Solution Combustion Synthesis of Hafnium-Doped Indium Oxide Thin Films for Transparent Conductors
Abstract
1. Introduction
2. Materials and Methods
2.1. Precursor Solutions Preparation and Characterization
2.2. Thin Film Deposition and Characterization
3. Results and Discussion
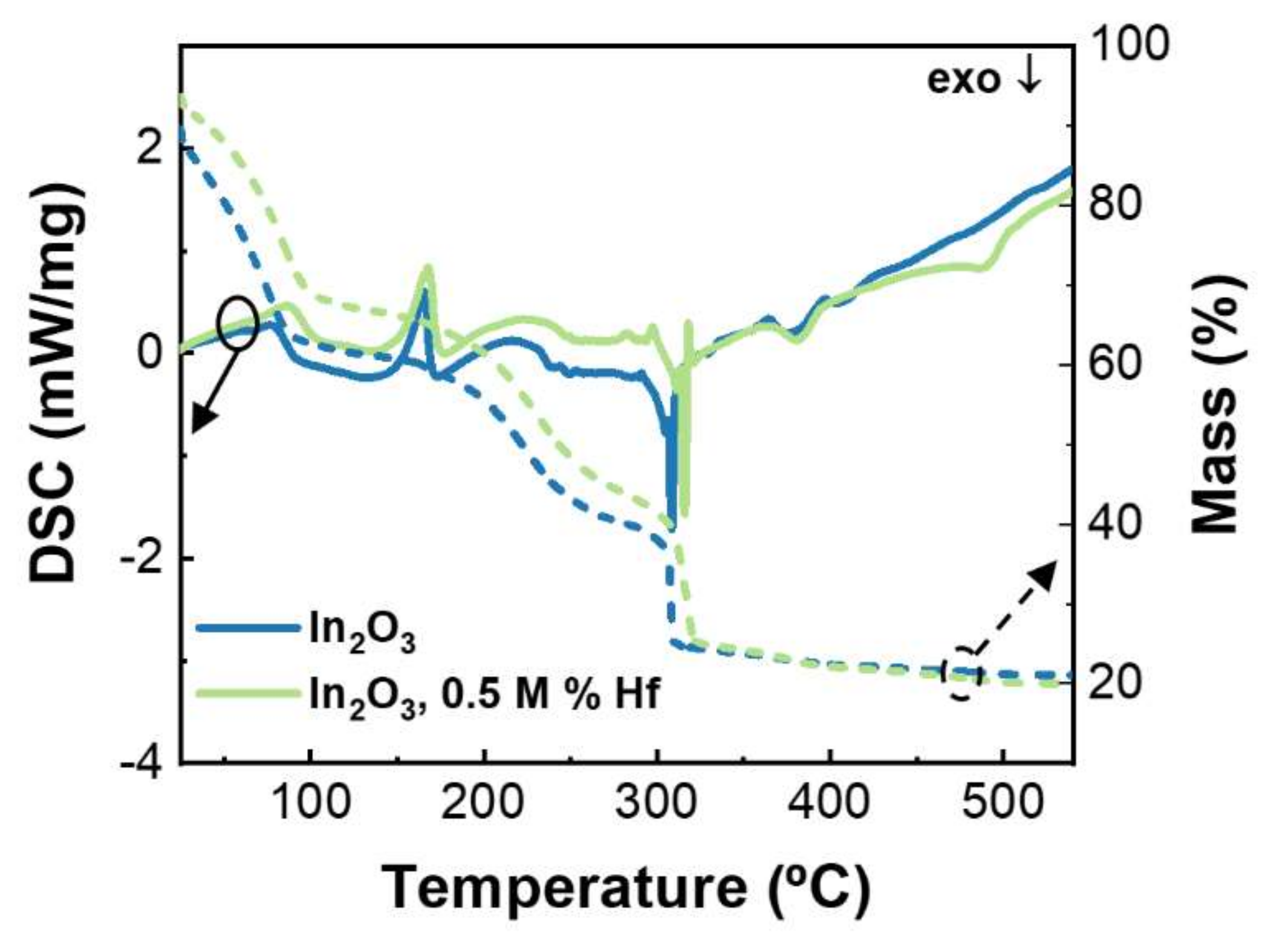
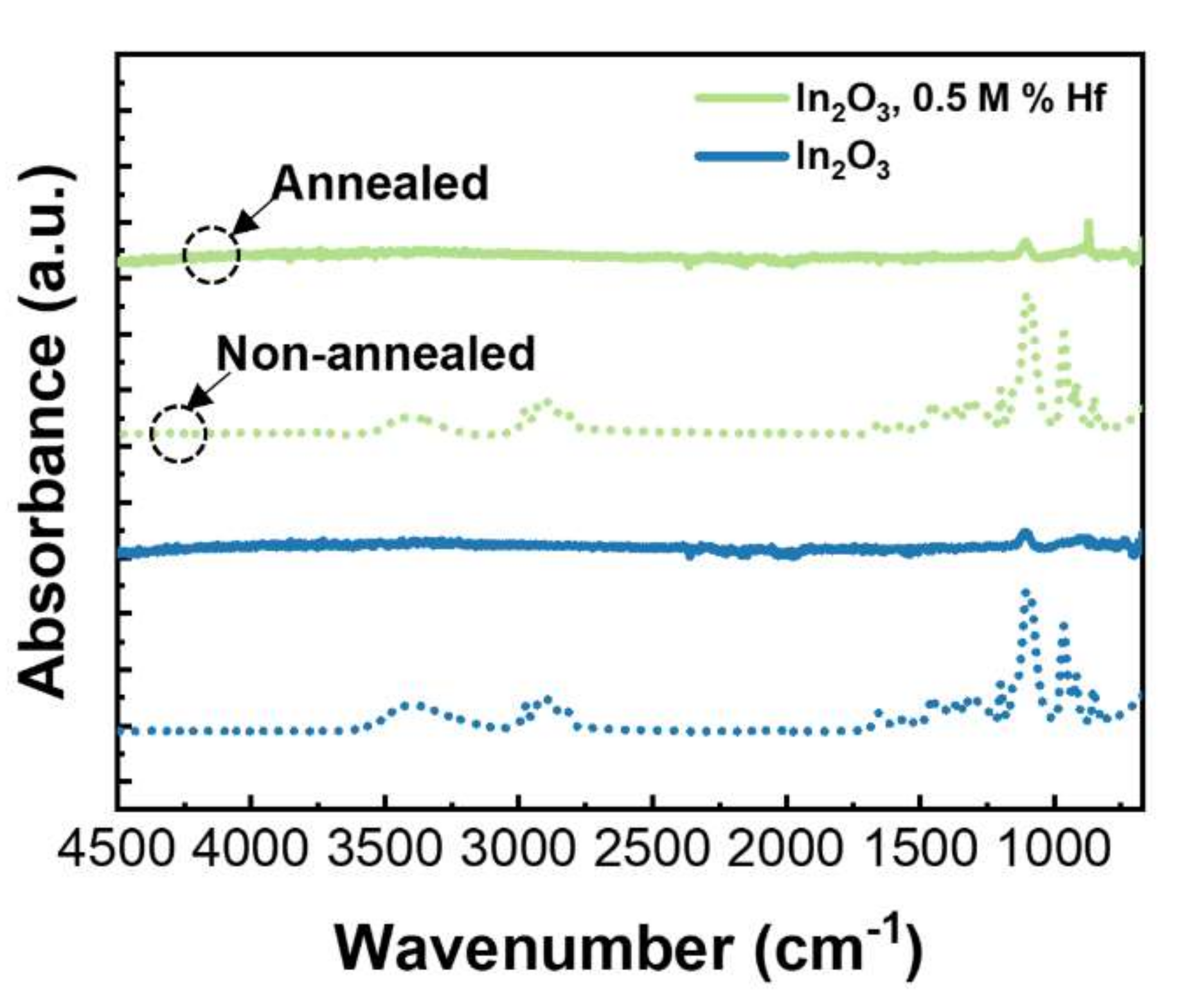
3.1. Precursor Solutions Characterization
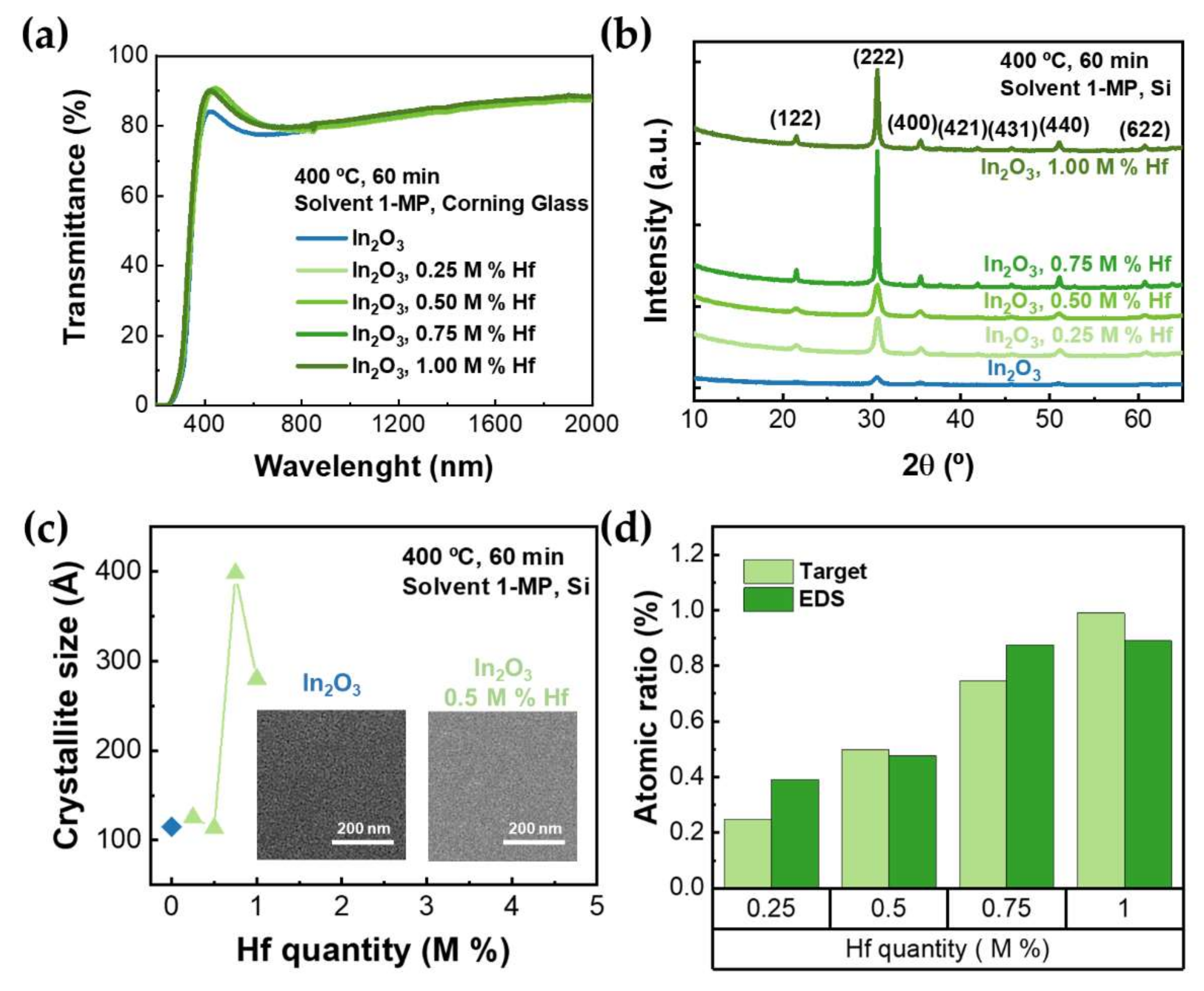
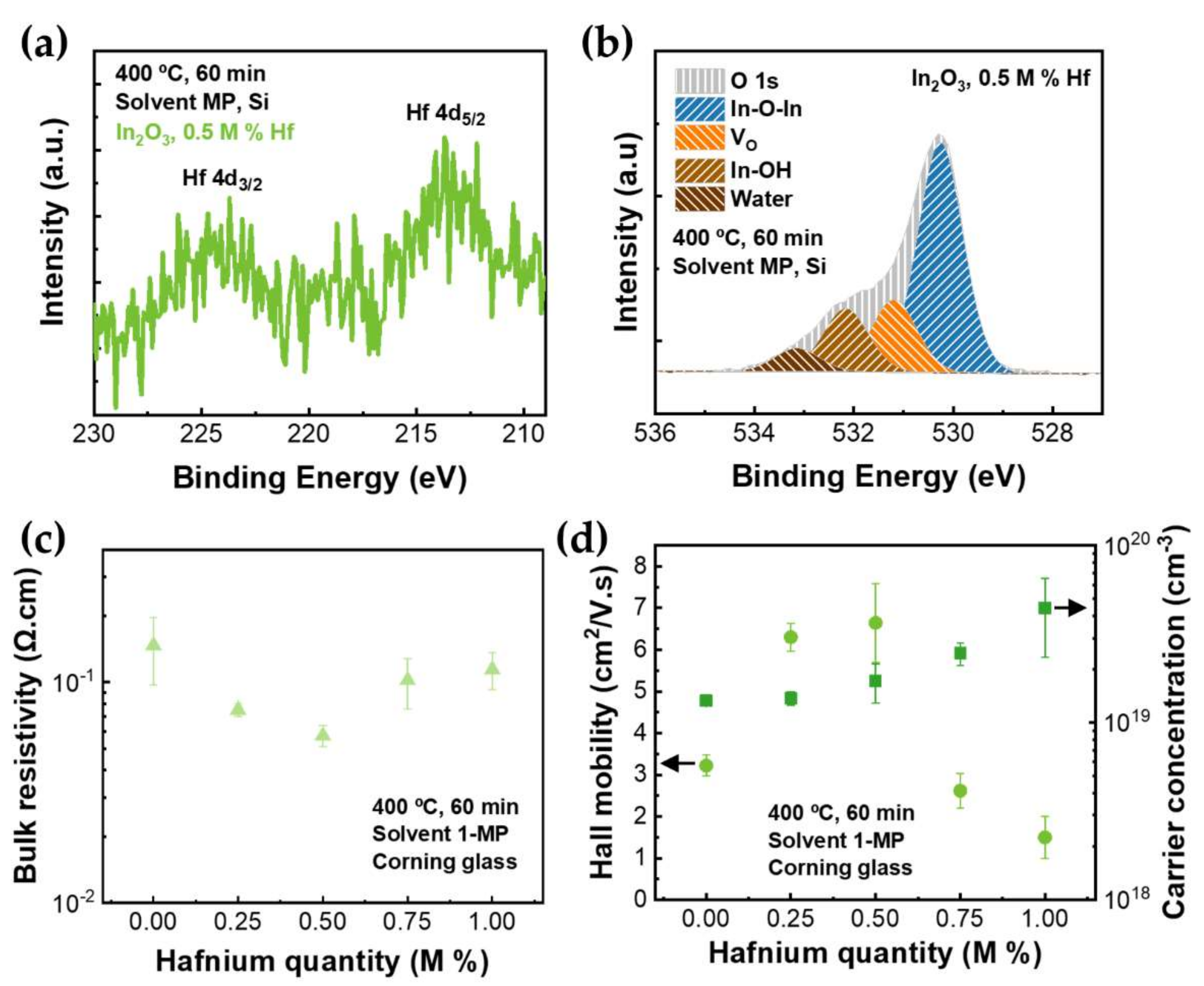
3.2. Doping of Indium Oxide Thin Films
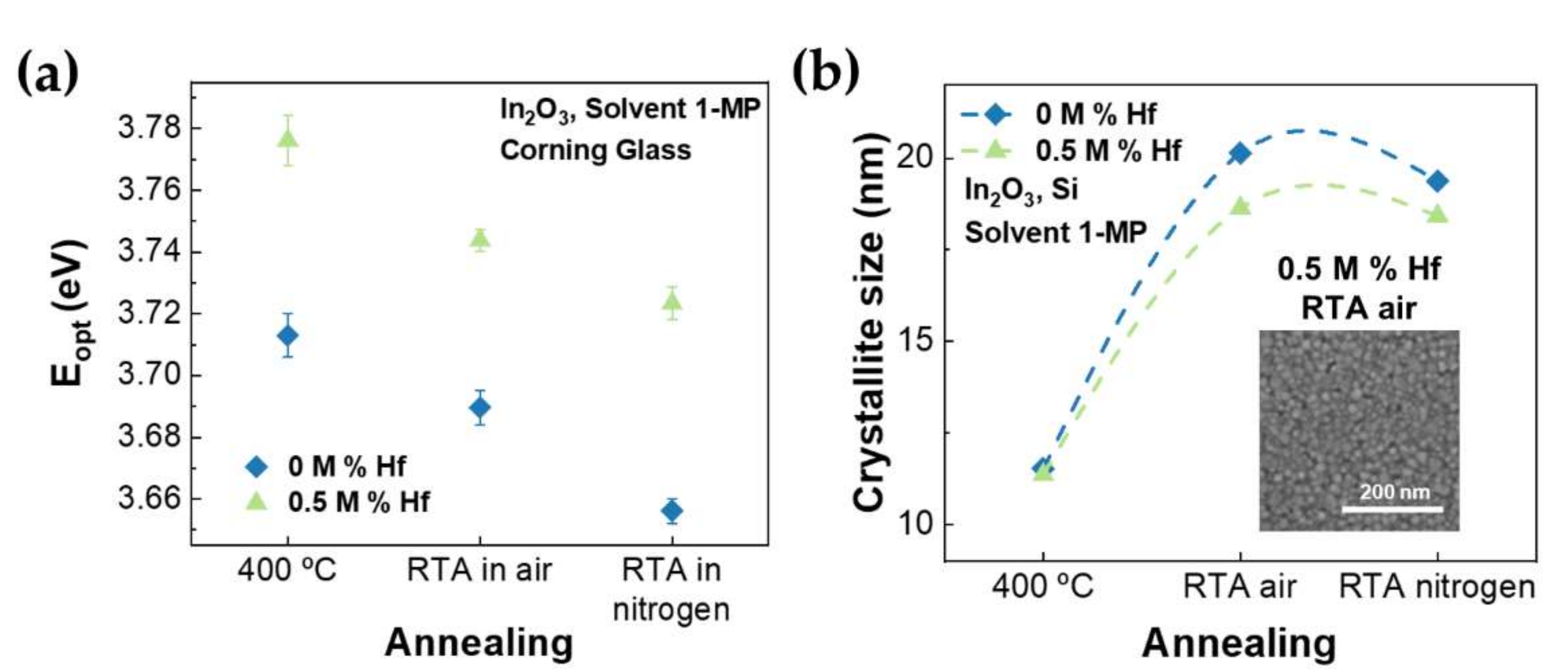
3.3. Effect of Post-Annealing in Undoped and Hf-Doped Indium Oxide Thin Films
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pujar, P.; Gandla, S.; Gupta, D.; Kim, S.; Kim, M. Trends in Low-Temperature Combustion Derived Thin Films for Solution-Processed Electronics. Adv. Electron. Mater. 2020, 6, 2000464. [Google Scholar] [CrossRef]
- Hosono, H.; Ueda, K. Transparent Conductive Oxides. In Handbook of Electronic and Photonic Materials; Kasap, S., Capper, P., Eds.; Springer: Cham, Switzerland, 2017; pp. 1391–1404. ISBN 9783319489339. [Google Scholar]
- Stadler, A. Transparent Conducting Oxides—An Up-To-Date Overview. Materials 2012, 5, 661–683. [Google Scholar] [CrossRef] [PubMed]
- Pasquarelli, R.M.; Ginley, D.S.; O’hayre, R. Solution processing of transparent conductors: From flask to film. Chem. Soc. Rev. 2011, 40, 5406–5441. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, G.P. Filmes Finos de Óxido de Índio e Zinco e sua Aplicação em Díodos Orgânicos Emissores de Luz. Ph.D. Thesis, Universidade Nova de Lisboa, Lisbon, Portugal, 2011. [Google Scholar]
- Barquinha, P. Transparent Oxide Thin-Film Transistors: Production, Characterization and Integration. Ph.D. Thesis, Universidade Nova de Lisboa, Lisbon, Portugal, 2010. [Google Scholar]
- Ullah, S.; Branquinho, R.; Mateus, T.; Martins, R.; Fortunato, E.; Rasheed, T.; Sher, F. Solution Combustion Synthesis of Transparent Conducting Thin Films for Sustainable Photovoltaic Applications. Sustainability 2020, 12, 10423. [Google Scholar] [CrossRef]
- Lee, S.; Jang, J.; Park, T.; Park, Y.M.; Park, J.S.; Kim, Y.K.; Lee, H.K.; Jeon, E.C.; Lee, D.K.; Ahn, B.; et al. Electrodeposited Silver Nanowire Transparent Conducting Electrodes for Thin-Film Solar Cells. ACS Appl. Mater. Interfaces 2020, 12, 6169–6175. [Google Scholar] [CrossRef]
