Nanotechnology-Based Bioactive Antifeedant for Plant Protection
Abstract
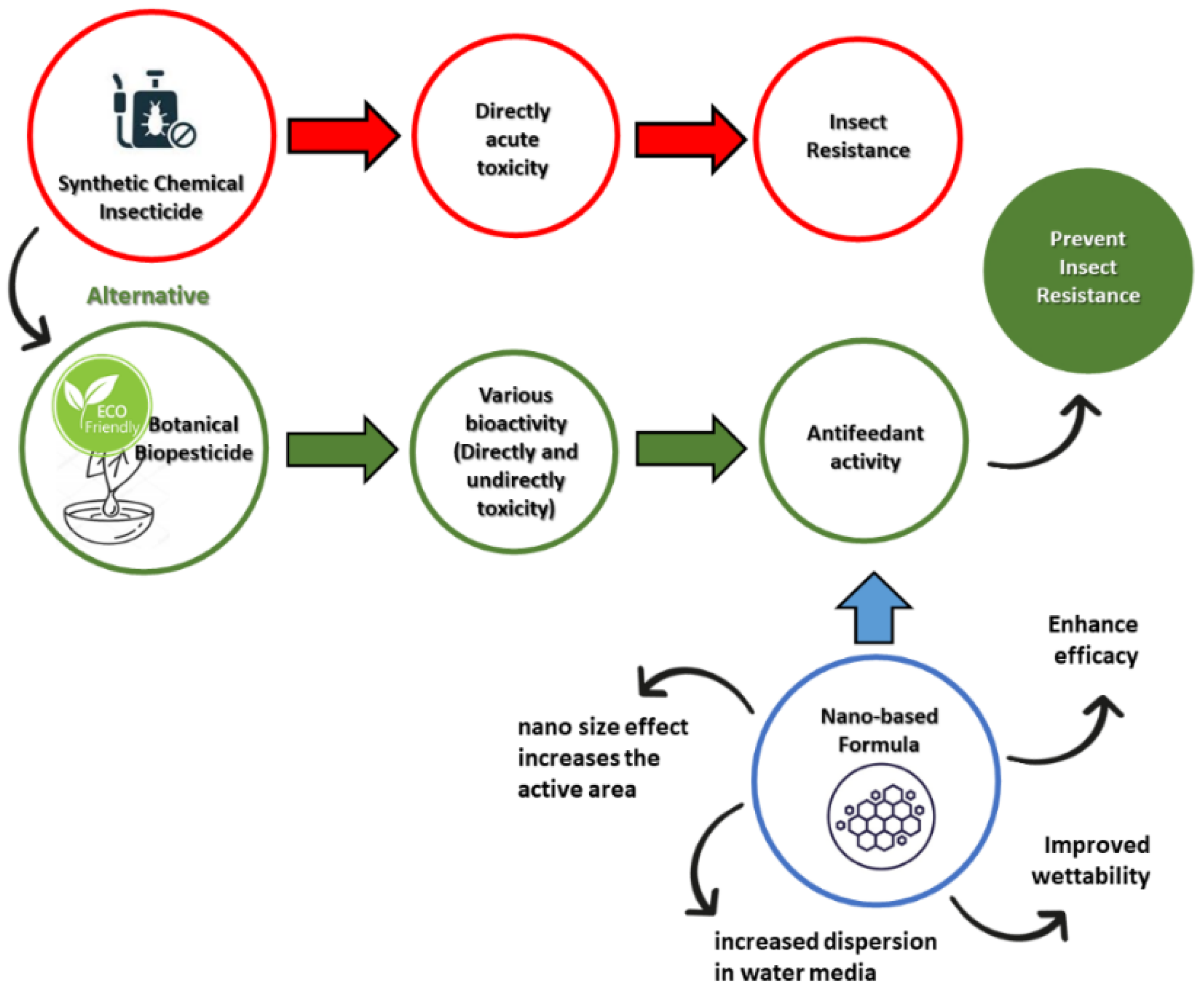
:1. Introduction
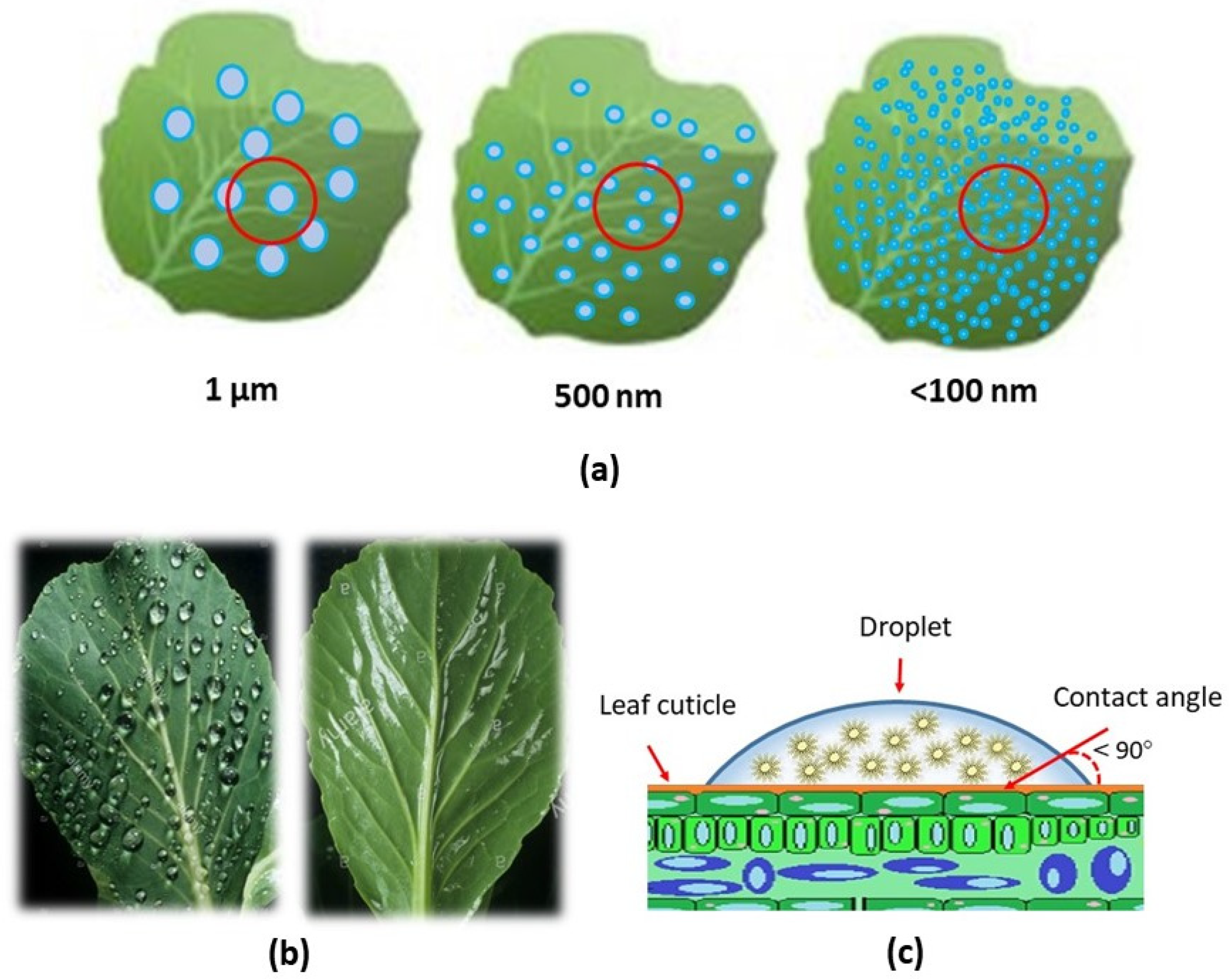
2. The Opportunity of Antifeedant Nano-Based Delivery Systems
2.1. Insect Pest Control Strategies Using Antifeedant Plant-Derived Pesticides: Antifeedant Management, Resources, and Reserve
| No. | Pest Control Strategies | Resources | Reserve/Formulations | Preparation Methods | Bioactive Compounds | Efficacy | Target Insect Pests | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | Stored grain pest manage-ment | Sunflower seed (Helianthus annuus) | Crude oil of sunflower seed hulls (SSH) | Pyrolysis reactor used to produce bio-oils | Acetic acid, furfural, methoxyphenol | Higher antifeedant FDI (79.3%) ECI (87.5%) Moderate antifeedant FDI (45.8%) ECI (16.6%) | Sitophilus oryzae, Lasioderma serricorne and Tribolium castaneum | [53] |
| Mentha piperita | Essential oil | Hydrodistillation | Menthone and menthol | Antifeedant activity S. oryzae FDI (29.68%, 18.81%, and 14.34%) T. castaneum FDI (20.67%, 13.73%, and 9.19%) | Sitophilus oryzae Linn. and T. castaneum | [54] | ||
| Gaultheria procumbens | Essential oil | Hydrodistillation | Methyl salicylate (MS), citral, Z-citral | Antifeedant activity FDI 8.26% (EO) FDI 5.33% (MS) | Sitophilus oryzae and Rhyzopertha Dominica | [55] | ||
| Zanthoxylum bungeanum, Z. schinifolium, Z. armatum and Z. dissitum | Methanolic stem bark extract | Screening active fraction continued by active compound isolation | Benzophenan-thridines | Antifeedant activity FDI (41.12%) EC50 62.67 ppm (norche chelerythrine) EC50 66.97 ppm (decarine) | Tribolium castaneum | [56] | ||
| 2 | Crop protection management | Melia volkensii | Methanolic crude extracts from the bark, leaves, pulp, and nuts | Dry crude extracts dissolved in methanol and topped up with water (96% of total volume) with several concentrations used for in vivo and in vitro assay | 1α,3α-Diacetylvilasinin, 1-cinnamoyl-trichilinin, 1-tigloyltrichilinin, 1-acetyltrichilinin, salannin, 1-detigloyl-1-isobutyl-salannin, 20,30-dihydro-salannin, ohchinin3-acetate, nimbolin B, volkensin, and toosen-danin | Antifeedant activity C. Puncticollis. FDI (51%) (nut and pulp) FDI (43%) (bark) FDI (44%) (leaf) S. exigua. FDI (57%) (nut and pulp) FDI (58%) (bark) FDI (50%) (leaf) | Cylas puncticollis, Spodoptera exigua, S. Frugiperda | [57] |
| Lantana Camara | Ethyl acetate active fraction (EAF) leaves L. camara | Nanosuspension formulation by reverse emulsion with the various components of Tween 80 as a surfactant | Alkaloids, saponins, and steroids | Strong antifeedant activity SOR 11 at the LC50 0.39% (D = 8.3 ± 1.3 nm), leading to wettability improvement on the leaf surface | Crocidolomia pavonana Fab. | [58] | ||
| Annona mucosa Jacq. | Ethanolic seed extract A. mucosa and biopesticide of A. squamosa extraction (Anosom® 1 EC) | Emulsifiable concentrate formulation of ESAM | Rolliniastatin-1 (ESAM), fatty acid-derived substances contain acetoge-nins (Anosom® 1 EC) | Strong antifeedant activity Decreased foliar consumption (>90%) at LC50 = 411.55 and 312.08 mg kg−1 in 168 h (crude extract and ESAM) | Helicoverpa armigera | [59] | ||
| Panax ginseng | Methanolic stems and leaves extract | Isolated pure compounds from the methanolic crude extract determined using the HPLC method | Ginsenoside | Moderate antifeedant activities (AFC50) are 4.98 and 5.03 mg mL−1 at 24 h and 48 h (no-choice assay), and 2.74 and 4.14 mg mL−1 (choice assay) (the pure compounds) | Plutella xylostella | [60] | ||
| Pilgerodendron uviferum | Essential oil (EO), petroleum ether extract (PEE), and dichloromethane extract (DCME) | Fractioned EO and both of the extracts by column chromatography with silica gel as a stationary phase, using a different gradient elution for each extract | Sesquiterpenes (circa 60%): (−)-trans-calamenene, cadalene, (−)-cubenol, (−)-epicubenol, (−)-torreyol, (−)-15-copaenol | Antifeedant activities (EO, DCME, and the sesquiterpenes isolated pure compounds) | Hylastinus obscurus | [61] | ||
| Cyperus eragrosits | Nature and synthetic compounds of dihydro benzofurans and aurones | Organic synthesis and electrochemical oxidation | Acetophenone-type dihydro benzofurans, coumaran (aurone derivatives) | Higher antifeedant activity of natural aurones compared with synthetic compounds | Spodoptera litura and Plutella xylostella | [62] | ||
| Caesalpinia bonduc (L.) Roxb. | Chloroform extract | Screening crude extracts and fractions solvents with increasing polarity | Coumarins, flavonoids, terpenoids, phenols, and quinones | Higher antifeedant of fraction 3 of chloroform EC50 = 357.13 ppm than the crude chloroform extract that shows a minimum EC50 3.07% | Helicoverpa armigera | [63] | ||
