Lipid Nanoparticles for Ocular Gene Delivery
Abstract
:1. Introduction
2. Uses and Advantages of Nanoparticles in Medicine
3. Gene Therapy and Viral Vectors
| Vector | Carrier | Delivery | Ref. |
|---|---|---|---|
| Virus | AAV | Local/systemic | [31,32,33,34,35] |
| Adenovirus | Local | [36] | |
| Baculovirus | Local | [37,38] | |
| Lentivirus | Local | [39] | |
| Non-virus | Liposome nanoparticles | Local | [40,41,42,43] |
| Solid lipid nanoparticles | Local | [9,13,16,17,21,22] | |
| LPD/lipoplexes | Local | [23,24,44] | |
| CK30-PEG | Local | [5,6,7,8] |
4. Lipid-Based Nanoparticles
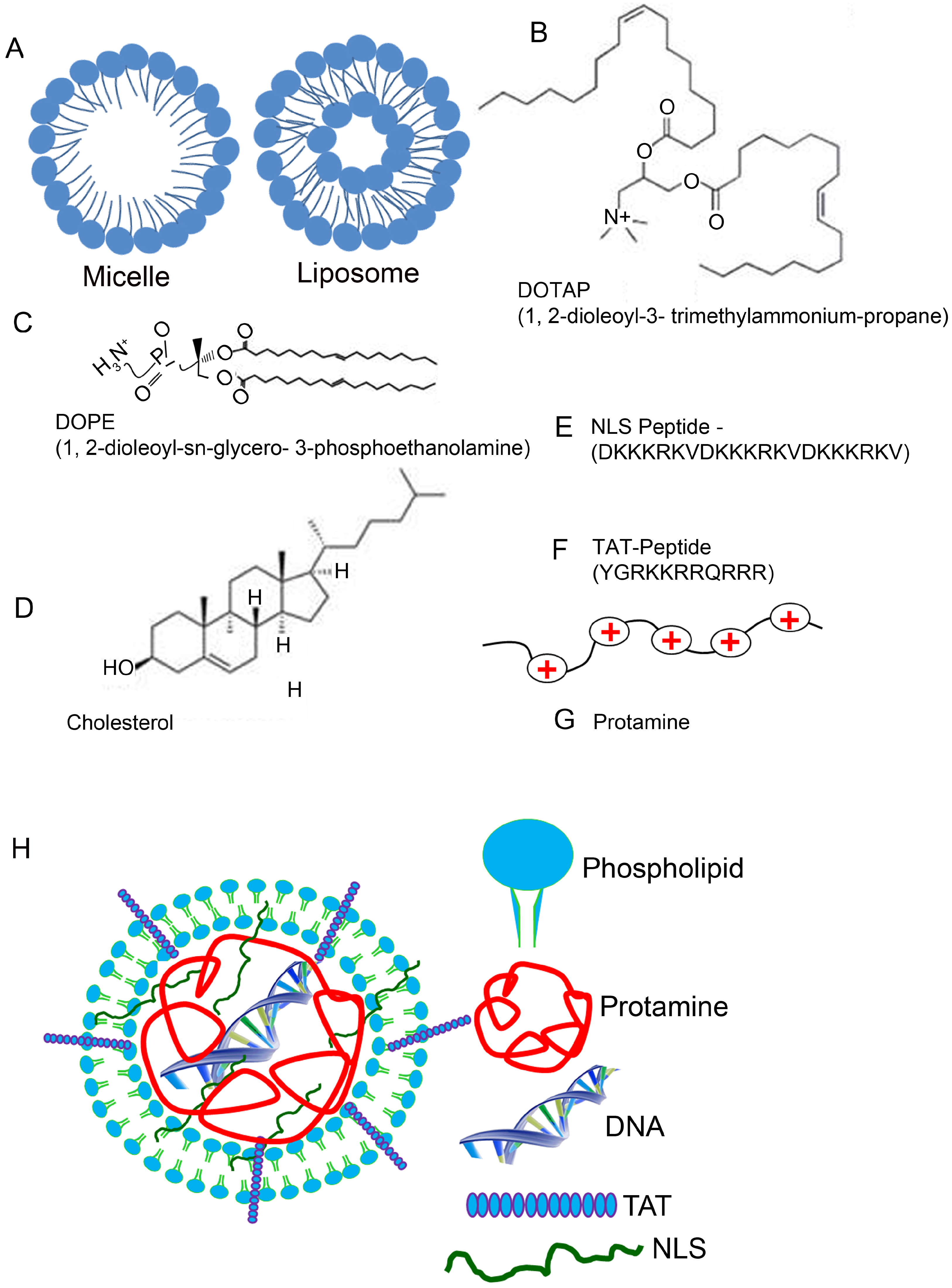
5. Composition of Lipid Nanoparticles
6. Transfer Mechanism of LPD Nanoparticles into Cells
7. Cellular Barriers in the Internalization of Lipid Nanoparticles
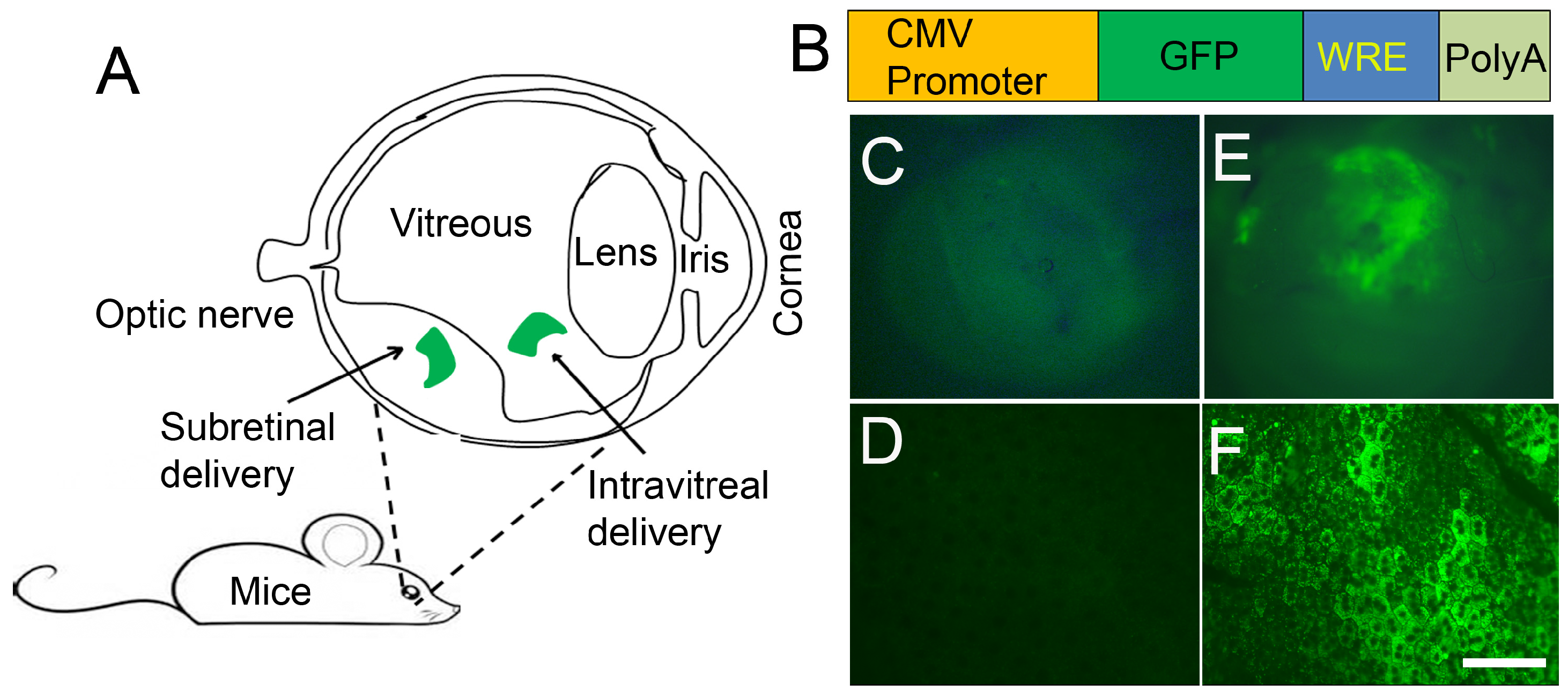
8. LPD Nanoparticle-Mediated Delivery of Genes to Eye Tissues
9. Cell-Specific Delivery of LPD Nanoparticles
10. Conclusions
Acknowledgements
Conflict of Interest
References
- Adijanto, J.; Naash, M.I. Nanoparticle-based technologies for retinal gene therapy. Eur. J. Pharm. Biopharm. 2015. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, N.R.; Vodovozova, E.L. Differential binding of plasma proteins by liposomes loaded with lipophilic prodrugs of methotrexate and melphalan in the bilayer. Biochemistry (Mosc.) 2014, 79, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Liu, J. Liposome supported metal oxide nanoparticles, interaction mechanism, light controlled content release, and intracellular delivery. Small 2014, 10, 3927–3931. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Szoka, F.C., Jr. Mechanism of DNA release from cationic liposome/DNA complexes used in cell transfection. Biochemistry 1996, 35, 5616–5623. [Google Scholar] [CrossRef] [PubMed]
- Koirala, A.; Makkia, R.S.; Conley, S.M.; Cooper, M.J.; Naash, M.I. S/MAR-containing DNA nanoparticles promote persistent RPE gene expression and improvement in RPE65-associated LCA. Hum. Mol. Genet. 2013, 22, 1632–1642. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Conley, S.M.; Makkia, R.S.; Cooper, M.J.; Naash, M.I. DNA nanoparticle-mediated ABCA4 delivery rescues Stargardt dystrophy in mice. J. Clin. Invest. 2012, 122, 3221–3226. [Google Scholar] [CrossRef] [PubMed]
- Rowe-Rendleman, C.L.; Durazo, S.A.; Kompella, U.B.; Rittenhouse, K.D.; Di, P.A.; Weiner, A.L.; Grossniklaus, H.E.; Naash, M.I.; Lewin, A.S.; Horsager, A.; et al. Drug and gene delivery to the back of the eye, from bench to bedside. Invest. Ophthalmol. Vis. Sci. 2014, 55, 2714–2730. [Google Scholar] [CrossRef] [PubMed]
