A Novel Multi-Phosphonate Surface Treatment of Titanium Dental Implants: A Study in Sheep
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Implants and Surface Characteristics
2.1.2. Surgery and Postoperative Period
2.1.3. Macroscopical and Radiological Evaluation
2.1.4. Microradiographic Evaluation
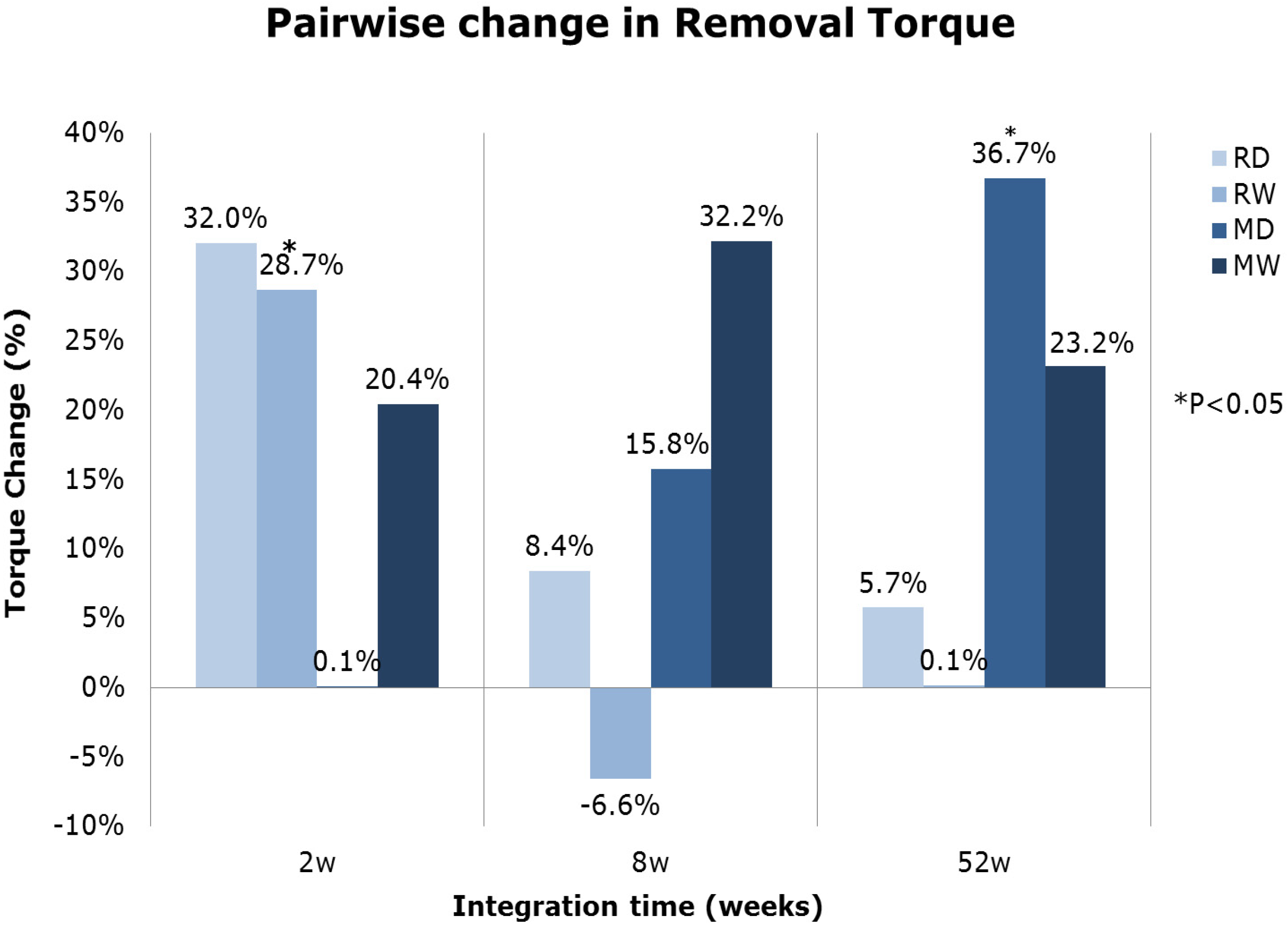
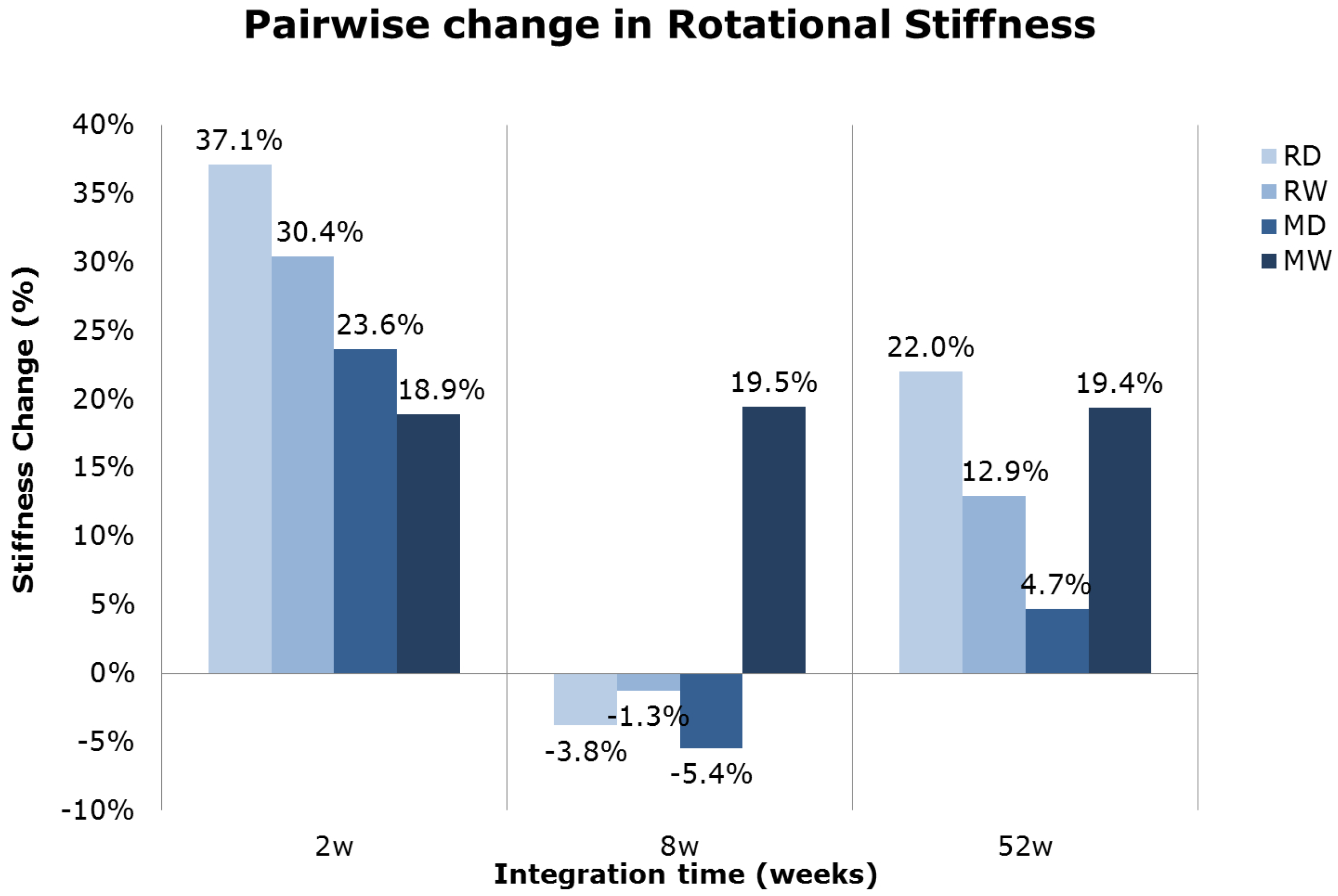
2.1.5. Torque-Test Evaluation
| Group | Removal Torque (N·mm) | Stiffness (N·mm/°) | |||
|---|---|---|---|---|---|
| Average | SE | Average | SE | ||
| RD | 2 weeks | 452.3 | 29.0 | 155.5 | 9.7 |
| 8 weeks | 1055.6 | 39.2 | 180.9 | 4.5 | |
| 52 weeks | 1286.9 | 71.0 | 155.5 | 12.5 | |
| RW | 2 weeks | 516.5 | 46.1 | 155.9 | 9.1 |
| 8 weeks | 920.4 | 73.2 | 186.7 | 10.1 | |
| 52 weeks | 1246.4 | 103.6 | 156.3 | 17.5 | |
| RC | 2 weeks | 450.8 | 37.8 | 140.0 | 9.1 |
| 8 weeks | 1035.0 | 45.4 | 194.2 | 6.9 | |
| 52 weeks | 1267.5 | 45.1 | 138.9 | 12.7 | |
| MD | 2 weeks | 251.5 | 35.0 | 125.6 | 17.9 |
| 8 weeks | 283.3 | 23.6 | 105.3 | 11.0 | |
| 52 weeks | 692.5 | 53.6 | 86.9 | 9.5 | |
| MW | 2 weeks | 277.4 | 31.9 | 117.4 | 13.7 |
| 8 weeks | 290.5 | 29.3 | 109.1 | 8.7 | |
| 52 weeks | 717.5 | 80.4 | 96.6 | 11.5 | |
| MC | 2 weeks | 274.4 | 22.4 | 123.5 | 14.3 |
| 8 weeks | 267.7 | 20.5 | 119.9 | 15.5 | |
| 52 weeks | 758.2 | 107.5 | 104.8 | 16.1 | |
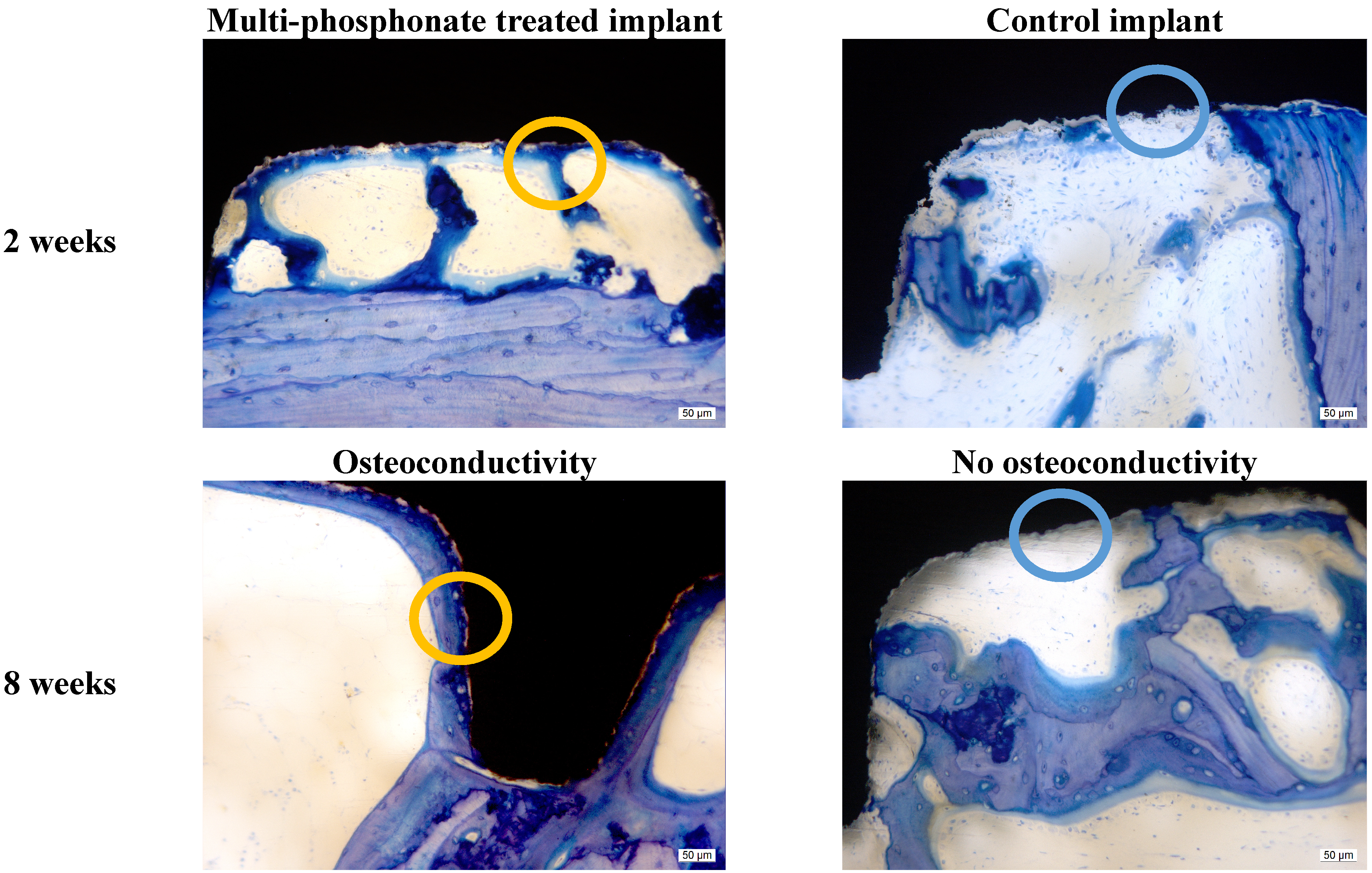
2.1.6. Histological Evaluation and BIC

| Group | Cortical | Cancellous | Total | ||||
|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | SE | ||
| RD | 2 weeks | 72.3 | 8.1 | 84.9 | 3.3 | 84.4 | 2.9 |
| 8 weeks | 61.9 | 6.1 | 72.1 | 4.5 | 66.0 | 3.6 | |
| 52 weeks | 84.0 | 4.5 | 73.9 | 7.0 | 76.4 | 4.4 | |
| RW | 2 weeks | 78.8 | 5.2 | 76.3 | 5.4 | 73.5 | 6.1 |
| 8 weeks | 63.5 | 4.6 | 76.8 | 4.0 | 71.5 | 5.4 | |
| 52 weeks | 72.3 | 7.2 | 80.6 | 5.3 | 77.5 | 3.4 | |
| RC | 2 weeks | 75.9 | 6.4 | 81.3 | 3.0 | 81.4 | 2.1 |
| 8 weeks | 65.2 | 5.9 | 71.9 | 4.5 | 68.8 | 4.7 | |
| 52 weeks | 80.7 | 4.3 | 76.1 | 5.3 | 76.0 | 3.9 | |
| MD | 2 weeks | 64.2 | 7.2 | 26.0 | 3.7 | 33.9 | 3.9 |
| 8 weeks | 44.0 | 5.7 | 27.9 | 4.5 | 32.1 | 3.6 | |
| 52 weeks | 44.5 | 5.3 | 52.2 | 4.8 | 46.6 | 4.7 | |
| MW | 2 weeks | 75.5 | 6.5 | 36.6 | 3.4 | 45.2 | 4.3 |