| Millettia pachycarpa | Hexane and methanol leaves extracts | Screening active fraction | Flavonoids and isoflavonoids (mille-wanins, prenylated isoflavones, and pyra-nochal-cones) | Strong antifeedant activity AI50 = 227.12 ppm at 24 h (the hexane) Good antifeedant activity AI50 = 340.87 ppm at 24 h (the methanol extract) | Spodoptera litura | [64] | ||
| Azadirachta indica | Crude neem gum from the incised bark of A. indica | Neem gum nano formulation (NGNF) | Hexadecanoic acid, oleic acid, and ricinoleic acid | Strong antifeedant activity FDI (100%) at LC50 10.20 ppm (NGNF) | Helicoverpa armigera and Spodoptera litura | [40] | ||
| Pinus kesiya Royle., Lantana Camara Linn., Litsea cubeba Lour., Gaultheria fragrantissima Wall., Mikania micrantha Kunth., Ambrosia artemisiifolia Linn., and Eupatorium riparium Regel., the indigenous plants of Meghalaya | The methanolic crude extracts of leaves and aerial parts of plants | Extraction by Soxhlet method | Alkaloids, flavonoids phenols, phytosterols, saponins, tannins, and terpenoids | Higher antifeedant activity FDI (50.92%, 70.61%) at 0.1% and 0.5% concentrations extract of G. Fragrantissima than extract of L. cubeba Moderate antifeedant activity Extract of P. kesiya | Helicoverpa armigera | [65] | ||
| Cabralea canjerana canjerana | Fruits and seeds extracts | Fractionation (ethyl acetate and ethanolic fractionation) | Dammarane triterpene, ocotillone 7,15 diol | Higher antifeedant activity of crude extract and ethyl acetate seed active fraction compared to pure compounds | Spodoptera frugiperda | [66] | ||
| Acalypha fruticosa Forssk. | Leaves extract | Dichloromethane extraction | Triterpenoids, steroids, tannins, saponins, flavonoid alkaloids | Significant antifeedant activity of the dichloro-methane extract against L. orbonalis (77.1%), H. armigera (66.2%), S. litura (74.8%), and E. Vittella (67.2%) followed by acetone, dimethyl sulfoxide, and aqueous extracts | Leucinodes orbonalis, Helicoverpa armigera, Spodoptera litura and Earias vittella | [67] | ||
| Hexane, chloroform, and ethyl acetate leaves extracts | Screening active fraction | Terpenoids, tannins, coumarins anthraquinones, and saponins | Antifeedant activity FDI (92.8%) at 5% concentration, LC50 1.86% (the cloroform extract) FDI (84.3%) at 1000 ppm concentration, LC50 385.7 ppm (the active fraction) | Plutella xylostella | [68] | |||
| Soymida febrifuga | Methanolic leaves extract | Isolated pure limonoids compounds determined using H-NMR spectra analysis | Limonoids (phragmalin-type) | Antifeedant activity AI (76.46 µg cm−2, 66.61 µg cm−2) against A. Janata, the pure isolated compounds of fissinolide AI (61.69 µg cm−2, 51.93 µg cm−2) against S. litura (Swietenitin) | Spodoptera litura and Achaea janata | [69] | ||
| Foeniculum vulgare, Anethum graveolens, Petroselinum crispum, Cuminum cyminum | Essential oil | Hydrodistillation | Estragole, fenchon, trans-anetholes, carvone, myristicin, cumin aldehyde | Antifeedant activity FDI (99.7%) (EO P. crispum) FDI (84.7%) (EO A. graveolens fruits) FDI (92.4%) (EO trans-anethole) FDI (84.7%) (EO cumin aldehyde) | Pseudaletia unipuncta | [70] | ||
| Syzygium aromaticum L., Cinnamomum zeylanicum Blume, Lavendula latifolia L., L. angustifolia L., Mentha crispa L., M. arvensis L. and M. piperita L. | Essential oil | Screening EO and pure compounds (single mixture and active compound) | Major constituents: eugenol, (E)-cinna-maldehyde, linalool, n-carvone, menthone, menthol Minor constituents: isoeugenol, β-caryo-phyllene, ceraniol | Higher antifeedant activity (DC50 = 12.5 and 16.4 μg cm−2) C. zeylanicum and S. aromaticum EO are better than pure compounds (eugenol, isoeugenol, and mixture minor compounds) | Trichoplusia ni Hubner | [71] | ||
| 3 | Oil palm plantation pest management | Syzygium aromaticum | Essential oil-based eugenol compounds | Isolated pure eugenol compounds from clove oil extract | 4-Allyl-2-methoxy-1-(4-tri-fluoromethyl-benzyloxy)- benzene | Highest antifeedant activity FDI (64.42%) of the pure compounds | Rhyncho-phorus ferrugineus | [72] |
| Cymbopogon nardus and C. martinii grown in Colombia | Essential oil | Hydrodistillation | Geraniol | The higher antifeedant activity of EO (C. nardus and C. martinii) is better than pure compound (geraniol) and synthetic repellent IR3535 | Euprosterna elaeasa and Acharia fusca | [73] |
2.2. The Role of Nanotechnology in Plant-Derived Pesticide Formulations
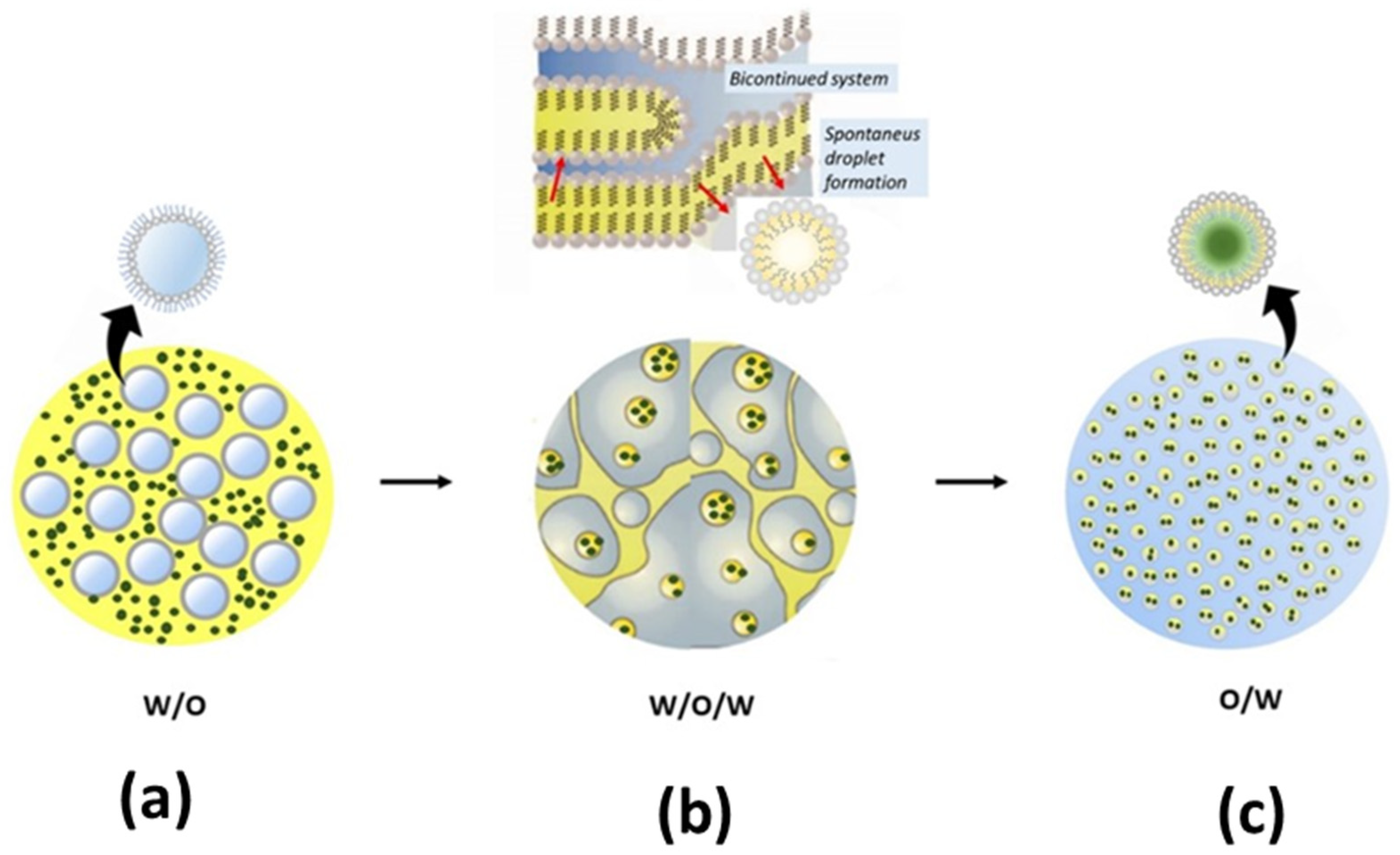
2.3. Nano-Delivery System of Antifeedant Formulation
3. Summary
4. Future Direction
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- FAO. Pesticides Use, Pesticides Trade and Pesticides Indicators. Global, Regional and Country Trends, 1990–2019. 2021. FAOSTAT Analytical Brief Series No. 29. Rome. Available online: https://www.fao.org/3/cb6034en/cb6034en.pdf. (accessed on 23 November 2021).
