Removal of Benzene and Toluene from Synthetic Wastewater by Adsorption onto Magnetic Zeolitic Imidazole Framework Nanocomposites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
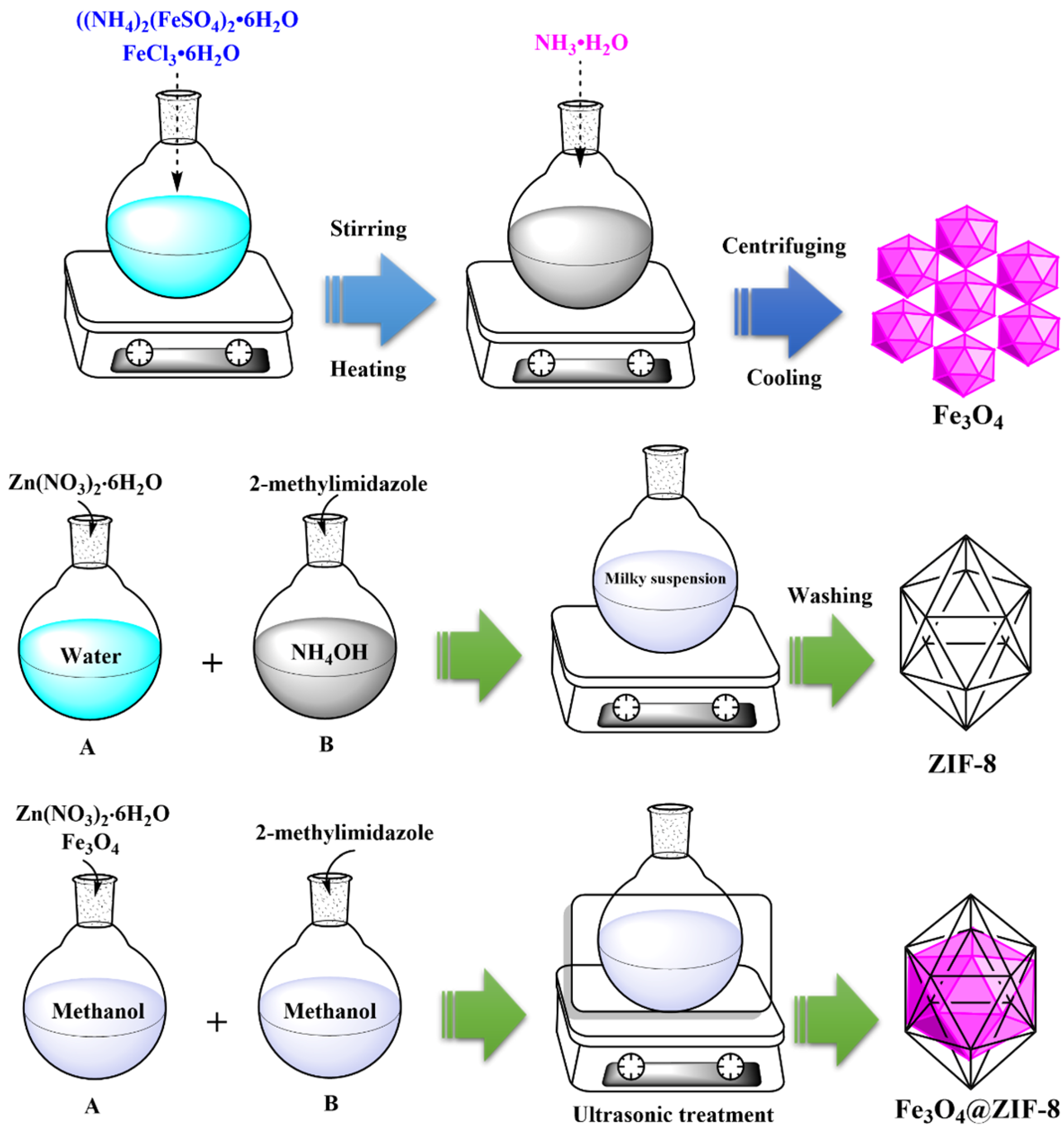
2.2. Synthesis of Fe3O4
2.3. Synthesis of Zeolitic Imidazole Framework-8 (ZIF-8)
2.4. Synthesis of Fe3O4@ZIF-8
2.5. Adsorption Experimental Design
2.6. Devices Used in the Study
3. Results and Discussion
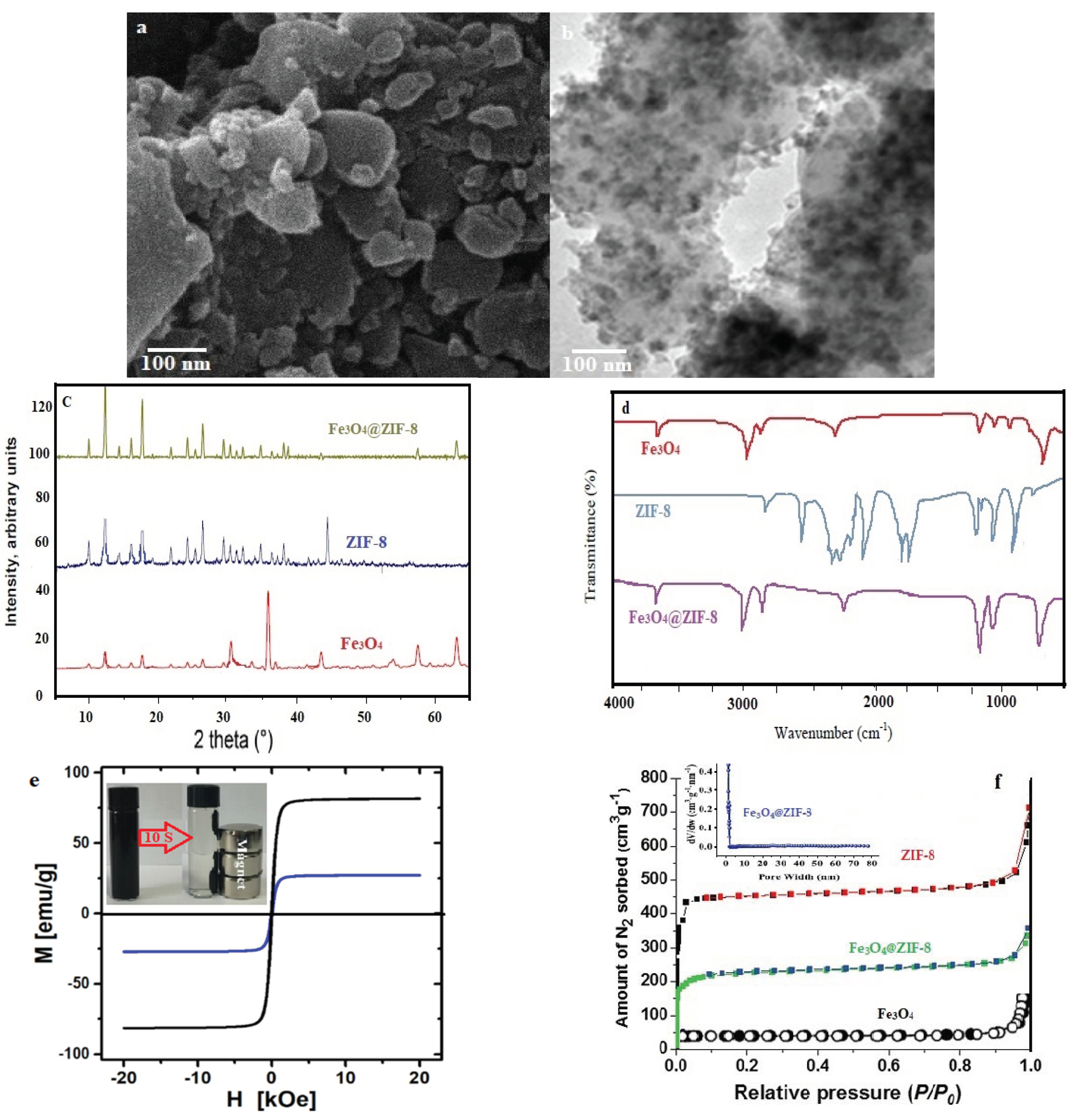
3.1. Characterizations
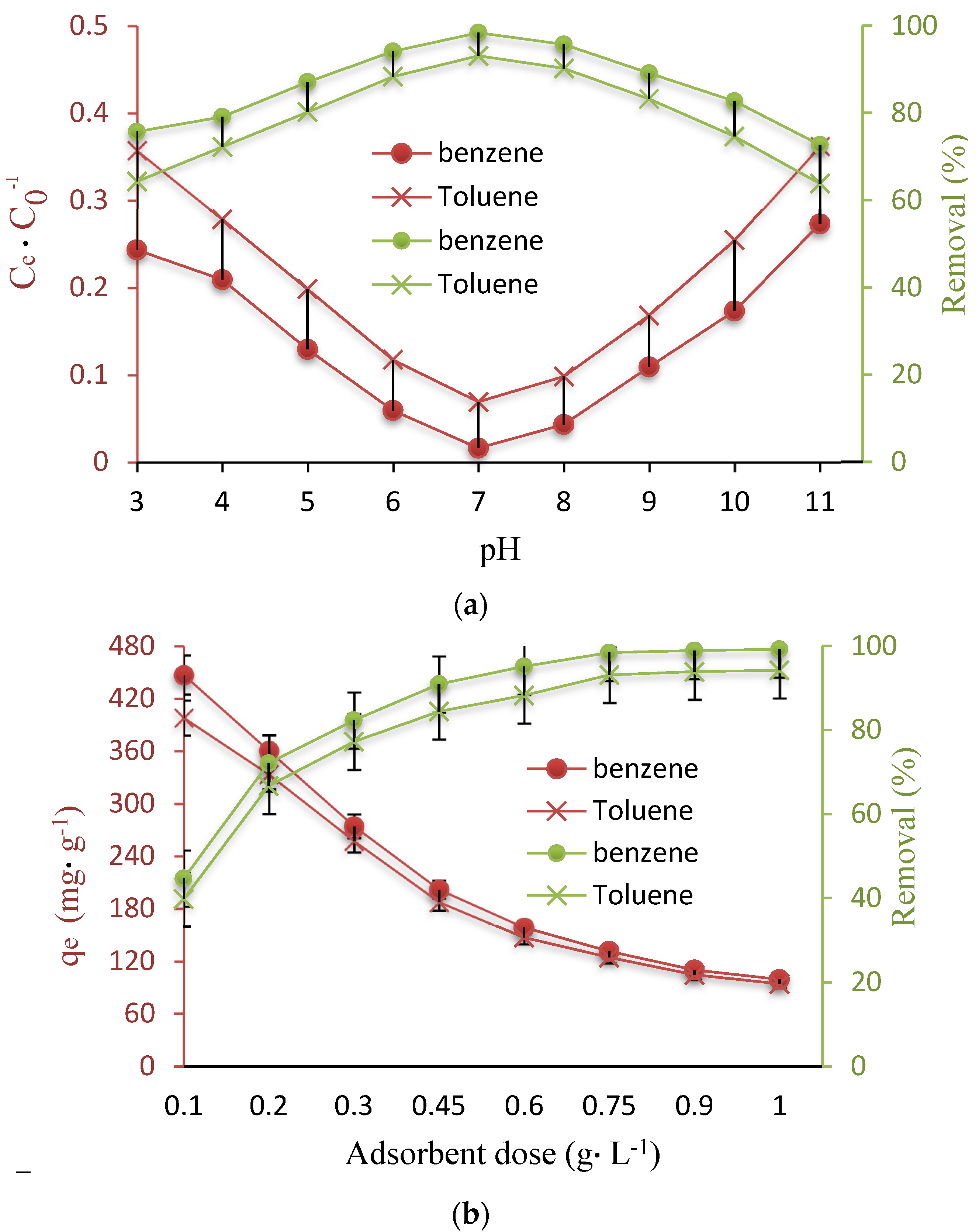
3.2. Influence of Parameters
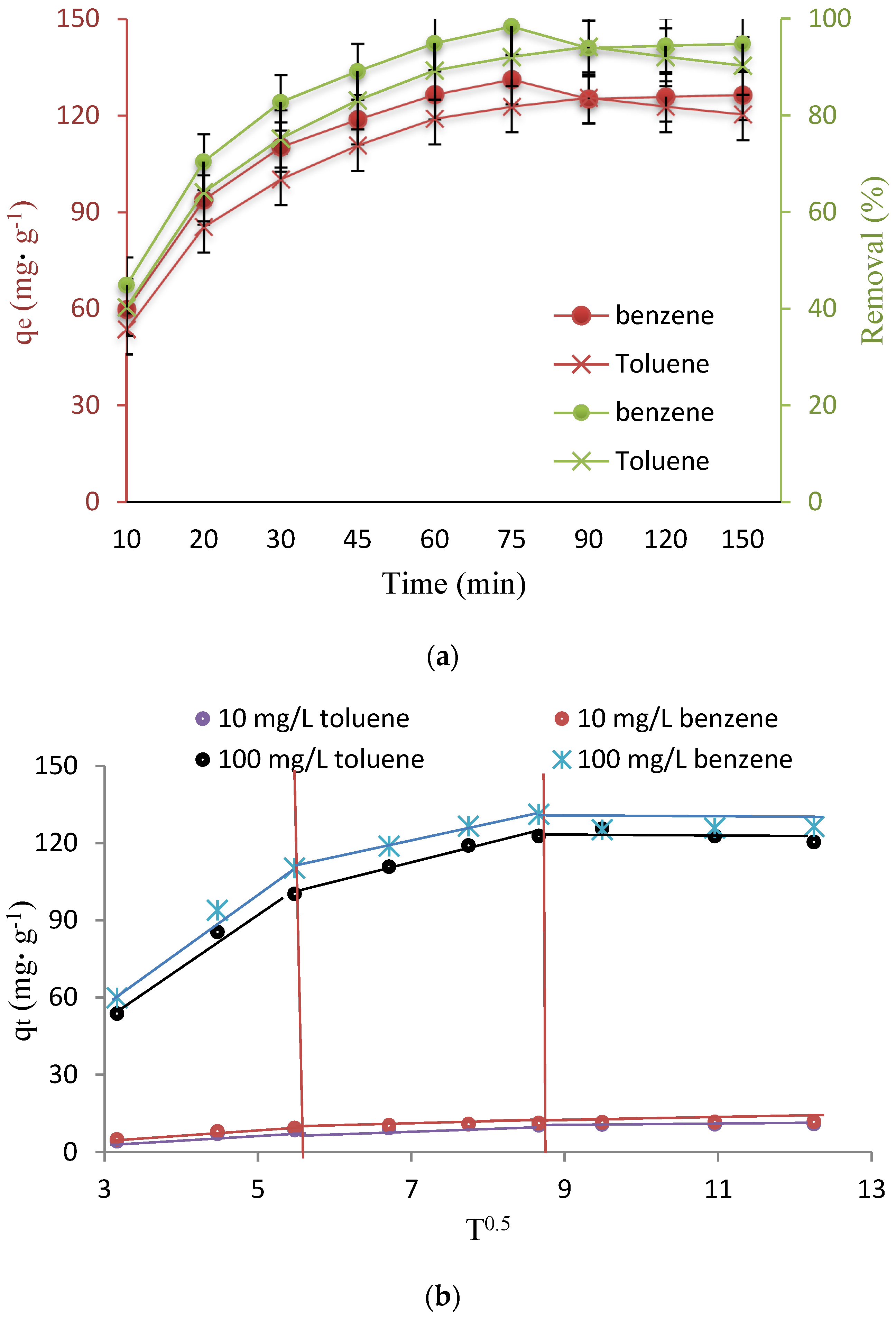
3.3. Investigation of Contact Time and Kinetic Studies
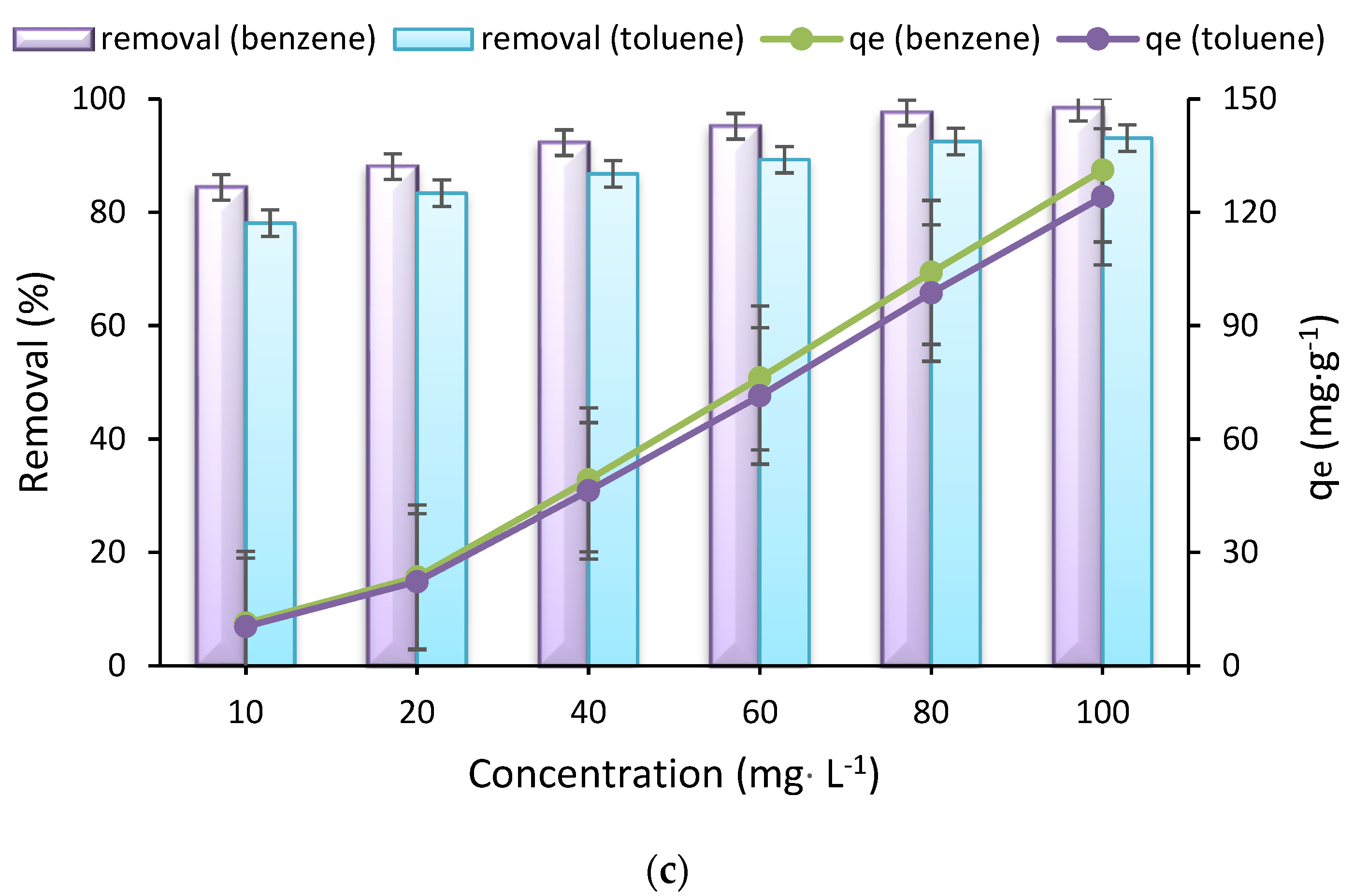
3.4. Isotherm Studies
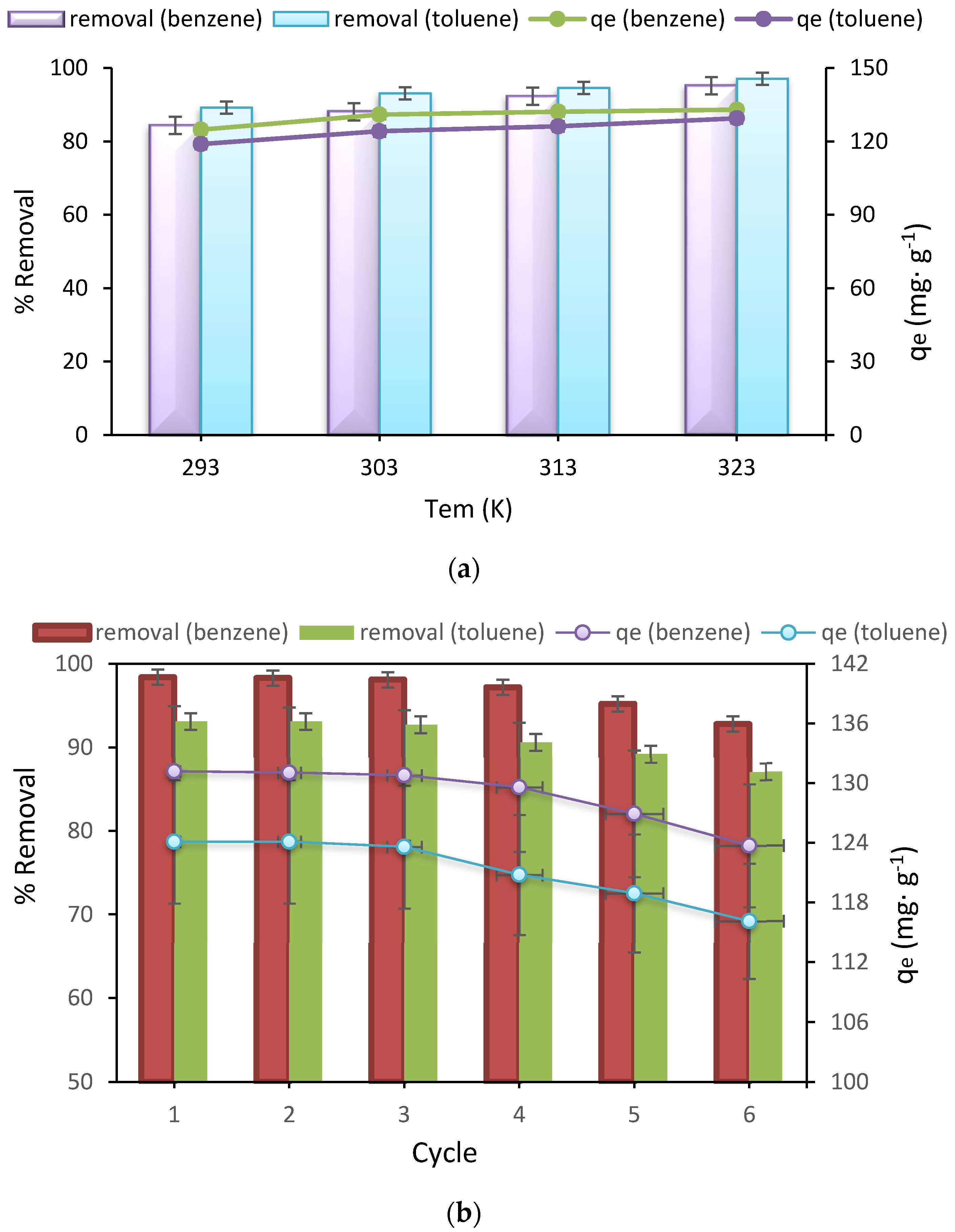
3.5. Effect of Temperature and Thermodynamics Study
3.6. Adsorbent Recyclability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| C0 = concentrations of benzene and toluene in mg L−1 at the start |
| Ce = concentrations of benzene and toluene in mg L−1 at equilibrium |
| V = benzene and toluene volume at mL |
| m = Fe3O4@ZIF-8 masses in mg |
| KL = (L mg−1) is the Langmuir adsorption constant |
| KF = Freundlich constant (L g−1) |
| n = heterogeneity factor (If the value of n < 1, the adsorption is a chemical process; if n > 1, then it is a physical process). |
| qe = adsorption capacity at equilibrium |
| qm = maximum adsorption capacity |
| RL = dimensionless separation factor |
| The values of RL show whether the biosorption process is irreversible (RL = 0), linear (RL = 1), favorable (0 < RL < 1) or undesirable (RL > 1) |
| ΔG° = Gibbs free energy (kJ mol−1) |
| ΔS° = entropy (kJ mol−1 K−1) |
| ΔH° = enthalpy (kJ mol−1) |
| K1 = equilibrium rate constant for pseudo-first order kinetics (PFO) (1.min−1) |
| K2 = equilibrium rate constant for pseudo-second order kinetics (PSO) (g mg−1 minmg) |
| Kb =Intra-Particle Diffusion (IPD) rate constant (mg g−1 min−1/2) |
| C= thickness of the boundary layer |
| D-R isotherm constant (mol2 kJ−2) |
| Polanyi potential |
| E = The adsorption energy (kJ.mol−1) |
| KT = constant of equilibrium binding (L.mg−1) |
| B = Temkin constant associated with the heat of adsorption (J/mol) |
| D-R = Dubinin-Radushkevich |
| XRD = X-ray Powder Diffraction |
| FTIR= Fourier Transform Infrared Spectrometer |
| SEM = Scanning electron microscope |
| TEM = Transmission Electron Microscope |
| VSM = Vibrating Sample Magnetometer |
| BET = Brunauer–Emmett–Teller |
| MOF = Metal-organic framework |
References
- Lakshman, M. Fe3O4@SiO2-Pip-SA nanocomposite: A novel and highly efficient reusable acidic catalyst for synthesis of rhodanine derivatives. J. Syn. Chem. 2022, 1, 48–51. [Google Scholar] [CrossRef]
