Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
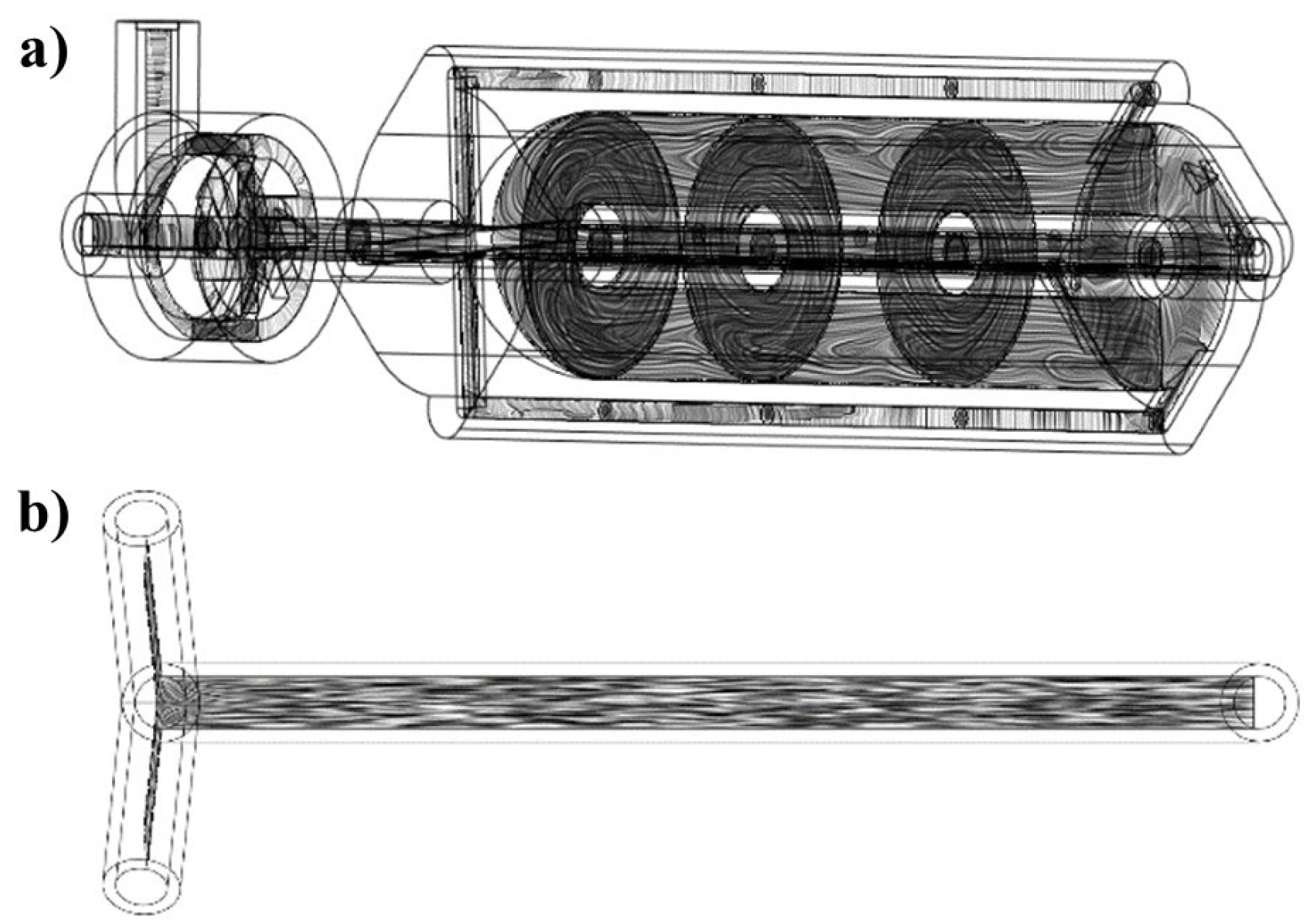
2.2. The Vortex Tube Reactor Model Design and Model Development according to Design of Experiment
2.3. Numerical Analysis
2.3.1. Mixing Index Evaluation
2.3.2. Reynolds Number Expression
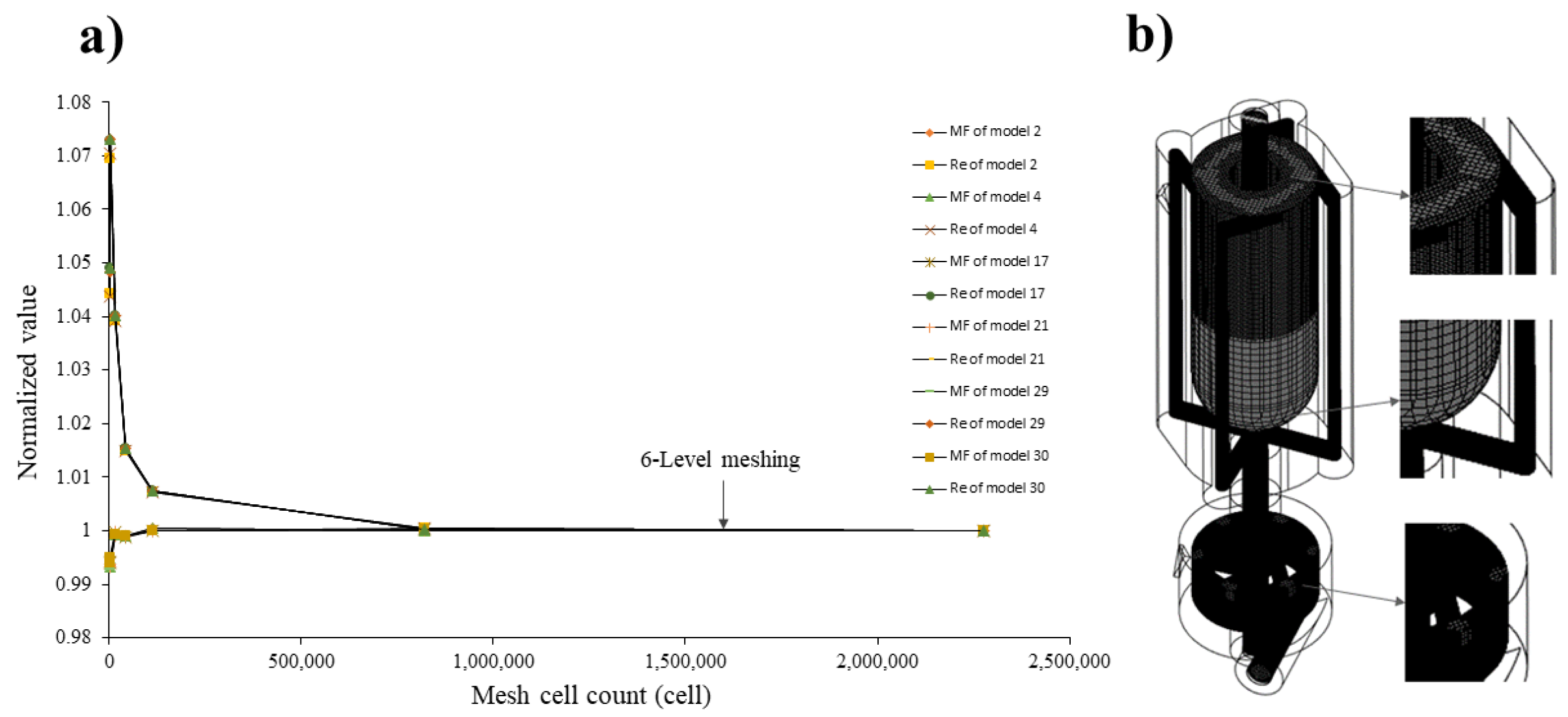
2.4. Mesh Independency Test
2.5. OptiMization of Geometrical Vortex Tube Reactor Model Design
2.6. Fabrication of 3D-Printed Vortex-Tube-Reactor-Based FDM
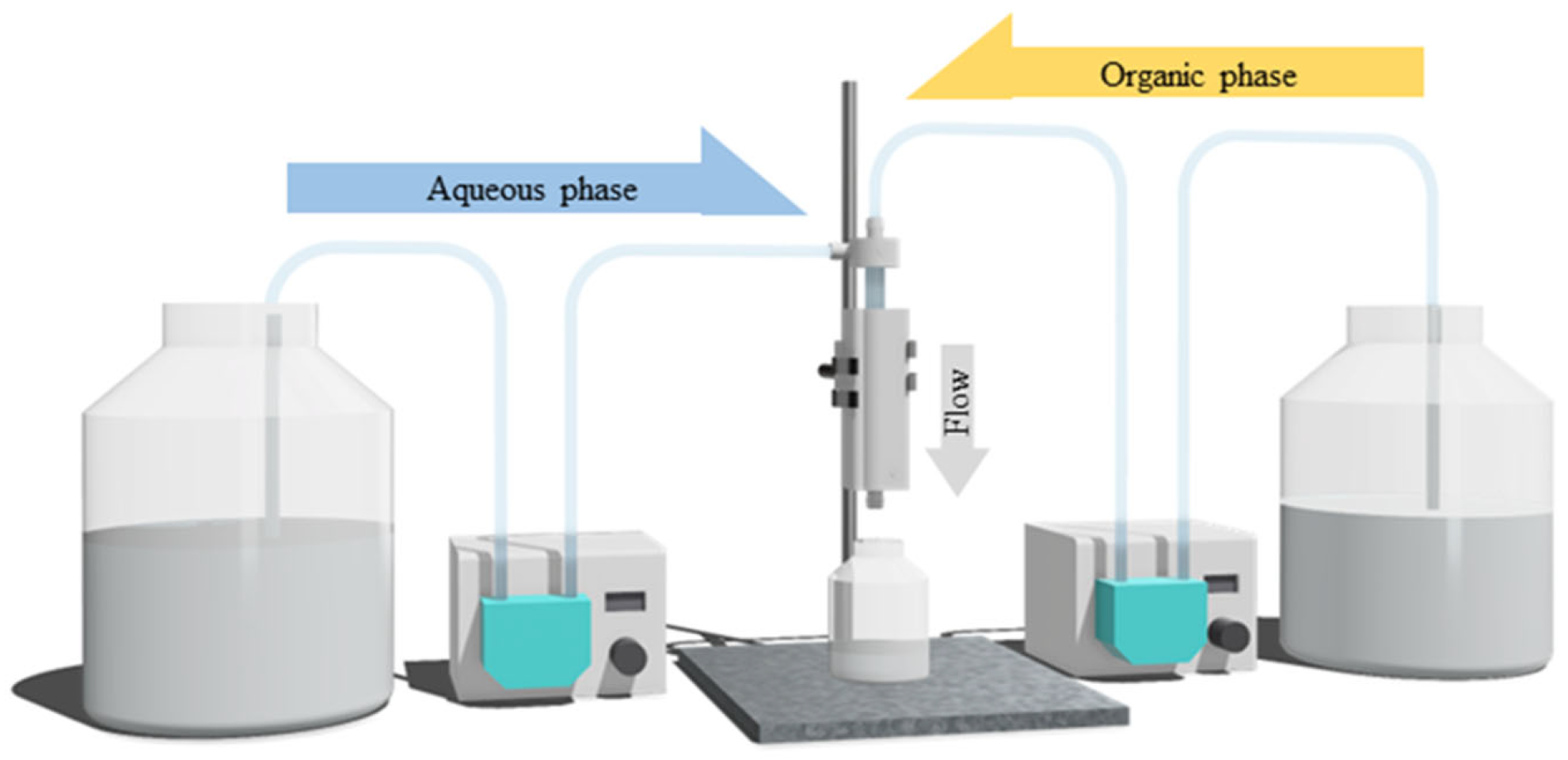
2.7. Dispersion Experiment
2.8. Synthesis and Evaluation of PGA Nanoparticles
2.9. The Physical Characteristics of PGA Nanoparticles
2.10. Statistical Analysis
3. Results and Discussion
3.1. CFD Mesh Sensitivity
3.2. Experimental Design of Vortex Tube Reactor
3.2.1. Mixing Performances of Vortex Tube Reactor (Y1)
3.2.2. Flow Structures of Vortex Tube Reactor (Y2)
3.3. OptiMized Vortex Tube Reactor
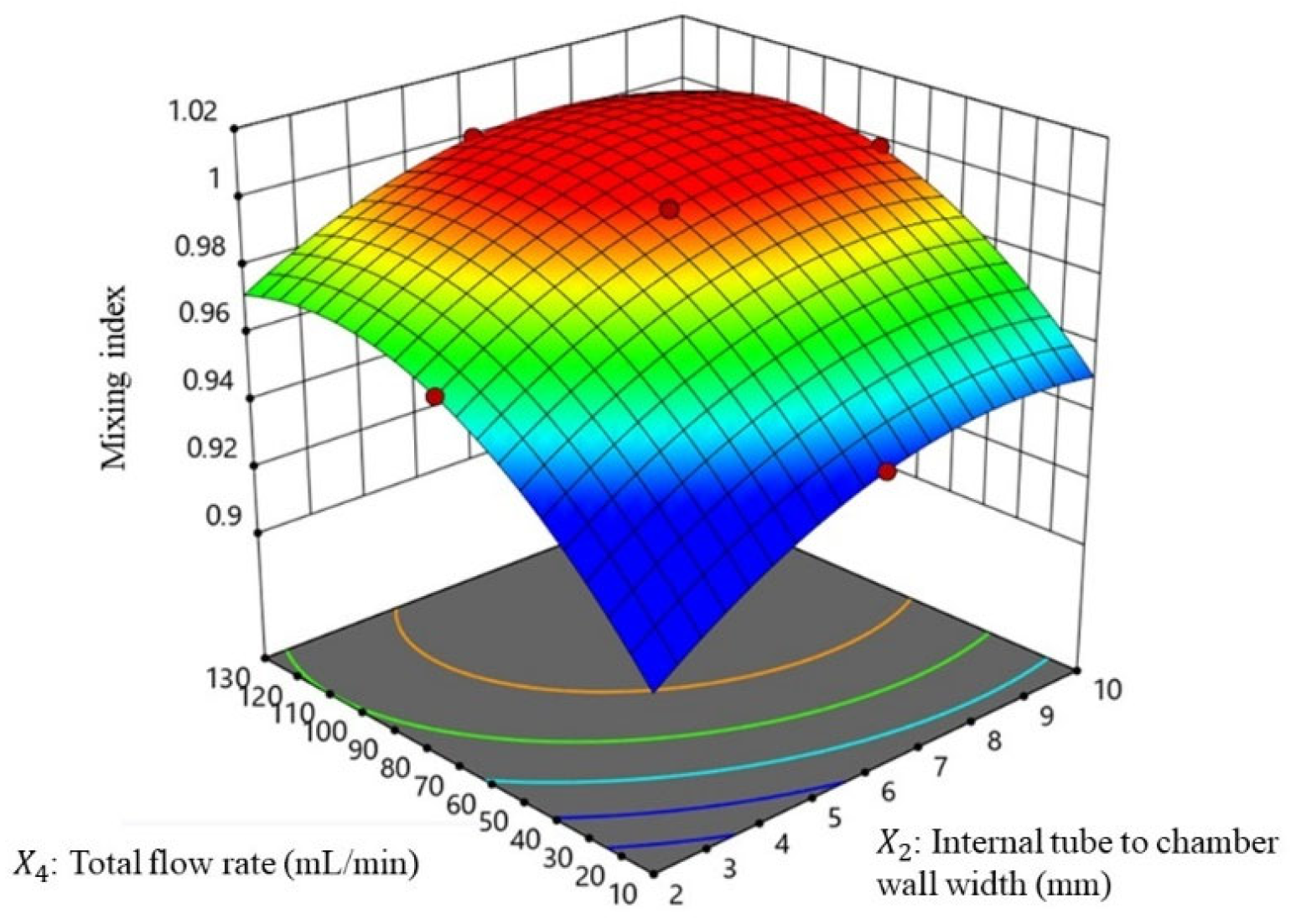
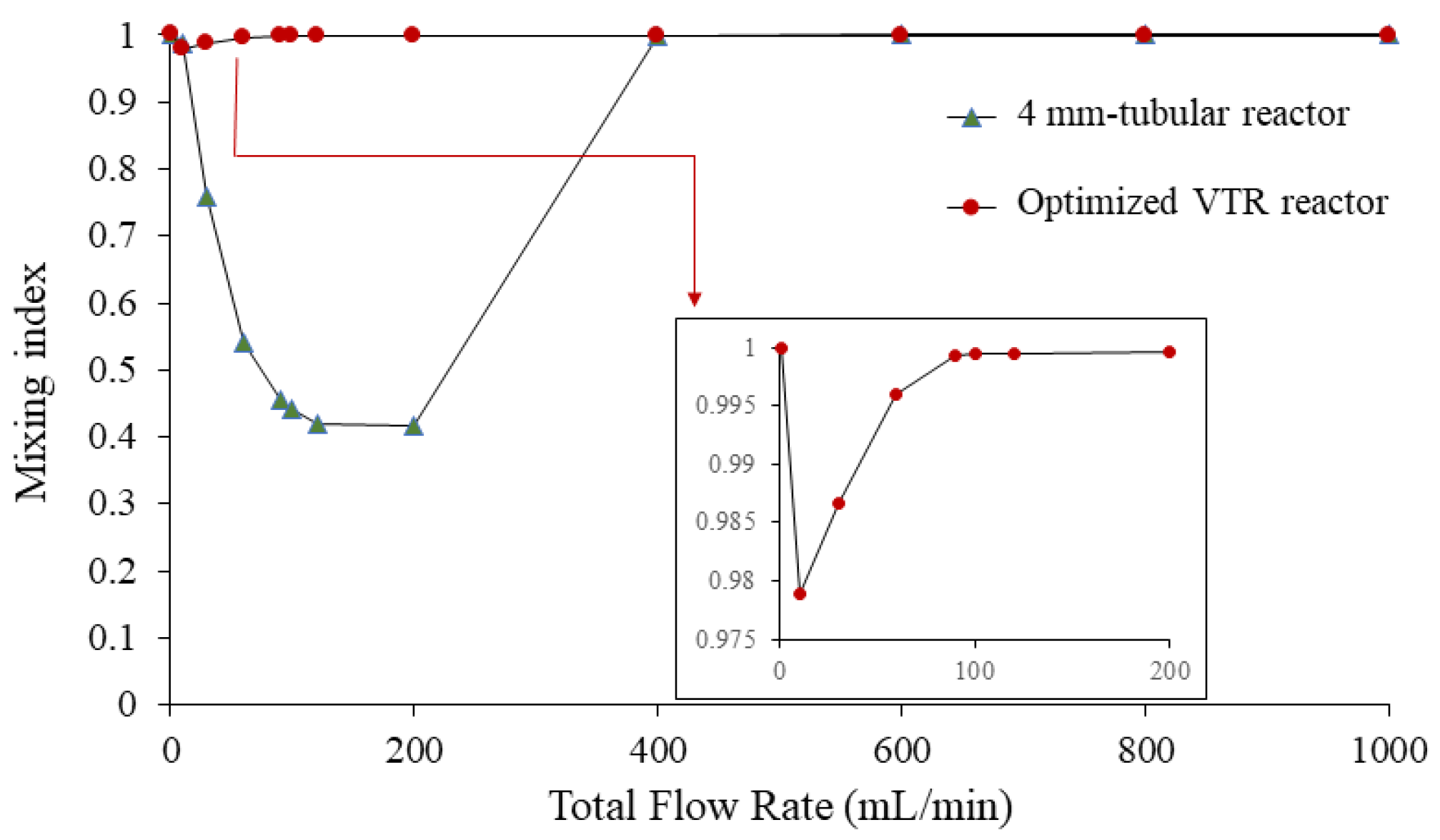
3.3.1. Mixing Performance Evaluation
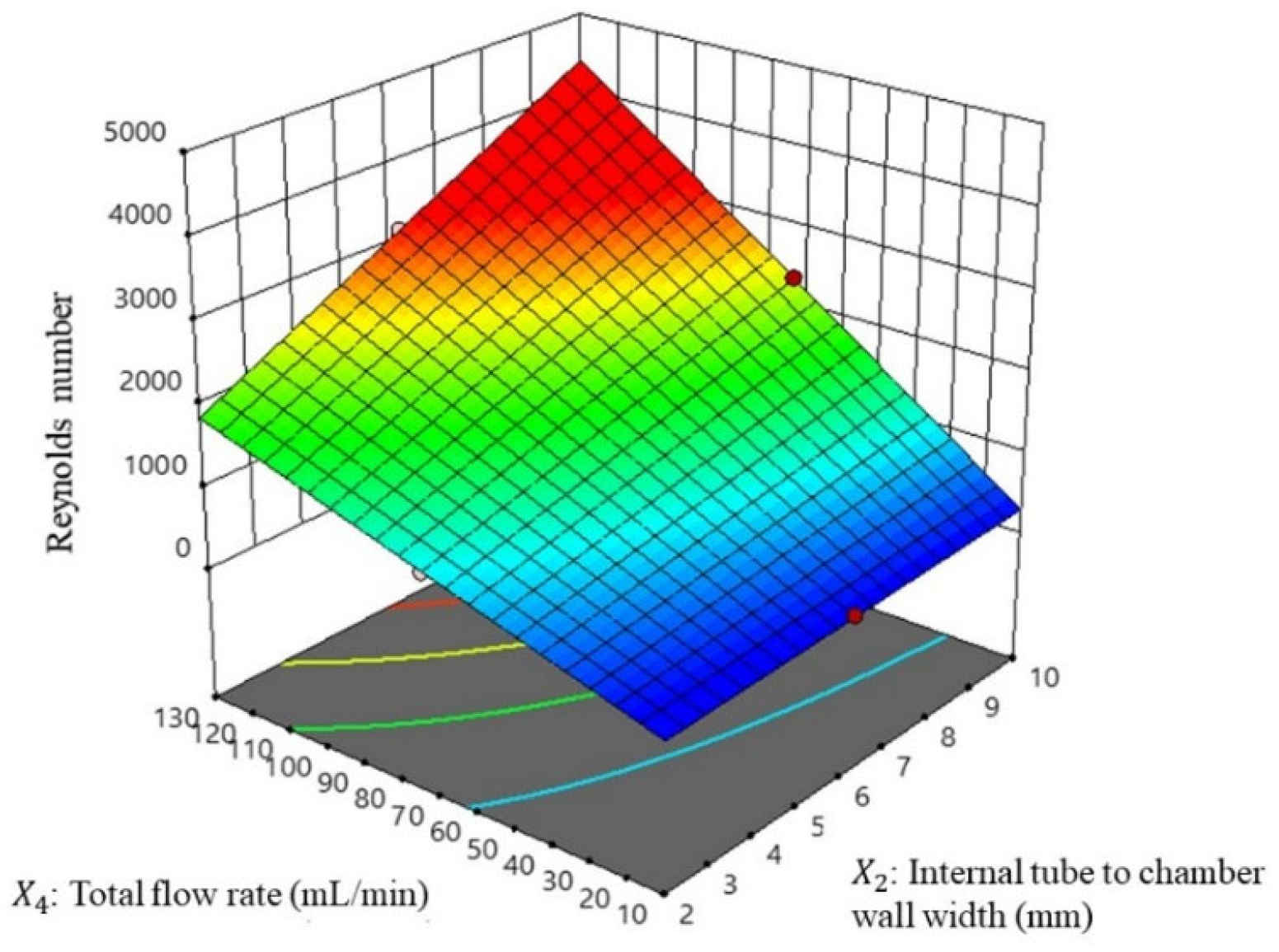
3.3.2. Reynolds Number Evaluation
3.3.3. Relationship of Mixing Index with Different Flow Rates
3.4. Fabrication of Vortex-Tube-Reactor-Based FDM
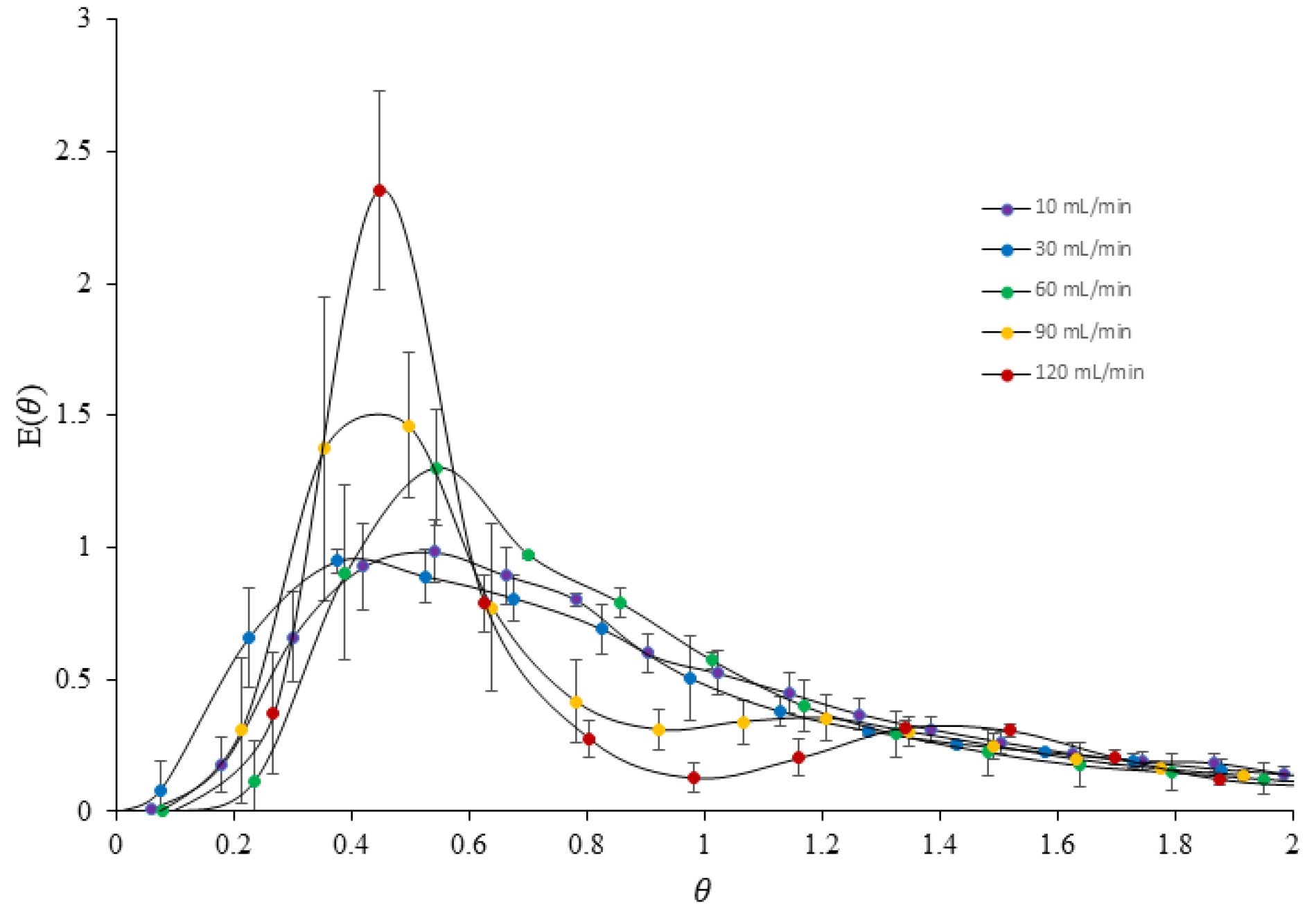
3.5. Dispersion Experiment
3.6. Formulation of PGA Nanoparticles
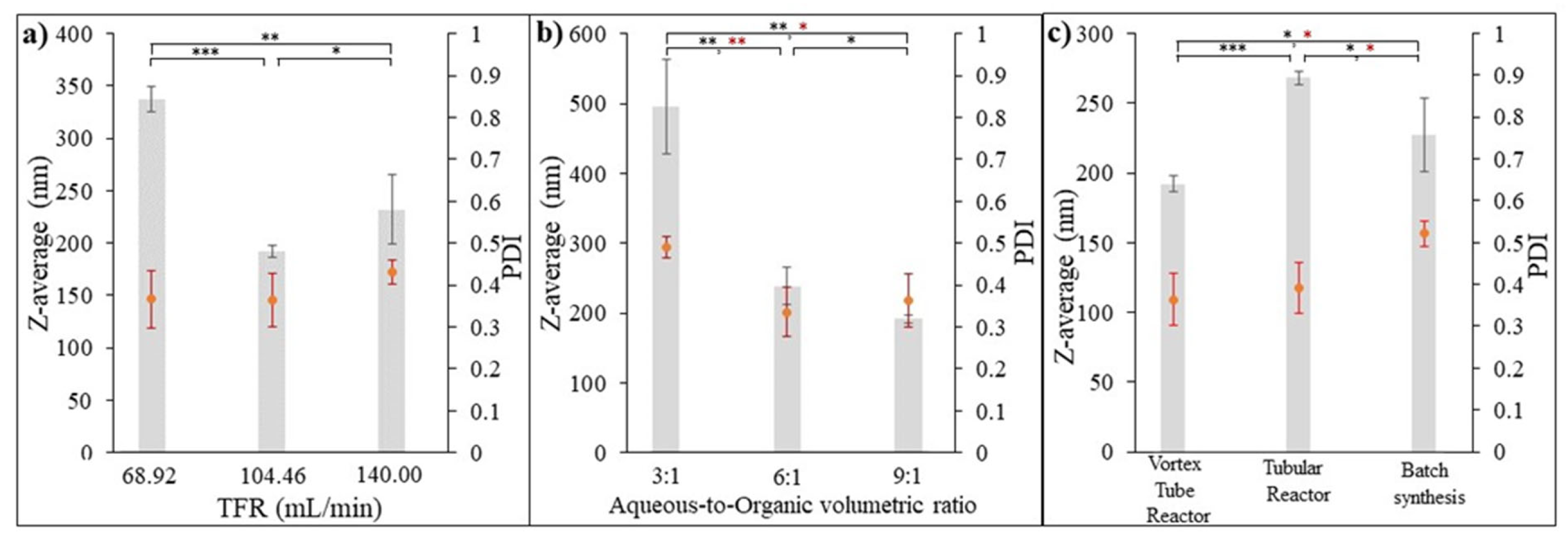
3.6.1. Effect of Input Flow Rate on Formulated PGA Nanoparticles
3.6.2. Effect of Aqueous-to-Organic Volumetric Ratio on Formulated PGA Nanoparticles
3.6.3. Comparison of the Formulated PGA Nanoparticles Using Three Different Synthesis Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Samantaray, P.K.; Little, A.; Haddleton, D.M.; McNally, T.; Tan, B.; Sun, Z.; Huang, W.; Ji, Y.; Wan, C. Poly (glycolic acid)(PGA): A versatile building block expanding high performance and sustainable bioplastic applications. Green Chem. 2020, 22, 4055–4081. [Google Scholar] [CrossRef]