- Koirala, A.; Conley, S.M.; Makkia, R.; Liu, Z.; Cooper, M.J.; Sparrow, J.R.; Naash, M.I. Persistence of non-viral vector mediated RPE65 expression, case for viability as a gene transfer therapy for RPE-based diseases. J. Control. Release 2013, 172, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Seyfoddin, A.; Al-Kassas, R. Development of solid lipid nanoparticles and nanostructured lipid carriers for improving ocular delivery of acyclovir. Drug Dev. Ind. Pharm. 2013, 39, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.A.; Liu, Y.L.; Ma, Z.Z.; Wang, J.C.; Zhang, Q. A lipid nanoparticle system improves siRNA efficacy in RPE cells and a laser-induced murine CNV model. Invest. Ophthalmol. Vis. Sci. 2011, 52, 4789–4794. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Jiang, S.; Liu, D.; Bi, X.; Wang, F.; Zhang, Q.; Xu, Q. A potential new therapeutic system for glaucoma, solid lipid nanoparticles containing methazolamide. J. Microencapsul. 2011, 28, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Souto, E.B.; Doktorovova, S.; Gonzalez-Mira, E.; Egea, M.A.; Garcia, M.L. Feasibility of lipid nanoparticles for ocular delivery of anti-inflammatory drugs. Curr. Eye Res. 2010, 35, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Seyfoddin, A.; Shaw, J.; Al-Kassas, R. Solid lipid nanoparticles for ocular drug delivery. Drug Deliv. 2010, 17, 467–489. [Google Scholar] [CrossRef] [PubMed]
- Gokce, E.H.; Sandri, G.; Bonferoni, M.C.; Rossi, S.; Ferrari, F.; Guneri, T.; Caramella, C. Cyclosporine A loaded SLNs: Evaluation of cellular uptake and corneal cytotoxicity. Int. J. Pharm. 2008, 364, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Gokce, E.H.; Sandri, G.; Egrilmez, S.; Bonferoni, M.C.; Guneri, T.; Caramella, C. Cyclosporine A-loaded solid lipid nanoparticles, ocular tolerance and in vivo drug release in rabbit eyes. Curr. Eye Res. 2009, 34, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Sun, M.; Ping, Q.; Ying, Z.; Liu, W. Incorporation of liquid lipid in lipid nanoparticles for ocular drug delivery enhancement. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Attama, A.A.; Reichl, S.; Muller-Goymann, C.C. Sustained release and permeation of timolol from surface-modified solid lipid nanoparticles through bioengineered human cornea. Curr. Eye Res. 2009, 34, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Del Pozo-Rodriguez, A.; Delgado, D.; Solinis, M.A.; Gascon, A.R.; Pedraz, J.L. Solid lipid nanoparticles for retinal gene therapy, transfection and intracellular trafficking in RPE cells. Int. J. Pharm. 2008, 360, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Montasser, I.; Shahgaldian, P.; Perret, F.; Coleman, A.W. Solid lipid nanoparticle-based calix[n]arenes and calix-resorcinarenes as building blocks, synthesis, formulation and characterization. Int. J. Mol. Sci. 2013, 14, 21899–21942. [Google Scholar] [CrossRef] [PubMed]
- Mashaghi, S.; Jadidi, T.; Koenderink, G.; Mashaghi, A. Lipid nanotechnology. Int. J. Mol. Sci. 2013, 14, 4242–4282. [Google Scholar] [CrossRef] [PubMed]
- Attama, A.A.; Reichl, S.; Muller-Goymann, C.C. Diclofenac sodium delivery to the eye, in vitro evaluation of novel solid lipid nanoparticle formulation using human cornea construct. Int. J. Pharm. 2008, 355, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, R.; Gasco, M.R.; Chetoni, P.; Burgalassi, S.; Saettone, M.F. Solid lipid nanoparticles (SLN) as ocular delivery system for tobramycin. Int. J. Pharm. 2002, 238, 241–245. [Google Scholar] [CrossRef]