- Sadeghi, M.; Mehdinejad, M.H.; Mengelizadeh, N.; Mahdavi, Y.; Pourzamani, H.; Hajizadeh, Y. Degradation of diclofenac by heterogeneous electro-Fenton process using magnetic single-walled carbon nanotubes as a catalyst. J. Water. Process. Eng. 2019, 31, 100852. [Google Scholar] [CrossRef]
- Ukhurebor, K.E.; Athar, H.; Adetunji, C.O.; Aigbe, U.O.; Onyancha, R.B.; Abifarin, O. Environmental implications of petroleum spillages in the Niger Delta region of Nigeria: A review. J. Environ. Manag. 2021, 293, 112872. [Google Scholar] [CrossRef]
- Sui, X.; Wang, X.; Li, Y.; Ji, H. Remediation of petroleum-contaminated soils with microbial and microbial combined methods: Advances, mechanisms, and challenges. Sustainability 2021, 13, 9267. [Google Scholar] [CrossRef]
- Kuranchie, F.A.; Angnunavuri, P.N.; Attiogbe, F.; Nerquaye-Tetteh, E.N. Occupational exposure of benzene, toluene, ethylbenzene and xylene (BTEX) to pump attendants in Ghana: Implications for policy guidance. Cogent Environ. Sci. 2019, 5, 1603418. [Google Scholar] [CrossRef]
- Logeshwaran, P.; Megharaj, M.; Chadalavada, S.; Bowman, M.; Naidu, R. Petroleum hydrocarbons (PH) in groundwater aquifers: An overview of environmental fate, toxicity, microbial degradation and risk-based remediation approaches. Environ. Technol. Innov. 2018, 10, 175–193. [Google Scholar] [CrossRef]
- Wibowo, N.; Setyadhi, L.; Wibowo, D.; Setiawan, J.; Ismadji, S. Adsorption of benzene and toluene from aqueous solutions onto activated carbon and its acid and heat treated forms: Influence of surface chemistry on adsorption. J. Hazard. Mater. 2007, 146, 237–242. [Google Scholar] [CrossRef]
- Loomis, D.; Guyton, K.Z.; Grosse, Y.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L. Carcinogenicity of benzene. Lancet Oncol. 2017, 18, 1574–1575. [Google Scholar] [CrossRef]
- Warden, H.; Richardson, H.; Richardson, L.; Siemiatycki, J.; Ho, V. Associations between occupational exposure to benzene, toluene and xylene and risk of lung cancer in Montréal. Occup. Environ. Med. 2018, 75, 696–702. [Google Scholar] [CrossRef]
- Bina, B.; Amin, M.M.; Rashidi, A.; Pourzamani, H. Benzene and toluene removal by carbon nanotubes from aqueous solution. Arch. Environ. Prot. 2012, 14, 22–31. [Google Scholar] [CrossRef]
- Flanagan, P.; Kelleher, B.; Allen, C. Assessment of anaerobic biodegradation of aromatic hydrocarbons: The impact of molecular biology approaches. Geomicrobiol. J. 2014, 31, 276–284. [Google Scholar] [CrossRef]
- Paixão, M.M.; Vianna, M.T.G.; Marques, M. Graphene and graphene nanocomposites for the removal of aromatic organic compounds from the water: Systematic review. Mater. Res. Exp. 2018, 5, 012002. [Google Scholar] [CrossRef]
