Binary Ni-Co-Based Layered Double Hydroxide Nanoneedle Arrays for High Performance of Oxygen Evolution Reaction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Electrocatalyst Synthesis
2.3. Structural Characterization
2.4. Electrochemical Characterization
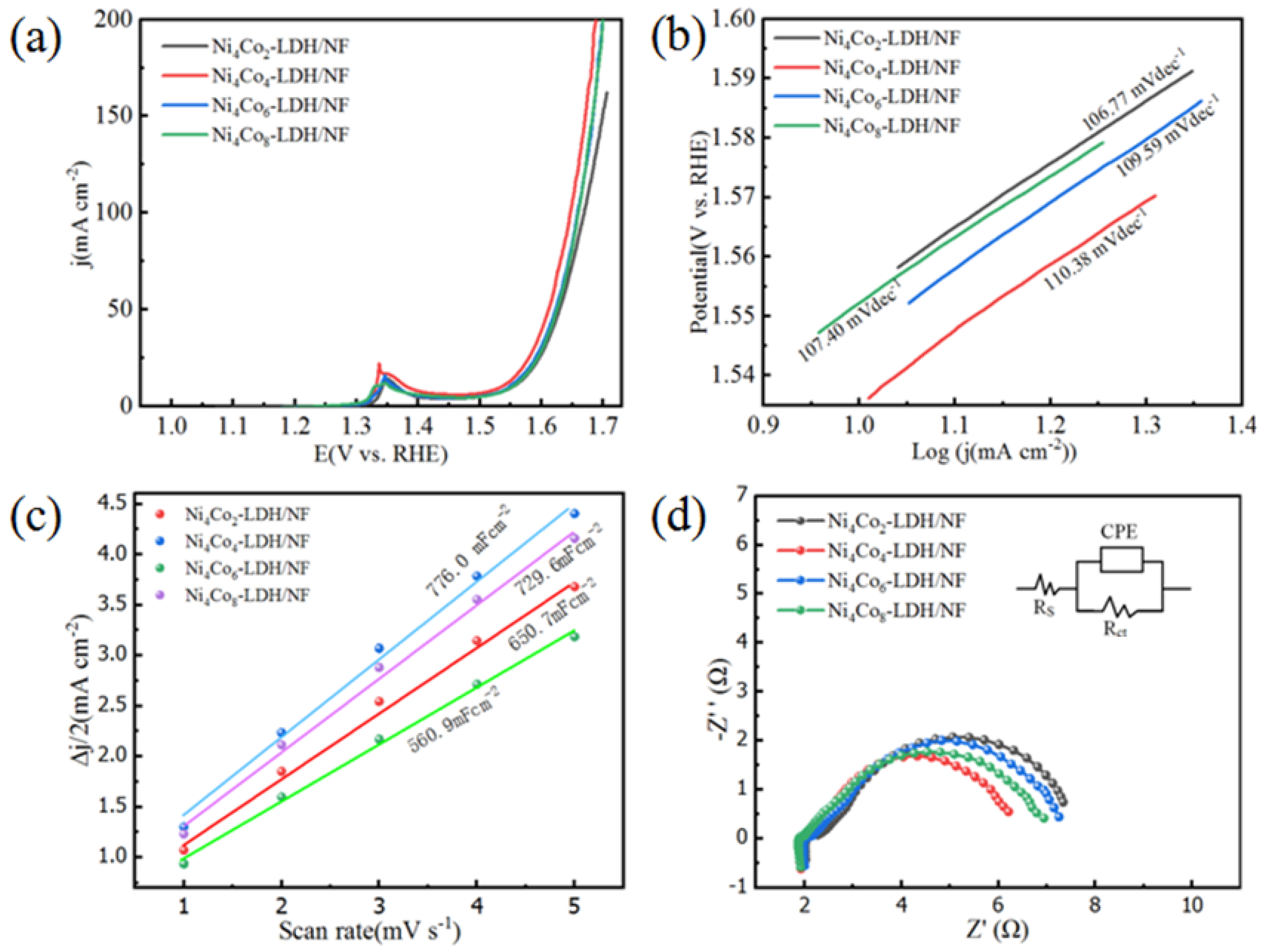
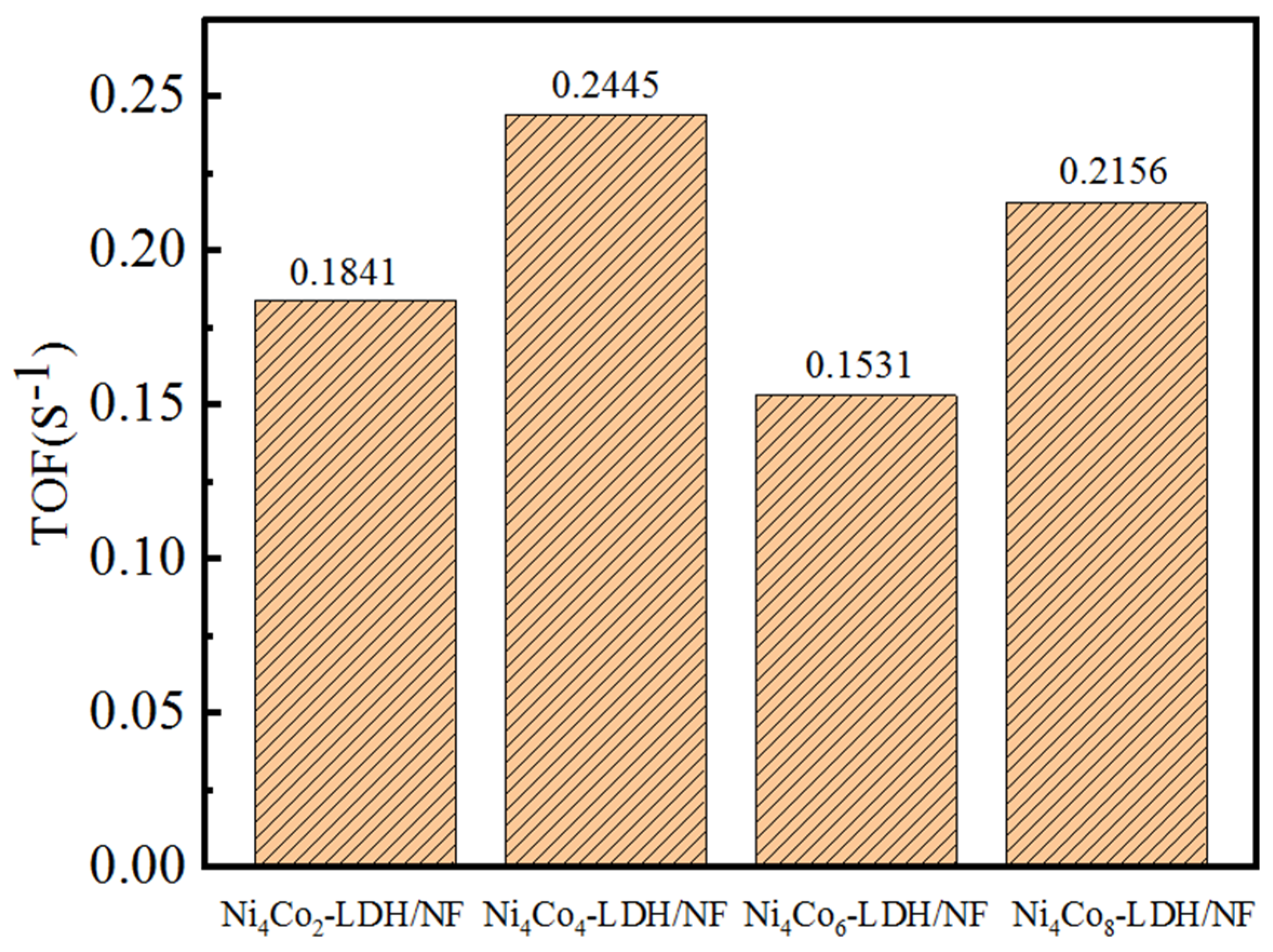
3. Results and Discussion
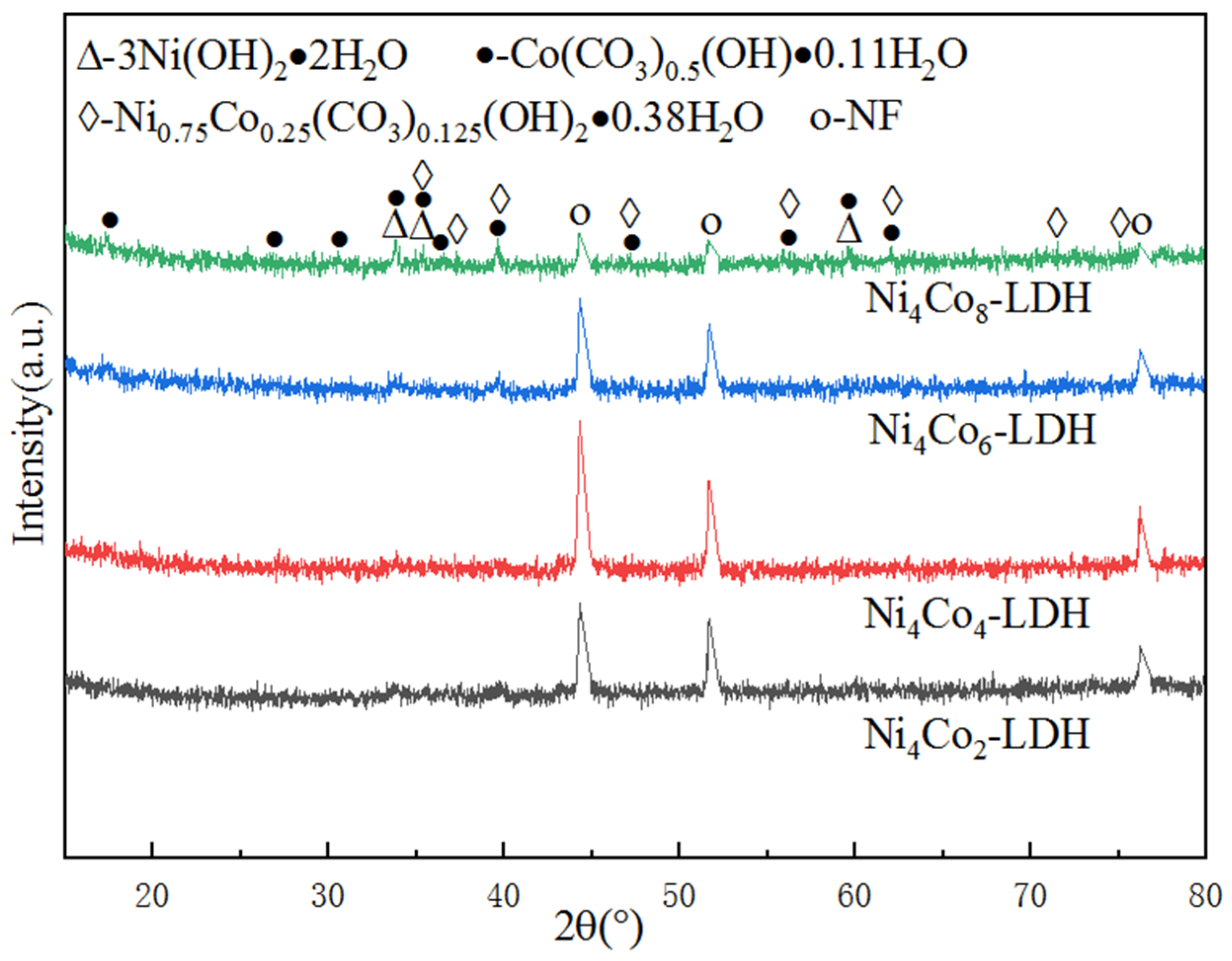
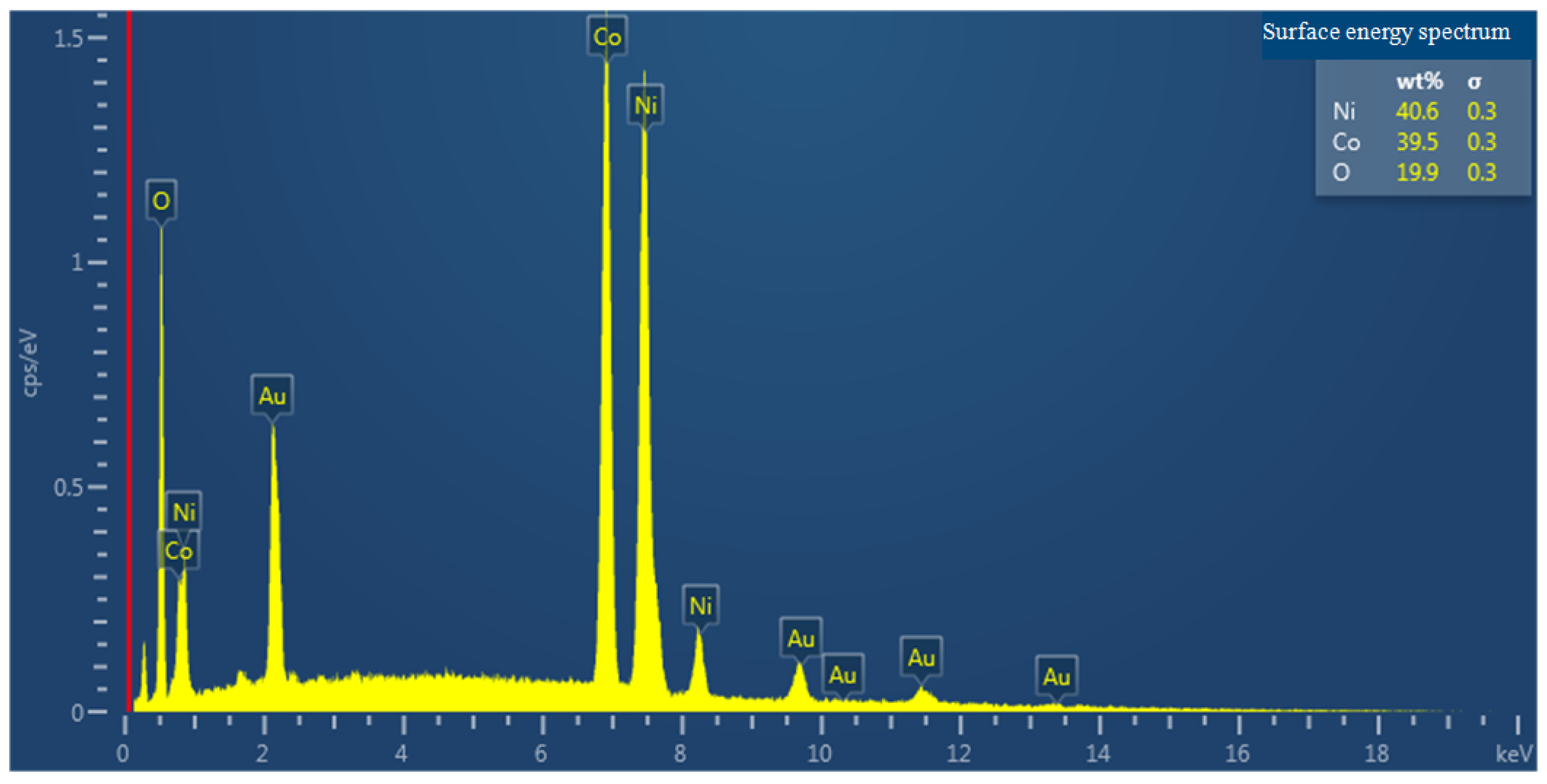
Structural Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Beall, C.E.; Fabbri, E.; Schmidt, T.J. Perovskite oxide based electrodes for the oxygen reduction and evolution reactions: The underlying mechanism. ACS Catal. 2021, 11, 3094–3114. [Google Scholar] [CrossRef]
- Song, F.; Bai, L.; Moysiadou, A.; Lee, S.; Hu, C.; Liardet, L.; Hu, X. Transition metal oxides as electrocatalysts for the oxygen evolution reaction in alkaline solutions: An application-inspired renaissance. J. Am. Chem. Soc. 2018, 140, 7748–7759. [Google Scholar] [CrossRef]
- Yin, H.; Xia, H.; Zhao, S.; Li, K.; Zhang, J.; Mu, S. Atomic level dispersed metal–nitrogen-carbon catalyst toward oxygen reduction reaction: Synthesis strategies and chemical environmental regulation. Energy Environ. Mater. 2021, 4, 5–18. [Google Scholar] [CrossRef]
- Cai, J.; Ding, J.; Wei, D.; Xie, X.; Li, B.; Lu, S.; Zhang, J.; Liu, Y.; Cai, Q.; Zang, S. Coupling of Ru and O-vacancy on 2D Mo-based electrocatalyst via a solid-phase interface reaction strategy for hydrogen evolution reaction. Adv. Energy Mater. 2021, 11, 2100141. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Z.; Qiu, J. Energy-saving hydrogen production by seawater electrolysis coupling sulfion degradation. Adv. Mater. 2022, 34, 2109321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yuan, H.; Li, X.; Wang, Y. Hydrothermal synthesis of NiCo2O4@NiCo2O4 core-shell nanostructures anchored on Ni foam for efficient oxygen evolution reactions catalysts. Coatings 2022, 12, 1240. [Google Scholar] [CrossRef]
