Literature Review: Clinical Data Interoperability Models
Abstract
1. Introduction
- The summary of main data standards used in a medical context;
- The study of the different interoperability models highlights these model’s pros and cons;
- The highlight of required features of a medical interoperability reference model.
2. Main Health System Interoperability Standards
2.1. Health Level Seven (HL7) Standards
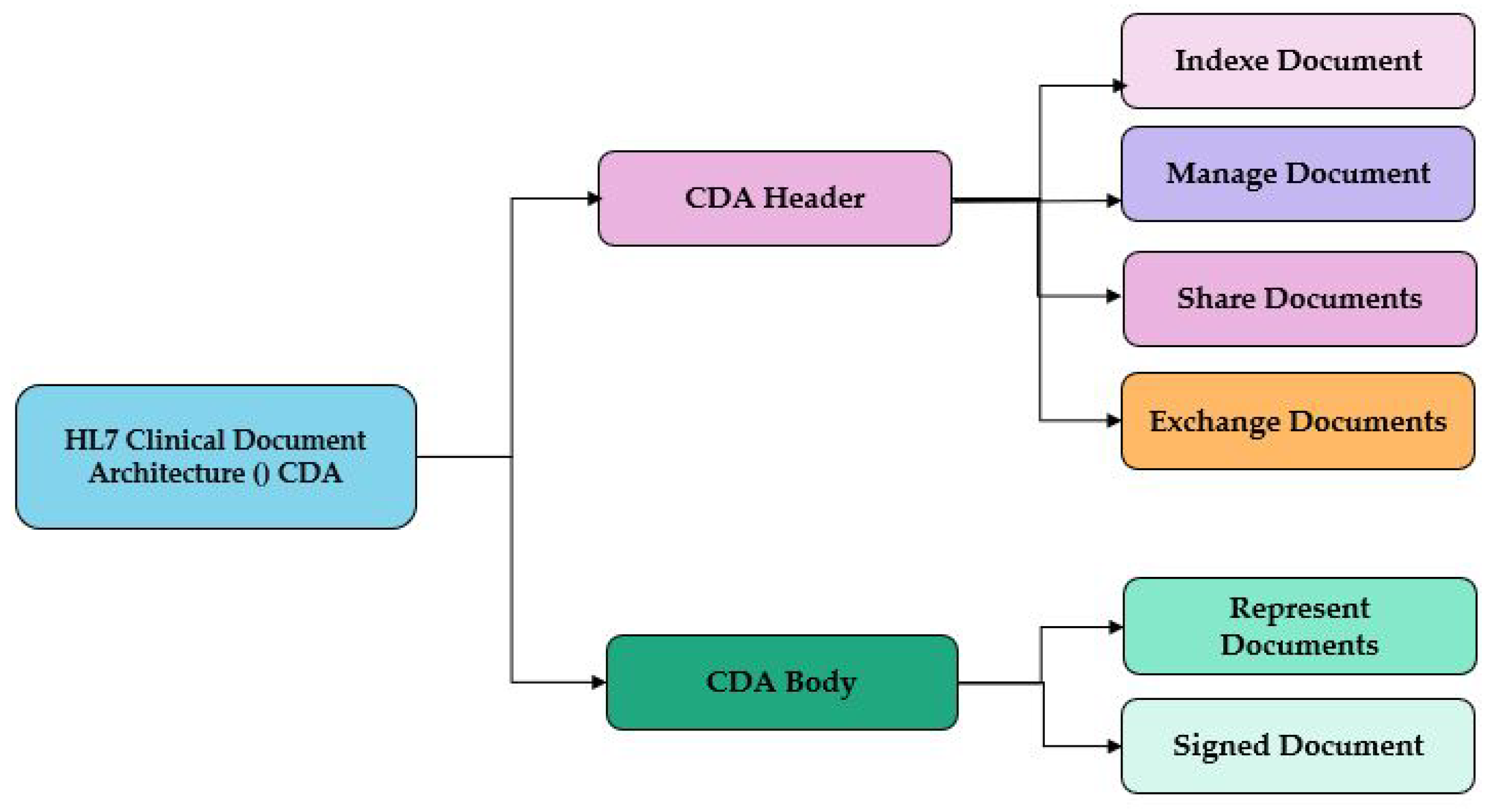
2.2. HL7 Clinical Document Architecture (CDA)
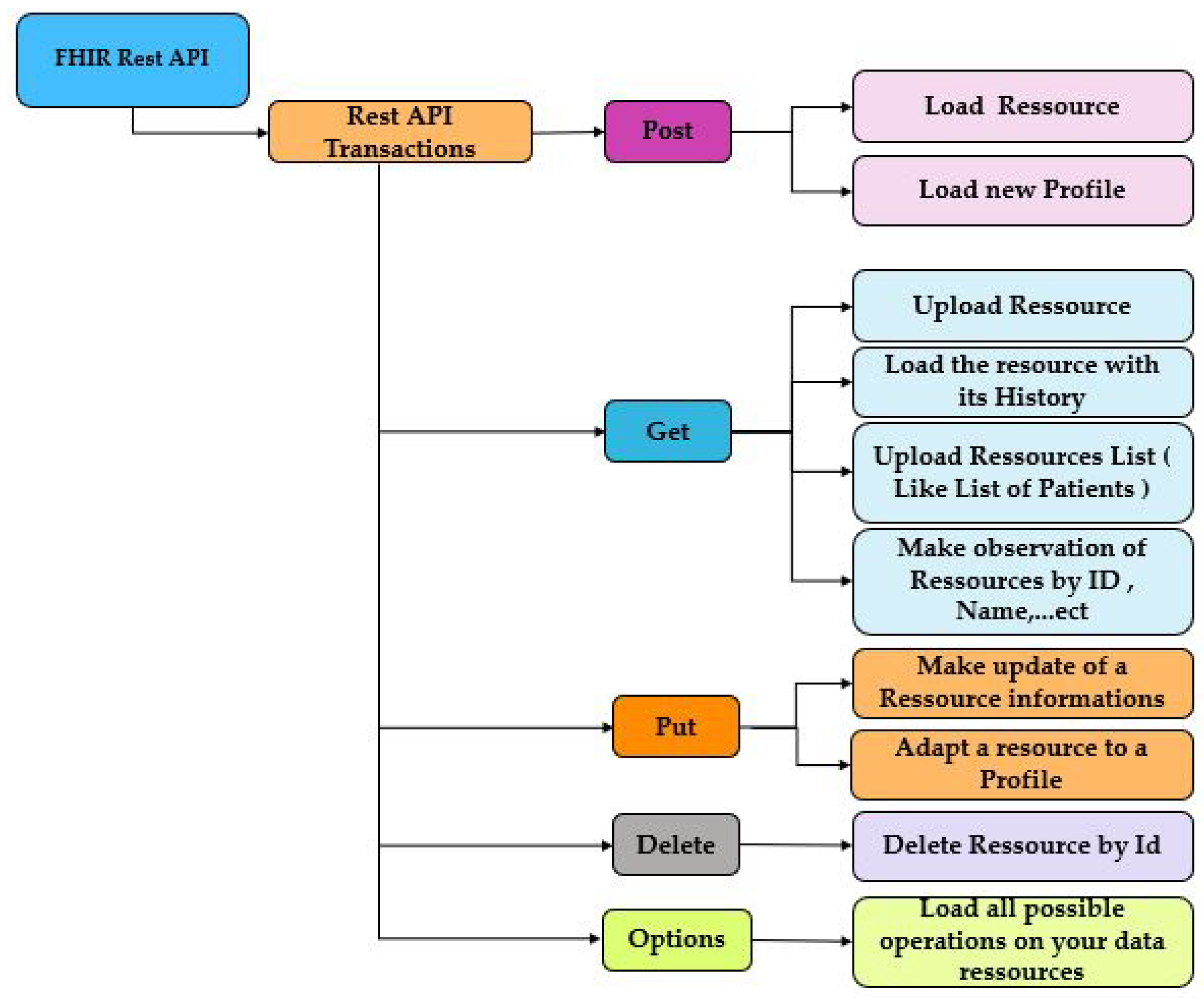
2.3. Fast Healthcare Interoperability Resources Standard (FHIR)
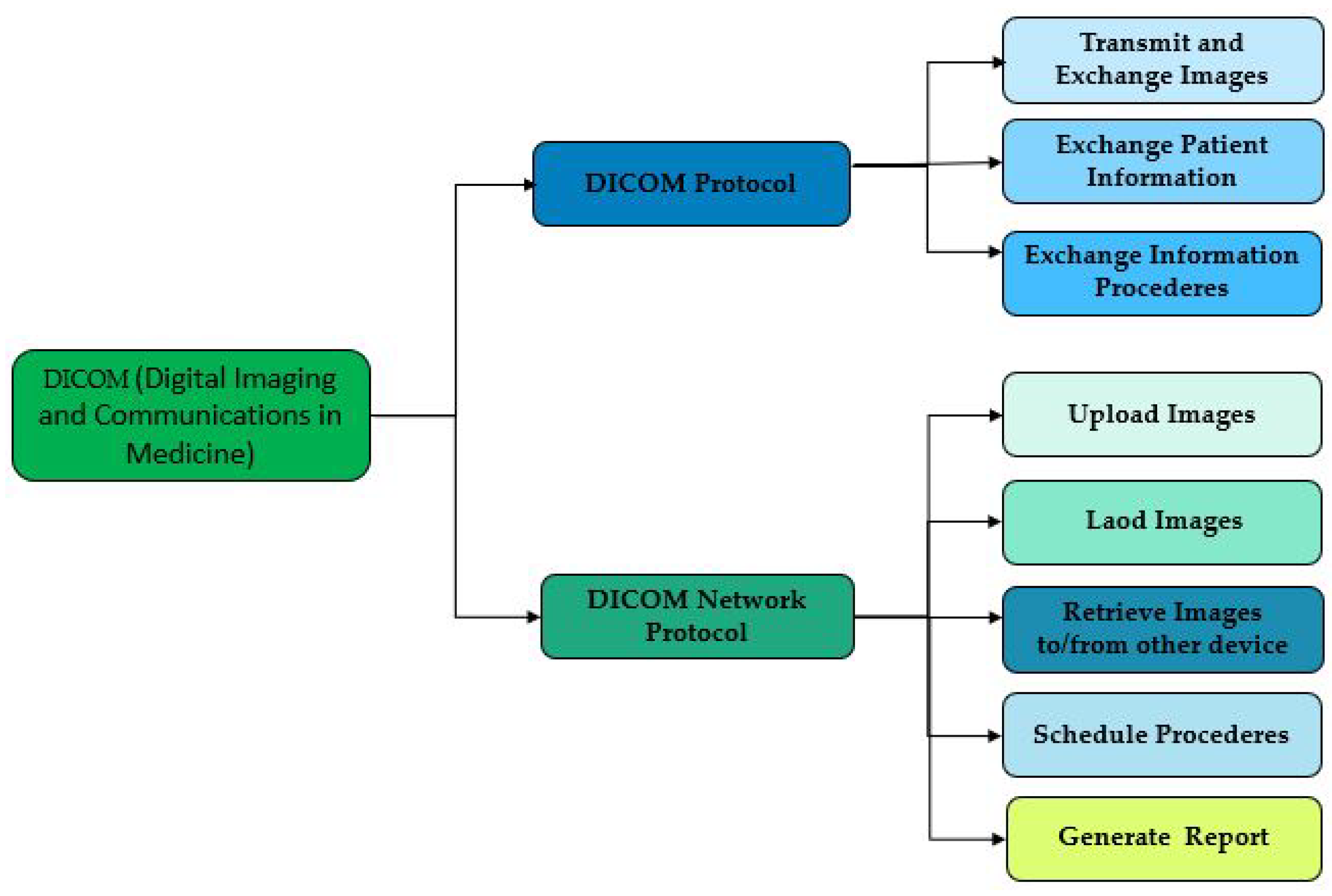
2.4. Digital Imaging and Communications in Medicine (DICOM)
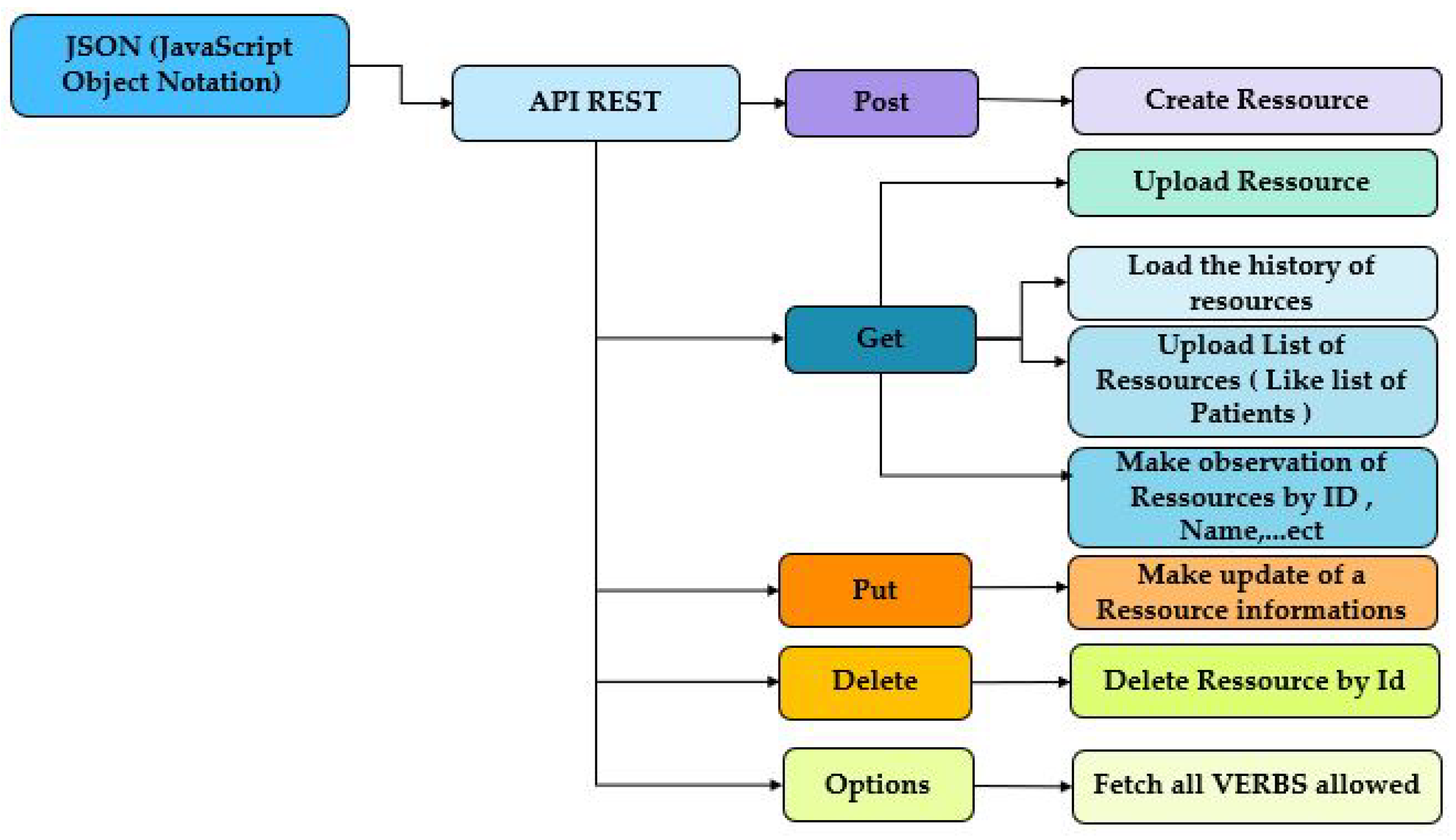
2.5. JavaScript Object Notation (JSON)
2.6. Data Interoperability Standards
3. State-of-the-Art Analysis and Comparative Study
3.1. State-of-the-Art
3.2. Comparative Study of Data Interoperability Models
3.2.1. Data Interoperability
3.2.2. Comparative Study between Data Interoperability Models
4. Discussion and Work Limitations
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 5GC | 5G Core |
| 5G OSM-MANO | Framework: 5G, Open Source Management and Orchestration Framework |
| ACP | Australian Colorectal Cancer Profile |
| AI | Artificial Intelligence |
| AJCC | American Joint Committee on Cancer |
| API | Application Programming Interface |
| BMI | Body mass index |
| CDA | Clinical Document Architecture |
| CDM | Clinical Document Management |
| CIOT | Cloud-centric IoT based disease diagnosis healthcare framework |
| CRF | Case Report Form |
| CSV | Comma-separated values |
| DICOM | Digital Imaging and Communications in Medicine |
| eConsent | electronic treatment consent |
| EDC | Electronic Data Capture |
| EHDS | European Health Data Space |
| EHR | Electronic Health Records |
| EIF | European Interoperability Framework |
| ETL | Extract, transform, load |
| FCAT | Federative Committee on Anatomical Terminology |
| FHIR | Fast Healthcare Interoperability Resources Standard |
| HSDF | Healthcare Sign Description Framework |
| HL7 | Health Level Seven |
| IA | Artificial Intelligence |
| IoMT | Internet of Medical Things |
| IoT | Internet of Things |
| IoTMD | Input-Data coming from any IoT device |
| IoT-SIM | IoT-based Semantic Interoperability Model |