- Aguillón, A.R.; Bezerra, M.A.d.M.; Gomez, M.R.; de Souza, R.O. Continuous-flow chemistry toward sustainable chemical synthesis. In Green Sustainable Process for CheMical and Environmental Engineering and Science; Elsevier: Amsterdam, The Netherlands, 2020; pp. 49–69. [Google Scholar]
- Malet-Sanz, L.; Susanne, F. Continuous flow synthesis. A pharma perspective. J. Med. Chem. 2012, 55, 4062–4098. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, A.R.; Dombrowski, A.W. Emerging trends in flow chemistry and applications to the pharmaceutical industry. J. Med. Chem. 2019, 62, 6422–6468. [Google Scholar] [CrossRef] [PubMed]
- Porta, R.; Benaglia, M.; Puglisi, A. Flow chemistry: Recent developments in the synthesis of pharmaceutical products. Org. Process Res. Dev. 2016, 20, 2–25. [Google Scholar] [CrossRef]
- Khositanon, C.; Adpakpang, K.; Bureekaew, S.; Weeranoppanant, N. Continuous-flow purification of silver nanoparticles and its integration with flow synthesis. J. Flow Chem. 2020, 10, 353–362. [Google Scholar] [CrossRef]
- Yu, C.; Youn, J.R.; Song, Y.S. Tunable electrical resistivity of carbon nanotube filled phase change material via solid-solid phase transitions. Fibers Polym. 2020, 21, 24–32. [Google Scholar] [CrossRef]
- Plutschack, M.B.; Pieber, B.u.; Gilmore, K.; Seeberger, P.H. The hitchhiker’s guide to flow chemistry. Chem. Rev. 2017, 117, 11796–11893. [Google Scholar] [CrossRef] [PubMed]
- Akwi, F.M.; Watts, P. Continuous flow chemistry: Where are we now? Recent applications, challenges and limitations. Chem. Commun. 2018, 54, 13894–13928. [Google Scholar] [CrossRef] [PubMed]