- Kilani-Morakchi, S.; Morakchi-Goudjil, H.; Sifi, K. Azadirachtin-Based Insecticide: Overview, Risk Assessments, and Future Directions. Front. Agron. 2021, 3, 32. [Google Scholar] [CrossRef]
- Karuppuchamy, P.; Venugopal, S. Integrated Pest Management; Academic Press: London, UK, 2016; pp. 651–684. ISBN 9780128032657. [Google Scholar]
- Lengai, G.M.W.; Muthomi, J.W.; Mbega, E.R. Phytochemical activity and role of botanical pesticides in pest management for sustainable agricultural crop production. Sci. Afr. 2020, 7, e00239. [Google Scholar] [CrossRef]
- Damalas, C.A.; Koutroubas, S.D. Current Status and Recent Developments in Biopesticide Use. Agriculture 2018, 8, 13. [Google Scholar] [CrossRef] [Green Version]
- Rani, L.; Thapa, K.; Kanojia, N.; Sharma, N.; Singh, S.; Grewal, A.S.; Srivastav, A.L.; Kaushal, J. An Extensive Review on the Consequences of Chemical Pesticides on Human Health and Environment. J. Clean. Prod. 2021, 283. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical Insecticides in the Twenty-First Century—Fulfilling Their Promise? Annu. Rev. Èntomol. 2020, 65, 233–249. [Google Scholar] [CrossRef] [Green Version]
- Sharma, A.; Shukla, A.; Attri, K.; Kumar, M.; Kumar, P.; Suttee, A.; Singh, G.; Barnwal, R.P.; Singla, N. Global trends in pesticides: A looming threat and viable alternatives. Ecotoxicol. Environ. Saf. 2020, 201, 110812. [Google Scholar] [CrossRef]
- Zhao, X.; Cui, H.; Wang, Y.; Sun, C.; Cui, B.; Zeng, Z. Development Strategies and Prospects of Nano-based Smart Pesticide Formulation. J. Agric. Food Chem. 2018, 66, 6504–6512. [Google Scholar] [CrossRef]
- Kala, S.; Sogan, N.; Agarwal, A.; Naik, S.; Patanjali, P.; Kumar, J. Biopesticides formulations and delivery techniques. In Natural Remedies for Pest, Disease and Weed Control, 1st ed.; Egbuna, C., Sawicka, B., Eds.; Elsevier; Academic Press: London, UK, 2020; ISBN 9780128193044. [Google Scholar] [CrossRef]
- Nuruzzaman, M.; Rahman, M.M.; Liu, Y.; Naidu, R. Nanoencapsulation, Nano-guard for Pesticides: A New Window for Safe Application. J. Agric. Food Chem. 2016, 64, 1447–1483. [Google Scholar] [CrossRef]
- De Oliveira, J.L.; Campos, E.V.R.; Fraceto, L.F. Recent Developments and Challenges for Nanoscale Formulation of Botanical Pesticides for Use in Sustainable Agriculture. J. Agric. Food Chem. 2018, 66, 8898–8913. [Google Scholar] [CrossRef]
- Mittal, D.; Kaur, G.; Singh, P.; Yadav, K.; Ali, S.A. Nanoparticle-Based Sustainable Agriculture and Food Science: Recent Advances and Future Outlook. Front. Nanotechnol. 2020, 2, 1–38. [Google Scholar] [CrossRef]
- Gasic, S.; Tanovic, B. Biopesticide formulations, possibility of application and future trends. Pestic. Fitomedicina 2013, 28, 97–102. [Google Scholar] [CrossRef]
- Chin, C.-P.; Lan, C.-W.; Wu, H.-S. Application of biodiesel as carrier for insecticide emulsifiable concentrate formulation. J. Taiwan Inst. Chem. Eng. 2012, 43, 578–584. [Google Scholar] [CrossRef]
- Patzke, H.; Schieber, A. Growth-inhibitory activity of phenolic compounds applied in an emulsifiable concentrate-ferulic acid as a natural pesticide against Botrytis cinerea. Food Res. Int. 2018, 113, 18–23. [Google Scholar] [CrossRef]
- Knowles, A. Recent developments of safer formulations of agrochemicals. Environmentalist 2007, 28, 35–44. [Google Scholar] [CrossRef]
- Pirzada, T.; de Farias, B.V.; Mathew, R.; Guenther, R.H.; Byrd, M.V.; Tim, L.; Sit, T.L.; Pal, L.; Opperman, C.H.; Khan, S.A. Recent advances in biodegradable matrices for active ingredient release in crop protection: Towards attaining sustainability in agriculture. J. Colloid Interface Sci. 2020, 48, 121–136. [Google Scholar] [CrossRef]
- Waghmare, J.T.; Ware, A.M.; Momin, S.A. Neem oil as pesticide. J. Dispers. Sci. Technol. 2007, 28, 323–328. [Google Scholar] [CrossRef]
- Puripattanavong, J.; Songkram, C.; Lomlim, L.; Amnuaikit, T. Development of concentrated emulsion containing nicotiana tabacum extract for use as a pesticide. J. Appl. Pharm. Sci. 2013, 3, 16–21. [Google Scholar] [CrossRef]
- Shao, H.; Xi, N.; Zhang, Y. Microemulsion formulation of a new biopesticide to control the diamondback moth (Lepidoptera: Plutellidae). Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Pavela, R.; Benelli, G.; Pavoni, L.; Bonacucina, G.; Cespi, M.; Cianfaglione, K.; Bajalan, I.; Morshedloo, M.R.; Lupidi, G.; Romano, D.; et al. Microemulsions for delivery of Apiaceae essential oils—Towards highly effective and eco-friendly mosquito larvicides? Ind. Crops Prod. 2019, 129, 631–640. [Google Scholar] [CrossRef]
- Pavoni, L.; Benelli, G.; Maggi, F.; Bonacucina, G. Green Nanoemulsion Interventions for Biopesticide Formulations; Elsevier BV: Amsterdam, The Netherlands, 2019; ISBN 9780128158296. [Google Scholar]
- Palermo, D.; Giunti, G.; Laudani, F.; Palmeri, V.; Campolo, O. Essential Oil-Based Nano-Biopesticides: Formulation and Bioactivity against the Confused Flour Beetle Tribolium confusum. Sustainability 2021, 13, 9746. [Google Scholar] [CrossRef]
- Isman, M. Insect antifeedants. Pestic. Outlook 2002, 13, 152–157. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miresmailli, S.; Isman, M.B. Botanical insecticides inspired by plant–herbivore chemical interactions. Trends Plant Sci. 2014, 19, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Koul, O. Insect Antifeedants; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Koul, O. Antifeedant Phytochemicals in Insect Management (so Close yet so Far). In Ecofriendly Pest Management for Food Security; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 525–544. ISBN 9780128032664. [Google Scholar]
- Usman, M.; Farooq, M.; Wakeel, A.; Nawaz, A.; Alam Cheema, S.A.; Rehman, H.U.; Ashraf, I.; Sanaullah, M. Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci. Total. Environ. 2020, 721, 137778. [Google Scholar] [CrossRef]
- Chaud, M.; Souto, E.B.; Zielinska, A.; Severino, P.; Batain, F.; Oliveira, J., Jr.; Alves, T. Nanopesticides in agriculture: Benefits and challenge in agricultural productivity, toxicological risks to human health and environment. Toxics 2021, 9, 131. [Google Scholar] [CrossRef]
- Mustafa, I.F.; Hussein, M.Z. Synthesis and technology of nanoemulsion-based pesticide formulation. Nanomaterials 2020, 10, 1608. [Google Scholar] [CrossRef]
- Oliveira, J.L.; Campos, E.; Bakshi, M.; Abhilash, P.; Fraceto, L. Application of nanotechnology for the encapsulation of botanical insecticides for sustainable agriculture: Prospects and promises. Biotechnol. Adv. 2014, 32, 1550–1561. [Google Scholar] [CrossRef]
- Shivanandappa, T.; Rajashekar, Y. Mode of action of plant-derived natural insecticides. In Advance in Plant Biopesticides; Springer: New Delhi, India, 2014; pp. 323–345. [Google Scholar] [CrossRef]
- Kumar, S.; Nehra, M.; Dilbaghi, N.; Marrazza, G.; Hassan, A.A.; Kim, K.-H. Nano-based smart pesticide formulations: Emerging opportunities for agriculture. J. Control. Release 2019, 294, 131–153. [Google Scholar] [CrossRef]
- Lade, B.D.; Google, D.P.; Lade, D.B.; Moon, G.M.; Nandeshwar, S.B.; Kumbhare, S.D. Nanobiopesticide Formulations: Application Strategies Today and Future Perspectives; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 179–206. ISBN 9780128158296. [Google Scholar]
