Visible-Light-Driven Reduced Graphite Oxide as a Metal-Free Catalyst for Degradation of Colored Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Graphite Oxide and Reduced Graphite Oxide
2.3. Characterization
2.4. Dye Degradation Experiments
2.5. The Recyclability and Stability Test of Reduced Graphite Oxide
3. Results and Discussion
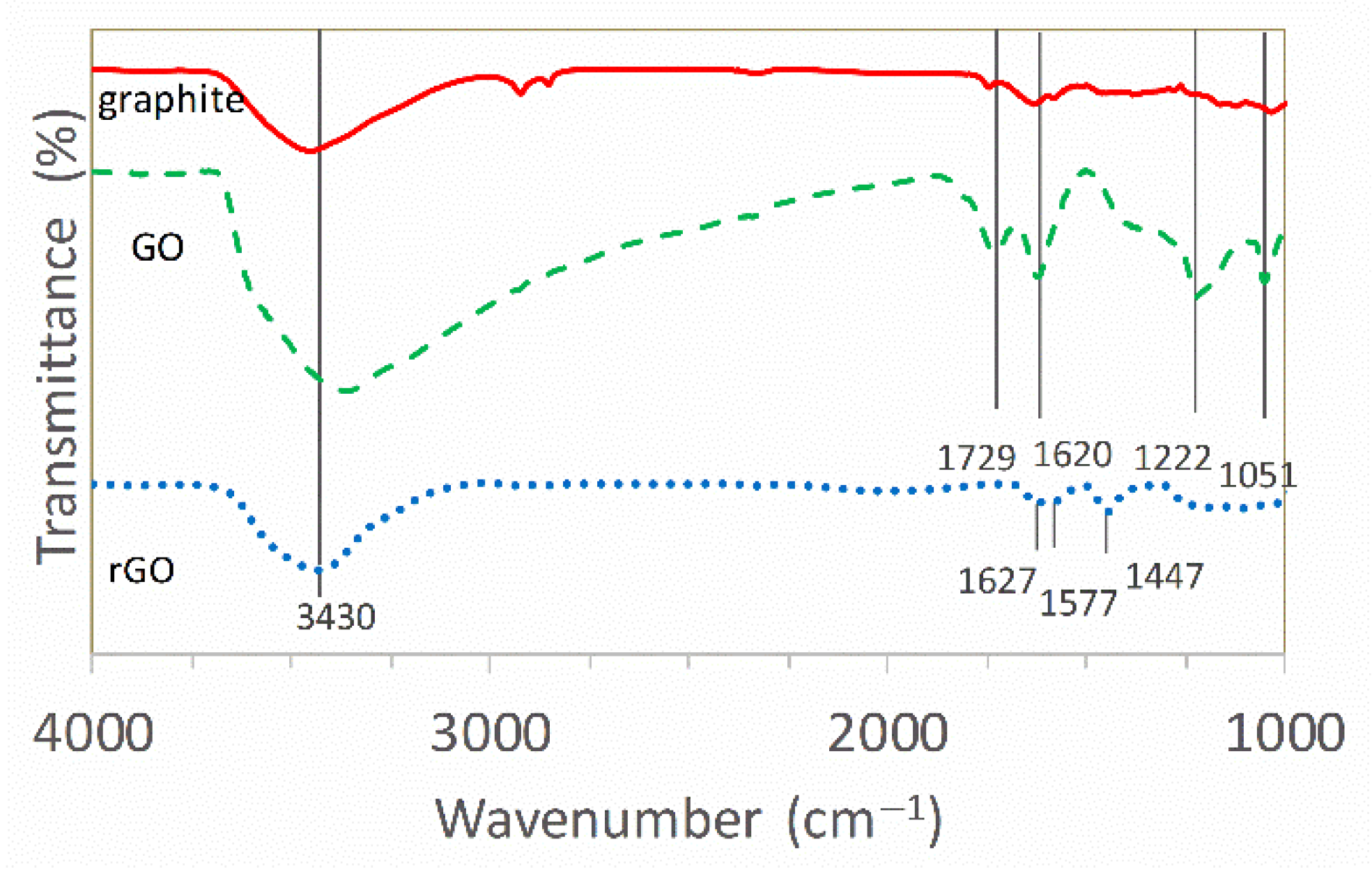
3.1. Characterization
3.2. Dye Degradation Experiments
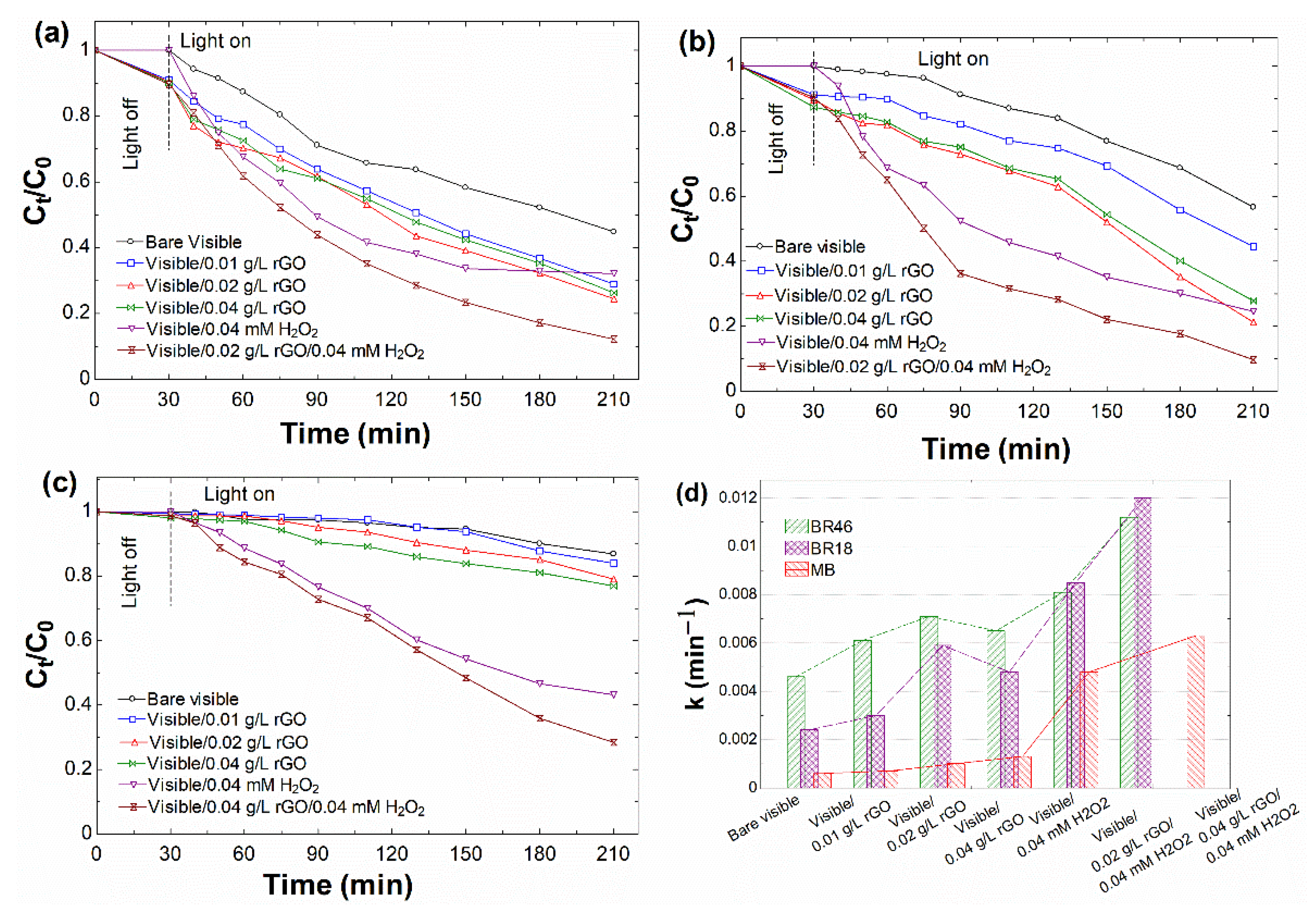
3.2.1. Effect of Catalyst Dosage
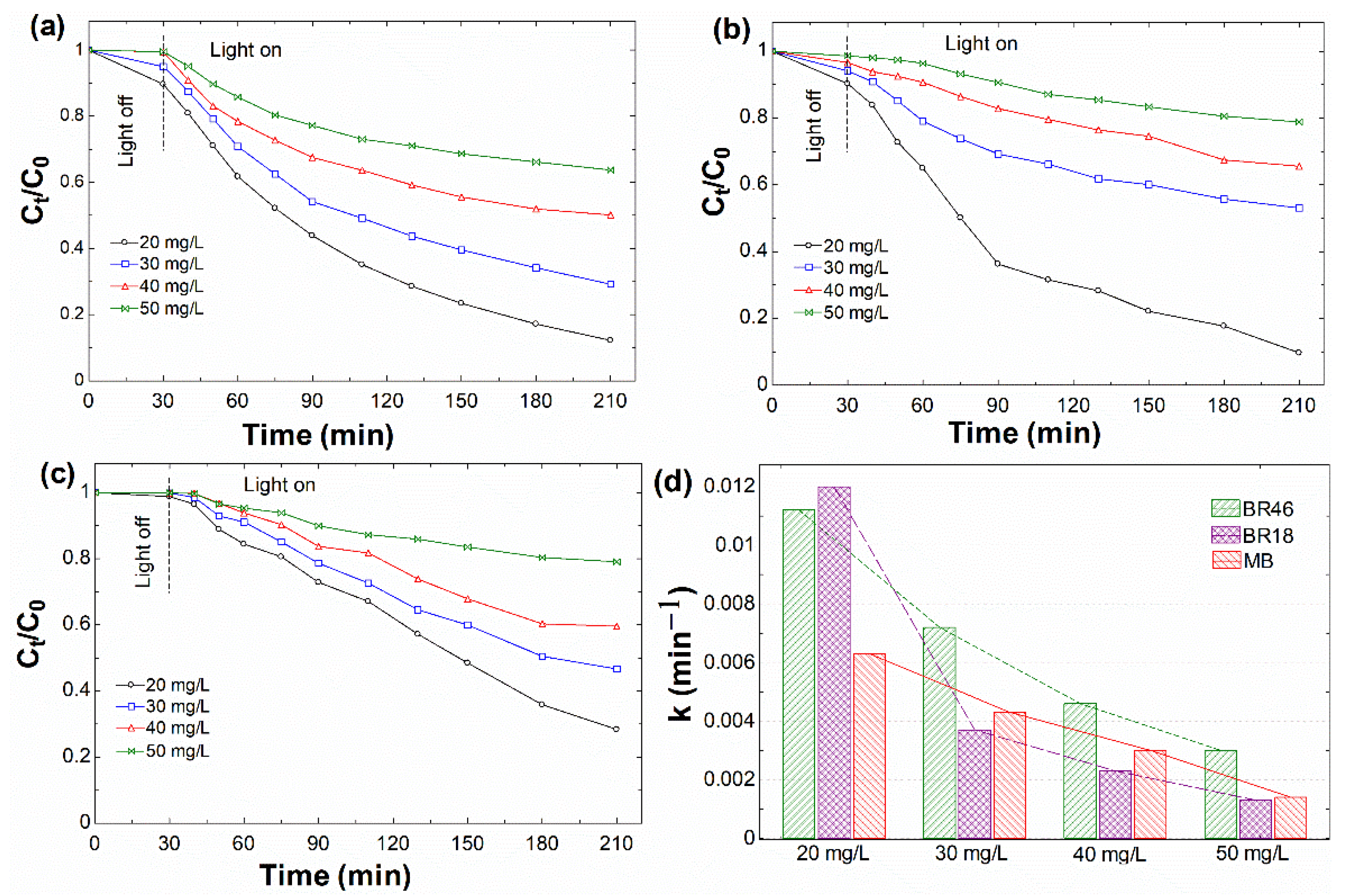
3.2.2. Effect of Initial Dye Concentration
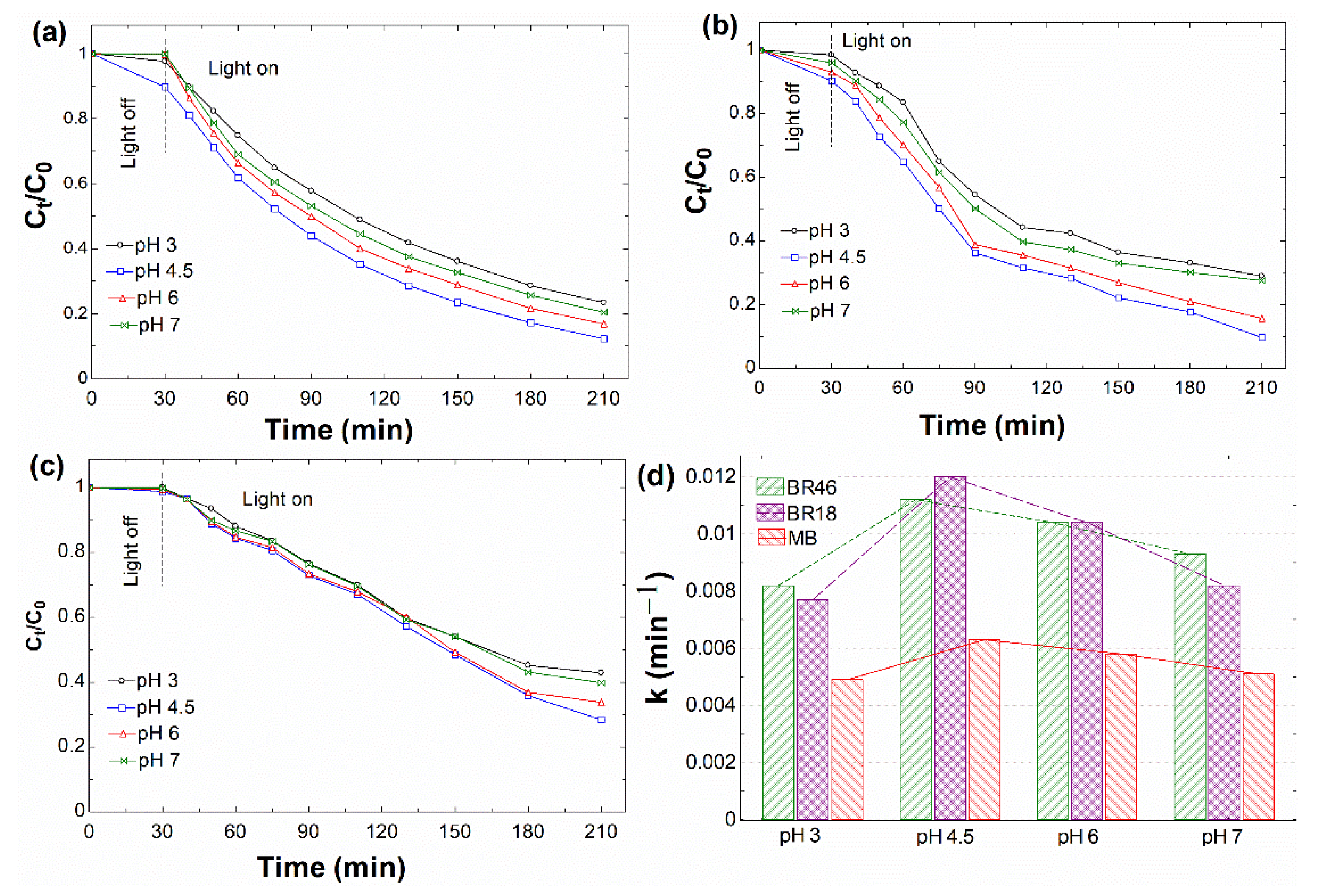
3.2.3. Effect of pH
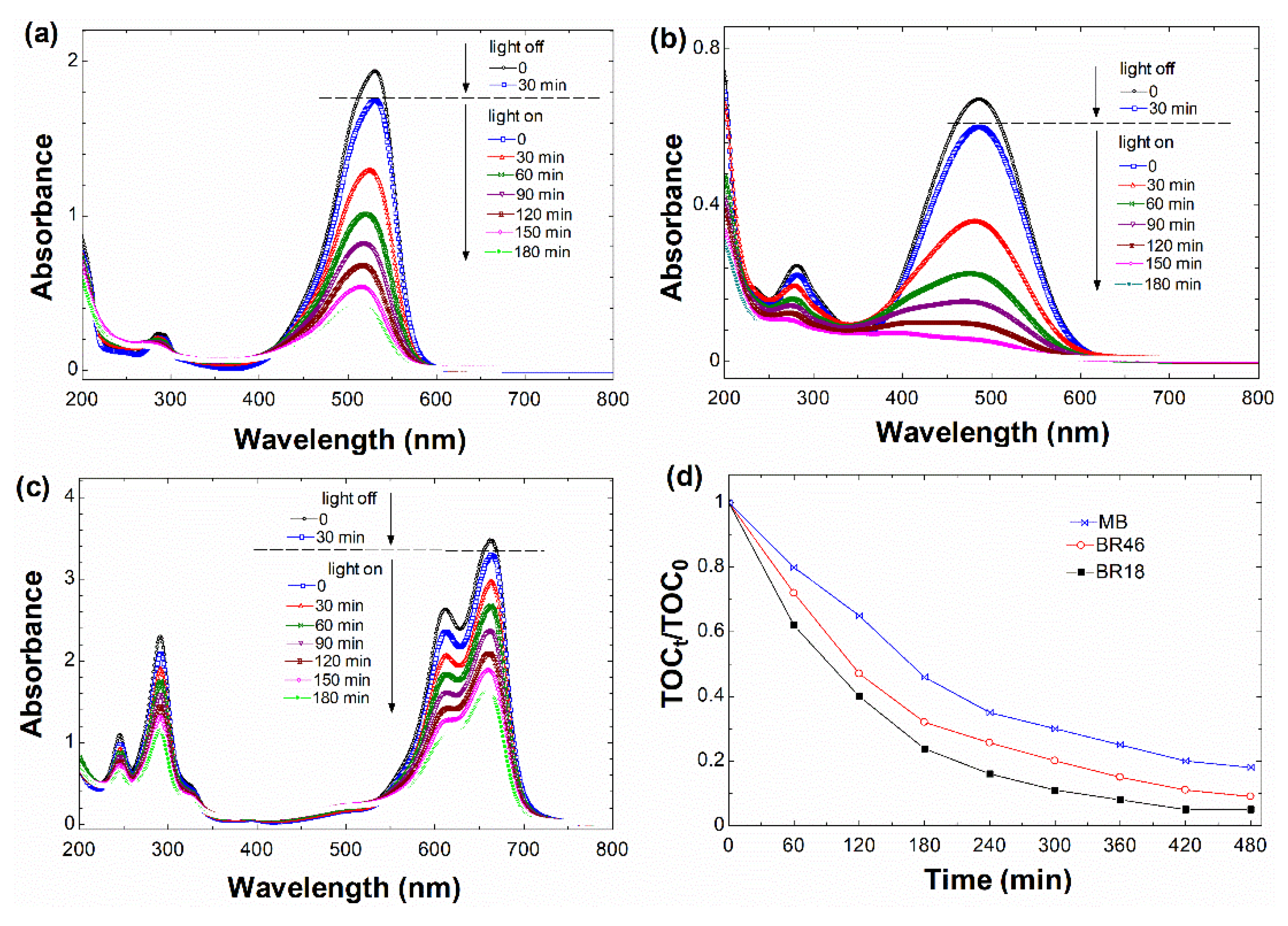
3.2.4. UV-Vis Spectral Changes of Dyes
3.3. Recyclability and Stability
3.4. Efficiency Comparison of the Metal-Free Catalytic Activity of Graphene Derivatives Reported in the Literature
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile finishing dyes and their impact on aquatic environs. Heliyon 2019, 5, e02711. [Google Scholar] [CrossRef] [PubMed]
- Rauf, M.A.; Ashraf, S.S. Fundamental principles and application of heterogeneous photocatalytic degradation of dyes in solution. Chem. Eng. J. 2009, 151, 10–18. [Google Scholar] [CrossRef]
- Saravanan, R.; Karthikeyan, N.; Gupta, V.K.; Thirumal, E.; Thangadurai, P.; Narayanan, V.; Stephen, A. ZnO/Ag nanocomposite: An efficient catalyst for degradation studies of textile effluents under visible light. Mater. Sci. Eng. C 2013, 33, 2235–2244. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.H.S.; Wu, T.Y.; Juan, J.C.; Teh, C.Y. Recent developments of metal oxide semiconductors as photocatalysts in advanced oxidation processes (AOPs) for treatment of dye waste-water. J. Chem. Technol. Biotechnol. 2011, 86, 1130–1158. [Google Scholar] [CrossRef]
- Chowdhury, S.; Balasubramanian, R. Graphene/semiconductor nanocomposites (GSNs) for heterogeneous photocatalytic decolorization of wastewaters contaminated with synthetic dyes: A review. Appl. Catal. B Environ. 2014, 160–161, 307–324. [Google Scholar] [CrossRef]
- Hosseinabadi-Farahani, Z.; Hosseini-Monfared, H.; Mahmoodi, N.M. Graphene oxide nanosheet: Preparation and dye removal from binary system colored wastewater. Desalin. Water Treat. 2015, 56, 2382–2394. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, H.; Hurren, C.; Zhao, J.; Pakdel, E.; Li, Z.; Wang, X. Journal of Environmental Chemical Engineering Degradation of organic dyes by P25-reduced graphene oxide: In fluence of inorganic salts and surfactants. Biochem. Pharmacol. 2015, 3, 1437–1443. [Google Scholar] [CrossRef]
- Chen, D.; Wang, D.; Ge, Q.; Ping, G.; Fan, M.; Qin, L.; Bai, L.; Lv, C.; Shu, K. Graphene-wrapped ZnO nanospheres as a photocatalyst for high performance photocatalysis. Thin Solid Films 2015, 574, 1–9. [Google Scholar] [CrossRef]
- Chorawala, K.; Mehta, M. Applications of nanotechnology in wastewater treatment. Int. J. Innov. Emerg. Res. Eng. 2015, 2, 21–26. [Google Scholar] [CrossRef]
