Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications
Abstract
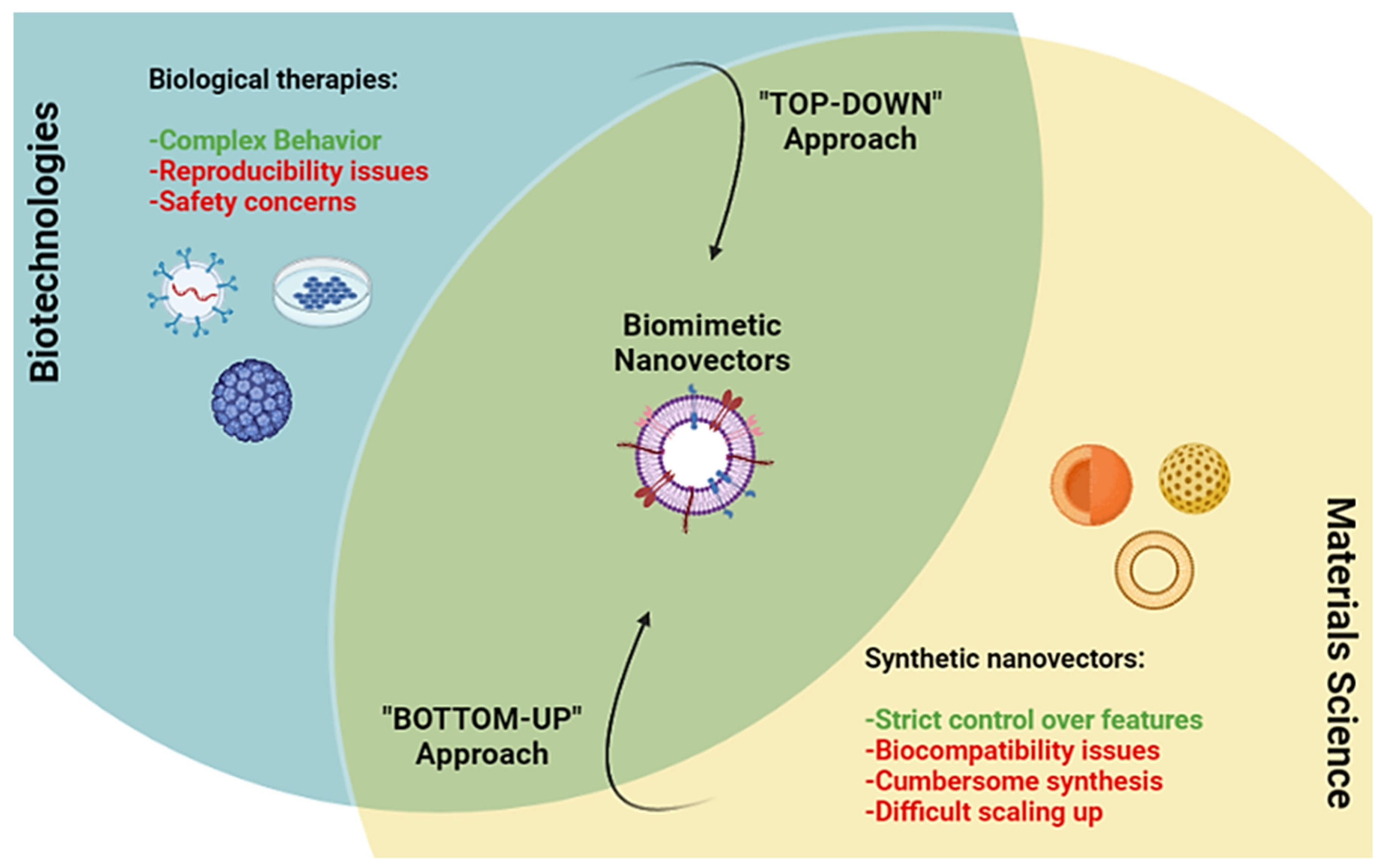
:1. Introduction
Overview of the Classes, Biological Sources, and Functions of Membrane Coated and Membrane Derived Biomimetic Nanovectros
2. Membrane Coated and Membrane-Based Nanomaterials
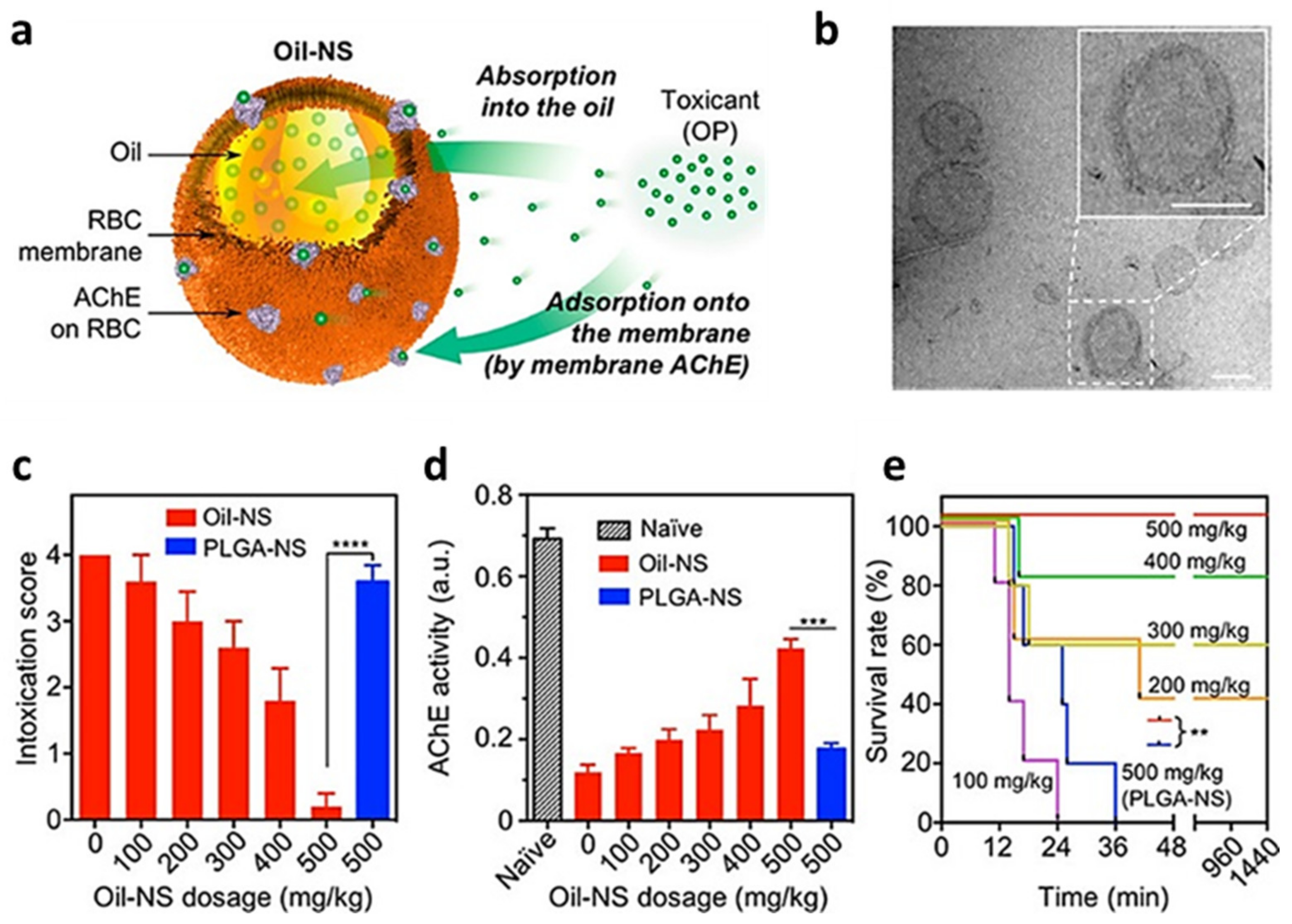
2.1. RBC-Coated NPs
2.2. PT-Coated NPs
2.3. Leukocytes-Coated NPs
2.4. Tumor Cell-Coated NPs
2.5. Multiple Membrane-Coated NPs
2.6. NPs Coated with Other Membranes
3. Cell Membrane and Membrane Derived Nanovesicles
3.1. Membrane Nanovesicles
3.2. Membrane Proteins-Based Nanovectors
3.3. Techniques Used for Membrane Coating or Membrane/Based Nanovectors Assembly
4. Current Hurdles and Future Perspectives on the Development of Biomimetic NPs
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Baig, I.K.N.; Falathabe, W. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the clinic: An update. Bioeng. Transl. Med. 2019, 4, e10143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deirram, N.; Zhang, C.; Kermaniyan, S.S.; Johnston, A.P.R.; Such, G.K. H-Responsive Polymer Nanoparticles for Drug Delivery. Macromol. Rapid Commun. 2019, 40, e1800917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Huang, Y.; Kumar, A.; Tan, A.; Jin, S.; Mozhi, A.; Liang, X.J. H-sensitive nano-systems for drug delivery in cancer therapy. Biotechnol. Adv. 2014, 32, 693–710. [Google Scholar] [CrossRef]
- Sadowska-Bartosz, I.; Bartosz, G. Redox nanoparticles: Synthesis, properties and perspectives of use for treatment of neurodegenerative diseases. J. Nanobiotechnol. 2018, 16, 87. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Zhao, G.; Su, W.-K.; Shuai, Q. Enzyme-Responsive Nanoparticles for Anti-tumor Drug Delivery. Front. Chem. 2020, 30, 647. [Google Scholar] [CrossRef]
- Increasingly translational delivery. Nat. Biomed. Eng. 2021, 5, 947–948. [CrossRef]
- Lu, R.-M.; Hwang, Y.-C.; Liu, I.-J.; Lee, C.-C.; Tsai, H.-Z.; Li, H.-J.; Wu, H.-C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1. [Google Scholar] [CrossRef]
- Bulcha, J.T.; Wang, Y.; Ma, H.; Tai, P.W.L.; Gao, G. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target. Ther. 2021, 8, 53. [Google Scholar] [CrossRef]
- Wiklander, O.P.B.; Brennan, M.Á.; Lötvall, J.; Breakefield, X.O.; El Andaloussi, S. Advances in therapeutic applications of extracellular vesicles. Sci. Transl. Med. 2019, 11, eaav8521. [Google Scholar] [CrossRef]
- Wang, L.L.; Janes, M.E.; Kumbhojkar, N.; Kapate, N.; Clegg, J.R.; Prakash, S.; Heavey, M.K.; Zhao, Z.; Anselmo, A.C.; Mitragotri, S. Cell therapies in the clinic. Bioeng. Transl. Med. 2021, 6, e10214. [Google Scholar] [CrossRef]
- Sathish, J.G.; Sethu, S.; Bielsky, M.-C.; de Haan, L.; French, N.S.; Govindappa, K.; Green, J.; Griffiths, C.E.M.; Holgate, S.; Jones, D.; et al. Challenges and approaches for the development of safer immunomodulatory biologics. Nat. Rev. Drug Discov. 2013, 12, 306–324. [Google Scholar] [CrossRef]
- Brennan, F.R.; Andrews, L.; Arulanandam, A.R.; Blumel, J.; Fikes, J.; Grimaldi, C.; Lansita, J.; Loberg, L.I.; MacLachlan, T.; Milton, M.; et al. Current strategies in the non-clinical safety assessment of biologics: New targets, new molecules, new challenges. Regul. Toxicol. Pharmacol. 2018, 98, 98–107. [Google Scholar] [CrossRef]
