NiO Nanofibers as a Candidate for a Nanophotocathode
Abstract
:1. Introduction
2. Results and Discussion
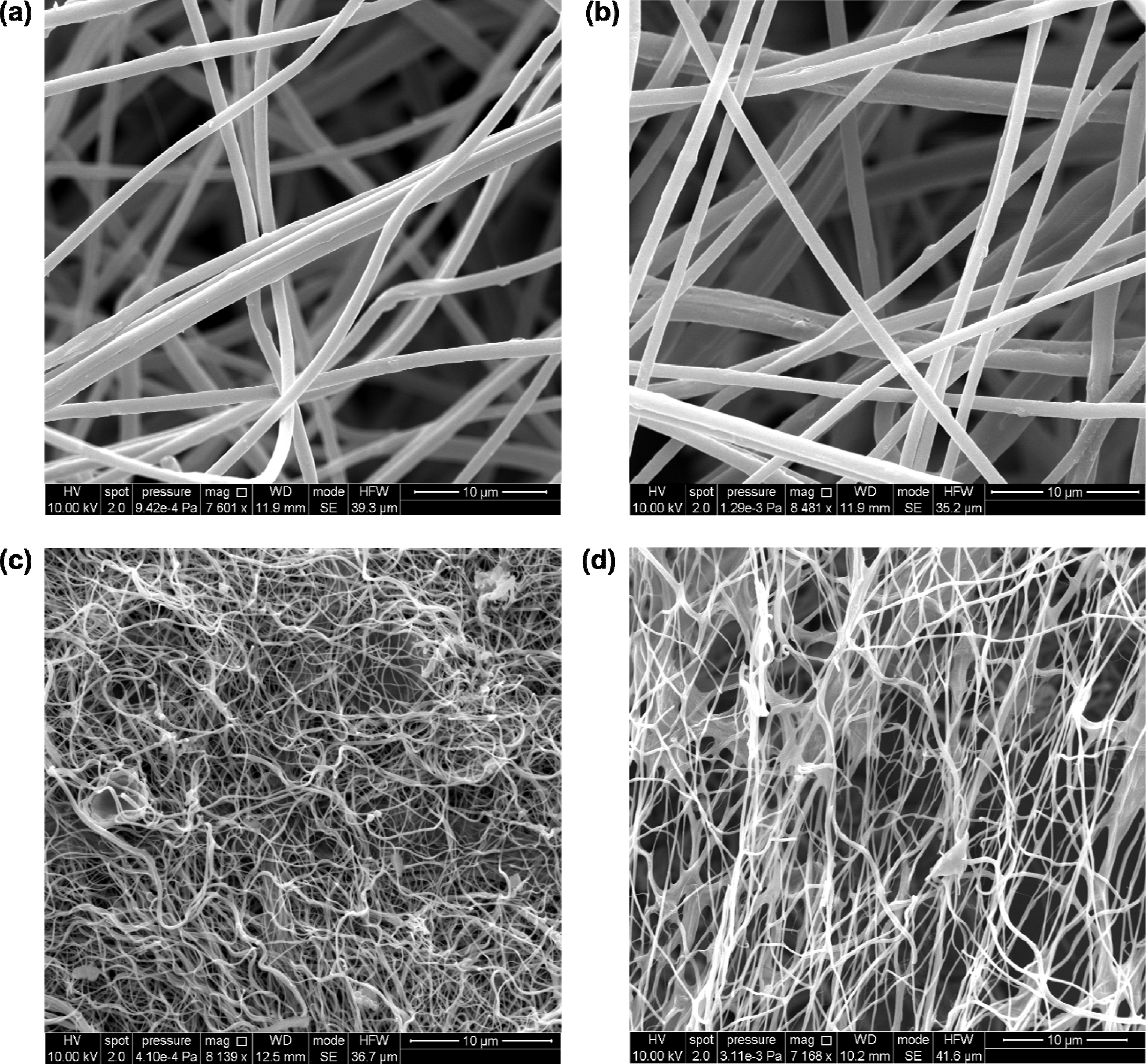
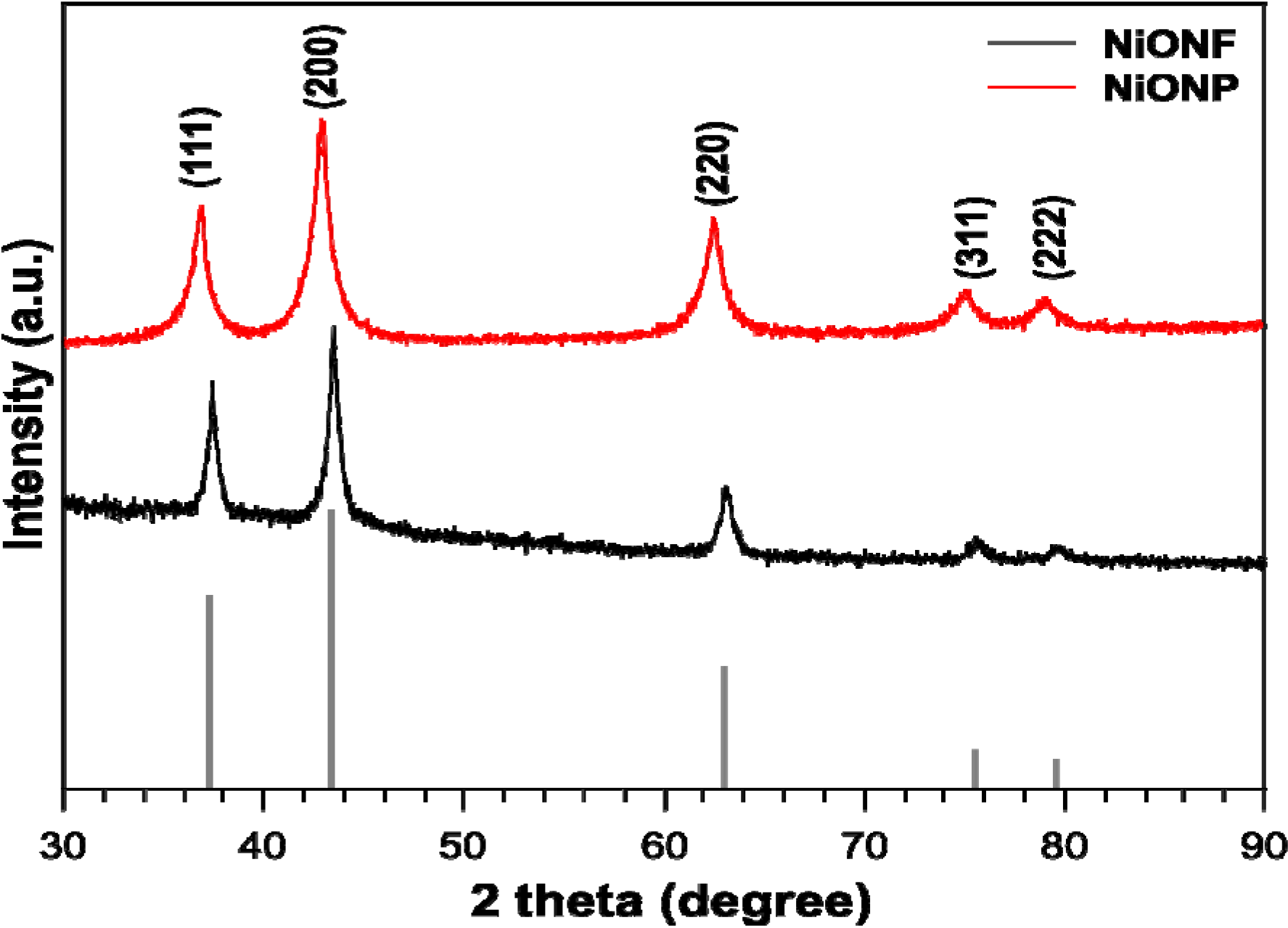
2.1. Structural and Surface Characterization

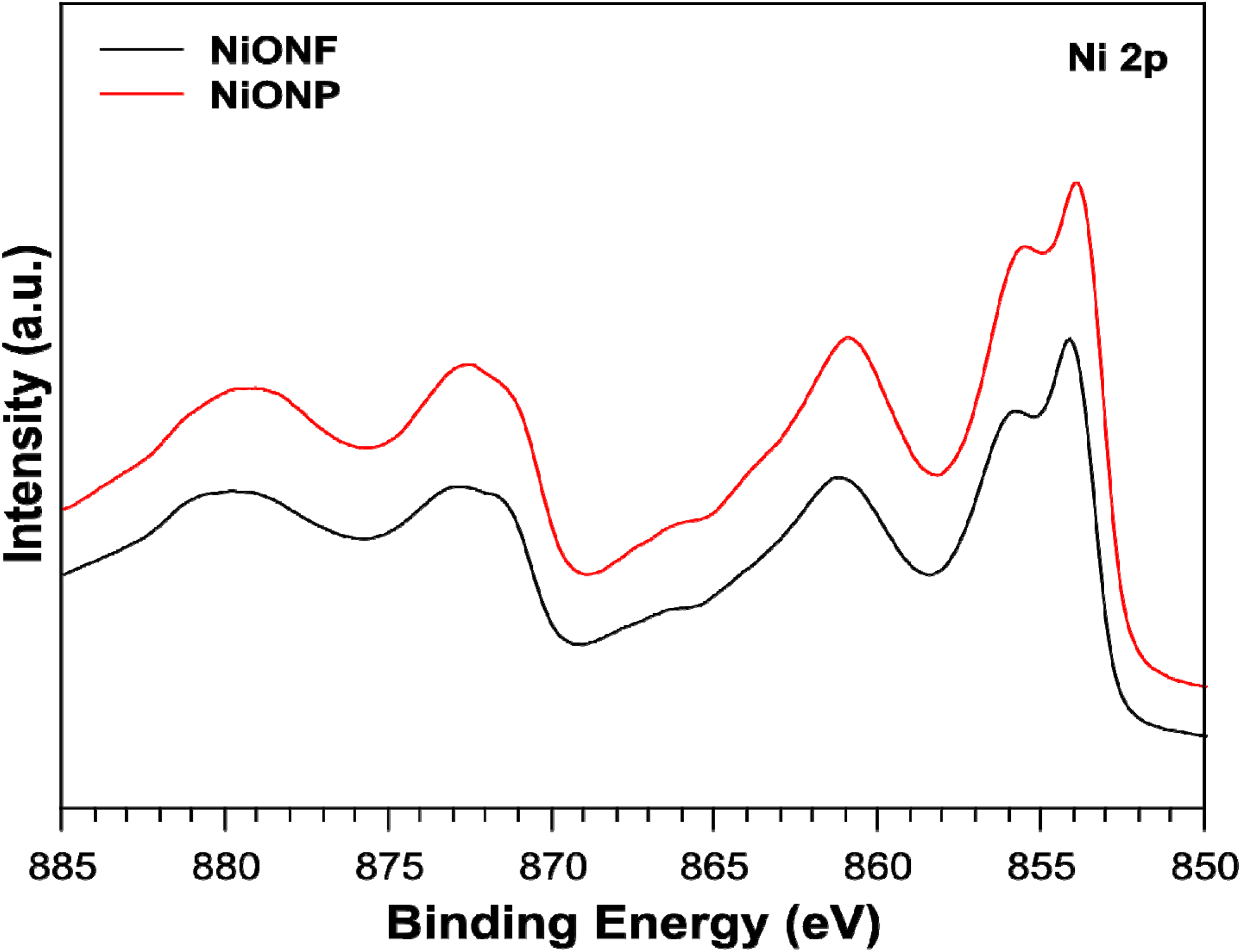
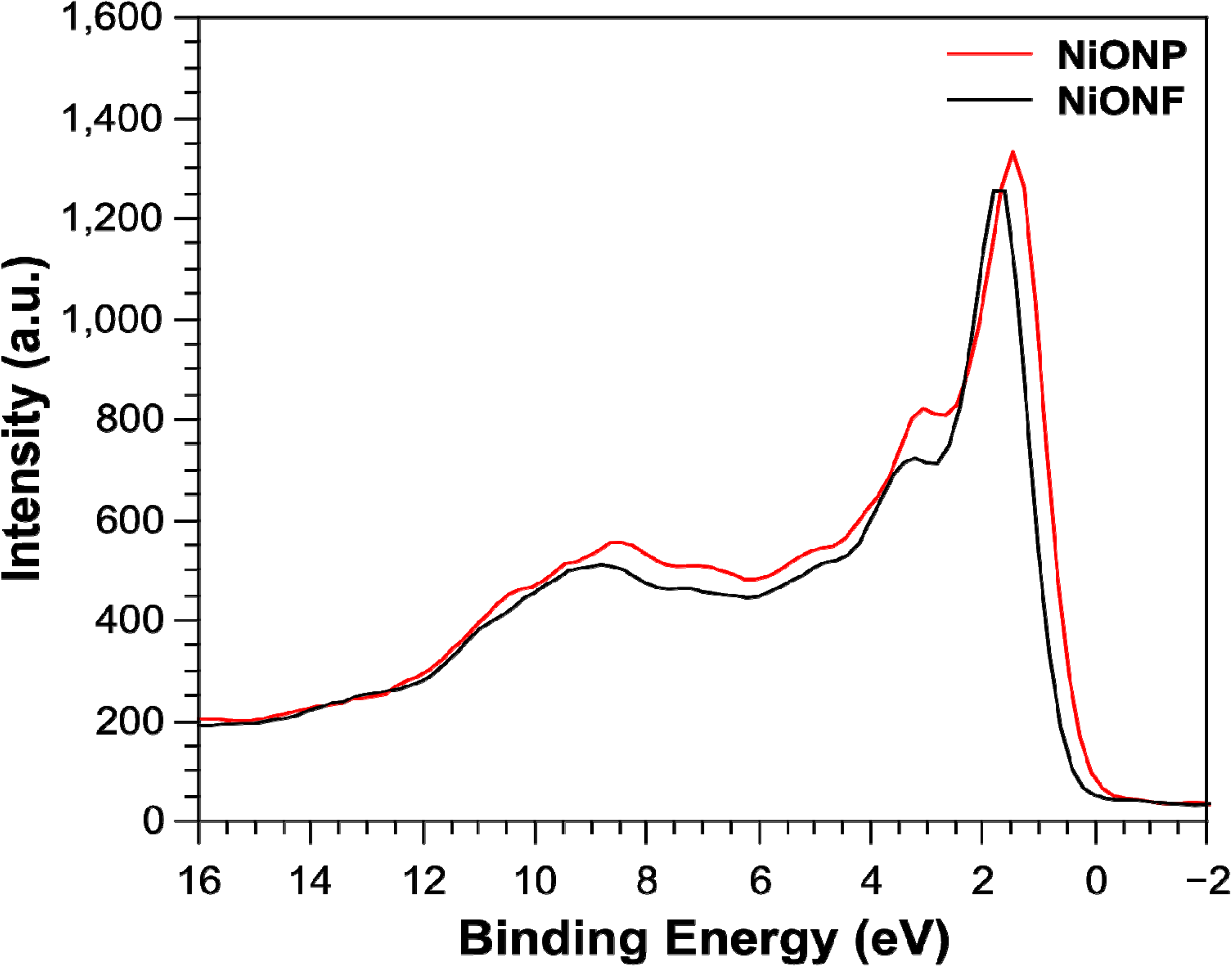
2.2. XPS
2.3. Photocurrent Response of NiO Nanoparticles
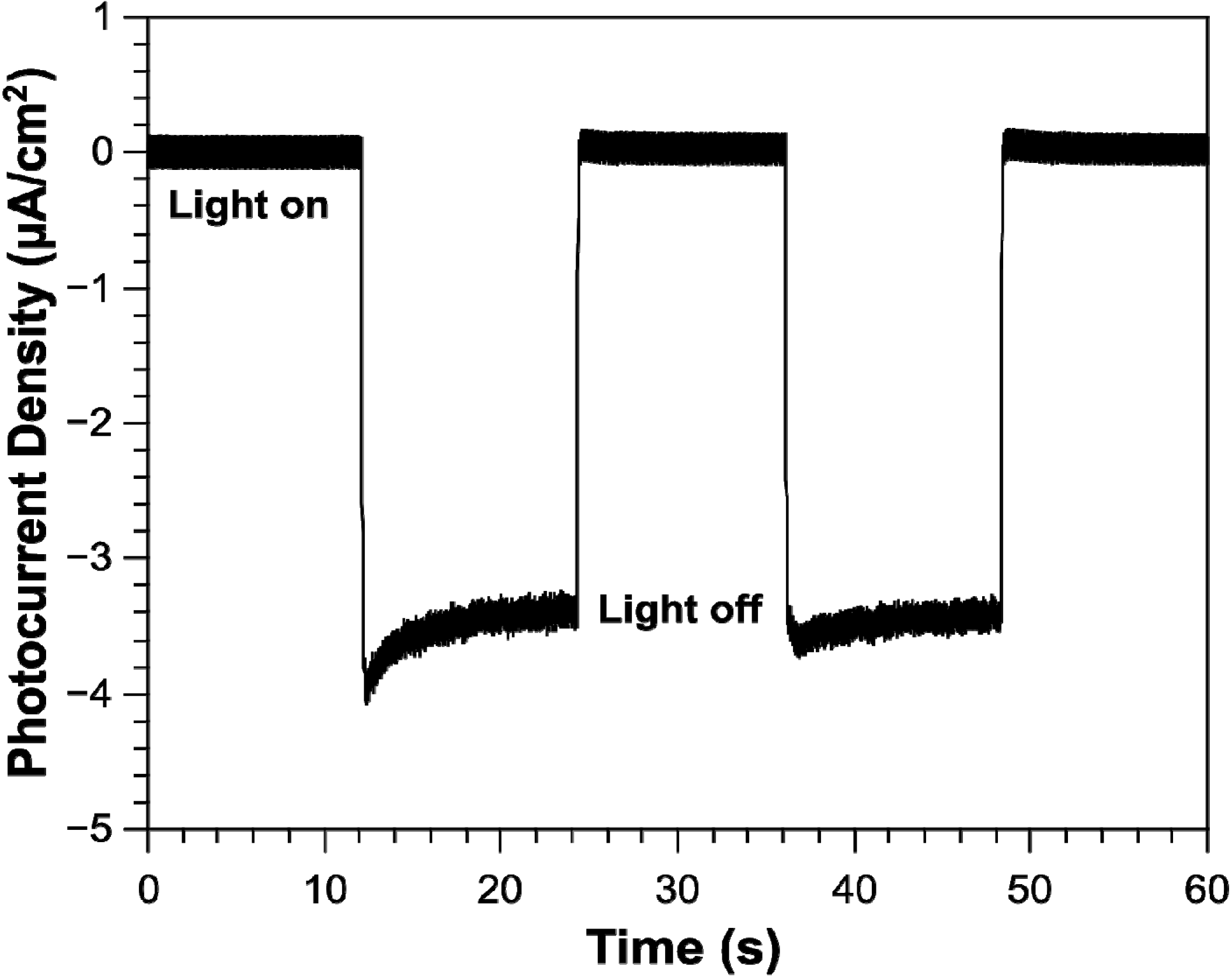
3. Experimental Section
3.1. Chemicals
3.2. Synthesis of NiO Nanofibers
3.3. Fabrication of NiO Nanofiber Electrode
3.4. Fabrication of NiO Particle Electrode
3.5. Photocurrent Measurements
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Conflicts of Interest
References
- Qu, Y.; Zhou, W.; Miao, X.; Li, Y.; Jiang, L.; Pan, K.; Tian, G.; Ren, Z.; Wang, G.; Fu, H. A new layered photocathode with porous NiO nanosheets: An effective candidate for p-type dye-sensitized solar cells. Chemistry 2013, 8, 3085–3090. [Google Scholar]
- Liu, H.; Wang, G.; Liu, J.; Qiao, S.; Ahn, H. Highly ordered mesoporous NiO anode material for lithium ion batteries with an excellent electrochemical performance. J. Mater. Chem. 2011, 21, 3046–3052. [Google Scholar] [CrossRef]