- Liang, J. Graphene-based materials: Fabrication, characterization and application for the decontamination of wastewater and wastegas and hydrogen storage/generation. Adv. Colloid Interface Sci. 2014, 195–196, 19–40. [Google Scholar] [CrossRef]
- Chong, M.N.; Jin, B.; Chow, C.W.; Saint, C. Recent developments in photocatalytic water treatment technology: A review. Water Res. 2010, 44, 2997–3027. [Google Scholar] [CrossRef] [PubMed]
- Sudha, D.; Sivakumar, P. Review on the photocatalytic activity of various composite catalysts. Chem. Eng. Process. Process Intensif. 2015, 97, 112–133. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, Q.; Chen, P.; Chen, L.; Ding, F.; Tang, J.; Li, Y.J.; Au, C.T.; Yin, S.F. Copper-mediated metal-organic framework as efficient photocatalyst for the partial oxidation of aromatic alcohols under visible-light irradiation: Synergism of plasmonic effect and schottky junction. Appl. Catal. B Environ. 2019, 248, 380–387. [Google Scholar] [CrossRef]
- Tang, S.; Fu, Z.; Li, Y.; Li, Y. Study on boron and fluorine-doped C3N4 as a solid activator for cyclohexane oxidation with H2O2 catalyzed by 8-quinolinolato ironIII complexes under visible light irradiation. Appl. Catal. A Gen. 2020, 590, 117342. [Google Scholar] [CrossRef]
- Xiong, S.; Yin, Z.; Zhou, Y.; Peng, X.; Yan, W.; Liu, Z.; Zhang, X. The dual-frequency (20/40 kHz) ultrasound assisted photocatalysis with the active carbon fiber-loaded Fe3+-TiO2 as photocatalyst for degradation of organic dye. Bull. Korean Chem. Soc. 2013, 34, 3039–3045. [Google Scholar] [CrossRef][Green Version]
- Cheng, T.; Gao, H.; Liu, G.; Pu, Z.; Wang, S.; Yi, Z.; Wu, X.; Yang, H. Preparation of core-shell heterojunction photocatalysts by coating CdS nanoparticles onto Bi4Ti3O12 hierarchical microspheres and their photocatalytic removal of organic pollutants and Cr(VI) ions. Colloids Surf. A Physicochem. Eng. Asp. 2022, 633, 127918. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, L.J. Advanced oxidation processes for wastewater treatment: Formation of hydroxyl radical and application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Touati, A.; Hammedi, T.; Najjar, W.; Ksibi, Z.; Sayadi, S. Journal of Industrial and Engineering Chemistry Photocatalytic degradation of textile wastewater in presence of hydrogen peroxide: Effect of cerium doping titania. J. Ind. Eng. Chem. 2016, 35, 36–44. [Google Scholar] [CrossRef]
- Maruthamani, D.; Divakar, D.; Kumaravel, M. Journal of Industrial and Engineering Chemistry Enhanced photocatalytic activity of TiO2 by reduced graphene oxide in mineralization of Rhodamine B dye. J. Ind. Eng. Chem. 2015, 30, 33–43. [Google Scholar] [CrossRef]
- Rong, X.; Qiu, F.; Zhang, C.; Fu, L.; Wang, Y.; Yang, D. Preparation of Ag-AgBr/TiO2-graphene and its visible light photocatalytic activity enhancement for the degradation of polyacrylamide. J. Alloys Compd. 2015, 639, 153–161. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.; Chen, Z.; Hu, J.; Li, S.; Wang, Z.; Liu, J.; Wang, X. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances. Chem. Soc. Rev. 2014, 43, 5234–5244. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K.; Soin, N.; Roy, S.S. Role of graphene/metal oxide composites as photocatalysts, adsorbents and disinfectants in water treatment: A review. RSC Adv. 2014, 4, 3823–3851. [Google Scholar] [CrossRef]
