Efficient Photocatalytic Hydrogen Evolution over TiO2-X Mesoporous Spheres-ZnO Nanorods Heterojunction
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of ZnO Nanorods (NRs)
2.2. Preparation of ZnO Nanorods (NRs)/TiO2-x Mesoporous Spheres (MSs) Composites
2.3. Characterizations
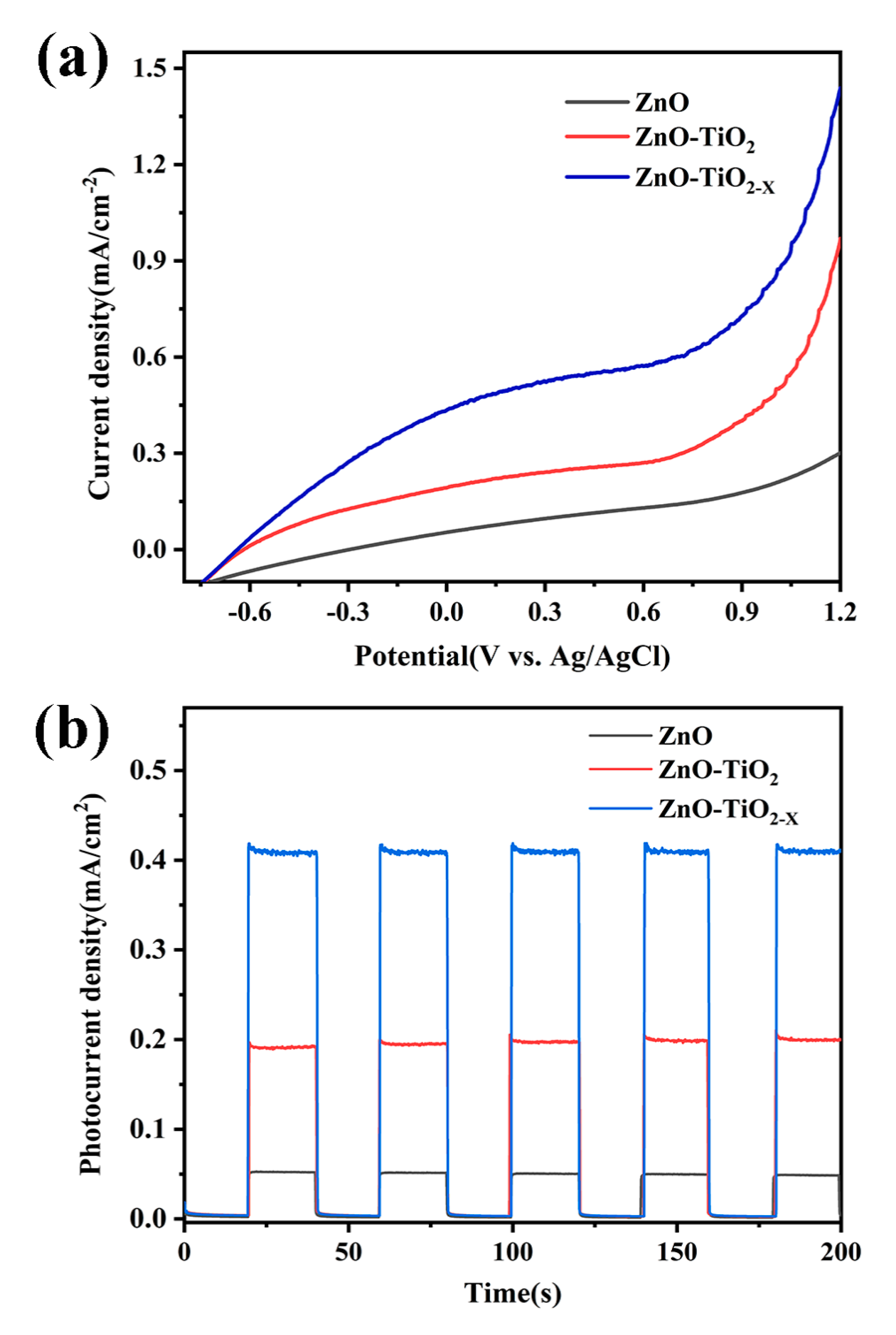
2.4. Photoelectrochemical (PEC) Measurements
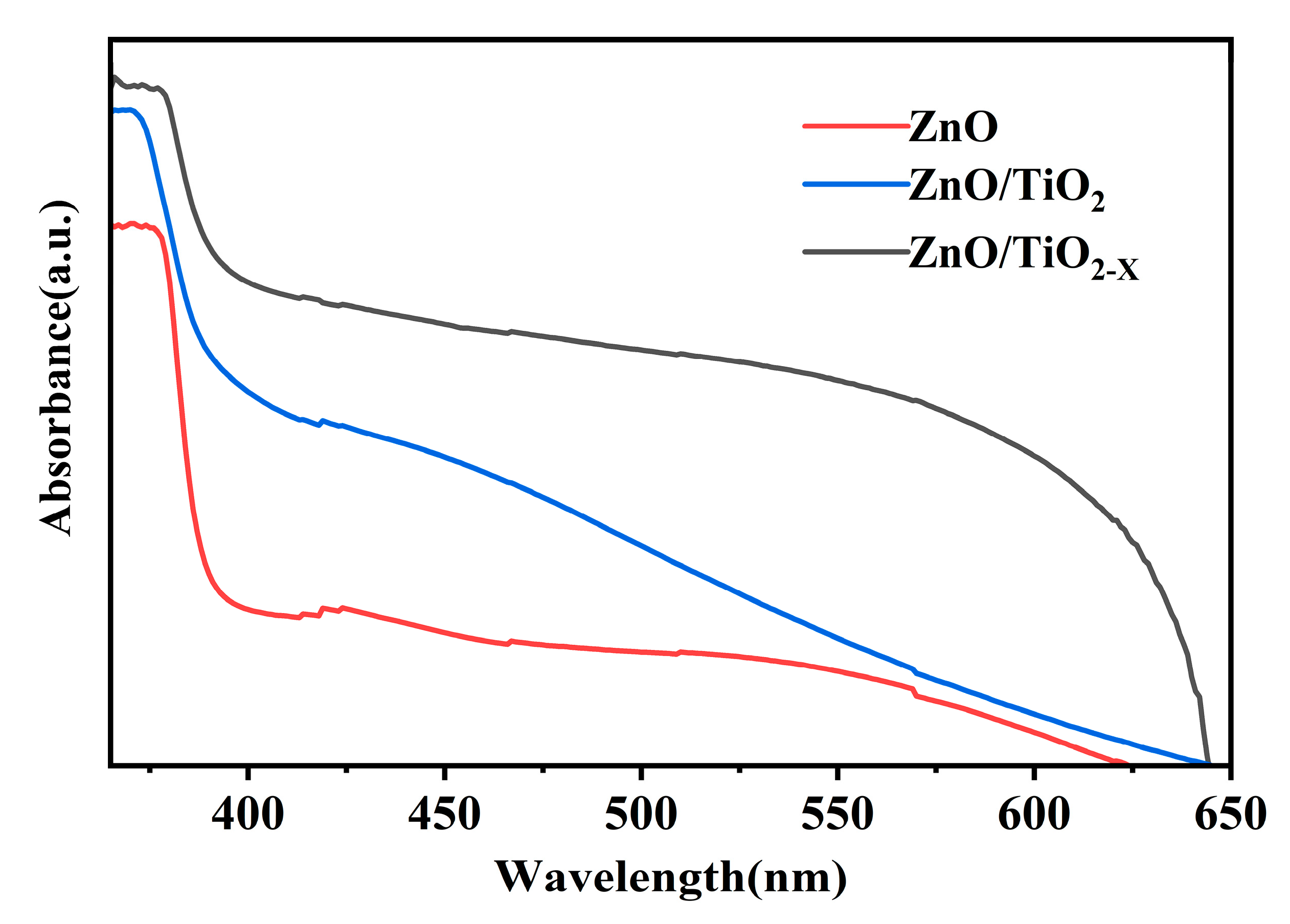
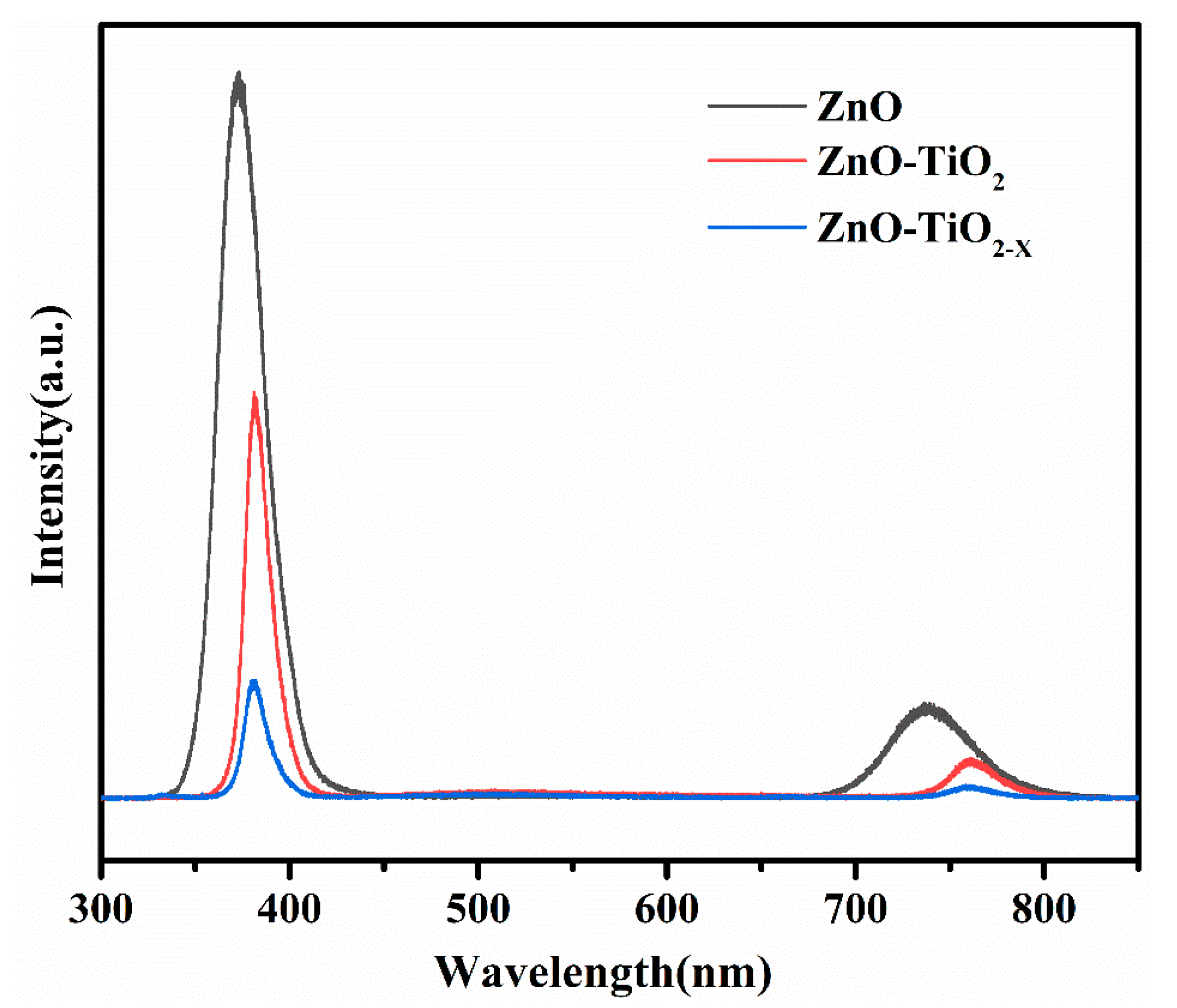
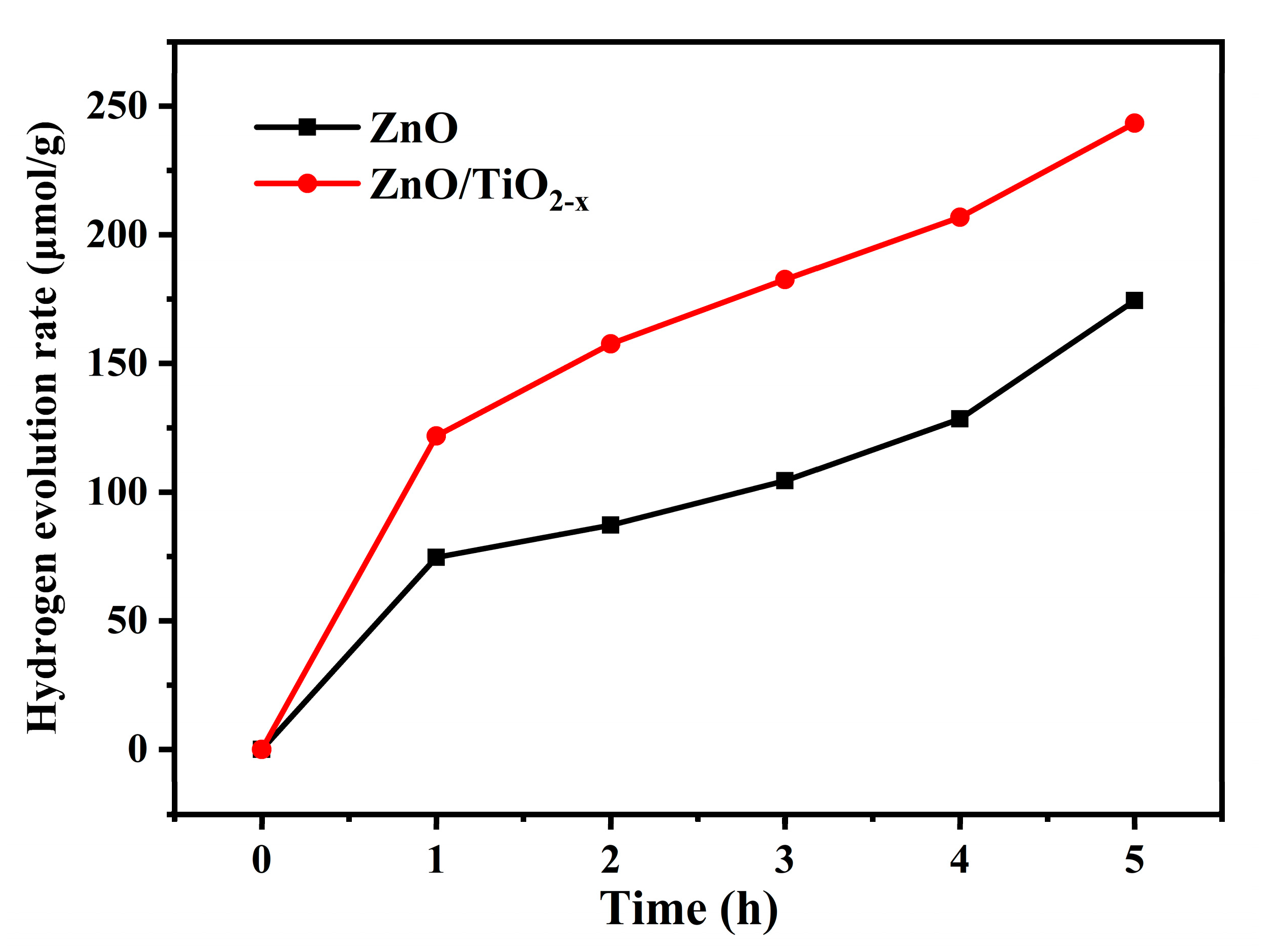
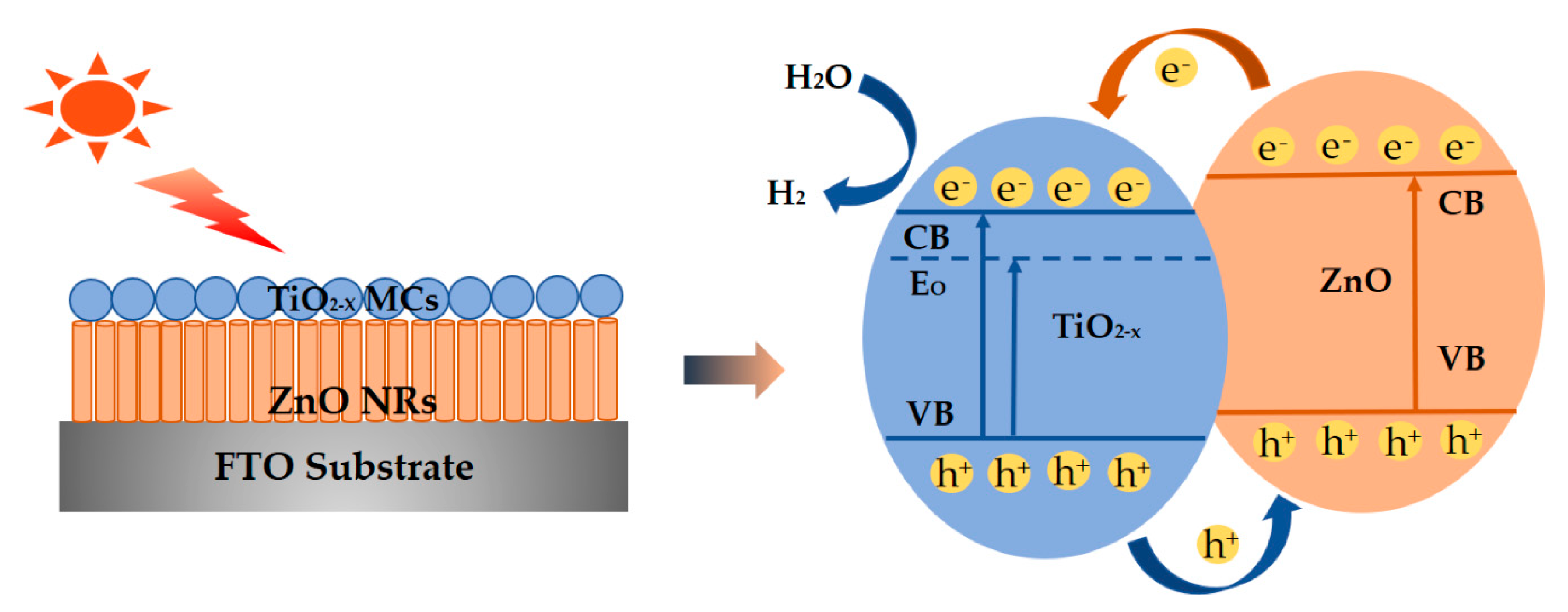
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tian, Z.R.; Voigt, J.A.; Liu, J.; Mckenzie, B.; Mcdermott, M.J.; Rodriguez, M.A.; Konishi, H.; Xu, H. Complex and oriented ZnO nanostructures. Nat. Mater. 2003, 2, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Hambali, N.A.; Hashim, A.M. Synthesis of zinc oxide nanostructures on graphene/glass substrate via electrochemical deposition: Effects of potassium chloride and hexamethylenetetramine as supporting reagents. Nano-Micro Lett. 2015, 7, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Zan, X.; Zhang, L.; Sun, D.D. Multi-functional CNT/ZnO/TiO2 nanocomposite membrane for concurrent filtration and photocatalytic degradation. Sep. Purif. Technol. 2015, 156, 922–930. [Google Scholar] [CrossRef]
- Van de Krol, R.; Liang, Y.; Schoonman, J. Solar hydrogen production with nanostructured metal oxides. J. Mater. Chem. 2008, 18, 2311. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, X.; Yeom, J. A floatable piezo-photocatalytic platform based on semi-embedded ZnO nanowire array for high-performance water decontamination. Nano-Micro Lett. 2019, 11, 11. [Google Scholar] [CrossRef]