- Sialvi, M.Z.; Mortimer, R.J.; Wilcox, G.D.; Teridi, A.M.; Varley, T.S.; Wijayantha, K.G.U.; Kirk, C.A. Electrochromic and colorimetric properties of Nickel(II) oxide Thin films prepared by aerosol-assisted chemical vapor deposition. ACS Appl. Mater. Interfaces 2013, 5, 5675–5682. [Google Scholar] [CrossRef]
- Liu, M.; Wang, Y.; Li, P.; Cheng, Z.; Zhang, Y.; Zhang, M.; Hu, M.; Li, J. Preparation and characterization of multilayer NiO nano-products via electrospinning. Appl. Surf. Sci. 2013, 284, 453–458. [Google Scholar]
- Mattei, G.; Mazzoldi, P.; Post, M.L.; Buso, D.; Guglielmi, M.; Martucci, A. Cookie-like Au/NiO nanoparticles with optical gas-sensing properties. Adv. Mater. 2007, 19, 561–564. [Google Scholar] [CrossRef]
- Pan, J.H.; Huang, Q.; Koh, Z.Y.; Neo, D.; Wang, X.Z.; Wang, Q. Scalable synthesis of urchin- and flowerlike hierarchical NiO microspheres and their electrochemical property for lithium storage. ACS Appl. Mater. Interfaces 2013, 5, 6292–6299. [Google Scholar] [CrossRef]