- Zhou, X.; Shi, T.; Zhou, H. Hydrothermal preparation of ZnO-reduced graphene oxide hybrid with high performance in photocatalytic degradation. Appl. Surf. Sci. 2012, 258, 6204–6211. [Google Scholar] [CrossRef]
- Li, J.; Zhou, S.L.; Hong, G.B.; Chang, C.T. Hydrothermal preparation of P25-graphene composite with enhanced adsorption and photocatalytic degradation of dyes. Chem. Eng. J. 2013, 219, 486–491. [Google Scholar] [CrossRef]
- Su, X.; Wang, G.; Li, W.; Bai, J.; Wang, H. A simple method for preparing graphene nano-sheets at low temperature. Adv. Powder Technol. 2013, 24, 317–323. [Google Scholar] [CrossRef]
- Julkapli, N.M.; Bagheri, S. Graphene supported heterogeneous catalysts: An overview. Int. J. Hydrogen. Energy 2015, 40, 948–979. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef]
- Espinosa, J.C.; Navalõn, S.; Primo, A.; Moral, M.; Sanz, J.F.; Álvaro, M.; García, H. Graphenes as Efficient Metal-Free Fenton Catalysts. Chem. Eur. J. 2015, 21, 11966–11971. [Google Scholar] [CrossRef]
- Espinosa, J.C.; Navalón, S.; Álvaro, M.; García, H. Reduced Graphene Oxide as a Metal-Free Catalyst for the Light-Assisted Fenton-Like Reaction. ChemCatChem 2016, 8, 2642–2648. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Qian, J. Graphene aerogel-based catalysts in Fenton-like reactions for water decontamination: A short review. Chem. Eng. J. Adv. 2021, 8, 100171. [Google Scholar] [CrossRef]
- Su, C.; Loh, K.P. Carbocatalysts: Graphene Oxide and Its Derivatives BT—Accounts of Chemical Research. Carbocatalysts 2012, 46, 2275–2285. [Google Scholar] [CrossRef]
- Primo, A.; Neatu, F.; Florea, M.; Parvulescu, V.; Garcia, H. Graphenes in the absence of metals as carbocatalysts for selective acetylene hydrogenation and alkene hydrogenation. Nat. Commun. 2014, 5, 5291. [Google Scholar] [CrossRef] [PubMed]
- Putri, L.K.; Ong, W.J.; Chang, W.S.; Chai, S.P. Heteroatom doped graphene in photocatalysis: A review. Appl. Surf. Sci. 2015, 358, 2–14. [Google Scholar] [CrossRef]
- Nd, M.; Analogue, S. S-TiO2 Decorated on Reduced Graphene Oxide for a Visible Light Active Photocatalyst for Dye Degradation: Compariso. Ind. Eng. Chem. Res. 2014, 53, 14329–14338. [Google Scholar] [CrossRef]
- Wong, C.P.P.; Lai, C.W.; Lee, K.M.; Abd Hamid, S.B. Advanced chemical reduction of reduced graphene oxide and its photocatalytic activity in degrading reactive black 5. Materials 2015, 8, 7118–7128. [Google Scholar] [CrossRef]
- Te-Fu, Y.; Jhih-Ming, S.; Ching, C.; Ting-Hsiang, C.; Hsisheng, T. Graphite Oxide as a Photocatalyst for Hydrogen Production from Water. Adv. Funct. Mater. 2010, 20, 2255–2262. [Google Scholar] [CrossRef]
- Yeh, T.F.; Chan, F.F.; Hsieh, C.T.; Teng, H. Graphite oxide with different oxygenated levels for hydrogen and oxygen production from water under illumination: The band positions of graphite oxide. J. Phys. Chem. C 2011, 115, 22587–22597. [Google Scholar] [CrossRef]
