Hemolytic Activity and Cytotoxicity of Synthetic Nanoclays with Montmorillonite Structure for Medical Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis
2.2. Characterization
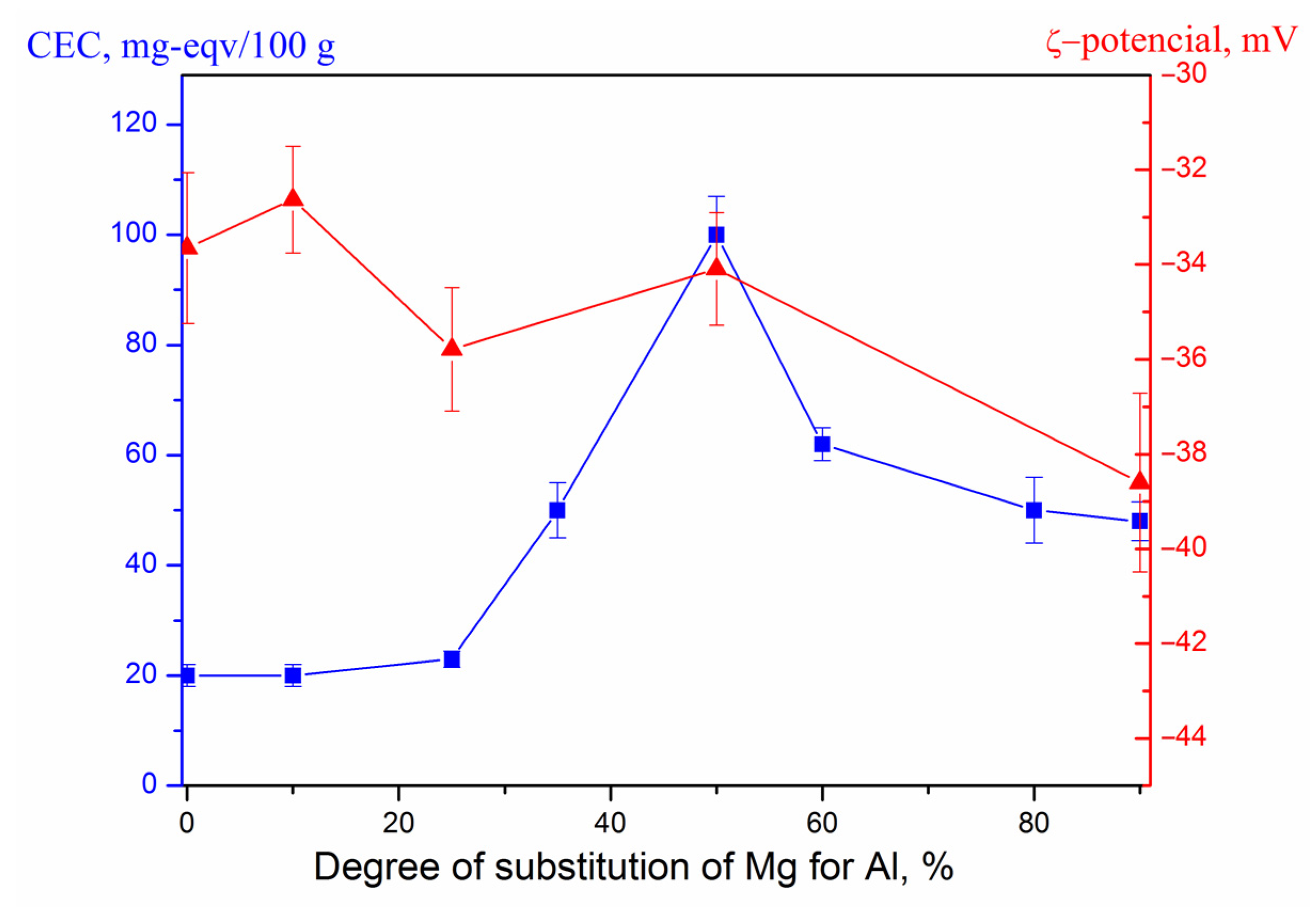
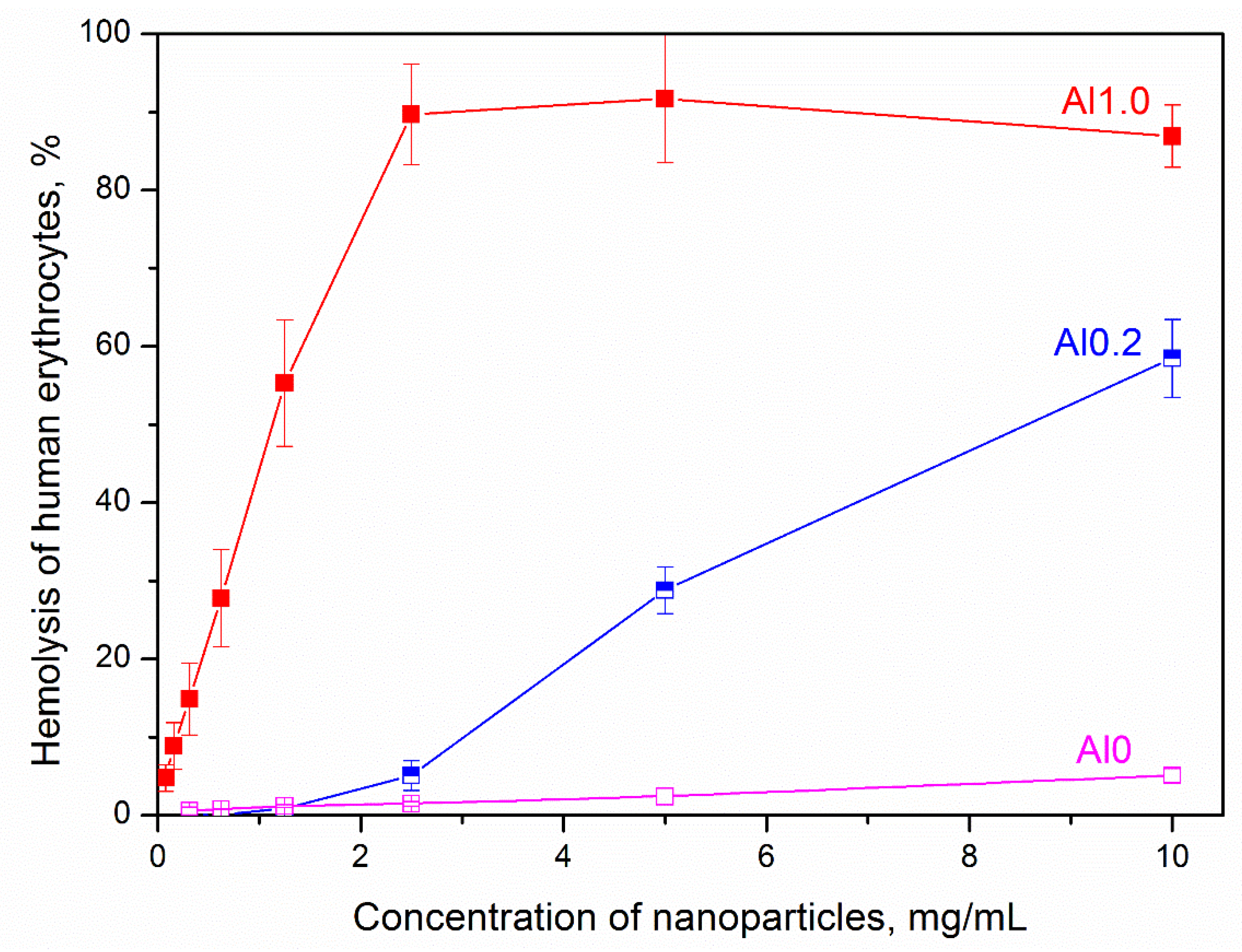
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ganapathy, D.; Shanmugam, R.; Pitchiah, S.; Murugan, P.; Chinnathambi, A.; Alharbi, S.A.; Durairaj, K.; Sundramoorthy, A.K. Potential Applications of Halloysite Nanotubes as Drug Carriers: A Review. J. Nanomater. 2022, 2022, 1068536. [Google Scholar] [CrossRef]
- Fizir, M.; Dramou, P.; Dahiru, N.S.; Ruya, W.; Huang, T.; He, H. Halloysite nanotubes in analytical sciences and in drug delivery: A review. Microchim. Acta 2018, 185, 389. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Brazovskaya, E.Y.; Shamova, O.V. Biological activity and sorption ability of synthetic montmorillonite modified by silver/lysozyme nanoparticles. Appl. Clay Sci. 2018, 163, 56–62. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Brazovskaya, E.Y.; Ul’yanova, N.Y.; Morozova, Y.A. Development of Approaches for Designing and Preparing Magnetic Nanocomposites Based on Zeolite Beta and Magnetite Nanoparticles under Hydrothermal Conditions. Glas. Phys. Chem. 2018, 44, 108–114. [Google Scholar] [CrossRef]
- Gao, X.; Miao, R.; Tao, Y.; Chen, X.; Wan, C.; Jia, R. Effect of Montmorillonite powder on intestinal mucosal barrier in children with abdominal Henoch–Schonlein purpura: A randomized controlled study. Medicine 2018, 97, e12577. [Google Scholar] [CrossRef]
- Park, J.-H.; Shin, H.-J.; Kim, M.H.; Kim, J.-S.; Kang, N.; Lee, J.-Y.; Kim, K.-T.; Lee, J.I.; Kim, D.-D. Application of montmorillonite in bentonite as a pharmaceutical excipient in drug delivery systems. J. Pharm. Investig. 2016, 46, 363–375. [Google Scholar] [CrossRef]
- Govea-Alonso, D.O.; García-Soto, M.J.; Betancourt-Mendiola, L.; Padilla-Ortega, E.; Rosales-Mendoza, S.; González-Ortega, O. Nanoclays: Promising Materials for Vaccinology. Vaccines 2022, 10, 1549. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Ng, E.-P.; Thirifays, C.; Lakiss, L.; Goupil, G.-M.; Mintova, S.; Burtea, C.; Oveisi, E.; Hébert, C.; de Vries, M.; et al. Corona protein composition and cytotoxicity evaluation of ultra-small zeolites synthesized from template free precursor suspensions. Toxicol. Res. 2013, 2, 270–279. [Google Scholar] [CrossRef]