| ISO | International Organization for Standardization |
| JSON | JavaScript Object Notation |
| KNN | K-Nearest Neighbors |
| LPU | Local Processing Unit |
| MeDic | Medical Data Interoperability through Collaboration of healthcare |
| devices Framework | |
| Medra | Repository: Medical Dictionary for Regulatory Activities |
| MedRec | Decentralized Medical record management system to handle EMRs |
| nusing blockchain technology | |
| NEMA | National Electrical Manufacturers Association |
| NLP | Natural Language Processing |
| OmH | Open Medical Health |
| OMOP | Observational Medical Outcomes Partnership |
| PH | Pulmonary Hypertension |
| PROMs | patient-reported outcome measures |
| PPR | Patient-Provider Relationship |
| RDF | Resource Description Framework |
| RIM | Reference Information Model |
| RWD | Real World Data |
| Shiny FHIR | Integrated framework leveraging Shiny R and HL7 FHIR |
| SIMB-IoT | Semantic Interoperability Model for Big-data in IoT |
| SPARQL | SPARQL Protocol and RDF Query Language |
| SWE | Sensor Web Enablement |
| TA | Terminologia Anatomica |
| UDR | User Diagnosis Result |
| UML | Unified Modeling Language |
| UMLs | Unified Medical Language System |
| VD | Virtual Doctor |
| XML | Extensible Markup Language |
| XPath | XML Path Language |
| XSLT | Extensible Stylesheet Language Transformations |
References
- Bender, D.; Sartipi, K. HL7 FHIR: An Agile and RESTful approach to healthcare information exchange. In Proceedings of the 26th IEEE International Symposium on Computer-Based Medical Systems, Porto, Portugal, 20–22 June 2013; pp. 326–331. [Google Scholar]
- Schweitzer, M.; Steger, B.; Hoerbst, A.; Augustin, M.; Pfeifer, B.; Hausmann, U.; Baumgarten, D. Data Exchange Standards in Teleophthalmology: Current and Future Developments. In dHealth 2022; IOS Press: Hall in Tyrol, Austria, 2022; pp. 270–277. [Google Scholar]
- Smith, B.; Ceusters, W. Hl7 Rim: An Incoherent Standard. Stud. Health Technol. Inform. 2006, 124, 133–138. [Google Scholar] [PubMed]