- Rajala, A.; Wang, Y.; Zhu, Y.; Ranjo-Bishop, M.; Ma, J.X.; Mao, C.; Rajala, R.V. Nanoparticle-assisted targeted delivery of eye-specific genes to eyes significantly improves the vision of blind mice in vivo. Nano. Lett. 2014, 14, 5257–5263. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Chen, Q.; Rajala, R.V.; Ma, J. Micro RNA-184 modulates cannocial Wnt signaling through regulation of frizzled-7 expression in the retina with ischemia-induced neovascularization. FEBS Lett. 2015, 589, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Qiu, P.; Mao, C. Mesoporous iron oxide nanoparticles prepared by polyacrylic acid etching and their application in gene delivery to mesenchymal stem cells. Microsc. Res. Tech. 2013, 76, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, R.C. The basic science of gene therapy. Science 1993, 260, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Herz, J.; Gerard, R.D. Adenovirus-mediated transfer of low density lipoprotein receptor gene acutely accelerates cholesterol clearance in normal mice. Proc. Natl. Acad. Sci USA 1993, 90, 2812–2816. [Google Scholar] [CrossRef] [PubMed]
- Simon, R.H.; Engelhardt, J.F.; Yang, Y.; Zepeda, M.; Weber-Pendleton, S.; Grossman, M.; Wilson, J.M. Adenovirus-mediated transfer of the CFTR gene to lung of nonhuman primates, toxicity study. Hum. Gene Ther. 1993, 4, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Lemoine, N.R.; Ring, C.J. The use of DNA viruses as vectors for gene therapy. Gene Ther. 1994, 1, 367–384. [Google Scholar] [PubMed]
- Wu, Z.; Yang, H.; Colosi, P. Effect of genome size on AAV vector packaging. Mol. Ther. 2010, 18, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Acland, G.M.; Aguirre, G.D.; Ray, J.; Zhang, Q.; Aleman, T.S.; Cideciyan, A.V.; Pearce-Kelling, S.E.; Anand, V.; Zeng, Y.; Maguire, A.M.; et al. Gene therapy restores vision in a canine model of childhood blindness. Nat. Genet. 2001, 28, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, W.; Dai, X.; Kong, F.; Zheng, Q.; Zhou, X.; Lu, F.; Chang, B.; Rohrer, B.; Hauswirth, W.W.; et al. Gene therapy rescues cone structure and function in the 3-month-old rd12 mouse, a model for midcourse RPE65 leber congenital amaurosis. Invest. Ophthalmol. Vis. Sci. 2011, 52, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Beltran, W.A.; Cideciyan, A.V.; Lewin, A.S.; Iwabe, S.; Khanna, H.; Sumaroka, A.; Chiodo, V.A.; Fajardo, D.S.; Roman, A.J.; Deng, W.T.; et al. Gene therapy rescues photoreceptor blindness in dogs and paves the way for treating human X-linked retinitis pigmentosa. Proc. Natl. Acad. Sci USA 2012, 109, 2132–2137. [Google Scholar] [CrossRef] [PubMed]
- Cepko, C.L. Emerging gene therapies for retinal degenerations. J. Neurosci. 2012, 32, 6415–6420. [Google Scholar] [CrossRef] [PubMed]