- Powar, S.; Wu, Q.; Weidelener, M.; Nattestad, A.; Hu, Z.; Mishra, A.; Bauerle, P.; Spiccia, L.; Cheng, Y.-B.; Bach, U. Improved photocurrents for p-type dye-sensitized solar cells using nano-structured Nickel(II) oxide microballs. Energy Environ. Sci. 2012, 5, 8896–8900. [Google Scholar] [CrossRef]
- Gibson, E.A.; Smeigh, A.L.; Le Pleux, L.; Fortage, J.; Boschloo, G.; Blart, E.; Pellegrin, Y.; Odobel, F.; Hagfeldt, A.; Hammarström, L. A p-type NiO-based dye-sensitized solar cell with an open-circuit voltage of 0.35 V. Angew. Chem. Int. Ed. 2009, 48, 4402–4405. [Google Scholar] [CrossRef]
- Salam Mahmood, A.; Venkatakrishnan, K.; Tan, B. Silicon nano network p/n sandwich solar cell. Sol. Energy Mater. Sol. Cells 2013, 115, 58–63. [Google Scholar] [CrossRef]
- Boschloo, G.; Hagfeldt, A. Spectroelectrochemistry of nanostructured NiO. J. Phys. Chem. B 2001, 105, 3039–3044. [Google Scholar] [CrossRef]
- Lee, M.K.; Park, J.J.; Rhee, C.K. Synthesis and structural properties of Ni–20Cr–2Y2O3 nanocomposite alloy prepared by a very high energy mechanical milling. Mater. Chem. Phys. 2012, 137, 129–134. [Google Scholar] [CrossRef]