- Hu, D.W.; Song, C.F.; Jin, X.; Huang, Q. Polymer solution-assisted assembly of hierarchically nano-structured ZnO onto 2D neat graphene sheets with excellent photocatalytic performance. J. Alloys Compd. 2020, 843, 156030. [Google Scholar] [CrossRef]
- Kayaci, F.; Vempati, S.; Ozgit-Akgun, C.; Donmez, I.; Biyikli, N.; Uyar, T. Transformation of polymer-ZnO core-shell nanofibers into ZnO hollow nanofibers: Intrinsic defect reorganization in ZnO and its influence on the photocatalysis. Appl. Catal. B Environ. 2015, 176, 646–653. [Google Scholar] [CrossRef]
- Zheng, H.B.; Wu, D.; Wang, Y.L.; Liu, X.P.; Gao, P.Z.; Liu, W.; Wen, J.; Rebrov, E.V. One-step synthesis of ZIF-8/ZnO composites based on coordination defect strategy and its derivatives for photocatalysis. J. Alloys Compd. 2020, 838, 155219. [Google Scholar] [CrossRef]
- Shimpi, N.T.; Rane, Y.N.; Shende, D.A.; Gosavi, S.R.; Ahirrao, P.B. Synthesis of rod-like ZnO nanostructure: Study of its physical properties and visible -light driven photocatalytic activity. Optik 2020, 217, 164916. [Google Scholar] [CrossRef]
- Mohd, A.M.A.; Julkapli, N.M.; Abd, H.S.B. Review on ZnO hybrid photocatalyst: Impact on photocatalytic activities of water pollutant degradation. Rev. Inorg. Chem. 2016, 36, 77–104. [Google Scholar]
- Vaiano, V.; Jaramillo-Paez, C.A.; Matarangolo, M.; Navio, J.A.; Hidalgo, M.D. UV and visible-light driven photocatalytic removal of caffeine using ZnO modified with different noble metals (Pt, Ag and Au). Mater. Res. Bull. 2019, 112, 251–260. [Google Scholar] [CrossRef]
- Pankaj, R.; Anita, S.; Pardeep, S. Photocatalytic water decontamination using graphene and ZnO coupled photocatalysts: A review. Mater. Sci. Energy Technol. 2019, 2, 509–525. [Google Scholar]