| 8 weeks | 47.3 | 9.0 | 33.4 | 4.8 | 37.1 | 6.6 | |
| 52 weeks | 45.5 | 7.6 | 50.8 | 6.5 | 47.1 | 6.0 | |
| MC | 2 weeks | 67.3 | 8.2 | 36.0 | 4.8 | 38.0 | 4.3 |
| 8 weeks | 51.7 | 6.2 | 27.2 | 4.0 | 34.4 | 4.1 | |
| 52 weeks | 42.3 | 3.7 | 53.6 | 5.4 | 48.6 | 5.2 | |
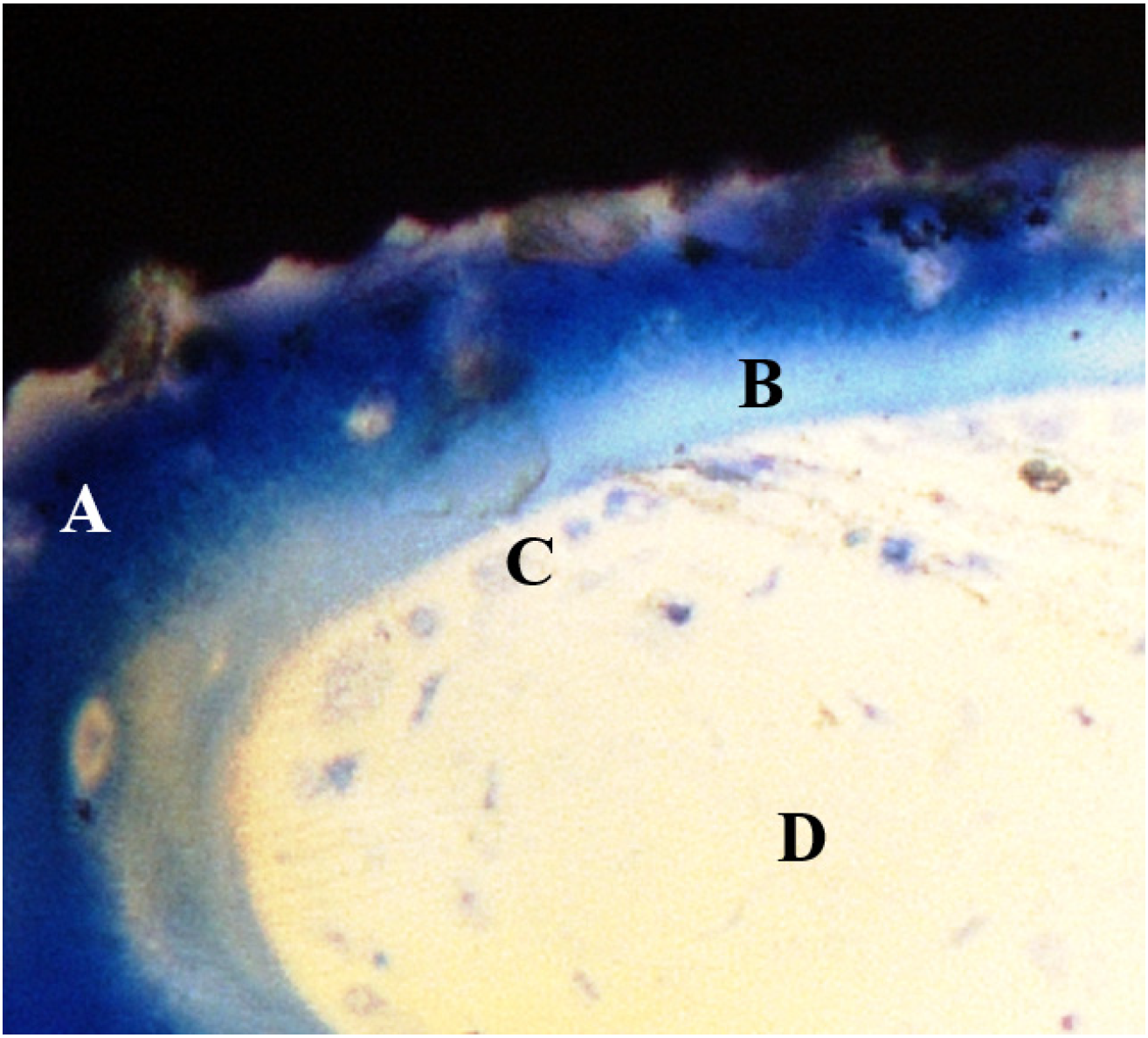
2.1.7. Histomorphometric Evaluation
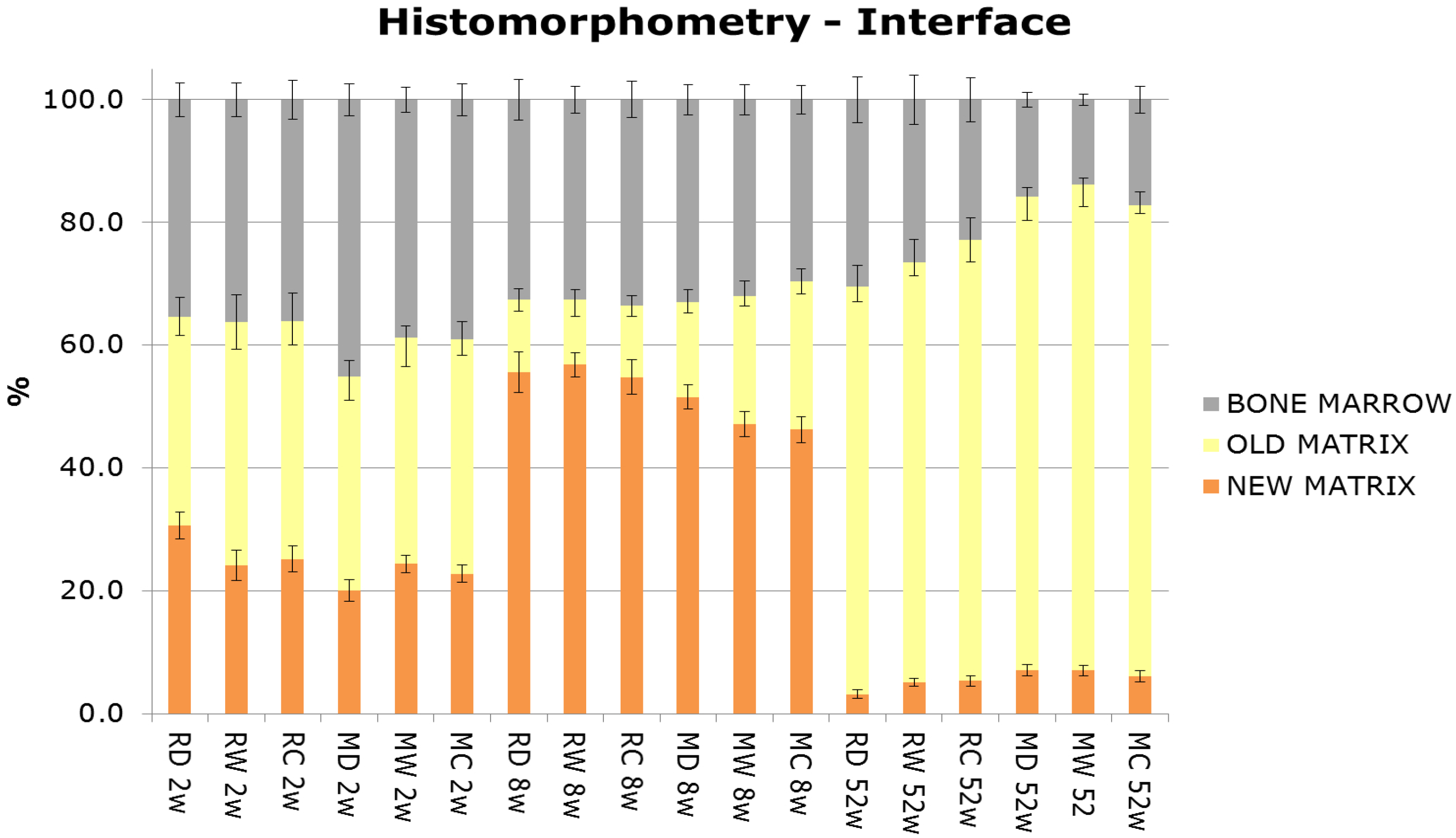
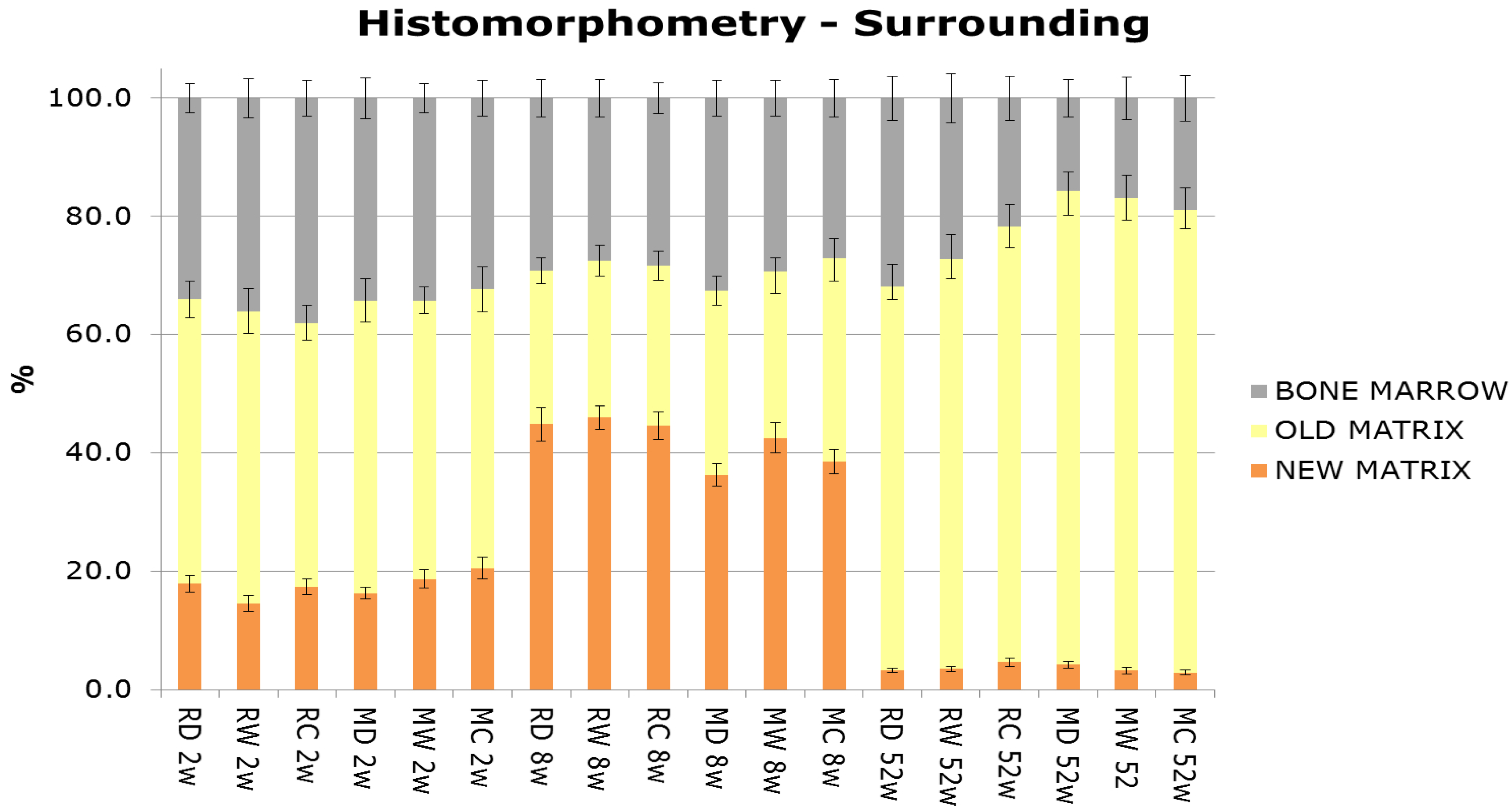
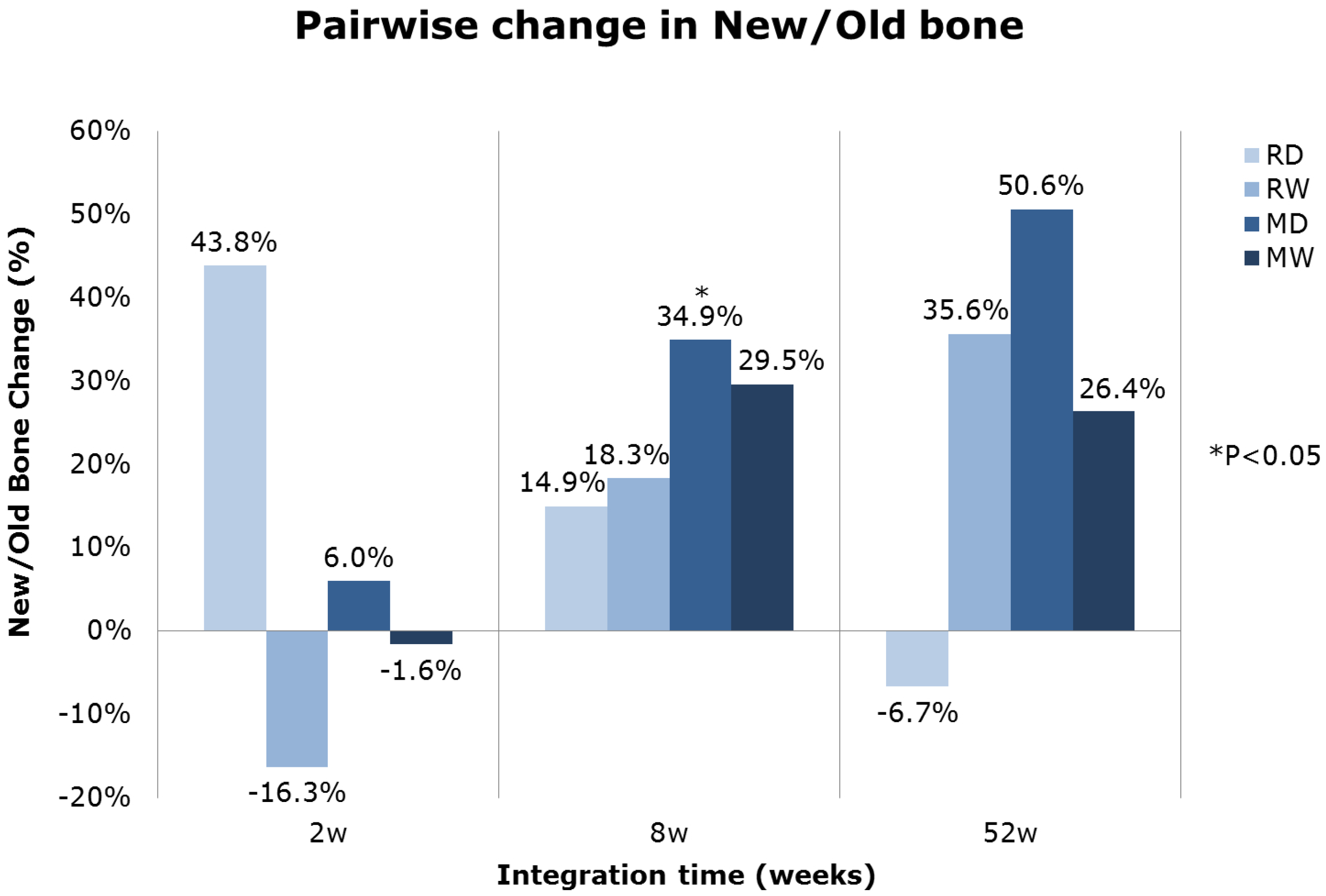
2.1.8. Scanning Electron Microscopy (SEM) Evaluation of Torque Tested Implants

2.2. Discussion
3. Experimental Section
3.1. Implants and Surface Characteristics
| Group ID | Surface | Treatment | Storage |
|---|---|---|---|
| RD | Rough | Multi-phosphonate | Dry |
| RW | Multi-phosphonate | Wet | |
| RC | Control | Dry | |
| MD | Machined | Multi-phosphonate | Dry |
| MW | Multi-phosphonate | Wet | |
| MC | Control | Dry |
3.2. Animals and Surgical Technique
3.3. Anaesthesia
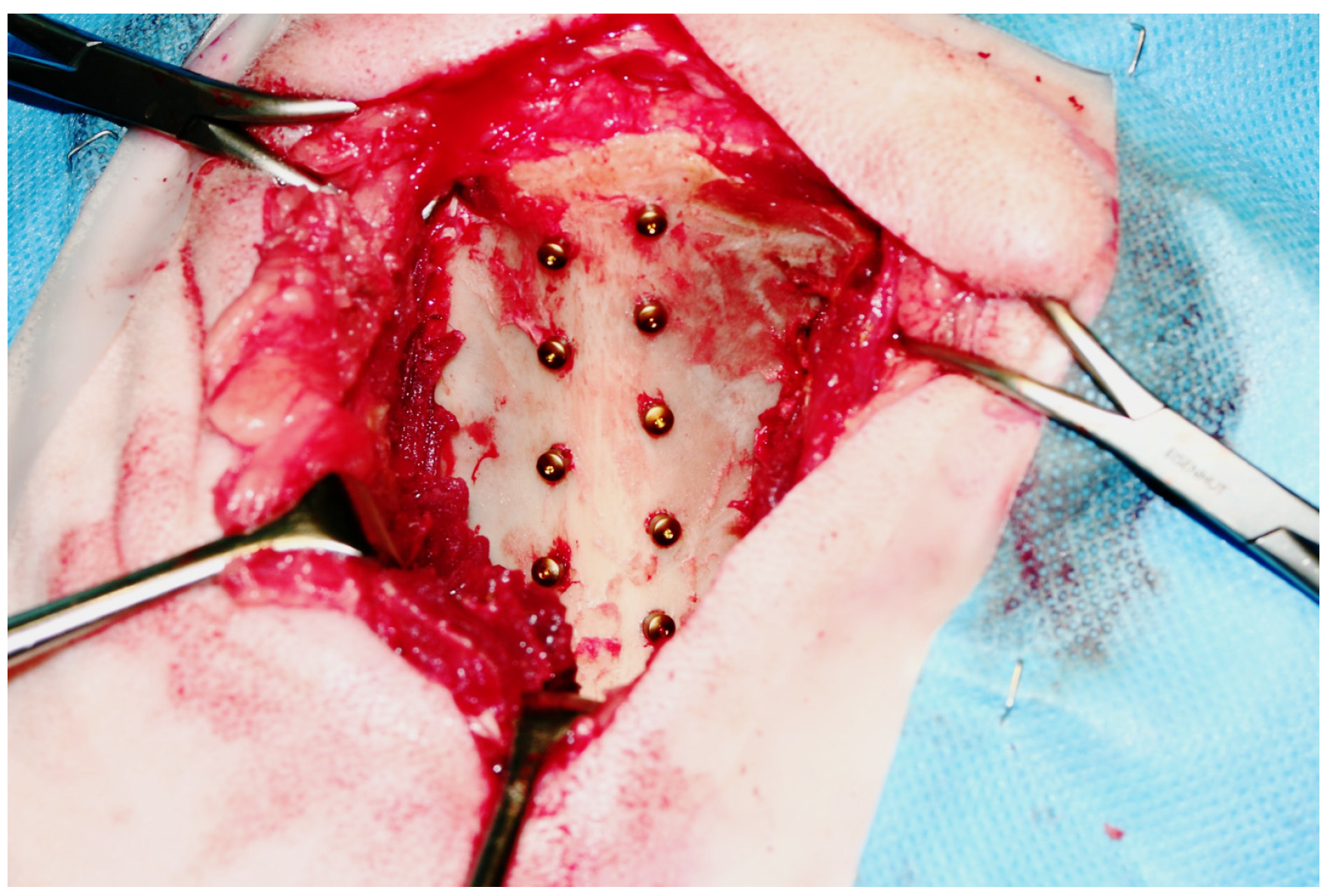
3.4. Surgical Procedure
3.5. Animal Sacrifice and Preparation of Bone Samples for Torque-Test and Histology
3.6. Analysis of Removal Torque-Tests
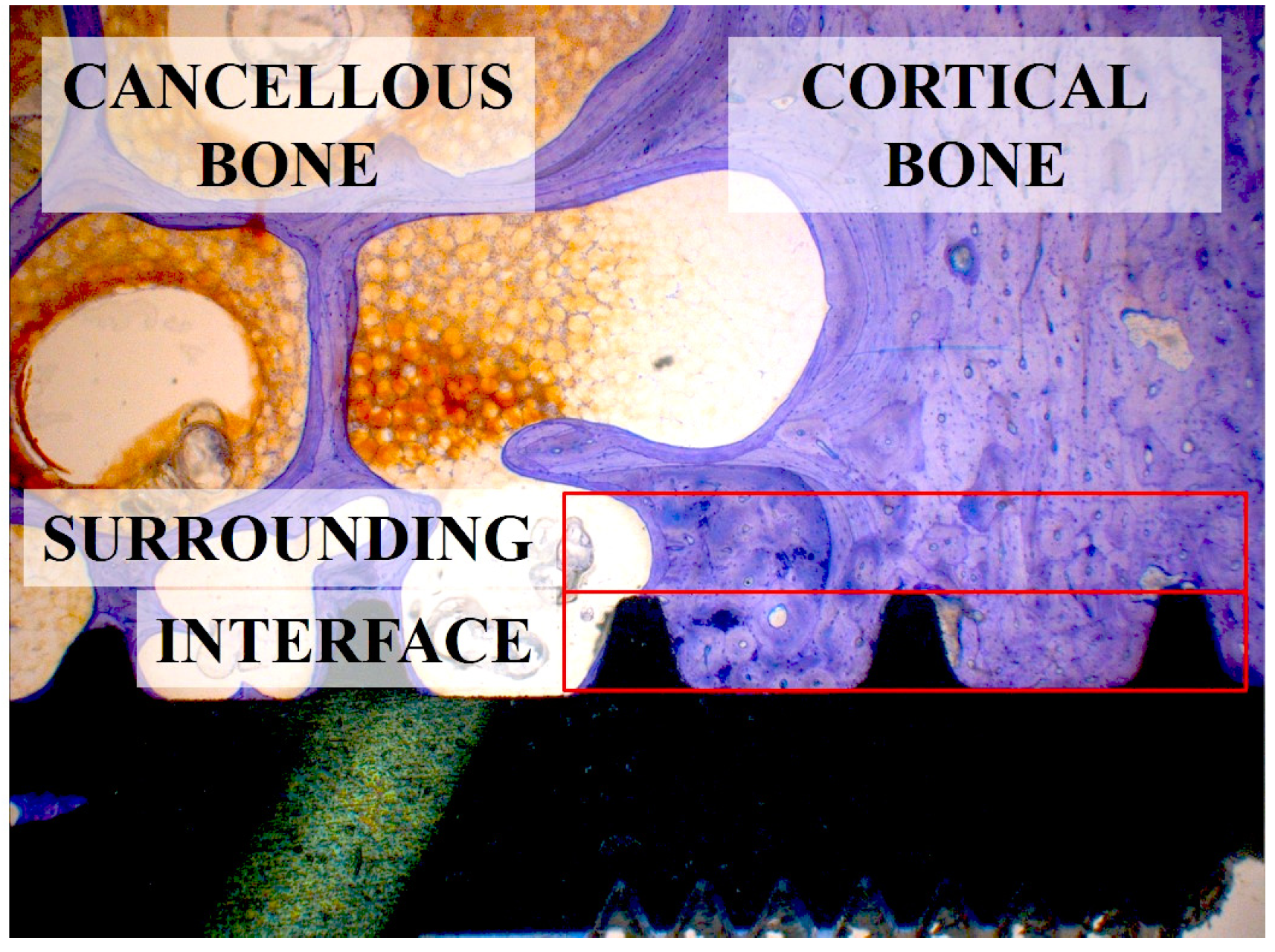
3.7. Histological Analysis
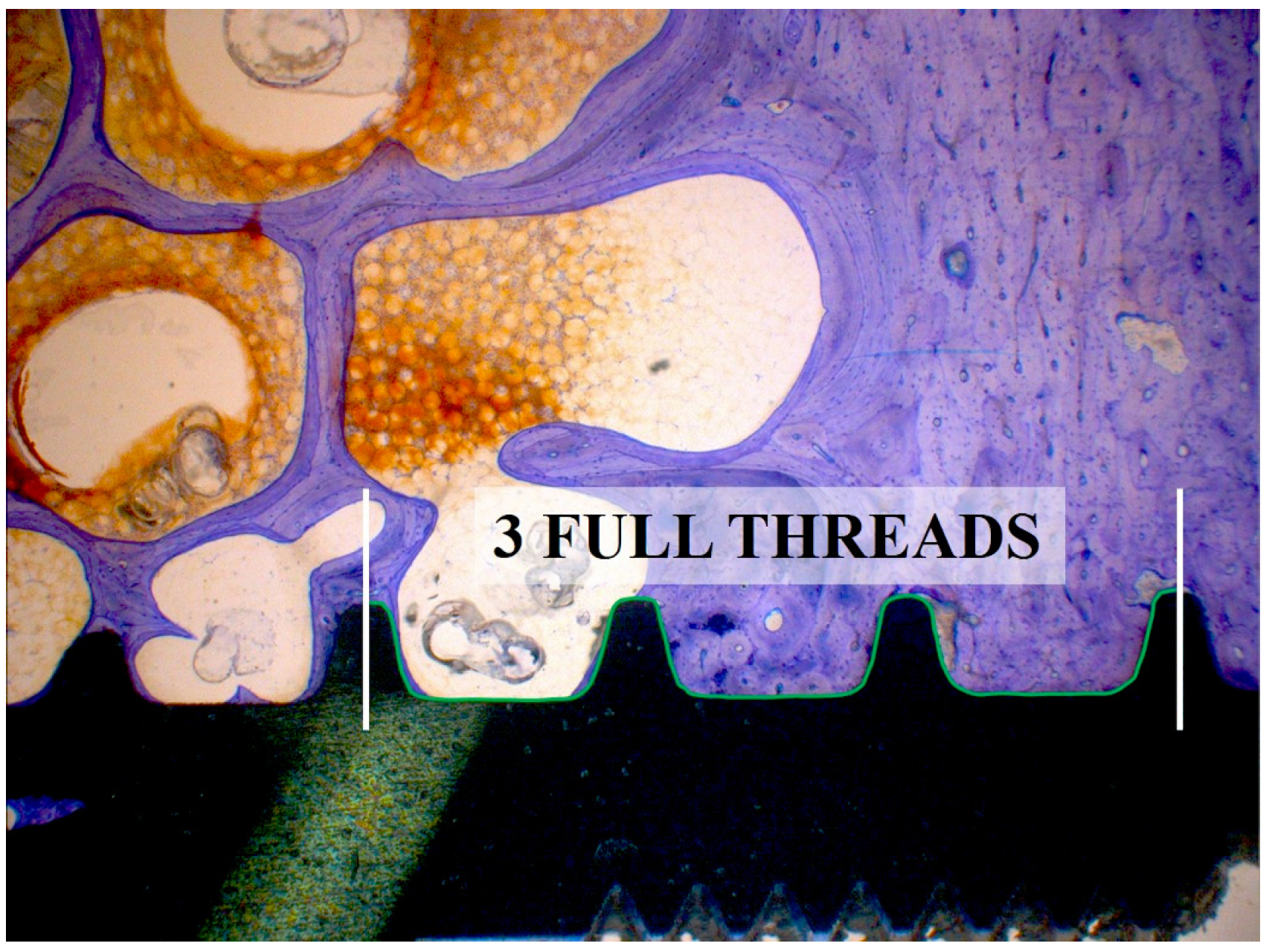
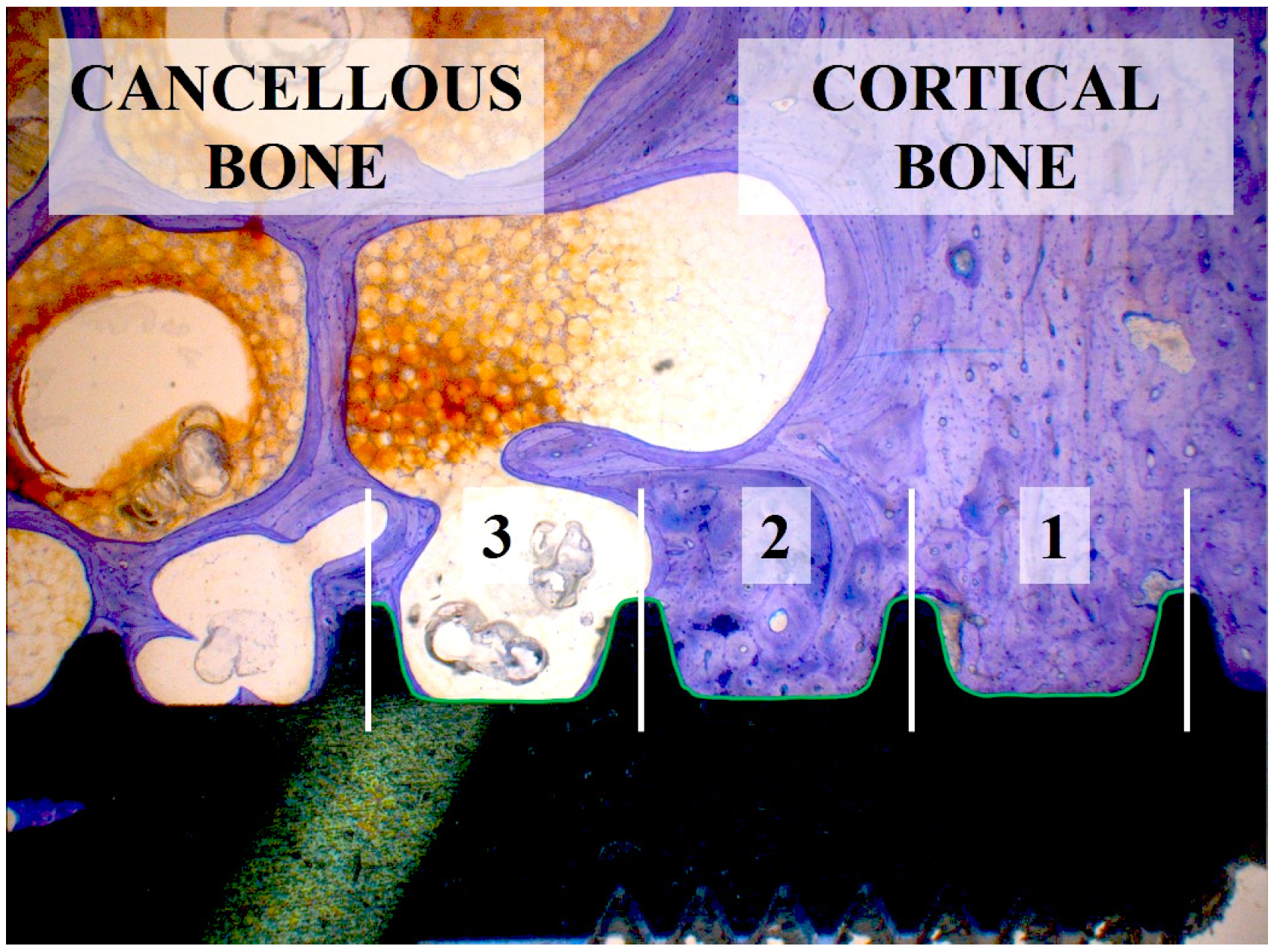
3.8. Histomorphometrical Analysis
3.9. Scanning Electron Microscopy
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sennerby, L.; Wennerberg, A. State of the art of oral implants. Periodontol 2000 2008, 47, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Ekelund, J.-A.; Lindquist, L.W.; Carlsson, G.E.; Jemt, T. Implant treatment in the edentulous mandible: A prospective study on Brånemark system implants over more than 20 years. Int. J. Prosthodont. 2003, 16, 602–608. [Google Scholar] [PubMed]
- Adell, R.; Lekholm, U.; Rockler, B.; Brånemark, P.I. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int. J. Oral Surg. 1981, 10, 387–416. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Dahl, E.; Enbom, L.; Engevall, S.; Engquist, B.; Eriksson, A.R.; Feldmann, G.; Freiberg, N.; Glantz, P.O.; Kjellman, O. Osseointegrated oral implants. A Swedish multicenter study of 8139 consecutively inserted Nobelpharma implants. J. Periodontol. 1988, 59, 287–296. [Google Scholar]