- Kim, J.G.; Lee, J.E.; Jo, S.M.; Chin, B.D.; Baek, J.Y.; Ahn, K.J.; Kang, S.J.; Kim, H.K. Room temperature processed high mobility W-doped In2O3 electrodes coated via in-line arc plasma ion plating for flexible OLEDs and quantum dots LEDs. Sci. Rep. 2018, 8, 12019. [Google Scholar] [CrossRef]
- Kim, M.G.; Kanatzidis, M.G.; Facchetti, A.; Marks, T.J. Low-temperature fabrication of high-performance metal oxide thin-film electronics via combustion processing. Nat. Mater. 2011, 10, 382–388. [Google Scholar] [CrossRef]
- Koo, B.R.; Bae, J.W.; Ahn, H.J. Optoelectronic multifunctionality of combustion-activated fluorine-doped tin oxide films with high optical transparency. Ceram. Int. 2019, 45, 10260–10268. [Google Scholar] [CrossRef]
- Chae, G.S. Modified transparent conducting oxide for flat panel displays only. Jpn. Soc. Appl. Phys. 2001, 40, 1282–1286. [Google Scholar] [CrossRef]
- Sohn, S.; Soo, Y. Transparent Conductive Oxide (TCO) Films for Organic Light Emissive Devices (OLEDs). In Organic Light Emitting Diode—Material, Process and Devices; Ko, S.H., Ed.; InTech: Vienna, Austria, 2011; pp. 233–274. ISBN 9789533072739. [Google Scholar]
- Anouar, Y. Optimization of Indium Oxide Thin Films Properties Prepared by Sol Gel Spin Coating Process for Optoelectronic Applications. Ph.D. Thesis, University Mohamed Khider of Biskra, City, Algeria, 2020. [Google Scholar]
- Liu, Y.; Zhu, S.; Wei, R.; Hu, L.; Tang, X.; Yang, J.; Song, W.; Dai, J.; Zhu, X.; Sun, Y. Solution processed W-doped In2O3 thin films with high carrier mobility. Ceram. Int. 2020, 46, 2173–2177. [Google Scholar] [CrossRef]
- Seki, S.; Sawada, Y.; Ogawa, M.; Yamamoto, M.; Kagota, Y.; Shida, A.; Ide, M. Highly conducting indium-tin-oxide transparent films prepared by dip-coating with an indium carboxylate salt. Surf. Coatings Technol. 2003, 169–170, 525–527. [Google Scholar] [CrossRef]
- Deokate, R.J. Influence of Spray Solution Quantity on Microstructural and Optical Properties of In2O3 Thin Films Prepared by Spray Pyrolysis. Macromol. Symp. 2020, 392, 1900152. [Google Scholar] [CrossRef]
