The Effect of Co Content and Annealing Temperatures on the Resistivity in Ag-Co Films
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
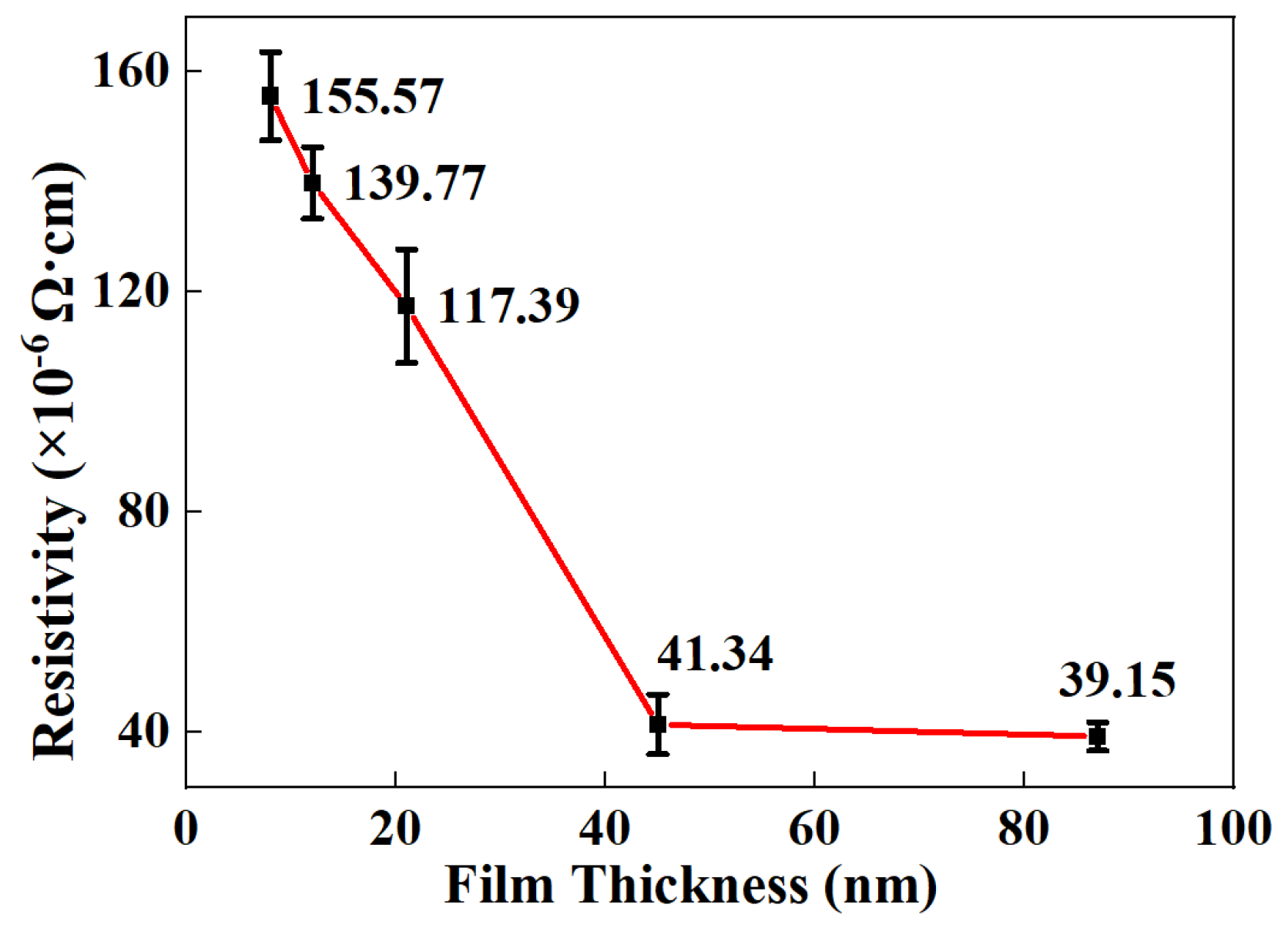
3.1. Resistivity of Ag-Co Films with Different Thicknesses
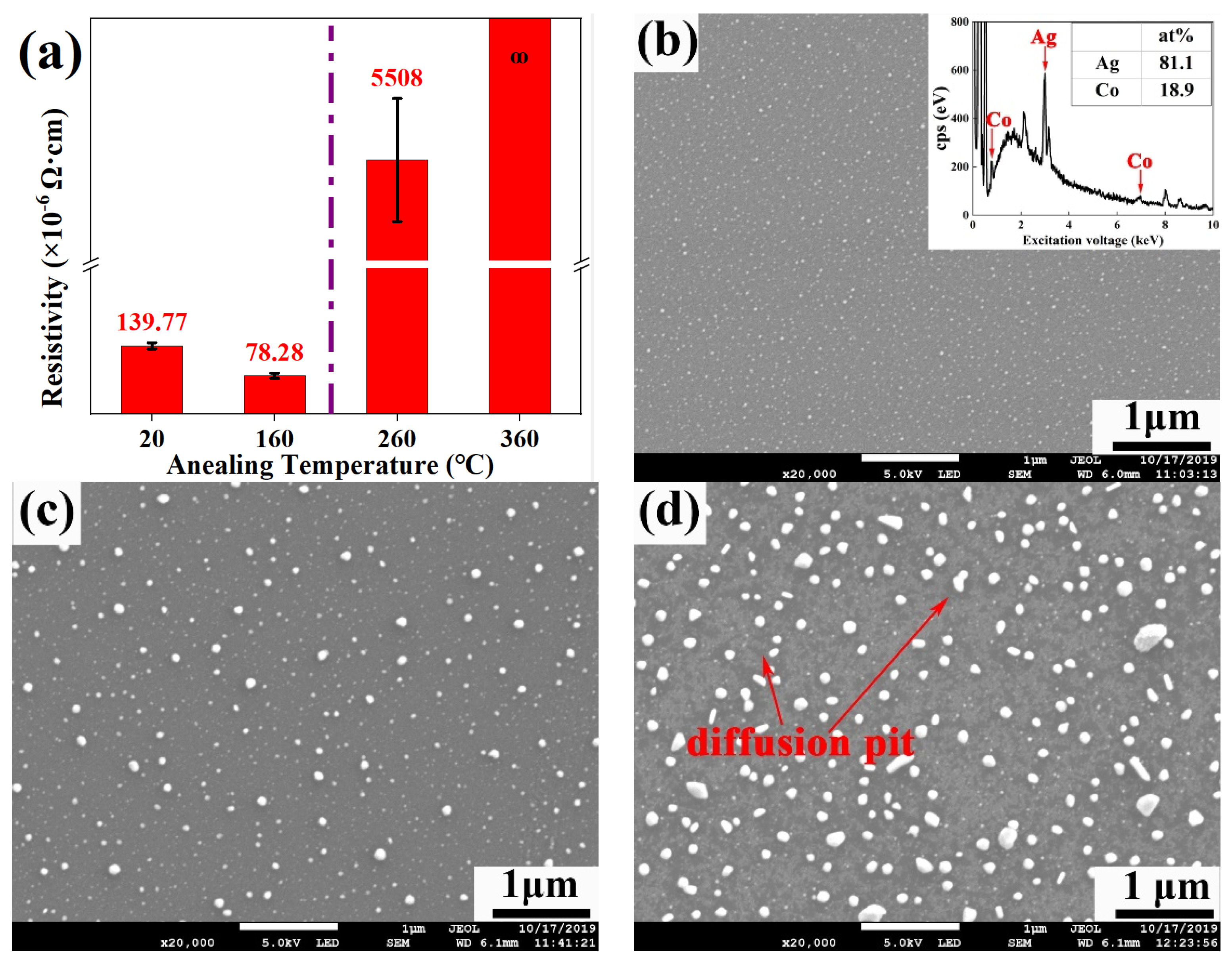
3.2. Resistivity of Ag-Co Films Annealed at Different Temperatures
3.3. The Analysis of Ultra-High Resistivity of Ag-Co Films
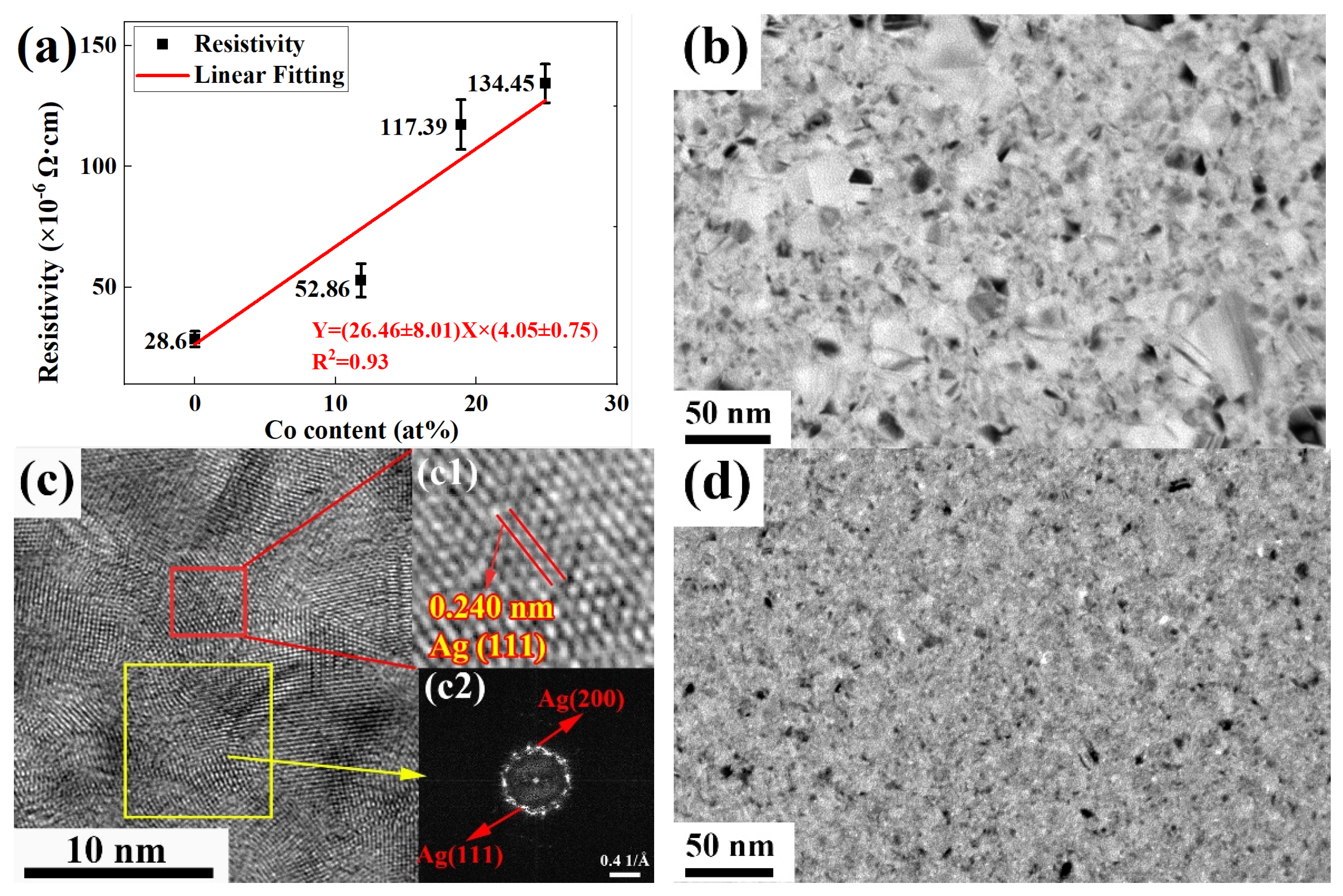
3.4. Resistivity of Annealed Ag-Co Films with Different Thicknesses
3.5. Resistivity of Ag-Co Films with Different Co Contents
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, W.; Chen, T.P.; Liu, Y.; Fung, S. Conduction Mechanisms at Low- and High-Resistance States in Aluminum/Anodic Aluminum Oxide/Aluminum Thin Film Structure. J. Appl. Phys. 2012, 112, 063706. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Bai, T.; Liu, Z. Thin Film Chip Resistors with High Resistance and Low Temperature Coefficient of Resistance. Trans. Tianjin Univ. 2010, 16, 348–353. [Google Scholar] [CrossRef]
- Abeles, B.; Pinch, H.L.; Gittleman, J.I. Percolation Conductivity in W-Al2O3Granular Metal Films. Phys. Rev. Lett. 1975, 35, 247–250. [Google Scholar] [CrossRef]