- O’Regan, B.; Gratzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Yella, A.; Lee, H.-W.; Tsao, H.N.; Yi, C.; Chandiran, A.K.; Nazeeruddin, M.K.; Diau, E.W.-G.; Yeh, C.-Y.; Zakeeruddin, S.M.; Grätzel, M. Porphyrin-sensitized solar cells with Cobalt(II/III)-based redox electrolyte exceed 12 percent efficiency. Science 2011, 334, 629–634. [Google Scholar] [CrossRef]
- Wolcott, A.; Smith, W. A.; Kuykendall, T. R.; Zhao, Y.; Zhang, J. Z. Photoelectrochemical study of nanostructured ZnO thin films for hydrogen generation from water splitting. Adv. Funct. Mater. 2009, 19, 1849–1856. [Google Scholar] [CrossRef]
- Chen, H.M.; Chen, C.K.; Chang, Y.-C.; Tsai, C.-W.; Liu, R.-S.; Hu, S.-F.; Chang, W.-S.; Chen, K.-H. Quantum dot monolayer sensitized ZnO nanowire-array photoelectrodes: True efficiency for water splitting. Angew. Chem. Int. Ed. 2010, 49, 5966–5969. [Google Scholar] [CrossRef]
- Ji, Z.; Natu, G.; Huang, Z.; Wu, Y. Linker effect in organic donor-acceptor dyes for p-type NiO dye sensitized solar cells. Energy Environ. Sci. 2011, 4, 2818–2821. [Google Scholar] [CrossRef]
- Nattestad, A.; Mozer, A.J.; Fischer, M.K.R.; Cheng, Y.-B.; Mishra, A.; Bauerle, P.; Bach, U. Highly efficient photocathodes for dye-sensitized tandem solar cells. Nat. Mater. 2010, 9, 31–35. [Google Scholar] [CrossRef]