- Beeler, G.W. HL7 Version 3—An object-oriented methodology for collaborative standards development. Int. J. Med. Inform. 1998, 48, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Dolin, R.H.; Alschuler, L.; Boyer, S.; Beebe, C.; Behlen, F.M.; Biron, P.V.; Shabo, A. HL7 clinical document architecture, release 2. J. Am. Med. Inform. Assoc. 2006, 13, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.A.; Langer, S.G.; Kohli, M. Learning HL7 FHIR using the HAPI FHIR server and its use in medical imaging with the SIIM dataset. J. Digit. Imaging 2018, 31, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Duda, S.N.; Kennedy, N.; Conway, D.; Cheng, A.C.; Nguyen, V.; Zayas-Cabán, T.; Harris, P.A. HL7 FHIR-based tools and initiatives to support clinical research: A scoping review. J. Am. Med. Inform. Assoc. 2022, 29, 1642–1653. [Google Scholar] [CrossRef] [PubMed]
- Mukhiya, S.K.; Rabbi, F.; Pun, V.K.I.; Rutle, A.; Lamo, Y. A GraphQL approach to healthcare information exchange with HL7 FHIR. Procedia Comput. Sci. 2019, 160, 338–345. [Google Scholar] [CrossRef]
- Ammar, M.; Mahmoudi, S.; Chikh, M.A.; Abbou, A. Endocardial border detection in cardiac magnetic resonance images using level set method. J. Digit. Imaging 2012, 25, 294–306. [Google Scholar] [CrossRef]
- Tang, S.T.; Tjia, V.; Noga, T.; Febri, J.; Lien, C.Y.; Chu, W.C.; Chen, C.Y.; Hsiao, C.H. Creating a Medical Imaging Workflow Based on FHIR, DICOMweb, and SVG. J. Digit. Imaging 2023, 36, 794–803. [Google Scholar] [CrossRef]
- Bidgood, W.D., Jr.; Horii, S.C.; Prior, F.W.; Van Syckle, D.E. Understanding and using DICOM, the data interchange standard for biomedical imaging. J. Am. Med. Inform. Assoc. 1997, 4, 199–212. [Google Scholar] [CrossRef]
- Blazona, B.; Koncar, M. HL7 and DICOM based integration of radiology departments with healthcare enterprise information systems. Int. J. Med. Inform. 2007, 76, S425–S432. [Google Scholar] [CrossRef]
- Indrajit, I.; Verma, B. DICOM, HL7 and IHE: A basic primer on Healthcare Standards for Radiologists. Indian J. Radiol. Imaging 2007, 17, 66. [Google Scholar] [CrossRef]
- Cui, Y.; Shi, G.; Liu, X.S.; Zhao, W.; Li, Y.Q. Research on data communication between intelligent terminals of medical internet of things. In Proceedings of the 2015 International Conference on Computer Science and Applications (CSA), Wuhan, China, 20–22 November 2015; pp. 357–359. [Google Scholar]
- Sun, C.; Zeng, X.; Sun, C.; Si, H.; Li, Y. Research and Application of Data Exchange based on JSON. In Proceedings of the 2020 Asia-Pacific Conference on Image Processing, Electronics and Computers (IPEC), Dalian, China, 14–16 April 2020; pp. 349–355. [Google Scholar]