- Byrne, L.C.; Dalkara, D.; Luna, G.; Fisher, S.K.; Clerin, E.; Sahel, J.A.; Leveillard, T.; Flannery, J.G. Viral-mediated RdCVF and RdCVFL expression protects cone and rod photoreceptors in retinal degeneration. J. Clin. Invest. 2015, 125, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Park, K. Cornea-targeted gene therapy using adenovirus vector. J. Control. Release 2014, 181. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, M.U.; Yla-Herttuala, S.; Airenne, K.J. How to avoid complement attack in baculovirus-mediated gene delivery. J. Invertebr. Pathol. 2011, 107 Suppl, S71–S79. [Google Scholar] [CrossRef] [PubMed]
- Luz-Madrigal, A.; Clapp, C.; Aranda, J.; Vaca, L. In vivo transcriptional targeting into the retinal vasculature using recombinant baculovirus carrying the human flt-1 promoter. Virol. J. 2007, 4. [Google Scholar] [CrossRef] [PubMed]
- Bemelmans, A.P.; Kostic, C.; Crippa, S.V.; Hauswirth, W.W.; Lem, J.; Munier, F.L.; Seeliger, M.W.; Wenzel, A.; Arsenijevic, Y. Lentiviral gene transfer of RPE65 rescues survival and function of cones in a mouse model of Leber congenital amaurosis. PLoS. Med. 2006, 3, e347. [Google Scholar] [CrossRef] [PubMed]
- Vadlapudi, A.D.; Mitra, A.K. Nanomicelles, an emerging platform for drug delivery to the eye. Ther. Deliv. 2013, 4, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Vadlapudi, A.D.; Vadlapatla, R.K.; Earla, R.; Sirimulla, S.; Bailey, J.B.; Pal, D.; Mitra, A.K. Novel biotinylated lipid prodrugs of acyclovir for the treatment of herpetic keratitis (HK), transporter recognition, tissue stability and antiviral activity. Pharm. Res. 2013, 30, 2063–2076. [Google Scholar] [CrossRef] [PubMed]
- Vaishya, R.D.; Gokulgandhi, M.; Patel, S.; Minocha, M.; Mitra, A.K. Novel dexamethasone-loaded nanomicelles for the intermediate and posterior segment uveitis. AAPS PharmSciTech. 2014, 15, 1238–1251. [Google Scholar] [CrossRef] [PubMed]
- Gaudana, R.; Ananthula, H.K.; Parenky, A.; Mitra, A.K. Ocular drug delivery. AAPS. J. 2010, 12, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Del Pozo-Rodriguez, A.; Delgado, D.; Gascon, A.R.; Solinis, M.A. Lipid nanoparticles as drug/gene delivery systems to the retina. J. Ocul. Pharmacol. Ther. 2013, 29, 173–188. [Google Scholar] [CrossRef] [PubMed]
- Paphadjopoulos, D.; Wilson, T.; Taber, R. Liposomes as vehicles for cellular incorporation of biologically active macromolecules. In Vitro 1980, 16, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Sessa, G.; Weissmann, G. Phospholipid spherules (liposomes) as a model for biological membranes. J. Lipid Res. 1968, 9, 310–318. [Google Scholar] [PubMed]
- Torchilin, V.P. Multifunctional nanocarriers. Adv. Drug Deliv. Rev. 2006, 58, 1532–1555. [Google Scholar] [CrossRef] [PubMed]
- Hamley, I.W. Nanotechnology with soft materials. Angew. Chem Int. Ed. Engl. 2003, 42, 1692–1712. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.K.; Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Noble metals on the nanoscale, optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem Res. 2008, 41, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Immordino, M.L.; Dosio, F.; Cattel, L. Stealth liposomes, review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomedicine. 2006, 1, 297–315. [Google Scholar] [PubMed]