- Biswal, R.; Castañeda, L.; Moctezuma, R.; Vega-Pérez, J.; Olvera, M.D.L.L.; Maldonado, A. Formation of indium-doped zinc oxide thin films using ultrasonic spray pyrolysis: The importance of the water content in the aerosol solution and the substrate temperature for enhancing electrical transport. Materials 2012, 5, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Hwang, M.S.; Jeong, B.Y.; Moon, J.; Chun, S.K.; Kim, J. Inkjet-printing of indium tin oxide (ITO) films for transparent conducting electrodes. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2011, 176, 1128–1131. [Google Scholar] [CrossRef]
- Branquinho, R.; Salgueiro, D.; Santa, A.; Kiazadeh, A.; Barquinha, P.; Pereira, L.; Martins, R.; Fortunato, E. Towards environmental friendly solution-based ZTO/AlOx TFTs. Semicond. Sci. Technol. 2015, 30, 024007. [Google Scholar] [CrossRef]
- Wen, W.; Wu, J.M. Nanomaterials via solution combustion synthesis: A step nearer to controllability. RSC Adv. 2014, 4, 58090–58100. [Google Scholar] [CrossRef]
- Carlos, E.; Martins, R.; Fortunato, E.; Branquinho, R. Solution Combustion Synthesis: Towards a Sustainable Approach for Metal Oxides. Chem. A Eur. J. 2020, 26, 397–417. [Google Scholar] [CrossRef]
- Carlos, E.; Branquinho, R.; Barquinha, P.; Martins, R.; Fortunato, E. New strategies toward high performance and low-temperature processing of solution-based metal oxide TFTs. In Chemical Solution Synthesis for Materials Design and Thin Film Device Applications; Das, S., Dhara, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 585–621. ISBN 9780128197189. [Google Scholar]
- Carlos, E.; Branquinho, R.; Martins, R.; Kiazadeh, A.; Fortunato, E. Recent Progress in Solution-Based Metal Oxide Resistive Switching Devices. Adv. Mater. 2020, 33, 2004328. [Google Scholar] [CrossRef]
- Ruske, F. Deposition and Properties of TCOs; Springer: Berlin/Heidelberg, Germany, 2012; pp. 301–330. [Google Scholar]
- Xu, J.; Liu, J.B.; Liu, B.X.; Li, S.N.; Wei, S.H.; Huang, B. Design of n-Type Transparent Conducting Oxides: The Case of Transition Metal Doping in In2O3. Adv. Electron. Mater. 2018, 4, 1700553. [Google Scholar] [CrossRef]
- Medvedeva, J.E. Combining Optical Transparency with Electrical Conductivity: Challenges and Prospects. In Transparent Electronics: From Synthesis to Applications; Facchetti, A., Marks, T.J., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2010; pp. 1–29. [Google Scholar]
