Synthesis and Characterization of Fatty Acid/Amino Acid Self-Assemblies
Abstract
:1. Introduction
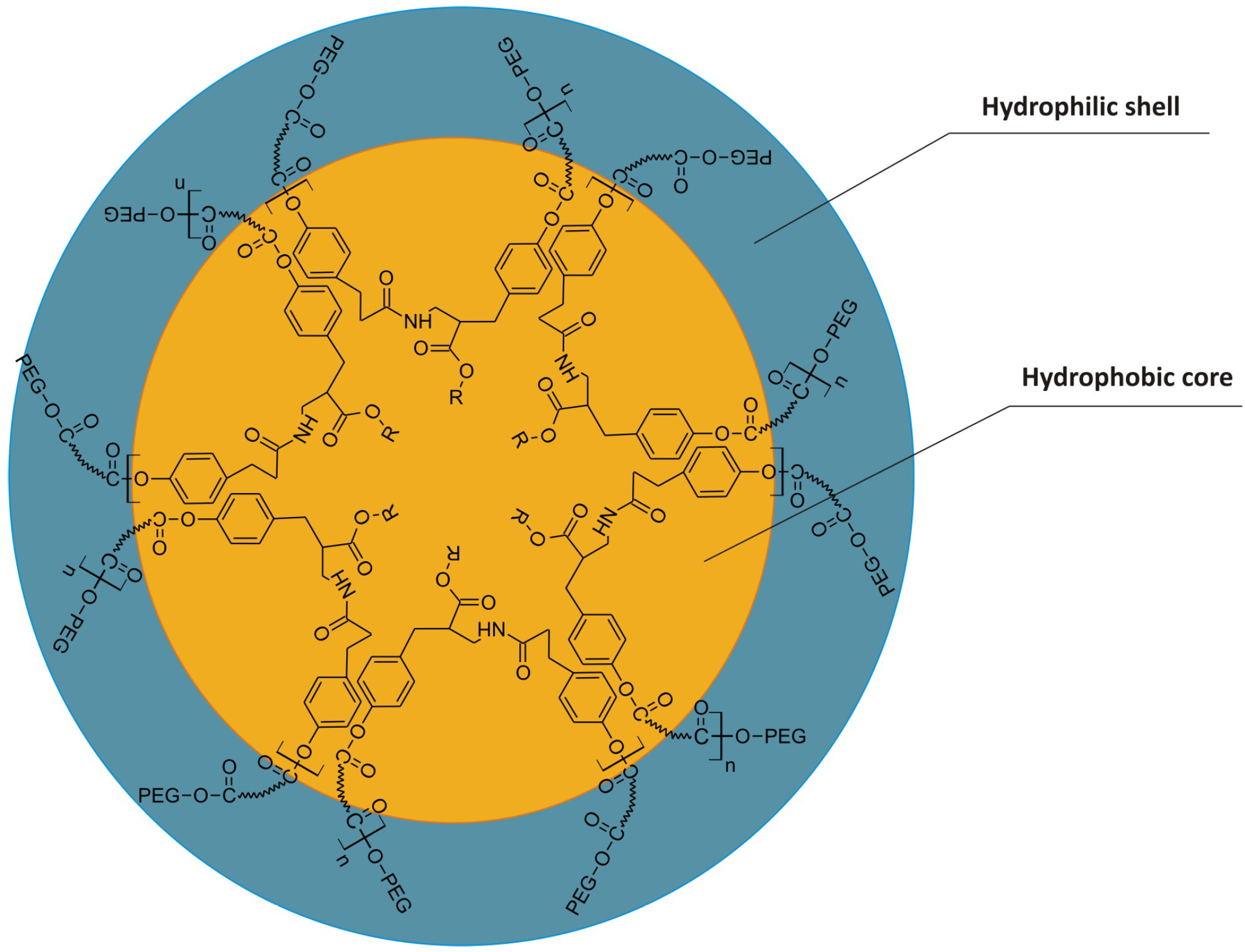
2. Experimental Section
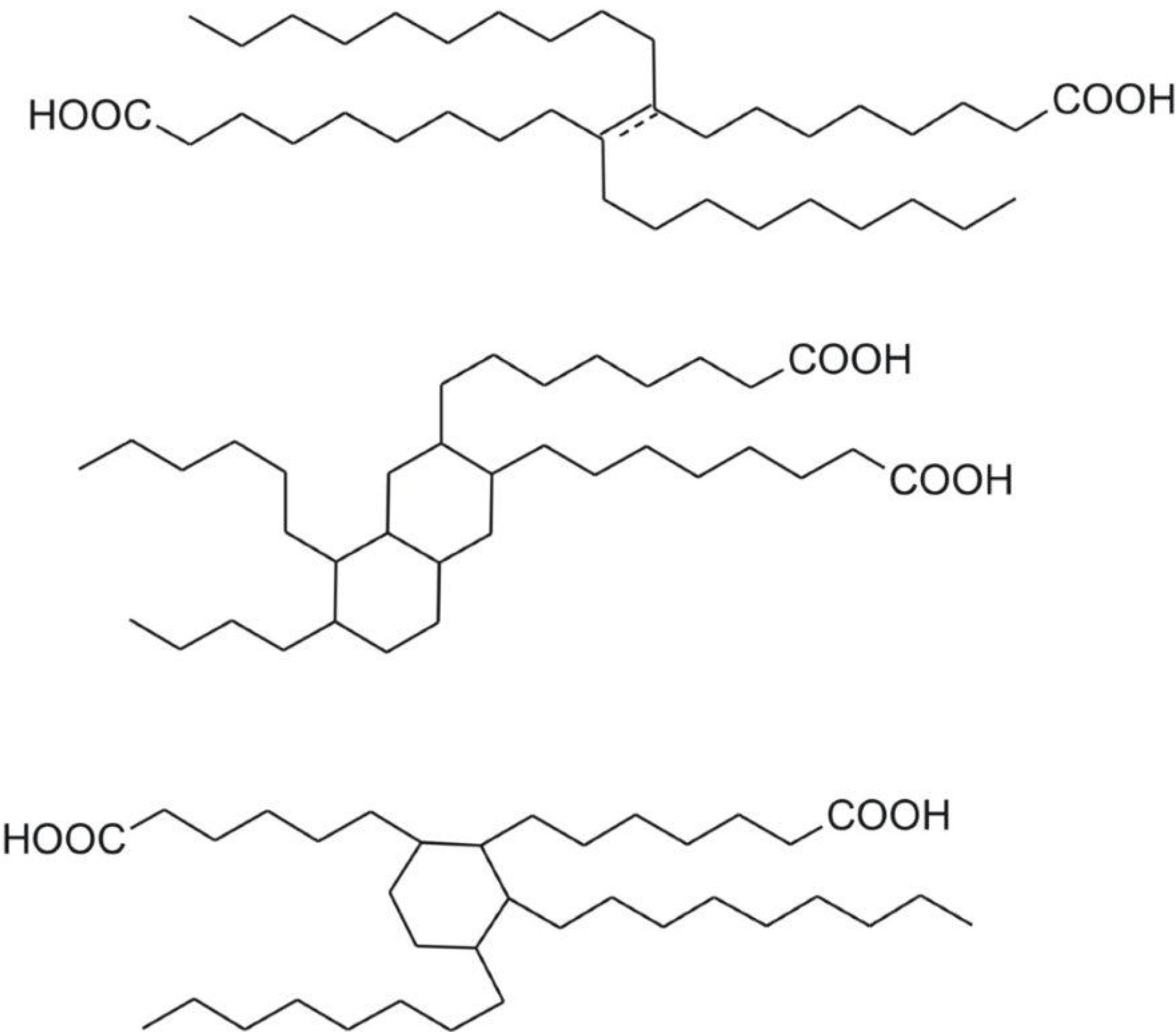
2.1. Materials
| Polymer | Mn(Da) | Mw (Da) | Dispersity index |
|---|---|---|---|
| DTE_DLA_mPEG5000 | 38,700 | 83,300 | 2.15 |
| DTE_DLA_PEG6000 | 35,400 | 66,300 | 1.87 |
| DTH_DLA_mPEG5000 | 21,400 | 48,800 | 2.28 |
| DTH_DLA_PEG6000 | 17,100 | 40,000 | 2.32 |
2.2. Synthesis
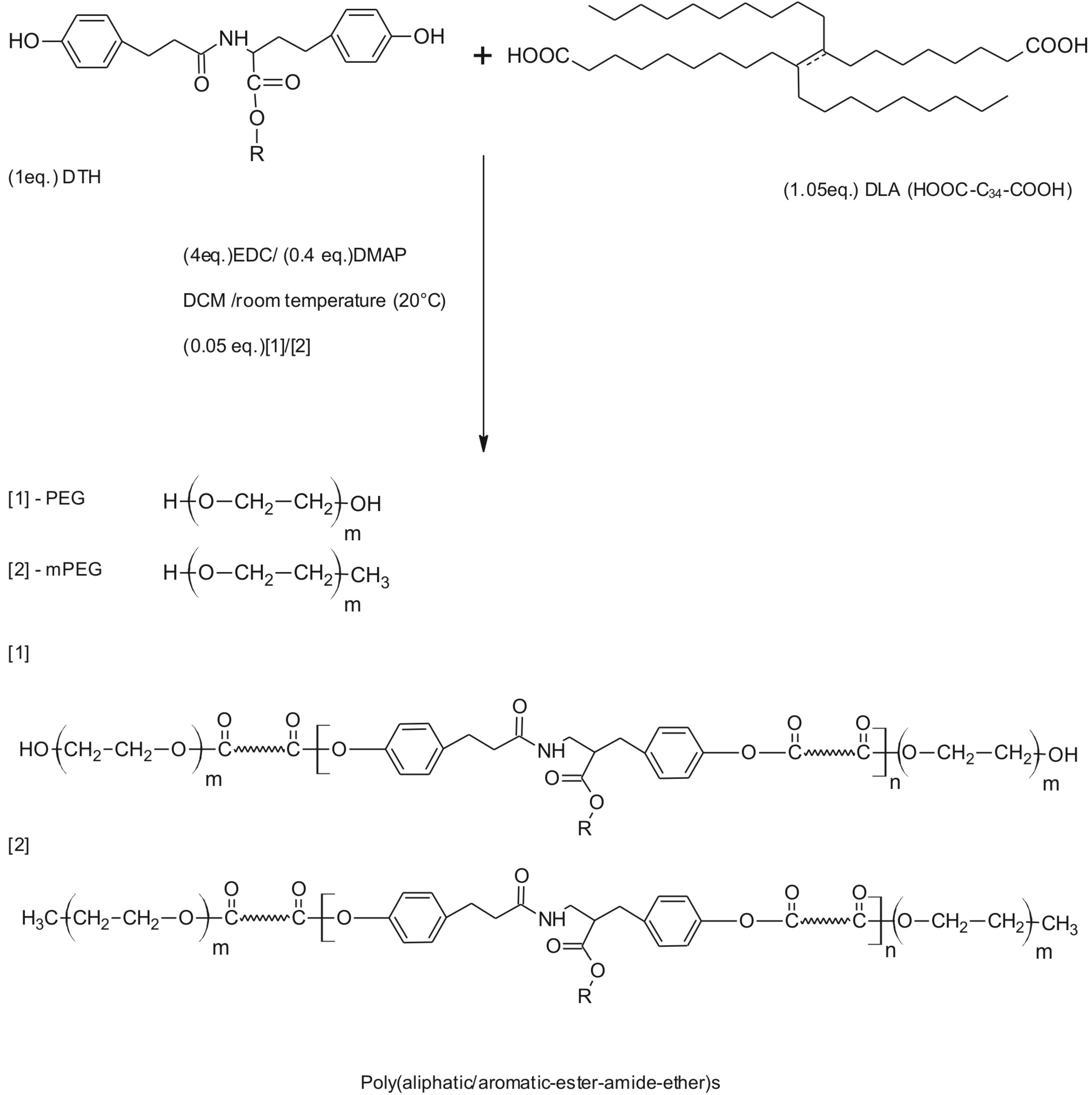
2.3. Preparation of Nanospheres
2.4. Characterization Methods
2.4.1. FTIR/1H NMR
2.4.2. Gel Permeation Chromatography (GPC)
2.4.3. UV-Vis for CMC Estimation
2.4.4. Dynamic Light Scattering (DLS)
2.4.5. TEM
3. Results and Discussion
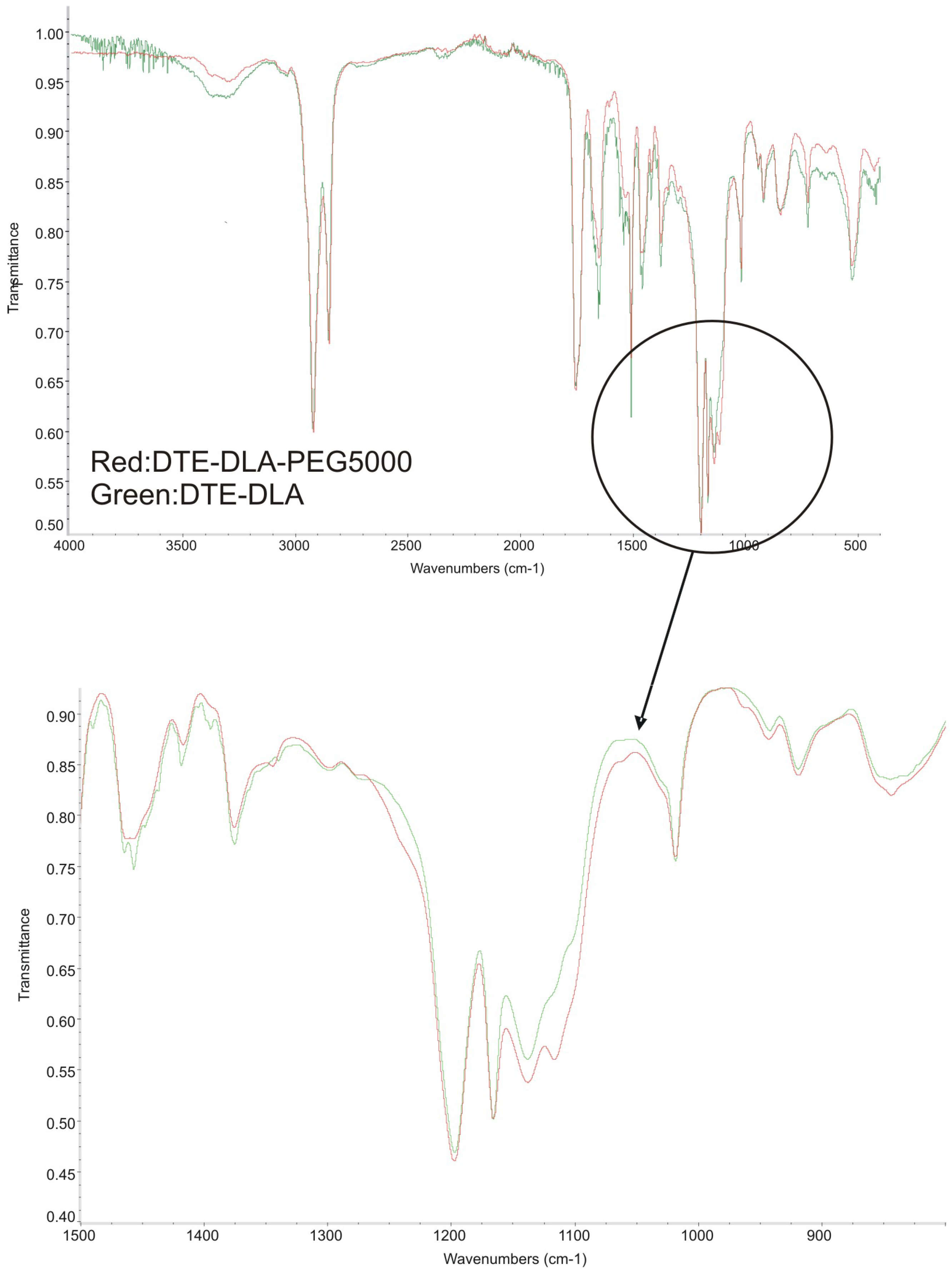
3.1. Infrared Spectroscopy
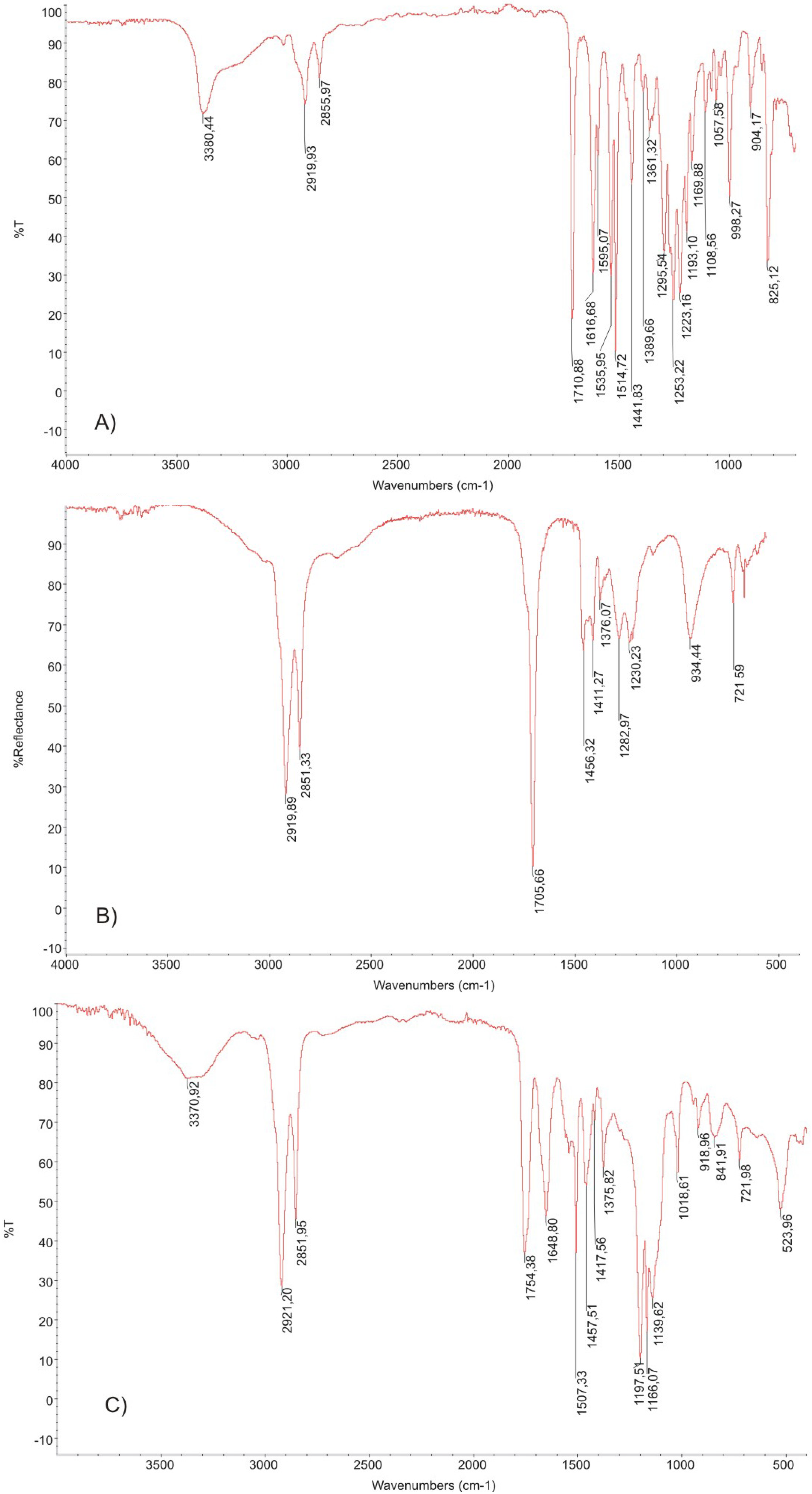
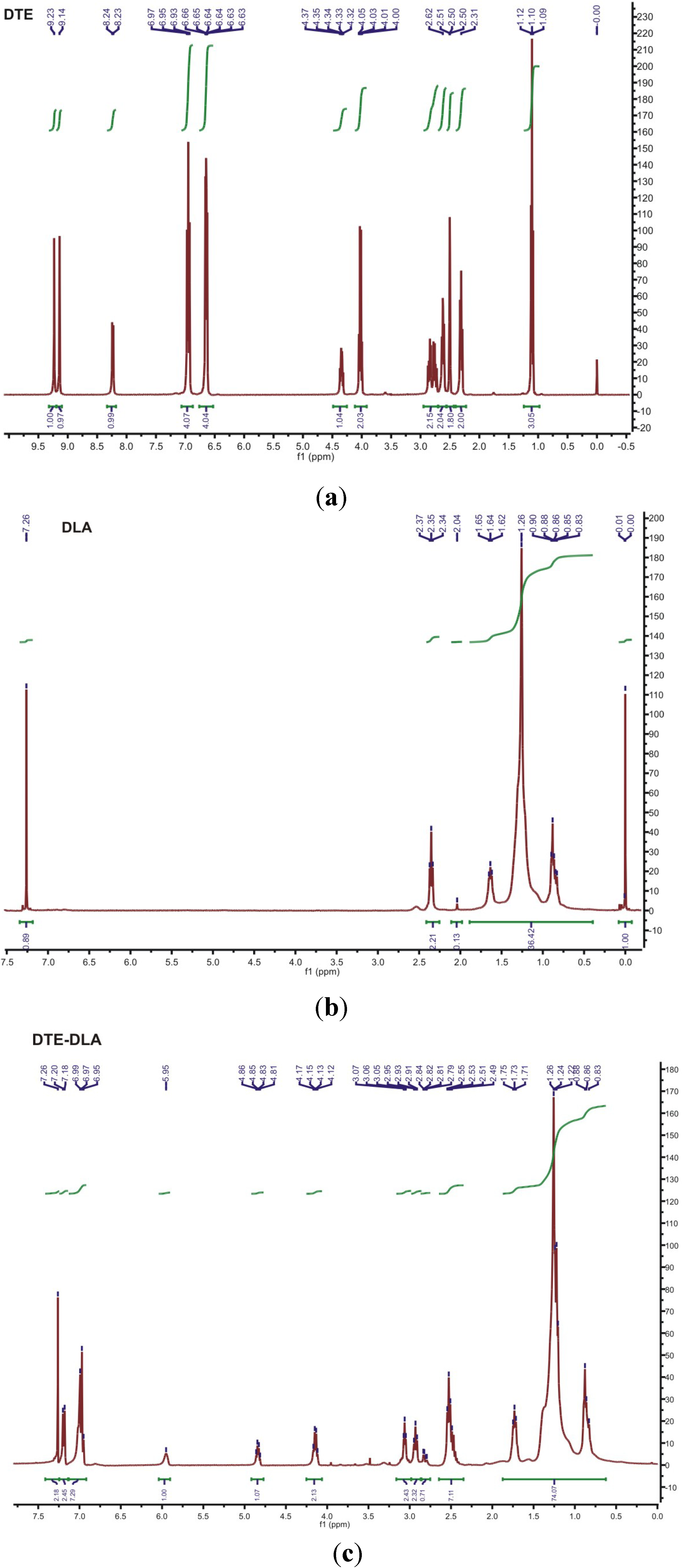
3.2. 1H NMR
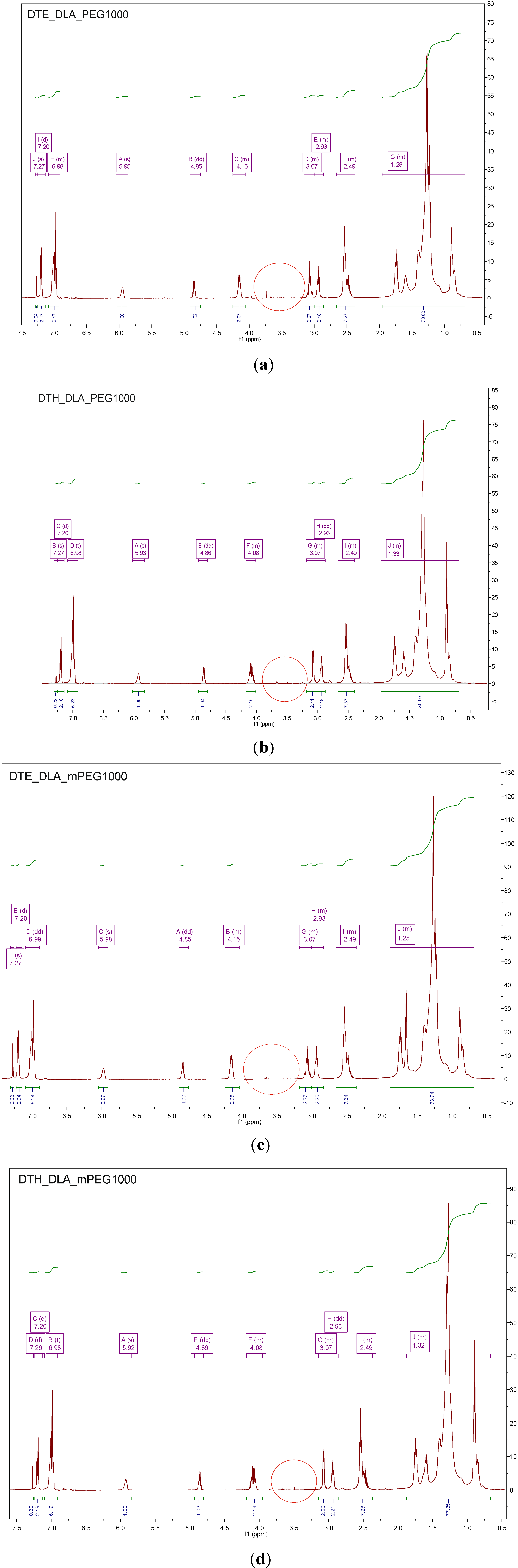
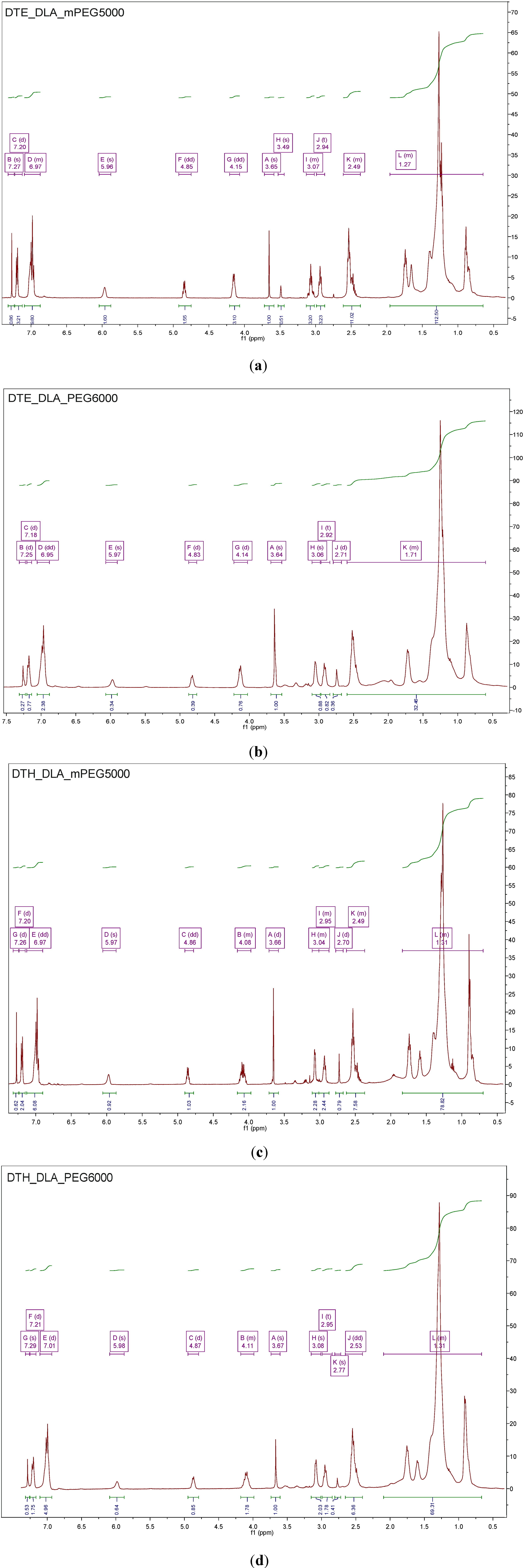
3.3. GPC
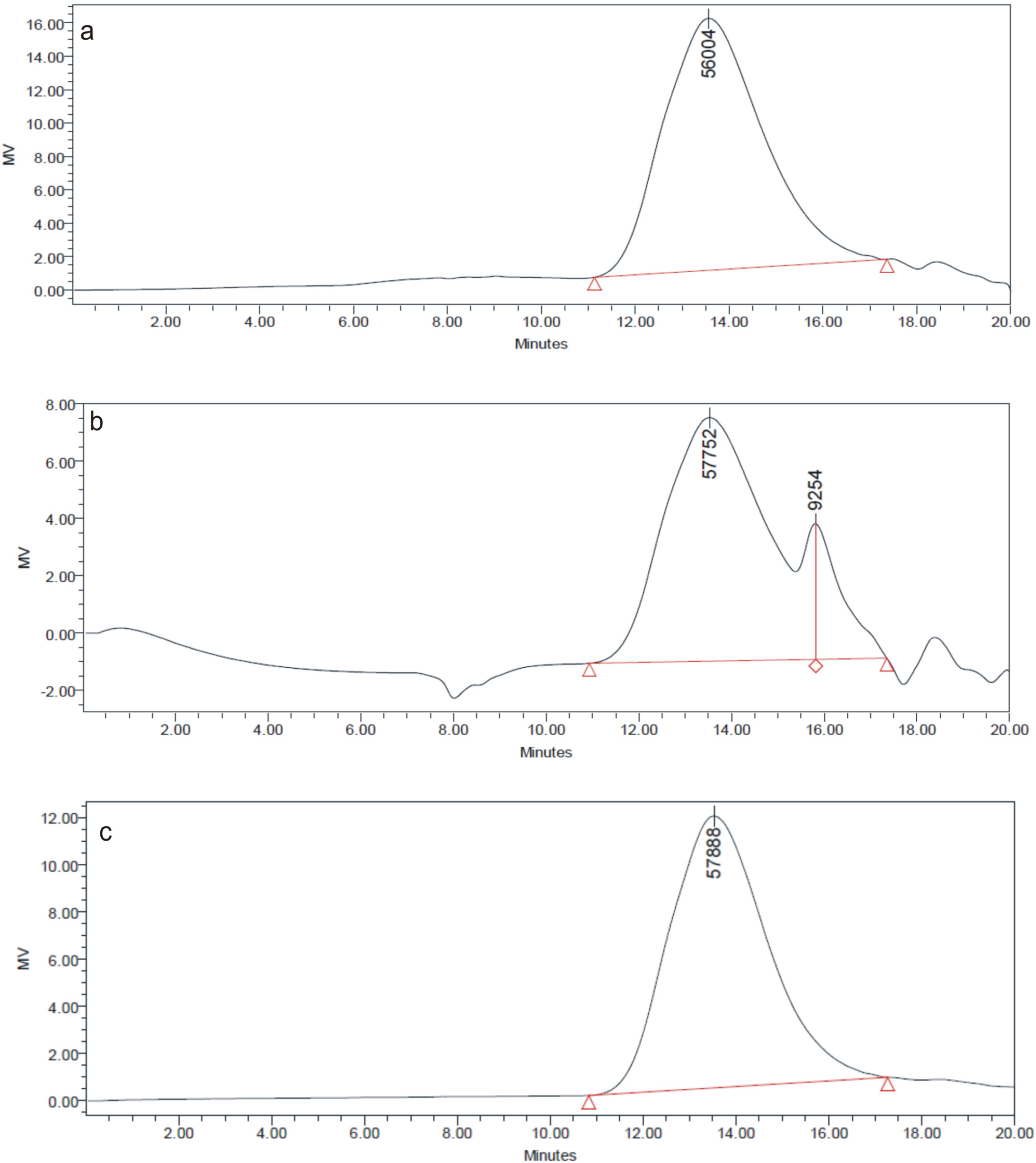
3.4. UV-Vis
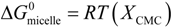
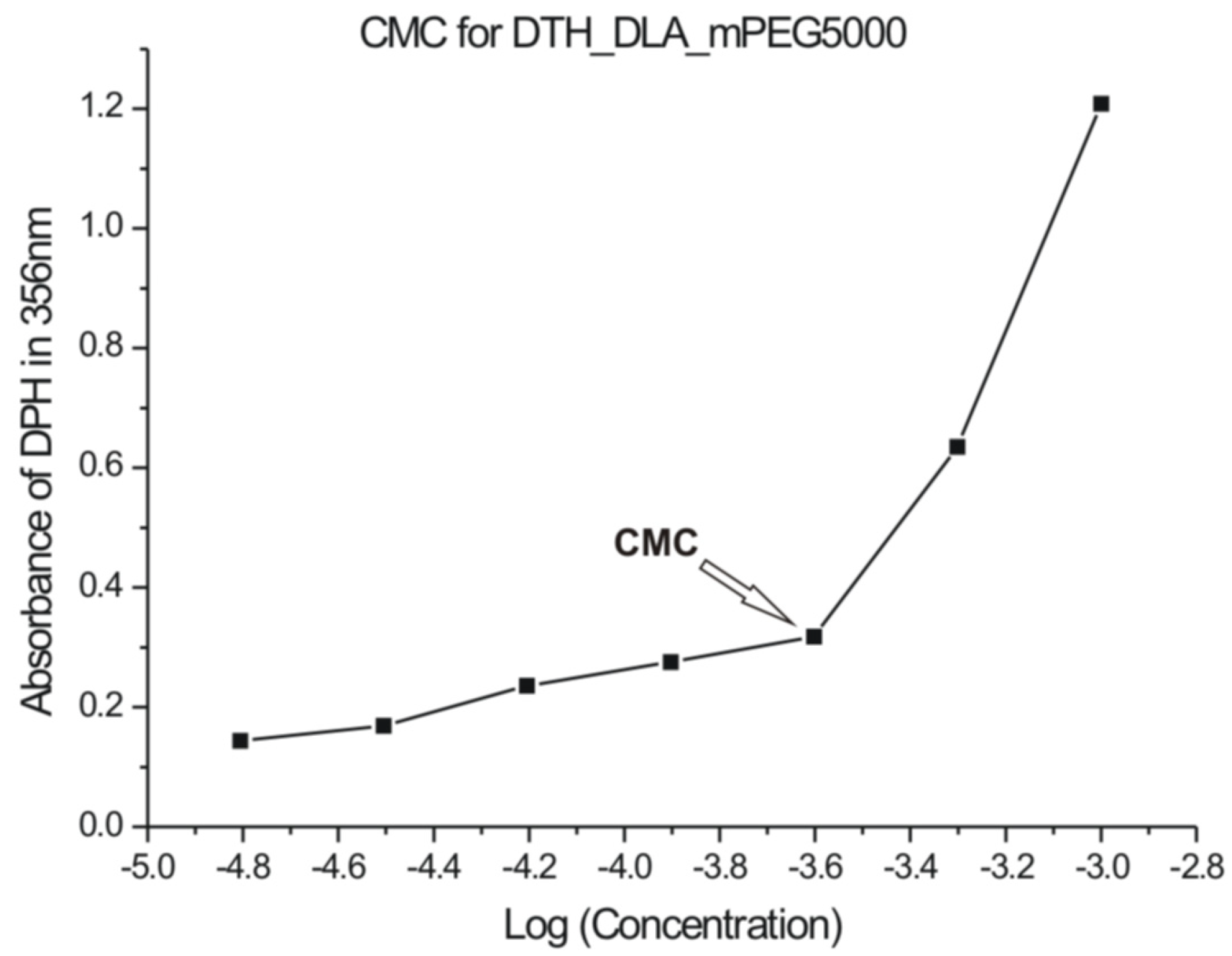
| Polymer | a CMC (g/mL) | CMC (mol fr) | Mn (Da) |  (kJ/mol) (kJ/mol) |
|---|---|---|---|---|
| DTE_DLA_mPEG5000 | 1.25 × 10−4 | 3.23 × 10−9 | 38,700 | −41.3 |
| DTE_DLA_PEG6000 | 1.25 × 10−4 | 3.53 × 10−9 | 35,400 | −41.1 |