- Liu, J.L.; Wang, Y.H.; Ma, J.Z.; Peng, Y.; Wang, A.Q. A review on bidirectional analogies between the photocatalysis and antibacterial properties of ZnO. J. Alloys Compd. 2019, 783, 898–918. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, F.Z.; Zhu, C.Q.; Li, Q.; Song, J.N.; Zheng, M.J.; Ma, L.; Shen, W.Z. A facile self-assembly synthesis of hexagonal ZnO nanosheet films and their photoelectrochemical properties. Nano-Micro Lett. 2016, 8, 137–142. [Google Scholar] [CrossRef]
- Li, Q.; An, M.; Li, D. In situ growth of ZnO nanowire film on nanocrystalline zinc electrodeposit via a low-temperature hydrothermal reaction. Results Phys. 2019, 12, 1446–1449. [Google Scholar] [CrossRef]
- Reinosa, J.J.; Docio, C.M.A.; Ramirez, V.Z.; Fernandez, J.F. Hierarchical nano ZnO-micro TiO2 composites: High UV protection yield lowering photodegradation in sunscreens. Ceram. Int. 2018, 44, 2827–2834. [Google Scholar] [CrossRef]
- Hernandez, S.; Hidalgo, D.; Sacco, A.; Chiodoni, A.; Lamberti, A.; Cauda, V.; Tresso, E.; Saracco, G. Comparison of photocatalytic and transport properties of TiO2 and ZnO nanostructures for solar-driven water splitting. Phys. Chem. 2015, 17, 7775–7786. [Google Scholar] [CrossRef]
- Haghighatzadeh, A.; Hosseini, M.; Mazinani, B.; Shokouhimehr, M. Improved photocatalytic activity of ZnO-TiO2 nanocomposite catalysts by modulating TiO2 thickness. Mater. Res. Express 2019, 6, 115060. [Google Scholar] [CrossRef]