- Brunette, D.; Tengvall, P.; Textor, M.; Thomsen, P. Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications; Springer-Berlin: Berlin, Germany, 2013. [Google Scholar]
- Akagawa, Y.; Abe, Y. Titanium: The ultimate solution or an evolutionary step? Int. J. Prosthodont. 2003, 16, 28–29; 47–51. [Google Scholar]
- Esposito, M.; Murray-Curtis, L.; Grusovin, M.G.; Coulthard, P.; Worthington, H.V. Interventions for replacing missing teeth: Different types of dental implants. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Junker, R.; Dimakis, A.; Thoneick, M.; Jansen, J.A. Effects of implant surface coatings and composition on bone integration: A systematic review. Clin. Oral Implants Res. 2009, 20, 185–206. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, G.; Mendonça, D.B.S.; Aragão, F.J.L.; Cooper, L.F. Advancing dental implant surface technology—From micron- to nanotopography. Biomaterials 2008, 29, 3822–3835. [Google Scholar]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2007, 23, 844–854. [Google Scholar]
- Creugers, N.H.; Kreulen, C.M.; Snoek, P.A.; de Kanter, R.J. A systematic review of single-tooth restorations supported by implants. J. Dent. 2000, 28, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, D. Implants for life? A critical review of implant-supported restorations. J. Dent. 2007, 35, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Friberg, B. The posterior maxilla: Clinical considerations and current concepts using Brånemark System implants. Periodontol. 2000 2008, 47, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Puleo, D.A.; Thomas, M.V. Implant surfaces. Dent. Clin. North Am. 2006, 50, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Suvan, J.; Petrie, A.; Moles, D.R.; Nibali, L.; Patel, K.; Darbar, U.; Donos, N.; Tonetti, M.; D’Aiuto, F. Body mass index as a predictive factor of periodontal therapy outcomes. J. Dent. Res. 2014, 93, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Scully, C.; Hobkirk, J.; Dios, P.D. Dental endosseous implants in the medically compromised patient. J. Oral Rehabil. 2007, 34, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Lekholm, U. Immediate/early loading of oral implants in compromised patients. Periodontol 2000 2003, 33, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Cano, J.; Campo, J.; Vaquero, J.J.; Martínez González, J.M.; Bascones, A. High resolution image in bone biology II. Review of the literature. Med. Oral Patol. Oral Cir. Bucal 2008, 13, E31–E35. [Google Scholar]