| DTH_DLA_mPEG5000 | 2.50 × 10−4 | 1.17 × 10−8 | 21,400 | −38.1 |
| DTH_DLA_PEG6000 | 2.50 × 10−4 | 1.46 × 10−8 | 17,100 | −37.6 |
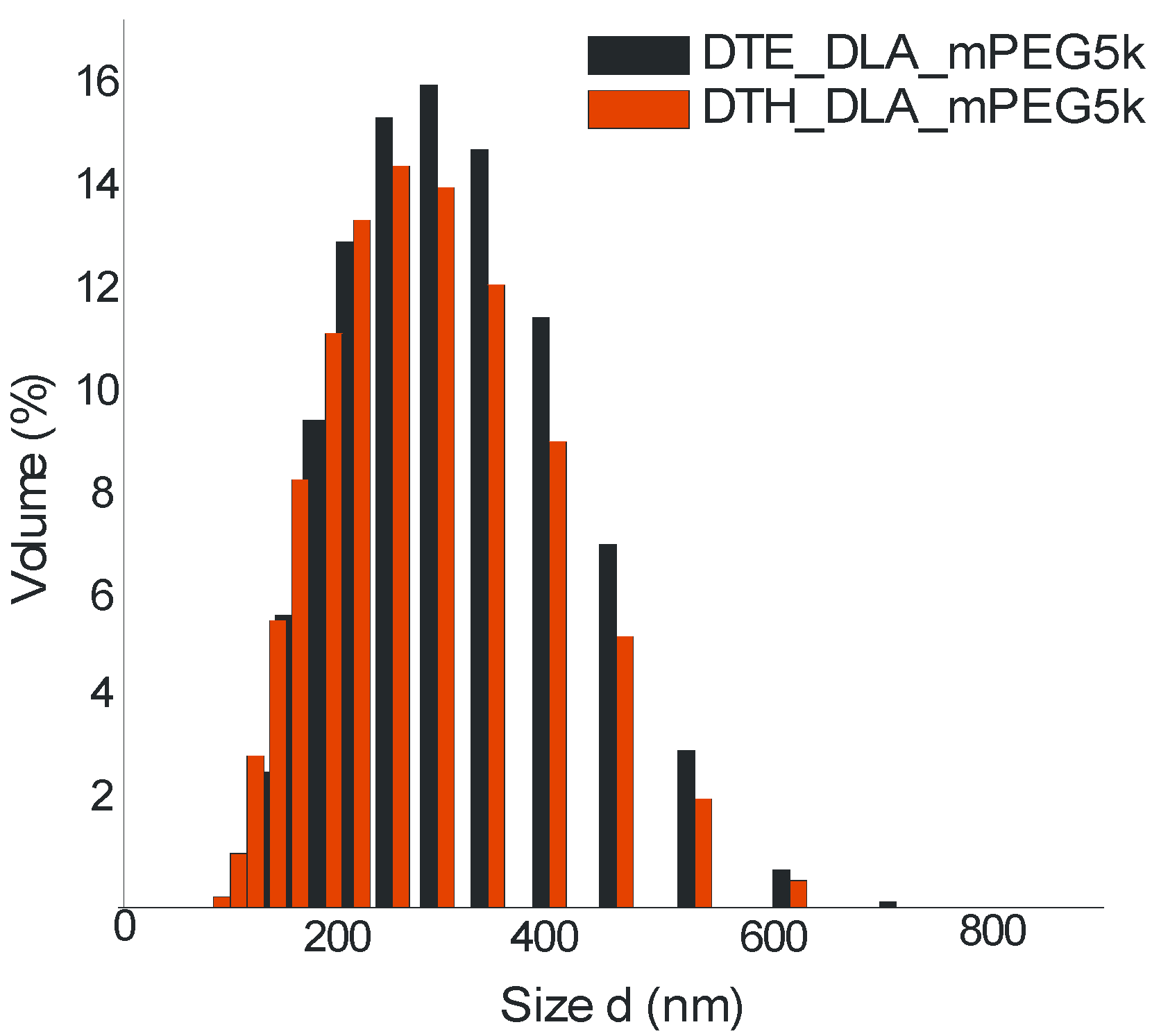
3.5. Dynamic Light Scattering (DLS)
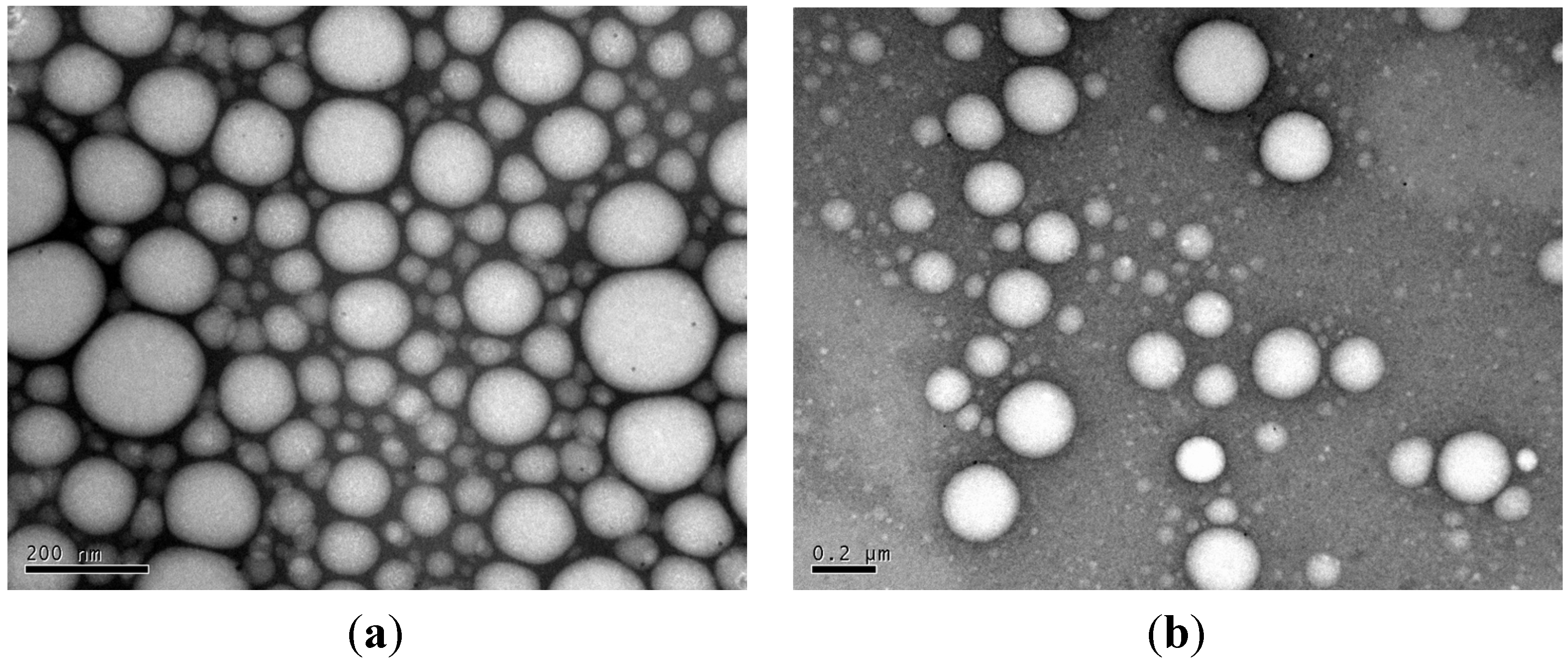
3.6. TEM
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.-X.; Robertson, J.L.; Spillman, J.; Claus, R.O. Effects of the chemical structure and the surface properties of polymeric biomaterials on their biocompatibility. Pharm. Res. 2004, 21, 1362–1373. [Google Scholar] [CrossRef]
- Gilmore, B.F.; Gorman, S.P. Antimicrobial devices. In Russell, Hugo & Ayliffe’s: Principles and Practice of Disinfection, Preservation and Sterilization, 5th ed.; Fraise, A.P., Maillard, J.-Y., Sattar, S.A., Eds.; Wiley: Hoboken, NJ, USA, 2012; pp. 500–513. [Google Scholar]
- Zhou, X.; Zhang, T.; Jiang, X.; Gu, N. The surface modification of medical polyurethane to improve the hydrophilicity and lubricity: The effect of pretreatment. J. Appl. Polym. Sci. 2010, 116, 1284–1290. [Google Scholar]
- Hoffman, A.S. Surface modification of polymers: Physical, chemical, mechanical and biological methods. Macromol. Symp. 1996, 101, 443–454. [Google Scholar] [CrossRef]
- He, C.; Wang, M.; Cai, X.; Huang, X.; Li, L.; Zhu, H.; Shen, J.; Yuan, J. Chemically induced graft copolymerization of 2-hydroxyethyl methacrylate onto polyurethane surface for improving blood compatibility. Appl. Surf. Sci. 2011, 258, 755–760. [Google Scholar] [CrossRef]