- Abdelouahid, R.A.; Marzak, A.; Sae, N. Towards a new meta-model of io lnteroperability. In Proceedings of the 2018 IEEE 5th International Congress on Information Science and Technology (CiSt), Marrakech, Morocco, 21–27 October 2018; pp. 54–63. [Google Scholar]
- Gomes, Y.F.; Santos, D.F.; Almeida, H.O.; Perkusich, A. Integrating MQTT and ISO/IEEE 11073 for health information sharing in the Internet of Things. In Proceedings of the 2015 IEEE International Conference on Consumer Electronics (ICCE), Las Vegas, NV, USA, 9–12 January 2015; pp. 200–201. [Google Scholar]
- Nemlaha, E.; Střelec, P.; Horák, T.; Kováč, S.; Tanuška, P. Suitability of MQTT and REST Communication Protocols for AIoT or IIoT Devices Based on ESP32 S3. In Software Engineering Application in Systems Design: Proceedings of 6th Computational Methods in Systems and Software 2022, Volume 1; Springer: Cham, Switzerland, 2023; pp. 225–233. [Google Scholar]
- Sarierao, B.S.; Prakasarao, A. Smart healthcare monitoring system using mqtt protocol. In Proceedings of the 2018 3rd iNternational Conference for Convergence in Technology (I2CT), Pune, India, 6–8 April 2018; pp. 1–5. [Google Scholar]
- Mavrogiorgou, A.; Kiourtis, A.; Touloupou, M.; Kapassa, E.; Kyriazis, D. Internet of medical things (IoMT): Acquiring and transforming data into HL7 FHIR through 5G network slicing. Emerg. Sci. J. 2019, 3, 64–77. [Google Scholar] [CrossRef]
- Zhang, S.; Cheng, D.; Deng, Z.; Zong, M.; Deng, X. A novel kNN algorithm with data-driven k parameter computation. Pattern Recognit. Lett. 2018, 109, 44–54. [Google Scholar] [CrossRef]
- Verma, P.; Sood, S.K. Cloud-centric IoT based disease diagnosis healthcare framework. J. Parallel Distrib. Comput. 2018, 116, 27–38. [Google Scholar] [CrossRef]
- Guarda, T.; Augusto, M.F.; Barrionuevo, O.; Pinto, F.M. Internet of Things in pervasive healthcare systems. In Next-Generation Mobile and Pervasive Healthcare Solutions; IGI Global: Hershey, PA, USA, 2018; pp. 22–31. [Google Scholar]
- Azaria, A.; Ekblaw, A.; Vieira, T.; Lippman, A. Using blockchain for medical data access and permission management. In Proceedings of the 2nd International Conference on Open and Big Data (OBD), Vienna, Austria, 22–24 August 2016; pp. 1–2. [Google Scholar]