- Al-Sabbagh, M. Implants in the esthetic zone. Dent. Clin. North Am. 2006, 50, 391–407. [Google Scholar]
- Wennerberg, A.; Albrektsson, T. Effects of titanium surface topography on bone integration: A systematic review. Clin. Oral Implants Res. 2009, 20, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Shalabi, M.M.; Gortemaker, A.; Van’t Hof, M.A.; Jansen, J.A.; Creugers, N.H.J. Implant surface roughness and bone healing: A systematic review. J. Dent. Res. 2006, 85, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.F. A role for surface topography in creating and maintaining bone at titanium endosseous implants. J. Prosthet. Dent. 2000, 84, 522–534. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Wennerberg, A. The impact of oral implants-past and future, 1966–2042. J. Can. Dent. Assoc. 2005, 71, 327:1–327:5. [Google Scholar]
- Jäger, M.; Zilkens, C.; Zanger, K.; Krauspe, R. Significance of nano- and microtopography for cell-surface interactions in orthopaedic implants. J. Biomed. Biotechnol. 2007, 69036:1–69036:19. [Google Scholar]
- Maekawa, K.; Yoshida, Y.; Mine, A.; Fujisawa, T.; Van Meerbeek, B.; Suzuki, K.; Kuboki, T. Chemical interaction of polyphosphoric acid with titanium and its effect on human bone marrow derived mesenchymal stem cell behavior. J. Biomed. Mater. Res. A 2007, 82, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, K.; Shimono, K.; Oshima, M.; Yoshida, Y.; Van Meerbeek, B.; Suzuki, K.; Kuboki, T. Polyphosphoric acid treatment promotes bone regeneration around titanium implants. J. Oral Rehabil. 2009, 36, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Kawazoe, Y.; Shiba, T.; Nakamura, R.; Mizuno, A.; Tsutsumi, K.; Uematsu, T.; Yamaoka, M.; Shindoh, M.; Kohgo, T. Induction of calcification in MC3T3-E1 cells by inorganic polyphosphate. J. Dent. Res. 2004, 83, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Itoh, H.; Kawazoe, Y.; Miyazaki, S.; Doi, K.; Kubo, T.; Akagawa, Y.; Shiba, T. Polyphosphate-mediated inhibition of tartrate-resistant acid phosphatase and suppression of bone resorption of osteoclasts. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Russell, R.G.G.; Watts, N.B.; Ebetino, F.H.; Rogers, M.J. Mechanisms of action of bisphosphonates: Similarities and differences and their potential influence on clinical efficacy. Osteoporos. Int. 2008, 19, 733–759. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Scheideler, L.; Rupp, F.; Hyzy, S.L.; Geis-Gerstorfer, J.; Schwartz, Z.; Boyan, B.D. A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomater. 2014, 10, 2907–2918. [Google Scholar] [CrossRef] [PubMed]
- Shannon, F.J.; Cottrell, J.M.; Deng, X.-H.; Crowder, K.N.; Doty, S.B.; Avaltroni, M.J.; Warren, R.F.; Wright, T.M.; Schwartz, J. A novel surface treatment for porous metallic implants that improves the rate of bony ongrowth. J. Biomed. Mater. Res. A 2008, 86, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Auernheimer, J.; Zukowski, D.; Dahmen, C.; Kantlehner, M.; Enderle, A.; Goodman, S.L.; Kessler, H. Titanium implant materials with improved biocompatibility through coating with phosphonate-anchored cyclic RGD peptides. Chembiochem. Eur. J. Chem. Biol. 2005, 6, 2034–2040. [Google Scholar] [CrossRef]
- Heijink, A.; Schwartz, J.; Zobitz, M.E.; Nicole Crowder, K.; Lutz, G.E.; Sibonga, J.D. Self-assembled monolayer films of phosphonates for bonding RGD to titanium. Clin. Orthop. 2008, 466, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Viornery, C.; Chevolot, Y.; Léonard, D.; Aronsson, B.-O.; Péchy, P.; Mathieu, H.J.; Descouts, P.; Grätzel, M. Surface modification of titanium with phosphonic acid to improve bone bonding: Characterization by XPS and ToF-SIMS. Langmuir 2002, 18, 2582–2589. [Google Scholar] [CrossRef]
- Esposito, M.; Grusovin, M.G.; Achille, H.; Coulthard, P.; Worthington, H.V. Interventions for replacing missing teeth: Different times for loading dental implants. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef]
- Viornery, C.; Guenther, H.L.; Aronsson, B.-O.; Péchy, P.; Descouts, P.; Grätzel, M. Osteoblast culture on polished titanium disks modified with phosphonic acids. J. Biomed. Mater. Res. 2002, 62, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Terheyden, H.; Lang, N.P.; Bierbaum, S.; Stadlinger, B. Osseointegration–communication of cells. Clin. Oral Implants Res. 2012, 23, 1127–1135. [Google Scholar] [CrossRef]