- Kyomoto, M.; Moro, T.; Miyaji, F.; Hashimoto, M.; Kawaguchi, H.; Takatori, Y.; Nakamura, K.; Ishihara, K. Effect of 2-methacryloyloxyethyl phosphorylcholine concentration on photo-induced graft polymerization of polyethylene in reducing the wear of orthopaedic bearing surface. J. Biomed. Mater. Res. A 2008, 86, 439–447. [Google Scholar] [CrossRef]
- Abbasi, F.; Mirzadeh, H.; Katbab, A.A. Modification of polysiloxane polymers for biomedical applications: A review. Polym. Int. 2001, 50, 1279–1287. [Google Scholar] [CrossRef]
- George, P.A.; Donose, B.C.; Cooper-White, J.J. Self-assembling polystyrene-block-poly(ethylene oxide) copolymer surface coatings: Resistance to protein and cell adhesion. Biomaterials 2009, 30, 2449–2456. [Google Scholar] [CrossRef]
- Fukazawa, K.; Ishihara, K. Simple surface treatment using amphiphilic phospholipid polymers to obtain wetting and lubricity on polydimethylsiloxane-based substrates. Colloids Surfaces B Biointerfaces 2012, 97, 70–76. [Google Scholar] [CrossRef]
- Kato, K.; Uchida, E.; Kang, E.T.; Uyama, Y.; Ikada, Y. Polymer surface with graft chains. Prog. Polym. Sci. 2003, 28, 209–259. [Google Scholar] [CrossRef]