- Bierwagen, O. Indium oxide—A transparent, wide-band gap semiconductor for (opto)electronic applications. Semicond. Sci. Technol. 2015, 30, 024001. [Google Scholar] [CrossRef]
- Swallow, J.E.N.; Williamson, B.A.D.; Sathasivam, S.; Birkett, M.; Featherstone, T.J.; Murgatroyd, P.A.E.; Edwards, H.J.; Lebens-Higgins, Z.W.; Duncan, D.A.; Farnworth, M.; et al. Resonant doping for high mobility transparent conductors: The case of Mo-doped In2O3. Mater. Horizons 2020, 7, 236–243. [Google Scholar] [CrossRef]
- Bright, C.I. Deposition and Performance Challenges of Transparent Conductive Oxides on Plastic Substrates. In Transparent Electronics: From Synthesis to Applications; Facchetti, A., Marks, T.J., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2010; pp. 103–140. [Google Scholar]
- Kaleemulla, S.; Madhusudhana Rao, N.; Girish Joshi, M.; Sivasankar Reddy, A.; Uthanna, S.; Sreedhara Reddy, P. Electrical and optical properties of In2O3:Mo thin films prepared at various Mo-doping levels. J. Alloys Compd. 2010, 504, 351–356. [Google Scholar] [CrossRef]
- Wang, G.H.; Shi, C.Y.; Zhao, L.; Diao, H.W.; Wang, W.J. Transparent conductive Hf-doped In2O3 thin films by RF sputtering technique at low temperature annealing. Appl. Surf. Sci. 2017, 399, 716–720. [Google Scholar] [CrossRef]
- Averill, B.; Eldredge, P. The Periodic Table and Periodic Trends. In General Chemistry: Principles, Patterns, and Applications; Saylor Academy: Washington, DC, USA, 2012; pp. 590–672. ISBN 9781453322307. [Google Scholar]
- Wang, F.; Zhao, X.; Duan, L.; Wang, Y.; Niu, H.; Ali, A. Structural, optical and electrical properties of Hf-doped ZnO transparent conducting films prepared by sol–gel method. J. Alloys Compd. 2015, 623, 290–297. [Google Scholar] [CrossRef]
- Cheynet, M.C.; Pokrant, S.; Tichelaar, F.D.; Rouvière, J.-L. Crystal structure and band gap determination of HfO2 thin films. J. Appl. Phys. 2007, 101, 054101. [Google Scholar] [CrossRef]
- Fleming, R.M.; Lang, D.V.; Jones, C.D.W.; Steigerwald, M.L.; Murphy, D.W.; Alers, G.B.; Wong, Y.H.; Van Dover, R.B.; Kwo, J.R.; Sergent, A.M. Defect dominated charge transport in amorphous Ta2O5 thin films. J. Appl. Phys. 2000, 88, 850. [Google Scholar] [CrossRef]