- Fang, R.H.; Kroll, A.V.; Gao, W.W.; Zhang, L.F. Cell Membrane Coating Nanotechnology. Adv. Mater. 2018, 30, e1706759. [Google Scholar] [CrossRef]
- Zhang, M.; Du, Y.; Wang, S.; Chen, B. A Review of Biomimetic Nanoparticle Drug Delivery Systems Based on Cell Membranes. Drug Des. Dev. Ther. 2020, 14, 5495–5503. [Google Scholar] [CrossRef]
- Fam, S.Y.; Chee, C.F.; Yong, C.Y.; Ho, K.L.; Mariatulqabtiah, A.R.; Tan, W.S. Stealth Coating of Nanoparticles in Drug-Delivery Systems. Nanomaterials 2020, 10, 787. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Fang, R.H.; Zhang, L. Biomimetic Nanosponges for Treating Antibody-Mediated Autoimmune Diseases. Bioconjug. Chem. 2018, 29, 870–877. [Google Scholar] [CrossRef]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef]
- De Castilla, P.E.M.; Tong, L.; Huang, C.; Sofias, A.M.; Pastorin, G.; Chen, X.; Storm, G.; Schiffelers, R.M.; Wang, J.-W. Extracellular vesicles as a drug delivery system: A systematic review of preclinical studies. Adv. Drug Deliv. Rev. 2021, 175, 113801. [Google Scholar] [CrossRef]
- Khodabakhsh, F.; Salimian, M.; Hedayati, M.H.; Ahangari Cohan, R.; Norouzian, D. Challenges and advancements in the pharmacokinetic enhancement of therapeutic proteins. Prep. Biochem. Biotechnol. 2021, 51, 519–529. [Google Scholar] [CrossRef]
- Luk, B.T.; Zhang, L. Cell membrane-camouflaged nanoparticles for drug delivery. J. Control. Release 2015, 220, 600–607. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Wang, F.; Lu, Y.; Qi, J.; Deng, L.; Sousa, F.; Sarmento, B.; Xu, X.; Cui, W. Recent advances of erythrocyte-mimicking nanovehicles: From bench to bedside. J. Control. Release 2019, 314, 81–91. [Google Scholar] [CrossRef]
- Li, Z.; Hu, S.; Cheng, K. Platelets and their biomimetics for regenerative medicine and cancer therapies. J. Mater. Chem. B 2018, 6, 7354–7365. [Google Scholar] [CrossRef]
- Löwenberg, E.C.; Meijers, J.; Levi, M. Platelet-vessel wall interaction in health and disease. Neth. J. Med. 2012, 68, 242–251. [Google Scholar]
- Ley, K.; Laudanna, C.; Cybulsky, M.I.; Nourshargh, S. Getting to the site of inflammation: The leukocyte adhesion cascade updated. Nat. Rev. Immunol. 2007, 7, 678–689. [Google Scholar] [CrossRef]
- Rossin, A.; Miloro, G.; Hueber, A.-O. TRAIL and FasL Functions in Cancer and Autoimmune Diseases: Towards an Increasing Complexity. Cancers 2019, 11, 639. [Google Scholar] [CrossRef] [Green Version]
- Sharpe, A.H.; Pauken, K.E. The diverse functions of the PD1 inhibitory pathway. Nat. Rev. Immunol. 2018, 18, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, M.; Abualrous, E.T.; Sticht, J.; Álvaro-Benito, M.; Stolzenberg, S.; Noé, F.; Freund, C. Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation. Front. Immunol. 2017, 8, 292. [Google Scholar] [CrossRef] [Green Version]
- Aravindhan, S.; Ejam, S.S.; Lafta, M.H.; Markov, A.; Yumashev, A.V.; Ahmadi, M. Mesenchymal stem cells and cancer therapy: Insights into targeting the tumour vasculature. Cancer Cell 2021, 21, 158. [Google Scholar] [CrossRef] [PubMed]