- Athanassiou, C.G.; Kavallieratos, N.G.; Benelli, G.; Losic, D.; Rani, P.U.; Desneux, N. Nanoparticles for pest control: Current status and future perspectives. J. Pest Sci. 2018, 91, 1–15. [Google Scholar] [CrossRef]
- Saini, R.K.; Patel, S.; Bajpai, J.; Bajpai, A.K. Advanced controlled nanopesticide delivery systems for managing insect pests. In Controlled Release of Pesticides for Sustainable Agriculture; Rakhimol, K.R., Thomas, S., Volova, T., Jayachandran, K., Eds.; Springer: Cham, Switzerland, 2020; pp. 155–184. [Google Scholar] [CrossRef]
- Forim, M.R.; Costa, E.S.; Silva, M.F.D.G.F.D.; Fernandes, J.B.; Mondego, J.M.; Junior, A.L.B. Development of a New Method To Prepare Nano-/microparticles Loaded with Extracts of Azadirachta indica, Their Characterization and Use in Controlling Plutella xylostella. J. Agric. Food Chem. 2013, 61, 9131–9139. [Google Scholar] [CrossRef]
- Kamaraj, C.; Gandhi, P.R.; Elango, G.; Karthi, S.; Chung, I.-M.; Rajakumar, G. Novel and environmental friendly approach; Impact of Neem (Azadirachta indica) gum nano formulation (NGNF) on Helicoverpa armigera (Hub.) and Spodoptera litura (Fab.). Int. J. Biol. Macromol. 2018, 107, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Thomine, E.; Mumford, J.; Rusch, A.; Desneux, N. Using crop diversity to lower pesticide use: Socio-ecological approaches. Sci. Total Environ. 2021, 804, 150156. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.P.; Green, T.A.; Loker, A.J. Biological control and integrated pest management in organic and conventional systems. Biol. Control 2020, 140, 104095. [Google Scholar] [CrossRef]
- Rezaei, R.; Safa, L.; Ganjkhanloo, M.M. Understanding farmers’ ecological conservation behavior regarding the use of integrated pest management—An application of the technology acceptance model. Glob. Ecol. Conserv. 2020, 22, e00941. [Google Scholar] [CrossRef]
- Jiang, X.; Hansen, H.C.B.; Strobel, B.W.; Cedergreen, N. What is the aquatic toxicity of saponin-rich plant extracts used as biopesticides? Environ. Pollut. 2018, 236, 416–424. [Google Scholar] [CrossRef]
- Koul, O. Phytochemicals and Insect Control: An Antifeedant Approach. Crit. Rev. Plant Sci. 2008, 27, 1–24. [Google Scholar] [CrossRef]
- Purrington, C. Antifeedant Substances in Plants. Encycl. Appl. Plant Sci. 2017, 2, 364–367. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Smart, L.E.; Birch, A.N.E.; Blok, V.C.; MacKenzie, K.; Guerrieri, E.; Cascone, P.; Luna, E.; Ton, J. Prospects for plant defence activators and biocontrol in IPM: Concepts and lessons learnt so far. J. Crop Prot. 2016, 97, 128–134. [Google Scholar] [CrossRef] [Green Version]
- Sandjo, L.P.; Kuete, V. Triterpenes and Steroids from the Medicinal Plants of Africa; Elsevier BV: Amsterdam, The Netherlands, 2013; ISBN 9780124059276. [Google Scholar]
- Suckling, D.M.; Conlong, D.E.; Carpenter, J.E.; Bloem, K.A.; Rendon, P.; Vreysen, M.J.B. Global range expansion of pest Lepidoptera requires socially acceptable solutions. Biol. Invasions 2017, 19, 1107–1119. [Google Scholar] [CrossRef] [Green Version]
- Kłyś, M.; Malejky, N.; Nowak-Chmura, M. The repellent effect of plants and their active substances against the beetle storage pests. J. Stored Prod. Res. 2017, 74, 66–77. [Google Scholar] [CrossRef]
- Lucia, A.; Guzmán, E. Emulsions containing essential oils, their components or volatile semiochemicals as promising tools for insect pest and pathogen management. Adv. Colloid Interface Sci. 2021, 287, 102330. [Google Scholar] [CrossRef]
- Stanley, V.; Hickerson, K.; Daley, M. Supermarket Supply Chains in Horticulture in India: The Novel Marketing Models, Effects and Policies. Agrotechnology 2012, S11, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Urrutia, R.I.; Yeguerman, C.; Jesser, E.; Gutierrez, V.S.; Volpe, M.A.; González, J.O.W. Sunflower seed hulls waste as a novel source of insecticidal product: Pyrolysis bio-oil bioactivity on insect pests of stored grains and products. J. Clean. Prod. 2021, 287, 125000. [Google Scholar] [CrossRef]
- Rajkumar, V.; Gunasekaran, C.; Christy, I.K.; Dharmaraj, J.; Chinnaraj, P.; Paul, C.A. Toxicity, antifeedant and biochemical efficacy of Mentha piperita L. essential oil and their major constituents against stored grain pest. Pestic. Biochem. Physiol. 2019, 156, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Kiran, S.; Prakash, B. Assessment of Toxicity, Antifeedant Activity, and Biochemical Responses in Stored-Grain Insects Exposed to Lethal and Sublethal Doses of Gaultheria procumbens L. Essential Oil. J. Agric. Food Chem. 2015, 63, 10518–10524. [Google Scholar] [CrossRef]
- Wang, C.-F.; You, C.-X.; Yang, K.; Guo, S.-S.; Geng, Z.-F.; Fan, L.; Du, S.-S.; Deng, Z.-W.; Wang, Y.-Y. Antifeedant activities of methanol extracts of four Zanthoxylum species and benzophenanthridines from stem bark of Zanthoxylum schinifolium against Tribolium castaneum. Ind. Crops Prod. 2015, 74, 407–411. [Google Scholar] [CrossRef]
- Jaoko, V.; Taning, C.N.T.; Backx, S.; Motti, P.; Mulatya, J.; Vandenabeele, J.; Magomere, T.; Olubayo, F.; Mangelinckx, S.; Werbrouck, S.P.O.; et al. Laboratory and Greenhouse Evaluation of Melia volkensii Extracts for Potency against African Sweet Potato Weevil, Cylas puncticollis, and Fall Armyworm, Spodoptera frugiperda. Agronomy 2021, 11, 1994. [Google Scholar] [CrossRef]
- Melanie, M.; Kosasih, F.Y.; Kasmara, H.; Malini, D.M.; Panatarani, C.; Joni, I.M.; Husodo, T.; Hermawan, W. Antifeedant activity of Lantana camara nano suspension prepared by reverse emulsion of ethyl acetate active fraction at various surfactant organic-phase ratio. Biocatal. Agric. Biotechnol. 2020, 29, 101805. [Google Scholar] [CrossRef]
- de Souza, C.M.; Baldin, E.L.L.; Ribeiro, L.D.P.; dos Santos, T.L.B.; da Silva, I.F.; Morando, R.; Vendramim, J.D. Antifeedant and growth inhibitory effects of Annonaceae derivatives on Helicoverpa armigera (Hübner). Crop Prot. 2019, 121, 45–50. [Google Scholar] [CrossRef]
- Yang, H.; Piao, X.; Zhang, L.; Song, S.; Xu, Y. Ginsenosides from the stems and leaves of Panax ginseng show antifeedant activity against Plutella xylostella (Linnaeus). Ind. Crops Prod. 2018, 124, 412–417. [Google Scholar] [CrossRef]
- Espinoza, J.; Urzua, A.; Bardehle, L.; Quiroz, A.; Echeverría, J.; González-Teuber, M. Antifeedant Effects of Essential Oil, Extracts, and Isolated Sesquiterpenes from Pilgerodendron uviferum (D. Don) Florin Heartwood on Red Clover Borer Hylastinus obscurus (Coleoptera: Curculionidae). Molecules 2018, 23, 1282. [Google Scholar] [CrossRef] [Green Version]
- Morimoto, M. Insect Antifeedant Activities and Preparation of Dihydrobenzofurans from Cyperus spp. ACS Symp. Ser. 2018, 1294, 11–21. [Google Scholar] [CrossRef]
- Baskar, K.; Maheswaran, R.; Pavunraj, M.; Packiam, S.M.; Ignacimuthu, S.; Duraipandiyan, V.; Benelli, G. Toxicity and antifeedant activity of Caesalpinia bonduc (L.) Roxb. (Caesalpiniaceae) extracts and fractions against the cotton bollworm Helicoverpa armigera Hub. (Lepidoptera: Noctuidae). Physiol. Mol. Plant Pathol. 2018, 101, 69–74. [Google Scholar] [CrossRef]
- Ningombam, A.; Ahluwalia, V.; Srivastava, C.; Walia, S. Antifeedant activity and phytochemical investigation of Millettia pachycarpa extracts against Tobacco Leaf Eating Caterpillar, Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae). J. Asia-Pac. Èntomol. 2017, 20, 381–385. [Google Scholar] [CrossRef]