- Patel, U.S.; Patel, K.H.; Chauhan, K.V.; Chawla, A.K.; Rawal, S.K. Investigation of Various Properties for Zirconium Oxide Films Synthesized by Sputtering. Procedia Technol. 2016, 23, 336–343. [Google Scholar] [CrossRef]
- Miyazaki, S.; Narasaki, M.; Ogasawara, M.; Hirose, M. Characterization of ultrathin zirconium oxide films on silicon using photoelectron spectroscopy. Microelectron. Eng. 2001, 59, 373–378. [Google Scholar] [CrossRef]
- Mangla, O.; Roy, S. Monoclinic Zirconium Oxide Nanostructures Having Tunable Band Gap Synthesized under Extremely Non-Equilibrium Plasma Conditions. Proceedings 2018, 3, 10. [Google Scholar] [CrossRef]
- Prathap, P.; Revathi, N.; Reddy, K.T.R.; Miles, R.W. Thickness dependence of structure and optoelectronic properties of In2O3:Mo films prepared by spray pyrolysis. Thin Solid Films 2009, 518, 1271–1274. [Google Scholar] [CrossRef]
- Beji, N.; Souli, M.; Azzaza, S.; Alleg, S.; Kamoun Turki, N. Study on the zinc doping and annealing effects of sprayed In2O3 thin films. J. Mater. Sci. Mater. Electron. 2016, 27, 4849–4860. [Google Scholar] [CrossRef]
- Beji, N.; Ajili, M.; Turki, N.K. Effect of Heat Treatment Under Nitrogen Atmosphere on Sprayed Fluorine Doped In2O3 Thin Films. J. Electron. Mater. 2016, 45, 3251–3258. [Google Scholar] [CrossRef]
- Pujar, P.; Gandla, S.; Singh, M.; Gupta, B.; Tarafder, K.; Gupta, D.; Noh, Y.-Y.; Mandal, S. Development of low temperature stoichiometric solution combustion derived transparent conductive ternary zinc tin co-doped indium oxide electrodes †. RSC Adv. 2017, 7, 48253–48262. [Google Scholar] [CrossRef]
- Pujar, P.; Vardhan, R.V.; Gupta, D.; Mandal, S. A balancing between super transparency and conductivity of solution combustion derived titanium doped indium oxide: Effect of charge carrier density and mobility. Thin Solid Films 2018, 660, 267–275. [Google Scholar] [CrossRef]
- Ullah, S.; Branquinho, R.; Santa, A.; De Matteis, F.; Martins, R.; Davoli, I.; Goncalves, G.; Fortunato, E. Boosting highly transparent and conducting indium zinc oxide thin films through solution combustion synthesis: Influence of rapid thermal annealing. Semicond. Sci. Technol. 2018, 33, 105004. [Google Scholar] [CrossRef]
