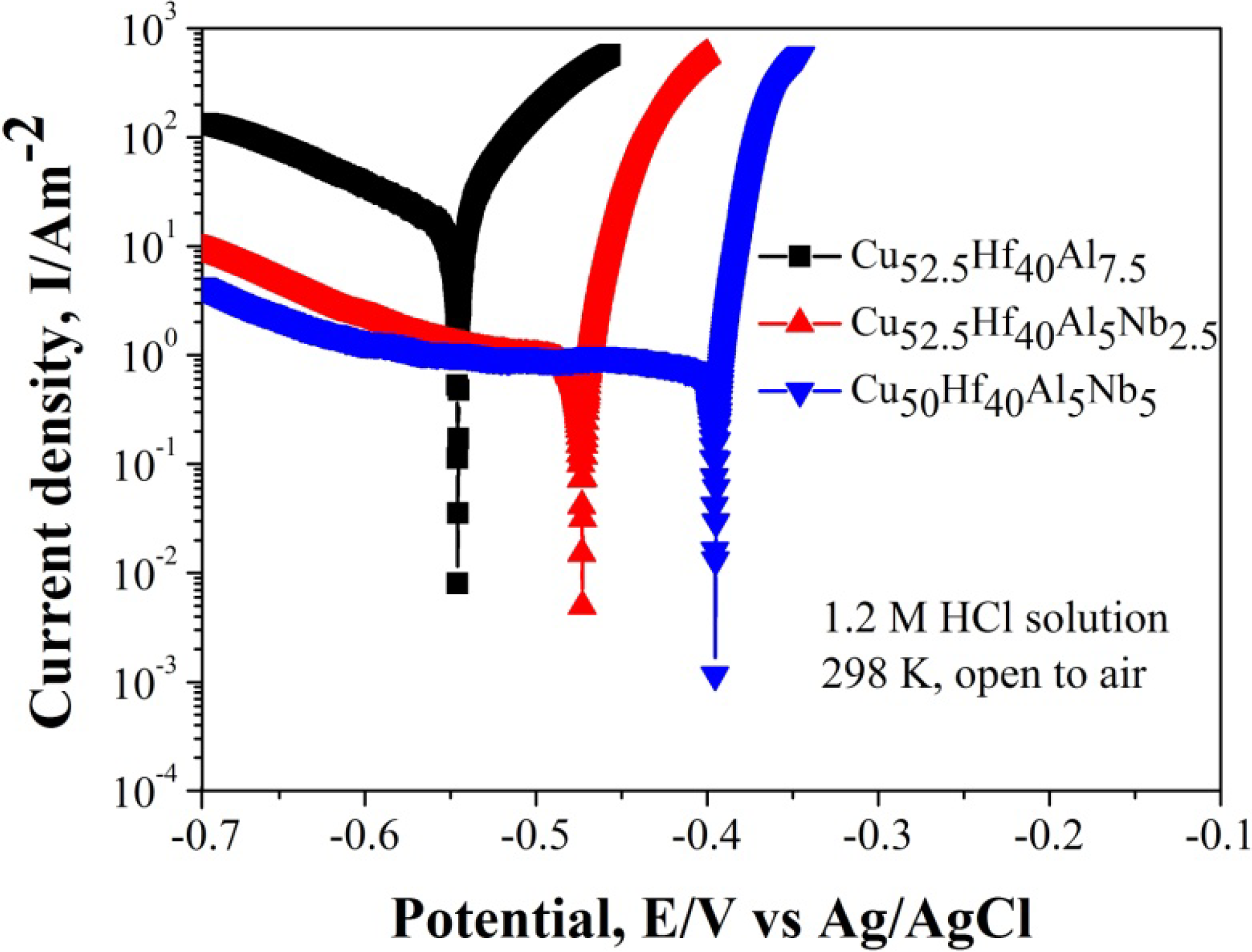
| Etching Solution | Dealloying Products | MG | Dealloying Conditions | References | |
|---|---|---|---|---|---|
| Inner | Surface | ||||
| HF | NPC | NPC | Cu52.5Hf40Al7.5 | 0.5 M HF, 300 s | [30] |
| NPC | Regular Cu2O particles | Cu52.5Hf40Al7.5 | oxygen-enriched 0.65 M HF, 420 s | This study | |
| HCl | MG | Regular Cu2O particles | Cu52.5Hf40Al7.5 | 0.05 M HCl, 4~24 h | [32,33] |
| MG | Cu2O micro-flowers | Cu50Hf40Al5Nb5 | 1.2 M HCl, 14 h | This study | |
5. Applications of Dealloying Products
| Products | Application Field | References |
|---|---|---|
| NPC | Surface-enhanced Raman scattering (SERS) | [22,89] |
| Immobilization of horseradish peroxidase | [84] | |
| NPC/Ag core–shell composite | SERS | [90] |
| NPC/Pt(Pd) core–shell composite | Methanol electro-oxidation | [91] |
| NPC/Au core-shell composite | Electrocatalysis and nonenzymatic biosensing | [87] |
| Ni-B amorphous nanoparticles modified NPC | Ethanol oxidation | [88,92] |
| NPC/Cu composite | Electro-oxidation of hydrazine | [85] |
| Nanoporous Cu–O system | Catalysts towards CO oxidation | [93] |
| NPC/Cu2O nanocomposite | Adsorption of methyl orange | [94] |
| NPC/MG composite | Degradation of azo dye | [86] |
| Degradation of phenol | [50] | |
| NPC/Si composite | Lithium-ion battery anodes | [95] |
| NPC/Cu2O composite | [70] | |
| Cu/NPC/MnO2 composite | [96] | |
| NPC/MnO2 composite | Supercapacitor electrodes | [30] |
| NPC/(Fe,Cu)3O4 composites | Excellent magnetic/electrical properties for potential applications in sensors, information storage, and so on | [76] |
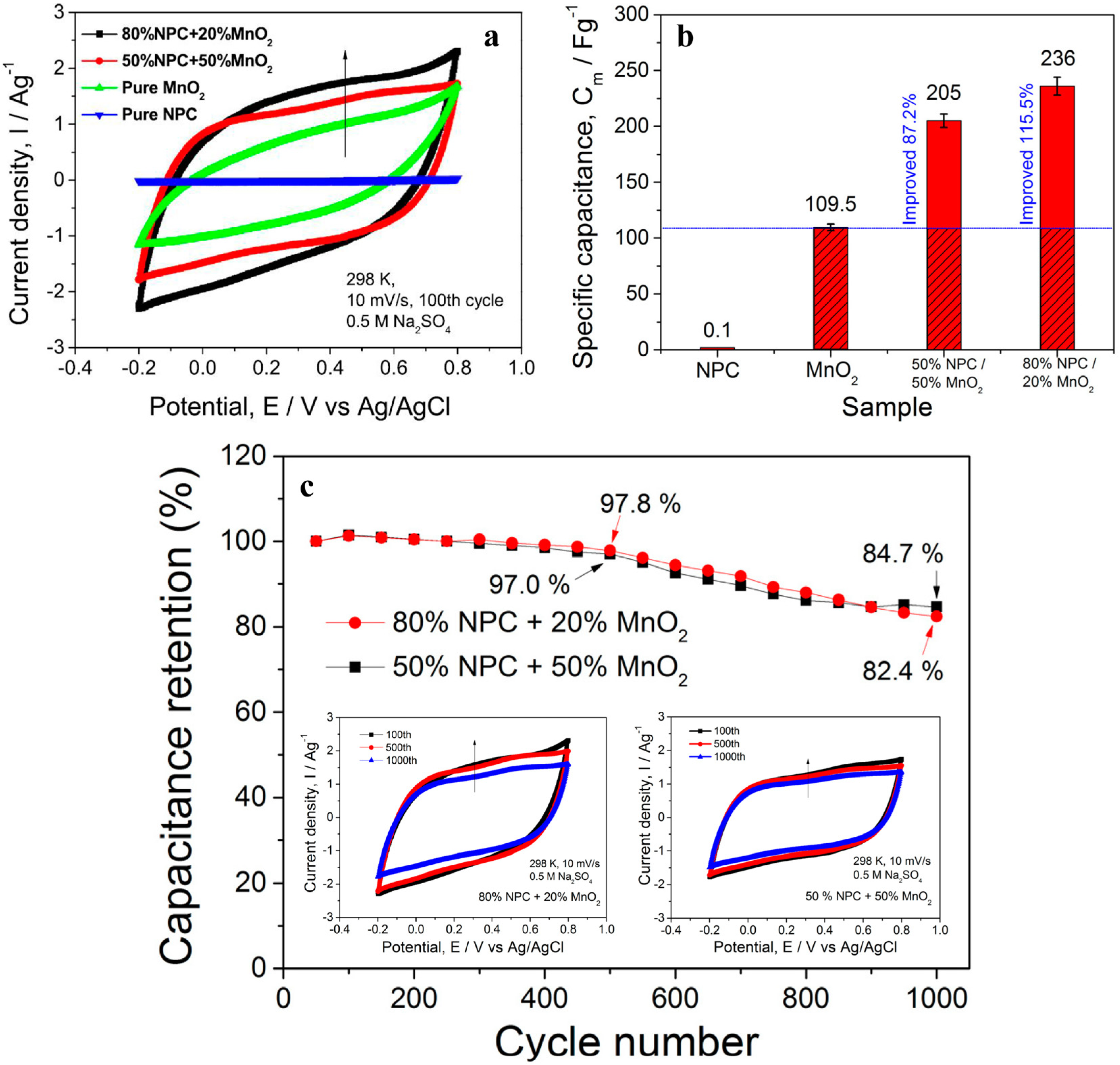
6. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, C.; Sun, J.; Xu, J.; Wang, X.; Ji, H.; Zhao, C.; Zhang, Z. Formation and microstructure of nanoporous silver by dealloying rapidly solidified Zn–Ag alloys. Electrochim. Acta 2012, 63, 302–311. [Google Scholar] [CrossRef]
- Darque-Ceretti, E.; Felder, E.; Aucouturier, M. Foil and leaf gilding on cultural artifacts: Forming and adhesion. Matéria 2011, 16, 540–559. [Google Scholar]
- Ding, Y.; Kim, Y.; Erlebacher, J. Nanoporous gold leaf: “Ancient technology”/advanced material. Adv. Mater. 2004, 16, 1897–1900. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Y.; Wang, J.; Geng, H.; Qiu, H. Nanoporous PdCu alloy for formic acid electro-oxidation. J. Power Sources 2012, 199, 124–131. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Y.; Zhou, C.; Wang, L.; Geng, H.; Ding, Y. An in situ dealloying and oxidation route to Co3O4 nanosheets and their ambient-temperature CO oxidation activity. ChemCatChem 2011, 3, 399–407. [Google Scholar] [CrossRef]
- Lang, X.; Fu, H.; Hou, C.; Han, G.; Yang, P.; Liu, Y.; Jiang, Q. Nanoporous gold supported cobalt oxide microelectrodes as high-performance electrochemical biosensors. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Pickering, H.; Wagner, C. Electrolytic dissolution of binary alloys containing a noble metal. J. Electrochem. Soc. 1967, 114, 698–706. [Google Scholar] [CrossRef]
- Seker, E.; Reed, M.; Begley, M. Nanoporous gold: Fabrication, characterization, and applications. Materials 2009, 2, 2188–2215. [Google Scholar] [CrossRef]
- Hakamada, M.; Nakano, H.; Furukawa, T.; Takahashi, M.; Mabuchi, M. Hydrogen storage properties of nanoporous palladium fabricated by dealloying. J. Phys. Chem. C 2010, 114, 868–873. [Google Scholar] [CrossRef]
- Jin, H.; Kramer, D.; Ivanisenko, Y.; Weissmüller, J. Macroscopically strong nanoporous Pt prepared by dealloying. Adv. Eng. Mater. 2007, 9, 849–854. [Google Scholar] [CrossRef]