- Jindrova, E.; Chocova, M.; Demnerova, K.; Brenner, V. Bacterial aerobic degradation of benzene, toluene, ethylbenzene and xylene. Folia Microbiol. 2002, 47, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.; Mat, H.; Yunus, M.A.C. (Eds.) Batch Adsorptive Removal of BTEX From Aqueous Solution: A Review. In Proceedings of the Third International Conference on Separation Technology 2020 (ICoST 2020), Senai, Kualalumpur, Malaysia, 15–16 August 2020; Atlantis Press: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Moura, C.P.; Vidal, C.B.; Barros, A.L.; Costa, L.S.; Vasconcellos, L.C.; Dias, F.S. Adsorption of BTX (benzene, toluene, o-xylene, and p-xylene) from aqueous solutions by modified periodic mesoporous organosilica. J. Colloid. Interface Sci. 2011, 363, 626–634. [Google Scholar] [CrossRef]
- Lima, E.C. Removal of emerging contaminants from the environment by adsorption. Ecotoxicol. Environ. Saf. 2018, 150, 1–17. [Google Scholar]
- Rashed, M.N. Adsorption technique for the removal of organic pollutants from water and wastewater. Org. Pollut. Monit. Risk. Treat. 2013, 7, 167–194. [Google Scholar]
- Largitte, L.; Pasquier, R. A review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem. Eng. Res. Des. 2016, 109, 495–504. [Google Scholar] [CrossRef]
- Yu, J.-G.; Yu, L.-Y.; Yang, H.; Liu, Q.; Chen, X.-H.; Jiang, X.-Y. Graphene nanosheets as novel adsorbents in adsorption, preconcentration and removal of gases, organic compounds and metal ions. Sci. Total Environ. 2015, 502, 70–79. [Google Scholar] [CrossRef]
- Bilal, M.; Ihsanullah, I.; Younas, M.; Shah, M.U.H. Recent advances in applications of low-cost adsorbents for the removal of heavy metals from water: A critical review. Sep. Purif. Technol. 2021, 278, 119510. [Google Scholar] [CrossRef]
- Pyrzynska, K. Removal of cadmium from wastewaters with low-cost adsorbents. J. Environ. Chem. Eng. 2019, 7, 102795. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, X.; Li, T.; Zhang, Y.; Xu, H.; Sun, Y. MIL series of metal organic frameworks (MOFs) as novel adsorbents for heavy metals in water: A review. J. Hazard. Mater. 2022, 429, 128271. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, L.; Bazrafshan, E.; Noroozifar, M.; Ansari-Moghaddam, A.; Barahuie, F. Adsorptive removal of benzene and toluene from aqueous environments by cupric oxide nanoparticles: Kinetics and isotherm studies. J. Chem. 2017, 69, 113434. [Google Scholar] [CrossRef] [Green Version]
- Jjagwe, J.; Olupot, P.W.; Menya, E.; Kalibbala, H.M. Synthesis and Application of Granular Activated Carbon from Biomass Waste Materials for Water Treatment: A Review. J. Bioresour. Bioprod. 2021, 6, 292–322. [Google Scholar] [CrossRef]
- Chen, S.; Jiang, S.; Jiang, H. A review on conversion of crayfish-shell derivatives to functional materials and their environmental applications. J. Bioresour. Bioprod. 2020, 5, 238–247. [Google Scholar] [CrossRef]
- Mahmoud, A.S.; Mostafa, M.K.; Abdel-Gawad, S.A. Artificial intelligence for the removal of benzene, toluene, ethyl benzene and xylene (BTEX) from aqueous solutions using iron nanoparticles. Water Supply 2018, 18, 1650–1663. [Google Scholar] [CrossRef]