- Xiang, W.; Yang, N.; Li, X.; Linnemann, J.; Hagemann, U.; Ruediger, O.; Heidelmann, M.; Falk, T.; Aramini, M.; DeBeer, S. 3D atomic- scale imaging of mixed Co-Fe spinel oxide nanoparticles during oxygen evolution reaction. Nat.Commnn. 2022, 13, 179. [Google Scholar] [CrossRef]
- Qiu, Y.; Feng, Z.; Ji, X.; Liu, J. Surface self-reconstruction of nickel foam triggered by hydrothermal corrosion for boosted water oxidation. Int. J. Hydrogen Energy 2021, 46, 1501. [Google Scholar] [CrossRef]
- Liu, Z.; Zeng, L.; Yu, J.; Yang, L.; Zhang, J.; Zhang, X.; Han, F.; Zhao, L.; Li, X.; Liu, H.; et al. Charge redistribution of Ru nanoclusters on Co3O4 porous nanowire via the oxygen regulation for enhanced hydrogen evolution reaction. Nano Energy 2021, 85, 105940. [Google Scholar] [CrossRef]
- Amirzhanova, A.; Akmansen, N.; Karakaya, I.; Dag, O. Mesoporous MnCo2O4, NiCo2O4, and ZnCo2O4 thin-film electrodes as electro-catalysts for the oxygen evolution reaction in alkaline solutions. ACS Appl. Mater. Interfaces 2021, 4, 2769–2785. [Google Scholar]
- Han, L.; Dong, S.; Wang, E.K. Transition-metal(Co, Ni, and Fe)-based electrocatalysts for the water oxidation reaction. Adv. Mater. 2016, 28, 9266–9291. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Ji, S.; Wang, H.; Linkov, V.; Gai, H.; Liu, F.; Liu, Q.; Wang, R. N-doped 3D porous Ni/C bifunctional electro-catalysts for alkaline water electrolysis. ACS Sustain. Chem. Eng. 2019, 7, 3974–3981. [Google Scholar] [CrossRef]