- Santhiran, A.; Iyngaran, P.; Abiman, P.; Kuganathan, N. Graphene Synthesis and Its Recent Advances in Applications—A Review. C 2021, 7, 76. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, M.; Hedtke, T.; Deshmukh, A.; Zhou, X.; Weon, S.; Elimelech, M.; Kim, J.H. Mechanism of Heterogeneous Fenton Reaction Kinetics Enhancement under Nanoscale Spatial Confinement. Environ. Sci. Technol. 2020, 54, 10868–10875. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Lv, R.; Zhang, W.; Lu, J.; Ren, Y.; Li, X.; Lv, L.; Hua, M.; Pan, B. Exploring mechanisms of different active species formation in heterogeneous Fenton systems by regulating iron chemical environment. Appl. Catal. B Environ. 2021, 295, 120282. [Google Scholar] [CrossRef]
- Wang, G.; Xiang, J.; Lin, J.; Xiang, L.; Chen, S.; Yan, B.; Fan, H.; Zhang, S.; Shi, X. Sustainable Advanced Fenton-like Catalysts Based on Mussel-Inspired Magnetic Cellulose Nanocomposites to Effectively Remove Organic Dyes and Antibiotics. ACS Appl. Mater. Interfaces 2020, 12, 51952–51959. [Google Scholar] [CrossRef] [PubMed]
- Waite, T.D.; Chen, Y.; Miller, C.J. Heterogeneous Fenton chemistry revisited: Mechanistic insights from ferrihydrite-mediated oxidation of formate and oxalate. Environ. Sci. Technol. 2021, 55, 14414–14425. [Google Scholar] [CrossRef]
- Vigo, T.L. (Ed.) Chapter 3—Methods of Applying Dyes to Textiles. In Textile Processing and Properties; Elsevier: Amsterdam, The Netherlands, 1994; Volume 11, pp. 112–192. ISBN 0920-4083. [Google Scholar]
- Oh, S.W.; Kang, M.N.; Cho, C.W.; Lee, M.W. Detection of carcinogenic amines from dyestuffs or dyed substrates. Dyes Pigment. 1997, 33, 119–135. [Google Scholar] [CrossRef]
- Pei, S.; Cheng, H.M. The reduction of graphene oxide. Carbon N. Y. 2012, 50, 3210–3228. [Google Scholar] [CrossRef]
- Bosch-Navarro, C.; Coronado, E.; Martí-Gastaldo, C.; Sánchez-Royo, J.F.; Gómez, M.G. Influence of the pH on the synthesis of reduced graphene oxide under hydrothermal conditions. Nanoscale 2012, 4, 3977–3982. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi, N.M. Binary catalyst system dye degradation using photocatalysis. Fibers Polym. 2014, 15, 273–280. [Google Scholar] [CrossRef]
- Mahmoodi, N.M. Photodegradation of Dyes Using Multiwalled Carbon Nanotube and Ferrous Ion. J. Environ. Eng. 2013, 139, 1368–1374. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Yao, W.; Zong, R.; Zhu, Y. New insights into the relationship between photocatalytic activity and TiO2-GR composites. RSC Adv. 2015, 5, 29201–29208. [Google Scholar] [CrossRef]
- Gao, W.; Ran, C.; Wang, M.; Li, L.; Sun, Z.; Yao, X. The role of reduction extent of graphene oxide in the photocatalytic performance of Ag/AgX (X = Cl, Br)/rGO composites and the pseudo-second-order kinetics reaction nature of the Ag/AgBr system. Phys. Chem. Chem. Phys. 2016, 18, 18219–18226. [Google Scholar] [CrossRef]
- Loryuenyong, V.; Totepvimarn, K.; Eimburanapravat, P.; Boonchompoo, W.; Buasri, A. Preparation and characterization of reduced graphene oxide sheets via water-based exfoliation and reduction methods. Adv. Mater. Sci. Eng. 2013, 2013, 923403. [Google Scholar] [CrossRef]