- Cimbaluk, G.V.; Ramsdorf, W.A.; Perussolo, M.C.; Santos, H.K.F.; De Assis, H.C.D.S.; Schnitzler, M.C. Evaluation of multiwalled carbon nanotubes toxicity in two fish species. Ecotoxicol. Environ. Saf. 2018, 150, 215–223. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Liu, Y.; Ramesh, M.; Deepa, C. Metal-organic frameworks and their composites. In Metal-Organic Frameworks for Chemical Reactions; Elsevier: Amsterdam, The Netherlands, 2021; pp. 1–18. ISBN 9780128220993. [Google Scholar]
- Gurusamy, L.; Anandan, S.; Wu, J. Nanomaterials derived from metal-organic frameworks for energy storage supercapacitor application. Met.-Org. Framew. Chem. React. 2020, 11, 441–470. [Google Scholar] [CrossRef]
- Jian, M.; Liu, B.; Zhang, G.; Liu, R.; Zhang, X. Adsorptive removal of arsenic from aqueous solution by zeolitic imidazolate framework-8 (ZIF-8) nanoparticles. Colloids Surf. A 2015, 465, 67–76. [Google Scholar] [CrossRef]
- Lee, Y.R.; Do, X.H.; Cho, K.Y.; Jeong, K.; Baek, K.Y. Amine-functionalized zeolitic imidazolate framework-8 (ZIF-8) nanocrystals for adsorption of radioactive iodine. ACS Appl. Nano Mater. 2020, 3, 9852–9861. [Google Scholar] [CrossRef]
- Hajializadeh, A.; Ansari, M.; Foroughi, M.; Jahani, S.; Kazemipour, M. Zeolite Imidazolate Framework Nanocrystals Electrodeposited on Stainless Steel Fiber for Determination of Polycyclic Aromatic Hydrocarbons. Iran. J. Chem. Chem. Eng. 2022, 41, 368–379. [Google Scholar] [CrossRef]
- Jiang, X.; Su, S.; Rao, J.; Li, S.; Lei, T.; Bai, H. Magnetic metal-organic framework (Fe3O4@ ZIF-8) core-shell composite for the efficient removal of Pb (II) and Cu (II) from water. J. Environ. Chem. Eng. 2021, 9, 105959. [Google Scholar] [CrossRef]
- Wu, Y.; Li, B.; Wang, X.; Yu, S.; Pang, H.; Liu, Y. Magnetic metal-organic frameworks (Fe3O4@ ZIF-8) composites for U (VI) and Eu (III) elimination: Simultaneously achieve favorable stability and functionality. Chem. Eng. J. 2019, 378, 122105. [Google Scholar] [CrossRef]
- El-Desouky, M.; El-Bindary, A. Magnetic metal-organic framework (Fe3O4@ZIF-8) nanocomposites for adsorption of anionic dyes from wastewater. Inorg. Nano-Met. Chem. 2021, 51, 1–15. [Google Scholar] [CrossRef]
- Qu, Y.; Qin, L.; Liu, X.; Yang, Y. Magnetic Fe3O4/ZIF-8 composite as an effective and recyclable adsorbent for phenol adsorption from wastewater. Sep. Purif. Technol. 2022, 294, 121169. [Google Scholar] [CrossRef]
- Ma, X.; Zhao, S.; Tian, Z.; Duan, G.; Pan, H.; Yue, Y.; Li, S.; Jian, S.; Yang, W.; Liu, K.; et al. MOFs meet wood: Reusable magnetic hydrophilic composites toward efficient water treatment with super-high dye adsorption capacity at high dye concentration. Chem. Eng. J. 2022, 446, 136851. [Google Scholar] [CrossRef]