- Li, X.F.; Lv, K.L.; Deng, K.J.; Tang, J.F.; Su, R.; Sun, J.; Chen, L.Q. Synthesis and characterization of ZnO and TiO2 hollow spheres with enhanced photoreactivity. Mater. Sci. Eng. B 2009, 158, 40–47. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Zhu, S.P.; Chen, X.R.; Tang, Y.G.; Jiang, Y.F.; Peng, Z.G.; Wang, H.Y. One-step template-free fabrication of mesoporous ZnO/TiO2 hollow microspheres with enhanced photocatalytic activity. Appl. Surf. Sci. 2014, 307, 263–271. [Google Scholar] [CrossRef]
- Tan, H.; Zhao, Z.; Niu, M.; Mao, C.; Cao, D.; Cheng, D.; Feng, P.; Sun, Z.A. A facile and versatile method for preparation of colored TiO2 with enhanced solar-driven photocatalytic activity. Nanoscale 2014, 6, 10216–10223. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, L.; Yu, P.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, L.; Huang, F. Black titanium dioxide (TiO2) nanomaterials. Chem. Soc. Rev. 2015, 44, 1861–1885. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhu, G.; Wang, X.; Yuan, X.; Lin, T.; Huang, F. Progress in black titania: A new material for advanced photocatalysis. Adv. Energy Mater. 2016, 6, 1600452. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.L.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Dahl, M.; Liu, Y.; Yin, Y. Composite titanium dioxide nanomaterials. Chem. Rev. 2014, 114, 9853–9889. [Google Scholar] [CrossRef]
- Liu, G.; Kong, L.P.; Hu, Q.Y.; Zhang, S.J. Diffused morphotropic phase boundary in relaxor-PbTiO3 crystals: High piezoelectricity with improved thermal stability. Appl. Phys. Rev. 2020, 7, 021405. [Google Scholar] [CrossRef]
- Chen, D.; Huang, F.; Cheng, Y.-B.; Caruso, R.A. Mesoporous anatase TiO2 beads with high surface areas and controllable pore sizes: A superior candidate for high-performance dye-sensitized solar cells. Adv. Mater. 2009, 21, 2206–2210. [Google Scholar] [CrossRef]