- He, J.; Lindström, H.; Hagfeldt, A.; Lindquist, S.-E. Dye-sensitized nanostructured tandem cell-first demonstrated cell with a dye-sensitized photocathode. Sol. Energy Mater. Sol. Cells 2000, 62, 265–273. [Google Scholar]
- Lefebvre, J.-F.; Sun, X.-Z.; Calladine, J.A.; George, M.W.; Gibson, E.A. Promoting charge-separation in p-type dye-sensitized solar cells using bodipy. Chem. Commun. 2014, 2014. [Google Scholar] [CrossRef]
- Smeigh, A.L.; Pleux, L.L.; Fortage, J.; Pellegrin, Y.; Blart, E.; Odobel, F.; Hammarstrom, L. Ultrafast recombination for NiO sensitized with a series of perylene imide sensitizers exhibiting Marcus normal behaviour. Chem. Commun. 2012, 48, 678–680. [Google Scholar]
- Zhang, X.L.; Zhang, Z.; Huang, F.; Bauerle, P.; Bach, U.; Cheng, Y.-B. Charge transport in photocathodes based on the sensitization of NiO nanorods. J. Mater. Chem. 2012, 22, 7005–7009. [Google Scholar] [CrossRef]
- Li, J.; Zhao, W.; Huang, F.; Manivannan, A.; Wu, N. Single-crystalline Ni(OH)2 and NiO nanoplatelet arrays as supercapacitor electrodes. Nanoscale 2011, 3, 5103–5109. [Google Scholar] [CrossRef]