- Balarak, D.; Taheri, Z.; Shim, M.J.; Lee, S.M.; Jeon, C. Adsorption kinetics and thermodynamics and equilibrium of ibuprofen from aqueous solutions by activated carbon prepared from Lemna minor. Desal Water Treat. 2021, 215, 183–193. [Google Scholar] [CrossRef]
- Al-Musawi, T.J.; Mengelizadeh, N.; Al Rawi, O. Capacity and Modeling of Acid Blue 113 Dye Adsorption onto Chitosan Magnetized by Fe2O3 Nanoparticles. J. Polym. Environ. 2022, 30, 344–359. [Google Scholar] [CrossRef]
- Kaya, A.; Şahin, S.A. Acid Orange 7 adsorption onto quaternized pistachio shell powder from aqueous solutions. Biomass Conv. Bioref. 2022, 12, 1–18. [Google Scholar] [CrossRef]
- Almeida, E.J.R.; Corso, C.R. Decolorization and removal of toxicity of textile azo dyes using fungal biomass pelletized. Int. J. Environ. Sci. Technol. 2019, 16, 1319–1328. [Google Scholar] [CrossRef]
- Ashouri, R.; Shirkhanloo, H.; Rashidi, A.M. Dynamic and static removal of benzene from air based on task-specific ionic liquid coated on MWCNTs by sorbent tube-headspace solid-phase extraction procedure. Int. J. Environ. Sci. Technol. 2021, 18, 2377–2390. [Google Scholar] [CrossRef]
- Wong, G.K.S.; Lim, L.Z.; Lim, M.J.W.; Ong, L.L.; Khezri, B.; Pumera, M.; Webster, R.D. Evaluation of the sorbent properties of single and multiwalled carbon nanotubes for volatile organic compounds through thermal desorption gaschromatography/mass spectrometry. Chem. Plus Chem. 2015, 80, 1279–1287. [Google Scholar]
- Wang, X.; Ma, C.; Xiao, J.; Xia, Q.; Wu, J.; Li, L. Benzene/toluene/water vapor adsorption and selectivity of novel C-PDA adsorbents with high uptakes of benzene and toluene. Chem. Eng. J. 2018, 335, 970–978. [Google Scholar] [CrossRef]
- Konicki, W.; Aleksandrzak, M.; Moszyn, D.; Mijowska, E. Adsorption of anionic azo-dyes from aqueous solution onto graphene oxide: Equilibrium, kinetic and thermodynamic studies. J. Colloid. Interface Sci. 2017, 496, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Balarak, D.; Zafariyan, M.; Igwegbe, C.A. Adsorption of Acid Blue 92 Dye from Aqueous Solutions by Single-Walled Carbon Nanotubes: Isothermal, Kinetic, and Thermodynamic Studies. Environ. Process. 2021, 8, 869–888. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, G.; An, C.; Xin, X.; Liu, X.; Raman, M.; Yao, Y.; Wang, W.; Doble, M. Transport of anionic azo dyes from aqueous solution to Gemini surfactant-modified wheat bran: Synchrotron infrared, molecular interaction and adsorption studies. Sci. Total Environ. 2017, 595, 723–732. [Google Scholar] [CrossRef]
- Vahdat, P.Z.; Asilian, H.; Jonidi Jafari, A. Adsorption of xylene from air by natural Iranian zeolite. Health Scope 2014, 3, 17528. [Google Scholar] [CrossRef]
- Balarak, D.; Mostafapour, F.K. Adsorption of acid red 66 dye from aqueous solution by heat-treated rice husk. Res. J. Chem. Environ. 2018, 22, 80–84. [Google Scholar]