- Rice, H.P.; He, Y.; Muller, F.L.; Bayly, A.E.; Ashe, R.; Karras, A.; Hassanpour, A.; Bourne, R.A.; Fairweather, M.; Hunter, T.N. Physical and numerical characterisation of an agitated tubular reactor (ATR) for intensification of chemical processes. Chem. Eng. Process.-Process Intensif. 2022, 179, 109067. [Google Scholar] [CrossRef]
- Khalde, C.M.; Ramanan, V.; Sangwai, J.S.; Ranade, V.V. Passive mixer cum reactor using threaded inserts: Investigations of flow, mixing, and heat transfer characteristics. Ind. Eng. Chem. Res. 2019, 59, 3943–3961. [Google Scholar] [CrossRef]
- Kouadri, A.; Douroum, E.; Lasbet, Y.; Naas, T.T.; Khelladi, S.; Makhlouf, M. Comparative study of mixing behaviors using non-Newtonian fluid flows in passive micromixers. Int. J. Mech. Sci. 2021, 201, 106472. [Google Scholar] [CrossRef]
- Plouffe, P.; Roberge, D.M.; Macchi, A. Liquid–liquid flow regimes and mass transfer in various micro-reactors. Chem. Eng. J. 2016, 300, 9–19. [Google Scholar] [CrossRef]
- White, F.M. Fluid Mechanics; McGraw-Hill: New York, NY, USA, 1966. [Google Scholar]
- Alimi, O.A.; Akinnawo, C.A.; Onisuru, O.R.; Meijboom, R. 3-D printed microreactor for continuous flow oxidation of a flavonoid. J. Flow Chem. 2020, 10, 517–531. [Google Scholar] [CrossRef]
- Howard, M.P.; Gautam, A.; Panagiotopoulos, A.Z.; Nikoubashman, A. Axial dispersion of Brownian colloids in microfluidic channels. Phys. Rev. Fluids 2016, 1, 044203. [Google Scholar] [CrossRef]
- Desai, D.; Guerrero, Y.A.; Balachandran, V.; Morton, A.; Lyon, L.; Larkin, B.; Solomon, D.E. Towards a microfluidics platform for the continuous manufacture of organic and inorganic nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2021, 35, 102402. [Google Scholar] [CrossRef]