- Paul, D.; Choudhury, M. Larvicidal and antifeedant activity of some indigenous plants of Meghalaya against 4th instar Helicoverpa armigera (Hübner) larvae. J. Crop Prot. 2016, 5, 447–460. [Google Scholar] [CrossRef]
- Magrini, F.E.; Specht, A.; Gaio, J.; Girelli, C.P.; Migues, I.; Heinzen, H.; Saldaña, J.; Sartori, V.C.; Cesio, V. Antifeedant activity and effects of fruits and seeds extracts of Cabralea canjerana canjerana (Vell.) Mart. (Meliaceae) on the immature stages of the fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae). Ind. Crops Prod. 2015, 65, 150–158. [Google Scholar] [CrossRef]
- Manickam, P.; Kathirvelu, B.; Sundaram, J.; Munusamy, A. Bio-efficacy of crude leaf extracts of Acalypha fruticosa Forssk. against some agriculturally important insect pests. Asian Pac. J. Trop. Dis. 2014, 4, S890–S894. [Google Scholar] [CrossRef]
- Lingathurai, S.; Vendan, S.E.; Paulraj, M.G.; Ignacimuthu, S. Antifeedant and larvicidal activities of Acalypha fruticosa Forssk. (Euphorbiaceae) against Plutella xylostella L. (Lepidoptera: Yponomeutidae) larvae. J. King Saud Univ. Sci. 2011, 23, 11–16. [Google Scholar] [CrossRef] [Green Version]
- Yadav, P.A.; Suresh, G.; Rao, M.S.A.; Shankaraiah, G.; Rani, P.U.; Babu, K.S. Limonoids from the leaves of Soymida febrifuga and their insect antifeedant activities. Bioorg. Med. Chem. Lett. 2014, 24, 888–892. [Google Scholar] [CrossRef]
- Sousa, R.M.O.F.; Rosa, J.S.; Oliveira, L.; Cunha, A.; Fernandes-Ferreira, M. Activities of Apiaceae Essential Oils against Armyworm, Pseudaletia unipuncta (Lepidoptera: Noctuidae). J. Agric. Food Chem. 2013, 61, 7661–7672. [Google Scholar] [CrossRef]
- Akhtar, Y.; Pages, E.; Stevens, A.; Bradbury, R.; da Camara, C.A.G.; Isman, M.B. Effect of chemical complexity of essential oils on feeding deterrence in larvae of the cabbage looper. Physiol. Èntomol. 2012, 37, 81–91. [Google Scholar] [CrossRef]
- Yan, T.K.; Asari, A.; Abdullah, S.; Ismail, M.; Azmi, W.A. The dataset for antifeedant activity of eugenol derived compounds against red palm weevil (Rhynchophorus ferrugineus, Olivier) larvae. Data Brief 2019, 25, 104227. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Lambraño, R.; Caballero-Gallardo, K.; Olivero-Verbel, J. Toxicity and antifeedant activity of essential oils from three aromatic plants grown in Colombia against Euprosterna elaeasa and Acharia fusca (Lepidoptera: Limacodidae). Asian Pac. J. Trop. Biomed. 2014, 4, 695–700. [Google Scholar] [CrossRef] [Green Version]
- Patra, J.K.; Das, G.; Lee, S.; Kang, S.-S.; Shin, H.-S. Selected commercial plants: A review of extraction and isolation of bioactive compounds and their pharmacological market value. Trends Food Sci. Technol. 2018, 82, 89–109. [Google Scholar] [CrossRef]
- Nawrot, J.; Harmatha, J. Phytochemical feeding deterrents for stored product insect pests. Phytochem. Rev. 2012, 11, 543–566. [Google Scholar] [CrossRef]
- Khandelwal, N.; Barbole, R.S.; Banerjee, S.; Chate, G.P.; Biradar, A.V.; Khandare, J.J.; Giri, A.P. Budding trends in integrated pest management using advanced micro- and nano-materials: Challenges and perspectives. J. Environ. Manag. 2016, 184, 157–169. [Google Scholar] [CrossRef]
- Kapinder; Dangi, K.; Verma, A.K. Efficient & eco-friendly smart nano-pesticides: Emerging prospects for agriculture. Mater. Today Proc. 2021, 45, 3819–3824. [Google Scholar] [CrossRef]
- Suresh, U.; Murugan, K.; Panneerselvam, C.; Aziz, A.T.; Cianfaglione, K.; Wang, L.; Maggi, F. Encapsulation of sea fennel (Crithmum maritimum) essential oil in nanoemulsion and SiO2 nanoparticles for treatment of the crop pest Spodoptera litura and the dengue vector Aedes aegypti. Ind. Crops Prod. 2020, 158, 113033. [Google Scholar] [CrossRef]
- Mohafrash, S.M.; Fallatah, S.A.; Farag, S.M.; Mossa, A.-T. Mentha spicata essential oil nanoformulation and its larvicidal application against Culex pipiens and Musca domestica. Ind. Crops Prod. 2020, 157, 112944. [Google Scholar] [CrossRef]
- Campolo, O.; Giunti, G.; Laigle, M.; Michel, T.; Palmeri, V. Essential oil-based nano-emulsions: Effect of different surfactants, sonication and plant species on physicochemical characteristics. Ind. Crops Prod. 2020, 157, 112935. [Google Scholar] [CrossRef]
- Lopes, A.I.F.; Monteiro, M.; Araújo, A.R.L.; Rodrigues, A.R.O.; Castanheira, E.M.S.; Pereira, D.M.; Olim, P.; Gil Fortes, A.; Gonçalves, M.S.T. Cytotoxic Plant Extracts towards Insect Cells: Bioactivity and Nanoencapsulation Studies for Application as Biopesticides. Molecules 2020, 25, 5855. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, V.; Gunasekaran, C.; Dharmaraj, J.; Chinnaraj, P.; Paul, C.A.; Kanithachristy, I. Structural characterization of chitosan nanoparticle loaded with Piper nigrum essential oil for biological efficacy against the stored grain pest control. Pestic. Biochem. Physiol. 2020, 166, 104566. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, Z.; Saber, M.; Bagheri, M.; Mahdavinia, G.R. Achillea millefolium essential oil and chitosan nanocapsules with enhanced activity against Tetranychus urticae. J. Pest Sci. 2018, 91, 837–848. [Google Scholar] [CrossRef]
- Yang, L.; Kaziem, A.E.; Lin, Y.; Li, C.; Tan, Y.; Huang, S.; Cheng, D.; Xu, H.; Zhang, Z. Carboxylated β-cyclodextrin anchored hollow mesoporous silica enhances insecticidal activity and reduces the toxicity of indoxacarb. Carbohydr. Polym. 2021, 266, 118150. [Google Scholar] [CrossRef]
- Pereira, K.D.C.; Quintela, E.D.; Da Silva, D.J.; Nascimento, V.A.D.; Da Rocha, D.V.M.; Silva, J.F.A.E.; Forim, M.R.; Silva, F.G.; Cazal, C.D.M. Characterization of Nanospheres Containing Zanthoxylum riedelianum Fruit Essential Oil and Their Insecticidal and Deterrent Activities against Bemisia tabaci (Hemiptera: Aleyrodidae). Molecules 2018, 23, 2052. [Google Scholar] [CrossRef] [Green Version]
- Rajkumar, V.; Gunasekaran, C.; Paul, C.A.; Dharmaraj, J. Development of encapsulated peppermint essential oil in chitosan nanoparticles: Characterization and biological efficacy against stored-grain pest control. Pestic. Biochem. Physiol. 2020, 170, 104679. [Google Scholar] [CrossRef]
- Giunti, G.; Palermo, D.; Laudani, F.; Algeri, G.M.; Campolo, O.; Palmeri, V. Repellence and acute toxicity of a nano-emulsion of sweet orange essential oil toward two major stored grain insect pests. Ind. Crops Prod. 2019, 142, 111869. [Google Scholar] [CrossRef]
- Oliveira, J.L.; Campos, E.V.R.; Pereira, A.; Nunes, L.E.S.; Da Silva, C.C.L.; Pasquoto, T.; Lima, R.; Smaniotto, G.; Polanczyk, R.A.; Fraceto, L.F. Geraniol Encapsulated in Chitosan/Gum Arabic Nanoparticles: A Promising System for Pest Management in Sustainable Agriculture. J. Agric. Food Chem. 2018, 66, 5325–5334. [Google Scholar] [CrossRef]
- Chen, H.; Chen, L.; Shen, Z.; Zhou, H.; Hao, L.; Xu, H.; Zhou, X. Synthesis of mesoporous silica post-loaded by methyl eugenol as an environment-friendly slow-release bio pesticide. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Attia, R.G.; Rizk, S.A.; Hussein, M.A.; Fattah, H.M.A.; Khalil, M.; Ma’Moun, S.A. Effect of cinnamon oil encapsulated with silica nanoparticles on some biological and biochemical aspects of the rice moth, Corcyra cephalonica (Staint.) (Lepidoptera: Pyralidae). Ann. Agric. Sci. 2020, 65, 1–5. [Google Scholar] [CrossRef]