- Li, Z.; Wang, X.; Lu, X. Refinement of nanoporous silver by adding surfactant to the electrolyte. ECS Electrochem. Lett. 2014, 3, C13–C16. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Yamaura, S.; Xie, G.; Makino, A.; Hara, N. Refinement of nanoporous copper by dealloying MgCuY amorphous alloys in sulfuric acids containing polyvinylpyrrolidone. J. Electrochem. Soc. 2014, 161, C120–C125. [Google Scholar] [CrossRef]
- Gu, X.; Xu, L.; Tian, F.; Ding, Y. Au–Ag alloy nanoporous nanotubes. Nano Res. 2009, 2, 386–393. [Google Scholar] [CrossRef]
- Koczkur, K.; Yi, Q.; Chen, A. Nanoporous Pt–Ru networks and their electrocatalytical properties. Adv. Mater. 2007, 19, 2648–2652. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Y.; Su, F.; Liu, A.; Qiu, H. Nanoporous PtAg and PtCu alloys with hollow ligaments for enhanced electrocatalysis and glucose biosensing. Biosens. Bioelectron. 2011, 27, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, Y.; Wang, X. Nanoporous bimetallic Pt–Au alloy nanocomposites with superior catalytic activity towards electro-oxidation of methanol and formic acid. Nanoscale 2011, 3, 1663–1674. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Jiang, Y.; Sun, J.; Jin, C.; Zhang, Z. Highly active nanoporous Pt-based alloy as anode and cathode catalyst for direct methanol fuel cells. J. Power Sources 2014, 267, 212–218. [Google Scholar] [CrossRef]
- Tang, Y.; Tang, B.; Qing, J.; Li, Q.; Lu, L. Nanoporous metallic surface: Facile fabrication and enhancement of boiling heat transfer. Appl. Surf. Sci. 2012, 258, 8747–8751. [Google Scholar] [CrossRef]
- Biener, J.; Wittstock, A.; Zepeda-Ruiz, L.A.; Biener, M.M.; Zielasek, V.; Kramer, D.; Viswanath, R.N.; Weissmüller, J.; Bäumer, M.; Hamza, A.V. Surface-chemistry-driven actuation in nanoporous gold. Nat. Mater. 2009, 8, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Hirata, A.; Qiu, H.; Chen, L.; Ge, X.; Fujita, T.; Chen, M. Self-grown oxy-hydroxide@nanoporous metal electrode for high-performance supercapacitors. Adv. Mater. 2014, 26, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Fujita, T.; Chen, M. Biofunctionalized nanoporous gold for electrochemical biosensors. Electrochim. Acta 2012, 67, 1–5. [Google Scholar] [CrossRef]
- Chen, L.; Yu, J.; Fujita, T.; Chen, M. Nanoporous copper with tunable nanoporosity for SERS applications. Adv. Funct. Mater. 2009, 19, 1221–1226. [Google Scholar] [CrossRef]
- Erlebacher, J.; Aziz, M.; Karma, A.; Dimitrov, N.; Sieradzki, K. Evolution of nanoporosity in dealloying. Nature 2001, 410, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Pia, G.; Delogu, F. Nanoporous Au: Statistical analysis of morphological features and evaluation of their influence on the elastic deformation behavior by phenomenological modeling. Acta Mater. 2015, 85, 250–260. [Google Scholar] [CrossRef]
- Detsi, E.; Punzhin, S.; Rao, J.; Onck, P.; Hosson, J. Enhanced strain in functional nanoporous gold with a dual microscopic length scale structure. ACS Nano 2012, 6, 3734–3744. [Google Scholar] [CrossRef] [PubMed]
- Detsi, E.; Chen, Z.; Vellinga, W.; Onck, P.; Hosson, J. Reversible strain by physisorption in nanoporous gold. Appl. Phys. Lett. 2011, 99, 083104. [Google Scholar] [CrossRef]
- Biener, J.; Hodge, A.; Hayes, J.; Volkert, C.; Zepeda-Ruiz, L.; Hamza, A.; Abraham, F. Size effects on the mechanical behavior of nanoporous Au. Nano Lett. 2006, 6, 2379–2382. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Lian, L.; Liu, Y.; Zhang, J. Fabrication and compression properties of bulk hierarchical nanoporous copper with fine ligament. Mater. Lett. 2014, 127, 59–62. [Google Scholar] [CrossRef]