- Manivannan, M.; Rajendran, S. Investigation of inhibitive action of urea-Zn2+ system in the corrosion control of carbon steel in sea water. Int. J. Eng. Sci. Technol. 2011, 3, 8038–8060. [Google Scholar]
- Epifani, M.; Melissano, E.; Pace, G.; Schioppa, M. Precursors for the combustion synthesis of metal oxides from the sol-gel processing of metal complexes. J. Eur. Ceram. Soc. 2007, 27, 115–123. [Google Scholar] [CrossRef]
- Senthamilselvi, V.; Saravanakumar, K.; Jabena Begum, N.; Anandhi, R.; Ravichandran, A.T.; Sakthivel, B.; Ravichandran, K. Photovoltaic properties of nanocrystalline CdS films deposited by SILAR and CBD techniques—A comparative study. J. Mater. Sci. Mater. Electron. 2012, 23, 302–308. [Google Scholar] [CrossRef]
- Xu, L.; Wang, R.; Liu, Y.; Zhang, D.; Xiao, Q. Influence of post-annealing on the properties of Ta-doped In2O3 transparent conductive films. Chinese Sci. Bull. 2011, 56, 1535–1538. [Google Scholar] [CrossRef][Green Version]
- Stroescu, H.; Anastasescu, M.; Preda, S.; Nicolescu, M.; Stoica, M.; Stefan, N.; Kampylafka, V.; Aperathitis, E.; Modreanu, M.; Zaharescu, M.; et al. Influence of thermal treatment in N2 atmosphere on chemical, microstructural and optical properties of indium tin oxide and nitrogen doped indium tin oxide rf-sputtered thin films. Thin Solid Films 2013, 541, 121–126. [Google Scholar] [CrossRef]
- Reddy, I.N.; Reddy, C.V.; Cho, M.; Shim, J.; Kim, D. Structural, optical and XPS study of thermal evaporated In2O3 thin films. Mater. Res. Express 2017, 4, 086406. [Google Scholar] [CrossRef]
- Tohsophon, T.; Dabirian, A.; De Wolf, S.; Morales-Masis, M.; Ballif, C. Environmental stability of high-mobility indium-oxide based transparent electrodes. APL Mater. 2015, 3, 116105. [Google Scholar] [CrossRef]
- Yan, Z.Y.; Run, X.; Bo, F.Z. Effects of annealing in N2 and air after CdCl2 treatment on the properties of CdS thin films. Adv. Mater. Res. 2012, 608–609, 1363–1365. [Google Scholar] [CrossRef]
- Won-Jae, L.; Chae-Ryong, C.; Kyung-Mok, C.; Se-Young, J. Rapid Thermal Annealing Effect of Al-Doped ZnO Thin Films. J. Korean Phys. Soc. 2005, 47, 296. [Google Scholar] [CrossRef]