- Dayer, R.; Rizzoli, R.; Péchy, P.; Vig, T.; Aronsson, B.-O.; Descouts, P.; Ammann, P. Chemical modification of smooth titanium implant surface by coating with propylene-tetra-phosphonic acid increases their osseointegration. Bone 2005, 36. [Google Scholar] [CrossRef]
- Esposito, M.; Dojcinovic, I.; Germon, L.; Lévy, N.; Curno, R.; Buchini, S.; Péchy, P.; Aronsson, B.-O. Safety and efficacy of a biomimetic monolayer of permanently bound multi-phosphonic acid molecules on dental implants: 1 year post-loading results from a pilot quadruple-blinded randomised controlled trial. Eur. J. Oral Implantol. 2013, 6, 227–236. [Google Scholar] [PubMed]
- Langhoff, J.D.; Voelter, K.; Scharnweber, D.; Schnabelrauch, M.; Schlottig, F.; Hefti, T.; Kalchofner, K.; Nuss, K.; von Rechenberg, B. Comparison of chemically and pharmaceutically modified titanium and zirconia implant surfaces in dentistry: A study in sheep. Int. J. Oral Maxillofac. Surg. 2008, 37, 1125–1132. [Google Scholar] [CrossRef] [PubMed]
- Martini, L.; Fini, M.; Giavaresi, G.; Giardino, R. Sheep model in orthopedic research: A literature review. Comp. Med. 2001, 51, 292–299. [Google Scholar] [PubMed]
- Apelt, D.; Theiss, F.; El-Warrak, A.O.; Zlinszky, K.; Bettschart-Wolfisberger, R.; Bohner, M.; Matter, S.; Auer, J.A.; von Rechenberg, B. In vivo behavior of three different injectable hydraulic calcium phosphate cements. Biomaterials 2004, 25, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Kuttenberger, J.J.; Waibel, A.; Stübinger, S.; Werner, M.; Klasing, M.; Ivanenko, M.; Hering, P.; von Rechenberg, B.; Sader, R.; Zeilhofer, H.-F. Bone healing of the sheep tibia shaft after carbon dioxide laser osteotomy: Histological results. Lasers Med. Sci. 2010, 25, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Newman, E.; Turner, A.S.; Wark, J.D. The potential of sheep for the study of osteopenia: Current status and comparison with other animal models. Bone 1995, 16, S277–S284. [Google Scholar] [CrossRef]