- Weissmüller, J.; Wang, K. Composites of nanoporous gold and polymer. Adv. Mater. 2013, 25, 1280–1284. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, J.; Qin, C.; Liu, L.; Zhao, W.; Inoue, A. Fabrication and new electrochemical properties of nanoporous Cu by dealloying amorphous Cu–Hf–Al alloys. Intermetallics 2015, 56, 48–55. [Google Scholar] [CrossRef]
- Qin, C.; Wang, Z.; Liu, H.; Liu, L.; Wang, H.; Ding, J.; Zhao, W. Monolithic nanoporous copper with novel electrochemical properties fabricated by dealloying Cu–Zr(–Al) metallic glasses. Mater. Sci. Forum 2014, 783–786, 1925–1930. [Google Scholar] [CrossRef]
- Wang, Z.; Qin, C.; Zhao, W.; Jia, J. Tunable Cu2O nanocrystals fabricated by free dealloying of amorphous ribbons. J. Nanomater. 2012, 2012, 126715. [Google Scholar]
- Wang, Z.; Wang, L.; Qin, C.; Liu, J.; Li, Y.; Zhao, W. Tailored dealloying products of Cu-based metallic glasses in hydrochloric acid solutions. Mater. Res. 2014, 17, 1003–1009. [Google Scholar] [CrossRef]
- Wang, Z.; Qin, C.; Liu, L.; Wang, L.; Ding, J.; Zhao, W. Synthesis of CuxO (x = 1,2)/amorphous compounds by dealloying and spontaneous oxidation method. Mater. Res. 2014, 17, 33–37. [Google Scholar] [CrossRef]
- Li, M.; Zhou, Y.Z.; Geng, H. Fabrication of nanoporous copper ribbons by dealloying of Al–Cu alloys. J. Porous Mater. 2012, 19, 791–796. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, S.; Li, N.; Zheng, J.; An, S.; Li, G. Influence of dealloying solution on the microstructure of monolithic nanoporous copper through chemical dealloying of Al 30 at.% Cu alloy. Int. J. Electrochem. Sci. 2012, 7, 7993–8006. [Google Scholar]
- Tan, X.; Li, K.; Niu, G.; Yi, Z.; Luo, J.; Liu, Y.; Han, S.; Wu, W.; Tang, Y. Effect of heat treatment of Mn–Cu precursors on morphology of dealloyed nanoporous copper. J. Cent. South Univ. 2012, 19, 17–21. [Google Scholar] [CrossRef]
- Rizzi, P.; Scaglione, F.; Battezzati, L. Nanoporous gold by dealloying of an amorphous precursor. J. Alloy. Compd. 2014, 586, S117–S120. [Google Scholar] [CrossRef]
- Scaglione, F.; Rizzi, P.; Celegato, F.; Battezzati, L. Synthesis of nanoporous gold by free corrosion of an amorphous precursor. J. Alloy. Compd. 2014, 615, S142–S147. [Google Scholar] [CrossRef]
- Qiu, H.; Peng, L.; Li, X.; Xu, H.; Wang, Y. Using corrosion to fabricate various nanoporous metal structures. Corros. Sci. 2015, 92, 16–31. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, S.; Xing, Y.; Liu, W. Preparation and characterization of nanoporous Cu6Sn5/Cu composite by chemical dealloying of Al–Cu–Sn ternary alloy. J. Mater. Sci. 2012, 47, 5911–5917. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, S.; Li, N.; Zheng, J.; An, S.; Xing, Y. Formation of nanoporous copper with hybrid-modal pore size distributions related to surface diffusion of copper atoms during dealloying of Mg 13.5 at.% Cu alloy in an acidic solution. Int. J. Electrochem. Sci. 2012, 7, 6365–6377. [Google Scholar]
- Hayes, J.; Hodge, A.; Biener, J.; Hamza, A.; Sieradzki, K. Monolithic nanoporous copper by dealloying Mn–Cu. J. Mater. Res. 2006, 21, 2611–2616. [Google Scholar] [CrossRef]
- Qi, Z.; Zhao, C.; Wang, X.; Lin, J.; Shao, W.; Zhang, Z.; Bian, X. Formation and characterization of monolithic nanoporous copper by chemical dealloying of Al–Cu alloys. J. Phys. Chem. C 2009, 113, 6694–6698. [Google Scholar] [CrossRef]