- Honda, M.; Asai, T.; Oku, N.; Araki, Y.; Tanaka, M.; Ebihara, N. Liposomes and nanotechnology in drug development, focus on ocular targets. Int. J. Nanomed. 2013, 8, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Berestein, G.; Mehta, R.; Hopfer, R.; Mehta, K.; Hersh, E.M.; Juliano, R. Effects of sterols on the therapeutic efficacy of liposomal amphotericin B in murine candidiasis. Cancer Drug Deliv. 1983, 1, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Van Rooijen, N.; van Nieuwmegen, R. Liposomes in immunology, multilamellar phosphatidylcholine liposomes as a simple, biodegradable and harmless adjuvant without any immunogenic activity of its own. Immunol. Commun. 1980, 9, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Schnyder, A.; Huwyler, J. Drug transport to brain with targeted liposomes. NeuroRx. 2005, 2, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Oberle, V.; Bakowsky, U.; Zuhorn, I.S.; Hoekstra, D. Lipoplex formation under equilibrium conditions reveals a three-step mechanism. Biophys. J. 2000, 79, 1447–1454. [Google Scholar] [CrossRef]
- Bragonzi, A.; Dina, G.; Villa, A.; Calori, G.; Biffi, A.; Bordignon, C.; Assael, B.M.; Conese, M. Biodistribution and transgene expression with nonviral cationic vector/DNA complexes in the lungs. Gene Ther. 2000, 7, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Kollen, W.J.; Mulberg, A.E.; Wei, X.; Sugita, M.; Raghuram, V.; Wang, J.; Foskett, J.K.; Glick, M.C.; Scanlin, T.F. High-efficiency transfer of cystic fibrosis transmembrane conductance regulator cDNA into cystic fibrosis airway cells in culture using lactosylated polylysine as a vector. Hum. Gene Ther. 1999, 10, 615–622. [Google Scholar] [CrossRef] [PubMed]
- El-Aneed, A. An overview of current delivery systems in cancer gene therapy. J. Control. Release 2004, 94, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Huang, L. In vivo gene transfer via intravenous administration of cationic lipid-protamine-DNA (LPD) complexes. Gene Ther. 1997, 4, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Rizzo, M.A.; Bhattacharya, S.; Huang, L. Characterization of cationic lipid-protamine-DNA (LPD) complexes for intravenous gene delivery. Gene Ther. 1998, 5, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Wang, D.D.; Lin, Y.; Wang, J.; Petrenko, V.; Mao, C. Synergetic Targeted Delivery of Sleeping-Beauty Transposon System to Mesenchymal Stem Cells Using LPD Nanoparticles Modified with a Phage-Displayed Targeting Peptide. Adv. Funct. Mater. 2013, 23, 1172–1181. [Google Scholar] [CrossRef] [PubMed]