- Nicole Boutros-Saikali, N.; Saikali, K.; Abou Naoum, R. An IoMT platform to simplify the development of healthcare monitoring applications. In Proceedings of the 2018 Third International Conference on Electrical and Biomedical Engineering, Clean Energy and Green Computing (EBECEGC), Beirut, Lebanon, 25–27 April 2018; pp. 6–11. [Google Scholar]
- Sony, P.; Nagarajan, S. Semantic Interoperability Model in Healthcare Internet of Things Using Healthcare Sign Description Framework. IAJIT 2022, 19, 589–596. [Google Scholar]
- Jabbar, S.; Ullah, F.; Khalid, S.; Khan, M.; Han, K. Semantic interoperability in heterogeneous IoT infrastructure for healthcare. Wirel. Commun. Mob. Comput. 2017, 2017, 9731806. [Google Scholar] [CrossRef]
- Jaleel, A.; Mahmood, T.; Hassan, M.A.; Bano, G.; Khurshid, S.K. Towards medical data interoperability through collaboration of healthcare devices. IEEE Access 2020, 8, 132302–132319. [Google Scholar] [CrossRef]
- Fischer, P.; Stöhr, M.R.; Gall, H.; Michel-Backofen, A.; Majeed, R.W. Data integration into OMOP CDM for heterogeneous clinical data collections via HL7 FHIR bundles and XSLT. In Digital Personalized Health and Medicine; IOS Press: Hall in Tyrol, Austria, 2020; pp. 138–142. [Google Scholar]
- Zong, N.; Wen, A.; Stone, D.J.; Sharma, D.K.; Wang, C.; Yu, Y.; Liu, H.; Shi, Q.; Jiang, G. Developing an FHIR-based computational pipeline for automatic population of case report forms for colorectal cancer clinical trials using electronic health records. JCO Clin. Cancer Inform. 2020, 4, 201–209. [Google Scholar] [CrossRef]
- Hong, N.; Prodduturi, N.; Wang, C.; Jiang, G. Shiny FHIR: An integrated framework leveraging Shiny R and HL7 FHIR to empower standards-based clinical data applications. Stud. Health Technol. Inform. 2017, 245, 868. [Google Scholar]
- Ullah, F.; Habib, M.A.; Farhan, M.; Khalid, S.; Durrani, M.Y.; Jabbar, S. Semantic interoperability for big-data in heterogeneous IoT infrastructure for healthcare. Sustain. Cities Soc. 2017, 34, 90–96. [Google Scholar] [CrossRef]
- Costa, C.M.; Menárguez-Tortosa, M.; Fernández-Breis, J.T. Clinical data interoperability based on archetype transformation. J. Biomed. Inform. 2011, 44, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Baskaya, M.; Yuksel, M.; Erturkmen, G.B.L.; Cunningham, M.; Cunningham, P.M. Health4Afrika-Implementing HL7 FHIR Based Interoperability. In Proceedings of the MedInfo 2019, Lyon, France, 25–30 August 2019; pp. 20–24. [Google Scholar]