- El-Batsh, H.M. Improving cyclone performance by proper selection of the exit pipe. Appl. Math. Model. 2013, 37, 5286–5303. [Google Scholar] [CrossRef]
- Overcamp, T.J.; Scarlett, S.E. Effect of Reynolds number on the Stokes number of cyclones. Aerosol Sci. Technol. 1993, 19, 362–370. [Google Scholar] [CrossRef]
- Rudyak, V.; Minakov, A. Modeling and optimization of Y-type micromixers. Micromachines 2014, 5, 886–912. [Google Scholar] [CrossRef]
- Le The, H.; Ta, B.Q.; Le-Thanh, H.; Karlsen, F.; Nguyen-Thoi, T. A novel design of passive split and recombination micromixer with trapezoidal zigzag channels. In Proceedings of the 10th IEEE International Conference on Nano/Micro Engineered and Molecular Systems, Xi’an, China, 7–11 April 2015; pp. 28–31. [Google Scholar]
- Hossain, S.; Kim, K.-Y. Mixing performance of a serpentine micromixer with non-aligned inputs. Micromachines 2015, 6, 842–854. [Google Scholar] [CrossRef]
- Kim, K.; Shah, I.; Ali, M.; Aziz, S.; Khalid, M.A.U.; Kim, Y.S.; Choi, K.H. Experimental and numerical analysis of three Y-shaped split and recombination micromixers based on cantor fractal structures. Microsyst. Technol. 2020, 26, 1783–1796. [Google Scholar] [CrossRef]
- Ferziger, J.H.; Perić, M.; Street, R.L. Computational Methods for Fluid DynaMics; Springer: Berlin/Heidelberg, Germany, 2002; Volume 3. [Google Scholar]
- Madana, V.S.T.; Ashraf Ali, B. Numerical investigation of engulfment flow at low Reynolds numbers in a T-shaped microchannel. Phys. Fluids 2020, 32, 072005. [Google Scholar] [CrossRef]
- Missen, R.W. Introduction to CheMical Reaction Engineering and Kinetics; John Wiley & Sons: Hoboken, NJ, USA, 1928. [Google Scholar]