- Futaki, S.; Suzuki, T.; Ohashi, W.; Yagami, T.; Tanaka, S.; Ueda, K.; Sugiura, Y. Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J. Biol. Chem. 2001, 276, 5836–5840. [Google Scholar] [CrossRef] [PubMed]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Zhu, N.; Liggitt, D.; Liu, Y.; Debs, R. Systemic gene expression after intravenous DNA delivery into adult mice. Science 1993, 261, 209–211. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, V.; Moore, A.T.; Ali, R.R.; Bainbridge, J.W. Retinal dystrophies and gene therapy. Eur. J. Pediatr. 2012, 171, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Triantafillou, G.; Hertlein, E.; Towns, W.; Stefanovski, M.; Mo, X.; Jarjoura, D.; Phelps, M.; Marcucci, G.; Lee, L.J.; et al. Milatuzumab-conjugated liposomes as targeted dexamethasone carriers for therapeutic delivery in CD74+ B-cell malignancies. Clin. Cancer Res. 2013, 19, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Gandra, N.; Wang, D.D.; Zhu, Y.; Mao, C. Virus-mimetic cytoplasm-cleavable magnetic/silica nanoclusters for enhanced gene delivery to mesenchymal stem cells. Angew. Chem Int. Ed. Engl. 2013, 52, 11278–11281. [Google Scholar] [CrossRef] [PubMed]
- Delgado, D.; del Pozo-Rodriguez, A.; Solinis, M.A.; Rodriguez-Gascon, A. Understanding the mechanism of protamine in solid lipid nanoparticle-based lipofection, the importance of the entry pathway. Eur. J. Pharm. Biopharm. 2011, 79, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Conley, S.M.; Naash, M.I. AAV and compacted DNA nanoparticles for the treatment of retinal disorders, challenges and future prospects. Invest. Ophthalmol. Vis. Sci 2011, 52, 3051–3059. [Google Scholar] [CrossRef] [PubMed]
- Day, T.P.; Byrne, L.C.; Schaffer, D.V.; Flannery, J.G. Advances in AAV vector development for gene therapy in the retina. Adv. Exp. Med. Biol. 2014, 801, 687–693. [Google Scholar] [PubMed]
- Koirala, A.; Conley, S.M.; Naash, M.I. A review of therapeutic prospects of non-viral gene therapy in the retinal pigment epithelium. Biomaterials 2013, 34, 7158–7167. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Conley, S.M.; Makkia, R.; Guo, J.; Cooper, M.J.; Naash, M.I. Comparative analysis of DNA nanoparticles and AAVs for ocular gene delivery. PLoS. On. 2012, 7, e52189. [Google Scholar] [CrossRef] [PubMed]
- Powell, S.K.; Rivera-Soto, R.; Gray, S.J. Viral expression cassette elements to enhance transgene target specificity and expression in gene therapy. Discov. Med. 2015, 19, 49–57. [Google Scholar] [PubMed]
- Pozzi, D.; Marchini, C.; Cardarelli, F.; Salomone, F.; Coppola, S.; Montani, M.; Zabaleta, M.E.; Digman, M.A.; Gratton, E.; Colapicchioni, V.; et al. Mechanistic evaluation of the transfection barriers involved in lipid-mediated gene delivery, interplay between nanostructure and composition. Biochim. Biophys. Acta 2014, 1838, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Conley, S.M.; Naash, M.I. Nanoparticles for retinal gene therapy. Prog. Retin. Eye Res. 2010, 29, 376–397. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P.; Rammohan, R.; Weissig, V.; Levchenko, T.S. TAT peptide on the surface of liposomes affords their efficient intracellular delivery even at low temperature and in the presence of metabolic inhibitors. Proc. Natl. Acad. Sci. USA 2001, 98, 8786–8791. [Google Scholar] [CrossRef] [PubMed]