- Han, S.Z.; Lim, S.H.; Kim, S.; Lee, J.; Goto, M.; Kim, H.G.; Han, B.; Kim, K.H. Increasing Strength and Conductivity of Cu Alloy Through Abnormal Plastic Deformation of an Intermetallic Compound. Sci. Rep. 2016, 6, 30907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Sun, L.X.; Tao, N.R. Nanostructures and Nanoprecipitates Induce High Strength and High Electrical Conductivity in a CuCrZr Alloy. J. Mater. Sci. Technol. 2020, 48, 18–22. [Google Scholar] [CrossRef]
- Hayashi, T.; Suzuki, T.; Ema, Y. A Very-High-Conductivity of In-Doped CdTe Film. J. Appl. Phys. 1988, 27, 1626–1629. [Google Scholar] [CrossRef]
- Sakai, Y.; Schneider-Muntau, H.J. Ultra-High Strength, High Conductivity Cu-Ag Alloy Wires. Acta Mater. 1997, 45, 1017–1023. [Google Scholar] [CrossRef]
- Lu, K.; Yao, R.; Hu, S.; Liu, X.; Wei, J.; Wu, W.; Ning, H.; Xu, M.; Lan, L.; Peng, J. High-Performance and Flexible Neodymium-Doped Oxide Semiconductor Thin-Film Transistors with Copper Alloy Bottom-Gate Electrode. IEEE Electron Device Lett. 2018, 39, 839–842. [Google Scholar] [CrossRef]
- Wang, X.Y.; Zhang, Z.S.; Bai, T. Investigation on Powder Metallurgy Cr–Si–Ta–Al Alloy target for High-Resistance Thin Film Resistors with Low Temperature Coefficient of Resistance. Mater. Des. 2010, 31, 1302–1307. [Google Scholar] [CrossRef]