- Zeng, Z.; Wang, D.B.; Wang, J.Z.; Jiao, S.J.; Huang, Y.W.; Zhao, S.X.; Zhang, B.K.; Ma, M.Y.; Gao, S.Y.; Feng, X.G.; et al. Self-Assembly Synthesis of the MoS2/PtCo Alloy Counter Electrodes for High-Efficiency and Stable Low-Cost Dye-Sensitized Solar Cells. Nanomaterials 2020, 10, 1725. [Google Scholar] [CrossRef]
- Sauvage, F.; Chen, D.; Huang, P.; Comte, F.; Heiniger, L.-P.; Cheng, Y.-B.; Caruso, R.A.; Grätzel, M. Dye-sensitized solar cells employing a single film of mesoporous TiO2 beads achieve power conversion efficiencies over 10%. ACS Nano 2010, 4, 4420–4425. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Bai, L.C.; Liu, H.Y.; Yu, X.F.; Yin, Y.D.; Gao, C.B. A Unique Disintegration-Reassembly Route to Mesoporous Titania Nanocrystalline Hollow Spheres with Enhanced Photocatalytic Activity. Adv. Funct. Mater. 2018, 28, 1704208. [Google Scholar] [CrossRef]
- Huang, Y.W.; Yu, Q.J.; Wang, J.Z.; Wang, J.A.; Yu, C.L.; Abdalla, J.T.; Zeng, Z.; Jiao, S.J.; Wang, D.B.; Gao, S.Y. Plasmon-Enhanced Self-Powered UV Photodetectors Assembled by Incorporating Ag@SiO2 Core-Shell Nanoparticles into TiO2 Nanocube Photoanodes. ACS Sustain. Chem. Eng. 2018, 6, 438–446. [Google Scholar] [CrossRef]
- Chen, J.Q.; Huang, S.L.; Long, Y.J.; Wu, J.H.; Li, H.; Li, Z.; Zeng, Y.J.; Ruan, S.C. Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting. Nanomaterials 2018, 8, 835. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, S.; Gargiulo, V.; Alfe, M.; Amati, M.; Zeller, P.; Maraloiu, V.A.; Borbone, F.; Pavone, M.; Munoz-Garcia, A.B.; Maddalena, P. Simple Ethanol Refluxing Method for Production of Blue-Colored Titanium Dioxide with Oxygen Vacancies and Visible Light-Driven Photocatalytic Properties. J. Phys. Chem. C 2020, 124, 3564–3576. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, C.Y. TiO2 Coated ZnO Nanorods by Mist Chemical Vapor Deposition for Application as Photoanodes for Dye-Sensitized Solar Cells. Nanomaterials 2019, 9, 1339. [Google Scholar] [CrossRef]