- Lokhov, P.G.; Balashova, E. Design of universal cancer vaccines using natural tumor vessel-specific antigens (SANTAVAC). Hum. Vaccin. Immunother. 2015, 11, 689–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garofalo, M.; Villa, A.; Crescenti, D.; Marzagalli, M.; Kuryk, L.; Limonta, P.; Mazzaferro, V.; Ciana, P. Heterologous and cross-species tropism of cancer-derived extracellular vesicles. Theranostics 2019, 9, 5681–5693. [Google Scholar] [CrossRef]
- Hu, C.-M.J.; Zhang, L.; Aryal, S.; Cheung, C.; Fang, R.H.; Zhang, L. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc. Natl. Acad. Sci. USA 2011, 108, 10980–10985. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Zhang, B.; Zhang, L.; Wu, X.; Li, H.; Shen, S.; Luo, Z.; Liu, X.; Hu, Y.; Pang, Z.; et al. Biomimetic nanoparticles delivered hedgehog pathway inhibitor to modify tumour microenvironment and improved chemotherapy for pancreatic carcinoma. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1088–1101. [Google Scholar] [CrossRef] [Green Version]
- Zou, Y.; Liu, Y.; Yang, Z.; Zhang, D.; Lu, Y.; Zheng, M.; Xue, X.; Geng, J.; Chung, R.; Shi, B. Effective and Targeted Human Orthotopic Glioblastoma Xenograft Therapy via a Multifunctional Biomimetic Nanomedicine. Adv. Mater. 2018, 30, e1803717. [Google Scholar] [CrossRef]
- Fu, S.; Liang, M.; Wang, Y.; Cui, L.; Gao, C.; Chu, X.; Liu, Q.; Feng, Y.; Gong, W.; Yang, M.; et al. Dual-Modified Novel Biomimetic Nanocarriers Improve Targeting and Therapeutic Efficacy in Glioma. ACS Appl. Mater. Interfaces 2019, 11, 1841–1854. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, W.; Wang, Q.; Hang, L.; Wang, Y.; Wang, Y. ROS-sensitive biomimetic nanocarriers modulate tumor hypoxia for synergistic photodynamic chemotherapy. Biomater. Sci. 2019, 7, 3706–3716. [Google Scholar] [CrossRef]
- Li, H.; Jin, K.; Luo, M.; Wang, X.; Zhu, X.; Liu, X.; Jiang, T.; Zhang, Q.; Wang, S.; Pang, Z. Size Dependency of Circulation and Biodistribution of Biomimetic Nanoparticles: Red Blood Cell Membrane-Coated Nanoparticles. Cells 2019, 8, 881. [Google Scholar] [CrossRef] [Green Version]
- Yu, W.; He, X.; Yang, Z.; Yang, X.; Xiao, W.; Liu, R.; Xie, R.; Qin, L.; Gao, H. Sequentially responsive biomimetic nanoparticles with optimal size in combination with checkpoint blockade for cascade synergetic treatment of breast cancer and lung metastasis. Biomaterials 2019, 217, 119309. [Google Scholar] [CrossRef]
- Zhang, Z.; Qian, H.; Huang, J.; Sha, H.; Zhang, H.; Yu, L.; Liu, B.; Hua, D.; Qian, X. Anti-EGFR-iRGD recombinant protein modified biomimetic nanoparticles loaded with gambogic acid to enhance targeting and antitumor ability in colorectal cancer treatment. Int. J. Nanomed. 2018, 13, 4961–4975. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Vyas, C.; Kim, G.G.; Choi, P.S.; Hur, M.G.; Yang, S.D.; Kong, Y.B.; Lee, E.J.; Park, J.H. Red Blood Cell Membrane Bioengineered Zr-89 Labelled Hollow Mesoporous Silica Nanosphere for Overcoming Phagocytosis. Sci. Rep. 2019, 9, 7419. [Google Scholar] [CrossRef]