- Yang, Y.C.; Tsau, C.H.; Yeh, J.W. TiFeCoNi Oxide Thin Film—A New Composition with Extremely Low Electrical Resistivity at Room Temperature. Scr. Mater. 2011, 64, 173–176. [Google Scholar] [CrossRef]
- Tsau, C.H.; Yang, Y.C.; Lee, C.C.; Wu, L.Y.; Huang, H.J. The Low Electrical Resistivity of the High-entropy Alloy Oxide Thin Films. Procedia Eng. 2012, 36, 246–252. [Google Scholar] [CrossRef]
- Klissurski, D.G. On the Approximate Correlation between the Bonding Energy of Oxygen in Metal Oxides and the Width of Their Forbidden Zones. Solid State Commun. 1974, 15, 1789–1792. [Google Scholar] [CrossRef]
- Fischer, D.W. Vanadium LII, IIIX-Ray Emission and Absorption Spectra from Metal, Oxides, Nitride, Carbide, and Boride. J. Appl. Phys. 1969, 40, 4151–4163. [Google Scholar] [CrossRef]
- Zhang, Q.-F.; Wang, L.-S.; Wang, X.-Z.; Zheng, H.-F.; Liu, X.; Xie, J.; Qiu, Y.-L.; Chen, Y.; Peng, D.-L. Electrical Transport Properties in Co Nanocluster-Assembled Granular Film. J. Appl. Phys. 2017, 121, 103901. [Google Scholar] [CrossRef]
- Bai, G.; Wu, C.; Jin, J.; Yan, M. Structural, Electron Transportation and Magnetic Behavior Transition of Metastable FeAlO Granular Films. Sci. Rep. 2016, 6, 24410. [Google Scholar] [CrossRef] [PubMed]
- Delimova, L.A.; Gushchina, E.V.; Zaitseva, N.V.; Seregin, D.S.; Vorotilov, K.A.; Sigov, A.S. Effect of the Crystal Structure on the Electrical Properties of Thin-Film PZT Structures. Phys. Solid State 2018, 60, 553–558. [Google Scholar] [CrossRef]