- Chawla, K.; Lee, S.; Lee, B.P.; Dalsin, J.L.; Messersmith, P.B.; Spencer, N.D. A novel low-friction surface for biomedical applications: modification of poly(dimethylsiloxane) (PDMS) with polyethylene glycol(PEG)-DOPA-lysine. J. Biomed. Mater. Res. A 2009, 90, 742–749. [Google Scholar] [CrossRef]
- Ho, S.P.; Nakabayashi, N.; Iwasaki, Y.; Boland, T.; LaBerge, M. Frictional properties of poly(MPC-co-BMA) phospholipid polymer for catheter applications. Biomaterials 2003, 24, 5121–5129. [Google Scholar] [CrossRef]
- Kobayashi, M.; Terayama, Y.; Hosaka, N.; Kaido, M.; Suzuki, A.; Yamada, N.; Torikai, N.; Ishihara, K.; Takahara, A. Friction behavior of high-density poly(2-methacryloyloxyethyl phosphorylcholine) brush in aqueous media. Soft Matter 2007, 3, 740–746. [Google Scholar] [CrossRef]
- Yang, S.H.; Lee, Y.S.; Lin, F.H.; Yang, J.M.; Chen, K.S. Chitosan/poly(vinyl alcohol) blending hydrogel coating improves the surface characteristics of segmented polyurethane urethral catheters. J. Biomed. Mater. Res. B Appl Biomater. 2007, 83, 304–313. [Google Scholar] [CrossRef]
- Zheng, C.J.; Yoo, J.-S.; Lee, T.-G.; Cho, H.-Y.; Kim, Y.-H.; Kim, W.-G. Fatty acid synthesis is a target for antibacterial activity of unsaturated fatty acids. FEBS Lett. 2005, 579, 5157–5162. [Google Scholar] [CrossRef]
- Bergsson, G.; Steingrímsson, Ó.; Thormar, H. Bactericidal effects of fatty acids and monoglycerides on Helicobacter pylori. Int. J. Antimicrob. Agents 2002, 20, 258–262. [Google Scholar] [CrossRef]