- Satish, S.; Tharmavaram, M.; Rawtani, D. Halloysite nanotubes as a nature’s boon for biomedical applications. Nanobiomedicine 2019, 6, 1849543519863625. [Google Scholar] [CrossRef]
- Kraljević Pavelić, S.; Simović Medica, J.; Gumbarević, D.; Filošević, A.; Pržulj, N.; Pavelić, K. Critical Review on Zeolite Clinoptilolite Safety and Medical Applications in vivo. Front. Pharmacol. 2018, 9, 1350. [Google Scholar] [CrossRef]
- Bacakova, L.; Vandrovcova, M.; Kopova, I.; Jirka, I. Applications of zeolites in biotechnology and medicine—A review. Biomater. Sci. 2018, 6, 974–989. [Google Scholar] [CrossRef] [PubMed]
- Servatan, M.; Zarrintaj, P.; Mahmodi, G.; Kim, S.-J.; Ganjali, M.R.; Saeb, M.R.; Mozafari, M. Zeolites in drug delivery: Progress, challenges and opportunities. Drug Discov. Today 2020, 25, 642–656. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.; Wang, M.-C.; Lin, J.-J. Biocompatibility and antimicrobial evaluation of montmorillonite/chitosan nanocomposites. Appl. Clay Sci. 2012, 56, 53–62. [Google Scholar] [CrossRef]
- Ijagbemi, C.O.; Baek, M.-H.; Kim, D.-S. Montmorillonite surface properties and sorption characteristics for heavy metal removal from aqueous solutions. J. Hazard. Mater. 2009, 166, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Golubeva, O.Y.; Maslennikova, T.P.; Ulyanova, N.Y.; Dyakina, M.P. Sorption of lead(II) ions and water vapors by synthetic hydro- and aluminosilicates with layered, framework, and nanotube morphology. Glas. Phys. Chem. 2014, 40, 250–255. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Alikina, Y.; Brazovskaya, E.; Ugolkov, V.V. Peculiarities of the 5-fluorouracil adsorption on porous aluminosilicates with different morphologies. Appl. Clay Sci. 2019, 184, 105401. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Alikina, Y.A.; Brazovskaya, E.Y.; Vasilenko, N.M. Adsorption Properties and Hemolytic Activity of Porous Aluminosilicates in a Simulated Body Fluid. ChemEngineering 2022, 6, 78. [Google Scholar] [CrossRef]
- Funes, I.G.A.; Peralta, M.E.; Pettinari, G.R.; Carlos, L.; Parolo, M.E. Facile modification of montmorillonite by intercalation and grafting: The study of the binding mechanisms of a quaternary alkylammonium surfactant. Appl. Clay Sci. 2020, 195, 105738. [Google Scholar] [CrossRef]