- Meng, Q.F.; Cheng, Y.; Huang, Q.; Zan, M.; Xie, W.; Sun, Y.; Li, R.; Wei, X.; Guo, S.S.; Zhao, X.Z.; et al. Biomimetic Immunomagnetic Nanoparticles with Minimal Nonspecific Biomolecule Adsorption for Enhanced Isolation of Circulating Tumor Cells. ACS Appl. Mater. Interfaces 2019, 11, 28732–28739. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, Y.; Chen, M.; Zhuang, J.; Fang, R.H.; Gao, W.; Zhang, L. Biomimetic Nanosponges Suppress in vivo Lethality Induced by the Whole Secreted Proteins of Pathogenic Bacteria. Small 2019, 15, e1804994. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, Y.; Zhuang, J.; Lee, J.H.; Wang, L.; Fang, R.H.; Gao, W.; Zhang, L. Cell-Membrane-Cloaked Oil Nanosponges Enable Dual-Modal Detoxification. ACS Nano 2019, 13, 7209–7215. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Shen, Q.; Huang, K.; Zheng, T.; Cheng, L.; Zhang, Z.; Yu, Y.; Liao, G.; Wang, X.; Li, C. Oriented Assembly of Cell-Mimicking Nanoparticles via a Molecular Affinity Strategy for Targeted Drug Delivery. ACS Nano 2019, 13, 5268–5277. [Google Scholar] [CrossRef]
- Luk, B.T.; Jiang, Y.; Copp, J.A.; Hu, C.J.; Krishnan, N.; Gao, W.; Li, S.; Fang, R.H.; Zhang, L. Biomimetic Targeting of Nanoparticles to Immune Cell Subsets via Cognate Antigen Interactions. Mol. Pharm. 2018, 15, 3723–3728. [Google Scholar] [CrossRef]
- Liang, H.; Huang, K.; Su, T.; Li, Z.; Hu, S.; Dinh, P.U.; Wrona, E.A.; Shao, C.; Qiao, L.; Vandergriff, A.C.; et al. Mesenchymal Stem Cell/Red Blood Cell-Inspired Nanoparticle Therapy in Mice with Carbon Tetrachloride-Induced Acute Liver Failure. ACS Nano 2018, 12, 6536–6544. [Google Scholar] [CrossRef]
- Wang, H.; Wu, J.; Williams, G.R.; Fan, Q.; Niu, S.; Wu, J.; Xie, X.; Zhu, L.M. Platelet-membrane-biomimetic nanoparticles for targeted antitumor drug delivery. J. Nanobiotechnol. 2019, 17, 60. [Google Scholar] [CrossRef]
- Bahmani, B.; Gong, H.; Luk, B.T.; Haushalter, K.J.; DeTeresa, E.; Previti, M.; Zhou, J.; Gao, W.; Bui, J.D.; Zhang, L.; et al. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat. Commun. 2021, 12, 1999. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Chen, J.; Liu, Y.; Cheng, X.; Yang, F.; Gu, N. Platelet Membrane Biomimetic Magnetic Nanocarriers for Targeted Delivery and in Situ Generation of Nitric Oxide in Early Ischemic Stroke. ACS Nano 2020, 14, 2024–2035. [Google Scholar] [CrossRef]
- Wang, B.; Chen, G.; Urabe, G.; Xie, R.; Wang, Y.; Shi, X.; Guo, L.W.; Gong, S.; Kent, K.C. A paradigm of endothelium-protective and stent-free anti-restenotic therapy using biomimetic nanoclusters. Biomaterials 2018, 178, 293–301. [Google Scholar] [CrossRef]
- Kim, J.K.; Uchiyama, S.; Gong, H.; Stream, A.; Zhang, L.; Nizet, V. Engineered Biomimetic Platelet Membrane-Coated Nanoparticles Block Staphylococcus aureus Cytotoxicity and Protect against Lethal Systemic Infection. Engineering 2021, 7, 1149–1156. [Google Scholar] [CrossRef]
- Wei, X.; Gao, J.; Fang, R.H.; Luk, B.T.; Kroll, A.V.; Dehaini, D.; Zhou, J.; Kim, H.W.; Gao, W.; Lu, W.; et al. Nanoparticles camouflaged in platelet membrane coating as an antibody decoy for the treatment of immune thrombocytopenia. Biomaterials 2016, 111, 116–123. [Google Scholar] [CrossRef] [Green Version]
- Wei, X.; Zhang, G.; Ran, D.; Krishnan, N.; Fang, R.H.; Gao, W.; Spector, S.A.; Zhang, L. T-Cell-Mimicking Nanoparticles Can Neutralize HIV Infectivity. Adv. Mater. 2018, 30, e1802233. [Google Scholar] [CrossRef]
- Zhang, Q.; Honko, A.; Zhou, J.; Gong, H.; Downs, S.N.; Vasquez, J.H.; Fang, R.H.; Gao, W.; Griffiths, A.; Zhang, L. Cellular Nanosponges Inhibit SARS-CoV-2 Infectivity. Nano Lett. 2020, 8, 5570–5574. [Google Scholar] [CrossRef]