- Teimoori, S.; Hassani, A.H.; Panahi, M.; Mansouri, N. Review: Methods for removal and adsorption of volatile organic compounds from environmental matrixes. Anal. Methods Environ. Chem J. 2020, 3, 34–58. [Google Scholar] [CrossRef]
- Takahashi, A.; Yang, F.H.; Yang, R.T. new sorbents for desulfurization by π-complexation: Thiophene/benzene adsorption. Ind Eng. Chem. Res. 2020, 41, 2487–2496. [Google Scholar] [CrossRef]
- Seifi, L.; Torabian, A.; Kazemian, H.; Bidhendi, G.N.; Azimi, A.; Nazmara, S.; Mohammadi, M. Adsorption of BTEX on surfactant modified granulated natural zeolite nano particles, parameters optimizing by applying taguchi experimental design method. Clean Soil Air Water. 2011, 39, 939–948. [Google Scholar] [CrossRef]
- Wang, D.; Repo, E.; He, F.; Zhang, X. Dual functional sites strategies toward enhanced heavy metal remediation: Interlayer expanded Mg-Al layered double hydroxide by intercalation with L-cysteine. J. Hazard. Mater. 2022, 439, 129693. [Google Scholar] [CrossRef]
- Saner, A.; Carvalho, P.N.; Catalano, J.; Anastasakis, K. Renewable adsorbents from the solid residue of sewage sludge hydrothermal liquefaction for wastewater treatment. Sci. Total Environ. 2022, 838, 156418. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Chen, S.; Xiang, H.; Gao, T. Selectively capacitive recovery of rare earth elements from aqueous solution onto Lewis base sites of pyrrolic-N doped activated carbon electrodes. Carbon 2022, 197, 282–291. [Google Scholar] [CrossRef]
- Chan, K.; Zinchenko, A. Conversion of waste bottle PET to magnetic microparticles adsorbent for dye-simulated wastewater treatment. J. Environ. Chem. Eng. 2022, 10, 108055. [Google Scholar] [CrossRef]
- Yilmaz, M.; Al-Musawi, T.J.; Saloot, M.K. Synthesis of activated carbon from Lemna minor plant and magnetized with iron (III) oxide magnetic nanoparticles and its application in removal of Ciprofloxacin. Biomass Conv. Bioref. 2022, 12, 1–14. [Google Scholar] [CrossRef]
- Dyanati-Tilaki, R.A.; Yousefi, Z.; Yazdani-Cherati, J. The ability of azollaand lemna minor biomass for adsorption of phenol from aqueous solutions. J. Mazand. Univ. Med. Sci. 2013, 23, 140–146. [Google Scholar]
- Al-Musawi, T.J.; Mahvi, A.H.; Khatibi, A.D. Effective adsorption of ciprofloxacin antibiotic using powdered activated carbon magnetized by iron(III) oxide magnetic nanoparticles. J. Porous. Mater. 2021, 28, 835–852. [Google Scholar] [CrossRef]
- Abdul-Hameed, H.M. A coated of Ca/Fe layered hydroxide onto a synthesized adsorbent from (banana peels) for removal of cadmium from simulated wastewater. Caspian. J. Environ. Sci. 2021, 19, 825–827. [Google Scholar] [CrossRef]