- Zhao, C.; Qi, Z.; Wang, X.; Zhang, Z. Fabrication and characterization of monolithic nanoporous copper through chemical dealloying of Mg–Cu alloys. Corros. Sci. 2009, 51, 2120–2125. [Google Scholar] [CrossRef]
- Lin, B.; Kong, L.; Hodgson, P.; Dumée, L. Impact of the de-alloying kinetics and alloy microstructure on the final morphology of de-alloyed meso-porous metal films. Nanomaterials 2014, 4, 856–878. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Fabrication of nanoporous copper by dealloying amorphous binary Ti–Cu alloys in hydrofluoric acid solutions. Intermetallics 2012, 29, 14–20. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Dependency of the formation of Au-stabilized nanoporous copper on the dealloying temperature. Microporous Mesoporous Mater. 2014, 186, 181–186. [Google Scholar] [CrossRef]
- Luo, X.; Li, R.; Liu, Z.; Huang, L.; Shi, M.; Xu, T.; Zhang, T. Three-dimensional nanoporous copper with high surface area by dealloying Mg–Cu–Y metallic glasses. Mater. Lett. 2012, 76, 96–99. [Google Scholar] [CrossRef]
- Deng, Z.; Zhang, C.; Liu, L. Chemically dealloyed MgCuGd metallic glass with enhanced catalytic activity in degradation of phenol. Intermetallics 2014, 52, 9–14. [Google Scholar] [CrossRef]
- Abe, H.; Sato, K.; Nishikawa, H.; Takemoto, T.; Fukuhara, M.; Inoue, A. Dealloying of Cu–Zr–Ti bulk metallic glass in hydrofluoric acid solution. Mater. Trans. 2009, 50, 1255–1258. [Google Scholar] [CrossRef]
- Aburada, T.; Fitz-Gerald, J.; Scully, J. Synthesis of nanoporous copper by dealloying of Al–Cu–Mg amorphous alloys in acidic solution: The effect of nickel. Corros. Sci. 2011, 53, 1627–1632. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Makino, A.; Sugawara, Y.; Muto, I.; Hara, N. Fabrication of nanoporous copper by dealloying of amorphous Ti–Cu–Ag alloys. J. Alloy. Compd. 2014, 586, S134–S138. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, Z. On the electrochemical dealloying of Al-based alloys in a NaCl aqueous solution. Phys. Chem. Chem. Phys. 2010, 12, 1453–1472. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, W.; Inoue, A. Nanoporous Cu wide ribbons with good mechanical integrity. Mater. Sci. Eng. B 2012, 177, 532–535. [Google Scholar] [CrossRef]
- Luo, X.; Li, R.; Huang, L.; Zhang, T. Nucleation and growth of nanoporous copper ligaments during electrochemical dealloying of Mg-based metallic glasses. Corros. Sci. 2013, 67, 100–108. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Makino, A.; Hara, N. Nickel-stabilized nanoporous copper fabricated from ternary TiCuNi amorphous alloys. Mater. Lett. 2013, 94, 128–131. [Google Scholar] [CrossRef]
- Cheng, I.; Hodge, A. Morphology, oxidation, and mechanical behavior of nanoporous Cu foams. Adv. Eng. Mater. 2012, 14, 219–226. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Liu, Y.; Zhang, H. Fabrication of a bimodal micro/nanoporous metal by the Gasar and dealloying processes. Mater. Lett. 2013, 92, 448–451. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, S.; Li, N.; Zheng, J.; Xing, Y. Influence of phase constituent and proportion in initial Al–Cu alloys on formation of monolithic nanoporous copper through chemical dealloying in an alkaline solution. Corros. Sci. 2011, 53, 809–814. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, X.; Qi, Z.; Ji, H.; Zhang, Z. On the electrochemical dealloying of Mg–Cu alloys in a NaCl aqueous solution. Corros. Sci. 2010, 52, 3962–3972. [Google Scholar] [CrossRef]