- Zhuang, J.; Duan, Y.; Zhang, Q.; Gao, W.; Li, S.; Fang, R.H.; Zhang, L. Multimodal Enzyme Delivery and Therapy Enabled by Cell Membrane-Coated Metal-Organic Framework Nanoparticles. Nano Lett. 2020, 20, 4051–4058. [Google Scholar] [CrossRef]
- Chen, F.; Geng, Z.; Wang, L.; Zhou, Y.; Liu, J. Biomimetic Nanoparticles Enabled by Cascade Cell Membrane Coating for Direct Cross-Priming of T Cells. Small 2022, 18, e2104402. [Google Scholar] [CrossRef]
- Kang, M.; Hong, J.; Jung, M.; Kwon, S.P.; Song, S.Y.; Kim, H.Y.; Lee, J.R.; Kang, S.; Han, J.; Koo, J.H.; et al. T-Cell-Mimicking Nanoparticles for Cancer Immunotherapy. Adv. Mater. 2020, 32, e2003368. [Google Scholar] [CrossRef]
- Zhai, Y.; Wang, J.; Lang, T.; Kong, Y.; Rong, R.; Cai, Y.; Ran, W.; Xiong, F.; Zheng, C.; Wang, Y.; et al. T lymphocyte membrane-decorated epigenetic nanoinducer of interferons for cancer immunotherapy. Nat. Nanotechnol. 2021, 16, 1271–1280. [Google Scholar] [CrossRef]
- Jin, J.; Krishnamachary, B.; Barnett, J.D.; Chatterjee, S.; Chang, D.; Mironchik, Y.; Wildes, F.; Jaffee, E.M.; Nimmagadda, S.; Bhujwalla, Z.M. Human Cancer Cell Membrane-Coated Biomimetic Nanoparticles Reduce Fibroblast-Mediated Invasion and Metastasis and Induce T-Cells. ACS Appl. Mater. Interfaces 2019, 11, 7850–7861. [Google Scholar] [CrossRef]
- Fang, R.H.; Kroll, A.; Zhang, L. Nanoparticle-Based Manipulation of Antigen-Presenting Cells for Cancer Immunotherapy. Small 2015, 11, 5483–5496. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Krishnan, N.; Zhou, J.; Chekuri, S.; Wei, X.; Kroll, A.V.; Yu, C.L.; Duan, Y.; Gao, W.; Fang, R.H.; et al. Engineered Cell-Membrane-Coated Nanoparticles Directly Present Tumor Antigens to Promote Anticancer Immunity. Adv. Mater. 2020, 32, e2001808. [Google Scholar] [CrossRef]
- Kroll, A.V.; Fang, R.; Jiang, Y.; Zhou, J.; Wei, X.; Yu, C.L.; Gao, J.; Luk, B.T.; Dehaini, D.; Gao, W.; et al. Nanoparticulate Delivery of Cancer Cell Membrane Elicits Multiantigenic Antitumor Immunity. Adv. Mater. 2017, 29, 1703969. [Google Scholar] [CrossRef]
- Gong, C.; Yu, X.; You, B.; Wu, Y.; Wang, R.; Han, L.; Wang, Y.; Gao, S.; Yuan, Y. Macrophage-cancer hybrid membrane-coated nanoparticles for targeting lung metastasis in breast cancer therapy. J. Nanobiotechnol. 2020, 18, 92. [Google Scholar] [CrossRef]
- Soto, F.; Wang, J.; Ahmed, R.; Demirci, U. Medical Micro/Nanorobots in Precision Medicine. Adv. Sci. 2020, 7, 2002203. [Google Scholar] [CrossRef]
- De Ávila, B.E.-F.; Angsantikul, P.; Ramírez-Herrera, D.E.; Soto, F.; Teymourian, H.; Dehaini, D.; Chen, Y.; Zhang, L.; Wang, J. Hybrid biomembrane-functionalized nanorobots for concurrent removal of pathogenic bacteria and toxins. Sci. Robot. 2018, 18, eaat0485. [Google Scholar] [CrossRef] [Green Version]
- Gao, W.; Fang, R.; Thamphiwatana, S.; Luk, B.T.; Li, J.; Angsantikul, P.; Zhang, Q.; Hu, C.M.; Zhang, L. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles. Nano Lett. 2015, 15, 1403–1409. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Chen, Y.; Lo, C.; Zhuang, J.; Angsantikul, P.; Zhang, Q.; Wei, X.; Zhou, Z.; Obonyo, M.; Fang, R.H.; et al. Inhibition of Pathogen Adhesion by Bacterial Outer Membrane-Coated Nanoparticles. Angewandte Chemie Int. Ed. Engl. 2019, 58, 11404–11408. [Google Scholar] [CrossRef]