- Felgner, P.L.; Gadek, T.R.; Holm, M.; Roman, R.; Chan, H.W.; Wenz, M.; Northrop, J.P.; Ringold, G.M.; Danielsen, M. Lipofection, a highly efficient, lipid-mediated DNA-transfection procedure. Proc. Natl. Acad. Sci. USA 1987, 84, 7413–7417. [Google Scholar] [CrossRef] [PubMed]
- Hoare, M.; Greiser, U.; Schu, S.; Mashayekhi, K.; Aydogan, E.; Murphy, M.; Barry, F.; Ritter, T.; O’Brien, T. Enhanced lipoplex-mediated gene expression in mesenchymal stem cells using reiterated nuclear localization sequence peptides. J. Gene Med. 2010, 12, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Becker-Hapak, M.; McAllister, S.S.; Dowdy, S.F. TAT-mediated protein transduction into mammalian cells. Methods 2001, 24, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Gump, J.M.; Dowdy, S.F. TAT transduction, the molecular mechanism and therapeutic prospects. Trends Mol. Med. 2007, 13, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Schwarze, S.R.; Ho, A.; Vocero-Akbani, A.; Dowdy, S.F. In vivo protein transduction, delivery of a biologically active protein into the mouse. Science 1999, 285, 1569–1572. [Google Scholar] [CrossRef] [PubMed]
- Schwarze, S.R.; Dowdy, S.F. In vivo protein transduction, intracellular delivery of biologically active proteins, compounds and DNA. Trends Pharmacol. Sci. 2000, 21, 45–48. [Google Scholar] [CrossRef]
- Farkhani, S.M.; Valizadeh, A.; Karami, H.; Mohammadi, S.; Sohrabi, N.; Badrzadeh, F. Cell penetrating peptides, efficient vectors for delivery of nanoparticles, nanocarriers, therapeutic and diagnostic molecules. Peptides 2014, 57, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Loewenstein, P.M. Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell 1988, 55, 1179–1188. [Google Scholar] [CrossRef]
- Green, M.; Ishino, M.; Loewenstein, P.M. Mutational analysis of HIV-1 Tat minimal domain peptides, identification of trans-dominant mutants that suppress HIV-LTR-driven gene expression. Cell 1989, 58, 215–223. [Google Scholar] [CrossRef]
- Suzuki, T.; Futaki, S.; Niwa, M.; Tanaka, S.; Ueda, K.; Sugiura, Y. Possible existence of common internalization mechanisms among arginine-rich peptides. J. Biol. Chem. 2002, 277, 2437–2443. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.H.; Niu, J.; Zhang, C.Z.; Yu, W.; Wu, J.H.; Shan, Y.H.; Wang, X.R.; Shen, Y.Q.; Mao, Z.W.; Liang, W.Q.; et al. TAT conjugated cationic noble metal nanoparticles for gene delivery to epidermal stem cells. Biomaterials 2014, 35, 5605–5618. [Google Scholar] [CrossRef] [PubMed]
- Redmond, T.M.; Yu, S.; Lee, E.; Bok, D.; Hamasaki, D.; Chen, N.; Goletz, P.; Ma, J.X.; Crouch, R.K.; Pfeifer, K. Rpe65 is necessary for production of 11-cis-vitamin A in the retinal visual cycle. Nat. Genet. 1998, 20, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Maguire, A.M.; Simonelli, F.; Pierce, E.A.; Pugh, E.N., Jr.; Mingozzi, F.; Bennicelli, J.; Banfi, S.; Marshall, K.A.; Testa, F.; Surace, E.M.; et al. Safety and efficacy of gene transfer for Leber’s congenital amaurosis. N. Engl. J. Med. 2008, 358, 2240–2248. [Google Scholar] [CrossRef] [PubMed]