- Aburada, T.; Unlu, N.; Fitz-Gerald, J.; Scully, J. Effect of Ni as a minority alloying element on the corrosion behavior in Al–Cu–Mg–(Ni) metallic glasses. Scr. Mater. 2008, 58, 623–626. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Uniform evolution of nanoporosity on amorphous Ti–Cu alloys. J. Nanosci. Nanotechnol. 2014, 14, 7879–7883. [Google Scholar] [CrossRef]
- Lan, G.; Xie, Z.; Huang, Z.; Yang, S.; Zhang, X.; Zeng, Y.; Jiang, J. Amorphous alloy: Promising precursor to form nanoflowerpot. Adv. Mater. Sci. Eng. 2014, 2014, 263681. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Hara, N. Refinement of nanoporous copper: A summary of micro-alloying of Au-group and Pt-group elements. Mater. Trans. 2014, 55, 796–800. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Fabrication of ultrafine nanoporous copper by the minor addition of gold. Mater. Trans. 2012, 53, 1765–1769. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Elaboration of nanoporous copper by modifying surface diffusivity by the minor addition of gold. Microporous Mesoporous Mater. 2013, 165, 257–264. [Google Scholar] [CrossRef]
- Dan, Z.H.; Qin, F.X.; Hara, N. Polyvinylpyrrolidone macromolecules function as a diffusion barrier during dealloying. Mater. Chem. Phys. 2014, 146, 277–282. [Google Scholar] [CrossRef]
- Xu, Q. Nanoporous Materials: Synthesis and Applications; CRC Press: Boca Raton, FL, USA, 2013; Chapter 5. [Google Scholar]
- Liu, D.; Yang, Z.; Wang, P.; Li, F.; Wang, D.; He, D. Preparation of 3D nanoporous copper-supported cuprous oxide for high-performance lithium ion battery anodes. Nanoscale 2013, 5, 1917–1921. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Wang, R.; Zhang, Y.; Ding, Y. A general corrosion route to nanostructured metal oxides. Nanoscale 2010, 2, 906–909. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Li, M.; Jia, S.; Zhao, X.; Xu, C. Controllable preparation of Co3O4 nanosheets and their electrochemical performance for Li-ion batteries. RSC Adv. 2013, 3, 7850–7854. [Google Scholar] [CrossRef]
- Jia, S.; Song, T.; Zhao, B.; Zhai, Q.; Gao, Y. Regular Fe3O4 octahedrons with excellent soft magnetic properties prepared by dealloying technique. J. Alloy. Compd. 2014, 585, 580–586. [Google Scholar] [CrossRef]
- Liu, Z.; Yamazaki, T.; Shen, Y.; Meng, D.; Kikuta, T.; Nakatani, N.; Kawabata, T. Dealloying derived synthesis of W nanopetal films and their transformation into WO3. J. Phys. Chem. C 2008, 112, 1391–1395. [Google Scholar] [CrossRef]
- Zhao, Z.; Xu, J.; Liaw, P.; Wu, B.; Wang, Y. One-step formation and photocatalytic performance of spindle-like TiO2 nanorods synthesized by dealloying amorphous Cu50Ti50 alloy. Corros. Sci. 2014, 84, 66–73. [Google Scholar] [CrossRef]
- Qi, Z.; Gong, Y.; Zhang, C.; Xu, J.; Wang, X.; Zhao, C.; Ji, H.; Zhang, Z. Fabrication and characterization of magnetic nanoporous Cu/(Fe,Cu)3O4 composites with excellent electrical conductivity by one-step dealloying. J. Mater. Chem. 2011, 21, 9716–9724. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, M.; Su, J.; Liu, Y. Fabrication of porous CuO nanoplate-films by oxidation-assisted dealloying method. Surf. Coat. Technol. 2014, 249, 19–23. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, Y.; Guo, H. Electrochemical behavior of Cu–Hf–Al amorphous films. ECS Electrochem. Lett. 2014, 3, C1–C3. [Google Scholar] [CrossRef]
- Zahrani, E.; Alfantazi, A. Molten salt induced corrosion of Inconel 625 superalloy in PbSO4–Pb3O4–PbCl2–Fe2O3–ZnO environment. Corros. Sci. 2012, 65, 340–359. [Google Scholar] [CrossRef]