- Paulraj, M.G.; Ignacimuthu, S.; Gandhi, M.R.; Shajahan, A.; Ganesan, P.; Packiam, S.M.; Al-Dhabi, N.A. Comparative studies of tripolyphosphate and glutaraldehyde cross-linked chitosan-botanical pesticide nanoparticles and their agricultural applications. Int. J. Biol. Macromol. 2017, 104, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhu, Y.; Zhang, C.; Lei, J.; Ma, Y.; Du, F. Positive charge pesticide nanoemulsions prepared by the phase inversion composition method with ionic liquids. RSC Adv. 2017, 7, 48586–48596. [Google Scholar] [CrossRef]
- Nuruzzaman, M.; Liu, Y.; Rahman, M.M.; Dharmarajan, R.; Duan, L.; Uddin, A.F.M.J.; Naidu, R. Nanobiopesticides: Composition and Preparation Methods; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128158296. [Google Scholar]
- de Oliveira, C.; Mulinari, J.; Reichert, F.; Júnior, A. Nano-Delivery Systems of Pesticides Active Agents for Agriculture Applications—An Overview. In Ciência, Tecnologia e Inovação: Do Campo à Mesa, Proceeding of International Agribusiness Congress, Recife, Brazil, 25–27 September 2020; Editora IIDV: Recife, Brasil, 2020. [Google Scholar] [CrossRef]
- Kashyap, P.L.; Xiang, X.; Heiden, P. Chitosan nanoparticle based delivery systems for sustainable agriculture. Int. J. Biol. Macromol. 2015, 77, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Pretty, J.; Bharucha, Z.P. Integrated pest management for sustainable intensification of agriculture in Asia and Africa. Insects 2015, 6, 152–182. [Google Scholar] [CrossRef] [PubMed]
- Koul, O.; Cuperus, G.W.; Elliott, N. Areawide Pest Management: Theory and Implementation; CABI: Wallingford, UK, 2008; ISBN 1845933729. [Google Scholar]
- Kamaraj, C.; Rajakumar, G.; Rahuman, A.A.; Velayutham, K.; Bagavan, A.; Zahir, A.A.; Elango, G. Feeding deterrent activity of synthesized silver nanoparticles using Manilkara zapota leaf extract against the house fly, Musca domestica (Diptera: Muscidae). Parasitol. Res. 2011, 111, 2439–2448. [Google Scholar] [CrossRef]
- Siva, C.; Kumar, M.S.; Nagar, G.; Nadu, T.; Nagar, G.; Nadu, T. Pesticidal activity of eco-friendly synthesized silver nano-particles using Aristolochia indica extract against Helicoverpa armigera Hubner (Lepidoptera: Noctuidae). Int. J. Adv. Sci. Tech. Res. 2015, 2, 197–226. [Google Scholar]
- Manimegalai, T.; Raguvaran, K.; Kalpana, M.; Maheswaran, R. Green synthesis of silver nanoparticle using Leonotis nepetifolia and their toxicity against vector mosquitoes of Aedes aegypti and Culex quinquefasciatus and agricultural pests of Spodoptera litura and Helicoverpa armigera. Environ. Sci. Pollut. Res. 2020, 27, 43103–43116. [Google Scholar] [CrossRef]
- González, J.O.W.; Gutiérrez, M.M.; Ferrero, A.A.; Band, B.F. Essential oils nanoformulations for stored-product pest control—Characterization and biological properties. Chemosphere 2014, 100, 130–138. [Google Scholar] [CrossRef]
- Giongo, A.M.M.; Vendramim, J.D.; Forim, M.R. Evaluation of neem-based nanoformulations as alternative to control fall armyworm. Ciência Agrotecnologia 2016, 40, 26–36. [Google Scholar] [CrossRef] [Green Version]
- Yeguerman, C.; Jesser, E.; Massiris, M.; Delrieux, C.; Murray, A.; González, J.W. Insecticidal application of essential oils loaded polymeric nanoparticles to control German cockroach: Design, characterization and lethal/sublethal effects. Ecotoxicol. Environ. Saf. 2020, 189, 110047. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Wang, R.; Qin, D.; Yang, L.; Lin, S.; Cheng, D.; Huang, S.; Zhang, Z. Insecticidal efficacy and mechanism of nanoparticles synthesized from chitosan and carboxymethyl chitosan against Solenopsis invicta (Hymenoptera: Formicidae). Carbohydr. Polym. 2021, 260, 117839. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, K.B.; Sakthivel, N. Biological synthesis of metal nanoparticles by microbes. Adv. Colloid Interface Sci. 2010, 156, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Villalobos, M.J.; Guirao, P.; Díaz-Baños, F.G.; Cantó-Tejero, M.; Villora, G. Oil in water nanoemulsion formulations of botanical active substances. In Nano-Biopesticides Today and Future Perspectives; Elsevier: Amsterdam, The Netherlands, 2019; pp. 223–247. ISBN 9780128158296. [Google Scholar]
- McClements, D.J.; Rao, J. Food-Grade Nanoemulsions: Formulation, Fabrication, Properties, Performance, Biological Fate, and Potential Toxicity. Crit. Rev. Food Sci. Nutr. 2011, 51, 285–330. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, F.; Weiss, J.; McClements, D.J. Low-energy formation of edible nanoemulsions: Factors influencing droplet size produced by emulsion phase inversion. J. Colloid Interface Sci. 2012, 388, 95–102. [Google Scholar] [CrossRef]
- Solans, C.; Solé, I. Nano-emulsions: Formation by low-energy methods. Curr. Opin. Colloid Interface Sci. 2012, 17, 246–254. [Google Scholar] [CrossRef]
- An, C.; Sun, C.; Li, N.; Huang, B.; Jiang, J.; Shen, Y.; Wang, C.; Zhao, X.; Cui, B.; Wang, C.; et al. Nanomaterials and nanotechnology for the delivery of agrochemicals: Strategies towards sustainable agriculture. J. Nanobiotech. 2022, 20, 11. [Google Scholar] [CrossRef]
- Abdollahdokht, D.; Gao, Y.; Faramarz, S.; Poustforoosh, A.; Abbasi, M.; Asadikaram, G.; Nematollahi, M.H. Conventional agrochemicals towards nano-biopesticides: An overview on recent advances. Chem. Biol. Technol. Agric. 2022, 9, 13. [Google Scholar] [CrossRef]
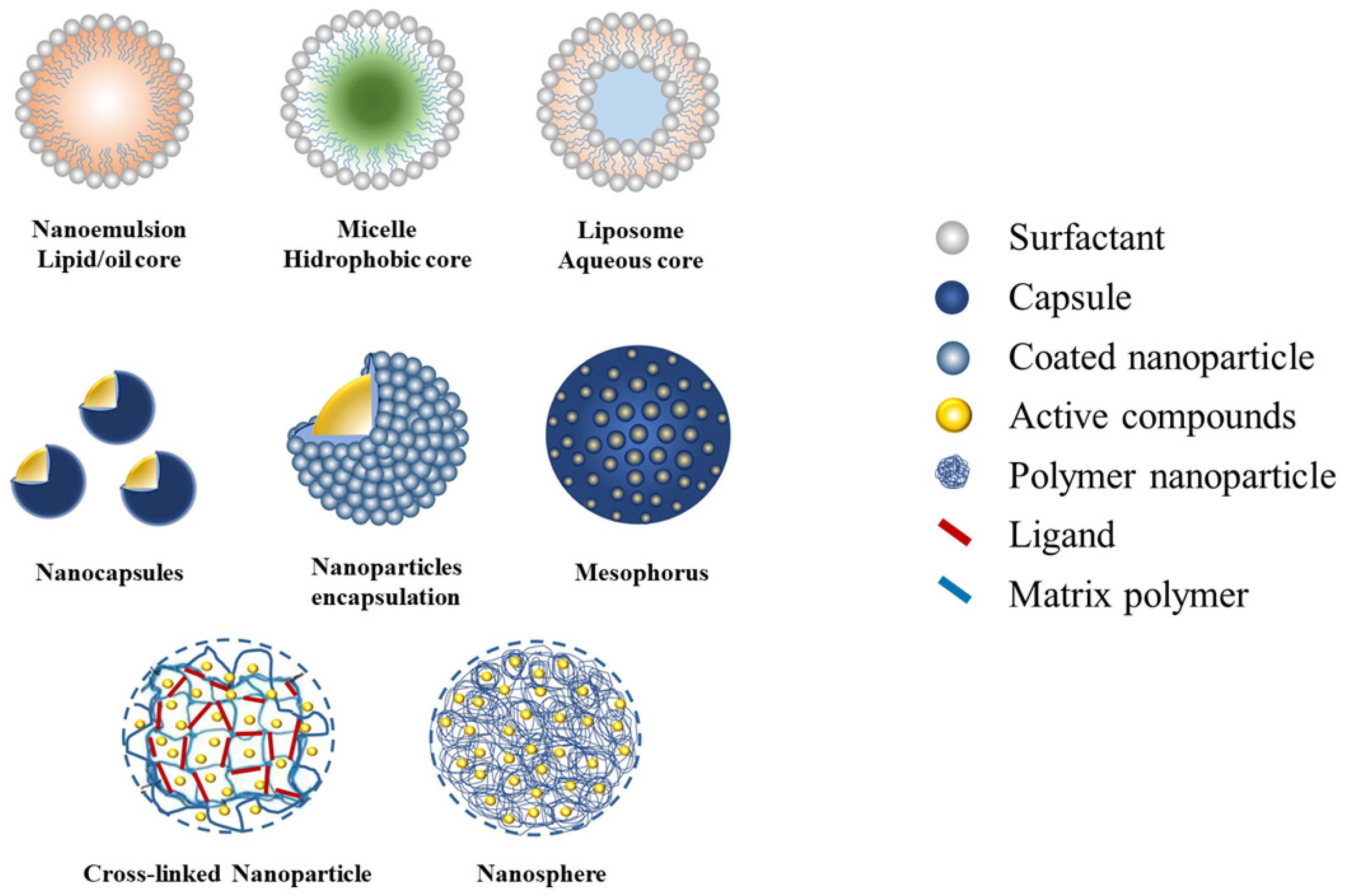
| No. | Carrier System | Preparation and Matrix/Polymers, Surfactant | Properties | Active Compounds | Efficacy/Bioactivities | Ref. |
|---|---|---|---|---|---|---|
| 1 | Nanoemulsion | Oil-in-water by low energy emulsification produce nanoemulsion loaded by EO sea fennel in core (EO:Tween 20 = 1:3) | D = 50–70 nm ZP = −18.3 mV | Seed sea fennel essential oil (EO) (dillapiole and γ-terpinene) | Nanoemulsion enhances the toxicity, inhibits longevity and fecundity of Aedes aegypti and Spodoptera litura. Nanoemulsion is more effective compared to crude EO but less effective compared to pure EO | [78] |
| 2 | Nanoemulsion | Oil-in-water nanoemulsion loaded by EO Mentha spicata in core (EO:Tween 80 = 1:1) | D = 97.8 nm EE = 52.0% | Mentha spicata EO with major compounds 8-cineole (19.55%) and menthol acetate (14%) | The higher larvicidal toxicity against Culex pipiens and Musca domestica larvae compared with the normal mint oil and lambda-cyhalothrin (synthetic) | [79] |
| 3 | Nanoemulsion | Oil-in-water by spontaneous emulsification produce nanoemulsion loaded by EO in core (EO:Tween 80 = 3:1) | D = 131.37 ± 0.29 nm ZP = −30 mV | Sweet orange EO with major monoterpene compounds (R-limonene, β-myrcene, α-pinene, linalool, and sabinene) | Enhance repellency, fumigant, and acute toxicity against Tribolium confusum and Cryptolestes ferrugineus | [87] |
| 4 | Micelle | Droplet emulsion loaded by ethyl acetate fraction (EAF) Lantana camara nano-suspension (EAF:Tween 80 = 3:2) | D = 8.3 ± 1.3 nm, ZP = −8.9 mV | Saponins, alkaloids, and steroids | Moderate toxicity and strong antifeedant activity against Crocidolomia pavonana lavae | [58] |