- Thomas, R.T.; Rasheed, P.A.; Sandhyarani, N. Synthesis of nanotitania decorated few-layer graphene for enhanced visible light driven photocatalysis. J. Colloid Interface Sci. 2014, 428, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; An, J.; Potts, J.R.; Velamakanni, A.; Murali, S.; Ruoff, R.S. Hydrazine-reduction of graphite- and graphene oxide. Carbon N. Y. 2011, 49, 3019–3023. [Google Scholar] [CrossRef]
- Kavitha, M.K.; Pillai, S.C.; Gopinath, P.; John, H. Journal of Environmental Chemical Engineering Hydrothermal synthesis of ZnO decorated reduced graphene oxide: Understanding the mechanism of photocatalysis. Biochem. Pharmacol. 2015, 3, 1194–1199. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.T.; Ruoff, R.S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon N. Y. 2007, 45, 1558–1565. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, Y.; Zhou, M.; Qian, H.; Hu, X. Microwave-assisted non-aqueous route to deposit well-dispersed ZnO nanocrystals on reduced graphene oxide sheets with improved photoactivity for the decolorization of dyes under visible light. Appl. Catal. B Environ. 2012, 125, 425–431. [Google Scholar] [CrossRef]
- Nethravathi, C.; Rajamathi, M. Chemically modified graphene sheets produced by the solvothermal reduction of colloidal dispersions of graphite oxide. Carbon N. Y. 2008, 46, 1994–1998. [Google Scholar] [CrossRef]
- Kellici, S.; Acord, J.; Ball, J.; Reehal, H.S.; Morgan, D.; Saha, B. A single rapid route for the synthesis of reduced graphene oxide with antibacterial activities. RSC Adv. 2014, 4, 14858–14861. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, Q.; Xu, Q.; Li, M.; Tang, N.; Du, Y. Synthesis and photoluminescence of F and N co-doped reduced graphene oxide. Carbon N. Y. 2013, 61, 436–440. [Google Scholar] [CrossRef]
- Saxena, S.; Tyson, T.A.; Negusse, E. Investigation of the local structure of graphene oxide. J. Phys. Chem. Lett. 2010, 1, 3433–3437. [Google Scholar] [CrossRef]
- Sun, L.; Wang, G.; Hao, R.; Han, D.; Cao, S. Solvothermal fabrication and enhanced visible light photocatalytic activity of Cu2O-reduced graphene oxide composite microspheres for photodegradation of Rhodamine B. Appl. Surf. Sci. 2015, 358, 91–99. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Mohan, R.; Kim, S. Graphene oxide as a photocatalytic material Advertisement: Graphene oxide as a photocatalytic material. Appl. Phys. Lett. 2011, 98, 244101. [Google Scholar] [CrossRef]
- Thema, F.T.; Moloto, M.J.; Dikio, E.D.; Nyangiwe, N.N.; Kotsedi, L.; Maaza, M.; Khenfouch, M. Synthesis and Characterization of Graphene Thin Films by Chemical Reduction of Exfoliated and Intercalated Graphite Oxide. J. Chem. 2013, 2013, 150536. [Google Scholar] [CrossRef]
- Loh, K.P.; Zhou, Y.; Bao, Q.L.; Tang, L.A.L.; Zhong, Y.L. Hydrothermal Dehydration for the “Green” Reduction of Exfoliated Graphene Oxide to Graphene and Demonstration of Tunable Optical Limiting Properties. Chem. Mater. 2009, 21, 2950–2956. [Google Scholar]