- Zhang, H.-X.; Wang, J.-X.; Shao, L.; Chen, J.-F. Microfluidic fabrication of monodispersed pharmaceutical colloidal spheres of atorvastatin calcium with tunable sizes. Ind. Eng. Chem. Res. 2010, 49, 4156–4161. [Google Scholar] [CrossRef]
- Roces, C.B.; Christensen, D.; Perrie, Y. Translating the fabrication of protein-loaded poly (lactic-co-glycolic acid) nanoparticles from bench to scale-independent production using microfluidics. Drug Deliv. Transl. Res. 2020, 10, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, P.C.; dos Santos-Silva, E.; Streck, L.; Damasceno, I.Z.; Maia, A.M.S.; Fernandes-Pedrosa, M.F.; da Silva-Júnior, A.A. Cationic functionalized biocompatible polylactide nanoparticles for slow release of proteins. Colloids Surf. A: Physicochem. Eng. Asp. 2017, 513, 442–451. [Google Scholar] [CrossRef]
- Kašpar, O.; Koyuncu, A.; Hubatová-Vacková, A.; Balouch, M.; Tokárová, V. Influence of channel height on mixing efficiency and synthesis of iron oxide nanoparticles using droplet-based microfluidics. RSC Adv. 2020, 10, 15179–15189. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Giottonini, K.Y.; Rodríguez-Córdova, R.J.; Gutiérrez-Valenzuela, C.A.; Peñuñuri-Miranda, O.; Zavala-Rivera, P.; Guerrero-Germán, P.; Lucero-Acuña, A. PLGA nanoparticle preparations by emulsification and nanoprecipitation techniques: Effects of formulation parameters. Rsc Adv. 2020, 10, 4218–4231. [Google Scholar] [CrossRef] [PubMed]






| Factor (X) | Unit | DoE Code | ||||
|---|---|---|---|---|---|---|
| −α | −1 | 0 | 1 | +α | ||
| X1: Reactor length | mm | 10 | 30 | 45 | 60 | 90 |
| X2: Internal tube to chamber wall width | mm | 2 | 4 | 6 | 8 | 10 |
| X3: Internal tube length per reactor length (%) | % | 10 | 30 | 45 | 60 | 90 |