- Joint Committee on Powder Diffraction Standards (JCPDS) Card No. 04-0835. International Centre for Diffraction Data: Newtown Square, PA, USA.
- Li, X.; Xiong, S.; Li, J.; Bai, J.; Qian, Y. Mesoporous NiO ultrathin nanowire networks topotactically transformed from α-Ni(OH)2 hierarchical microspheres and their superior electrochemical capacitance properties and excellent capability for water treatment. J. Mater. Chem. 2012, 22, 14276–14283. [Google Scholar] [CrossRef]
- Luches, P.; Altieri, S.; Giovanardi, C.; Moia, T.S.; Valeri, S.; Bruno, F.; Floreano, L.; Morgante, A.; Santaniello, A.; Verdini, A.; et al. Growth, structure and epitaxy of ultrathin NiO films on Ag(001). Thin Solid Films 2001, 400, 139–143. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar water splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef]
- Tong, L.; Iwase, A.; Nattestad, A.; Bach, U.; Weidelener, M.; Gotz, G.; Mishra, A.; Bauerle, P.; Amal, R.; Wallace, G.G.; et al. Sustained solar hydrogen generation using a dye-sensitised NiO photocathode/BiVO4 tandem photo-electrochemical device. Energy Environ. Sci. 2012, 5, 9472–9475. [Google Scholar] [CrossRef]