- Pereira, J.A.; Oliveira, I.; Sousa, A.; Ferreira, I.C.F.R.; Bento, A.; Estevinho, L. Bioactive properties and chemical composition of six walnut (Juglans regia L.) cultivars. Food Chem. Toxicol. 2008, 46, 2103–2111. [Google Scholar]
- Fukuoka, T.; Tachibana, Y.; Tonami, H.; Uyama, H.; Kobayashi, S. Enzymatic polymerization of tyrosine derivatives. Peroxidase- and protease-catalyzed synthesis of poly(tyrosine)s with different structures. Biomacromolecules 2002, 3, 768–774. [Google Scholar] [CrossRef]
- Silvia, V.; Baldisserotto, A.; Scalambra, E.; Malisardi, G.; Durini, E.; Manfredini, S. Novel molecular combination deriving from natural aminoacids and polyphenols: Design, synthesis and free-radical scavenging activities. Eur. J. Med. Chem. 2012, 50, 383–392. [Google Scholar] [CrossRef]
- Patel, M.N.; Patel, S.H.; Chhasatia, M.R.; Parmar, P.A. Five-coordinated oxovanadium(IV) complexes derived from amino acids and ciprofloxacin: Synthesis, spectral, antimicrobial, and DNA interaction approach. Bioorganic Med. Chem. Lett. 2008, 18, 6494–6500. [Google Scholar] [CrossRef]
- Bourke, S.L.; Kohn, J. Polymers derived from the amino acid l-tyrosine: Polycarbonates, polyarylates and copolymers with poly(ethylene glycol). Adv. Drug Deliv. Rev. 2003, 55, 447–466. [Google Scholar] [CrossRef]