- Block, K.A.; Trusiak, A.; Katz, A.; Alimova, A.; Wei, H.; Gottlieb, P.; Steiner, J.C. Exfoliation and intercalation of montmorillonite by small peptides. Appl. Clay Sci. 2015, 107, 173–181. [Google Scholar] [CrossRef]
- Pannirselvam, M.; Gupta, R.K.; Bhattacharya, S.N.; Shanks, R.A. Intercalation of Montmorillonite by Interlayer Adsorption and Complex Formation. Adv. Mater. Res. 2007, 29–30, 295–298. [Google Scholar] [CrossRef]
- Hou, H.; Hu Huang, Y.; Gui, R.; Zhang, L.; Tao, Q.; Zhang, C.; Tian, S.; Komarneni, S.; Ping, Q. Preparation and in vitro study of lipid nanoparticles encapsulating drug loaded montmorillonite for ocular delivery. Appl. Clay Sci. 2016, 119, 277–283. [Google Scholar] [CrossRef]
- Baldassari, S.; Komarneni, S.; Mariani, E.; Villa, C. Microwave versus conventional preparation of organoclays from natural and synthetic clays. Appl. Clay Sci. 2006, 31, 134–141. [Google Scholar] [CrossRef]
- Fatullayeva, S.; Tagiyev, D.; Zeynalov, N. A review on enterosorbents and their application in clinical practice: Removal of toxic metals. Colloid Interface Sci. Commun. 2021, 45, 100545. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Shamova, O.V.; Yakovlev, A.V.; Zharkova, M.S. Synthesis and study of the biologically active lysozyme–silver nanoparticles–montmorillonite K10 complexes. Glas. Phys. Chem. 2016, 42, 87–94. [Google Scholar] [CrossRef]
- Phillips, T.D.; Lemke, S.L.; Grant, P.G. Characterization of Clay-Based Enterosorbents for the Prevention of Aflatoxicosis BT—Mycotoxins and Food Safety; DeVries, J.W., Trucksess, M.W., Jackson, L.S., Eds.; Springer: Boston, MA, USA, 2002; pp. 157–171. [Google Scholar] [CrossRef]
- Lipson, S.M.; Stotzky, G. Specificity of virus adsorption to clay minerals. Can. J. Microbiol. 1985, 31, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Permpikul, C.; Sivakorn, C.; Tongyoo, S. In-Hospital Death after Septic Shock Reversal: A Retrospective Analysis of In-Hospital Death among Septic Shock Survivors at Thailand’s Largest National Tertiary Referral Center. Am. J. Trop. Med. Hyg. 2021, 104, 395–402. [Google Scholar] [CrossRef]
- Mitzner, S.R.; Stange, J.; Klammt, S.; Peszynski, P.; Schmidt, R.; Nöldge-Schomburg, G. Extracorporeal detoxification using the molecular adsorbent recirculating system for critically ill patients with liver failure. J. Am. Soc. Nephrol. 2001, 12 (Suppl. S1), S75–S82. [Google Scholar] [CrossRef]