- Ha, Y.; Shi, L.; Yan, X.; Chen, Z.; Li, Y.; Xu, W.; Wu, R. Multifunctional electro-catalysis on a porous N-doped NiCo2O4@C nanonetwork. ACS Appl. Mater. Interfaces 2019, 11, 45546–45553. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Kim, H.; Kim, J.; Kim, J.; Kim, S.; Ahn, S. Micro-nanoporous MoO2@CoMo heterostructure catalyst for hydrogen evolution reaction. Appl. Catal. B Environ. 2020, 270, 118895. [Google Scholar] [CrossRef]
- Sun, H.; Li, J.; Lv, L.; Li, Z.; Ao, X.; Xu, C.; Xue, X.; Hong, G.; Wang, C. Engineering hierarchical CoSe/NiFe layered-double-hydroxide nanoarrays as high efficient bifunctional electrocatalyst for overall water splitting. J. Power Sources 2019, 425, 138–146. [Google Scholar] [CrossRef]
- Peng, L.; Shen, J.; Zheng, X.; Xiang, R.; Deng, M.; Mao, Z.; Feng, Z.; Zhang, L.; Li, L.; Wei, Z. Rationally design of monometallic NiO-Ni3S2/NF heteronanosheets as bifunctional electrocatalysts for overall water splitting. J. Catal. 2019, 369, 345–351. [Google Scholar] [CrossRef]
- Yang, D.; Cao, L.; Feng, L.; Huang, J.; Kajiyoshi, K.; Feng, Y.; Liu, Q.; Li, W.; Feng, L.; Hai, G. Formation of hierarchical Ni3S2 nanohorn arrays driven by in-situ generation of VS4 nanocrystals for boosting alkaline water splitting. Appl. Catal. B Environ. 2019, 257, 117911. [Google Scholar] [CrossRef]
- Sumboj, A.; Chen, J.; Zong, Y.; Lee, P.S.; Liu, Z. NiMn Layered Double Hydroxide as efficient electrocatalyst for oxygen evolution reaction and its application in rechargeable Zn-Air batteries. Nanoscle 2017, 9, 774–780. [Google Scholar] [CrossRef]
- Wang, C.; Sui, G.; Guo, D.; Li, J.; Ma, X.; Zhuang, Y.; Chai, D. Oxygen vacancies-rich NiCo2O4-4x nanowires assembled on porous carbon derived from cigarette ash: A competitive candidate for hydrogen evolution reaction and supercapacitor. J. Energy Storage 2022, 50, 104280. [Google Scholar] [CrossRef]