- Kimmel, D.B.; Jee, W.S. A quantitative histologic study of bone turnover in young adult beagles. Anat. Rec. 1982, 203, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Al-Nawas, B.; Groetz, K.A.; Goetz, H.; Duschner, H.; Wagner, W. Comparative histomorphometry and resonance frequency analysis of implants with moderately rough surfaces in a loaded animal model. Clin. Oral Implants Res. 2008, 19, 1–8. [Google Scholar]
- Morand, M.; Irinakis, T. The challenge of implant therapy in the posterior maxilla: Providing a rationale for the use of short implants. J. Oral Implantol. 2007, 33, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, I.; Linder, E.; Larsson, L.; Berglundh, T. Deposition of nanometer scaled calcium-phosphate crystals to implants with a dual acid-etched surface does not improve early tissue integration. Clin. Oral Implants Res. 2013, 24, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, S.L. Eight-year clinical retrospective study of titanium plasma-sprayed and hydroxyapatite-coated cylinder implants. Int. J. Oral Maxillofac. Implants 1996, 11, 340–350. [Google Scholar] [PubMed]
- Van Oirschot, B.A.J.A.; Bronkhorst, E.M.; van den Beucken, J.J.J.P.; Meijer, G.J.; Jansen, J.A.; Junker, R. Long-term survival of calcium phosphate-coated dental implants: A meta-analytical approach to the clinical literature. Clin. Oral Implants Res. 2013, 24, 355–362. [Google Scholar]
- Albrektsson, T.O.; Johansson, C.B.; Sennerby, L. Biological aspects of implant dentistry: Osseointegration. Periodontol 2000 1994, 4, 58–73. [Google Scholar] [CrossRef] [PubMed]
- Gholami, H.; Mericske-Stern, R.; Kessler-Liechti, G.; Katsoulis, J. Radiographic bone level changes of implant-supported restorations in edentulous and partially dentate patients: 5-year results. Int. J. Oral Maxillofac. Implants 2014, 29, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Hirsch, J.M.; Lekholm, U.; Thomsen, P. Biological factors contributing to failures of osseointegrated oral implants. (I). Success criteria and epidemiology. Eur. J. Oral Sci. 1998, 106, 527–551. [Google Scholar]
- Kong, Y.-M.; Kim, D.-H.; Kim, H.-E.; Heo, S.-J.; Koak, J.-Y. Hydroxyapatite-based composite for dental implants: An in vivo removal torque experiment. J. Biomed. Mater. Res. 2002, 63, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Sabbatini, L.; Zambonin, P.G. XPS and SIMS surface chemical analysis of some important classes of polymeric biomaterials. J. Electron Spectrosc. Relat. Phenom. 1996, 81, 285–301. [Google Scholar] [CrossRef]