- Doğan, İ.; van de Sanden, M.C.M. Direct characterization of nanocrystal size distribution using Raman spectroscopy. J. Appl. Phys. 2013, 114, 134310. [Google Scholar] [CrossRef]
- Rajalakshmi, M.; Arora, A.K.; Bendre, B.S. Optical phonon confinement in zinc oxide nanoparticles. Phys. Rev. 2000, 87, 2445–2448. [Google Scholar] [CrossRef]
- Ursaki, V.V.; Tiginyanu, I.M.; Zalamai, V.V.; Rusu, E.V.; Emelchenko, G.A.; Masalov, V.M.; Samarov, E.N. Multiphonon resonant Raman scattering in ZnO crystals and nanostructured layers. Phys. Rev. B. 2004, 70, 155204. [Google Scholar] [CrossRef]
- Zhou, W.; Sun, F.F.; Pan, K.; Tian, G.H.; Jiang, B.J.; Ren, Z.Y.; Tian, C.G.; Fu, H.G. Well-Ordered Large-Pore Mesoporous Anatase TiO2 with Remarkably High Thermal Stability and Improved Crystallinity: Preparation, Characterization, and Photocatalytic Performance. Adv. Funct. Mater. 2011, 21, 1922–1930. [Google Scholar] [CrossRef]
- Vasu, K.; Sreedhara, M.B.; Ghatak, J.; Rao, C.N.R. Atomic layer deposition of p-type epitaxial thin films of undoped and N-doped anatase TiO2. ACS Appl. Mater. Interfaces 2016, 8, 7897–7901. [Google Scholar] [CrossRef]
- Hwang, Y.J.; Yang, S.; Lee, H. Surface analysis of N-doped TiO2 nanorods and their enhanced photocatalytic oxidation activity. Appl. Catal. B. 2017, 204, 209–215. [Google Scholar] [CrossRef]
- O’Brien, S.; Koh, L.H.K.; Crean, G.M. ZnO thin films prepared by a single step sol-gel process. Thin Solid Films 2008, 516, 1391–1395. [Google Scholar] [CrossRef]