- Cideciyan, A.V.; Aleman, T.S.; Boye, S.L.; Schwartz, S.B.; Kaushal, S.; Roman, A.J.; Pang, J.J.; Sumaroka, A.; Windsor, E.A.; Wilson, J.M.; et al. Human gene therapy for RPE65 isomerase deficiency activates the retinoid cycle of vision but with slow rod kinetics. Proc. Natl. Acad. Sci USA 2008, 105, 15112–15117. [Google Scholar] [CrossRef] [PubMed]
- Bainbridge, J.W.; Smith, A.J.; Barker, S.S.; Robbie, S.; Henderson, R.; Balaggan, K.; Viswanathan, A.; Holder, G.E.; Stockman, A.; et al. Effect of gene therapy on visual function in Leber’s congenital amaurosis. N. Engl. J. Med. 2008, 358, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Bemelmans, A.P.; Kostic, C.; Hornfeld, D.; Jaquet, M.; Crippa, S.V.; Hauswirth, W.W.; Lem, J.; Wang, Z.; Schorderet, D.E.; Munier, F.L.; et al. Lentiviral vectors containing a retinal pigment epithelium specific promoter for leber congenital amaurosis gene therapy. Lentiviral gene therapy for LCA. Adv. Exp. Med. Biol 2006, 572, 247–253. [Google Scholar] [PubMed]
- Lai, C.M.; Yu, M.J.; Brankov, M.; Barnett, N.L.; Zhou, X.; Redmond, T.M.; Narfstrom, K.; Rakoczy, P.E. Recombinant adeno-associated virus type 2-mediated gene delivery into the Rpe65−/− knockout mouse eye results in limited rescue. Genet. Vaccines. Ther. 2004, 2. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Moiseyev, G.; Takahashi, Y.; Ma, J.X. RPE65 gene delivery restores isomerohydrolase activity and prevents early cone loss in Rpe65−/− mice. Invest. Ophthalmol. Vis. Sci. 2006, 47, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.R. Gene therapy for retinal dystrophies, twenty years in the making. Hum. Gene Ther. 2012, 23, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Stein, L.; Roy, K.; Lei, L.; Kaushal, S. Clinical gene therapy for the treatment of RPE65-associated Leber congenital amaurosis. Expert. Opin. Biol. Ther. 2011, 11, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Mowat, F.M.; Breuwer, A.R.; Bartoe, J.T.; Annear, M.J.; Zhang, Z.; Smith, A.J.; Bainbridge, J.W.; Petersen-Jones, S.M.; Ali, R.R. RPE65 gene therapy slows cone loss in Rpe65-deficient dogs. Gene Ther. 2013, 20, 545–555. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Rajala, A.; Rajala, R.V.S. Lipid Nanoparticles for Ocular Gene Delivery. J. Funct. Biomater. 2015, 6, 379-394. https://doi.org/10.3390/jfb6020379
Wang Y, Rajala A, Rajala RVS. Lipid Nanoparticles for Ocular Gene Delivery. Journal of Functional Biomaterials. 2015; 6(2):379-394. https://doi.org/10.3390/jfb6020379
Chicago/Turabian StyleWang, Yuhong, Ammaji Rajala, and Raju V. S. Rajala. 2015. "Lipid Nanoparticles for Ocular Gene Delivery" Journal of Functional Biomaterials 6, no. 2: 379-394. https://doi.org/10.3390/jfb6020379
APA StyleWang, Y., Rajala, A., & Rajala, R. V. S. (2015). Lipid Nanoparticles for Ocular Gene Delivery. Journal of Functional Biomaterials, 6(2), 379-394. https://doi.org/10.3390/jfb6020379