- Park, J.-H.; Choi, Y.-Y.; Kim, H.-K.; Lee, H.H.; Na, S.-I. The effects of rapid thermal annealing on the electrical, optical, and structural properties of Nb:TiO2 multilayer electrodes with an inserted nanoscale Ag layer for organic solar cells. J. Appl. Phys. 2010, 108, 083509. [Google Scholar] [CrossRef]
- Ullah, S.; Matteis, F.D.E.; Davoli, I.; Yar Khan, R. Rapid Thermal Annealing for Solution Synthesis of Transparent Conducting Aluminum Zinc Oxide Thin Films. J. Electron. Mater. 2017, 46, 6609–6616. [Google Scholar] [CrossRef]
- Lyubchyk, A.; Vicente, A.; Soule, B.; Alves, P.U.; Mateus, T.; Mendes, M.J.; Águas, H.; Fortunato, E.; Martins, R. Mapping the Electrical Properties of ZnO-Based Transparent Conductive Oxides Grown at Room Temperature and Improved by Controlled Postdeposition Annealing. Adv. Electron. Mater. 2016, 2, 1500287. [Google Scholar] [CrossRef]
- Ayeshamariam, A.; Bououdina, M.; Sanjeeviraja, C. Optical, electrical and sensing properties of In2O3 nanopar-ticles. Mater. Sci. Semicond. Process. 2013, 16, 686–695. [Google Scholar] [CrossRef]
- Ahn, C.H.; Kim, J.H.; Cho, H.K. Tunable Electrical and Optical Properties in Composition Controlled Hf:ZnO Thin Films Grown by Atomic Layer Deposition. J. Electrochem. Soc. 2012, 159, H384–H387. [Google Scholar] [CrossRef]
- Geng, Y.; Xie, Z.Y.; Yang, W.; Xu, S.S.; Sun, Q.Q.; Ding, S.J.; Lu, H.L.; Zhang, D.W. Structural, optical, and elec-trical properties of Hf-doped ZnO films deposited by atomic layer deposition. Surf. Coatings Technol. 2013, 232, 41–45. [Google Scholar] [CrossRef]
- Alfakes, B.; Villegas, J.; Apostoleris, H.; Devarapalli, R.; Tamalampudi, S.; Lu, J.-Y.; Viegas, J.; Almansouri, I.; Chiesa, M. Optoelectronic Tunability of Hf-Doped ZnO for Photovoltaic Applications. J. Phys. Chem. C 2019, 123, 15258–15266. [Google Scholar] [CrossRef]
- Wang, G.H.; Shi, C.Y.; Zhao, L.; Mo, L.B.; Diao, H.W.; Wang, W.J. Efficiency improvement of the heterojunction solar cell using an antireflection Hf-doped In2O3 thin film prepared via glancing angle magnetron sputtering technology. Opt. Mater. 2020, 109, 110323. [Google Scholar] [CrossRef]

| Year | TCO | Tannealing (°C) | Time of Annealing | T550nm (%) | ρ (Ω.cm) | N (cm−3) | µ (cm2/V.s) |
|---|---|---|---|---|---|---|---|
| 2003 [16] | Sn (6.1 at%)—In2O3 | 600 | 1 h | 80 | 2.1 × 10−4 | 1.0 × 1021 | 28.0 |