- Gandhiraman, R.P.; Nordlund, D.; Javier, C.; Koehne, J.E.; Chen, B.; Meyyappan, M. X-ray absorption study of graphene oxide and transition metal oxide nanocomposites. J. Phys. Chem. C 2014, 118, 18706–18712. [Google Scholar] [CrossRef]
- Saranya, M.; Santhosh, C.; Ramachandran, R.; Nirmala Grace, A. Growth of CuS nanostructures by hydrothermal route and its optical properties. J. Nanotechnol. 2014, 2014, 321571. [Google Scholar] [CrossRef]
- Suram, S.K.; Newhouse, P.F.; Gregoire, J.M. High Throughput Light Absorber Discovery, Part 1: An Algorithm for Automated Tauc Analysis. ACS Comb. Sci. 2016, 18, 673–681. [Google Scholar] [CrossRef]
- Makuła, P.; Pacia, M.; Macyk, W. How To Correctly Determine the Band Gap Energy of Modified Semiconductor Photocatalysts Based on UV-Vis Spectra. J. Phys. Chem. Lett. 2018, 9, 6814–6817. [Google Scholar] [CrossRef]
- Coulter, J.B.; Birnie, D.P. Assessing Tauc Plot Slope Quantification: ZnO Thin Films as a Model System. Phys. Status Solidi Basic Res. 2018, 255, 1700393. [Google Scholar] [CrossRef]
- Herrmann, J.-M. Heterogeneous photocatalysis: Fundamentals and applications to the removal of various types of aqueous pollutants. Catal. Today 1999, 53, 115–129. [Google Scholar] [CrossRef]
- Mazarji, M.; Nabi-Bidhendi, G.; Mahmoodi, N.M. One-pot synthesis of a reduced graphene oxide–ZnO nanorod composite and dye decolorization modeling. J. Taiwan Inst. Chem. Eng. 2017, 80, 439–451. [Google Scholar] [CrossRef]
- Fan, H.; Zhao, X.; Yang, J.; Shan, X.; Yang, L.; Zhang, Y.; Li, X.; Gao, M. ZnO-graphene composite for photocatalytic degradation of methylene blue dye. Catal. Commun. 2012, 29, 29–34. [Google Scholar] [CrossRef]
- Omran, B.; Baek, K.-H. Graphene-derived antibacterial nanocomposites for water disinfection: Current and future perspectives. Environ. Pollut. 2022, 298, 118836. [Google Scholar] [CrossRef] [PubMed]
- Sanjaya, D.P.; Ruperto, G.M.; Khiem, V.; Nijem, N.; Oliver, S.; Yves, C.; Balkus, K.J., Jr. Hydrothermal Synthesis of Graphene-TiO2 Nanotube Composites with Enhanced Photocatalytic Activity. ACS Catal. 2012, 2, 949–956. [Google Scholar] [CrossRef]
- Liu, J.; Bai, H.; Wang, Y.; Liu, Z.; Zhang, X.; Sun, D.D. Self-assembling TiO2 nanorods on large graphene oxide sheets at a two-phase interface and their anti-recombination in photocatalytic applications. Adv. Funct. Mater. 2010, 20, 4175–4181. [Google Scholar] [CrossRef]
- Tseng, D.H.; Juang, L.C.; Huang, H.H. Effect of oxygen and hydrogen peroxide on the photocatalytic degradation of monochlorobenzene in TiO2 aqueous suspension. Int. J. Photoenergy 2012, 2012, 328526. [Google Scholar] [CrossRef]
- Pham, V.H.; Shin, E.W.; Pham, H.; Kim, S.; Chung, J.S.; Kim, E.J.; Hur, S.H. The role of graphene oxide content on the adsorption-enhanced photocatalysis of titanium dioxide/graphene oxide composites. Chem. Eng. J. 2011, 170, 226–232. [Google Scholar] [CrossRef]
- Ghaly, M.Y.; Farah, J.Y.; Fathy, A.M. Enhancement of decolorization rate and COD removal from dyes containing wastewater by the addition of hydrogen peroxide under solar photocatalytic oxidation. Desalination 2007, 217, 74–84. [Google Scholar] [CrossRef]