- Angsantikul, P.; Thamphiwatana, S.; Zhang, Q.; Spiekermann, K.; Zhuang, J.; Fang, R.H.; Gao, W.; Obonyo, M.; Zhang, L. Coating nanoparticles with gastric epithelial cell membrane for targeted antibiotic delivery against Helicobacter pylori infection. Adv. Ther. 2018, 1, 1800016. [Google Scholar] [CrossRef]
- Yang, F.; Meng, L.; Lin, S.; Wu, F.; Liu, J. Polyethyleneimine-complexed charge-reversed yeast cell walls for the enhanced oral delivery of pseudovirus-based antigens. Chem. Commun. 2021, 57, 12768–12771. [Google Scholar] [CrossRef]
- Oieni, J.; Levy, L.; Khait, N.L.; Yosef, L.; Schoen, B.; Fliman, M.; Shalom-Luxenburg, H.; Dayan, N.M.; D’Atri, D.; Anavy, N.C.; et al. Nano-Ghosts: Biomimetic membranal vesicles, technology and characterization. Methods 2020, 177, 126–134. [Google Scholar] [CrossRef]
- Oieni, J.; Lolli, A.; D’Atri, D.; Kops, N.; Yayon, A.; van Osch, G.J.V.M.; Machluf, M. Nano-ghosts: Novel biomimetic nano-vesicles for the delivery of antisense oligonucleotides. J. Control. Release 2021, 333, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liu, Y.; Fu, B.L.; Xu, L.L.; Wang, B. MicroRNA-21: An Emerging Player in Bone Diseases. Front. Pharmacol. 2021, 7, 722804. [Google Scholar] [CrossRef] [PubMed]
- Kaneti, L.; Bronshtein, T.; Dayan, N.M.; Kovregina, I.; Khait, N.L.; Lupu-Haber, Y.; Fliman, M.; Schoen, B.W.; Kaneti, G.; Machluf, M. Nanoghosts as a Novel Natural Nonviral Gene Delivery Platform Safely Targeting Multiple Cancers. Nano Lett. 2016, 16, 1574–1582. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, R.; Corbo, C.; Martinez, J.O.; Taraballi, F.; Evangelopoulos, M.; Minardi, S.; Yazdi, I.K.; Zhao, P.; De Rosa, E.; Sherman, M.B.; et al. Biomimetic proteolipid vesicles for targeting inflamed tissues. Nat. Mater. 2016, 15, 1037–1046. [Google Scholar] [CrossRef]
- Han, X.; Shen, S.; Fan, Q.; Chen, G.; Archibong, E.; Dotti, G.; Liu, Z.; Gu, Z.; Wang, C. Red blood cell-derived nanoerythrosome for antigen delivery with enhanced cancer immunotherapy. Sci. Adv. 2019, 5, eaaw6870. [Google Scholar] [CrossRef] [Green Version]
- Zou, M.Z.; Li, Z.H.; Bai, X.F.; Liu, C.J.; Zhang, X.Z. Hybrid Vesicles Based on Autologous Tumor Cell Membrane and Bacterial Outer Membrane to Enhance Innate Immune Response and Personalized Tumor Immunotherapy. Nano Lett. 2021, 21, 8609–8618. [Google Scholar] [CrossRef]
- Martinez, J.O.; Molinaro, R.; Hartman, K.A.; Boada, C.; Sukhovershin, R.; De Rosa, E.; Kirui, D.; Zhang, S.; Evangelopoulos, M.; Carter, A.M.; et al. Biomimetic nanoparticles with enhanced affinity towards activated endothelium as versatile tools for theranostic drug delivery. Theranostics 2018, 8, 1131–1145. [Google Scholar] [CrossRef]
- Zinger, A.; Baudo, G.; Naoi, T.; Giordano, F.; Lenna, S.; Massaro, M.; Ewing, A.; Kim, H.R.; Tasciotti, E.; Yustein, J.T.; et al. Reproducible and Characterized Method for Ponatinib Encapsulation into Biomimetic Lipid Nanoparticles as a Platform for Multi-Tyrosine Kinase-Targeted Therapy. ACS Appl. Bio Mater. 2020, 3, 6737–6745. [Google Scholar] [CrossRef]
- Molinaro, R.; Martinez, J.; Zinger, A.; De Vita, A.; Storci, G.; Arrighetti, N.; De Rosa, E.; Hartman, K.A.; Basu, N.; Taghipour, N.; et al. Leukocyte-mimicking nanovesicles for effective doxorubicin delivery to treat breast cancer and melanoma. Biomater. Sci. 2020, 8, 333–341. [Google Scholar] [CrossRef]