- Wang, Z. Fabrication of Nanoporous Copper and Nano/Micro Cuprous Oxide Particles by Dealloying Method; Hebei University of Technology: Tianjin, China, 2013. [Google Scholar]
- Chen, K.; Xue, D. Cu-based materials as high-performance electrodes toward electrochemical energy storage. Funct. Mater. Lett. 2014, 7, 1430001. [Google Scholar] [CrossRef]
- Poizot, P.; Laruelle, S.; Grugeon, S.; Dupont, L.; Tarascon, J. Nano-sized transition-metal oxides as negative-electrode materials for lithium-ion batteries. Nature 2000, 407, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, W.; Zhu, W. Shape evolution and size-controllable synthesis of Cu2O octahedra and their morphology-dependent photocatalytic properties. J. Phys. Chem. B 2006, 110, 13829–13834. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Lu, L.; Huang, X.; Zhang, Z.; Qu, Y. Immobilization of horseradish peroxidase on nanoporous copper and its potential applications. Bioresour. Technol. 2010, 101, 9415–9420. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Zhao, J.; Yu, X. Nanoporous Cu film/Cu plate with superior catalytic performance toward electro-oxidation of hydrazine. J. Power Sources 2013, 222, 135–139. [Google Scholar] [CrossRef]
- Luo, X.; Li, R.; Zong, J.; Zhang, Y.; Li, H.; Zhang, T. Enhanced degradation of azo dye by nanoporous-copper-decorated Mg–Cu–Y metallic glass powder through dealloying pretreatment. Appl. Surf. Sci. 2014, 305, 314–320. [Google Scholar] [CrossRef]
- Chen, L.; Fujita, T.; Ding, Y.; Chen, M. A three-dimensional gold-decorated nanoporous copper core–shell composite for electrocatalysis and nonenzymatic biosensing. Adv. Funct. Mater. 2010, 20, 2279–2285. [Google Scholar] [CrossRef]
- Zhang, S.; Zheng, Y.; Yuan, L.; Zhao, L. Ni–B amorphous alloy nanoparticles modified nanoporous Cu toward ethanol oxidation in alkaline medium. J. Power Sources 2014, 247, 428–436. [Google Scholar] [CrossRef]
- Li, M.; Su, Y.; Zhao, J.; Geng, H.; Zhang, J.; Zhang, L.; Yang, C.; Zhang, Y. One-pot preparation of thin nanoporous copper foils with enhanced light absorption and SERS properties. CrystEngComm 2015, 17, 1296–1304. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, L.; Fujita, T.; Chen, M. Surface-enhanced Raman scattering of silver@nanoporous copper core–shell composites synthesized by an in situ sacrificial template approach. J. Phys. Chem. C 2009, 113, 14195–14199. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Y.; Wang, J.; Geng, H.; Qiu, H. Fabrication of nanoporous Cu–Pt(Pd) core/shell structure by galvanic replacement and its application in electrocatalysis. ACS Appl. Mater. Interfaces 2011, 3, 4626–4632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zheng, Y.; Yuan, L.; Wang, X.; Zhao, L. In situ synthesis of nickel–boron amorphous alloy nanoparticles electrode on nanoporous copper film/brass plate for ethanol electro-oxidation. Int. J. Hydrog. Energy 2014, 39, 3100–3108. [Google Scholar] [CrossRef]
- Kou, T.; Si, C.; Gao, Y.; Frenzel, J.; Wang, H.; Yan, X.; Bai, Q.; Eggeler, G.; Zhang, Z. Large-scale synthesis and catalytic activity of nanoporous Cu–O system towards CO oxidation. RSC Adv. 2014, 4, 65004. [Google Scholar] [CrossRef]
- Kou, T.; Wang, Y.; Zhang, C.; Sun, J.; Zhang, Z. Adsorption behavior of methyl orange onto nanoporous core–shell Cu@Cu2O nanocomposite. Chem. Eng. J. 2013, 223, 76–83. [Google Scholar] [CrossRef]
- Li, G.; Song, Y.; Zhang, L.; Wei, X.; Song, X.; Sun, Z. Nanoporous copper silicon composite prepared by chemical dealloying as anode material for Lithium-ion batteries. Funct. Mater. Lett. 2013, 6, 1350033. [Google Scholar] [CrossRef]