- Greco, I.; Molchanova, N.; Holmedal, E.; Jenssen, H.; Hummel, B.D.; Watts, J.L.; Håkansson, J.; Hansen, P.R.; Svenson, J. Correlation between hemolytic activity, cytotoxicity and systemic in vivo toxicity of synthetic antimicrobial peptides. Sci. Rep. 2020, 10, 13206. [Google Scholar] [CrossRef]
- Chen, Z.; Duan, H.; Tong, X.; Hsu, P.; Han, L.; Morris-Natschke, S.L.; Yang, S.; Liu, W.; Lee, K.-H. Cytotoxicity, Hemolytic Toxicity, and Mechanism of Action of Pulsatilla Saponin D and Its Synthetic Derivatives. J. Nat. Prod. 2018, 81, 465–474. [Google Scholar] [CrossRef]
- Sæbø, I.P.; Bjørås, M.; Franzyk, H.; Helgesen, E.; Booth, J.A. Optimization of the Hemolysis Assay for the Assessment of Cytotoxicity. Int. J. Mol. Sci. 2023, 24, 2914. [Google Scholar] [CrossRef]
- Perderiset, M.; Etienne, L.S.; Bignon, J.; Jaurand, M.C. Interactions of attapulgite (fibrous clay) with human red blood cells. Toxicol. Lett. 1989, 47, 303–309. [Google Scholar] [CrossRef]
- Secchi, G.C.; Rezzonico, A. Hemolytic activity of asbestos dusts. Med. Lav. 1968, 59, 1–5. [Google Scholar] [PubMed]
- Mányai, S.; Kabai, J.; Kis, J.; Süveges, E.; Timár, M. The in vitro hemolytic effect of various clay minerals. Med. Lav. 1969, 60, 331–342. [Google Scholar]
- Mányai, S.; Kabai, J.; Kis, J.; Süveges, É.; Timár, M. The effect of heat treatment on the structure of kaolin and its in vitro hemolytic activity. Environ. Res. 1970, 3, 187–198. [Google Scholar] [CrossRef]
- Light, W.G.; Wei, E.T. Surface charge and asbestos toxicity. Nature 1977, 265, 537–539. [Google Scholar] [CrossRef]
- Harington, J.S.; Miller, K.; Macnab, G. Hemolysis by asbestos. Environ. Res. 1971, 4, 95–117. [Google Scholar] [CrossRef] [PubMed]
- Oscarson, D.W.; Van Scoyoc, G.E.; Ahlrichs, J.L. Lysis of Erythrocytes by Silicate Minerals. Clays Clay Miner. 1986, 34, 74–80. [Google Scholar] [CrossRef]
- Carretero, M.I.; Gomes, C.S.F.; Tateo, F. Chapter 11.5 Clays and Human Health. In Handbook of Clay Science; Bergaya, F., Theng, B.K.G., Lagaly, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 717–741. [Google Scholar] [CrossRef]
- Baek, M.; Lee, J.A.; Choi, S.J. Toxicological effects of a cationic clay, montmorillonite in vitro and in vivo. Mol. Cell. Toxicol. 2012, 8, 95–101. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, K.; Li, Z.; Zhang, H.; Guo, T.; Li, Y.; Zhao, J.; Feng, N. DOC-LS, a new liposome for dermal delivery, and its endocytosis by HaCaT and CCC-ESF-1 cells. IET Nanobiotechnol. 2018, 12, 1037–1041. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Pavlova, S.V.; Yakovlev, A.V. Adsorption and in vitro release of vitamin B1 by synthetic nanoclays with montmorillonite structure. Appl. Clay Sci. 2015, 112–113, 10–16. [Google Scholar] [CrossRef]