- Yang, Y.; Dang, L.; Shearer, M.; Sheng, H.; Li, W.; Chen, J.; Xiao, P.; Zhang, Y.; Hamers, R.; Jin, S. Highly Active Trimetallic NiFeCr Layered Double Hydroxide Electrocatalysts for Oxygen Evolution Reaction. Adv. Energy Mater. 2018, 12, 1703189. [Google Scholar] [CrossRef]
- Qayum, A.; Peng, X.; Yuan, J.; Qu, Y.; Zhou, J.; Huang, Z.; Xia, H.; Liu, Z.; Tan, D.; Chu, P.; et al. Highly durable and efficient Ni-FeOx/FeNi3 electrocatalysts synthesized by a facile in situ combustion-based method for overall water splitting with large current densities. ACS Appl. Mater. Interfaces 2022, 14, 27842–27853. [Google Scholar] [CrossRef]
- Chaudhari, N.; Jin, H.; Kim, B.; Lee, K. Nanostructured materials on 3D nickel foam as electrocatalysts for water splitting. Nanoscale 2017, 9, 12231–12247. [Google Scholar] [CrossRef]
- Cai, M.; Liu, Q.; Xue, Z.; Li, Y.; Fan, Y.; Huang, A.; Li, M.; Croft, M.; Tyson, T.A.; Ke, Z.; et al. Constructing 2D MOFs from 2D LDHs: A highly efficient and durable electrocatalyst for water oxidation. J. Mater. Chem. A 2020, 8, 190–195. [Google Scholar] [CrossRef]
- Wang, H.; Li, J.; Li, K.; Lin, Y.; Chen, J.; Gao, L.; Nicolosi, V.; Xiao, X.; Lee, J.M. Transition metal nitrides for electrochemical energy applications. Chem. Soc. Rev. 2021, 50, 1354–1390. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yan, X.; Guo, P.; Yang, Y.; He, Y.; Liu, J.; Chen, J.; Pan, H.; Wu, R. Inert Mg Incorporation to Break the Activity/Stability Relationship in High-Entropy Layered Hydroxides for the Electrocatalytic Oxygen Evolution Reaction. ACS Catal. 2023, 13, 7698–7706. [Google Scholar] [CrossRef]
- Ning, M.; Wu, L.; Zhang, F.; Wang, D.; Song, S.; Tong, T.; Bao, J.; Chen, S.; Yu, L.; Ren, Z. One-step spontaneous growth of NiFe layered double hydroxide at room temperature for seawater oxygen evolution. Mater. Today Phys. 2021, 19, 100419. [Google Scholar] [CrossRef]