- Kilfoyle, B.E.; Sheihet, L.; Zhang, Z.; Laohoo, M.; Kohn, J.; Michniak-Kohn, B.B. Development of paclitaxel-TyroSpheres for topical skin treatment. J Control. Release 2012, 163, 18–24. [Google Scholar] [CrossRef]
- Sheihet, L.; Dubin, R.A.; Devore, D.; Kohn, J. Hydrophobic drug delivery by self-assembling triblock copolymer-derived nanospheres. Biomacromolecules 2005, 6, 2726–2731. [Google Scholar] [CrossRef]
- Sheihet, L.; Garbuzenko, O.B.; Bushman, J.; Gounder, M.K.; Minko, T.; Kohn, J. Paclitaxel in tyrosine-derived nanospheres as a potential anti-cancer agent: In vivo evaluation of toxicity and efficacy in comparison with paclitaxel in Cremophor. Eur. J. Pharm. Sci. 2012, 45, 320–329. [Google Scholar] [CrossRef]
- Batheja, P.; Sheihet, L.; Kohn, J.; Singer, A.J.; Michniak-Kohn, B. Topical drug delivery by a polymeric nanosphere gel: Formulation optimization and in vitro and in vivo skin distribution studies. J. Control. Release 2011, 149, 159–167. [Google Scholar] [CrossRef]
- Prowans, P.; El Fray, M.; Slonecki, J. Biocompatibility studies of new multiblock poly(ester-ester)s composed of poly(butylene terephthalate) and dimerized fatty acid. Biomaterials 2002, 23, 2973–2978. [Google Scholar] [CrossRef]
- El Fray, M.; Altstädt, V. Fatigue behaviour of multiblock thermoplastic elastomers. 1. Stepwise increasing load testing of poly(aliphatic/aromatic-ester) copolymers. Polymer Guildf. 2003, 44, 4635–4642. [Google Scholar] [CrossRef]