- González-Castro, L.; Cal-González, V.M.; Del Fiol, G.; López-Nores, M. CASIDE: A data model for interoperable cancer survivorship information based on FHIR. J. Biomed. Inform. 2021, 124, 103953. [Google Scholar] [CrossRef] [PubMed]
- Lackerbauer, A.M.; Lin, A.C.; Krauss, O.; Hearn, J.; Helm, E. A model for implementing an interoperable electronic consent form for medical treatment using HL7 FHIR. Eur. J. Biomed. Inform. 2018, 14, 37–47. [Google Scholar] [CrossRef]
- Kiourtis, A.; Nifakos, S.; Mavrogiorgou, A.; Kyriazis, D. Aggregating the syntactic and semantic similarity of healthcare data towards their transformation to HL7 FHIR through ontology matching. Int. J. Med. Inform. 2019, 132, 104002. [Google Scholar] [CrossRef] [PubMed]
- Benson, T.; Grieve, G. Principles of Health Interoperability: SNOMED CT, HL7 and FHIR; Springer: Marseille, France, 2016. [Google Scholar]
- Setyawan, R.; Hidayanto, A.N.; Sensuse, D.I.; Suryono, R.R.; Abilowo, K. Data Integration and Interoperability Problems of HL7 FHIR Implementation and Potential Solutions: A Systematic Literature Review. In Proceedings of the 2021 5th International Conference on Informatics and Computational Sciences (ICICoS), Semarang, Indonesia, 24–25 November 2021; pp. 293–298. [Google Scholar]
- Holweg, F.; Achenbach, S.; Deppenwiese, N.; Gaede, L.; Prokosch, H.U. Towards a FHIR-Based Data Model for Coronary Angiography Observations. In Healthcare of the Future 2022; IOS Press: Hall in Tyrol, Austria, 2022; pp. 96–99. [Google Scholar]
| Organization | Standard | Description | Type of Data Supported |
|---|---|---|---|
| HL7 V2.8 | Exchange health data electronically between the IT systems | Electronic data | |
| HL7 1 | HL7 V3 (RIM 2) 2.36 | Specifications based on HL7’s RIM | Create reusable clinical data standards |
| HL7 CDA 3 | Stored health data in a structured format | Manage EHR 12 and clinical documents | |
| HL7 FHIR 4 (R4) | Specification for online health data exchange | Medical data in JSON 6 or XML 10 format | |
| NEMA 11 | DICOM 5 | This format is used to store, transmit, and display images | Digital medical imaging |
| Douglas Crockford, in 2002 | JSON 6 | Store and transmit health data in structured data format | Bills and medical reports |