- Kundu, R.; Biswas, C.; Ahmed, J.; Naime, J.; Ara, M. A Study on the Adsorption of Cadmium(II) from Aqueous Solution onto Activated Carbon Originated from Bombax ceiba Fruit Shell. J. Chem. Health. Risks 2020, 10, 243–252. [Google Scholar] [CrossRef]
- Davarnejad, R.; Azizi, A.; Asadi, S.; Mohammadi, M. Green Synthesis of Copper Nanoparticles using Centaurea cyanus Plant Extract: A Cationic Dye Adsorption Application. Iran. J. Chem. Chem. Eng. 2022, 41, 1–14. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Y.; Zheng, C.; Lin, H.; Xu, R.; Zhu, H.; Xu, X. Lanthanum carbonate grafted ZSM-5 for superior phosphate uptake: Investigation of the growth and adsorption mechanism. Chem. Eng. J. 2022, 430, 133166. [Google Scholar] [CrossRef]
- Yang, Y.; Zhu, H.; Xu, X.; Bao, L.; Wang, Y.; Lin, H. Zheng, C. Construction of a novel lanthanum carbonate-grafted ZSM-5 zeolite for effective highly selective phosphate removal from wastewater. Microporous Mesoporous Mater. 2021, 324, 111289. [Google Scholar] [CrossRef]
- Fadaei, S.; Noorisepehr, M.; Pourzamani, H.; Salari, M.; Moradnia, M.; Darvishmotevalli, M. Heterogeneous activation of peroxymonosulfate with Fe3O4 magnetic nanoparticles for degradation of Reactive Black 5: Batch and column study. J. Environ. Chem. Eng. 2021, 9, 4–10. [Google Scholar] [CrossRef]
- Papadopoulos, A.N.; Bikiaris, D.N.; Mitropoulos, A.C.; Kyzas, G.Z. Nanomaterials and chemical modifications for enhanced key wood properties: A review. Nanomaterials 2019, 9, 607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kyzas, G.Z.; Matis, K.A. Flotation in water and wastewater treatment. Processes 2018, 6, 116. [Google Scholar] [CrossRef]
- Trikkaliotis, D.G.; Christoforidis, A.K.; Mitropoulos, A.C.; Kyzas, G.Z. Adsorption of copper ions onto chitosan/poly(vinyl alcohol) beads functionalized with poly(ethylene glycol). Carbohydr. Polym. 2020, 234, 115890. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Nanaki, S.G.; Koltsakidou, A.; Papageorgiou, M.; Kechagia, M.; Bikiaris, D.N.; Lambropoulou, D.A. Effectively designed molecularly imprinted polymers for selective isolation of the antidiabetic drug metformin and its transformation product guanylurea from aqueous media. Anal. Chim. Acta 2015, 866, 27–40. [Google Scholar] [CrossRef]
| Models | Benzene (mg L−1) | Toluene (mg L−1) | Models | Benzene (mg L−1) | Toluene (mg L−1) | ||||
|---|---|---|---|---|---|---|---|---|---|
| 10 | 100 | 10 | 100 | 10 | 100 | 10 | 100 | ||
| PFO | Elovich | ||||||||
| qe (calf) K1 R2 | 4.2 0.027 0.895 | 83.1 0.091 0.871 | 3.9 0.019 0.925 | 77.2 0.044 0.897 | α × 106 b R2 | 0.041 7.24 0.921 | 0.73 19.6 0.898 | 0.025 5.32 0.903 | 0.57 16.2 0.987 |
| PSO | IPD | ||||||||
| qe (cal) K2 R2 | 13.8 0.002 0.998 | 126.4 0.003 0.996 | 11.2 0.001 0.994 | 114.4 0.002 0.992 | Kb C R2 | 2.13 24.2 0.876 | 6.16 65.8 0.658 | 1.94 17.4 0.805 | 6.68 55.5 0.714 |
| Benzene | Toluene | |||||||