| 2009 [40] | Mo (6 at%)—In2O3 | 400 | - | 75 | 8.1 × 10−4 | 1.9 × 1020 | 34.0 |
| 2015 [34] | Hf (3 at%)—ZnO | 550 | 2 h | 75 | 5.6 × 10−3 | 4.5 × 1019 | 2.5 |
| 2016 [41] | Zn (1 at%)—In2O3 | 250 | 2 h | 80 | 2.8 × 10−3 | 2.3 × 1020 | 1.0 |
| 2016 [42] | F (2 at%)—In2O3 | 400 | 45 min | 65 | 6.6 × 10−4 | 2.4 × 1021 | 11.3 |
| 2017 [43] | In2-2xZnxSnxO3; x = 0.3 | 400 | 30 min | 83 | 1.0 × 10−2 | 6.7 × 1019 | 6.0 |
| 2018 [44] | Ti (0.3 M)—In2O3 | 450 | 15 min | 97 | 5.0 × 10−2 | 1.0 × 1019 | 5.0 |
| 2020 [15] | W (0.5 at%)—In2O3 | 500 | 3 h | 77 | 5.4 × 10−4 | 5.2 × 1020 | 23.0 |
| This work | Hf (0.5 M%)—In2O3 | 600 | 10 min | 80 | 4.0 × 10−3 | 8.0 × 1019 | 21.0 |
| Hf (M%) | Thickness (nm) | T380–750nm (%) | Eopt (eV) |
|---|---|---|---|
| 0 | 104.0 ± 0.6 nm | 79.45 ± 2.26 | 3.72 ± 0.01 |
| 0.25 | 107.9 ± 1.2 nm | 83.33 ± 3.88 | 3.77 ± 0.01 |
| 0.50 | 110.9 ± 1.3 nm | 83.85 ± 4.06 | 3.78 ± 0.01 |
| 0.75 | 106.8 ± 0.5 nm | 83.53 ± 3.66 | 3.85 ± 0.01 |
| 1.00 | 101.8 ± 0.4 nm | 83.16 ± 3.63 | 3.85 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Firmino, R.; Carlos, E.; Pinto, J.V.; Deuermeier, J.; Martins, R.; Fortunato, E.; Barquinha, P.; Branquinho, R. Solution Combustion Synthesis of Hafnium-Doped Indium Oxide Thin Films for Transparent Conductors. Nanomaterials 2022, 12, 2167. https://doi.org/10.3390/nano12132167
Firmino R, Carlos E, Pinto JV, Deuermeier J, Martins R, Fortunato E, Barquinha P, Branquinho R. Solution Combustion Synthesis of Hafnium-Doped Indium Oxide Thin Films for Transparent Conductors. Nanomaterials. 2022; 12(13):2167. https://doi.org/10.3390/nano12132167
Chicago/Turabian StyleFirmino, Rita, Emanuel Carlos, Joana Vaz Pinto, Jonas Deuermeier, Rodrigo Martins, Elvira Fortunato, Pedro Barquinha, and Rita Branquinho. 2022. "Solution Combustion Synthesis of Hafnium-Doped Indium Oxide Thin Films for Transparent Conductors" Nanomaterials 12, no. 13: 2167. https://doi.org/10.3390/nano12132167
APA StyleFirmino, R., Carlos, E., Pinto, J. V., Deuermeier, J., Martins, R., Fortunato, E., Barquinha, P., & Branquinho, R. (2022). Solution Combustion Synthesis of Hafnium-Doped Indium Oxide Thin Films for Transparent Conductors. Nanomaterials, 12(13), 2167. https://doi.org/10.3390/nano12132167