- Boff, M.; Canto, B.; Hinrichs, R.; Pereira, L.; Mesquita, F.; Schmidt, J.; Fraga, G. Electrical Current Influence on Resistance and Localization Length of a Co–Al2O3 Granular Thin Film. Phys. B Condens. Matter 2011, 406, 4304–4306. [Google Scholar] [CrossRef][Green Version]
- Sun, H.; Song, Z.; Ma, F.; Zhan, J.; Xu, K. Microstructure, Formation Mechanism and Compression Plasticity of Regularly Faceted Cu Particles. Scr. Mater. 2009, 60, 305–308. [Google Scholar] [CrossRef]
- Sachan, R.; Yadavali, S.; Shirato, N.; Krishna, H.; Ramos, V.; Duscher, G.; Pennycook, S.J.; Gangopadhyay, A.K.; Garcia, H.; Kalyanaraman, R. Self-organized bimetallic Ag-Co nanoparticles with tunable localized surface plasmons showing high environmental stability and sensitivity. Nanotechnology 2012, 23, 275604. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, J.C.; Liu, C.J.; Ju, Y.H. Response Characteristics of Lead Phthalocyanine Gas Sensor: Effects of Film Thickness and Crystal Morphology. Thin Solid Film. 1998, 322, 98–103. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nozaki, K.; Yamaguchi, A.; Urakami, N. Molecular Simulation of Crystallization in N-Alkane Ultrathin Films: Effects of Film Thickness and Substrate Attraction. J. Chem. Phys. 2007, 127, 154704. [Google Scholar] [CrossRef] [PubMed]