- Yu, S.; Li, J.; Zhang, H. Nanosheet array-like NixMg3-xAl-LDH/rGO hybrids loaded atomically precise Aun nanoclusters for the solvent-free oxidation of benzyl alcohol. J. Catal. 2022, 413, 534–545. [Google Scholar] [CrossRef]
- Harish, S.; Chi, H.; Akbar, I.; Jonghoon, H.; Sunjung, P.; Sangeun, C.; Nabeen, K.; Sang, U.; Bo, H.; Hyunsik, I.; et al. Designing and Tuning the Electronic Structure of Nickel−Vanadium Layered Double Hydroxides for Highly E cient Oxygen Evolution Electrocatalysis. ACS Catal. 2022, 12, 3821–3831. [Google Scholar]
- Badreldin, A.; Nabeeh, A.; Youssef, E.; Mubarak, N.; ElSayed, H.; Mohsen, R.; Ahmed, F.; Wubulikasimu, Y.; Elsaid, K.; Abdel-Wahab, A. Adapting early transition metal and nonmetallic dopants on CoFe oxyhydroxides for enhanced alkaline and neutral pH saline water oxidation. ACS Appl. Energy Mater. 2021, 4, 6942–6956. [Google Scholar] [CrossRef]
- Zheng, K.; Xu, J.; Ruan, J.; Li, X.; Yuan, Z.; Yang, M.; Chen, J.; Xie, F.; Jin, Y.; Wang, N.; et al. Rapid synthesis of porous Ni/Co/Fe-LDHs nanosheets for effective electrochemical oxygen evolution reaction and zinc-air batteries. Int. J. Hydrogen Energy 2022, 47, 26865–26870. [Google Scholar] [CrossRef]
- Baz, A.; Holewinski, A. Predicting macro-kinetic observables in electrocatalysis using the generalized degree of rate control. J. Catal. 2021, 397, 233–244. [Google Scholar] [CrossRef]
- Li, Z.; Shao, M.; An, H.; Wang, Z.; Xu, S.; Wei, M.; Evansa, D.; Duan, X. Fast electrosynthesis of Fe-containing layered double hydroxides arrays toward highly efficient electrocatalytic oxidation reactions. Chem. Sci. 2015, 3, 115–122. [Google Scholar]
- Lv, J.; Wang, L.; Li, R.; Zhang, K.; Zhao, D.; Li, Y.; Li, X.; Huang, X.; Wang, G. Constructing a hetero-interface composed of oxygen vacancy-enriched Co3O4 and crystalline–amorphous NiFe-LDH for oxygen evolution reaction. ACS Catal. 2021, 11, 14338–14351. [Google Scholar] [CrossRef]