- Rathi, A.; Rajor, H.K.; Sharma, R.K. Photodegradation of direct yellow-12 using UV/H2O2/Fe2+. J. Hazard. Mater. 2003, 102, 231–241. [Google Scholar] [CrossRef]
- Kritikos, D.E.; Xekoukoulotakis, N.P.; Psillakis, E.; Mantzavinos, D. Photocatalytic degradation of reactive black 5 in aqueous solutions: Effect of operating conditions and coupling with ultrasound irradiation. Water Res. 2007, 41, 2236–2246. [Google Scholar] [CrossRef]
- Behnajady, M.A.; Modirshahla, N.; Hamzavi, R. Kinetic study on photocatalytic degradation of C.I. Acid Yellow 23 by ZnO photocatalyst. J. Hazard. Mater. 2006, 133, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi, N.M.; Oveisi, M.; Arabi, A.M.; Karimi, B. Cadmium selenide quantum dots: Synthesis, characterization, and dye removal ability with UV irradiation. Desalin. Water Treat. 2016, 57, 16552–16558. [Google Scholar] [CrossRef]
- Bansal, P.; Chaudhary, G.R.; Mehta, S.K. Comparative study of catalytic activity of ZrO2 nanoparticles for sonocatalytic and photocatalytic degradation of cationic and anionic dyes. Chem. Eng. J. 2015, 280, 475–485. [Google Scholar] [CrossRef]
- Iqbal, M.Z.; Pal, P.; Shoaib, M.; Abdala, A.A. Efficient removal of different basic dyes using graphene. Desalin. Water Treat. 2017, 68, 226–235. [Google Scholar] [CrossRef]
- Kashyap, S.; Mishra, S.; Behera, S.K. Aqueous Colloidal Stability of Graphene Oxide and Chemically Converted Graphene. J. Nanopart. 2014, 2014, 640281. [Google Scholar] [CrossRef]
- Muruganandham, M.; Swaminathan, M. Photochemical oxidation of reactive azo dye with UV-H2O2 process. Dyes Pigment. 2004, 62, 269–275. [Google Scholar] [CrossRef]
- Nanoparticles, U.T.; Radich, J.G.; Krenselewski, A.L.; Zhu, J.; Prashant, V. Supporting Information: Is Graphene a Stable Platform for Photocatalysis? Mineralization of Reduced Graphene Oxide with. Chem. Mater. 2014, 26, 4662–4668. [Google Scholar]
- Feng, Y.; Lu, K.; Mao, L.; Guo, X.; Gao, S.; Petersen, E.J. Degradation of 14C-labeled few layer graphene via Fenton reaction: Reaction rates, characterization of reaction products, and potential ecological effects. Water Res. 2015, 84, 49–57. [Google Scholar] [CrossRef]
- Lyu, L.; Yu, G.; Zhang, L.; Hu, C.; Sun, Y. 4-Phenoxyphenol-Functionalized Reduced Graphene Oxide Nanosheets: A Metal-Free Fenton-Like Catalyst for Pollutant Destruction. Environ. Sci. Technol. 2018, 52, 747–756. [Google Scholar] [CrossRef]
- Siong, V.L.E.; Lee, K.M.; Juan, J.C.; Lai, C.W.; Tai, X.H.; Khe, C.S. Removal of methylene blue dye by solvothermally reduced graphene oxide: A metal-free adsorption and photodegradation method. RSC Adv. 2019, 9, 37686–37695. [Google Scholar] [CrossRef]




| Parameters | BR46 | BR18 | MB | |||
|---|---|---|---|---|---|---|
| k (min−1) | R2 | k (min−1) | R2 | k (min−1) | R2 | |
| Catalyst dosage (g/L) | ||||||
| Without catalyst | 0.0046 | 0.9861 | 0.0024 | 0.8863 | 0.0006 | 0.9026 |
| 0.01 | 0.0061 | 0.9963 | 0.0030 | 0.8602 | 0.0007 | 0.8999 |
| 0.02 | 0.0071 | 0.9861 | 0.0059 | 0.8851 | 0.0010 | 0.9018 |
| 0.04 | 0.0065 | 0.9885 | 0.0048 | 0.8688 | 0.0013 | 0.9759 |