- Ullattil, S.G.; Narendranath, S.B.; Pillai, S.C.; Periyat, P. Black TiO2 Nanomaterials: A Review of Recent Advances. Chem. Eng. J. 2018, 343, 708–736. [Google Scholar] [CrossRef]
- Lima, S.A.M.; Sigoli, F.A.; Jafelicci, M.; Davolos, M.R. Luminescent properties and lattice correlation defects on zinc oxide. Int. J. Inorg. Mater. 2001, 3, 749–754. [Google Scholar] [CrossRef]
- Shi, Z.-F.; Zhang, Y.-T.; Cui, X.-J.; Zhuang, S.-W.; Wu, B.; Chu, X.-W.; Dong, X.; Zhang, B.-L.; Du, G.-T. Photoluminescence performance enhancement of ZnO/MgO heterostructured nanowires and their applications in ultraviolet laser diodes. Phys. Chem. Chem. Phys. 2015, 17, 13813–13820. [Google Scholar] [CrossRef]
- Ma, D.; Shi, J.-W.; Zou, Y.; Fan, Z.; Ji, X.; Niu, C.; Wang, L. Rational design of CdS@ZnO core-shell structure via atomic layer deposition for drastically enhanced photocatalytic H2 evolution with excellent photostability. Nano Energy 2017, 39, 183–191. [Google Scholar] [CrossRef]
- Amany, A.; Wang, D.B.; Wang, J.Z.; Zeng, Z.; Jiao, S.J.; Hao, C.L.; Zhao, Y.C.; Xie, Y.; Gao, S.Y.; Ni, S.M.; et al. Enhanced the UV response of AlN coated ZnO nanorods photodetector. J. Alloys Compd. 2018, 776, 111–115. [Google Scholar] [CrossRef]
- Pastore, M.; De Angelis, F. Computational Modeling of Stark Effects in Organic Dye-Sensitized TiO2 Heterointerfaces. J. Phys. Chem. Lett. 2011, 2, 1261–1267. [Google Scholar] [CrossRef]
- Sun, L.H.; Dong, A.N.; Li, J.M.; Hao, D.; Tang, X.Q.; Yan, S.C.; Guo, Y.; Shan, X.Y.; Lu, X.H. Optical Stark effect of a local defect on the TiO2 (110) surface. Phys. Rev. B. 2018, 98, 081402. [Google Scholar] [CrossRef]