- Sun, H.; Li, L.; Chen, Y.; Kim, H.; Xu, X.; Guan, D.; Hu, Z.; Zhang, L.; Shao, Z.; Jung, W.C. Boosting ethanol oxidation by NiOOH-CuO nano-heterostructure for energy-saving hydrogen production and biomass upgrading. Appl. Catal. B Environ. 2023, 325, 122388. [Google Scholar] [CrossRef]
- Yu, M.; Budiyanto, E.; Tüysüz, H. Principles of water electrolysis and recent progress in Cobalt-, Nickel-, and Iron-based oxides for the oxygen evolution reaction. Angew. Chem. Int. Ed. 2022, 61, e202103824. [Google Scholar]
- Sun, H.; Xu, X.; Kim, H.; Jung, W.; Zhou, W.; Shao, Z. Electrochemical water splitting: Bridging the gaps between fundamental research and industrial applications. Energy Environ. Mater. 2022, 1, e12441. [Google Scholar] [CrossRef]
- Pintado, S.; Goberna-Ferron, S.; Escudero-Adan, E.C.; Galan-Mascaros, J.R. Fast and persistent electrocatalytic water oxidation by Co-Fe prussian blue coordination polymers. J. Am. Chem. Soc. 2013, 135, 13270–13273. [Google Scholar] [CrossRef]
- Ge, R.; Ren, X.; Ji, X.; Liu, Z.; Du, G.; Asiri, A.M.; Sun, X.; Chen, L. Benzoate anion-intercalated layered cobalt hydroxide nanoarray: An efficient electrocatalyst for the oxygen evolution reaction. Chem. Sus. Chem. 2017, 10, 4004–4008. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, G.; Zhao, J.; Liang, J.; Sun, L.; Liu, G.; Ji, B.; Yan, X.; Zhang, J. Self-growth Ni2P nanosheet arrays with cationic vacancy defects as a highly efficient bifunctional electrocatalyst for overall water splitting. J. Colloid Interf. Sci. 2020, 561, 638–646. [Google Scholar] [CrossRef]