- El Fray, M.; Altstädt, V. Fatigue behaviour of multiblock thermoplastic elastomers. 2. Dynamic creep of poly(aliphatic/aromatic-ester) copolymers. Polymer Guildf. 2003, 44, 4643–4650. [Google Scholar] [CrossRef]
- El Fray, M.; Altstädt, V. Fatigue behaviour of multiblock thermoplastic elastomers. 3. Stepwise increasing strain test of poly (aliphatic/aromatic-ester) copolymers. Polymer Guildf. 2004, 45, 263–273. [Google Scholar] [CrossRef]
- El Fray, M.; Skrobot, J.; Bolikal, D.; Kohn, J. Synthesis and characterization of telechelic macromers containing fatty acid derivatives. React. Funct. Polym. 2012, 72, 781–790. [Google Scholar]
- Sheet, S.D. PRIPOL TM Polymerised Fatty Acids; Croda Group: Gouda, The Netherlands, 2008. [Google Scholar]
- Kinard, L.A.; Kasper, F.K.; Mikos, A.G. Synthesis of oligo(poly(ethylene glycol) fumarate). Nat. Protoc. 2012, 7, 1219–1227. [Google Scholar] [CrossRef]
- MestReNova; Mestrelab Research: Santiago de Compostela, Spain, 2009.
- Empower, version 2; Waters Associates: Milford, MA, USA, 2005.
- Fiordeliso, J.; Bron, S.; Kohn, J. Design, synthesis, and preliminary characterization of tyrosine-containing polyarylates: new biomaterials for medical applications. J. Biomater. Sci. Polym. Ed. 1994, 5, 497–510. [Google Scholar] [CrossRef]