|---|---|---|---|---|---|---|---|---|
| models | 293 K | 303 K | 313 K | 323 K | 293 K | 303 K | 313 K | 323 K |
| Qe exp (mg g−1) | 124.8 | 130.9 | 132.1 | 132.9 | 118.9 | 124.1 | 126.2 | 129.4 |
| Langmuir | ||||||||
| qm (mg g−1) KL (L mg−1) RL R2 | 129.4 0.0024 0.806 0.989 | 134.2 0.0028 0.781 0.993 | 137.3 0.0036 0.735 0.996 | 148.2 0.0041 0.709 0.997 | 118.4 0.0018 0.847 0.989 | 125.2 0.0021 0.826 0.989 | 129.6 0.0023 0.813 0.989 | 133.1 0.0026 0.793 0.989 |
| Freundlich | log Ce + log KF | |||||||
| KF | 7.02 0.389 0.862 | 6.51 0.354 0.891 | 7.23 0.342 0.873 | 7.89 0.425 0.823 | 10.1 0.342 0.834 | 12.3 0.453 0.786 | 9.82 0.511 0.821 | 8.41 0.562 0.792 |
| 1/n | ||||||||
| R2 | ||||||||
| Temkin | ||||||||
| KT (L g−1) Β (J mol−1) R2 | 2.16 12.1 0.841 | 1.76 17.4 0.834 | 1.73 24.8 0.865 | 1.22 38.3 0.871 | 1.84 8.22 0.812 | 1.32 13.4 0.832 | 1.05 19.1 0.846 | 0.924 19.9 0.802 |
| D-R | ε | |||||||
| qm (mg g−1) E R2 | 82.2 7.64 0.956 | 89.2 8.02 0.987 | 92.4 8.49 0.971 | 98.3 8.95 0.985 | 72.4 7.14 0.947 | 78.8 7.66 0.948 | 84.1 8.91 0.931 | 89.4 9.27 0.912 |
| Adsorbent | Pollutant | Reference | |
|---|---|---|---|
| Iron nanoparticles | benzene | 41.2 | [26] |
| Iron nanoparticles | toluene | 27.6 | [26] |
| CuO nanoparticles | benzene | 36.9 | [23] |
| CuO nanoparticles | toluene | 40.2 | [23] |
| Activated carbon | benzene | 51.2 | [7] |
| Activated carbon | toluene | 58.5 | [7] |
| MWCNT | benzene | 56.9 | [10] |
| MWCNT | toluene | 63.2 | [10] |
| periodic organosilica | benzene | 65.1 | [15] |
| periodic organosilica | toluene | 97. 6 | [15] |
| Fe3O4@ZIF-8 Fe3O4@ZIF-8 | benzene toluene | 148.2 133.1 | This study This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kyzas, G.Z.; McKay, G.; Al-Musawi, T.J.; Salehi, S.; Balarak, D. Removal of Benzene and Toluene from Synthetic Wastewater by Adsorption onto Magnetic Zeolitic Imidazole Framework Nanocomposites. Nanomaterials 2022, 12, 3049. https://doi.org/10.3390/nano12173049
Kyzas GZ, McKay G, Al-Musawi TJ, Salehi S, Balarak D. Removal of Benzene and Toluene from Synthetic Wastewater by Adsorption onto Magnetic Zeolitic Imidazole Framework Nanocomposites. Nanomaterials. 2022; 12(17):3049. https://doi.org/10.3390/nano12173049
Chicago/Turabian StyleKyzas, George Z., Gordon McKay, Tariq J. Al-Musawi, Sabereh Salehi, and Davoud Balarak. 2022. "Removal of Benzene and Toluene from Synthetic Wastewater by Adsorption onto Magnetic Zeolitic Imidazole Framework Nanocomposites" Nanomaterials 12, no. 17: 3049. https://doi.org/10.3390/nano12173049
APA StyleKyzas, G. Z., McKay, G., Al-Musawi, T. J., Salehi, S., & Balarak, D. (2022). Removal of Benzene and Toluene from Synthetic Wastewater by Adsorption onto Magnetic Zeolitic Imidazole Framework Nanocomposites. Nanomaterials, 12(17), 3049. https://doi.org/10.3390/nano12173049