- Chen, D.; Tang, Q.; Li, X.; Zhou, X.; Zang, J.; Xue, W.; Xiang, J.; Guo, C. Biocompatibility of magnetic Fe3O4 nanoparticles and their cytotoxic effect on MCF-7 cells. Int. J. Nanomed. 2012, 7, 4973–4982. [Google Scholar] [CrossRef] [PubMed]
- Thomas, F.; Michot, L.J.; Vantelon, D.; Montargès, E.; Prélot, B.; Cruchaudet, M.; Delon, J.F. Layer charge and electrophoretic mobility of smectites, Colloids Surfaces A Physicochem. Eng. Asp. 1999, 159, 351–358. [Google Scholar] [CrossRef]
- Dubinin, M.M. Equivalent equations for micropore adsorption. Bull. Acad. Sci. USSR Div. Chem. Sci. 1978, 27, 456–461. [Google Scholar] [CrossRef]
- Serpinski, V.V.; Jakubov, T.S. Dubinin–Radushkevich Equation as the Equation for the Excess Adsorption Isotherm. Adsorpt. Sci. Technol. 1993, 10, 85–92. [Google Scholar] [CrossRef]
- Bering, B.P.; Dubinin, M.M.; Serpinsky, V.V. Theory of volume filling for vapor adsorption. J. Colloid Interface Sci. 1966, 21, 378–393. [Google Scholar] [CrossRef]
- Shafer, W. Antibacterial Peptide Protocols (Methods in Molecular Biology); Humana: Totowa, NJ, USA, 1997; 269p. [Google Scholar]
- Kopeikin, P.M.; Zharkova, M.S.; Kolobov, A.A.; Smirnova, M.P.; Sukhareva, M.S.; Umnyakova, E.S.; Kokryakov, V.N.; Orlov, D.S.; Milman, B.L.; Balandin, S.V.; et al. Caprine Bactenecins as Promising Tools for Developing New Antimicrobial and Antitumor Drugs. Front. Cell. Infect. Microbiol. 2020, 10, 552905. [Google Scholar] [CrossRef] [PubMed]
- Golubeva, O.Y.; Alikina, Y.A.; Khamova, T.V.; Vladimirova, E.V.; Shamova, O.V. Aluminosilicate Nanosponges: Synthesis, Properties, and Application Prospects. Inorg. Chem. 2021, 60, 17008–17018. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Alikina, Y.A.; Kalashnikova, T.A. Influence of hydrothermal synthesis conditions on the morphology and sorption properties of porous aluminosilicates with kaolinite and halloysite structures. Appl. Clay Sci. 2020, 199, 105879. [Google Scholar] [CrossRef]
- Tolosa, L.; Donato, M.T.; Gómez-Lechón, M.J. General Cytotoxicity Assessment by Means of the MTT Assay. Methods Mol. Biol. 2015, 1250, 333–348. [Google Scholar] [CrossRef]