- Friebel, D.; Louie, M.W.; Bajdich, M.; Sanwald, K.E.; Cai, Y.; Wise, A.M.; Cheng, M.; Sokaras, D.; Weng, T.; Alonso-Mori, R.; et al. Identification of highly active Fe sites in (Ni, Fe)OOH for electrocatalytic water splitting. J. Am. Chem. Soc. 2015, 137, 1305. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Xia, Z.; Zhang, Z.; Ma, Y.; Qu, Y. One-step synthesis of multi-walled carbon nanotubes/ultra-thin Ni(OH)2 nanoplate composite as efficient catalysts for water oxidation. J. Mater. Chem. A 2014, 2, 11799–11806. [Google Scholar] [CrossRef]
- Rovetta, A.A.; Browne, M.P.; Harvey, A.; Godwin, I.J.; Coleman, J.N.; Lyons, M.E. Cobalt hydroxide nanoflakes and their application as supercapacitors and oxygen evolution catalysts. Nanotechnology 2017, 28, 375401. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, F.; Chen, W.; Xiang, Q.; Ma, Y.; Zhu, H.; Tao, P.; Song, C.; Shang, W.; Deng, T.; et al. Coupling interface constructions of MoS2/Fe5Ni4S8 heterostructures for efficient electrochemical water splitting. Adv. Mater. 2018, 30, 1803151. [Google Scholar] [CrossRef]
- Ling, C.; Shi, L.; Ouyang, Y.; Chen, Q.; Wang, J. Transition metal-promoted V2CO2(MXenes): A new and highly active catalyst for hydrogen evolution reaction. Adv. Sci. 2016, 3, 1600180. [Google Scholar] [CrossRef] [Green Version]
- Sayeed, M.A.; Herd, T.; O’Mullane, A.P. Direct electrochemical formation of nanostructured amorphous Co(OH)2 on gold electrodes with enhanced activity for the oxygen evolution reaction. J. Mater. Chem. A 2016, 4, 991–999. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Zhao, Y.; Li, M.; Fan, G.; Yang, L.; Li, F. A strong coupled 2D metal-organic framework and ternary layered double hydroxide hierarchical nanocomposite as an excellent electrocatalyst for the oxygen evolution reaction. Electrochim. Acta 2019, 307, 275–284. [Google Scholar] [CrossRef]






| Sample | SBET (m2/g) | Vtotal (cm3/g) |
|---|---|---|