| FCAT 8 | TA 7 | Anatomy terms in English and Latin | Text |
| ISO 9 | 21090:2011 | Harmonized data types for information exchange | Text |
| 13606 | High-level description of clinical information | JSON 6 | |
| IEEE 11073 [17] | Personal health data standards | HL7 1 format | |
| ISO 23903 | Representation of concepts on a semantic level | ICT-supported systems, EHDS 13 |
| References | Interoperability Model | Type of Data Source | Technologies | Architecture Type | FHIR Resources Type | FHIR Structure Type |
|---|---|---|---|---|---|---|
| Mavrogiorgou et al. in 2019 [20] | 5G OSM-MANO 10 Framework | IoMTs data, JSON, text | 5G Network Slicing, OpenCV, MAC Vendors API, KNN Algorithm, Levenshtein Distance | 5G Centralized Architecture | Structure definition, Observation | Patient |
| Verma et al. in 2018 [22] | CIoT 9 | IoMTs data, JSON, text | Cloud | Cloud Architecture | No FHIR Resources | User Diagnosis Result (UDR) |
| Azaria et al. in 2016 [24] | MedRec 12 | Patients Contracts | Ethereum blockchain, Gatekeeper | Blockchain Architecture (Decentralized) | Structure definition | Patient |
| Boutros-Saikali et al. in 2018 [25] | IoMT platform | Text, JSON, EHR 6 | VD algorithms, IA, Rest API | RESTful Service Architecture | Observation | Patient, OmH 1 |
| Sony et al. 2021, [26] | SIM-HIOT Model 11 | IoTMD 5, EHR, Home collection data | HSDF, UMLs [38] ontology, NLTK tool | Ontological Architecture | No FHIR Resources | Healthcare signs; Vital Sign, Medication Sign, and Symptom Sign |
| Jabbar, 2017, [27] | IoT-SIM 8 | IoMT data, JSON, Text | RDF, SWE framework, SPARQL query | RDF Architecture (Decentralized) | Structure Definition | IoT devices, Patients |
| Jaleel et al. in 2020 [28] | MeDic 7 | IoMTs Data; JSON, XML, Text | Cloud | Cloud Publish/Subscriber Architecture | No FHIR Resources | EHR Records, JSON |
| Fischer et al. in 2020 [29] | OHDSI OMOP Common Data Model | XML, JSON | ETL, OMOP CDM, XSLT, XPath | ETL Architecture | Structure Definition, Observation | Patient, Encounter, CSV |
| Zong et al. in 2020 [30] | FHIR-based method | EHRs, ACP 3, EDC 4 systems | ETL, NLP Tools | ETL Architecture (Centralized) | Structure definition | Profile, FHIR Questionnaire, Questionnaire Response Resources, Diagnostic Report, Observation |