| H2O2 addition (mM) | ||||||
| 0.04 without catalyst | 0.0081 | 0.8804 | 0.0085 | 0.9637 | 0.0048 | 0.9903 |
| 0.04 at optimum catalyst dosage | 0.0112 | 0.9982 | 0.0120 | 0.9825 | 0.0063 | 0.9679 |
| Dye (mg/L) | ||||||
| 20 | 0.0112 | 0.9982 | 0.0120 | 0.9825 | 0.0063 | 0.9679 |
| 30 | 0.0072 | 0.9605 | 0.0037 | 0.9132 | 0.0043 | 0.9915 |
| 40 | 0.0046 | 0.8952 | 0.0023 | 0.9906 | 0.0030 | 0.9751 |
| 50 | 0.0030 | 0.8789 | 0.0013 | 0.9802 | 0.0014 | 0.9757 |
| pH | ||||||
| 3.0 | 0.0082 | 0.9967 | 0.0077 | 0.9483 | 0.0049 | 0.9878 |
| 4.5 | 0.0112 | 0.9982 | 0.0120 | 0.9825 | 0.0063 | 0.9679 |
| 6.0 | 0.0104 | 0.9862 | 0.0104 | 0.9727 | 0.0058 | 0.9746 |
| 7.0 | 0.0093 | 0.9855 | 0.0082 | 0.9251 | 0.0051 | 0.9881 |
| Sample | Method of Synthesis | Target Pollution and Concentration | Light Source | H2O2 (mM) | rGO Dosage (g/L) | pH | Time | Efficiency (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| rGO | Oxidation by modified Hummers’ method following with thermal reduction at 200 °C | Phenol (100 mg/L) | Visible | 5.88 | 0.2 | 3 | 150 h | ≈90 | [28,29] |
| rGO | Oxidation by modified Hummers’ method following with thermal reduction at 350 °C | Bisphenol A (10 mg/L) | No light | 10 | 0.4 | 6.5 | 120 min | 20 | [90] |
| 2 chlorophenol (10 mg/L) | ≈40 | ||||||||
| rGO | Oxidation by modified Hummers’ method following with solvothermal reduction at 160 °C | Methylene blue (50 mg/L) | UV-C (light intensity: 60 W m−2) | 5.88 | 0.02 | 11 | 6 h | ≈90 | [91] |
| rGO | Oxidation by modified Hummers’ method and two-step reduction via hydrothermal and calcination in N2 atmosphere | Basic red 46 (20 mg/L) | Visible | 0.04 | 0.04 | 4.5 | 210 min | ≈88 | Present study |
| Basic red 18 (20 mg/L) | ≈92 | ||||||||
| Methylene blue (20 mg/L) | ≈70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazarji, M.; Mahmoodi, N.M.; Nabi Bidhendi, G.; Minkina, T.; Sushkova, S.; Mandzhieva, S.; Bauer, T.; Soldatov, A. Visible-Light-Driven Reduced Graphite Oxide as a Metal-Free Catalyst for Degradation of Colored Wastewater. Nanomaterials 2022, 12, 374. https://doi.org/10.3390/nano12030374
Mazarji M, Mahmoodi NM, Nabi Bidhendi G, Minkina T, Sushkova S, Mandzhieva S, Bauer T, Soldatov A. Visible-Light-Driven Reduced Graphite Oxide as a Metal-Free Catalyst for Degradation of Colored Wastewater. Nanomaterials. 2022; 12(3):374. https://doi.org/10.3390/nano12030374
Chicago/Turabian StyleMazarji, Mahmoud, Niyaz Mohammad Mahmoodi, Gholamreza Nabi Bidhendi, Tatiana Minkina, Svetlana Sushkova, Saglara Mandzhieva, Tatiana Bauer, and Alexander Soldatov. 2022. "Visible-Light-Driven Reduced Graphite Oxide as a Metal-Free Catalyst for Degradation of Colored Wastewater" Nanomaterials 12, no. 3: 374. https://doi.org/10.3390/nano12030374
APA StyleMazarji, M., Mahmoodi, N. M., Nabi Bidhendi, G., Minkina, T., Sushkova, S., Mandzhieva, S., Bauer, T., & Soldatov, A. (2022). Visible-Light-Driven Reduced Graphite Oxide as a Metal-Free Catalyst for Degradation of Colored Wastewater. Nanomaterials, 12(3), 374. https://doi.org/10.3390/nano12030374