- Tenailleau, C.; Aharon, S.; Cohen, B.E.; Etgar, L. Cell Refinement of CsPbBr3 Perovskite Nanoparticles and Thin Films. Nanoscale Adv. 2019, 1, 147–153. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, Y.; Sun, H.; Shi, P.; Lian, X.; Zhang, H.; Li, S.; Liang, S.; Wang, G.; Ma, F. The Effect of Co Content and Annealing Temperatures on the Resistivity in Ag-Co Films. Nanomaterials 2022, 12, 2297. https://doi.org/10.3390/nano12132297
Lv Y, Sun H, Shi P, Lian X, Zhang H, Li S, Liang S, Wang G, Ma F. The Effect of Co Content and Annealing Temperatures on the Resistivity in Ag-Co Films. Nanomaterials. 2022; 12(13):2297. https://doi.org/10.3390/nano12132297
Chicago/Turabian StyleLv, Yuanjiang, Haoliang Sun, Pengyan Shi, Xinxin Lian, Haoge Zhang, Saibo Li, Shihao Liang, Guangxin Wang, and Fei Ma. 2022. "The Effect of Co Content and Annealing Temperatures on the Resistivity in Ag-Co Films" Nanomaterials 12, no. 13: 2297. https://doi.org/10.3390/nano12132297
APA StyleLv, Y., Sun, H., Shi, P., Lian, X., Zhang, H., Li, S., Liang, S., Wang, G., & Ma, F. (2022). The Effect of Co Content and Annealing Temperatures on the Resistivity in Ag-Co Films. Nanomaterials, 12(13), 2297. https://doi.org/10.3390/nano12132297