| X4: Total flow rate | mL/min | 10 | 40 | 70 | 100 | 130 |
| Factor (X) and Response (Y) | Constraint | Criteria | |
|---|---|---|---|
| X1 | Reactor length | 10 < X1 < 90 | in range |
| X2 | Internal tube to chamber wall width | 2 < X2 < 10 | in range |
| X3 | Percentage of internal tube length/reactor length | 10 < X3 < 90 | in range |
| X4 | Total flow rate | 10 < X4 < 130 | in range |
| Y1 | Mixing index | 0.947361 < Y1 < 1 | target = 1 |
| Y2 | Reynolds number | 239.45 < Y2 < 3132.25 | maximize |
| Printing Condition | Parameter Setting |
|---|---|
| Polymer | PP Filament |
| Nozzle Diameter | 0.4 mm |
| Layer Height | 0.2 mm |
| Print Speed | 50 mm/s |
| Infill | 20% |
| Print Temperature | 210 °C |
| Bed Temperature | 50 °C |
| Run | Mesh No. of 6th Level Meshing (Cells) | Iteration | %Relative Error with 7th Level | |
|---|---|---|---|---|
| Mass Fraction of Water | Reynolds Number | |||
| 1 | 817,569 | 521 | 0.0537 | 0.0785 |
| 2 | 821,352 | 508 | 0.0000 | 0.0417 |
| 3 | 658,343 | 447 | 0.0179 | 0.0749 |
| 4 | 821,325 | 508 | 0.0000 | 0.0417 |
| 5 | 133,316 | 437 | 0.0538 | 0.0732 |
| 6 | 849,119 | 383 | 0.1439 | 0.0677 |
| 7 | 1,031,558 | 488 | 0.1436 | 0.0333 |
| 8 | 1,428,243 | 526 | 0.0717 | 0.0680 |
| 9 | 1,010,688 | 419 | 0.1799 | 0.0942 |
| 10 | 821,352 | 534 | 0.0896 | 0.2214 |
| 11 | 639,576 | 461 | 0.2689 | 0.4146 |
| 12 | 658,350 | 531 | 0.0358 | 0.0438 |
| 13 | 849,077 | 434 | 0.0716 | 0.5331 |
| 14 | 1,032,001 | 459 | 0.1076 | 0.0679 |
| 15 | 821,352 | 401 | 0.0000 | 0.0627 |
| 16 | 1,010,660 | 449 | 0.0539 | 0.3500 |
| 17 | 821,352 | 506 | 0.0179 | 0.0298 |
| 18 | 195,603 | 275 | 0.1073 | 1.4667 |
| 19 | 941,032 | 584 | 0.0000 | 0.0627 |
| 20 | 673,532 | 437 | 0.0179 | 0.0356 |