- Molinaro, R.; Evangelopoulos, M.; Hoffman, J.R.; Corbo, C.; Taraballi, F.; Martinez, J.O.; Hartman, K.A.; Cosco, D.; Costa, G.; Romeo, I.; et al. Design and Development of Biomimetic Nanovesicles Using a Microfluidic Approach. Adv. Mater. 2018, 30, e1702749. [Google Scholar] [CrossRef]
- Molinaro, R.; Pasto, A.; Corbo, C.; Taraballi, F.; Giordano, F.; Martinez, J.O.; Zhao, P.; Wang, X.; Zinger, A.; Boada, C.; et al. Macrophage-derived nanovesicles exert intrinsic anti-inflammatory properties and prolong survival in sepsis through a direct interaction with macrophages. Nanoscale 2019, 11, 13576–13586. [Google Scholar] [CrossRef]
- Corbo, C.; Cromer, W.; Molinaro, R.; Furman, N.E.T.; Hartman, K.A.; De Rosa, E.; Boada, C.; Wang, X.; Zawieja, D.C.; Agostini, M.; et al. Engineered biomimetic nanovesicles show intrinsic anti-inflammatory properties for the treatment of inflammatory bowel diseases. Nanoscale 2017, 9, 14581–14591. [Google Scholar] [CrossRef]
- Liu, L.; Bai, X.; Martikainen, M.-V.; Kårlund, A.; Roponen, M.; Xu, W.; Hu, G.; Tasciotti, E.; Lehto, V.-P. Cell membrane coating integrity affects the internalization mechanism of biomimetic nanoparticles. Nat. Commun. 2021, 12, 5726. [Google Scholar] [CrossRef]






| Starting Cells | Advantages | Disadvantages | Technology Used for Biological Material Derivation | Technology Used to Integrate the Membranes on Nanovectors or Induce Vesicles Formation | References |
|---|---|---|---|---|---|
| Red blood cells | -Abundant in the blood and easy to separate. -Ease of membrane isolation 1 -Provide longer circulation. -Provide decoy against RBCs targeting toxins. | -No intrinsic targeting moieties -Keep in mind the blood type used -HEME group residues potentially toxic | -Whole membranes: whole blood centrifugation to remove plasma, platelets, and other cells; hypotonic treatment to lyse isolated RBCs, followed by centrifugation to remove hemoglobin. | -Membrane coating: mixing with synthetic particles followed by sonication and/or extrusion to reduce size and make the particles homogeneous in size. -Vesicle formation: sonication and/or extrusion to reduce size and make the particles homogeneous in size. | Membrane coating: [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,65] Nanovesicles formulation: [75] |
| Platelets | -Relatively easy to separate. -Ease of membrane isolation 1 -Provide longer circulation. -Targeting to inflamed/damaged tissue | -Less abundant in the blood. | -Whole membranes: whole blood low speed centrifugation to remove red and white blood cells; higher speed centrifugation to wash away the plasma; followed by freeze and thaw cycles to separate the platelets membranes. | -Membrane coating: mixing with synthetic particles followed by sonication and/or extrusion to reduce size and make the particles homogeneous in size. | [47,48,49,50,51,52,65] |