| Ni4Co2-LDH/NF | 8.6960 | 0.009751 |
| Ni4Co4-LDH/NF | 10.1848 | 0.011258 |
| Ni4Co6-LDH/NF | 11.9787 | 0.013901 |
| Ni4Co8-LDH/NF | 7.3907 | 0.008607 |
| Sample | FWHM | Crystallite Size (nm) |
|---|---|---|
| Ni4Co2-LDH/NF | 0.306 | 29 |
| Ni4Co4-LDH/NF | 0.220 | 43 |
| Ni4Co6-LDH/NF | 0.187 | 68 |
| Ni4Co8-LDH/NF | 0.137 | 89 |
| Catalyst | Current Density (mA cm−2) | Overpotential (mV) | Reference |
|---|---|---|---|
| Ni(OH)2 | 10 | 595 | [41] |
| NiCo hydroxide | 10 | 460 | [39] |
| Co(OH)2/NF | 10 | 280 | [42] |
| NiCo-LDH | 10 | 367 | [43] |
| NiCo-NS | 10 | 334 | [44] |
| Co(OH)2 | 10 | 360 | [45] |
| ZIF-67/CoNiAl-LDH/NF | 10 | 303 | [46] |
| Ni-Co-based LDH arrays | 10 | 305 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Z.; Zhou, Z.; Li, S.; Tan, G.; Chen, H.; Ge, Z.; Chen, C.; Wang, G. Binary Ni-Co-Based Layered Double Hydroxide Nanoneedle Arrays for High Performance of Oxygen Evolution Reaction. Nanomaterials 2023, 13, 1941. https://doi.org/10.3390/nano13131941
Lu Z, Zhou Z, Li S, Tan G, Chen H, Ge Z, Chen C, Wang G. Binary Ni-Co-Based Layered Double Hydroxide Nanoneedle Arrays for High Performance of Oxygen Evolution Reaction. Nanomaterials. 2023; 13(13):1941. https://doi.org/10.3390/nano13131941
Chicago/Turabian StyleLu, Zhi, Zhihao Zhou, Shilin Li, Gongliang Tan, Hangtian Chen, Zishuo Ge, Chong Chen, and Guangxin Wang. 2023. "Binary Ni-Co-Based Layered Double Hydroxide Nanoneedle Arrays for High Performance of Oxygen Evolution Reaction" Nanomaterials 13, no. 13: 1941. https://doi.org/10.3390/nano13131941
APA StyleLu, Z., Zhou, Z., Li, S., Tan, G., Chen, H., Ge, Z., Chen, C., & Wang, G. (2023). Binary Ni-Co-Based Layered Double Hydroxide Nanoneedle Arrays for High Performance of Oxygen Evolution Reaction. Nanomaterials, 13(13), 1941. https://doi.org/10.3390/nano13131941