| 21 | 821,366 | 506 | 0.0179 | 0.0268 |
| 22 | 817,667 | 636 | 0.0000 | 0.0584 |
| 23 | 733,085 | 518 | 0.0358 | 0.3908 |
| 24 | 276,519 | 307 | 0.0896 | 0.1132 |
| 25 | 639,597 | 381 | 0.0179 | 0.0313 |
| 26 | 1,427,656 | 580 | 0.0538 | 0.0313 |
| 27 | 1,013,285 | 557 | 0.0179 | 0.0657 |
| 28 | 630,716 | 624 | 0.0179 | 0.0358 |
| 29 | 821,352 | 506 | 0.0358 | 0.0298 |
| 30 | 821,086 | 506 | 0.0179 | 0.0268 |
| Model | X1 | X2 | X3 | X4 | Y1 | Y2 |
|---|---|---|---|---|---|---|
| 1 | 70 | 8 | 70 | 40 | 0.9956 | 1070.28 |
| 2 | 50 | 6 | 50 | 70 | 0.9978 | 1677.65 |
| 3 | 70 | 8 | 30 | 40 | 0.9958 | 1148.58 |
| 4 | 50 | 6 | 50 | 70 | 0.9979 | 1677.65 |
| 5 | 30 | 8 | 70 | 40 | 0.9928 | 1148.88 |
| 6 | 30 | 4 | 30 | 40 | 0.9886 | 767.20 |
| 7 | 70 | 4 | 30 | 100 | 0.9989 | 1924.36 |
| 8 | 70 | 4 | 70 | 40 | 0.9863 | 765.72 |
| 9 | 30 | 4 | 70 | 40 | 0.9899 | 765.36 |
| 10 | 50 | 6 | 50 | 130 | 0.9997 | 3132.25 |
| 11 | 30 | 8 | 30 | 100 | 0.9995 | 2911.32 |
| 12 | 70 | 8 | 30 | 100 | 0.9997 | 2879.04 |
| 13 | 30 | 4 | 30 | 100 | 0.9998 | 1940.40 |
| 14 | 70 | 4 | 30 | 40 | 0.9943 | 766.20 |
| 15 | 50 | 6 | 50 | 10 | 0.9474 | 239.45 |
| 16 | 30 | 4 | 70 | 100 | 0.9973 | 1936.08 |
| 17 | 50 | 6 | 50 | 70 | 0.9988 | 1679.05 |
| 18 | 10 | 6 | 50 | 70 | 0.9974 | 1753.35 |
| 19 | 90 | 6 | 50 | 70 | 0.9963 | 1676.70 |
| 20 | 50 | 6 | 10 | 70 | 0.9989 | 1685.70 |
| 21 | 50 | 6 | 50 | 70 | 0.9983 | 1679.00 |
| 22 | 70 | 8 | 70 | 100 | 0.9997 | 2876.46 |
| 23 | 30 | 8 | 70 | 100 | 0.9998 | 2905.50 |
| 24 | 50 | 2 | 50 | 70 | 0.9668 | 1007.88 |
| 25 | 30 | 8 | 30 | 40 | 0.9952 | 1150.86 |
| 26 | 70 | 4 | 70 | 100 | 0.9982 | 1916.36 |
| 27 | 50 | 6 | 90 | 70 | 0.9957 | 1676.45 |
| 28 | 50 | 10 | 50 | 70 | 0.9981 | 2349.69 |