| Leukocytes | -Active targeting to inflamed tissue. -Wide variety of different cell populations. -Possible manipulation of membrane proteins. | -Not very abundant in the blood. -Requires removal of all non-membrane cellular contents. -Effect on plasmatic half-life variable. | -Whole membranes: homogenization, followed by sequential centrifugation to remove cell organelles. -Membrane proteins extraction: detergent-based cell permeabilization to remove intracellular components followed by plasma membrane solubilization using stronger, non denaturating detergents | -Membrane coating: mixing with synthetic particles followed by sonication and/or extrusion to reduce size and make the particles homogeneous in size. -Biomimetic nanovesicles formulations (leukosomes): membrane proteins integration through lipids hydration with membrane proteins suspension and subsequent extrusion; or single step integration of membrane proteins into the forming vesicles through microfluidics. | Membrane coating: [53,54,55,56,57,58] Leukosomes formulation: [77,78,79,80,81,82] |
| Cancer Cells | -Possible homologous targeting to tumor cells. -Tumor neo-antigens for nanovaccines. | -Difficult to obtain enough autologous, patient-specific cells. -Requires removal of all non-membrane cellular contents. -Possible safety concerns. | -Whole membranes: homogenization, followed by sequential centrifugation to remove cell organelles. In some cases, membranes are further purified using gradient centrifugation. | -Membrane coating: mixing with synthetic particles followed by sonication and/or extrusion to reduce size and make the particles homogeneous in size. | [59,60,61,62,63] |
| Bacterial cells | -Allow the display of bacterial antigens without risk of active infection -Antagonize bacterial adhesion to tissues | -Highly antigenic material | -Centrifugation of confluent bacterial cultures to separate bacteria from spontaneously released membrane vesicles and subsequent filtration to ensure the absence of living bacteria. | [67] | |
| Other mammalian cells | -Highly dependent on the cell type, but often based on the translation of specific receptor and binding proteins onto nanomaterials | -Requires removal of all non-membrane cellular contents. -Possible difficulties in having large amounts of starting materials | -Whole membranes: homogenization, followed by sequential centrifugation to remove cell organelles. | [68] | |
| Mesenchymal stem cells | -Provide active targeting to solid tumors. | -Requires removal of all non-membrane cellular contents. -Complex isolation or stabilized cell lines. | -Whole membranes: hypotonic lysis, homogenization with a dunce homogenizer, and differential centrifugation to separate membranes from cell debris. | -Vesicles formation (Nanoghosts): sonication and/or extrusion to reduce size and make the particles homogeneous in size. | [72,74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rampado, R.; Caliceti, P.; Agostini, M. Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications. Nanomaterials 2022, 12, 1543. https://doi.org/10.3390/nano12091543
Rampado R, Caliceti P, Agostini M. Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications. Nanomaterials. 2022; 12(9):1543. https://doi.org/10.3390/nano12091543
Chicago/Turabian StyleRampado, Riccardo, Paolo Caliceti, and Marco Agostini. 2022. "Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications" Nanomaterials 12, no. 9: 1543. https://doi.org/10.3390/nano12091543
APA StyleRampado, R., Caliceti, P., & Agostini, M. (2022). Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications. Nanomaterials, 12(9), 1543. https://doi.org/10.3390/nano12091543