- Ghasemi, M.; Turnbull, T.; Sebastian, S.; Kempson, I. The MTT Assay: Utility, Limitations, Pitfalls, and Interpretation in Bulk and Single-Cell Analysis. Int. J. Mol. Sci. 2021, 22, 12827. [Google Scholar] [CrossRef]




| Sample Denotation | Composition by Synthesis | Oxide Contents by Analysis, wt % | |||
|---|---|---|---|---|---|
| SiO2 | Al2O3 | MgO | Na2O | ||
| Al0 | NaxMg3Si4O10(OH)2·nH2O | 54.11 | - | 32.52 | 0.11 |
| Al0.2 | NaxAl0.2Mg1.8Si4O10(OH)2·nH2O | 58.10 | 5.32 | 18.31 | 3.52 |
| Al0.5 | NaxAl0.5Mg1.5Si4O10(OH)2·nH2O | 56.01 | 12.08 | 13.73 | 3.47 |
| Al1.0 | NaxAl1.0Mg1.0Si4O10(OH)2·nH2O | 53.89 | 22.82 | 8.04 | 3.2 |
| Al1.8 | NaxAl1.8Mg0.2Si4O10(OH)2·nH2O | 56.96 | 24.81 | 2.10 | 2.99 |
| Samples Denotation | SSA, m2/g | Ws, cm3/g | Vmic, cm3/g | Vmes, cm3/g |
|---|---|---|---|---|
| Al0.2 | 320 | 0.509 | 0.171 | 0.338 |
| Al0.5 | 207 | 0.569 | 0.109 | 0.460 |
| Al1.0 | 190 | 0.722 | 0.036 | 0.686 |
| Al1.8 | 106 | 0.474 | 0.066 | 0.408 |
| Sample Denotation | % Hemolysis before Heat Treatment | % Hemolysis after Heat Treatment |
|---|---|---|
| Al0 | 4.2 ± 0.3 | 19.5 ± 0.6 |
| Al0.5 | 34.3 ± 1.0 | 54.6 ± 1.2 |
| Al1.0 | 86.9 ± 1.2 | 69.1 ± 3.4 |
| Al1.8 | 71.2 ± 2.1 | 54.0 ± 0.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golubeva, O.Y.; Alikina, Y.A.; Brazovskaya, E.Y.; Vasilenko, N.M. Hemolytic Activity and Cytotoxicity of Synthetic Nanoclays with Montmorillonite Structure for Medical Applications. Nanomaterials 2023, 13, 1470. https://doi.org/10.3390/nano13091470
Golubeva OY, Alikina YA, Brazovskaya EY, Vasilenko NM. Hemolytic Activity and Cytotoxicity of Synthetic Nanoclays with Montmorillonite Structure for Medical Applications. Nanomaterials. 2023; 13(9):1470. https://doi.org/10.3390/nano13091470
Chicago/Turabian StyleGolubeva, Olga Yu., Yulia A. Alikina, Elena Yu. Brazovskaya, and Nadezhda M. Vasilenko. 2023. "Hemolytic Activity and Cytotoxicity of Synthetic Nanoclays with Montmorillonite Structure for Medical Applications" Nanomaterials 13, no. 9: 1470. https://doi.org/10.3390/nano13091470
APA StyleGolubeva, O. Y., Alikina, Y. A., Brazovskaya, E. Y., & Vasilenko, N. M. (2023). Hemolytic Activity and Cytotoxicity of Synthetic Nanoclays with Montmorillonite Structure for Medical Applications. Nanomaterials, 13(9), 1470. https://doi.org/10.3390/nano13091470