| 5 | Liposome | Ruta graveolens dichloromethane extract encapsulate in chitosan nanoemulsions and lipid nanosystems (liposomes) with ethanolic injection and thin-film hydration | Liposomes ethanolic D = 121 nm, PI = 0.256, EE = 93% Liposomes thin-film hydration D = 203 nm, PI = 0.185, EE = 73% | Dichloromethane extract of Ruta graveolens (quinolin, benzopyran, and acridone derivatives) | Cytotoxic activity against Spodoptera frugiperda (Sf9) insect cells is more effective than chlorpyrifos (synthetic insecticides) | [81] |
| 6 | Nanocapsules | Nanocapsules formed by chitosan precipitation loaded with Achillea millefolium essential oil (AEO) | D = 85–145 nm | Groups of terpenes: 1,8-cineol, camphor, borneol, phellandrene, and linalool-L | Improving the fumigant effectiveness and efficiency by slow and persistent release against the A. millefolium L. | [83] |
| 7 | Nanocapsules | Nanocapsules formed by SiO2 precipitation loaded by sea fennel EO (SiNPs) | D = 20–78 nm ZP = −11.7 to 34.3 mV | Seed sea fennel essential oil (EO) (dillapiole and γ-terpinene) | Higher toxicity, reduced longevity, and fecundity compared with the nanoemulsion and pure essential oil | [78] |
| 8 | Nanoparticles encapsulation | Chitosan nanoparticles (CSNPs) loaded by Piper nigrum essential oil (PNO) | D = 527.5 nm | Piper nigrum essential oil (PNO) | Toxicity activity, fumigant activity against Sitophilus oryzae and Tribolium castaneum. | [82] |
| 9 | Nanoparticles encapsulation | Zein nanoparticles loaded with geraniol | D = 172.3 ± 3.8 nm, PI = 0.351 ± 0.032, ZP = −12 to −25 mV, EE > 90% | Geraniol and R-citronellal | Decreasing toxicity and phytotoxicity but enhanced repellency against Tetranychus urticae mite. | [88] |
| 10 | Nanoparticles encapsulation | Chitosan and gum arabic nanoparticles containing geraniol | D = 200−300 nm, PI = 0.21−0.78, ZP = −21 to −35 mV, EE = 91−98% | Geraniol | Significant attraction activity (attractant) against whitefly (Bemisia tabaci) | [88] |
| 11 | Mesoporous | Hollow mesoporous silica (HMS) nanoparticles, using carboxylated β-cyclodextrin (CD) as a capping molecule HMS with Si–NH2 as gatekeeper (HMS–NH2), HMS with β-cyclodextrin (β-CD) as gatekeeper (HMS-CD) | HMS (D = 150.16 nm, PI = 0.036, ZP = −2.47 mV) HMS-NH2 (D = 153 nm, PI = 0.041, ZP = 7.21 mV) IDC loaded HMS–NH2 (D = 153.78 nm, PI = 0.034, ZP = −19.7 mV) IDC loaded HMS-CD (D = 193.26 nm, PI = 0.011) HMS average pore size 2.41 nm Loading efficiency 26.42% | Indoxacarb (IDC) carboxylated β-cyclodextrin | Toxic activity against Spodoptera frugiperda | [84] |
| 12 | Mesoporous | Mesoporous silica (MCM) nanoparticles modified by salicylaldimine (Sal-MCM), furfuralimine (Fur-MCM), and benzaldehyde imine (Ben-MCM) | Me (D = not available, ZP = −38.82 mV, SR (10 h) = 70.82%) MCM (D = 833 ± 11 nm, ZP = −20.16 mV, SR (10 h) = 82.88%) Sal-MCM (D = 789 ± 12 nm, ZP = 15.77 mV, SR (10 h) = 48.59%) Fur-MCM (D = 701 ± 12 nm, ZP = 29.89 mV, SR (10 h) = 56.63%) Ben-MCM (D = 763 ± 12 nm, ZP = 25.70 mV, SR (10 h) = 37.21%) | Methyl eugenol (Me) | Attraction activity against Bactrocera dorsalis The best formula shows the highest lure rate of Fur-MCM loading by Me equals 73% of the pure Me | [89] |
| 13 | Mesoporous | Mesoporous silica nanoparticles (MSNs) from tetraethyl orthosilicate (TEOS) hydrolysis modified by cinnamon oil encapsulated with silica nanoparticles (CESN) | Spherical silica nanoparticles, well dispersed in water, provide a maximal interface to load optimal cinnamon oil for the delivery target that induces the biological mechanism indicated by protein profiles | Cinnamon oil | Insect pest Corcyra cephalonica LC50 MSNs Total protein content = 28.88 mg mL−1 Inhibition (pupa = 47.50%, adult emergence = 28%) LC50 CESN Total protein content = 28.65 mg mL−1 Inhibition (pupa = 45.0%, adult emergence = 0%) LC50 cinnamon oil Total protein content = 28.2 mg mL−1 Inhibition (pupa = 37.7%, adult emergence = 0%) Control (−) untreated Total protein content = 32.56 mg mL−1 Inhibition (pupa = 90.0%, adult emergence = 87.5%) Control (+) silica gel Total protein content = 30.32 mg mL−1 Inhibition (pupa = 47.5%, adult emergence = 78.0%) | [90] |
| 14 | Cross-linked nanoparticle | Chitosan nanoparticles (CSNPs) cross-linked by ionic gelation of sodium tripolyphosphate (TPP), coating EO (1% Tween 80) | D < 563.3 nm, ZP = −12.12 mV) EE = 70% Loading capacity > 12.31%. | Peppermint essential oil: L-menthone (32.27%) menthol (23.47%) | The higher toxicity by the mechanism of acetylcholinesterase inhibition on S. oryzae and T. castaneum | [86] |
| 15 | Cross-linked nanoparticle | Chitosan nanoparticles (CSNPs) cross-linked by glutaraldehyde (GLA) and tripolyphosphate (TPP) | Electron micrograph measured: CSNPs (D = 32–90 nm) DLS measured: CSNPs-TPP-PONEEM (D = 122.7 nm, PI = 0.282, EE = 59.34%) CSNPs-GLA/TPP-PONEEM (D = 243.5 nm, PI = 0.57, EE = 65%) | Chitosan and azadirachtin | Effective as antifeedant, larvicidal, and growth-regulating activities, at very low concentrations | [91] |
| 16 | Nanosphere | Encapsulated Zanthoxylum riedelianum fruit EO in poly-ε-caprolactone (PCL) nanospheres | D = 106.7–129.2 nm, pH 6, ZP = < −19.0 mV, EE > 98% | Z. riedelianum fruit EO Major compounds: limo-nene (29.22%), β-myrcene (22.79%), bicyclogermacrene (18.13%) | Effective as oviposition deterrent and low toxicity against nymphs of Bemisia tabaci | [85] |
| No. | Nanoparticles/Nanocarriers | Resources and Compounds | Methods | Compositions | Properties and Performances | Antifeedant Activities | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | Silver nanoparticles | Manilkara zapota leaf aqueous crude extract | Chemical reaction of M. zapota leaf extract and AgNO3 | Manilkara zapota leaf aqueous crude extract = 12 mL Aqueous 1 mM AgNO3 = 88 mL | D = 70–140 nm, nano-size enhances bioactivity and reduces toxicity compared to control | Insect pest M. domestica Positive control: dichlorvos The feeding deterrent activity (3 h exposure): Crude extracts LD50 = 28.35 mg mL−1, LD90 = 89.19 mg mL−1 Synthesized AgNPs LD50 = 3.64 mg mL−1, LD90 = 7.74 mg mL−1 | [98] |
| 2 | Silver nanoparticles | Aristolochia indica leaf aqueous extract | Chemical reaction of A. indica leaf extract and AgNO3 | Aristolochia indica leaf aqueous extract = 15 mL Aqueous 3 mM AgNO3 = 85 mL | D = 112.35 nm, nano-size enhances bioactivity and reduces toxicity compared to control | Insect pest H. armigera larvae Positive control: azadirachtin pure compounds Negative control: Aqueous extract Synthesized Ag NPs Strong antifeedant (92.40%, LC50 365.72 mg mL−1) Azadirachtin pure compounds Strong antifeedant (97.28%, LC50 348.98 mg mL−1) Aqueous extract Good antifeedant (72.22%, LC50 623.3 mg mL−1) | [99] |
| 3 | Silver nanoparticles | Leonotis nepetifolia leaf aqueous extract | The chemical reaction of L. nepetifolia leaf extract and AgNO3 | Aristolochia indica leaf aqueous extract = 2.5 mL Aqueous 1 mM AgNO3 = 47.5 mL | AgNPs D = 37.5 nm (monodisperse and spherical structures) | Insect pest S. litura and H. armigera Positive control: azadirachtin pure compounds Negative control: L. nepetifolia plant extract AgNPs Strong antifeedant activities (FDI) at 150 ppm 78.77% against S. litura 82.16% against H. armigera Azadirachtin pure compounds Strong antifeedant activities (FDI) at 150 ppm 87.43% against S. litura 90.47% against H. armigera L. nepetifolia plant extract moderate antifeedant activities (FDI) at 150 ppm 48.17% against S. litura 50.92% against H. armigera | [100] |