- Hou, C.; Lang, X.; Han, G.; Li, Y.; Zhao, L.; Wen, Z.; Zhu, Y.; Zhao, M.; Li, J.; Lian, J.; et al. Integrated solid/nanoporous copper/oxide hybrid bulk electrodes for high-performance lithium-ion batteries. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.; Brodd, R. What are batteries, fuel cells, and supercapacitors? Chem. Rev. 2004, 104, 4245–4270. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Xu, H.; Liu, L.; Wang, Y. Correlation of the structure and applications of dealloyed nanoporous metals in catalysis and energy conversion/storage. Nanoscale 2015, 7, 386–400. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.; Simon, P. Electrochemical capacitors for energy management. Science 2008, 321, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Zhang, W.; Fu, Y.; Wang, X. Controlled growth of nanostructured MnO2 on carbon nanotubes for high-performance electrochemical capacitors. Electrochim. Acta 2015, 152, 480–488. [Google Scholar] [CrossRef]
- Miller, J.; Outlaw, R.; Holloway, B. Graphene electric double layer capacitor with ultra-high-power performance. Electrochim. Acta 2011, 56, 10443–10449. [Google Scholar] [CrossRef]
- Guo, Y.; Shi, Z.; Chen, M.; Wang, C. Hierarchical porous carbon derived from sulfonated pitch for electrical double layer capacitors. J. Power Sources 2014, 252, 235–243. [Google Scholar] [CrossRef]
- Liu, D.; Shen, J.; Liu, N.; Yang, H.; Du, A. Preparation of activated carbon aerogels with hierarchically porous structures for electrical double layer capacitors. Electrochim. Acta 2013, 89, 571–576. [Google Scholar] [CrossRef]
- Du, X.; Zhao, W.; Wang, Y.; Wang, C.; Chen, M.; Qi, T.; Hua, C.; Ma, M. Preparation of activated carbon hollow fibers from ramie at low temperature for electric double-layer capacitor applications. Bioresour. Technol. 2013, 149, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Kuratani, K.; Kiyobayashi, T.; Kuriyama, N. Influence of the mesoporous structure on capacitance of the RuO2 electrode. J. Power Sources 2009, 189, 1284–1291. [Google Scholar] [CrossRef]
- Cheng, S.; Yang, L.; Chen, D.; Ji, X.; Jiang, Z.; Ding, D.; Liu, M. Phase evolution of an alpha MnO2-based electrode for pseudo-capacitors probed by in operando Raman spectroscopy. Nano Energy 2014, 9, 161–167. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Shi, S.; Xiong, Q.; Zhao, X.; Wang, X.; Gu, C.; Tu, J. Synthesis of porous Co3O4 nanoflake array and its temperature behavior as pseudo-capacitor electrode. J. Power Sources 2014, 256, 200–205. [Google Scholar] [CrossRef]
- Yuan, C.; Hou, L.; Feng, Y.; Xiong, S.; Zhang, X. Sacrificial template synthesis of short mesoporous NiO nanotubes and their application in electrochemical capacitors. Electrochim. Acta 2013, 88, 507–512. [Google Scholar] [CrossRef]
- Bavio, M.; Acosta, G.; Kessler, T. Polyaniline and polyaniline–carbon black nanostructures as electrochemical capacitor electrode materials. Int. J. Hydrog. Energy 2014, 39, 8582–8589. [Google Scholar] [CrossRef]
- Lee, H.; Cho, M.; Kim, I.; Nam, J.; Lee, Y. RuOx/polypyrrole nanocomposite electrode for electrochemical capacitors. Synth. Met. 2010, 160, 1055–1059. [Google Scholar] [CrossRef]
- Aradilla, D.; Estrany, F.; Casellas, F.; Iribarren, J.; Alemán, C. All-polythiophene rechargeable batteries. Org. Electron. 2014, 15, 40–46. [Google Scholar] [CrossRef]
- Cortie, M.; Maaroof, A.; Smith, G. Electrochemical capacitance of mesoporous gold. Gold Bull. 2005, 38, 14–22. [Google Scholar] [CrossRef]