- Renaud, A.; Chavillon, B.; Cario, L.; Pleux, L.L.; Szuwarski, N.; Pellegrin, Y.; Blart, E.; Gautron, E.; Odobel, F.; Jobic, S. Origin of the black color of NiO used as photocathode in p-type dye-sensitized solar cells. J. Phys. Chem. C 2013, 117, 22478–22483. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Macdonald, T.J.; Xu, J.; Elmas, S.; Mange, Y.J.; Skinner, W.M.; Xu, H.; Nann, T. NiO Nanofibers as a Candidate for a Nanophotocathode. Nanomaterials 2014, 4, 256-266. https://doi.org/10.3390/nano4020256
Macdonald TJ, Xu J, Elmas S, Mange YJ, Skinner WM, Xu H, Nann T. NiO Nanofibers as a Candidate for a Nanophotocathode. Nanomaterials. 2014; 4(2):256-266. https://doi.org/10.3390/nano4020256
Chicago/Turabian StyleMacdonald, Thomas J., Jie Xu, Sait Elmas, Yatin J. Mange, William M. Skinner, Haolan Xu, and Thomas Nann. 2014. " NiO Nanofibers as a Candidate for a Nanophotocathode" Nanomaterials 4, no. 2: 256-266. https://doi.org/10.3390/nano4020256
APA StyleMacdonald, T. J., Xu, J., Elmas, S., Mange, Y. J., Skinner, W. M., Xu, H., & Nann, T. (2014). NiO Nanofibers as a Candidate for a Nanophotocathode. Nanomaterials, 4(2), 256-266. https://doi.org/10.3390/nano4020256