| 29 | 50 | 6 | 50 | 70 | 0.9983 | 1679.05 |
| 30 | 50 | 6 | 50 | 70 | 0.9983 | 1679.00 |
| PLS Models | p-Value | R2 | Adjusted R2 |
|---|---|---|---|
| 0.0003 * | 0.9998 | 0.9990 | |
| <0.0001 * | 0.9995 | 0.9995 |
| Mesh No. (Cells) | Iteration | Factor (X) and Response (Y) | Prediction Values | Actual Values | Percentage of Prediction Ability (%) |
|---|---|---|---|---|---|
| 880,082 | 743 | X1 | 79.81 | - | - |
| X2 | 8.95 | - | - | ||
| X3 | 86.74 | - | - | ||
| X4 | 104.46 | - | - | ||
| Y1 | 1.0000 | 0.9995 | 99.95 | ||
| Y2 | 3244.46 | 3240.55 | 99.88 |
| Flow Rate (mL/Min) | (s) | |
|---|---|---|
| 10 | 241.27 | 15,601.41 |
| 30 | 66.48 | 1787.27 |
| 60 | 26.95 | 244.34 |
| 90 | 21.12 | 218.23 |
| 120 | 16.78 | 146.42 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suwanpitak, K.; Sriamornsak, P.; Singh, I.; Sangnim, T.; Huanbutta, K. Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity. Nanomaterials 2023, 13, 2679. https://doi.org/10.3390/nano13192679
Suwanpitak K, Sriamornsak P, Singh I, Sangnim T, Huanbutta K. Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity. Nanomaterials. 2023; 13(19):2679. https://doi.org/10.3390/nano13192679
Chicago/Turabian StyleSuwanpitak, Kittipat, Pornsak Sriamornsak, Inderbir Singh, Tanikan Sangnim, and Kampanart Huanbutta. 2023. "Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity" Nanomaterials 13, no. 19: 2679. https://doi.org/10.3390/nano13192679
APA StyleSuwanpitak, K., Sriamornsak, P., Singh, I., Sangnim, T., & Huanbutta, K. (2023). Three-Dimensional-Printed Vortex Tube Reactor for Continuous Flow Synthesis of Polyglycolic Acid Nanoparticles with High Productivity. Nanomaterials, 13(19), 2679. https://doi.org/10.3390/nano13192679