- Sun, C.Y.; Xu, Q.H.; Xie, Y.; Ling, Y.; Hou, Y. Designed synthesis of anatase–TiO2(B) biphase nanowire/ZnO nanoparticle heterojunction for enhanced photocatalysis. J. Mater. Chem. A 2018, 6, 8289. [Google Scholar] [CrossRef]
- Han, H.S.; Wang, W.Z.; Yao, L.Z.; Hao, C.C.; Liang, Y.J.; Fu, J.L.; Zeng, P.B. Photostable 3D heterojunction photoanode made of ZnO nanosheets coated onto TiO2 nanowire arrays for photoelectrochemical solar hydrogen generation. Catal. Sci. Technol. 2019, 9, 1989. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, B.; Li, Q.; Wang, D.; Wang, J.; Jiang, B.; Jiao, S.; Liu, D.; Zeng, Z.; Zhao, C.; Liu, Y.; et al. Efficient Photocatalytic Hydrogen Evolution over TiO2-X Mesoporous Spheres-ZnO Nanorods Heterojunction. Nanomaterials 2020, 10, 2096. https://doi.org/10.3390/nano10112096
Zhang B, Li Q, Wang D, Wang J, Jiang B, Jiao S, Liu D, Zeng Z, Zhao C, Liu Y, et al. Efficient Photocatalytic Hydrogen Evolution over TiO2-X Mesoporous Spheres-ZnO Nanorods Heterojunction. Nanomaterials. 2020; 10(11):2096. https://doi.org/10.3390/nano10112096
Chicago/Turabian StyleZhang, BingKe, Qi Li, Dongbo Wang, Jinzhong Wang, Baojiang Jiang, Shujie Jiao, DongHao Liu, Zhi Zeng, ChenChen Zhao, YaXin Liu, and et al. 2020. "Efficient Photocatalytic Hydrogen Evolution over TiO2-X Mesoporous Spheres-ZnO Nanorods Heterojunction" Nanomaterials 10, no. 11: 2096. https://doi.org/10.3390/nano10112096
APA StyleZhang, B., Li, Q., Wang, D., Wang, J., Jiang, B., Jiao, S., Liu, D., Zeng, Z., Zhao, C., Liu, Y., Xun, Z., Fang, X., Gao, S., Zhang, Y., & Zhao, L. (2020). Efficient Photocatalytic Hydrogen Evolution over TiO2-X Mesoporous Spheres-ZnO Nanorods Heterojunction. Nanomaterials, 10(11), 2096. https://doi.org/10.3390/nano10112096