- Buser, D.; Nydegger, T.; Oxland, T.; Cochran, D.L.; Schenk, R.K.; Hirt, H.P.; Snétivy, D.; Nolte, L.P. Interface shear strength of titanium implants with a sandblasted and acid-etched surface: A biomechanical study in the maxilla of miniature pigs. J. Biomed. Mater. Res. 1999, 45, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, S.J.; Langhoff, J.D.; Voelter, K.; von Rechenberg, B.; Scharnweber, D.; Bierbaum, S.; Schnabelrauch, M.; Kautz, A.R.; Frauchiger, V.M.; Mueller, T.L.; et al. Biomechanical comparison of different surface modifications for dental implants. Int. J. Oral Maxillofac. Implants 2008, 23, 1037–1046. [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Von Salis-Soglio, M.; Stübinger, S.; Sidler, M.; Klein, K.; Ferguson, S.J.; Kämpf, K.; Zlinszky, K.; Buchini, S.; Curno, R.; Péchy, P.; et al. A Novel Multi-Phosphonate Surface Treatment of Titanium Dental Implants: A Study in Sheep. J. Funct. Biomater. 2014, 5, 135-157. https://doi.org/10.3390/jfb5030135
Von Salis-Soglio M, Stübinger S, Sidler M, Klein K, Ferguson SJ, Kämpf K, Zlinszky K, Buchini S, Curno R, Péchy P, et al. A Novel Multi-Phosphonate Surface Treatment of Titanium Dental Implants: A Study in Sheep. Journal of Functional Biomaterials. 2014; 5(3):135-157. https://doi.org/10.3390/jfb5030135
Chicago/Turabian StyleVon Salis-Soglio, Marcella, Stefan Stübinger, Michéle Sidler, Karina Klein, Stephen J. Ferguson, Käthi Kämpf, Katalin Zlinszky, Sabrina Buchini, Richard Curno, Péter Péchy, and et al. 2014. "A Novel Multi-Phosphonate Surface Treatment of Titanium Dental Implants: A Study in Sheep" Journal of Functional Biomaterials 5, no. 3: 135-157. https://doi.org/10.3390/jfb5030135
APA StyleVon Salis-Soglio, M., Stübinger, S., Sidler, M., Klein, K., Ferguson, S. J., Kämpf, K., Zlinszky, K., Buchini, S., Curno, R., Péchy, P., Aronsson, B.-O., & Von Rechenberg, B. (2014). A Novel Multi-Phosphonate Surface Treatment of Titanium Dental Implants: A Study in Sheep. Journal of Functional Biomaterials, 5(3), 135-157. https://doi.org/10.3390/jfb5030135