- Sheihet, L.; Piotrowska, K.; Dubin, R.A.; Kohn, J.; Devore, D. Effect of tyrosine-derived triblock copolymer compositions on nanosphere self-assembly and drug delivery. Biomacromolecules 2007, 8, 998–1003. [Google Scholar] [CrossRef]
- Lu, C.; Liu, L.; Guo, S.-R.; Zhang, Y.; Li, Z.; Gu, J. Micellization and gelation of aqueous solutions of star-shaped PEG–PCL block copolymers consisting of branched 4-arm poly(ethylene glycol) and polycaprolactone blocks. Eur. Polym. J. 2007, 43, 1857–1865. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gajowy, J.; Bolikal, D.; Kohn, J.; Fray, M.E. Synthesis and Characterization of Fatty Acid/Amino Acid Self-Assemblies. J. Funct. Biomater. 2014, 5, 211-231. https://doi.org/10.3390/jfb5040211
Gajowy J, Bolikal D, Kohn J, Fray ME. Synthesis and Characterization of Fatty Acid/Amino Acid Self-Assemblies. Journal of Functional Biomaterials. 2014; 5(4):211-231. https://doi.org/10.3390/jfb5040211
Chicago/Turabian StyleGajowy, Joanna, Durgadas Bolikal, Joachim Kohn, and Miroslawa El Fray. 2014. "Synthesis and Characterization of Fatty Acid/Amino Acid Self-Assemblies" Journal of Functional Biomaterials 5, no. 4: 211-231. https://doi.org/10.3390/jfb5040211
APA StyleGajowy, J., Bolikal, D., Kohn, J., & Fray, M. E. (2014). Synthesis and Characterization of Fatty Acid/Amino Acid Self-Assemblies. Journal of Functional Biomaterials, 5(4), 211-231. https://doi.org/10.3390/jfb5040211