| 4 | Essential oil (EO) nanoparticles | Geranium EO (contain citronellol and geraniol) or Bergamot EO (contain linalyl acetate), and polyethylene glycol (PEG) 6000 as surfactant | Melt-dispersion method | The ratio of essential oil (geranium EO or bergamot EO) to PEG (10%) | Geranium EO-PEG NPs D < 235 nm, PI = 0.280 loading efficacy = 75% Bergamot EO-PEG NPs D < 184 nm, PI = 0.309 loading efficacy = 71% | Insect pest Tribolium castaneum and Rhizopertha dominica Negative control: geranium EO and bergamot EO Geranium EO-PEG NPs Antifeedant activities (FDI) 60% against T. castaneum 48% against R. dominica Geranium EO Antifeedant activities (FDI) 11% against T. castaneum 57% against R. dominica Bergamot EO-PEG NPs Antifeedant activities (FDI) 54% against T. castaneum 39% against R. dominica Bergamot EO Antifeedant activities (FDI) 3% against T. castaneum 83% against R. dominica | [101] |
| 5 | Polymeric nanoparticles | Active compounds: the neem (Azadirachta indica) oil and neem seed kernel extract The polymers: Poly (ε-caprolactone) (PCL), poly (β–hydroxy-butyrate) (PHB), poly (methylmethacrylate) (PMMA) | Colloidal suspensions prepared by interfacial preformed polymer deposition Spray drying of colloidal suspensions | Solution polyvinyl alcohol (PVA) 0.5 mL of benzyl-benzoate containing 12.5 mg of neem oil phospholipids (250 mg) 0.25, 0.5, and 0.75 g PCL (Aldrich) and PHB (Fluka) dissolved in PVA 25 mL 0.25 g PMMA (Aldrich) dissolved in PVA 25 mL | Efficiencies nanocapsules (68%) nanospheres (33%) | Insect pest Spodoptera frugiperda Negative control: neem oil The nanoformulations Antifeedants at 1st DAS: [P]NC-PCL (0.25) Preference Index (PI) 0.77 [S]NS-PHB (0.25) Preference Index (PI) 0.77 neem oil Preference Index (PI) 0.59 Antifeedants at 3rd DAS [P]NC-PCL (0.25) Preference Index (PI) 0.81 [S]NC-PMMA (0.25) Preference Index (PI) 0.88 neem oil Preference Index (PI) 0.90 Antifeedants at 7th DAS [S]NS-PHB (0.25) Preference Index (PI) 0.50 neem oil Preference Index (PI) 0.89 | [102] |
| 6 | Polymeric nanoparticles | Essential oils (EOs) from peppermint and palmarosa Essential oil loaded polymeric nanoparticles (EOPN) Polymer: Polyethylene glycol 6000 | Melt-dispersion method | Solvent ethanol 20 g PEG and 2 g EO | 7 days post-formulation: EOPN-peppermint: D = 310 nm, PI = 0.61, LE = 93.75% EOPN-palmarosa: D = 203 nm, PI = 0.16, LE = 89.7% | Insect pest German cockroach Blatella germanica L. At doses of 4 mg disc−1, EOPN-palmarosa highly antifeedant (FDI = 76.9%) EOPN-peppermint highly antifeedant (FDI = 76.5%) | [103] |
| 7 | Polymeric nanoparticles | Piper nigrum essential oil (PNO) Chitosan nanoparticles (CS/PNO NPs) Polymer: Chitosan and sodium tripolyphosphate (TPP) as cross-linking agent | Emulsification method and ionic gelation | Total solution = 40 mL Chitosan solution 1% w/v + acetic acid solution 1% v/v Tween 80 (0.45 g) PNO (0.81 g) + dichloromethane (4 mL), TPP (4% w/v) | CS/PNO NPs D = 527.5 nm, ZP = −5.34 mV, LE (40.62 ± 0.26)% | Insect pest S. oryzae and T. castaneum Negative control: acetone CS/PNO NPs High antifeedant FDI 100% against S. oryzae and T. castaneum | [82] |
| 8 | Polymeric nanoparticles | PONNEEM® (neem oil, karanj oil, azadirachtin, and karanjin), Chitosan nanoparticles CSNs-TPP-PONNEEM NPs and CSNs-GLA-PONNEEM NPs Polymer: Chitosan and sodium tripolyphosphate (TPP) as cross-linking agent and glutaraldehyde (GLA) | Ultrasonic-added ionic gelation freeze-drying method | PONNEEM® 41% neem oil, 41% karanj oil, 1% azadirachtin, 1% karanjin, 13% emulsifier and stabilize Chitosan in 1% acetic acid (50 mg/50 mL), TPP or GLA (50 mg/50 mL) | CSNs-TPP-PONNEEM NPs (D = 122.7 nm; PI = 0.282, encapsulation efficiencies = 59.34%) CSNs-GLA-PONNEEM NPs (D = 243.5 nm; PI = 0.57, encapsulation efficiencies = 65%) | Insect pest H. armigera larvae Positive control: PONNEEM Negative control: CSNs-TPP and CSNs-GLA PONNEEM Antifeedant activities 100%; at 0.3% CSNs-TPP-PONNEEM Strong antifeedant activity (88.5%) CSNs-GLA-PONNEEM Medium antifeedant activity (72.3%) CSNs-TPP Medium antifeedant activity (76.4%) CSNs-GLA Medium antifeedant activity (65.3%) | [91] |
| 9 | Polymeric nanoparticles | Nanoparticles synthesized from chitosan and carbo-xymethyl chitosan Carboxymethyl chitosan (CS/CMCS-NPs) Cross-linking agent: amino groups glutaraldehyde (GA) | Emulsion chemical cross-linking method with ultrasonic-aided ionic gelation | 0.01% and 0.02% CS/CMCS-NPs, 5 mL of 0.4% CMCS + CS/CS-NPs (1.5 mL of 0.5% glutaralde-hyde solution (GA) + 5 mL 0.3% CS) | SEM (D = 30–50 nm) PSA (D = 142.1 ± 2.0 nm, PI = 0.171 ± 0.002) | Insect pest Solenopsis invicta After 4 days of treatments 0.01% CS/CMCS-NPs S-NPs antifeedant activity (40%) 0.02% CS/CMCS-NPs S-NPs antifeedant activity (50%) After 16 days of treatments 0.01% CS/CMCS-NPs S-NPs antifeedant activity (60%) 0.02% CS/CMCS-NPs S-NPs antifeedant activity (80%) | [104] |
| 10 | Polymeric nanoparticles | The extract neem gum (NGE) powder (the majority of oleic acid compounds 31.45%) Neem gum nano formulation (NGNF) | Simple mixing method | Neem gum aqueous suspension (0.5% w/v) TiCl4 (stabilizing agent) with mixing ratio of 5:95, 10:90, 15:85, 20:80, and 25:75 mL | D = 20–40.83 nm and the average size of 31.27 nm | Insect pest H. armigera and S. litura larvae. Positive control: azadiractin NGNF at 100 ppm Strong antifeedant activity 100% against H. armigera and S. litura larvae. NGE at 100 ppm Medium antifeedant activity 74.82% against H. armigera 82.21% against S. litura larvae. Azadiractin at 100 ppm Medium antifeedant activity 68.26% against H. armigera 76.80% against S. litura larvae. | [40] |
| 11 | Micelle | Ethyl acetate fraction of Lantana camara (saponins, alkaloids, and steroids) Surfactant = Tween 80 | Low energy phase inverse method aided sonication with the variation of surfactant–organic ratio (SOR) | Ethyl acetate fraction (EAF) Lantana camara (0.55% w/v) in aquadest Tween 80:EAF or SOR 1:11 | D = 8.3 ± 1.3 nm, distribution 77%, lowest contact angles (48.5°) on the cabbage leaf surface | Insect pest C. pavonana larvae Negative control: EAF pre-emulsion EAF nanosuspension SOR 11 strong antifeedant category LC50 (0.39%) EAF pre-emulsion weak antifeedant category LC50 (0.69%) | [58] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melanie, M.; Miranti, M.; Kasmara, H.; Malini, D.M.; Husodo, T.; Panatarani, C.; Joni, I.M.; Hermawan, W. Nanotechnology-Based Bioactive Antifeedant for Plant Protection. Nanomaterials 2022, 12, 630. https://doi.org/10.3390/nano12040630
Melanie M, Miranti M, Kasmara H, Malini DM, Husodo T, Panatarani C, Joni IM, Hermawan W. Nanotechnology-Based Bioactive Antifeedant for Plant Protection. Nanomaterials. 2022; 12(4):630. https://doi.org/10.3390/nano12040630
Chicago/Turabian StyleMelanie, Melanie, Mia Miranti, Hikmat Kasmara, Desak Made Malini, Teguh Husodo, Camellia Panatarani, I Made Joni, and Wawan Hermawan. 2022. "Nanotechnology-Based Bioactive Antifeedant for Plant Protection" Nanomaterials 12, no. 4: 630. https://doi.org/10.3390/nano12040630
APA StyleMelanie, M., Miranti, M., Kasmara, H., Malini, D. M., Husodo, T., Panatarani, C., Joni, I. M., & Hermawan, W. (2022). Nanotechnology-Based Bioactive Antifeedant for Plant Protection. Nanomaterials, 12(4), 630. https://doi.org/10.3390/nano12040630