- Snyder, J.; Asanithi, P.; Dalton, A.; Erlebacher, J. Stabilized nanoporous metals by dealloying ternary alloy precursors. Adv. Mater. 2008, 20, 4883–4886. [Google Scholar] [CrossRef]
- Lang, X.; Hirata, A.; Fujita, T.; Chen, M. Nanoporous metal/oxide hybrid electrodes for electrochemical supercapacitors. Nat. Nanotechnol. 2011, 6, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Lang, X.; Zhang, L.; Fujita, T.; Ding, Y.; Chen, M. Three-dimensional bicontinuous nanoporous Au/polyaniline hybrid films for high-performance electrochemical supercapacitors. J. Power Sources 2012, 197, 325–329. [Google Scholar] [CrossRef]
- Meng, F.; Ding, Y. Sub-micrometer-thick all-solid-state supercapacitors with high power and energy densities. Adv. Mater. 2011, 23, 4098–4102. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Balka, T. Synthesis of nanoporous nickel thin films from various precursors. Philos. Mag. Lett. 2014, 94, 573–581. [Google Scholar] [CrossRef]
- Dan, Z.; Qin, F.; Sugawara, Y.; Muto, I.; Hara, N. Bimodal nanoporous nickel prepared by dealloying Ni38Mn62 alloys. Intermetallics 2012, 31, 157–164. [Google Scholar] [CrossRef]
- Fukumizu, T.; Kotani, F.; Yoshida, A.; Katagiri, A. Electrochemical formation of porous nickel in zinc chloride-alkali chloride melts. J. Electrochem. Soc. 2006, 153, C629–C633. [Google Scholar] [CrossRef]
- Hakamada, M.; Mabuchi, M. Preparation of nanoporous Ni and Ni–Cu by dealloying of rolled Ni–Mn and Ni–Cu–Mn alloys. J. Alloy. Compd. 2009, 485, 583–587. [Google Scholar] [CrossRef]
- Qiu, H.; Kang, J.; Liu, P.; Hirata, A.; Fujita, T.; Chen, M. Fabrication of large-scale nanoporous nickel with a tunable pore size for energy storage. J. Power Sources 2014, 247, 896–905. [Google Scholar] [CrossRef]
- Dong, C.; Bai, Q.; Cheng, G.; Zhao, B.; Wang, H.; Gao, Y.; Zhang, Z. Flexible and ultralong-life cuprous oxide microsphere-nanosheets with superior pseudocapacitive properties. RSC Adv. 2015, 5, 6207–6214. [Google Scholar] [CrossRef]
- Dong, C.; Wang, Y.; Xu, J.; Cheng, G.; Yang, W.; Kou, T.; Zhang, Z.; Ding, Y. 3D binder-free Cu2O@Cu nanoneedle arrays for high-performance asymmetric supercapacitors. J. Mater. Chem. A 2014, 2, 18229–18235. [Google Scholar] [CrossRef]
- Momeni, M.; Nazari, Z.; Kazempour, A.; Hakimiyan, M.; Mirhoseini, S. Preparation of CuO nanostructures coating on copper as supercapacitor materials. Surf. Eng. 2014, 30, 775–778. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Liu, J.; Qin, C.; Yu, H.; Xia, X.; Wang, C.; Zhang, Y.; Hu, Q.; Zhao, W. Dealloying of Cu-Based Metallic Glasses in Acidic Solutions: Products and Energy Storage Applications. Nanomaterials 2015, 5, 697-721. https://doi.org/10.3390/nano5020697
Wang Z, Liu J, Qin C, Yu H, Xia X, Wang C, Zhang Y, Hu Q, Zhao W. Dealloying of Cu-Based Metallic Glasses in Acidic Solutions: Products and Energy Storage Applications. Nanomaterials. 2015; 5(2):697-721. https://doi.org/10.3390/nano5020697
Chicago/Turabian StyleWang, Zhifeng, Jiangyun Liu, Chunling Qin, Hui Yu, Xingchuan Xia, Chaoyang Wang, Yanshan Zhang, Qingfeng Hu, and Weimin Zhao. 2015. "Dealloying of Cu-Based Metallic Glasses in Acidic Solutions: Products and Energy Storage Applications" Nanomaterials 5, no. 2: 697-721. https://doi.org/10.3390/nano5020697
APA StyleWang, Z., Liu, J., Qin, C., Yu, H., Xia, X., Wang, C., Zhang, Y., Hu, Q., & Zhao, W. (2015). Dealloying of Cu-Based Metallic Glasses in Acidic Solutions: Products and Energy Storage Applications. Nanomaterials, 5(2), 697-721. https://doi.org/10.3390/nano5020697