| Hong et al. in 2017 [31] | Shiny FHIR framework 13 | CSV, XML, CDM 2 Database | HAPI FHIR API, Shiny API, R packages | RESTful service architecture | Structure Definition, Observation | Patient, Condition, Procedure |
| Ullah et al. in 2017 [32] | SIMB-IoT 16 | IoMT data, JSON, Text | RDF 14, SPARQL, Cloud, Big Data | Centralized Cloud Architecture | No FHIR Resources | Text(String), MedDRA 15 repository |
| Model | Data Interoperability | Standard (FHIR) | Centralized Architecture | Real-Time Monitoring | Ease of Implementation |
|---|---|---|---|---|---|
| Mavrogiorgou et al. [20] | Yes (limited to research) | FHIR | No | Yes | Moderate |
| Verma et al. [22] | No | N/A | Yes | Yes | Moderate |
| Azaria et al. [24] | Yes | N/A | No | No | Moderate |
| Boutros-Saikali et al. [25] | No | FHIR | No | Yes | Moderate |
| Sony et al. [26] | Yes | UMLS ontology | Yes | Yes | Moderate |
| Jaleel et al. [28] | Yes | N/A | No | Yes | Moderate |
| Fischer et al. [29] | No | N/A | No | No | Complex |
| Zong et al. [30] | No | N/A | No | No | Complex |
| Hong et al. [31] | No | N/A | No | No | Complex |
| Ullah et al. [32] | No | N/A | No | No | Moderate |
| Costa et al. [33] | Yes | Archetyped-data | No | No | Complex |
| Baskaya et al. [34] | Yes | FHIR | Yes | Yes | Moderate |
| Gonzales et al. [35] | Yes | Yes | Yes | Yes | Moderate |
| Lackerbauer et al. [36] | Yes | Yes | Yes | Yes | Complex |
| Kiourtis et al. [37] | yes | RDF Ontologies | Yes | No | Complex |
| Holweg Florian et al. [40] | Yes | HL7 FHIR, ICD-10, SNOMED CT | Yes | Yes | Moderate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ait Abdelouahid, R.; Debauche, O.; Mahmoudi, S.; Marzak, A. Literature Review: Clinical Data Interoperability Models. Information 2023, 14, 364. https://doi.org/10.3390/info14070364
Ait Abdelouahid R, Debauche O, Mahmoudi S, Marzak A. Literature Review: Clinical Data Interoperability Models. Information. 2023; 14(7):364. https://doi.org/10.3390/info14070364
Chicago/Turabian StyleAit Abdelouahid, Rachida, Olivier Debauche, Saïd Mahmoudi, and Abdelaziz Marzak. 2023. "Literature Review: Clinical Data Interoperability Models" Information 14, no. 7: 364. https://doi.org/10.3390/info14070364
APA StyleAit Abdelouahid, R., Debauche, O., Mahmoudi, S., & Marzak, A. (2023). Literature Review: Clinical Data Interoperability Models. Information, 14(7), 364. https://doi.org/10.3390/info14070364








